Abstract
Purpose
To evaluate the safety and efficacy of intravitreal dexamethasone (IVD) implants in eyes with diabetic macular edema that did not respond to previous treatment.
Methods
We included 46 eyes of 46 patients in this retrospective study. Each month, we recorded patient visual acuity with logarithm of the minimum angle of resolution using the Early Treatment Diabetic Retinopathy Study chart, central macular thickness measurements with optical coherence tomography, intraocular pressure (IOP), and posttreatment complication occurrence.
Results
The mean follow-up time was 8.95 ± 1.33 months (range, 6 to 12). Best-corrected visual acuity improved significantly in the first 4 months after IVD, but no statistically significant change was observed over the following 2 months. Although a statistically significant decrease in central macular thickness was observed in the first 3 months, the change was not statistically significant in the following 3 months. There was a statistically significant increase in IOP in the first 2 months, but no statistically significant change was observed in the following months. IOP was controlled with medication in all patients with elevated IOP. Of the 26 phakic patients, two had cataracts requiring surgery.
Conclusions
Cases of refractory diabetic macular edema that did not respond to previous treatment, such as anti-vascular endothelial growth factor injections and laser photocoagulation, exhibited improvements in visual acuity and decreases in retinal thickness after IVD implantation. Both functional and anatomical effects were observed in the first 3 months after injection. Repeat injections and frequent examination might be required for continued improvement. Side effects, such as cataracts and elevation of IOP, may require medical or surgical treatment.
Keywords: Diabetic macular edema, Intravitreal dexamethasone implants
The most common cause of vision loss in 20 to 74 years old in developed nations is diabetic retinopathy (DR), and 75% of this loss is due to diabetic macular edema (DME) [1]. The results of the Wisconsin DR Epidemiological Study showed that 20.1% of type 1 diabetes mellitus (DM) cases and 25.4% of type 2 insulin-dependent DM cases experienced vision loss within 10 years [2]. According to data from the Early Treatment Diabetic Retinopathy Study (ETDRS), DME causes severe and moderate vision loss in 6.4% and 24% of cases, respectively [3].
The pathogenesis of DME, which can emerge alongside both preproliferative and proliferative DR, is not well understood. This disorder is thought to arise as a result of several types of changes, including leakage associated with retinal vascular anomalies, localized ischemia, chronic inflammation, or vascular cell degeneration and death [4]. Earlier studies demonstrated that several cytokines play a role in this process, including vascular endothelial growth factor (VEGF), intercellular adhesion molecule-1 (an important mediator in leukostasis), tumor necrosis factor-alpha and interleukin-1 (which increase vasodilatation and vascular permeability), interleukin-6, angiopoietin-2, and fibroblastic growth factor-2 [5,6].
Corticosteroids block arachidonic acid secretion from cell membranes, reduce prostaglandin synthesis and block leukotriene formation. Corticosteroids in human vascular smooth muscle cells have been shown to suppress VEGF and decrease VEGF expression and vascular permeability in rats [7,8]. Moreover, they prevent the release of mediators that lead to inflammation, such as VEGF, tumor necrosis factor-alpha and leucocyte migration. Corticosteroids also reduce the release of molecules such as intercellular adhesion molecule-1, which is responsible for vascular endothelial cell activity, vascular cell adhesion molecule 1 and major histocompatibility complexes 1 and 2. Moreover, they suppress acute processes and inflammation processes [9].
The anti-inflammatory effects of corticosteroids are direct and fast when applied intravitreally. Corticosteroids stabilize tight bonds between endothelial cells in retinal veins and strengthen them by increasing their number and decreasing edema. Because of these properties, corticosteroids are considered to have anti-inflammatory, antiapoptotic, antiedematous, and antiangiogenic effects [9,10,11,12,13].
Devices that deliver long-release intravitreal steroids were developed after promising results from intravitreal steroid injections. Compared to intravitreal steroid injections, long-release delivery advantageously offers long-term effects and consistent drug levels.
