Abstract
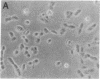
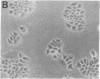
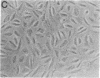
Somatic cell hybrids between MCF-7 human breast cancer cells and normal immortalized human mammary epithelial cells have been obtained by polyethylene glycol-mediated cell fusion. The hybrid cells are suppressed in their ability to form tumors in nude mice, as well as in traits specific to the tumorigenic MCF-7 parent: growth factor independence, tumor necrosis factor sensitivity, and pS2 gene expression. In addition, they display other characteristics of the "normal" parent, including increased expression relative to the MCF-7 cells of the genes for the extracellular matrix component fibronectin, the intermediate filament keratin 5, and the angiogenesis inhibitor thrombospondin. The levels of keratins 8 and 18 also resemble those of the nontumorigenic parent. These results provide evidence for the existence of tumor suppressor gene products in immortal mammary epithelial cells. We propose a characteristic "suppressed" tumor cell phenotype, which encompasses altered cytoarchitecture, angiogenesis capabilities, and growth factor requirements.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ali I. U., Lidereau R., Theillet C., Callahan R. Reduction to homozygosity of genes on chromosome 11 in human breast neoplasia. Science. 1987 Oct 9;238(4824):185–188. doi: 10.1126/science.3659909. [DOI] [PubMed] [Google Scholar]
- Band V., Sager R. Distinctive traits of normal and tumor-derived human mammary epithelial cells expressed in a medium that supports long-term growth of both cell types. Proc Natl Acad Sci U S A. 1989 Feb;86(4):1249–1253. doi: 10.1073/pnas.86.4.1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berry M., Nunez A. M., Chambon P. Estrogen-responsive element of the human pS2 gene is an imperfectly palindromic sequence. Proc Natl Acad Sci U S A. 1989 Feb;86(4):1218–1222. doi: 10.1073/pnas.86.4.1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brissenden J. E., Derynck R., Francke U. Mapping of transforming growth factor alpha gene on human chromosome 2 close to the breakpoint of the Burkitt's lymphoma t(2;8) variant translocation. Cancer Res. 1985 Nov;45(11 Pt 2):5593–5597. [PubMed] [Google Scholar]
- Callahan R., Campbell G. Mutations in human breast cancer: an overview. J Natl Cancer Inst. 1989 Dec 6;81(23):1780–1786. doi: 10.1093/jnci/81.23.1780. [DOI] [PubMed] [Google Scholar]
- Cavenee W., Leach R., Mohandas T., Pearson P., White R. Isolation and regional localization of DNA segments revealing polymorphic loci from human chromosome 13. Am J Hum Genet. 1984 Jan;36(1):10–24. [PMC free article] [PubMed] [Google Scholar]
- Creasey A. A., Doyle L. V., Reynolds M. T., Jung T., Lin L. S., Vitt C. R. Biological effects of recombinant human tumor necrosis factor and its novel muteins on tumor and normal cell lines. Cancer Res. 1987 Jan 1;47(1):145–149. [PubMed] [Google Scholar]
- Fingert H. J., Chen Z., Mizrahi N., Gajewski W. H., Bamberg M. P., Kradin R. L. Rapid growth of human cancer cells in a mouse model with fibrin clot subrenal capsule assay. Cancer Res. 1987 Jul 15;47(14):3824–3829. [PubMed] [Google Scholar]
- Folkman J., Klagsbrun M. Angiogenic factors. Science. 1987 Jan 23;235(4787):442–447. doi: 10.1126/science.2432664. [DOI] [PubMed] [Google Scholar]
