Abstract
For many years electromagnetic fields (EMFs) have been used clinically with various settings as an exogenous stimulation method to promote fracture healing. However, underlying mechanisms of action and EMF parameters responsible for certain effects remain unclear. Our aim was to investigate the influence of defined EMFs on human osteoblasts' and osteoclasts' viability and function. Primary human osteoblasts and osteoclasts were treated 3 times weekly for 21 days during their maturation process using the Somagen® device (Sachtleben GmbH, Hamburg, Germany), generating defined extremely low-frequency pulsed electromagnetic fields (ELF-PEMFs). Certain ELF-PEMF treatment significantly increased the total protein content (up to 66%), mitochondrial activity (up to 91.1%) and alkaline phosphatase (AP) activity (up to 129.9%) of human osteoblasts during the entire differentiation process. Furthermore, ELF-PEMF treatment enhanced formation of mineralized matrix (up to 276%). Interestingly, ELF-PEMF dependent induction of AP activity and matrix mineralization was strongly donor dependent — only osteoblasts with a poor initial osteoblast function responded to the ELF-PEMF treatment. As a possible regulatory mechanism, activation of the ERK1/2 signaling pathway was identified. Maturation of osteoclasts from human monocytes was not affected by the ELF-PEMF treatment. In summary the results indicate that a specific ELF-PEMF treatment with the Somagen® device improves viability and maturation of osteoblasts, while osteoclast viability and maturation was not affected. Hence, ELF-PEMF might represent an interesting adjunct to conventional therapy supporting bone formation during fracture healing or even for the treatment of osteoporosis.
Keywords: Extremely low-frequency pulsed electromagnetic fields (ELF-PEMF), Human osteoblasts, Human osteoclasts, ERK1/2, Specific EMF-responsiveness
Highlights
-
•
Exposure to extremely low-frequency pulsed electromagnetic fields (ELF-PEMFs) increases viability of human osteoblasts.
-
•
Exposure to specific ELF-PEMFs improves primary human osteoblasts’ function.
-
•
Especially osteoblasts with a low differentiation capacity profit from the ELF-PEMF exposure.
-
•
For the observed effects ERK1/2 activation is pivotal.
-
•
Osteoclast viability and function is not affected by the same ELF-PEMF.
1. Introduction
The biological process of fracture healing is complex and requires the interaction of many factors. If these mechanisms are inadequate or interrupted, healing is delayed or impaired. In 1974, Bassett and colleagues reported for the first time that electromagnetic fields (EMFs) may accelerate and improve bone healing (Bassett et al., 1974a). Since then various forms of electrical fields, including invasive direct current as well as non-invasive combined electromagnetic fields (EMFs) and pulsed electromagnetic fields (PEMFs) have been used as exogenous stimulation methods to promote bone formation, not only to support fracture healing but also for treatment of osteoporosis (Griffin et al., 2008).
Electrical Direct Current (DC) stimulators, which deliver the current through either implanted or percutaneously applied insulated electrodes, have the advantage of providing a constant challenge directly at the fracture site and increased patient compliance. However, due to the invasive nature, this method may result in higher infection rates and painful implants (Haddad et al., 2007). Therefore, non-invasive EMFs and PEMFs became more and more popular. Since the 1980s, the FDA has approved several EMF and PEMF devices. These have been used to treat delayed and non-union bone fractures (Griffin et al., 2008), for relieving postsurgical pain and edema (Thomas et al., 2007), to treat chronic wounds or to facilitate vasodilatation and angiogenesis (Strauch et al., 2009). The described EMFs range from frequencies of 20 Hz to 200 kHz, resulting in electrical fields in the tissue of approximately 1 to 100 mV/cm (Aaron et al., 2004). For PEMFs, the reported configurations of the applied magnetic fields vary in their amplitude, frequency, waveform, single pulse or pulse burst, resulting in magnetic fields of 10 to 2000 μT and voltage gradients of 1 to 100 mV/cm (Aaron et al., 2004). Hence, a broad range of configurations of EMFs and PEMFs has been applied clinically on fresh fractures and osteotomies, spine fusions, as well as delayed and non-union fractures. Despite this long history of clinical application the underlying mechanisms of action remain poorly understood. Hence, optimal parameters for the treatment of fractures have not been established yet.
Aim of the present project was to investigate the influence of selected extremely low-frequency pulsed electromagnetic fields (ELF-PEMFs) on primary human bone cells (osteoblasts and osteoclasts). ELF-PEMFs were generated with the help of the Somagen® (Sachtleben GmbH, Hamburg, Germany), a device certified for medical use according to European laws (CE 0482, compliant with EN ISO 13485:2012 + AC:2012). The Somagen® device generates ELF-PEMFs with specific magnetic field strengths and frequency spectra. For the present study 10 programs with frequencies between 10 and 90.6 Hz were chosen based on data from literature. There are several reports about the application of ELF-PEMFs inducing changes in cellular processes, in various cell types e.g. stem cells, immune cells, skin cells, tumor cells, tendon and bone cells (Hong et al., 2014, Seeliger et al., 2014, Tofani et al., 2001, Zhou et al., 2011, Zhou et al., 2014). The observed biological effects include amongst others cell differentiation, apoptosis, DNA synthesis, calcium flux, protein expression and phosphorylation, anti-inflammatory effects and hormone production. Focusing on the bone, both in vitro and in vivo studies, have demonstrated that EMF and PEMF treatment induces the synthesis of extracellular matrix proteins and growth factors, as well as initiating signaling cascades involved in proliferation and cell survival (Zhou et al., 2011, Zhou et al., 2014, Ciombor and Aaron, 2005, Yumoto et al., 2015). Therefore, as functional readout total protein content, mitochondrial activity, alkaline phosphatase (AP) and tartrate resistant acidic phosphatase (Trap5B) activity as well as matrix mineralization was assessed during the 3 weeks of maturation of primary human osteoblasts and osteoclasts. In order to better understand the regulatory mechanisms, gene expression changes and activation of mitogen activated protein kinases (MAPKinases), described to be involved in the osteogenic differentiation process, were examined.
2. Materials and methods
Human recombinant (rh)M-CSF and rhRANKL were purchased from Peprotech (Hamburg, Germany); cell culture medium and supplements were purchased from PAA (Cölbe, Germany); primary and secondary antibodies were obtained from Cell Signaling (Beverly, USA); chemicals were purchased from Sigma (Munich, Germany).
2.1. Ethics statement
All human studies were performed in accordance with the 1964 Declaration of Helsinki. Osteoblasts were isolated from bone tissue of patients undergoing total hip replacement or high tibial osteotomy, in accordance to the ethical vote (387/2012BO) of the University Clinic in Tübingen (Eberhard Karls Universität, Tübingen, Germany) and the patients' written consent. Bone tissue from (potential) tumor patients or patients with viral or bacterial infections was excluded from this study.
