Abstract
Avascular necrosis (AVN) of the femoral head (FH) causes 5% to 12% of total hip arthroplasties (THA). It especially affects active male adults between the third and fifth decades of life. The exact worldwide incidence is unknown. There are only few data related to each country, but most of it relates to the United States.
Non-surgical management has a very limited role in the treatment of AVN of the FH and only in its earliest stages. Core decompression (CD) of the hip is the most common procedure used to treat the early stages of AVN of the FH. Recently, surgeons have considered combining CD with autologous bone-marrow cells, demineralised bone matrix or bone morphogenetic proteins or methods of angiogenic potential to enhance bone repair in the FH.
Manuscripts were deemed eligible for our review if they evaluated treatment of early stage AVN of the FH with biotechnology implanted via CD. After application of eligibility criteria, we selected 19 reports for final analysis.
The principal results showed that only by correctly mastering the therapeutic principles and adopting proper methods specifically oriented to different stages can the best therapeutic effect be achieved. Combining CD with biotechnology could result in a novel long-lasting hip- preserving treatment option.
Furthermore, more refined clinical studies are needed to establish the effectiveness of biotechnology treatments in AVN of the FH.
Cite this article: EFORT Open Rev 2017;2:41-50. DOI: 10.1302/2058-5241.2.150006
Keywords: AVN, avascular necrosis, osteonecrosis, ONFH, core decompression, CD, mesenchymal stem cell, MSC, bone morphogenetic protein, BMP, biotechnology, scaffold, autologous bone marrow
Introduction
Avascular necrosis (AVN) of the femoral head (FH) causes 5% to 12% of total hip arthroplasties (THA). It especially affects active male adults between the third and fifth decades of life.1 The exact worldwide incidence is unknown; there are only few data related to individual countries (e.g. Germany 5000 to 7000 patients per year).2 Most of these data relate to the United States.3,4 If left untreated, AVN can lead to FH collapse and hip joint destruction. There is a 50% chance of bilateral presentation.5 The aetiology and pathogenesis are still unclear, but a lot of risk factors have been identified through the years. Trauma, embolisation, smoking, corticosteroids, alcohol abuse, haemoglobinopathy, hyperbaric exposure, auto-immune disease, anti-tumour chemotherapy, Gaucher’s disease and Caisson disease are some of the risk factors that could act individually or synergistically to produce death of bone cells.
Early presentation can be asymptomatic; when the disease becomes symptomatic the most common manifestation is deep pain in the groin referred to the buttock or knee. Physical examination may be normal until the FH collapses. The diagnosis of AVN is based on plain anteroposterior and frog-leg radiographs and MRI8,9 which are used for classification and staging, i.e. Ficat,10 Steinberg11 and Association Research Circulation Osseous (ARCO).12
Non-surgical management has a very limited role in the treatment of AVN of the FH and only in the earliest stages.
Core decompression (CD) of the hip is the most common procedure used to treat early stages of AVN of the FH.13-15 Due to the frequent young age of these patients, the consequences of AVN often severely impact on their leisure activities and work status. There is consequently great interest in trying to preserve the FH whenever possible. Recently, surgeons have considered combining CD with autologous bone-marrow cells, demineralised bone matrix,16 bone morphogenetic proteins (BMPs),17 or vascular endothelial growth factor (VEGF) to enhance bone repair in the FH.
We report a systematic review of the current medical literature on the treatment of early stage AVN of the FH using biotechnologies implanted via CD.
Materials and methods
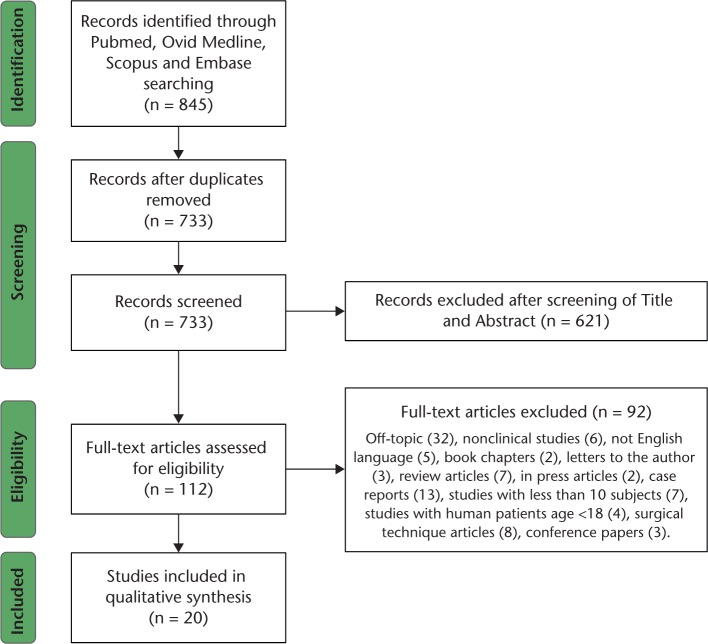
The PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) were followed to perform this systematic review of the literature and to present the results.18 A protocol was written stating the purpose of the review and the search strategy. A flow diagram according to PRISMA guidelines summarises our selection protocol (Fig. 1).
Fig. 1.
PRISMA flow diagram.
Eligibility criteria
We considered as eligible for our review manuscripts that used biotechnologies associated with CD for the treatment of the AVN of the FH in the ‘pre-collapse’ stage. Only clinical manuscripts were selected and we included studies only on human patients aged > 18 years. Only studies in the English language were eligible for inclusion for this review. Off-topic articles, book chapters, letters to the editor, review articles, press articles, case reports, studies with less than ten participants, surgical technique articles and conference papers were excluded.
