Abstract
The treatment of musculoskeletal neoplasms and infection is usually based on an initial diagnostic biopsy.
Prior to biopsy, a hypothesis should be formed about the most likely diagnosis and a differential diagnosis. These deliberations should consider whether the lesion is a primary benign or malignant tumour, a metastasis, a haematological problem or an infection.
A tactical plan should be developed which evaluates the necessity, the risk, the approach and finally defines the technique of biopsy most likely to achieve a representative result in the clinical case.
In developing this technical approach, the pitfalls should be anticipated, i.e. inadequate sampling, difficulty of pathological interpretation and contamination.
The tactical approach should be developed in conjunction with a multi-disciplinary team together with appropriate pre-biopsy imaging.
Cite this article: EFORT Open Rev 2017;2:51–57. DOI: 10.1302/2058-5241.2.160065
Keywords: tumours, musculoskeletal, multi-disciplinary approach, imaging, biopsy techniques, complications
Introduction
The basic concept of modern medicine is that treatment is based upon accurate diagnosis. In musculoskeletal problems and abnormalities, the diagnosis is frequently based upon pathological examination of a specimen taken from the lesion. This is termed a biopsy. The label of ‘biopsy’ refers to the process of removal of the piece of tissue itself, as well as the examination of that sample by an appropriate specialist.
It may be considered that biopsy itself is a simple technical procedure, but it can have the potential of adversely affecting the outcome of the case, particularly in malignant tumours. The importance of proper planning and performance of appropriate biopsy in musculoskeletal lesions is well-known and a recent literature review by Traina et al1 outlines the general approach to performing biopsies of bone and soft tissue, respectively. As there is no single standard approach for biopsy for very different conditions, personal opinions and judgement influence the decisions involved in a biopsy. Biopsies are inevitably based upon scientific discussion, particularly of pre-biopsy imaging and the personal experience and expertise of the multi-disciplinary team (MDT). A biopsy performed at a centre where there are experienced radiologists, pathologists, orthopaedic surgeons and oncologists will generally have greater accuracy and lower complication rates.2 This is particularly important with the increasing use of limb-salvage surgery in primary treatment involving musculoskeletal malignancies.
Obviously, a successful biopsy must cause minimal harm to the patient and ensure that sufficient viable and representative tissue samples can be presented in an appropriate form to the pathologist. Harm can occur from taking a biopsy from a lesion that does not require biopsy as appropriate management, by damage to neurovascular structures or by inadvertent contamination of an uninvolved anatomical compartment. These mistakes are minimised by pre-operative tactical discussion in a MDT setting. Acquisition of tissue can be via open biopsy or needle biopsy. The latter includes both fine needle aspiration (FNA) and core needle biopsy. At present the most popular method is core needle biopsy.
It can be assumed that the purpose of the biopsy is to produce an accurate diagnosis. The topic of accuracy has been diagnosed by Layfield et al.3 Diagnostic accuracy is assessed as the number of correct cases as a percentage of the total performed. Therapeutic accuracy reports the number of correctly treated cases as a percentage of the total cases under study. The question that the surgeon needs to answer from the biopsy is the usual question of benign versus malignant, is the diagnosis absolutely specific, is a grading of tissue possible? Obviously the amount of tissue available plays a role and most studies show a slight superiority in diagnostic accuracy for core needle biopsy over FNA.4 However, even FNA has a reported diagnostic accuracy above 90%.5 A combination of the two methods has been reported as giving an overall diagnostic accuracy of 96%.6
In a recent study, the highest rate of non-diagnostic biopsies was for bone lymphoma (22%).7 Out of our own last 14 cases with bone lymphoma, the first biopsy was not conclusive in four out of 14. Because of high diagnostic suspicion, repeat biopsy was performed and a diagnosis was gained in three cases, and in one case a third biopsy was required. It is also recognised that bone lymphoma also presents difficulties for the pathologist. Figure 1 depicts the characteristic compression artefacts resulting in failure to diagnose bone lymphoma by a pathologist not familiar with this artefact. Faced with the artefact an experienced pathologist will carefully search for cells that do not show the artefact and eventually lead to the correct diagnosis. This case illustrates the need for close cooperation between clinician and pathologist and the role of an MDT.
Fig. 1.