2.2. Isolation, expansion and maturation of primary human osteoblasts
Briefly, cancellous bone was removed mechanically from the femur head and washed 3–5 times with DPBS. After 1 h of collagenase digestion (DPBS, 0.07% Collagenase II — Biochrom AG, Berlin, Germany) at 37 °C, cancellous bone was washed with DPBS and released pre-osteoblasts were transferred to cell culture flasks in culture medium (MEM/Ham's F12, 10% FCS, 2 mM l-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 50 μM l-ascorbate-2-phosphate, 50 μM β-glycerol-phosphate) for expansion. Medium was changed every 4–5 days. Experiments were performed in passage 3 and 4, when a pure population of osteoblasts was reached (negative for CD14 and CD45 and positive for CD90 and CD105), as determined by flow cytometry (Ehnert et al., 2010). In order to favor osteogenic differentiation, cells were cultured in differentiation medium (MEM/Ham's F12, 1% FCS, 2 mM l-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 200 μM l-ascorbate-2-phosphate, 10 mM β-glycerol-phosphate, 25 mM HEPES, 1.5 mM CaCl2, 100 nM dexamethasone) during the experiments.
2.3. Generation of human osteoclasts
Briefly, human mononuclear cells were isolated from whole blood samples by density gradient centrifugation as described (Ehnert et al., 2011). Then, cells were plated in isolation medium (Alpha MEM, 10% FCS, 2 mM l-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin). Following overnight adherence, medium was changed to dedifferentiation medium (Alpha MEM, 10% FCS, 2 mM l-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 2.5 ng/ml rhM-CSF) for 6 days. Osteoclast differentiation was induced by differentiation medium (Alpha MEM, 10% FCS, 2 mM l-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 20 ng/ml rhRANKL) for 21 days (Schmitt et al., 2012).
2.4. Electromagnetic field application with the Somagen® device
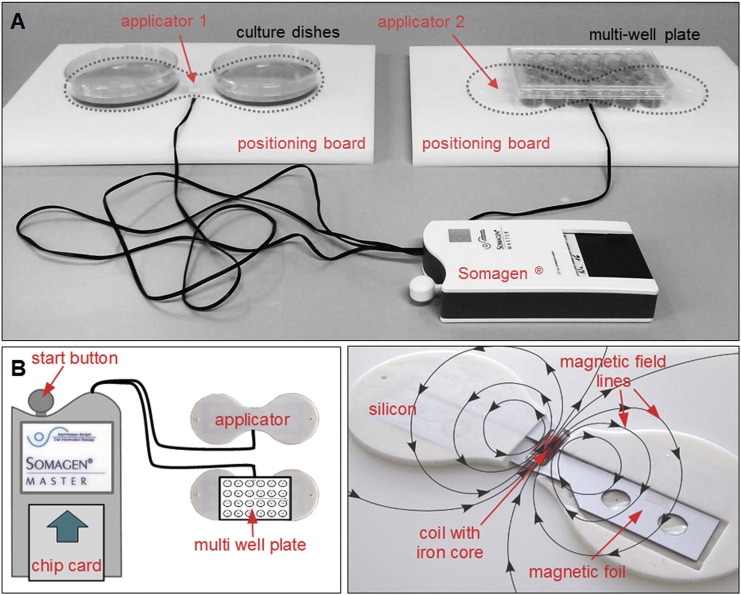
During the entire differentiation process primary human osteoblasts were exposed to selected ELF-PEMFs 3 times per week (Monday, Wednesday and Friday) for 7 min. The ELF-PEMFs were generated by the Somagen® device (Sachtleben GmbH, Hamburg, Germany) and emitted via two identical applicators located in a positioning board to ensure precise orientation of the culture dish/multi-well plate (Fig. 1A). In the present project 10 specific CIT programs (labeled CIT program #4, #10, #16, etc.) were selected each a combination of ELF-PEMF parameters such as fundamental frequency ( = pulse repetition rate), waveform (spike pulses), intensity (expressed as pulse amplitude and the corresponding RMS value) and send–pause intervals. The applied ELF-PEMF frequencies ranged from 10 to 90.6 Hz. For IP reasons more detailed data will not be published here. The ELF-PEMF applications were controlled via chip cards and started by simply pushing a button (Fig. 1B). Fig. 1C gives a simplified pattern of the magnetic field lines emitted by the applicator (magnetic dipole field).
Fig. 1.
Representation of the Somagen® device used to generate electromagnetic fields. (A) Primary human osteoblasts were exposed to ELF-PEMFs generated by the Somagen® device (Sachtleben GmbH, Hamburg, Germany). ELF-PEMF emission via two identical applicators located in a positioning board to ensure precise orientation of a culture dish/multi-well plate. (B) Control of specific ELF-PEMF parameters via chip cards, user-friendly by simply pushing the start button. (C) Simplified pattern of magnetic field lines emitted by the applicator (magnetic dipole field).
2.5. Sulforhodamine B (SRB) staining for total protein content
SRB staining on ethanol fixed cells was performed as reported (Skehan et al., 1990) in order to assess the total protein content. Resolved stain was quantified photometrically (λ = 565 nm–690 nm).
2.6. Resazurin conversion assay
Mitochondrial activity (viability) was measured by Resazurin conversion. Briefly, 1/10 volume of a 0.025% (w/v) Resazurin solution (in DPBS) was added to the cells. After 1 h of incubation at 37 °C, fluorescence intensity was measured (ex/em = 540/590 nm) and corrected to background control (solvent mixture without cells).
2.7. AP activity measurement
For assessing the AP activity primary human osteoblasts were incubated with reaction buffer (0.2% 4-nitrophenyl-phosphate, 50 mM glycine, 1 mM MgCl2, 100 mM TRIS, pH 10.5) at 37 °C. Resulting formation of 4-nitrophenol (yellow) was determined photometrically (λ = 405 nm). Signals were normalized to relative cell numbers by Resazurin conversion (Ehnert et al., 2010).
2.8. Von Kossa and Alizarin red staining
Briefly, ethanol fixed primary human osteoblasts were washed 3 times with tap water, then incubated with the corresponding staining solutions: (i) for von Kossa staining 3% silver-nitrate solution for 30 min at ambient temperature, followed by 3 additional washing steps and incubation with sodium-carbonate-formaldehyde solution (0.5 M sodium-carbonate, 10% formaldehyde) for color development (brownish-black). The staining was quantified by densitometric analysis using the ImageJ software (NIH, Bethesda, USA); (ii) for Alizarin Red staining 0.5% Alizarin Red solution (pH 4.0) for 30 min at ambient temperature. After 3 additional washing steps, Alizarin Red staining was resolved with 10% Cetylpyridiumchloride solution and quantified photometrically (λ = 562 nm) (Ehnert et al., 2010).
2.9. Semi-quantitative RT-PCR
Total cellular RNA was isolated using Trifast (Peqlab, Erlangen, Germany) according to the manufacturer's protocol. First-strand cDNA was synthesized from 2 μg of total RNA using the First Strand cDNA Synthesis Kit from Fermentas (St. Leon-Rot, Germany). Primer sequences and PCR conditions were as published (Ehnert et al., 2015, Freude et al., 2012). Products, separated by gel electrophoresis in a 1.8% (w/v) agarose gel, were visualized with ethidium bromide. Densitometric analysis of signals was performed using the ImageJ software (NIH, Bethesda, USA).