Search strategy
A systematic, computerised search for potential manuscripts was performed independently by two investigators (AC, SM). Any discrepancies were resolved by discussion. PubMed, Ovid Medline, SCOPUS and EMBASE databases were used to identify studies between January 2000 and July 2015. Keywords used for the search were: AVN or avascular necrosis or ON or osteonecrosis and core decompression; AVN or avascular necrosis or ON or osteonecrosis and stem cells; AVN or avascular necrosis or ON or osteonecrosis and bone morphogenetic proteins or BMP; AVN or avascular necrosis or ON or osteonecrosis and biotechnology; AVN or avascular necrosis or ON or osteonecrosis and scaffold; AVN or avascular necrosis or ON or osteonecrosis and autologous bone marrow between January 2000 and July 2015. The search results were reviewed for duplicates. We performed a preliminary selection of the manuscripts reading the abstracts considered relevant by title. We then analysed full-length articles in order to include them in our review. We focused our attention on the references of selected articles with the aim to search other potential relevant articles. We used a summary table to collect the following data extracted from retrieved articles: study type; study design; sample size; treatment protocol; AVN classification; mean follow-up; and primary results.
Results
Study selection
We identified 845 abstracts using our electronic search; 733 met the initial screening inclusion criteria and the full-length articles were retrieved and reviewed. Following the full-length reviews, 112 studies met our eligibility criteria and were retained for this review. We then selected 19 eligible reports which were included in the final analysis (Table 1).
Table 1.
Comparison of the studies evaluated in the review
| Authors | Treatment protocol | No. of patients/hips | Initial AVN class | Mean follow-up (range), years | Results | Conclusion |
|---|---|---|---|---|---|---|
| Yoon et al (2011)20 | CD | 39/39 | Ficat I-III | 5.1 | Patients who have Ficat II or III disease have an increased rate of conversion to a THA (n=17/22 hips) than those had Ficat I disease (n=5/17 hips). | The ideal candidate has pre-collapse disease with lesions less than 15% of the size of the FH. |
| Iorio et al (1998)21 |
CD | 23/33 | Ficat I-IIB | 5.3 | Patients who have Ficat I disease have markedly higher 5-year survivorship than those with stage IIA and IIB disease (75% vs 30% vs 17%, respectively). | Excellent survivorship occurs for patients with Ficat I disease. |
| Mazieres et al (1997)22 |
CD | 20/20 | Ficat II | 2 | After a mean of 24 months, 50% of the hips showed signs of radiographic progression. 1/8 hips with smaller lesions showed disease progression, while 9/12 hips with larger lesions showed radiographic progression. | All decisions regarding the procedure should take into account whether the femoral head has collapsed as well as the volume of the lesions. |
| Mont et al (1998)23 |
CD | 68/68 | Steinberg III-IV | (4-12), 12 | Only 29% of total hips treated post-collapse (n=20) had satisfactory outcomes (no additional surgeries and HHS ⩾75 points). 41% of the Steinberg III hips (n=18/44 hips) required a THA and 92% of the stage IV hips (n=22/24 hips) underwent a THA. | Diagnosis before femoral head collapse is crucial for CD to be effective. |
| Mont et al (2004)24 | Multiple drilling | 35/45 | Ficat I-II | 2 | There were 32/45 hips with successful clinical result at a mean follow-up of two years. 24/30 stage I hips had a successful outcome compared with 8/15 stage II hips. No surgical complications. | This technique is effective in delaying a THR in young patients in early stages of AVN of the FH. |
| Song et al (2006)25 | CD vs multiple drilling | 136/163 | Ficat I | 7.2 | After a minimum follow-up of 5 years 79% of patients with stage I and 77% of stage II had no additional surgery. All small lesions (15/15) and 84% of medium-sized lesions were considered successful. Hips with a large necrotic area had poor results. | Multiple drilling is straightforward with few complications and produces results comparable to results of other core decompression technique. |
| Al Omran et al (2013)26 |
CD vs multiple drilling | 9/94 | Ficat I-II | 2 | After a minimum follow-up of 2 years, patients had significant reductions in pain and improvement in HHS regardless of the technique. | Although the multiple drilling technique is less invasive, it has similar outcomes compared with conventional CD. |
| Lieberman et al (2004)17 |
CD + BMP | 15/17 | Ficat IIA, IIB and III | 4 | Clinical success in 14/15 hips (93%; 13 patients) with stage IIA disease. 3/17 hips (3 patients) had radiographic progression of the FH, then converted to a THA. | The results of the study are similar to CD associated to various techniques, but there are a number of limitations. The study suggests that further study of the efficacy of CD + BMP is required. |
| Sun et al (2014)33 |
Bone graft with or without hrBMP | 42/72 | ARCO II-III | (5-7.67) | No statistical difference between the two groups (survival rate of FH was 64.1% in the group treated with the bone graft alone and 69.7% in the group treated with bone graft + BMP-2). | Mid-term excellent and good function could be achieved in selected patients (ARCO stage II) with impacted bone graft operation. Although no statistical difference was obtained in clinical results, rhBMP2 might improve the speed and quality of the bone repair inside the lesion. |
| Hernigou et al (2002)42 |
CD + BM grafting | 116/189 | Steinberg I-II | 7 | 6% of patients with pre-collapsed hips required a THA. 57% of patients with collapse of the FH before the CD required a THA. Outcomes improved in hips received increased numbers of progenitor cells. | This technique could be used to improve repair in the osteonecrosis area, at least in the earlier stage. |
| Gangji et al (2005)43 |
CD + BMMCs | 13/18 | ARCO I-II | 2 | Bone-marrow-graft group: significant reduction in pain and in joint symptoms; only 1/10 hips had progressed to this stage. A significant difference in the time to collapse between the two groups. | Implantation of autologous bone-marrow mononuclear cells appears to be a safe and effective treatment for early stages of osteonecrosis of the femoral head. Study is limited by small number of cases and short follow-up. |
| Wang et al (2009)44 |
CD + concentrated BMMCs | 45/59 | ARCO I-IIIA | 2.3 | Pre- and post-operative evaluation showed that HHS increased from 71 to 83 points. Clinically, the overall success is 79.7% and THA was done in 7/59 hips (11.9%). Radiologically, 14/59 hips exhibited FH collapse or narrowing of the coxofemoral joint space, and the overall failure rate is 23.7%. | Autologous BMMCs implantation relieves hip pain and prevents the progression of osteonecrosis particularly in stages I-II. |