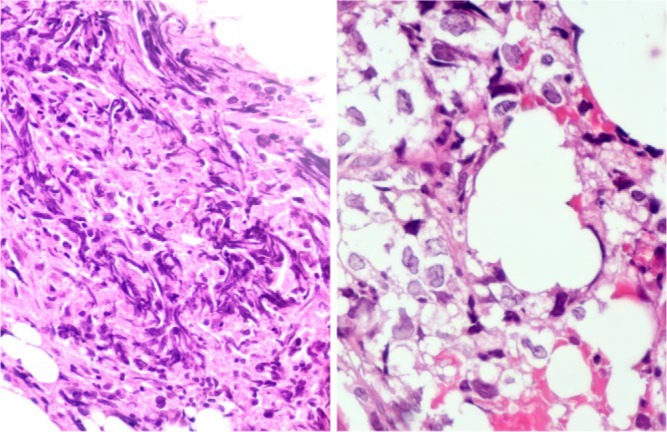
Microphotographs of haematoxylin and eosin (H&E)-stained histological section of a core biopsy of a classical bone lymphoma: low power view (left side, 50× magnification) shows the characteristic shearing artefact with streaks of disrupted tumour cell nuclei, obliterating the nuclear detail and often preventing diagnosis in most segments of the biopsy tissue (left side). Focal preservation of the characteristic cellular and nuclear detail with clear cytoplasm and polylobated nuclei with somewhat coarser chromatin at high power magnification (right side, 400× magnification) leads to the correct diagnosis of diffuse large cell lymphoma.
The indication for biopsy
Most primary bone lesions present with focal bone pain and/or swelling and with a focal or multi-focal radiological abnormality. However, with increasing radiological sophistication, a number of variations of normal radiological artefacts or non-neoplastic lesions arise. Our experience is that approximately 10% of MDT referrals are non-neoplastic. These may be congenital, traumatic, degenerative, infective or inflammatory in origin. Infected lesions are common, both acute8 and chronic, including chronic recurrent multi-focal osteomyelitis (CRMO),9 Brodie’s abscess10 and tuberculosis11,12 which can all mimic a primary neoplasm. Perhaps the most common lesion-mimicking bone tumour or infection is a stress fracture.13
Similarly, many sarcomata, particularly Ewing’s sarcoma, are well documented to mimic infection and wherever infection is thought a possibility, biopsy of the lesion should be sent for microbiological culture to assess sensitivity to antibiotics and for exclusion of the diagnosis of fungal infection or tuberculosis.14,15
Tactical approach and MDT discussion
As stated above, the indication for biopsy is usually a radiological abnormality. However, the MDT need to take into account the patient’s history and clinical findings prior to developing a working hypothesis. Many lesions, such as non-ossifying fibroma, fibrous dysplasia and osteoma, do not require a biopsy and biopsy of such lesions may even be misleading and distracting for the pathologist. As part of this hypothesis, the following questions should be discussed:
Does the lesion have its origin in the bone (primary bone tumour) or is it a metastasis?
Could the lesion be an expression of an underlying metabolic disease (Brown tumour caused by hyperparathyroidism) or could the process itself be causing a metabolic aberration?16
Could the process be an infection, especially in spinal lesions where tuberculosis can be more common?
Has the lesion a haematogenous origin, e.g. chloroma, lymphoma, plasmocytoma? In such cases, immune electrophoresis may be helpful prior to biopsy.
We consider that unselected screening blood tests are not usually very helpful but the simple metabolic studies, i.e. serum calcium, phosphorus, alkaline phosphatase, together with immune electrophoresis may add important information. Traditionally, plain radiographs are analysed according to the criteria of Lodwick,17 which can help to differentiate a destructive lesion, a bone-forming lesion, calcification within a chondrogenic process or a reaction to a neoplastic process or periosteal reaction. Although localisation of the lesion within a bone is also an important differential, it suggests that growth dynamics are best assessed by utilisation of PET-CT for systemic screening and assessment of local metabolic activity of a tumour. Whether PET-CT is performed before or after the biopsy, depends on the type of lesion suspected. Most clinicians, however, would prefer that it was performed prior to biopsy if at all possible.
Pre-biopsy imaging
Once a decision has been made to perform a biopsy, planning should occur in an MDT setting. Although initial referral imaging may be adequate for deciding whether a biopsy is needed, imaging may have to be repeated where the whole lesion has not been covered or in the case of malignant disease the whole bone has not been imaged. For both bone and soft-tissue sarcomata, MRI is the technique of choice and this will give additional information on the type of biopsy to be considered in order to supply sufficient tissue for the pathologist, the approach and the appropriate part of the lesion to target. It will also define areas which need to be avoided, such as the neurovascular bundle and the team will also be able to decide which imaging technique to use for guidance of the biopsy itself. In cases where an additional lesion is discovered within the bone, which does not have the typical appearance of a skip metastasis, the second lesion may also require biopsy. MRI is also critical in defining whether there is local joint involvement.