2.10. Western blot
Cells were lyzed in freshly prepared ice-cold RIPA buffer (50 mM TRIS, 250 mM NaCl, 2% Nonidet-P40, 2.5 mM EDTA, 0.1% SDS, 0.5% DOC, protease and phosphatase inhibitors, pH 7.2). Protein concentration was determined by micro-Lowry (Lowry et al., 1951). 30 μg of total protein were separated by SDS-PAGE and transferred to nitrocellulose membranes (Roth, Karlsruhe, Germany). Unspecific binding sites were blocked with 5% BSA in TBST solution (25 mM Tris, 137 mM NaCl, 2.7 mM KCl, 0.05% Tween-20, pH 7.4) for 1 h at RT. After overnight incubation with primary antibodies (Cell Signaling, Beverly, USA) diluted 1:1000 in TBST at 4 °C, membranes were incubated with the corresponding HRP-labeled secondary antibodies (1:10,000 in TBST) for 2 h at RT. For signal development, membranes were incubated for 1 min with ECL substrate solution (1.25 mM luminol, 0.2 mM p-coumaric acid, 0.03% H2O2 in 100 mM TRIS, pH 8.5). Chemiluminescent signals were detected with a CCD camera. Signal intensities were determined by densitometry using the ImageJ software (NIH, Bethesda, USA) and normalized to the corresponding GAPDH signals (Wang et al., 2014).
2.11. Trap5B activity assay and staining
Trap5B activity was measured in culture supernatants. 30 μl of culture supernatant was mixed with 90 μl of Trap5B activity assay buffer (0.2% 4-nitrophenyl-phosphate, 100 mM Na-acetate, 50 mM Na2-tartrate, pH 5.5) and incubated for 1 h at 37 °C. Reaction was stopped by adding 90 μl of 1 M NaOH. Resulting formation of 4-nitrophenol (yellow) was determined photometrically (λ = 405 nm). Signals were normalized to relative cell numbers by Resazurin conversion. Trap5B staining was performed as described (Schmitt et al., 2012).
2.12. Statistics
Results are expressed as mean or median ± SEM of at least 5 independent experiments (N ≥ 5) measured as triplicates or quadruplicates (n = 3 or 4). Details are given in the figure legends. Data sets with two groups were compared by Wilcoxon matched pairs test. Data sets with more than two groups were compared by non-parametric one-way analysis of variance (Kruskal Wallis) followed by Dunn's multiple comparison test (GraphPad Prism Software, El Camino Real, USA). p < 0.05 was taken as minimum level of significance.
3. Results
3.1. ELF-PEMF application improves viability and maturation of primary human osteoblasts
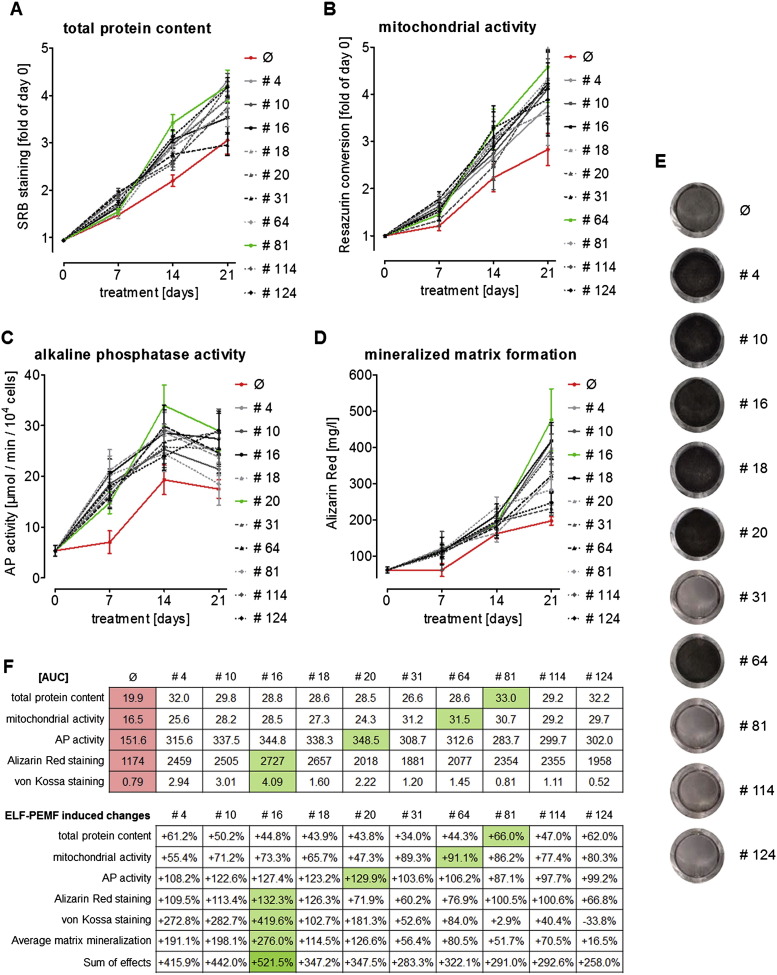
Using the Somagen® device primary human osteoblasts (N = 12) were exposed to 10 selected ELF-PEMFs 3 times per week (Monday, Wednesday and Friday) over the course of 21 days. Total protein content, mitochondrial activity (viability), AP activity and matrix mineralization were determined on day 0, 7, 14, and 21 of culture (Fig. 2A–E). In order to better reflect changes of different functions over the entire differentiation process, the area under the curve (AUC) was determined (Fig. 2F).
Fig. 2.
ELF-PEMF application with the Somagen® device improved viability and function of primary human osteoblasts. Primary human osteoblasts (N = 12, n = 4) were exposed to 10 specific ELF-PEMFs 3 times per week (Monday, Wednesday and Friday), for 7 min each day. ELF-PEMFs with 10 specific frequencies were generated using the Somagen® device. On day 0, 7, 14 and 21 of culture (A) total protein content was determined by SRB staining, (B) mitochondrial activity (viability) was determined by Resazurin conversion, (C) AP activity was determined by pNPP conversion and matrix mineralization was determined by (D) Alizarin Red and (E) von Kossa staining. In the graphs data are represented as median ± SEM. The red curve represents untreated cells (Ø). The green curve marks the program with the strongest effect over the entire observation period. (F) To evaluate the ELF-PEMF effect over the entire culture period the area under the curve (AUC) was determined. %—changes were calculated based on the AUC.
Throughout the entire maturation process total protein content and viability (mitochondrial activity) of primary human osteoblasts were increased by the ELF-PEMF application. On average the total protein content increased by + 49.7% (+ 34% to + 66%) over the entire duration of the experiment determined by the relative increase of the area under the curve (AUC). The highest protein content (+ 66% increase as compared to untreated cells) was observed in primary human osteoblasts exposed to ELF-PEMF CIT #81 (Fig. 2A & F). Similar to the total protein content the viability (mitochondrial activity) increased. The highest mitochondrial activity (+ 91.1% increase as compared to untreated cells) was measured in primary human osteoblasts exposed to ELF-PEMF CIT #64 (Fig. 2B & F).