Ozurdex (Allergan, Irvine, CA, USA) is a biodegradable intravitreal implant that slowly releases dexamethasone, which has potent anti-inflammatory and anti-VEGF effects. Ozurdex is inserted into the vitreous cavity through the pars plana using a single use 22-gauge applicator device [14]. Direct intravitreal dexamethasone (IVD) administration results in high intraocular concentration with minimal systemic absorption [14,15]. The implant consists of a biodegradable copolymer matrix of lactic acid and glycolic acid that enables the slow release of dexamethasone [14]. Dexamethasone implantation resulted in sustained levels of dexamethasone and biological activity for 6 months, with the drug at peak levels during the first 2 months [16].
The aim of this study was to evaluate the results of dexamethasone (Ozurdex) implantation for treating eyes with DME that did not respond to previous treatment.
Materials and Methods
We retrospectively searched the medical records of 46 DME patients who received IVD implants and were followed up for 12 months between January 2013 and December 2014 at Istanbul Research and Training Hospital.
The local ethics committee approved this study, which was conducted in accordance with the Declaration of Helsinki. Before IVD application, we informed all patients about the potential adverse effects of treatment and obtained their consent.
Our patients underwent complete ophthalmoscopic examination before treatment. These examinations involved determination of best-corrected visual acuity (BCVA; logarithm of the minimum angle of resolution [logMAR]) using the ETDRS scale, slit-lamp examination and intraocular pressure (IOP) measurement with Goldmann applanation tonometry. Patients' pupils were dilated with 2.5% phenylephrine and 1% tropicamide, and the fundus was examined using a 90-D lens. Central macular thickness (CMT, µm) was measured via fundus photography, fundus fluorescein angiography, and spectral domain optic coherence tomography (Optovue OCT V 5.1, RTVue 100-2; Optovue, Fremont, CA, USA). Gonioscopic examination was conducted before and after injection. Hemoglobin A1c levels in the 3 and 6 months were evaluated before and after IVD application.
DME diagnosis, defined as clinically significant macular edema, was determined based on criteria developed in the ETDRS [3]. Despite at least three administrations of an intravitreal anti-VEGF drug (ranibizumab [Lucentis] or bevacizumab [Avastin]; Genentech Inc., South San Francisco, CA, USA), DME persisted beyond 6 months in many patients; those with CMT thickness ≥300 µm were included in this study. Performance in a focal photocoagulation test was assessed 3 months before the first injection. If CMT thickness remained >300 µm, or if a fall in BCVA was detected after 6 months following the first injection, a second injection was given.
Patients were excluded from this study if they ever underwent eye surgery (except cataract surgery), had a trauma history, epiretinal membrane or vitreomacular traction in optic coherence tomography, macular ischemia in fluorescein angiography, glaucoma, IOP over 21 mmHg or used topical and/or systemic steroids within the last 3 months. Also, patients who had a hemorrhage disorder, active ocular infections, a recent history of myocardial infarction, uncontrolled hypertension or who were pregnant at the time of treatment were excluded. Finally, patients who underwent eye surgery or who received DME treatment other than a dexamethasone implant, such as intravitreal anti-VEGF, or who underwent photocoagulation during their follow-up were not included in the study. During the 6-month study period, patients were only injected with a dexamethasone implant and did not receive intravitreal anti-VEGF or photocoagulation.
A single vitreoretinal surgeon (EU) conducted all applications under sterile conditions after the pupils were dilated. After applying topical proparacaine HCL 0.5% (Alcaine; Alcon, Mississauga, ON, Canada), eyelids and the supraorbital area were cleaned with 10% povidone iodine. Povidone iodine (5%) was applied to the conjunctiva after eyelids were opened with a blepharostat. Intravitreal injections were performed in the upper temporal quadrant and positioned 3.0 to 4.0 mm from the limbus. The patients remained seated immediately after injection and the fundus was examined with indirect ophthalmoscopy. Optical nerve perfusion was controlled. The patients were cautioned not to go to bed for at least 1 hour. Following injection, patients were prescribed lomefloxacin HCL (Okacin; Novartis, Basel, Switzerland) four times a day for a week.