The approach to be used by the biopsy must be discussed with the surgical team who will perform the actual appropriate resection of the tumour. This is because it is generally considered that needle track resection at the time of the definitive surgery is strongly advised.18 The small biopsy scar associated with core needle biopsy is usually visible for two to three months following performance, but if there is going to be significant delay, marking of the biopsy site with Indian ink should be considered.
Analgesia
The majority of bone and soft-tissue tumours in the appendicular skeleton can be biopsied under local anaesthetic without or without conscious sedation. Extensive local anaesthetic infiltration of the periosteum at the biopsy site is necessary to achieve adequate analgesia and this carries a theoretical risk of spread of the tumour. In children and in cases associated with pathological fracture, a local anaesthetic is unlikely to be tolerated. The biopsy of lesions in the posterior capsule of the knee, pelvic lesions and vertebral lesions may also be best performed on occasion from the use of general anaesthesia or sedation.
The types of biopsy and associated techniques
The biopsy should be as small as possible but as large as is needed to obtain material and make a diagnosis. Small biopsies have the advantage of being less invasive, but the diagnostic yield is smaller. Large biopsies provide more material for more studies and allow preservation of material where required for research. Statistically they result in higher accuracy but carry a risk of more local complications.
Excisional biopsy
An excisional biopsy is the complete removal of the lesion. It is clear that this is permitted in potentially malignant processes only, if definitive treatment without the risk of contamination or marginality (penetration of the margins of the lesion) can be achieved. Furthermore, in cases which should receive chemotherapy, it may be important to have the tumour in place to observe the effects of the neo-adjuvant chemotherapy prior to resection.
Incisional biopsy
Incisional biopsy has long been considered to be to be the gold standard. Certainly, large samples of tumour tissue can be achieved and the accuracy is reported as up to 95%.20 It carries with it the increased risks of haematoma which may delay treatment as well as an increased risk of local dissemination of tumour. It usually requires an inpatient setting and greater complications are reported.21
Core biopsies
Core biopsies provide a cylinder of tissue for histological examination by a pathologist. They provide more material than FNA and hence more pathological analysis can be performed. Presently, it is the most frequently used technique in the diagnostic work-up of soft-tissue and bone lesions. It usually uses systems of Gauge 8 to 18.
Where soft tissue is involved, the instruments are usually spring-loaded, e.g. Achieve® Automatic biopsy device by CareFusion (Fig. 2). It releases a cannula and stilette sequentially at high speed. It can be used within a guiding needle (co-axial system) to take several specimens with only one skin penetration. In addition, the co-axial system can be used for decontamination of the tract by radiofrequency.
Fig. 2.
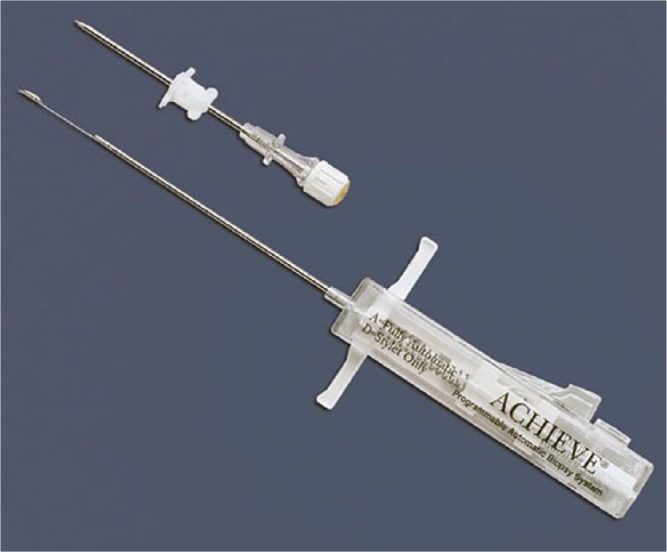
The spring-loaded CareFusion® Core Biopsy device with the cannula for co-axial procedures.