On average AP activity was induced by ELF-PEMF treatment over the entire maturation process. The strongest induction of AP activity in primary human osteoblasts was observed in cells exposed to ELF-PEMF CIT #16 and #20 (+ 127.4% and + 129.9% increase as compared to untreated cells/Fig. 2C & F). As the strongest differences were observed early in the differentiation process, ELF-PEMF effects on AP activity were determined on day 7 in subsequent experiments. Similar to the AP activity also matrix mineralization was induced by ELF-PEMF treatment, as determined by von Kossa and Alizarin Red staining. Strongest induction of matrix mineralization was observed in primary human osteoblasts exposed to ELF-PEMF CIT #16 (+ 276% increase as compared to untreated cells/Fig. 2D–F).
Taken into account the effects for both viability and functionality ELF-PEMF CIT #16 treatment showed the strongest effects. These results led to the application of ELF-PEMF CIT #16 for all subsequent experiments (Fig. 2F). If not otherwise stated, subsequently the term ELF-PEMF refers to this specific CIT program.
3.2. Human osteoblasts with low initial AP activity respond well to the ELF-PEMF treatment
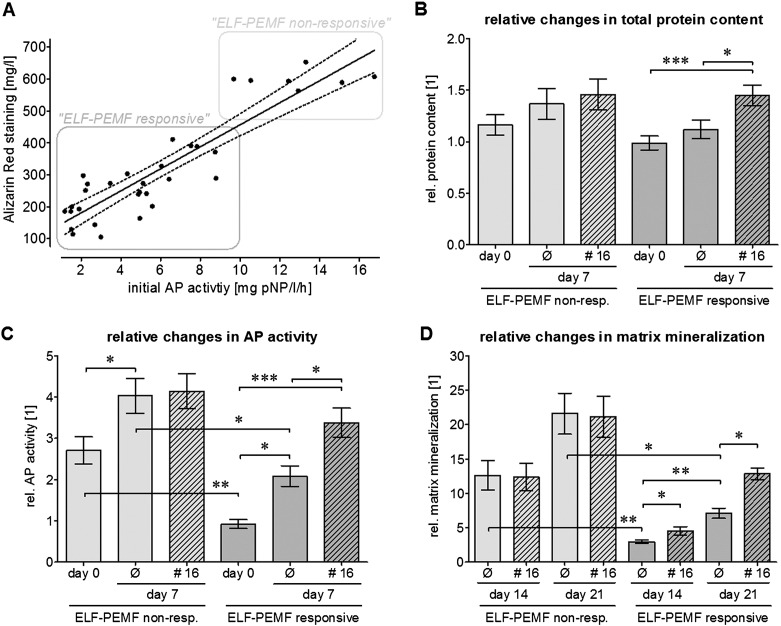
However, the observed induction of AP activity and matrix mineralization was strongly donor-dependent. While some donors responded strongly to the ELF-PEMF treatment, other donors were not affected at all. Donors with a high initial (day 0) AP activity formed lots of mineralized matrix, which could not be further improved by the ELF-PEMF treatment. Thus, these donors (N = 9) were defined as “ELF-PEMF non-responsive”. On the other hand, donors with low initial (day 0) AP activity and resulting deficits in matrix mineralization responded to treatment with the ELF-PEMF. Thus, these donors (N = 28) were termed “ELF-PEMF responsive” (Fig. 3A).
Fig. 3.
Primary human osteoblasts with low initial AP activity respond well to the ELF-PEMF CIT #16 exposure. (A) Correlation between initial AP activity (day 0) and matrix mineralization after 21 days in untreated cells (N = 37). Cells with a high initial AP activity (N = 9, n = 4) produced huge amounts of mineralized matrix. The function of these cells could not be further improved by ELF-PEMF CIT #16 exposure (ELF-PEMF non-responsive cells/light bars). Cells with a low initial AP activity (N = 28, n = 4) produced little mineralized matrix. The function of these cells could be significantly improved by ELF-PEMF CIT #16 exposure (ELF-PEMF responsive cells/dark bars). To further distinguish between the two responder groups primary human osteoblasts were exposed to ELF-PEMF CIT #16, generated with the Somagen® device, 3 times per week (Monday, Wednesday and Friday), for 7 min each day (hatched bars). After 0 and 7 days (B) total protein content/SRB staining and (C) AP activity were determined. (D) On day 0, 14 and 21 matrix mineralization was quantified by Alizarin Red staining. Data are calculated relative to the average of all cells on day 0 and displayed as mean ± SEM. * p < 0.05, ** p < 0.01 *** p < 0.001 as indicated.
Considering these two groups, the initial (day 0) total protein content was slightly lower in ELF-PEMF responsive cells as compared to ELF-PEMF non-responsive cells. In these ELF-PEMF responsive cells the total protein content was increased by 46.3% (as compared to untreated cells) upon exposure to the ELF-PEMF (CIT #16/day 7), reaching total protein levels comparable to ELF-PEMF non-responsive cells (Fig. 3B). The basal mitochondrial activity (viability/day 0) was comparable between both responder groups. Furthermore, mitochondrial activity was comparably induced by ELF-PEMF treatment (data not shown).
Differences in the functional parameter AP activity and matrix mineralization between the two responder groups were more pronounced. On day 0, the AP activity in ELF-PEMF responsive osteoblasts measured about 1/3 of the AP activity in ELF-PEMF non-responsive osteoblasts. Within 7 days of differentiation the AP activity increased approximately 2 fold in both the responder and non-responder group. In the non-responder group AP activity of cells could not be further increased by ELF-PEMF (CIT #16). In the ELF-PEMF responsive cells the AP activity was further increased by 62.5% by CIT #16, reaching almost the level of the ELF-PEMF non-responsive osteoblasts (Fig. 3C). Similar to AP activity matrix mineralization was found to be significantly lower in ELF-PEMF responsive osteoblasts. While in the ELF-PEMF non-responsive osteoblasts matrix mineralization could not be further increased by ELF-PEMF exposure, ELF-PEMF responsive osteoblasts produced significantly more mineralized matrix when exposed to CIT #16 (Fig. 3D).