IOP was regarded as high when IOP ≥22 mmHg. Timolol, dorzolamide, brinzolamide, or a dorzolamide-timolol combination were the first options for patients who had IOP ≥25 mmHg. When necessary, brimonidine was added.
Statistical analysis
The relationships between BCVA, IOP, and CMT, which were measured prior to injection and during initial examinations, were analyzed using paired t-tests with SPSS ver. 15.0 for Windows (SPSS Inc., Chicago, IL, USA). A p < 0.05 was considered statistically significant.
Results
In total, 46 eyes of 46 patients (30 females [65.2%] and 16 males [34.8%]) with a mean age of 61.69 ± 8.5 years (range, 47 to 78) were included in the study (Table 1). The mean duration of diabetes was 15.69 ± 8.73 years (range, 4 to 31), the average follow-up period was 8.95 ± 1.33 months (range, 6 to 12), and the average injection number was 1.08 ± 0.28 (range, 1 to 2) (Table 1). The mean period between primary treatments and Ozurdex treatment was 1.66 ± 0.92 months (range, 1 to 4).
Table 1. Characteristics of patients.
Values are presented as mean ± standard deviation (range) or mean ± standard deviation.
DM = diabetes mellitus; HbA1c = hemoglobin A1c; inj = injection.
Although a second injection was recommended for all patients, only four received the second injection within 6 months. Other patients refused the second injection due to financial or other reasons.
Efficacy
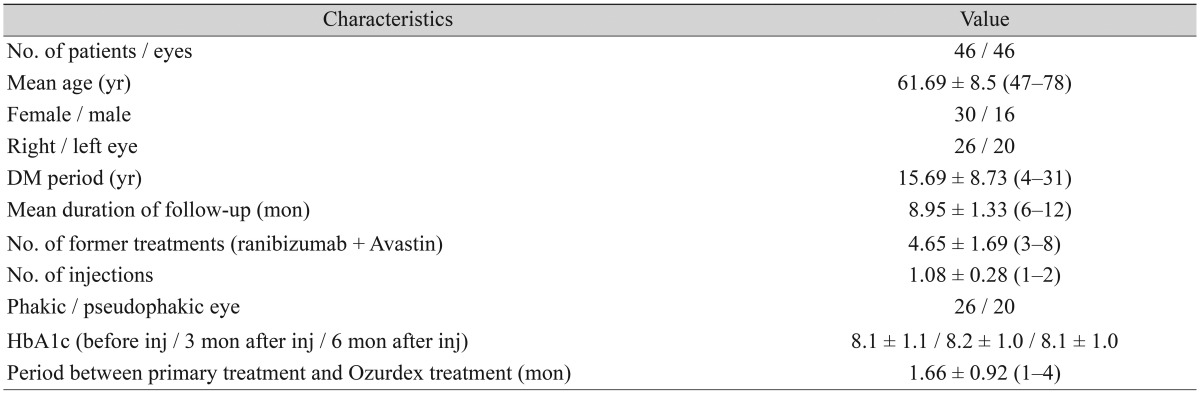
Although a statistically significant increase was detected (p < 0.05) during months 1 to 4 of the follow-up period when compared to patient condition before BCVA treatment, no statistically significant changes were detected during months 5 and 6 (p > 0.05) (Table 2 and Fig. 1A).
Table 2. Comparison of VA, CMT, and IOP at study onset and during follow-up.
Values are presented as mean ± standard deviation (range).
VA = visual acuity; CMT = central macular thickness; IOP = intraocular pressure; BCVA = best-corrected visual acuity; logMAR = logarithm of the minimum angle of resolution.