For bone biopsy, our preferred instrument is the T-handle Jamshidi biopsy needle by CareFusion. An acquisition cradle increases the potential of removing a biopsy cylinder (Fig. 3). The needle can be partially withdrawn and re-directed within the lesion for more probes to be removed. Because occasionally very hard bone is encountered we have developed a power-driven cannulated special drill system with an external diameter of 8 mm and an internal diameter of 5 mm (Fig. 4). If no tissue can be removed either with the needle or the drill system, a fine rongeur (Figs 5 and 6) can be inserted through the 8 Gauge Jamshidi needle or an even larger standard rongeur can be inserted through the opening created by the drill to allow material to be acquired. This is particularly useful in myxoid and cystic lesions. In suspected aneurysmal bone cysts, the use of contrast medium to confirm the venous drainage of the lesion can be considered and a sclerosant injection performed at the time of the core needle biopsy. The reported accuracy of core needle biopsy is in the range of 74%22 to 93%23-26 with complication rates in the range of 1%26 to 2%.27 No significant difference in accuracy of diagnosing soft-tissue or bone lesions has ever been demonstrated.27 The paediatric literature and meta-analysis has a documented accuracy of 94% with complications of 1%.28 However, it is worth noticing that the accuracy of this technique has been shown to decrease in the biopsy of spinal lesions (61%) and in cases of infection (50%).22 In a study of 151 consecutive core needle biopsies, factors affecting diagnostic yield included the type of lesion (87%), lytic bone lesion (57%), sclerotic bone lesions, lesion size (54%), less than 2 cm (86%), lesions greater than 5 cm, longer length specimens and increasing numbers of specimens obtained. Again, no difference was identified between soft-tissue and bone lesions by imaging modality used or by needle size. The minimum of three specimens for bone lesions and four for soft-tissue lesions has been proposed.29
Fig. 3.
CareFusion® T-handle Jamshidi® bone biopsy set.
Fig. 4.

The cannulated power drill system for larger core bone biopsies with rongeurs that can be inserted through the drill holes after removal, especially in cystic lesions.
Fig. 5.

Fine rongeur, that can be inserted through the cannulated drill or even through the 8G Jamshidi needle shown in Fig. 2.
Fig. 6.
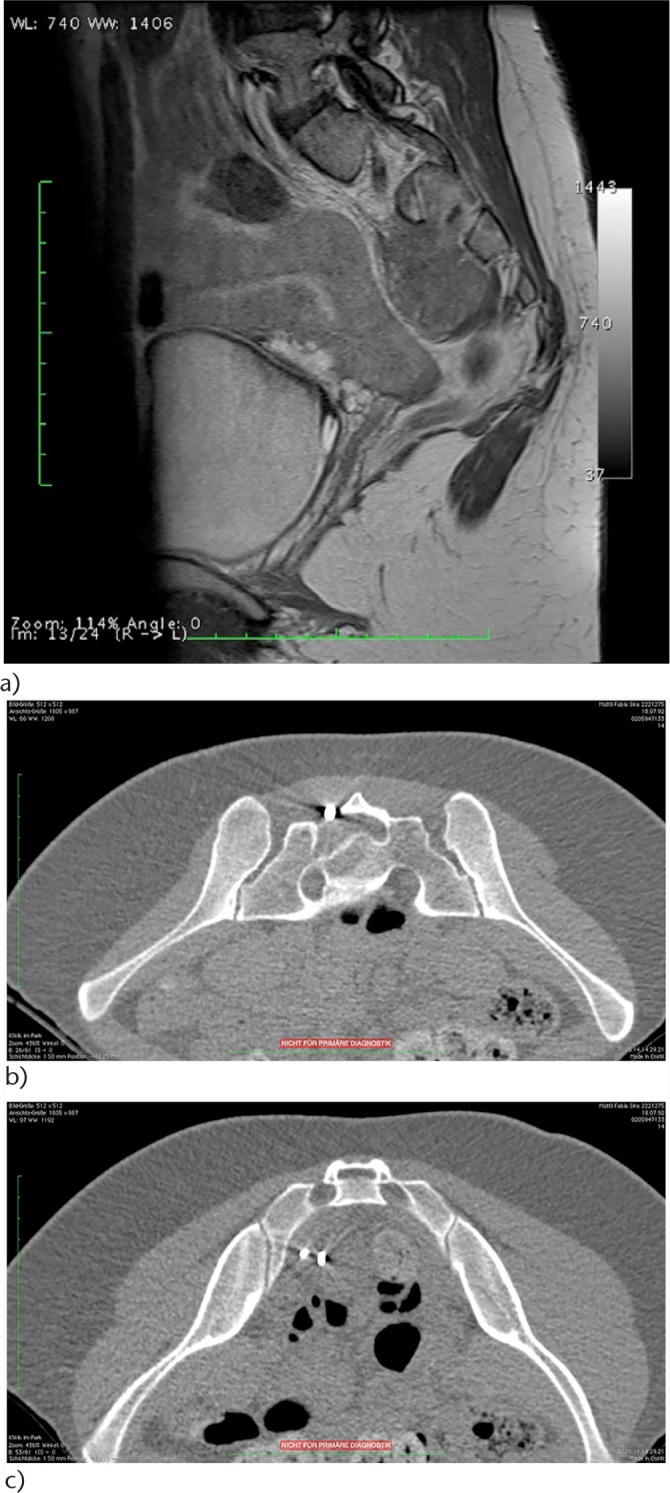
a) Mass involving the sacrum. Dilated neurogenic bladder. Two non-diagnostic posterior core biopsies, one non-diagnostic open posterior biopsy. b) Nine months following the first non-diagnostic needle and open posterior attempts the diagnostic transforaminal (S2) CT-guided rongeur biopsy was performed by the author. Ewing sarcoma: top: posterior entry of the rongeur; bottom: rongeur within the pre-sacral lesion.