3.3. ELF-PEMF application alters the expression of osteogenic transcription factors
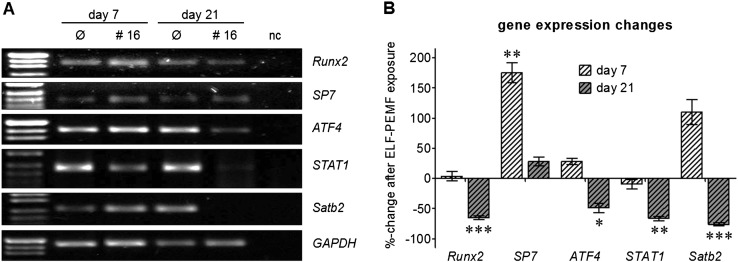
Subsequently the effect of ELF-PEMF (CIT #16) on the expression of osteogenic transcription factors Runx2, SP7 (osterix), ATF4, STAT1 and Satb2 was investigated by semiquantitative RT-PCR in ELF-PEMF responsive osteoblasts. For this purpose primary human osteoblasts were differentiated in the presence or absence of ELF-PEMF (CIT #16) for 7 and 21 days (N = 5, n = 4 respectively). Expression of the early osteogenic transcription factor Runx2 was not affected after 7 days of differentiation, but expression of the late osteogenic transcription factor SP7 (osterix) was significantly induced (approx. + 250%). At this time point expression of the regulatory transcription factors ATF4, STAT1 and Satb2 were not significantly affected, but Satb2 showed an increasing trend. Interestingly, after 21 days of differentiation a significant down-regulation of Runx2 and its regulators ATF4, STAT1, and Satb2 were detected in the ELF-PEMF (CIT #16) exposed cells compared to untreated controls (Fig. 4A & B).
Fig. 4.
ELF-PEMF CIT #16 application induces the expression of the late osteogenic transcription factor SP7 (osterix) in primary human osteoblasts. (A) Representative RT-PCR picture for Runx2, SP7 (osterix), ATF4, STAT1 and Satb2 for primary human osteoblasts (ELF-PEMF-responsive) after 7 and 21 days of differentiation with or without ELF-PEMF CIT #16 exposure. GAPDH was used as house-keeping gene. (B) Densitometric analysis (N = 5, n = 4) of the RT-PCR signals was performed using the ImageJ software. Data are represented as %-change in relative gene expression as compared to untreated cells. * p < 0.05, ** p < 0.01 *** p < 0.001 as compared to untreated cells.
3.4. ERK1/2 activation is pivotal for improving the osteogenic function by ELF-PEMF exposure with the Somagen® device
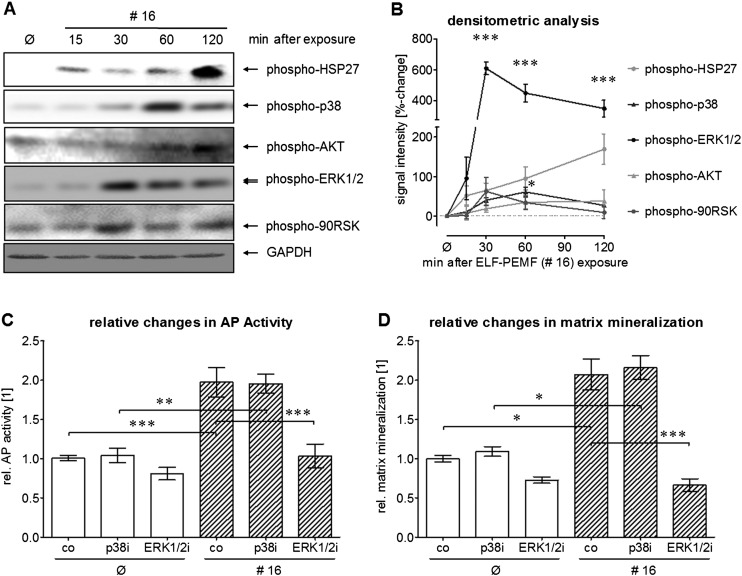
In order to elucidate possible underlying molecular mechanisms of action activated by ELF-PEMF, the following experiments were performed with primary human osteoblasts exhibiting a low initial AP activity (N = 5/ELF-PEMF responsive). 15, 30, 60 and 120 min after exposure to CIT #16 activation/phosphorylation of signaling molecules (HSP27, p38, AKT, ERK1/2, JNK and 90RSK) were investigated (Fig. 5A). Activation of JNK could not be detected (data not shown). Densitometric analysis (n = 4) of the Western blot signals revealed a continuous but not significant increase of phospho-HSP27 and phospho-AKT in primary human osteoblasts exposed to ELF-PEMF CIT #16. Phospho-p38 was slightly increased due to ELF-PEMF exposure, with a peak signal 60 min after exposure. Signals for phospho-90RSK also showed an increasing trend following ELF-PEMF exposure with a signal peak at 30 min. A similar pattern was observed for its upstream signaling partner phospho-ERK1/2 which was significantly (approx. + 600%) increased upon ELF-PEMF CIT #16 exposure (Fig. 5B).
Fig. 5.
Regulation of MAPKinases by ELF-PEMF CIT #16. (A) Representative Western blot for phospho-HSP27, phospho-p38, phospho-AKT, phospho-ERK1/2 and phospho-90RSK in primary human osteoblasts (ELF-PEMF-responsive) 15, 30, 60, 120 min after exposure to the ELF-PEMF CIT #16 using the Somagen® device. GAPDH was used as loading control. (B) Densitometric analysis (N = 5, n = 4) of the Western blot signals was performed using the ImageJ Software. Intensities are given as %-change of untreated cells. To investigate the role of the MAPKinase activation in ELF-PEMF induced osteoblast function, primary human osteoblasts were differentiated with and without exposure to the ELF-PEMF CIT #16 in the presence and absence of chemical inhibitors for p38 (SB203580) and ERK1/2 (U0126) signaling. (C) After 7 days AP activity was measured and (D) after 21 days matrix mineralization was quantified by Alizarin Red staining. All data were normalized to untreated cells. * p < 0.05, ** p < 0.01 *** p < 0.001 as compared to untreated cells.
As p38 and ERK1/2 signaling was significantly activated by ELF-PEMF CIT #16 exposure, their effect on the osteogenic function after ELF-PEMF treatment was investigated. Inhibition of p38 signaling by the chemical inhibitor SB203580 did not show an influence on the AP activity or matrix mineralization. However, inhibition of the ERK1/2 signal transduction with inhibitor U0126 significantly reduced both AP activity as well as matrix mineralization (Fig. 5C & D).
3.5. ELF-PEMF (CIT #16) does not alter the viability or function of osteoclasts
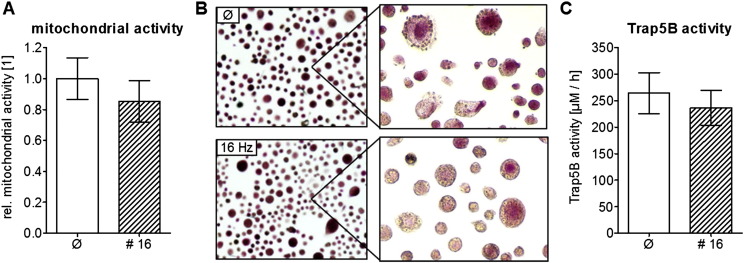
After observing the positive effects of ELF-PEMF CIT #16 on osteoblast viability and function, we sought to further investigate the effect of this ELF-PEMF on human osteoclasts. Hence, we generated osteoclasts from peripheral blood monocytes (N = 4). During the 3 week differentiation process, the cells were exposed to the ELF-PEMF CIT #16 3 times per week (Monday, Wednesday and Friday). After 21 days of differentiation, cell viability (mitochondrial activity), Trap5B staining and activity were determined. No significant differences for viability or Trap5B activity were observed between ELF-PEMF treated and untreated cells (Fig. 6A–C).