Paired t-test (p < 0.05 indicates statistical significance).
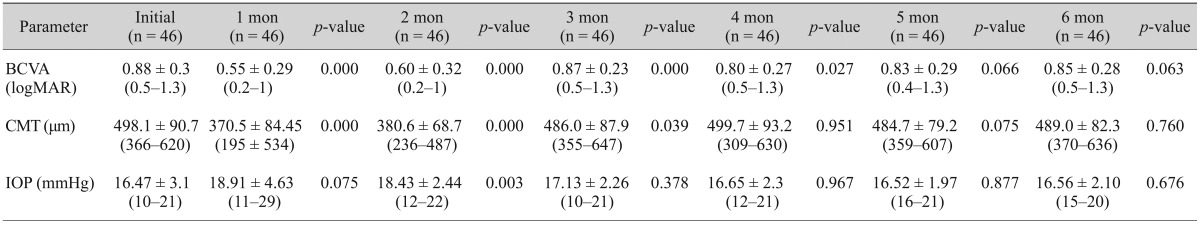
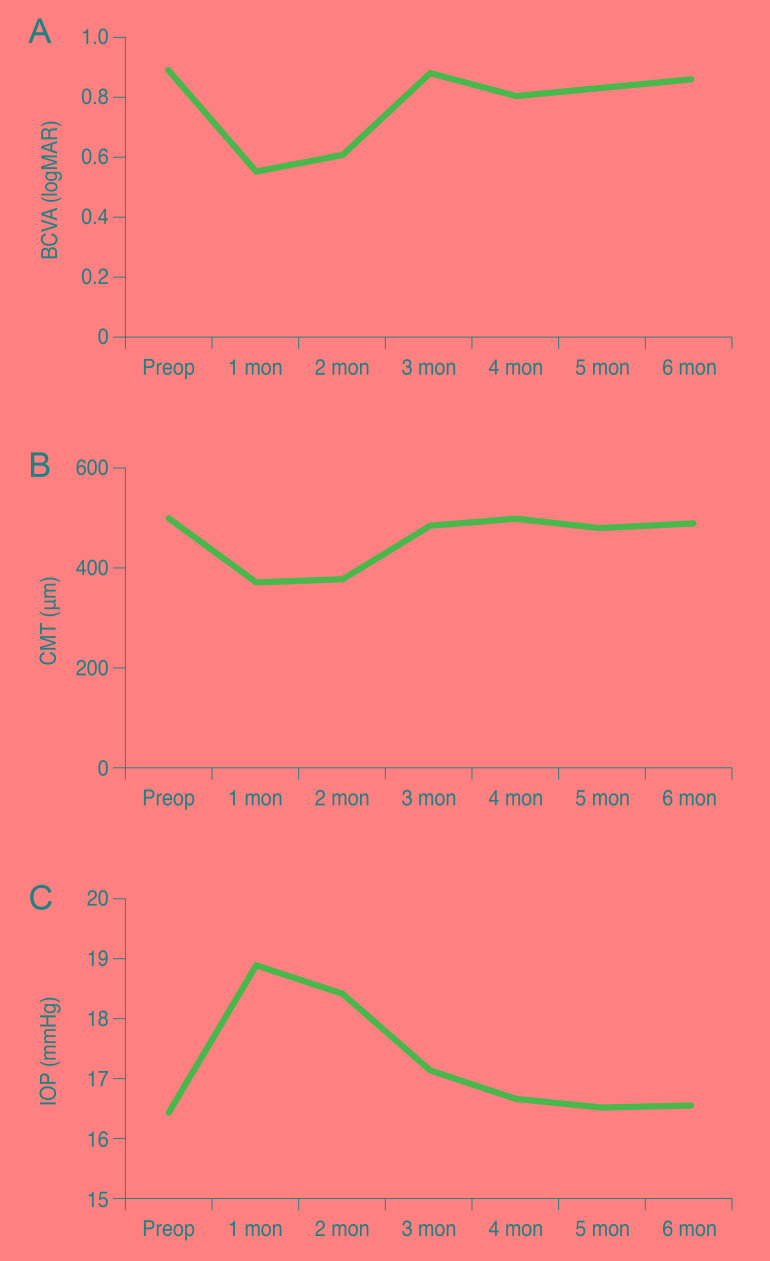
Fig. 1. Mean BCVA, CMT, and IOP at study onset and during follow-up. (A) Mean BCVA (logMAR), (B) mean CMT, (C) mean IOP before and after treatment. BCVA = best-corrected visual acuity; logMAR = logarithm of the minimum angle of resolution; Preop = preoperative; CMT = central macular thickness; IOP = intraocular pressure.