Fine needle aspiration
In this technique, a fine hollow needle is inserted into the lesion and a syringe applied and aspirating material for pathological examination of the cells (cytology and/or cultures). Usually the needle is guided by ultrasound and several passes may be used to increase yield. This is usually performed under local anaesthetic. This technique is particularly useful in high-grade soft-tissue sarcoma where typing of individual cells or small tissue aggregates by conventional staining, immunohistochemistry or fluorescent in-situ hybridisation (FISH) will provide a diagnosis. It is the least invasive of the biopsy procedures and has the lowest risk of seeding of tumour along the needle tract.30,31 Studies have shown that in bone an excellent accuracy can be achieved from 97% to 100%32,33 and therefore it has been suggested as an effective initial investigation.34 The main disadvantage, of course, is that it only offers cytological features of the lesion. Although this can be accurate in assessing metastatic bone lesions,35 a more recent comparison with core needle biopsy shows that the latter appears to be better in establishing the histological type and grade and achieving a specific diagnosis.20,25
The type of imaging technique utilised during the biopsy can vary dependent upon a number of circumstances. Soft-tissue lesions or extra-osseous components of bone lesions in the appendicular skeleton can usually be assessed by ultrasound guidance. The additional use of colour Doppler can identify neovascularity within the tumour and such areas are more likely to be viable and should therefore be targeted. An adjacent neurovascular bundle can also be identified and avoided. Deep soft tissues around the pelvis or shoulder girdles or small lesions near neurovascular structures are perhaps more appropriately biopsied under CT guidance. CT is also perhaps best for intra-cortical lesions. Fluoroscopy is a very useful technique, particularly in the periphery and in lytic lesions. In studies comparing these three imaging modalities, accuracy has been shown to be very similar.35
Complications
Tumour seeding and joint contamination
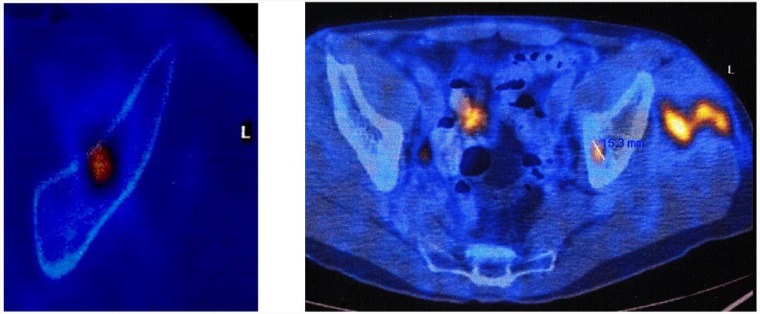
Although there are a number of individual studies which have reported seeding in a biopsy tract, the true incidence is unknown.36 In a variety of malignant tumours, the incidence of needle tract seeding has been estimated at 0.003% and 0.009%.37 As can be expected, given the rarity, several studies38,39 reported no risk of recurrence related to a biopsy tract, although tumour contamination in the biopsy tract, even in percutaneous techniques, has to be considered a real risk, re-inforcing the need for removal of the biopsy tract during tumour resection or already at biopsy (Fig. 7). This contamination of the biopsy tract and the need to remove it en bloc with the tumour resection was established in 1982.40 An alternative way of sterilising the biopsy tract is by the radiofrequency technique described above (Fig. 7).