Fig. 6.
Osteoclast differentiation was not affected by ELF-PEMF CIT #16. To investigate the effect of ELF-PEMF CIT #16 on osteoclast maturation and function, we differentiated peripheral blood monocytes (N = 4, n = 4) to osteoclasts in the presence or absence of the CIT Technology. After 3 weeks of differentiation, no significant difference in (A) the mitochondrial activity/resazurin conversion, (B) the Trap5b staining (100 × and 200 × magnification) and (C) the Trap5b activity was observed.
4. Discussion
In the early 1970s, Bassett and colleagues reported for the first time that electromagnetic fields may accelerate and improve bone healing (Bassett et al., 1974b). Since then, the FDA has approved several EMF and PEMF devices. These devices have been used clinically as exogenous stimulation methods to promote bone formation, for many years (Griffin et al., 2008). Although positive effects of EMFs and PEMFs on osteotomies, spine fusions, as well as delayed and non-union fractures are documented, little is known about the underlying mechanisms of action. In vivo, electrical and electromagnetic fields are assumed to play a role in bone healing through the same principles as mechanical stress applications. When mechanical load is applied to bone, a strain gradient develops. Subsequent pressure gradients in the interstitial fluid cause flow-related shear stress and electrical potentials subsequent to the compensating fluid streaming (Hannouche et al., 2001). However, the underlying molecular mechanisms are not explained as easily. Several theories are favored within the bioelectromagnetic society, e.g. the molecular gyroscope model (Binhi and Savin, 2002), Lorentz models (Muehsam and Pilla, 2009a, Muehsam and Pilla, 2009b), DNA antenna model (Blank and Goodman, 2011), radical pair model (Foley et al., 2011), and ion cyclotron resonance (Bawin et al., 1975). It might well be that effects triggered by the ELF-PEMFs described here can be explained by a combination of these theories.
In the present project, we investigated the effect of ELF-PEMFs on the viability and function of primary human osteoblasts and osteoclasts. ELF-PEMFs were generated with the Somagen® device which is certified for medical use according to European laws. In the first project phase 10 ELF-PEMF programs with frequencies ranging between 10 Hz and 90.6 Hz were applied in order to identify the most potent CIT program; in a second step further analysis was performed with the identified ELF-PEMF CIT #16 in order to identify possible regulatory mechanisms.
Focusing on the bone there are both in vitro and in vivo studies suggesting that EMFs and PEMFs might induce protein synthesis and activate signaling cascades involved in proliferation and cell survival using such devices (Aaron et al., 2004, Zhou et al., 2014, Ciombor and Aaron, 2005, Hannouche et al., 2001). In our experiments, the ELF-PEMF treatment induced protein synthesis and mitochondrial activity of primary human osteoblasts, suggesting a slight induction of proliferation in EMF-PEMF exposed cells. This is in clear contrast to the observations of Zhou et al., who investigated different EMF waveforms showing an inhibition of proliferation in rat osteoblasts exposed to sinusoidal EMFs (50 Hz/0.9–4.8 mT) (Zhou et al., 2011, Zhou et al., 2014). In this study the effects on viability/proliferation seem to be independent of the applied frequency. This is in line with the series of preclinical and clinical studies on electrical and electromagnetic energy on bones that Aaron et al. reviewed (Aaron et al., 2004). Most frequently, the application of PEMF and their role on regulation of structural extra-cellular matrix proteins have been explored. Ciombor and colleagues suggest that the application of electromagnetic fields, both in vitro and in vivo, stimulate the synthesis of structural proteins of the extracellular matrix (Ciombor and Aaron, 2005). Production of extracellular matrix, however, is a measure of the differentiation potential of osteoblasts.
Contrary to general stimulation of protein synthesis and mitochondrial activity, in our experiments the ELF-PEMF mediated modulation of differentiation potential of osteoblasts was strongly donor dependent. The strongest induction of AP activity and matrix mineralization (von Kossa and Alizarin Red staining) was observed in primary human osteoblasts exposed to two specific ELF-PEMFs (CIT programs #16 and #10).
Expression of many of the matrix proteins, e.g. collagen type I, fibronectin or osteocalcin, as well as the following matrix mineralization are controlled by a defined interplay between the key osteogenic transcription factors RUNX2, ATF4, SATB2, STAT1 and osterix (SP7) (Ducy et al., 1997). Using the Somagen® device, we observed an early up-regulation of osterix (SP7), ATF, and SATB2. SATB2 is an adapter molecule between ATF4 and RUNX2, increasing their activity (Dobreva et al., 2006). Osterix (SP7) acts downstream of RUNX2 and is mainly expressed during late osteogenesis (Nakashima et al., 2002). This points towards an accelerated osteogenic maturation caused by the ELF-PEMF (CIT #16). Contrary to ATF4 and SATB2, STAT1 inhibits the activity of RUNX2 by inducing conformational changes in the nuclear localization sequence of this transcription factor. Thus, STAT1 knock-out mice display a predominant increase in osteoblast differentiation leading to a significantly higher bone mineral density (Kim et al., 2003). In the primary human osteoblasts treated with the CIT Technology, we observed an early down-regulation of STAT1 gene expression. Our results are in line with several studies showing increased expression of transcription factors and growth factors, which enhance cellular repair and the synthesis of extra-cellular matrix proteins (Hannouche et al., 2001), which might explain the observed increase in AP activity and matrix mineralization. This result is supported by the work of Zhang and colleagues who showed an increased calcium up-take in osteoblasts treated with ELF-PEMF in the range of 50 Hz and 0.8 mT (Zhang et al., 2010). Also Zhou et al., showed an improved AP activity and matrix mineralization evoked in rat calvarial osteoblasts by sinusoidal EMFs (50 Hz) with an intensity of 1.8 mT and 3.6 mT (Zhou et al., 2014).
It has been suggested that the application of electromagnetic fields may activate transmembrane receptors, e.g. parathyroid hormone, insulin, transferrin or calcitonin receptors, thus initiating signaling cascades (Ciombor and Aaron, 2005). In our experiments, we observed a strong activation of the ERK1/2 signaling pathway in primary human osteoblasts after ELF-PEMF (CIT #16) treatment. Inhibition of ERK1/2 signaling with the inhibitor U0126 prevented activation of AP activity and matrix mineralization by ELF-PEMF (CIT #16) treatment, leading to the conclusion that activation of the ERK1/2 signaling cascade is pivotal for the observed positive effects of this ELF-PEMF treatment on osteoblast function. Our result is supported by the work of Goodman and colleagues showing an ELF-PEMF-dependent activation of the ERK1/2 signaling cascade in planaria, which is correlated to an increased regeneration rate (Goodman et al., 2009). Also Yumoto and colleagues found in mouse calvaria cell line MC3T3-E1 that electromagnetic wave irradiation increases osteoblastic cell activity and the expression of growth factors via ERK1/2 and p38 MAPK pathways (Yumoto et al., 2015). Taken together these observations from planaria, rodents and human leads to the assumption that EMF mediated ERK1/2 activation is a general principle. A possible mechanistic explanation to this phenomenon is given by Friedman et. al linking ERK1/2 activation to radical pair mechanism mediated through plasma membrane by NADH oxidase generating ROS (Friedman et al., 2007).