Macular thickness
Although macular thickness was found (p < 0.05) to decrease at a statistically significant rate during months 1, 2 and, 3 when compared to pretreatment conditions, the rate of decrease was not statistically significant during months 4 to 6 (p > 0.05) (Table 2 and Fig. 1B).
Decreases in CMT were greatest (370 µm) after the first injection during month 1 (Fig. 1B).
Safety
Although IOP increased at a statistically significant rate during the follow-up month 2 (p < 0.05) compared to pre-IVD treatment conditions, rate increases were not statistically significant after that point (p > 0.05) (Table 2 and Fig. 1C).
The first day after injection, one eye had an IOP of 27 mmHg; two eyes had an IOP of 26 mmHg; three eyes had an IOP of 25 mmHg; and one eye had an IOP of 29 mmHg. In total, eight eyes had an IOP >25 mmHg in the month following injection. All of these patients' IOP values decreased to within normal limits (<22 mmHg) after administration of topical antiglaucomatous drugs and did not require surgery. Twelve patients (26%) experienced an IOP increase >5 mmHg after injection.
With the exception of subconjunctival hemorrhage, which occurred after 6 of the 50 injections (12%) given to patients, no associated complications, such as endophthalmitis, retinal detachment or any IVD-dependent systemic adverse effects, were observed.
During follow-up, four patients were diagnosed with cataracts in month 11 and two of these patients underwent cataract surgery.
Discussion
A major cause of DR-associated visual loss is DME. Capillary leakage resulting from destruction of the blood-retina barrier, fluid, increases in macular thickness, inflammatory mediators and VEGF are the main causes of DME pathogenesis [17,18].
The number of treatment modalities that exist parallels the number of pathogenic etiologies. Visual acuity loss can be prevented by application of focal/grid laser photocoagulation [19] in microaneurysms and diffuse capillary layer leakage. However, sufficient visual acuity cannot be achieved through this method alone [3].
Repeat injections of anti-VEGF are required and determining which patients are partial responders or non-responders is a major challenge. Detection of ranibizumab and BCVA below 20 / 40 in 40% of the patients in RISE and RIDE (a study of Ranibizumab Injection in Subjects with Clinically Significant Macular Edema with Center Involvement Secondary to Diabetes Mellitus) studies revealed that add-on therapies are required to achieve sufficient improvement [20].
Intravitreal steroids have positive effects on DME through VEGF inhibition, anti-inflammatory effects and vascular disorder reduction via tight bonds among endothelial cells. The most frequent complications from steroids are IOP increase and cataract formation. Infectious endophthalmitis and retinal detachment have been reported as well [21,22].
Dexamethasone is a potent water-soluble corticosteroid. Because its intravitreal half-life is short, it is applied to the vitreous through a drug release system. Ozurdex (Allergan) is a biodegradable dexamethasone implant that contains lactic acid and glycolic acid and can be injected through a 22-gauge applicator. The implant undergoes hydrolysis over time and decomposes into carbon dioxide and water components [14] and was designed to reduce the number of repeat injections, which last for 6 months at most [16]. Dexamethasone is less lipophilic than triamcinolone and fluocinolone acetonide, so it decreases IOP and prevents cataract formation by reducing accumulation in the trabecular meshwork and lens [23].