Fig. 7.
Metastasis of cholangiocarcinoma (left). The metastasis was successfully inactivated by radiofrequency. The needle was removed after cooling. A new metastasis and biopsy proven contamination of the biopsy path is shown on the right.
Cells showing characteristic Ewing’s sarcoma translocations have been demonstrated in circulating blood during uncontaminated tumour removal, but no relationship to survival has been established.41 Whether such dissemination occurs during biopsy is unknown. In animal studies, after core needle biopsy an increased frequency of metastases from experimental carcinoma has been demonstrated.42,43 Undoubtedly, there must be some protective effect from chemotherapy against problems related to biopsy tract contamination. It can be assumed that this is likely in tumours which are successfully treated with chemotherapy and radiotherapy alone, such as lymphomas and some Ewing’s sarcoma. During needle biopsy, particular care must be taken as to avoid joint contamination. This is a particular risk in the supra-patellar pouch of the knee joint.
Bleeding
Bleeding is a well-recognised risk of any biopsy technique and particular care should be taken with those tumours which have a particular vascular structure.44 Bleeding into adjacent dead spaces can be difficult to control and the use of a pressure bandage should be considered.
Neural damage
It is well recognised that neural structures can be damaged by a cutting needle; however, pre-procedure planning and use of CT can usually avoid these problems. Instrument breakage that retains metal, smaller biopsy needles can break. Procedures should be performed at centres where orthopaedic surgeons can resect retained foreign bodies without difficulty.
Fracture
Great care has to be exhibited in lesions which are very lytic and on the verge of fracture. A biopsy can be the last straw in the integrity of the bone. Again, biopsy of such lesions in treating centres is ideal.
It can be seen that biopsy of bone and soft-tissue neoplasms is an essential component of the management of these difficult conditions. Although there have been algorithms presented for recommended biopsy approaches, we consider that the use of a MDT approach is essential to ensure that biopsy is, first, necessary, and, second, will ensure that it is successful. One should proceed with the least-invasive method following determination of the biopsy path and this can be based on an agreement between the treating surgeon, the radiologist, the pathologist and oncologist in the MDT setting. Pre-operative MRI to stage the lesion means that the highest-grade component of the lesion can be targeted by the biopsy clinician. The technical procedure and the steps required can be planned and executed based on the appropriate imaging. Such an approach should result in the highest diagnostic yield for the patient and ensure the lowest possible rate of complications.
Footnotes
ICMJE Conflict of interest statement: None
Funding
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
References
- 1. Traina F, Errani C, Toscano A, et al. Current concepts in the biopsy of musculoskeletal tumours. AAOS Exhibit Selection. J Bone & Joint Surg [Am] 2015;97:e7. [DOI] [PubMed] [Google Scholar]
- 2. Mankin HJ, Mankin CJ, Simon MA. The hazards of the biopsy, revisited. Members of the Musculoskeletal Tumor Society. J Bone Joint Surg [Am] 1996;78-A:656-663. [DOI] [PubMed] [Google Scholar]
- 3. Layfield LJ, Schmidt RL, Sangle N, Crim JR. Diagnostic accuracy and clinical utility of biopsy in musculoskeletal lesions: a comparison of fine-needle aspiration, core, and open biopsy techniques. Diagn Cytopathol 2014;42:476-486. [DOI] [PubMed] [Google Scholar]