Considering proliferation and differentiation of osteoblasts as equally important for a future clinical application to support bone formation, ELF-PEMF exposure with the Somagen® device applying CIT #16 showed the strongest effects on primary human osteoblasts over the entire observation period. However, in our study only osteoblasts from donors with an initially low AP activity and consequently little matrix mineralization capacity were sensitive towards the ELF-PEMF treatment. These results indicate a possible clinical use of the Somagen® device applying ELF-PEMF CIT #16 for fracture healing or treatment of osteoporosis. The correlation between low initial AP activity and little matrix mineralization after the differentiation process, was also described in jaw periosteal cells (Alexander et al., 2010). Thus, circulating bone-specific AP levels might be used as a possible predictive marker to identify patients' responsiveness to the ELF-PEMF treatment.
Hong and co-workers (Hong et al., 2014) could show that EMF controlled osteoclast activity can be inversely correlated to the osteoblasts activity. Also Rubin and colleagues suggest that osteoclast formation might be suppressed by low frequency, low intensity electric fields (Rubin et al., 1996). Interestingly, ELF-PEMF exposure with the Somagen® device (CIT #16) did not significantly affect osteoclast viability or function in our hands.
5. Conclusion
Summarizing our results, specific ELF-PEMF treatment with the Somagen® device (CIT #16) improves the viability/proliferation and accelerates the osteogenic differentiation of primary human osteoblasts. Strikingly, only osteoblasts with an initial low AP activity and consequently little matrix mineralization after differentiation were responsive to the ELF-PEMF treatment. Seeing also that osteoclast maturation was not affected by ELF-PEMF CIT #16 this technology might present an interesting adjunct to conventional therapy supporting bone formation during fracture healing or even during the treatment of osteoporosis.
Acknowledgments
The authors thank Patrysya Lacorte for her excellent technical assistance and Dr. Ingo Fricke for critically reviewing the manuscript.
References
- Aaron R.K., Ciombor D.M., Simon B.J. Treatment of nonunions with electric and electromagnetic fields. Clin. Orthop. Relat. Res. 2004;21–9 doi: 10.1097/00003086-200402000-00005. [DOI] [PubMed] [Google Scholar]
- Alexander D., Schafer F., Olbrich M., Friedrich B., Buhring H.J., Hoffmann J., Reinert S. MSCA-1/TNAP selection of human jaw periosteal cells improves their mineralization capacity. Cell. Physiol. Biochem. 2010;26:1073–1080. doi: 10.1159/000323985. [DOI] [PubMed] [Google Scholar]
- Bassett C.A., Pawluk R.J., Pilla A.A. Acceleration of fracture repair by electromagnetic fields. A surgically noninvasive method. Ann. N. Y. Acad. Sci. 1974;238:242–262. doi: 10.1111/j.1749-6632.1974.tb26794.x. [DOI] [PubMed] [Google Scholar]
- Bassett C.A., Pawluk R.J., Pilla A.A. Augmentation of bone repair by inductively coupled electromagnetic fields. Science. 1974;184:575–577. doi: 10.1126/science.184.4136.575. [DOI] [PubMed] [Google Scholar]
- Bawin S.M., Kaczmarek L.K., Adey W.R. Effects of modulated VHF fields on the central nervous system. Ann. N. Y. Acad. Sci. 1975;247:74–81. doi: 10.1111/j.1749-6632.1975.tb35984.x. [DOI] [PubMed] [Google Scholar]
- Binhi V.N., Savin A.V. Molecular gyroscopes and biological effects of weak extremely low-frequency magnetic fields. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2002;65:051912. doi: 10.1103/PhysRevE.65.051912. [DOI] [PubMed] [Google Scholar]
- Blank M., Goodman R. DNA is a fractal antenna in electromagnetic fields. Int. J. Radiat. Biol. 2011;87:409–415. doi: 10.3109/09553002.2011.538130. [DOI] [PubMed] [Google Scholar]
- Ciombor D.M., Aaron R.K. The role of electrical stimulation in bone repair. Foot Ankle Clin. 2005;10:579–593. doi: 10.1016/j.fcl.2005.06.006. vii. [DOI] [PubMed] [Google Scholar]
- Dobreva G., Chahrour M., Dautzenberg M., Chirivella L., Kanzler B., Farinas I., Karsenty G., Grosschedl R. SATB2 is a multifunctional determinant of craniofacial patterning and osteoblast differentiation. Cell. 2006;125:971–986. doi: 10.1016/j.cell.2006.05.012. [DOI] [PubMed] [Google Scholar]
- Ducy P., Zhang R., Geoffroy V., Ridall A.L., Karsenty G. Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation. Cell. 1997;89:747–754. doi: 10.1016/s0092-8674(00)80257-3. [DOI] [PubMed] [Google Scholar]
- Ehnert S., Baur J., Schmitt A., Neumaier M., Lucke M., Dooley S., Vester H., Wildemann B., Stockle U., Nussler A.K. TGF-beta1 as possible link between loss of bone mineral density and chronic inflammation. PLoS ONE. 2010;5:e14073. doi: 10.1371/journal.pone.0014073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehnert S., Seeliger C., Vester H., Schmitt A., Saidy-Rad S., Lin J., Neumaier M., Gillen S., Kleeff J., Friess H., Burkhart J., Stockle U., Nussler A.K. Autologous serum improves yield and metabolic capacity of monocyte-derived hepatocyte-like cells: possible implication for cell transplantation. Cell Transplant. 2011;20:1465–1477. doi: 10.3727/096368910X550224. [DOI] [PubMed] [Google Scholar]
- Ehnert S., Freude T., Ihle C., Mayer L., Braun B., Graeser J., Flesch I., Stöckle U., Nussler A.K., Pscherer S. Factors circulating in the blood of type 2 diabetes mellitus patients affect osteoblast maturation — description of a novel in vitro model. Exp. Cell Res. 2015;332:247–258. doi: 10.1016/j.yexcr.2014.12.011. [DOI] [PubMed] [Google Scholar]
- Foley L.E., Gegear R.J., Reppert S.M. Human cryptochrome exhibits light-dependent magnetosensitivity. Nat. Commun. 2011;2:356. doi: 10.1038/ncomms1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freude T., Braun K.F., Haug A., Pscherer S., Stockle U., Nussler A.K., Ehnert S. Hyperinsulinemia reduces osteoblast activity in vitro via upregulation of TGF-beta. J. Mol. Med. 2012;90:1257–1266. doi: 10.1007/s00109-012-0948-2. [DOI] [PubMed] [Google Scholar]