| Gangji et al (2011)45 | Concentrated BMMCs vs CD | 19/24 | ARCO I-II | 5 | BM cell implantation afforded a significant reduction in pain and in joint symptoms and reduced the incidence of fractural stage. At 60 month, only 3/13 hips in the BM graft group had progressed to that stage. | Implantation of autologous BM cells in the necrotic lesion might be an effective treatment for patients with early stages of AVN of FH. |
| Sen et al (2012)40 |
Concentrated BMMCs vs CD | 40/51 | ARCO I-II | (0.4-2) | The clinical score and mean hip survival were significantly better in the CD+BMMCs group than in the CD group. Patients with adverse prognostic features at initial presentation had significantly better clinical outcomes and hip survival in the CD+BMMCs group than in the CD group. | Instillation of autologous BMMCs into the core tract after multiple CD in the femoral head osteonecrosis can result in better clinical outcome and hip survival. |
| Zhao et al (2012)39 |
Cultured BMSCs vs CD | 100/104 | ARCO IC-IIC | 5 | At 60 months, only 2/53 BMSCs-treated hips progressed and underwent vascularised bone grafting. In CD group, 7 hips were lost to follow-up, and 10 of the remaining 44 hips progressed and underwent vascularised bone grafting (5 hips) or THA. | BMSCs treatment significantly improved the HHS as well as decreased the volume of FH of the hips pre-operatively classified at stage IC, IIB and IIC. |
| Lim et al (2013)46 |
Multiple drilling + BMSCs vs CD and bone graft | 128/190 | Ficat I-IIB | 5 | No difference in the success rate between the two groups. Statistically significant differences were observed in the success rate between the pre-collapse state (Ficat II) and the collapsed state (Ficat III). | Multiple drilling and stem cell implantation produce outcomes comparable to other CD techniques and do not change the natural course of AVN. Significant differences were observed in patients who had more cells transplanted. |
| Rastogi et al (2013)47 |
CD + isolated mononuclear cells vs unprocessed BM injection | 40/60 | ARCO I-III | 2 | Considerable improvement in the hip function as measured by the HHS in both the groups. On MRI, there was a decrease in the size of the lesion in CD + isolated mononuclear cells group. 3/30 hips in unprocessed BM injection group required a THA. | Implantation of autologous BM stem cells has a better outcome than BM for early stage of AVN of the FH. |
| Liu et al (2013)48 |
CD + BMMCs vs CD | 34/53 | ARCO I-IIIA | 2.15 | The increase of HHS, the decrease of VAS, radiological and clinical success rates were significantly higher in the BMMC group compared with the control group. Post-operative collapse of the FH was less common in the BMMC group compared with the control group. | Both CD with or without implantation of BMMC are effective treatments, but CD with implantation of BMMCs and porous hydroxyapatite bone filler may be more effective treatment for AVN of the FH. |
| Ma et al (2014)49 |
CD + BM buffy coat grafting | 53 | Ficat I-III | 2 | A significant relief in pain and clinical joint symptoms as measured by the Lequesne index and WOMAC index in the treatment group. 33.3% of the hips in the control group had deteriorated to the next stage, whereas only 8% in the treatment group had further deterioration. The non-progression rates for stage I/II hips were 100% in the treatment group and 66.7% in the control group. | Implantation of the autologous BM buffy coat grafting combined with CD is effective to prevent further progression for the early stages of AVN of the FH. |
| Calori et al (2014)50 | CD + polytherapy | 38/40 | Ficat I-III | 4 | Clinical and radiographic healing occurred in 33/38 patients (86.84%); the mean time to clinical success was 1.8 ± 0.99 months. The radiological success was 86.84% at 36 months. HHS was 78.5 pre-op, 82.97 at 1 month and 81.39 at 36 months after surgery. Five treatments failed with collapse of the femoral head. | CD technique with autologous BMSCs, growth factors (BMP-7) and flexible xenograft bone substitute implantation reduces the incidence of fractural stage non-traumatic AVN of the FH. |
BM, bone morphogenetic; BMMCs, BM containing mononuclear cells; BMP-7, bone morphogenetic protein 7; BMSCs, BM stromal cells or BM-derived mesenchymal stem cells; CD, core decompression; AVN, avascular necrosis; FH, femoral head; HHS, Harris Hip Score; THA, total hip arthroplasty; VAS, visual analogue scale.
Core decompression (CD)
The aim of CD is to attempt to reduce intra-osseous pressure in the FH, restore vascular flow and improve pain which has been in use for over 40 years.
A current review of the literature on CD was published in 2015 by Pierce et al.19 The ideal lesion to be treated with CD is pre-collapse and small. Standard CD procedure is typically performed by the drilling and removal of an 8- to 10-mm cylindrical core from the necrotic lesion. In an attempt to minimise complications due to the procedure, such as violation of the articular cartilage or subtrochanteric fractures, a number of surgeons started to use a small diameter pin for multiple passes through the lesion. Pierce and colleagues showed how more recent studies on CD reported better results than older studies; this could be due to a surgical improvement and a better selection of patients.19
Yoon et al20 in 2001 evaluated the clinical outcome of the disease when treated at different stages. After a mean follow-up of 61 months, they concluded that patients classified as Ficat stage II or III (n = 17 out of 22 hips) have an increased rate of conversion to a THA than those classified as Ficat I (n = 5 out of 17 hips). In the same study, it was shown that even the localisation of the lesion could be predictive of the outcome, particularly with better results being observed in lesions 15% smaller than the FH, located medially. Those findings were also supported by Iorio et al21 and Mazieres et al.22
Mont et al23 highlighted the importance of making the diagnosis before head collapse occurred to have a significant chance to avoid a THA with CD. In a cohort of 68 hips treated post-collapse, only 29% had satisfactory outcomes.
In 2004, Mont et al24 treated 35 patients (45 hips) using percutaneous multiple drilling with clinical success in 32/45 hips (71%). Their conclusion was that this technique was effective in delaying a THR in young patients in the early stages of AVN of the FH.