In a randomized, sham-controlled 3-year study (n = 1,048), Boyer et al. [24] observed an average decrease in CMT of 111.6 µ after a mean of 4.1 injections and 22.2% of the patients had a visual acuity gain of more than 15 letters. Meanwhile, 59.2% of the patients underwent cataract operation, and almost one-third exhibited a significant increase in IOP. Only one patient required surgery to control IOP, whereas other patients only needed medical therapy. Intravitreal hemorrhage, which does not require surgery, was observed in 6.9% of the patients and endophthalmitis was observed in one patient [24].
In our retrospective study, a mean CMT decrease of 128 µ after an average of 1.08 ± 0.28 injections was achieved; 78.2% and 21.7% of patients demonstrated visual gains of more than 10 and 15 letters, respectively. Two patients underwent cataract operations and significant IOP increases were detected in 10 patients. Neither hemorrhage nor endophthalmitis was observed among our patients. Furthermore, none of our patients experienced neovascularization. The low incidence of both cataract surgery and significant IOP increases might be due to the short-term follow-up period and small sample size.
In a retrospective study of nine patients, Zucchiatti et al. [25] observed CMT decreases in refractory DME patients who had treatments at an average of 13.4 months that did not include Ozurdex injections. On day 3, by applying a single Ozurdex injection, Zucchiatti et al. found that BCVA improvements started in month 1 and continued for 4 months. They reported that 88.8% of their patients required a second injection in month 6. In this study, the average hemoglobin A1c value was 8.2 [25]. In our study, the average hemoglobin A1c level was 8.1. Because the last treatment before Ozurdex injection was performed 1.66 months prior, 100% of the patients required a second injection in month 6, but only 9.09% of the patients underwent a second injection.
Similar to our study, Scaramuzzi et al. [26] determined that the effect of dexamethasone implantation on BCVA and CMT continued for 4 months. In contrast, in a retrospective study of 58 patients, Dutra Medeiros et al. [27] found that patients with DME refractory to other treatments, were under the effect of dexamethasone implants during 6 months of follow-up and the maximum effect was observed in month 3. CMT decreases of 37% CMT and 0.44 ± 0.27 logMAR BCVA improvement were detected during the period of maximum effect after a single injection of Ozurdex. In our study, the maximum effect was observed in month 1 and it continued for 4 months. CMT decreases of 25% and 0.33 logMAR BCVA improvement were detected during the period of maximum effect.
In the PLACID study of patients with diffuse macular edema, laser photocoagulation treatment was compared to a combination treatment of laser photocoagulation and dexamethasone; no significant differences were observed among groups at the end of the first year, although vision improvements were greater among the combination group [28]. However, DME patients who were refractory to focal laser photocoagulation and anti-VEGF treatment were susceptible to the dexamethasone implant in our study and a significant increase in visual acuity was observed in the first 4 months.
In their randomized control trial, Haller et al. [29] determined that the percentage of eyes with DME that achieved a BCVA improvement of 10 letters or more by day 90 after receiving 700 µg of dexamethasone was similar to the overall study population, which also included eyes with macular edema due to uveitis, Irvine-Gass syndrome and retinal vein occlusion. Rishi et al. [30], however, concluded that the effect of Ozurdex implantation lasted for a shorter period in patients with refractory DME (4 months) than in patients with retinal vein occlusion dependent-macular edema (6 months). Furthermore, they suggested that CMT decreases in month 1 did not affect BCVA increases, which could be due to the chronic effect of DM on the macula. Even though CMT changed in month 4 among patients who had been diagnosed with DM at least 15 years prior in our study, continued BCVA increases do not support this theory.