- Friedman J., Kraus S., Hauptman Y., Schiff Y., Seger R. Mechanism of short-term ERK activation by electromagnetic fields at mobile phone frequencies. Biochem. J. 2007;405:559–568. doi: 10.1042/BJ20061653. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Goodman R., Lin-Ye A., Geddis M.S., Wickramaratne P.J., Hodge S.E., Pantazatos S.P., Blank M., Ambron R.T. Extremely low frequency electromagnetic fields activate the ERK cascade, increase hsp70 protein levels and promote regeneration in Planaria. Int. J. Radiat. Biol. 2009;85:851–859. doi: 10.1080/09553000903072488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin X.L., Warner F., Costa M. The role of electromagnetic stimulation in the management of established non-union of long bone fractures: what is the evidence? Injury. 2008;39:419–429. doi: 10.1016/j.injury.2007.12.014. [DOI] [PubMed] [Google Scholar]
- Haddad J.B., Obolensky A.G., Shinnick P. The biologic effects and the therapeutic mechanism of action of electric and electromagnetic field stimulation on bone and cartilage: new findings and a review of earlier work. J. Altern. Complement. Med. 2007;13:485–490. doi: 10.1089/acm.2007.5270. [DOI] [PubMed] [Google Scholar]
- Hannouche D., Petite H., Sedel L. Current trends in the enhancement of fracture healing. J. Bone Joint Surg. (Br.) 2001;83:157–164. doi: 10.1302/0301-620x.83b2.12106. [DOI] [PubMed] [Google Scholar]
- Hong J.M., Kang K.S., Yi H.G., Kim S.Y., Cho D.W. Electromagnetically controllable osteoclast activity. Bone. 2014;62:99–107. doi: 10.1016/j.bone.2014.02.005. [DOI] [PubMed] [Google Scholar]
- Kim S., Koga T., Isobe M., Kern B.E., Yokochi T., Chin Y.E., Karsenty G., Taniguchi T., Takayanagi H. Stat1 functions as a cytoplasmic attenuator of Runx2 in the transcriptional program of osteoblast differentiation. Genes Dev. 2003;17:1979–1991. doi: 10.1101/gad.1119303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowry O.H., Rosebrough N.J., Farr A.L., Randall R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- Muehsam D.J., Pilla A.A. A Lorentz model for weak magnetic field bioeffects: part I—thermal noise is an essential component of AC/DC effects on bound ion trajectory. Bioelectromagnetics. 2009;30:462–475. doi: 10.1002/bem.20494. [DOI] [PubMed] [Google Scholar]
- Muehsam D.J., Pilla A.A. A Lorentz model for weak magnetic field bioeffects: part II—secondary transduction mechanisms and measures of reactivity. Bioelectromagnetics. 2009;30:476–488. doi: 10.1002/bem.20493. [DOI] [PubMed] [Google Scholar]
- Nakashima K., Zhou X., Kunkel G., Zhang Z., Deng J.M., Behringer R.R., de Crombrugghe B. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell. 2002;108:17–29. doi: 10.1016/s0092-8674(01)00622-5. [DOI] [PubMed] [Google Scholar]
- Rubin J., McLeod K.J., Titus L., Nanes M.S., Catherwood B.D., Rubin C.T. Formation of osteoclast-like cells is suppressed by low frequency, low intensity electric fields. J. Orthop. Res. 1996;14:7–15. doi: 10.1002/jor.1100140104. [DOI] [PubMed] [Google Scholar]
- Schmitt A., Ehnert S., Schyschka L., Buschner P., Kuhnl A., Dobele S., Siebenlist S., Lucke M., Stockle U., Nussler A.K. Monocytes do not transdifferentiate into proper osteoblasts. ScientificWorldJournal. 2012;2012:384936. doi: 10.1100/2012/384936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeliger C., Falldorf K., Sachtleben J., Griensven M. Low-frequency pulsed electromagnetic fields significantly improve time of closure and proliferation of human tendon fibroblasts. Eur. J. Med. Res. 2014;19:37. doi: 10.1186/2047-783X-19-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skehan P., Storeng R., Scudiero D., Monks A., McMahon J., Vistica D., Warren J.T., Bokesch H., Kenney S., Boyd M.R. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst. 1990;82:1107–1112. doi: 10.1093/jnci/82.13.1107. [DOI] [PubMed] [Google Scholar]
- Strauch B., Herman C., Dabb R., Ignarro L.J., Pilla A.A. Evidence-based use of pulsed electromagnetic field therapy in clinical plastic surgery. Aesthet. Surg. J. 2009;29:135–143. doi: 10.1016/j.asj.2009.02.001. [DOI] [PubMed] [Google Scholar]
- Thomas A.W., Graham K., Prato F.S., McKay J., Forster P.M., Moulin D.E., Chari S. A randomized, double-blind, placebo-controlled clinical trial using a low-frequency magnetic field in the treatment of musculoskeletal chronic pain. Pain Res. Manag. 2007;12:249–258. doi: 10.1155/2007/626072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tofani S., Barone D., Cintorino M., de Santi M.M., Ferrara A., Orlassino R., Ossola P., Peroglio F., Rolfo K., Ronchetto F. Static and ELF magnetic fields induce tumor growth inhibition and apoptosis. Bioelectromagnetics. 2001;22:419–428. doi: 10.1002/bem.69. [DOI] [PubMed] [Google Scholar]
- Wang Z., Ehnert S., Ihle C., Schyschka L., Pscherer S., Nussler N.C., Braun K.F., Van Griensven M., Wang G., Burgkart R., Stöckle U., Gebhard F., Vester H., Nussler A.K. Increased oxidative stress response in granulocytes from older patients with a hip fracture may account for slow regeneration. Oxidative Med. Cell. Longev. 2014;2014:819847. doi: 10.1155/2014/819847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yumoto H., Hirao K., Tominaga T., Bando N., Takahashi K., Matsuo T. Electromagnetic wave irradiation promotes osteoblastic cell proliferation and up-regulates growth factors via activation of the ERK1/2 and p38 MAPK pathways. Cell. Physiol. Biochem. 2015;35:601–615. doi: 10.1159/000369722. [DOI] [PubMed] [Google Scholar]
- Zhang X., Liu X., Pan L., Lee I. Magnetic fields at extremely low-frequency (50 Hz, 0.8 mT) can induce the uptake of intracellular calcium levels in osteoblasts. Biochem. Biophys. Res. Commun. 2010;396:662–666. doi: 10.1016/j.bbrc.2010.04.154. [DOI] [PubMed] [Google Scholar]
- Zhou J., Ming L.G., Ge B.F., Wang J.Q., Zhu R.Q., Wei Z., Ma H.P., Xian C.J., Chen K.M. Effects of 50 Hz sinusoidal electromagnetic fields of different intensities on proliferation, differentiation and mineralization potentials of rat osteoblasts. Bone. 2011;49:753–761. doi: 10.1016/j.bone.2011.06.026. [DOI] [PubMed] [Google Scholar]
- Zhou J., Wang J.Q., Ge B.F., Ma X.N., Ma H.P., Xian C.J., Chen K.M. Different electromagnetic field waveforms have different effects on proliferation, differentiation and mineralization of osteoblasts in vitro. Bioelectromagnetics. 2014;35:30–38. doi: 10.1002/bem.21794. [DOI] [PubMed] [Google Scholar]