- 4. Kasraeian S, Allison DC, Ahlmann ER, Fedenko AN, Menendez LR. A comparison of fine-needle aspiration, core biopsy, and surgical biopsy in the diagnosis of extremity soft tissue masses. Clin Orthop Relat Res 2010;468:2992-3002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Dey P, Mallik MK, Gupta SK, Vasishta RK. Role of fine needle aspiration cytology in the diagnosis of soft tissue tumours and tumour-like lesions. Cytopathology 2004;15:32-37. [DOI] [PubMed] [Google Scholar]
- 6. Logan PM, Connell DG, O’Connell JX, Munk PL, Janzen DL. Image-guided percutaneous biopsy of musculoskeletal tumors: an algorithm for selection of specific biopsy techniques. AJR Am J Roentgenol 1996;166:137-141. [DOI] [PubMed] [Google Scholar]
- 7. Chang CY, Huang AJ, Bredella MA, et al. Percutaneous CT-guided needle biopsies of musculoskeletal tumors: a 5-year analysis of non-diagnostic biopsies. Skeletal Radiol 2015;44:1795-1803. [DOI] [PubMed] [Google Scholar]
- 8. Sheppard JE, Switlick DN. Coccidioides immitis osteomyelitis of the radius presenting as Ewing’s sarcoma. Orthopedics 2008;31:607. [PubMed] [Google Scholar]
- 9. Sundaram M, McDonald D, Engel E, Rotman M, Siegfried EC. Chronic recurrent multifocal osteomyelitis: an evolving clinical and radiological spectrum. Skeletal Radiol 1996;25:333-336. [DOI] [PubMed] [Google Scholar]
- 10. Abril JC, Castillo F, Casas J, Díaz A. Brodie’s abscess of the hip simulating osteoid osteoma. Orthopedics 2000;23:285-287. [DOI] [PubMed] [Google Scholar]
- 11. Yip KM, Lin J, Leung PC. Cystic tuberculosis of the bone mimicking osteogenic sarcoma. Tuber Lung Dis 1996;77:566-568. [DOI] [PubMed] [Google Scholar]
- 12. Kalbermatten DF, Kalbermatten NT, Siebenrock KA. Osteolytic lesion in the greater trochanter mimicking tumor. Arch Orthop Trauma Surg 2002;122:53-55. [DOI] [PubMed] [Google Scholar]
- 13. Solomon L. Stress fractures of the femur and tibia simulating malignant bone tumous. S Afr J Surg 1974;12:19-25. [PubMed] [Google Scholar]
- 14. Jordanov MI, Block JJ, Gonzalez AL, Green NE. Transarticular spread of Ewing sarcoma mimicking septic arthritis. Pediatr Radiol 2009;39:381-384. [DOI] [PubMed] [Google Scholar]
- 15. Datir A, Lidder S, Pollock R, Tirabosco R, Saifuddin A. High-grade chondrosarcoma mimicking Brodie’s abscess. Clin Radiol 2009;64:944-947. [DOI] [PubMed] [Google Scholar]
- 16. Koplas MC, Rubin BP, Sundaram M. Phosphaturic mesenchymal tumor: two contrasting cases. Skeletal Radiol 2014;43:841-845. [DOI] [PubMed] [Google Scholar]
- 17. Lodwick GS, Wilson AJ, Farrell C, Virtama P, Dittrich F. Determining growth rates of focal lesions of bone from radiographs. Radiology 1980;134:577-583. [DOI] [PubMed] [Google Scholar]
- 18. Schwartz HS, Spengler DM. Needle tract recurrences after closed biopsy for sarcoma: three cases and review of the literature. Ann Surg Oncol 1997;4:228-236. [DOI] [PubMed] [Google Scholar]
- 19. Huvos AG. The importance of the open surgical biopsy in the diagnosis and treatment of bone and soft-tissue tumors. Hematol Oncol Clin North Am 1995;9:541-544. [PubMed] [Google Scholar]
- 20. Rougraff BT, Aboulafia A, Biermann JS, Healey J. Biopsy of soft tissue masses: evidence-based medicine for the musculoskeletal tumor society. Clin Orthop Relat Res 2009;467:2783-2791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ferguson PC, Sommerville S, Grimer RJ. Possible metastasis of osteosarcoma to a remote biopsy site: a case report. Clin Orthop Relat Res 2004;424:216-220. [DOI] [PubMed] [Google Scholar]
- 22. Hau A, Kim I, Kattapuram S, et al. Accuracy of CT-guided biopsies in 359 patients with musculoskeletal lesions. Skeletal Radiol 2002;31:349-353. [DOI] [PubMed] [Google Scholar]
- 23. van der Bijl AE, Taminiau AH, Hermans J, Beerman H, Hogendoorn PC. Accuracy of the Jamshidi trocar biopsy in the diagnosis of bone tumors. Clin Orthop Relat Res 1997;334:233-243. [PubMed] [Google Scholar]
- 24. Quinn SF, Demlow T, Dunkley B. Temno biopsy needle: evaluation of efficacy and safety in 165 biopsy procedures. AJR Am J Roentgenol 1992;158:641-643. [DOI] [PubMed] [Google Scholar]