- Frazier W. A. Thrombospondin: a modular adhesive glycoprotein of platelets and nucleated cells. J Cell Biol. 1987 Aug;105(2):625–632. doi: 10.1083/jcb.105.2.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham K. A., Trent J. M., Osborne C. K., McGrath C. M., Minden M. D., Buick R. N. The use of restriction fragment polymorphisms to identify the cell line MCF-7. Breast Cancer Res Treat. 1986;8(1):29–34. doi: 10.1007/BF01805922. [DOI] [PubMed] [Google Scholar]
- Hammond S. L., Ham R. G., Stampfer M. R. Serum-free growth of human mammary epithelial cells: rapid clonal growth in defined medium and extended serial passage with pituitary extract. Proc Natl Acad Sci U S A. 1984 Sep;81(17):5435–5439. doi: 10.1073/pnas.81.17.5435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen M. F., Cavenee W. K. Genetics of cancer predisposition. Cancer Res. 1987 Nov 1;47(21):5518–5527. [PubMed] [Google Scholar]
- Harris H. The analysis of malignancy by cell fusion: the position in 1988. Cancer Res. 1988 Jun 15;48(12):3302–3306. [PubMed] [Google Scholar]
- Hennessy S. W., Frazier B. A., Kim D. D., Deckwerth T. L., Baumgartel D. M., Rotwein P., Frazier W. A. Complete thrombospondin mRNA sequence includes potential regulatory sites in the 3' untranslated region. J Cell Biol. 1989 Feb;108(2):729–736. doi: 10.1083/jcb.108.2.729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Höppener J. W., Steenbergh P. H., Zandberg J., Bakker E., Pearson P. L., Geurts van Kessel A. H., Jansz H. S., Lips C. J. Localization of the polymorphic human calcitonin gene on chromosome 11. Hum Genet. 1984;66(4):309–312. doi: 10.1007/BF00287635. [DOI] [PubMed] [Google Scholar]
- Klein G. The approaching era of the tumor suppressor genes. Science. 1987 Dec 11;238(4833):1539–1545. doi: 10.1126/science.3317834. [DOI] [PubMed] [Google Scholar]
- Knudson A. G., Jr Hereditary cancer, oncogenes, and antioncogenes. Cancer Res. 1985 Apr;45(4):1437–1443. [PubMed] [Google Scholar]
- Lundberg C., Skoog L., Cavenee W. K., Nordenskjöld M. Loss of heterozygosity in human ductal breast tumors indicates a recessive mutation on chromosome 13. Proc Natl Acad Sci U S A. 1987 Apr;84(8):2372–2376. doi: 10.1073/pnas.84.8.2372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majack R. A., Mildbrandt J., Dixit V. M. Induction of thrombospondin messenger RNA levels occurs as an immediate primary response to platelet-derived growth factor. J Biol Chem. 1987 Jun 25;262(18):8821–8825. [PubMed] [Google Scholar]
- Mumby S. M., Abbott-Brown D., Raugi G. J., Bornstein P. Regulation of thrombospondin secretion by cells in culture. J Cell Physiol. 1984 Sep;120(3):280–288. doi: 10.1002/jcp.1041200304. [DOI] [PubMed] [Google Scholar]
- Nunez A. M., Jakowlev S., Briand J. P., Gaire M., Krust A., Rio M. C., Chambon P. Characterization of the estrogen-induced pS2 protein secreted by the human breast cancer cell line MCF-7. Endocrinology. 1987 Nov;121(5):1759–1765. doi: 10.1210/endo-121-5-1759. [DOI] [PubMed] [Google Scholar]
- Pienta K. J., Partin A. W., Coffey D. S. Cancer as a disease of DNA organization and dynamic cell structure. Cancer Res. 1989 May 15;49(10):2525–2532. [PubMed] [Google Scholar]
- Rastinejad F., Polverini P. J., Bouck N. P. Regulation of the activity of a new inhibitor of angiogenesis by a cancer suppressor gene. Cell. 1989 Feb 10;56(3):345–355. doi: 10.1016/0092-8674(89)90238-9. [DOI] [PubMed] [Google Scholar]