In 2006, Song et al25 retrospectively reviewed 136 patients (163 hips) with a multiple drilling technique. After a minimum follow-up of five years, 31/39 (79%) patients with Ficat stage I and 62/81 (77%) patients with Ficat stage II disease had no additional surgery; all small lesions had successful outcomes. Their results, comparing their experience with the classical CD literature, show relatively few complications and results comparable with other CD techniques.
In a recent paper, Al Omran et al26 compared the traditional CD technique with the multiple drilling technique.
To treat larger lesions of the FH, surgeons have been trying to find new ways to enhance bone formation in the FH and to prevent fracture of the core tract. Non-vascularised (allograft bone or demineralised bone matrix) and vascularised bone grafts (fibula or iliac crest) have been used to try to improve the results of CD. The goal of this type of bone grafting is to decompress the osteonecrotic lesion, remove necrotic bone and provide structural support and scaffolding for repair of the subchondral bone in the FH. Vascularised grafts potentially have the advantage of re-vascularisation and promotion of osteogenesis of the FH. The non-vascularised cortical graft was originally popularised by Phemister,27 Boettcher et al28 and Bonfiglio et al;29 but currently it is not commonly used. In this technique, the core tract is filled with cortical strut grafts that were harvested from either the fibular, ilium or tibia. Successful results also have been reported with vascularised fibular grafts; the hypothetical advantage of this strategy is that the vascularised bone graft provides a strut to protect the subchondral region of the FH and a scaffold for new bone repair.30-32 However, it has not been established if the vascularised graft is the critical element in the success of this procedure.
Bone morphogenetic proteins (BMPs)
In 2004, Lieberman et al17 reported a retrospective evaluation of 15 patients (17 hips) with symptomatic osteonecrosis of the hip treated with CD combined with an allogeneic antigen-extracted, autolysed fibular allograft and 50 mg of partially purified human BMP and non-collagenous protein. The 15 hips were classified as Ficat Stage IIA, one hip as Ficat Stage IIB and one hip as Ficat Stage III, according to the modified Ficat staging system. The procedures were a clinical success in 14/15 hips (93%; 13 patients) with stage IIA disease. Three of 17 hips (three patients) had radiographic progression (Ficat Stages IIA, IIB and III) of the FH and were converted to a THA.
In 2014, Sun et al33 evaluated clinical outcomes of impacted bone graft with or without human recombinant BMP-2 for osteonecrosis of the FH (ONFH) on 42 patients (72 hips). After a mean follow-up of 6.3 years, the survival rate of the FH was 64.1% in the group treated with the bone graft alone and 69.7% for those patients treated with bone graft + BMP-2, although no statistical difference was found. However, the efficacy of this treatment was improved only in patients with ARCO stage II.
The risk of cancer should be considered in patients receiving a high dose of BMP-2, as shown in Carrage et al34 and Mesfin et al.35
Autologous bone-marrow cells
More and more studies have shown the association between bone mesenchymal stem cells (BMSCs) and non-traumatic AVN, which might have resulted from the decreased number and minor proliferative activity of BMSCs containing osteoprogenitor cells in the FH.36-38 Therefore, it may be beneficial to treat non-traumatic AVN with CD and implantation of autologous bone mononuclear cells (BMMCs), which contained high concentration of BMSCs.
Autologous bone marrow aspirates combined with CD have the clinical potential to enhance bone repair due to osteoprogenitor cells. Mesenchymal stem cell (MSC) transplantation based on CD surgery can provide seed cells to promote repair and restoration of the FH, accelerate reconstruction and produce creeping substitution of new bone.39,40 It is thought that MSCs implanted into the necrotic FH may differentiate into osteoblasts or vascular endothelial cells, thereby promoting bone repair and regeneration.41
Hernigou et al42 did a prospective study of 189 hips in 116 patients treated with autologous bone marrow grafting and CD, followed for seven years after surgery. In patients with pre-collapsed hips (University of Pennsylvania classification Stages I and II), 9/145 hips (6%) required a THA. THA was done in 25/44 hips (57%) in patients that had collapse of the FH before the CD. The authors concluded that outcomes were improved in hips that received increased numbers of progenitor cells and they believed that this technique could be used to improve repair in the area of osteonecrosis, at least in the earlier stage before mechanical failure of the FH occurs.
Gangji et al43 studied 13 patients (18 hips) with stage I or II AVN, according to the ARCO classification system. The hips were allocated to a programme of either CD (control group) or CD and implantation of autologous BMMCs (bone-marrow-graft group). After 24 months of follow-up, there was a significant reduction in pain and in joint symptoms measured with the Lequesne index and the Western Ontario and McMaster universities (WOMAC) index within the bone-marrow-graft group. Five of the eight hips in the control group had deteriorated to stage III, whereas only one of the ten hips in the bone-marrow-graft group had progressed to this stage. Survival analysis showed a significant difference in the time to collapse between the two groups.
In 2010, Wang et al44 published the results of a clinical study of 45 patients (59 hips) with stage I to IIIA AVN of the FH, according to the ARCO classification. All patients were treated using the CD technique with implantation of autologous BMMCs. The outcome was assessed by changes in Harris Hip Score (HHS), by progression in radiographic stages and by the need for a THA. The mean follow-up was 27.6 months. Pre- and post-operative evaluation showed that HHS increased from 71 to 83 points. Clinically, the overall success was 79.7% and THA was done in 7/59 hips (11.9%). Radiologically, 14 of the 59 hips exhibited FH collapse or narrowing of the coxo-femoral joint space and the overall failure rate was 23.7%. Therefore, for the authors, the autologous BMMCs implantation relieves hip pain and prevents the progression of osteonecrosis particularly in stages I to II.