- 25. Yang YJ, Damron TA. Comparison of needle core biopsy and fine-needle aspiration for diagnostic accuracy in musculoskeletal lesions. Arch Pathol Lab Med 2004;128:759-764. [DOI] [PubMed] [Google Scholar]
- 26. Dupuy DE, Rosenberg AE, Punyaratabandhu T, Tan MH, Mankin HJ. Accuracy of CT-guided needle biopsy of musculoskeletal neoplasms. AJR Am J Roentgenol 1998;171:759-762. [DOI] [PubMed] [Google Scholar]
- 27. Puri A, Shingade VU, Agarwal MG, et al. CT-guided percutaneous core needle biopsy in deep seated musculoskeletal lesions: a prospective study of 128 cases. Skeletal Radiol 2006;35:138-143. [DOI] [PubMed] [Google Scholar]
- 28. Sebire NJ, Roebuck DJ. Pathological diagnosis of paediatric tumours from image-guided needle core biopsies: a systematic review. Pediatr Radiol 2006;36:426-431. [DOI] [PubMed] [Google Scholar]
- 29. Wu JS, Goldsmith JD, Horwich PJ, Shetty SK, Hochman MG. Bone and soft-tissue lesions: what factors affect diagnostic yield of image-guided core-needle biopsy? Radiology 2008;248:962-970. [DOI] [PubMed] [Google Scholar]
- 30. Metcalfe MS, Bridgewater FHG, Mullin EJ, Maddern GJ. Useless and dangerous–fine needle aspiration of hepatic colorectal metastases. BMJ 2004;328:507-508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Akerman M. Fine-needle aspiration cytology of soft tissue sarcoma: benefits and limitations. Sarcoma 1998;2:155-161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Bommer KK, Ramzy I, Mody D. Fine-needle aspiration biopsy in the diagnosis and management of bone lesions: a study of 450 cases. Cancer 1997;81:148-156. [DOI] [PubMed] [Google Scholar]
- 33. Layfield LJ, Glasgow BJ, Anders KH, Mirra JM. Fine needle aspiration cytology of primary bone lesions. Acta Cytol 1987;31:177-184. [PubMed] [Google Scholar]
- 34. Wedin R, Bauer HC, Skoog L, Söderlund V, Tani E. Cytological diagnosis of skeletal lesions. Fine-needle aspiration biopsy in 110 tumours. J Bone Joint Surg [Br] 2000;82-B:673-678. [DOI] [PubMed] [Google Scholar]
- 35. Sung KS, Seo SW, Shon MS. The diagnostic value of needle biopsy for musculoskeletal lesions. Int Orthop 2009;33:1701-1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Davies NM, Livesley PJ, Cannon SR. Recurrence of an osteosarcoma in a needle biopsy track. J Bone Joint Surg [Br] 1993;75-B:977-978. [DOI] [PubMed] [Google Scholar]
- 37. Iemsawatdikul K, Gooding CA, Twomey EL, et al. Seeding of osteosarcoma in the biopsy tract of a patient with multifocal osteosarcoma. Pediatr Radiol 2005;35:717-721. [DOI] [PubMed] [Google Scholar]
- 38. Kaffenberger BH, Wakely PE, Jr, Mayerson JL. Local recurrence rate of fine-needle aspiration biopsy in primary high-grade sarcomas. J Surg Oncol 2010;101:618-621. [DOI] [PubMed] [Google Scholar]
- 39. Saghieh S, Masrouha KZ, Musallam KM, et al. The risk of local recurrence along the core-needle biopsy tract in patients with bone sarcomas. Iowa Orthop J 2010;30:80-83. [PMC free article] [PubMed] [Google Scholar]
- 40. Mankin HJ, Lange TA, Spanier SS. The hazards of biopsy in patients with malignant primary bone and soft-tissue tumors. J Bone Joint Surg [Am] 1982;64-A:1121-1127. [PubMed] [Google Scholar]
- 41. Grohs JG, Zoubek A, Jugovic D, Kovar H, Windhager R. Intraoperative dissemination of tumour cells in patients with Ewing tumours detected by RT-PCR. Int Orthop 2004;28:222-225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Mathenge EG, Dean CA, Clements D, et al. Core needle biopsy of breast cancer tumors increases distant metastases in a mouse model. Neoplasia 2014;16:950-960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kusukawa J, Suefuji Y, Ryu F, et al. Dissemination of cancer cells into circulation occurs by incisional biopsy of oral squamous cell carcinoma. J Oral Pathol Med 2000;29:303-307. [DOI] [PubMed] [Google Scholar]
- 44. Sheikh R, Bode B, Simovitch R, Exner GU. Hemangioma of the proximal humerus: bleeding hazard at surgery–a case report. J Shoulder Elbow Surg 2007;16:e11-e14. [DOI] [PubMed] [Google Scholar]