The effect of a single a nti-VEGF injection appears to last 6 to 8 weeks [31]; in our study, the average period between the treatments before Ozurdex and receipt of Ozurdex treatment was 1.66 ± 0.92 months (range, 1 to 4), indicating that anti-VEGF might affect the early follow-up period after switching from anti-VEGF to Ozurdex.
The limitations of our study are its retrospective nature and the absence of a control group. In future studies, randomization of patients not responding adequately to anti-VEGF either to a continued anti-VEGF treatment group or to a dexamethasone implantation group could help address the question of whether dexamethasone implantation is superior to anti-VEGF treatment.
The advantages of our study are that we were able to determine the clinical effect of dexamethasone implantation on DME refractory to anti-VEGF treatment and focal laser photocoagulation.
In summary, treatment of DME patients who do not respond to anti-VEGF and laser photocoagulation treatment and who have prolonged duration DME can be a challenge. Herein, we analyzed the effect of Ozurdex implantation in this difficult patient group. Consequently, for patients who require DM-dependent refractory macular edema treatment, IVD implantation is effective and well-tolerated; it increases visual acuity and decreases macular edema. Although its effective period has been observed to be longer than that of anti-VEGF treatments, repeat injections may still be needed if the effective period is <6 months. The effective period and reliability of Ozurdex intravitreal injection should be determined in future randomized studies.
Footnotes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
- 1.Williams GA. Diabetic macular edema. In: Bandello F, Querques G, editors. ESASO course series. Vol. 1. Medical retina. Basel: Karger; 2012. pp. 99–104. [Google Scholar]
- 2.Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. XV: the long-term incidence of macular edema. Ophthalmology. 1995;102:7–16. doi: 10.1016/s0161-6420(95)31052-4. [DOI] [PubMed] [Google Scholar]
- 3.Photocoagulation for diabetic macular edema: Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985;103:1796–1806. [PubMed] [Google Scholar]
- 4.Adamis AP. Is diabetic retinopathy an inflammatory disease? Br J Ophthalmol. 2002;86:363–365. doi: 10.1136/bjo.86.4.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Grant MB, Afzal A, Spoerri P, et al. The role of growth factors in the pathogenesis of diabetic retinopathy. Expert Opin Investig Drugs. 2004;13:1275–1293. doi: 10.1517/13543784.13.10.1275. [DOI] [PubMed] [Google Scholar]
- 6.Nguyen QD, Shah SM, Van Anden E, et al. Supplemental oxygen improves diabetic macular edema: a pilot study. Invest Ophthalmol Vis Sci. 2004;45:617–624. doi: 10.1167/iovs.03-0557. [DOI] [PubMed] [Google Scholar]
- 7.Nauck M, Karakiulakis G, Perruchoud AP, et al. Corticosteroids inhibit the expression of the vascular endothelial growth factor gene in human vascular smooth muscle cells. Eur J Pharmacol. 1998;341:309–315. doi: 10.1016/s0014-2999(97)01464-7. [DOI] [PubMed] [Google Scholar]
- 8.Heiss JD, Papavassiliou E, Merrill MJ, et al. Mechanism of dexamethasone suppression of brain tumor-associated vascular permeability in rats: involvement of the glucocorticoid receptor and vascular permeability factor. J Clin Invest. 1996;98:1400–1408. doi: 10.1172/JCI118927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McGhee CN. Pharmacokinetics of ophthalmic corticosteroids. Br J Ophthalmol. 1992;76:681–684. doi: 10.1136/bjo.76.11.681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Parke DW, 3rd, Sisk RA, Houston SK, Murray TG. Ocular hypertension after intravitreal triamcinolone with vitrectomy and phacoemulsification. Clin Ophthalmol. 2012;6:925–931. doi: 10.2147/OPTH.S32934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Massin P, Audren F, Haouchine B, et al. Intravitreal triamcinolone acetonide for diabetic diffuse macular edema: preliminary results of a prospective controlled trial. Ophthalmology. 2004;111:218–224. doi: 10.1016/j.ophtha.2003.05.037. [DOI] [PubMed] [Google Scholar]