Gangji et al45 reported the results of a five-year follow-up of a prospective controlled study of the efficacy of bone marrow cells implantation into the necrotic lesion of the FH on clinical symptoms and the progression of AVN in comparison with CD. They studied 19 patients and 24 hips with early stage AVN (ARCO stage 1: 4 hips – ARCO stage 2: 20 hips). Visual analogue scale (VAS) was used for patient pain assessment. The severity of hip disease was gauged using Lequesne algofunctional index. Symptoms of osteonecrosis were also assessed by the WOMAC score. Antero-posterior and frog-leg lateral radiographs and MRI of the affected hip were taken at the time of each clinical assessment. Radiological progression of osteonecrosis was measured by reference to ARCO-defined stages. Results of this study showed that bone marrow cell implantation afforded a significant reduction in pain and in joint symptoms and reduced the incidence of the fractural stage. At 60 months of follow-up, 8/11 hips in the control group had deteriorated to the fractural stage, whereas only 3/13 hips in the bone-marrow-graft group had progressed to that stage. Survival analysis showed a significant difference in the time to failure between the two groups. Patients had only minor side-effects after the treatments.
Sen et al40 showed the results of a prospective randomised controlled study. The 51 osteonecrotic hips in 40 patients (ARCO stage I to II) were randomly divided into two treatment groups (A and B). Patients in group A (25 hips) were treated with CD and those in group B (26 hips) received autologous BMMCs instillation into the core tract after CD. Outcome between the two groups were compared clinically (HHS), radiographically (radiographs and MRI) and by Kaplan-Meier survival analysis after 12 and 24 months of surgical intervention. The clinical score and mean hip survival were significantly better in group B than in group A. Patients with adverse prognostic features at initial presentation (i.e. poor HHS, radiograph changes, oedema and/or effusion on MRI) had significantly better clinical outcomes and hip survival in group B than in group A.
Zhao et al39 reported the results of a further randomised clinical study. The goal was to assess the efficacy of cultured BMSCs implantation into the FH as treatment against early stage AVN of the FH. One hundred patients (104 hips) were recruited (ARCO stages IC to IIC) and were randomly divided into a CD group alone (CD group: 50 patients, 51 hips) and a second group with FH autologous implantation of cultured BMSCs group (BMSCs group: 50 patients, 53 hips). Only two of the 53 BMSC-treated hips progressed and underwent vascularised bone grafting at five years after the operation. In the CD group, seven hips were lost to follow-up, and ten of the remaining 44 hips progressed and underwent vascularised bone grafting (five hips) or THA. BMSC treatment significantly improved the HHS as well as decreased the volume of FH of the hips pre-operatively classified as stage IC, IIB and IIC.
In 2013, Lim et al46 investigated whether multiple drilling and stem cell implantation in the treatment of AVN would improve clinical and radiographic results, compared with conventional CD and bone graft in the early stages of AVN. There were 128 patients (190 hips) who had undergone surgery were divided into two groups based on which treatment they received: a) multiple drilling and stem cell implantation (107 patients, 159 hips) or b) CD, curettage and a bone graft (21 patients, 31 hips). Indications for surgery were restricted primarily to modified Ficat stages I, IIA and IIB, although some patients with stage III disease were included if the size of the lesion was greater than 30% and if the lesion was located laterally. Among the stem cell implantation group, 21 patients (31 hips) were lost to follow-up; therefore, 86 patients (128 hips) were available for the study. The authors observed no difference in the success rate between the stem cell group and the CD group, 73/128 hips and 17/31 hips, respectively, in the Kaplan-Meier survivorship curves at the five-year follow-up. They observed statistically significant differences in the success rate between the pre-collapse state (Ficat II) and the collapsed state (Ficat III): 64/102 hips and 24/57 hips, respectively, in the Kaplan-Meier survivorship curves at the five-year follow-up. In the stem cell group, 59 hips were considered unsuccessful at the last follow-up, while 47 of 59 hips required additional surgery, and 12 hips had a HHS below 75 points and showed additional collapse during the follow-up period. In the CD group, 14 hips were considered unsuccessful at the last follow-up. Additional surgery has been required in 11/14 hips and three hips had a HHS below 75 points and showed additional collapse during the follow-up period.
Rastogi et al47 performed a prospective clinical trial in 40 patients (60 hips) with stage I, II or III (ARCO system) AVN, followed up for a mean period of 24 months. The patients were allocated to two groups: a) CD and isolated mononuclear cells (group A: 30 hips) or b) CD and unprocessed bone marrow injection (group B: 30 hips). The functional outcome was assessed in terms of HHS, and disease progression was assessed radiologically by comparing the pre-operative and follow-up MRI. At the two-year follow-up, there was considerable improvement in the hip function as measured by the HHS in both the groups (p = 0.031). On MRI, there was a decrease in the size of the lesion in group A. Three of 30 hips in group B required THA.
Liu et al48 showed the results of a retrospective analysis that investigated the effectiveness of CD and implantation of BMMCs with porous hydroxyapatite bone filler for the treatment AVN of the FH. Patients were eligible for inclusion in the study if they had ARCO stage I, II or IIIA AVN of the FH. There were two groups of patients in the study: a group who received CD and implantation of BMMCs (17 patients, 26 hips) and a group who received CD without implantation of BMMCs (17 patients, 27 hips). HHS was significantly increased in both groups of patients after surgery. The magnitude of increase was significantly greater in the BMMCs group as compared with the control group. VAS score was significantly decreased in both groups after surgery. The magnitude of decrease was significantly greater in the BMMCs group as compared with the control group. Radiological and clinical success rates were significantly higher in the BMMCs group as compared with the control group. Post-operative collapse of the FH was less common in the BMMCs group as compared with the control group.
Ma et al49 conducted a prospective double-blinded randomised controlled trial to examine the effect of bone-marrow buffy coat grafting combined with CD for the treatment of AVN of the FH. The 45 patients (53 hips) with Ficat stage I to III AVN of the FH were recruited. The hips were allocated to the control group (CD + autologous bone graft) or treatment group (CD + autologous bone graft with bone-marrow buffy coat). At the 24-month follow-up, there was a significant relief in pain and clinical joint symptoms as measured by the Lequesne index and WOMAC index in the treatment group. Of the hips in the control group, 33.3% deteriorated to the next stage after 24 months post procedure, whereas only 8% in the treatment group had further deterioration. The non-progression rates for stage I/II hips were 100% in the treatment group and 66.7% in the control group. Therefore, for the authors, implantation of the autologous bone marrow buffy coat grafting combined with CD is effective in preventing further progression of the early stages of AVN of the FH.