- Rio M. C., Bellocq J. P., Gairard B., Rasmussen U. B., Krust A., Koehl C., Calderoli H., Schiff V., Renaud R., Chambon P. Specific expression of the pS2 gene in subclasses of breast cancers in comparison with expression of the estrogen and progesterone receptors and the oncogene ERBB2. Proc Natl Acad Sci U S A. 1987 Dec;84(24):9243–9247. doi: 10.1073/pnas.84.24.9243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sager R. Genetic suppression of tumor formation. Adv Cancer Res. 1985;44:43–68. doi: 10.1016/s0065-230x(08)60025-1. [DOI] [PubMed] [Google Scholar]
- Sager R. Tumor suppressor genes: the puzzle and the promise. Science. 1989 Dec 15;246(4936):1406–1412. doi: 10.1126/science.2574499. [DOI] [PubMed] [Google Scholar]
- Spriggs D., Imamura K., Rodriguez C., Horiguchi J., Kufe D. W. Induction of tumor necrosis factor expression and resistance in a human breast tumor cell line. Proc Natl Acad Sci U S A. 1987 Sep;84(18):6563–6566. doi: 10.1073/pnas.84.18.6563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spurr N. K., Solomon E., Jansson M., Sheer D., Goodfellow P. N., Bodmer W. F., Vennstrom B. Chromosomal localisation of the human homologues to the oncogenes erbA and B. EMBO J. 1984 Jan;3(1):159–163. doi: 10.1002/j.1460-2075.1984.tb01777.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stampfer M. R., Bartley J. C. Induction of transformation and continuous cell lines from normal human mammary epithelial cells after exposure to benzo[a]pyrene. Proc Natl Acad Sci U S A. 1985 Apr;82(8):2394–2398. doi: 10.1073/pnas.82.8.2394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steel D. M., Harris H. The effect of antisense RNA to fibronectin on the malignancy of hybrids between melanoma cells and normal fibroblasts. J Cell Sci. 1989 Jul;93(Pt 3):515–524. doi: 10.1242/jcs.93.3.515. [DOI] [PubMed] [Google Scholar]
- Sugarman B. J., Aggarwal B. B., Hass P. E., Figari I. S., Palladino M. A., Jr, Shepard H. M. Recombinant human tumor necrosis factor-alpha: effects on proliferation of normal and transformed cells in vitro. Science. 1985 Nov 22;230(4728):943–945. doi: 10.1126/science.3933111. [DOI] [PubMed] [Google Scholar]
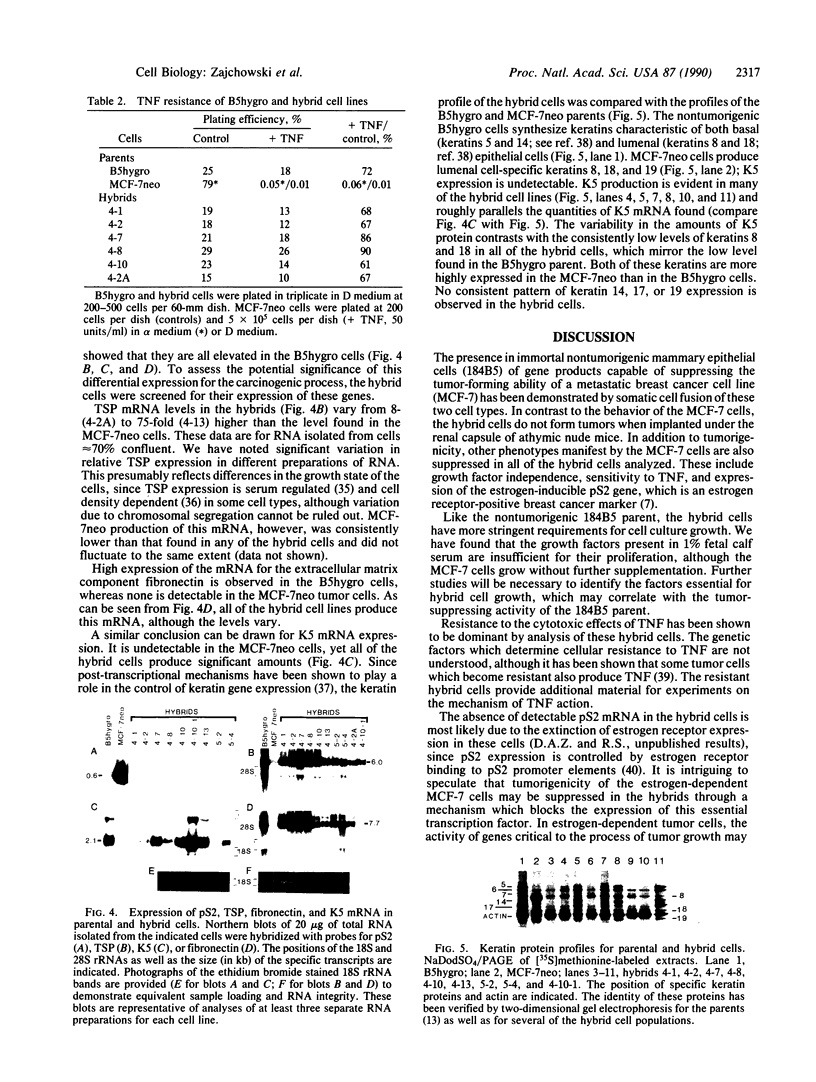
- Trask D. K., Band V., Zajchowski D. A., Yaswen P., Suh T., Sager R. Keratins as markers that distinguish normal and tumor-derived mammary epithelial cells. Proc Natl Acad Sci U S A. 1990 Mar;87(6):2319–2323. doi: 10.1073/pnas.87.6.2319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyner A. L., Fuchs E. Evidence for posttranscriptional regulation of the keratins expressed during hyperproliferation and malignant transformation in human epidermis. J Cell Biol. 1986 Nov;103(5):1945–1955. doi: 10.1083/jcb.103.5.1945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walen K. H., Stampfer M. R. Chromosome analyses of human mammary epithelial cells at stages of chemical-induced transformation progression to immortality. Cancer Genet Cytogenet. 1989 Feb;37(2):249–261. doi: 10.1016/0165-4608(89)90056-3. [DOI] [PubMed] [Google Scholar]
- Wigler M., Pellicer A., Silverstein S., Axel R., Urlaub G., Chasin L. DNA-mediated transfer of the adenine phosphoribosyltransferase locus into mammalian cells. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1373–1376. doi: 10.1073/pnas.76.3.1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zajchowski D., Band V., Pauzie N., Tager A., Stampfer M., Sager R. Expression of growth factors and oncogenes in normal and tumor-derived human mammary epithelial cells. Cancer Res. 1988 Dec 15;48(24 Pt 1):7041–7047. [PubMed] [Google Scholar]