Combined techniques
Many researchers explored the combination of CD with stimulation and promotion of bone regeneration (i.e. artificial bone, autologous fibular bone, allograft fibular bone, iliac bone or osseous growth factor, and MSC-rich autologous bone marrow) to achieve better efficacy than CD alone.
In 2014, Calori et al50 performed a retrospective clinical study to determine the efficacy of the CD technique combined with recombinant BMP-7, BMSCs and xenograft bone substitute, placed into the necrotic lesion of the FH on the clinical symptoms and on the progression of AVN. A total of 38 patients (40 hips) with early stages of AVN of the FH were studied over a four-year period. According to the Ficat classification criteria, there were seven hips with stage I osteonecrosis, 25 hips with stage II and eight hips with stage III changes. Clinical and radiographic healing occurred in 33/38 (87%) patients. In all the five failed treatments, there was a collapse of the FH with progression of the osteonecrotic lesion at the 24-month follow-up. THA was performed in these patients.
Discussion
The treatment of AVN of the FH is based on patient age, periodisation, area of osteonecrosis, position and collapse risk, and individual choice. Only by correctly mastering the therapeutic principles and adopting proper methods specifically oriented to different stages can the best therapeutic effect be achieved.
Although there are many studies concerning CD in the treatment of non-traumatic AVN of the FH, there have been inconsistencies in the reports of its clinical success (from 47% to 83%), mainly related to many different factors including: stage of AVN; skill of procedure; and duration of follow-up.
Recently, Papakostidis et al51 performed a meta-analysis to investigate whether implantation of autologous bone marrow aspirate, containing MSCs, into the CD track would improve the clinical and radiological results of AVN of the FH compared with the classical method of CD alone. They showed that the application of autologous bone marrow concentrate (autologous cell therapy) in combination with CD is superior to CD treatment alone, as it was found to markedly decelerate the progression of the disease to the stage of FH collapse. This procedure also limited the need for THA, particularly when employed in the early (pre-collapse) stages of AVN of the FH. In this meta-analysis, the authors demonstrated that autologous cell therapy in addition to CD for the treatment of AVN of the FH resulted in reduction of painful joint symptoms and improvement in HHS compared with CD treatment alone.
However, this new therapeutic approach presents some limitations, such as the limited data on the number of cells required and the sources of MSCs that can be used (currently autologous iliac bone marrow).
CD and BMMC transplantation have shown good results in the treatment of the early stages of AVN of the FH. On the contrary, for advanced stages of AVN of the FH, the therapeutic effect of this procedure is poor and the treatment method must be selected with discretion. When the collapse has already occurred, (collapse > 4 mm and duration > 6 months) no other useful treatment can be proposed other than THA.
AVN of the FH often affects young active patients with an important negative effect on their activity and quality of life. The long-term prognosis of THA is as yet unclear. A prosthetic implantation in such patients produces important financial repercussions on the national health system also. Many cases of revision surgery will result. A treatment that can avoid or at least delay prosthetic implantation is necessary.
The most important limitation of this systematic review is the quality of the available papers. In particular, clinical studies included in our review did not provide sample, size and power calculations. Therefore, there were limited number of comparative trials and only three randomised controlled trials. We included all types of clinical studies, potentially introducing confounding and selection bias, but we felt that inclusion of these studies would provide a more comprehensive review of the literature surrounding this difficult topic. Furthermore, meta-analysis was not performed due to the limited number of comparative trials.
AVN of the FH primarily affects younger and working age individuals, thus leading to increased morbidity and functional disability in the population. Innovative treatments aimed to halt or delay progression of disease would provide a welcome alternative to those faced with progression to joint collapse and hip replacement surgery. Combining CD with biotechnologies could result in a novel long-lasting hip-preserving solution.
Current clinical studies have suggested beneficial effects on patient-reported outcomes, but definitive conclusions regarding hip survival and disease progression cannot be drawn.
This systematic review of the literature shows that there is not at present one treatment which is superior to the others in the treatment of AVN of the FH. CD alone may still have therapeutic potential, since it can stimulate bone regeneration reducing pain and improving function. Moreover, the association of biologically active substances could improve the effectiveness of CD in increasing bone healing.
These biotechnologies have demonstrated their effectiveness in the treatment of long bone nonunion and bone defects as evidenced by many studies.52-58 Their application in cases of AVN of FH may give similar good results.
Further studies are needed to clarify the more effective biotechnologies to use in the treatment of AVN of the FH and the best combination.
Overall, considering the results of this review, the surgical treatment with CD associated with biotechnologies is advisable in early stages of AVN of the FH. In those cases in which it is not possible to save the FH, the treatment with the best outcome for the patient’s quality of life remains prosthetic arthroplasty.
Footnotes
ICMJE Conflict of Interest Statement: None
Funding
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
References
- 1. Mont MA, Carbone JJ, Fairbank AC. Core decompression versus nonoperative management for osteonecrosis of the hip. Clin Orthop Relat Res 1996;324:169-178. [DOI] [PubMed] [Google Scholar]
- 2. Arbab D, König DP. Atraumatic femoral head necrosis in adults. Dtsch Arztebl Int 2016;113:31-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lieberman JR, Berry DJ, Mont MA, et al. Osteonecrosis of the hip: management in the 21st century. Instr Course Lect 2003;52:337-355. [PubMed] [Google Scholar]
- 4. Petrigliano FA, Lieberman JR. Osteonecrosis of the hip: novel approaches to evaluation and treatment. Clin Orthop Relat Res 2007;465:53-62. [DOI] [PubMed] [Google Scholar]
- 5. Aldridge JM, III, Urbaniak JR. Avascular necrosis of the femoral head: etiology, pathophysiology, classification, and current treatment guidelines. Am J Orthop (Belle Mead NJ) 2004;33:327-332. [PubMed] [Google Scholar]
- 6. Zalavras CG, Lieberman JR. Osteonecrosis of the femoral head: evaluation and treatment. J Am Acad Orthop Surg 2014;22:455-464. [DOI] [PubMed] [Google Scholar]
- 7. Zalavras C, Dailiana Z, Elisaf M, et al. Potential aetiological factors concerning the development of osteonecrosis of the femoral head. Eur J Clin Invest 2000;30:215-221. [DOI] [PubMed] [Google Scholar]
- 8. Steinberg ME. Diagnostic imaging and role of stage and lesion size in determining outcome in osteonecrosis of the femoral head. Tech Orthop 2001;16:6-15 [Google Scholar]
- 9. Lang P, Jergesen HE, Moseley ME, et al. Avascular necrosis of the femoral head: high-field-strength MR imaging with histologic correlation. Radiology 1988;169:517-524. [DOI] [PubMed] [Google Scholar]
- 10. Ficat RP. Idiopathic bone necrosis of the femoral head. Early diagnosis and treatment. J Bone Joint Surg [Br] 1985;67-B:3-9. [DOI] [PubMed] [Google Scholar]
- 11. Steinberg ME, Hayken GD, Steinberg DR. A quantitative system for staging avascular necrosis. J Bone Joint Surg [Br] 1995;77-B:34-41. [PubMed] [Google Scholar]
- 12. Sugano N, Atsumi T, Ohzono K, et al. The 2001 revised criteria for diagnosis, classification, and staging of idiopathic osteonecrosis of the femoral head. J Orthop Sci 2002;7:601-605. [DOI] [PubMed] [Google Scholar]
- 13. Hungerford DS. Treatment of osteonecrosis of the femoral head: everything’s new. J Arthroplasty 2007;22:91-94. [DOI] [PubMed] [Google Scholar]
- 14. Lieberman JR. Core decompression for osteonecrosis of the hip. Clin Orthop Relat Res 2004;418:29-33. [DOI] [PubMed] [Google Scholar]
- 15. Soohoo NF, Vyas S, Manunga J, et al. Cost-effectiveness analysis of core decompression. J Arthroplasty 2006;21:670-681. [DOI] [PubMed] [Google Scholar]
- 16. Lau RL, Perruccio AV, Evans HM, et al. Stem cell therapy for the treatment of early stage avascular necrosis of the femoral head: a systematic review. BMC Musculoskelet Disord 2014;15:156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lieberman JR, Conduah A, Urist MR. Treatment of osteonecrosis of the femoral head with core decompression and human bone morphogenetic protein. Clin Orthop Relat Res 2004;429:139-145. [DOI] [PubMed] [Google Scholar]
- 18. Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Pierce TP, Jauregui JJ, Elmallah RK, et al. A current review of core decompression in the treatment of osteonecrosis of the femoral head. Curr Rev Musculoskelet Med 2015;8:228-232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Yoon TR, Song EK, Rowe SM, Park CH. Failure after core decompression in osteonecrosis of the femoral head. Int Orthop 2001;24:316-318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Iorio R, Healy WL, Abramowitz AJ, Pfeifer BA. Clinical outcome and survivorship analysis of core decompression for early osteonecrosis of the femoral head. J Arthroplasty 1998;13:34-41. [DOI] [PubMed] [Google Scholar]
- 22. Mazières B, Marin F, Chiron P, et al. Influence of the volume of osteonecrosis on the outcome of core decompression of the femoral head. Ann Rheum Dis 1997;56:747-750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Mont MA, Jones LC, Pacheco I, Hungerford DS. Radiographic predictors of outcome of core decompression for hips with osteonecrosis stage III. Clin Orthop Relat Res 1998;354:159-168. [DOI] [PubMed] [Google Scholar]
- 24. Mont MA, Ragland PS, Etienne G. Core decompression of the femoral head for osteonecrosis using percutaneous multiple small-diameter drilling. Clin Orthop Relat Res 2004;429:131-138. [DOI] [PubMed] [Google Scholar]
- 25. Song WS, Yoo JJ, Kim YM, Kim HJ. Results of multiple drilling compared with those of conventional methods of core decompression. Clin Orthop Relat Res 2007;454:139-146. [DOI] [PubMed] [Google Scholar]
- 26. Al Omran A. Multiple drilling compared with standard core decompression for avascular necrosis of the femoral head in sickle cell disease patients. Arch Orthop Trauma Surg 2013;133:609-613. [DOI] [PubMed] [Google Scholar]
- 27. Phemister DB. Treatment of the necrotic head of the femur in adults. Dallas Burton Phemister (1882-1951). Clin Orthop Relat Res 2000;381:4-8. [PubMed] [Google Scholar]
- 28. Boettcher WG, Bonfiglio M, Smith K. Non-traumatic necrosis of the femoral head. II. Experiences in treatment. J Bone Joint Surg [Am] 1970;52-A:322-329. [PubMed] [Google Scholar]
- 29. Bonfiglio M. Technique of core biopsy and tibial bone grafting (Phemister procedure) for treatment of aseptic necrosis of the femoral head. Iowa Orthop J 1982;2:57-62. [Google Scholar]
- 30. Marker DR, Seyler TM, Ulrich SD, Srivastava S, Mont MA. Do modern techniques improve core decompression outcomes for hip osteonecrosis? Clin Orthop Relat Res 2008;466:1093-1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Nelson LM, Clark CR. Efficacy of phemister bone grafting in nontraumatic aseptic necrosis of the femoral head. J Arthroplasty 1993;8:253-258. [DOI] [PubMed] [Google Scholar]
- 32. Pierce TP, Elmallah RK, Jauregui JJ, et al. A current review of non-vascularized bone grafting in osteonecrosis of the femoral head. Curr Rev Musculoskelet Med 2015;8:240-245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Sun W, Li Z, Gao F, et al. Recombinant human bone morphogenetic protein-2 in debridement and impacted bone graft for the treatment of femoral head osteonecrosis. PLoS One 2014;9:e100424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Carragee EJ, Chu G, Rohatgi R, et al. Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg [Am] 2013;95:1537-1545. [DOI] [PubMed] [Google Scholar]
- 35. Mesfin A, Buchowski JM, Zebala LP, et al. High-dose rhBMP-2 for adults: major and minor complications: a study of 502 spine cases. J Bone Joint Surg [Am] 2013;95:1546-1553. [DOI] [PubMed] [Google Scholar]
- 36. Hernigou P, Beaujean F, Lambotte JC. Decrease in the mesenchymal stem-cell pool in the proximal femur in corticosteroid-induced osteonecrosis. J Bone Joint Surg [Br] 1999;81-B:349-355. [DOI] [PubMed] [Google Scholar]
- 37. Wang GJ, Cui Q, Balian G. The pathogenesis and prevention of steroid-induced osteonecrosis. Clin Orthop Relat Res 2000;370:295-310. [DOI] [PubMed] [Google Scholar]
- 38. Suh KT, Kim SW, Roh HL, Youn MS, Jung JS. Decreased osteogenic differentiation of mesenchymal stem cells in alcohol-induced osteonecrosis. Clin Orthop Relat Res 2005;431:220-225. [DOI] [PubMed] [Google Scholar]
- 39. Zhao D, Cui D, Wang B, et al. Treatment of early stage osteonecrosis of the femoral head with autologous implantation of bone marrow-derived and cultured mesenchymal stem cells. Bone 2012;50:325-330. [DOI] [PubMed] [Google Scholar]
- 40. Sen RK, Tripathy SK, Aggarwal S, et al. Early results of core decompression and autologous bone marrow mononuclear cells instillation in femoral head osteonecrosis: a randomized control study. J Arthroplasty 2012;27:679-686. [DOI] [PubMed] [Google Scholar]
- 41. Yan Z, Hang D, Guo C, Chen Z. Fate of mesenchymal stem cells transplanted to osteonecrosis of femoral head. J Orthop Res 2009;27:442-446. [DOI] [PubMed] [Google Scholar]
- 42. Hernigou P, Beaujean F. Treatment of osteonecrosis with autologous bone marrow grafting. Clin Orthop Relat Res 2002;405:14-23. [DOI] [PubMed] [Google Scholar]
- 43. Gangji V, Hauzeur JP. Treatment of osteonecrosis of the femoral head with implantation of autologous bone-marrow cells. Surgical technique. J Bone Joint Surg [Am] 2005;87-A:106-112. [DOI] [PubMed] [Google Scholar]
- 44. Wang BL, Sun W, Shi ZC, et al. Treatment of nontraumatic osteonecrosis of the femoral head with the implantation of core decompression and concentrated autologous bone marrow containing mononuclear cells. Arch Orthop Trauma Surg 2010;130:859-865. [DOI] [PubMed] [Google Scholar]
- 45. Gangji V, De Maertelaer V, Hauzeur JP. Autologous bone marrow cell implantation in the treatment of non-traumatic osteonecrosis of the femoral head: five year follow-up of a prospective controlled study. Bone 2011;49:1005-1009. [DOI] [PubMed] [Google Scholar]
- 46. Lim YW, Kim YS, Lee JW, Kwon SY. Stem cell implantation for osteonecrosis of the femoral head. Exp Mol Med 2013;45:e61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Rastogi S, Sankineani SR, Nag HL, et al. Intralesional autologous mesenchymal stem cells in management of osteonecrosis of femur: a preliminary study. Musculoskelet Surg 2013;97:223-228. [DOI] [PubMed] [Google Scholar]
- 48. Liu Y, Liu S, Su X. Core decompression and implantation of bone marrow mononuclear cells with porous hydroxylapatite composite filler for the treatment of osteonecrosis of the femoral head. Arch Orthop Trauma Surg 2013;133:125-133. [DOI] [PubMed] [Google Scholar]
- 49. Ma Y, Wang T, Liao J, et al. Efficacy of autologous bone marrow buffy coat grafting combined with core decompression in patients with avascular necrosis of femoral head: a prospective, double-blinded, randomized, controlled study. Stem Cell Res Ther 2014;5:115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Calori GM, Mazza E, Colombo M, et al. Treatment of AVN using the induction chamber technique and a biological-based approach: indications and clinical results. Injury 2014;45:369-373. [DOI] [PubMed] [Google Scholar]
- 51. Papakostidis C, Tosounidis TH, Jones E, Giannoudis PV. The role of “cell therapy” in osteonecrosis of the femoral head. Acta Orthop 2016;87:72-78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Calori GM, Colombo M, Bucci M, et al. Clinical effectiveness of Osigraft in long-bones non-unions. Injury 2015;46:S55-S64. [DOI] [PubMed] [Google Scholar]
- 53. Calori GM, Colombo M, Mazza E, et al. Monotherapy vs. polytherapy in the treatment of forearm non-unions and bone defects. Injury 2013;44:S63-S69. [DOI] [PubMed] [Google Scholar]
- 54. Blokhuis TJ, Calori GM, Schmidmaier G. Autograft versus BMPs for the treatment of non-unions: what is the evidence? Injury 2013;44:S40-S42. [DOI] [PubMed] [Google Scholar]
- 55. Kanakaris NK, Calori GM, Verdonk R, et al. Application of BMP-7 to tibial non-unions: a 3-year multicenter experience. Injury 2008;39:S83-S90. [DOI] [PubMed] [Google Scholar]
- 56. Calori GM, Tagliabue L, Gala L, et al. Application of rhBMP-7 and platelet-rich plasma in the treatment of long bone non-unions: a prospective randomised clinical study on 120 patients. Injury 2008;39:1391-1402. [DOI] [PubMed] [Google Scholar]
- 57. Kanakaris NK, Lasanianos N, Calori GM, et al. Application of bone morphogenetic proteins to femoral non-unions: a 4-year multicentre experience. Injury 2009;40:S54-S61. [DOI] [PubMed] [Google Scholar]
- 58. Calori GM, Colombo M, Mazza E, et al. Monotherapy vs. polytherapy in the treatment of forearm non-unions and bone defects. Injury 2013;44:S63-S69. [DOI] [PubMed] [Google Scholar]