- 12.Park CH, Jaffe GJ, Fekrat S. Intravitreal triamcinolone acetonide in eyes with cystoid macular edema associated with central retinal vein occlusion. Am J Ophthalmol. 2003;136:419–425. doi: 10.1016/s0002-9394(03)00228-9. [DOI] [PubMed] [Google Scholar]
- 13.Edelman JL, Lutz D, Castro MR. Corticosteroids inhibit VEGF-induced vascular leakage in a rabbit model of blood-retinal and blood-aqueous barrier breakdown. Exp Eye Res. 2005;80:249–258. doi: 10.1016/j.exer.2004.09.013. [DOI] [PubMed] [Google Scholar]
- 14.Haller JA, Bandello F, Belfort R, Jr, et al. Randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with macular edema due to retinal vein occlusion. Ophthalmology. 2010;117:1134–1146. doi: 10.1016/j.ophtha.2010.03.032. [DOI] [PubMed] [Google Scholar]
- 15.Goldfein A. Adrenocorticosteroids and adrenocortical antagonists. In: Katzung BG, editor. Basic and clinical pharmacology. 6th ed. London: Prentice Hall; 1995. pp. 592–607. [Google Scholar]
- 16.Chang-Lin JE, Attar M, Acheampong AA, et al. Pharmacokinetics and pharmacodynamics of a sustained-release dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci. 2011;52:80–86. doi: 10.1167/iovs.10-5285. [DOI] [PubMed] [Google Scholar]
- 17.Yau JW, Rogers SL, Kawasaki R, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35:556–564. doi: 10.2337/dc11-1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bhagat N, Grigorian RA, Tutela A, Zarbin MA. Diabetic macular edema: pathogenesis and treatment. Surv Ophthalmol. 2009;54:1–32. doi: 10.1016/j.survophthal.2008.10.001. [DOI] [PubMed] [Google Scholar]
- 19.American Diabetes Association. Standards of medical care in diabetes--2009. Diabetes Care. 2009;32(Suppl 1):S13–S61. doi: 10.2337/dc09-S013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Brown DM, Nguyen QD, Marcus DM, et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials. RISE and RIDE. Ophthalmology. 2013;120:2013–2022. doi: 10.1016/j.ophtha.2013.02.034. [DOI] [PubMed] [Google Scholar]
- 21.Gillies MC, Sutter FK, Simpson JM, et al. Intravitreal triamcinolone for refractory diabetic macular edema: two-year results of a double-masked, placebo-controlled, randomized clinical trial. Ophthalmology. 2006;113:1533–1538. doi: 10.1016/j.ophtha.2006.02.065. [DOI] [PubMed] [Google Scholar]
- 22.Jonas JB, Degenring R, Kreissig I, Akkoyun I. Safety of intravitreal high-dose reinjections of triamcinolone acetonide. Am J Ophthalmol. 2004;138:1054–1055. doi: 10.1016/j.ajo.2004.06.041. [DOI] [PubMed] [Google Scholar]
- 23.Thakur A, Kadam R, Kompella UB. Trabecular meshwork and lens partitioning of corticosteroids: implications for elevated intraocular pressure and cataracts. Arch Ophthalmol. 2011;129:914–920. doi: 10.1001/archophthalmol.2011.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Boyer DS, Yoon YH, Belfort R, Jr, et al. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology. 2014;121:1904–1914. doi: 10.1016/j.ophtha.2014.04.024. [DOI] [PubMed] [Google Scholar]
- 25.Zucchiatti I, Lattanzio R, Querques G, et al. Intravitreal dexamethasone implant in patients with persistent diabetic macular edema. Ophthalmologica. 2012;228:117–122. doi: 10.1159/000336225. [DOI] [PubMed] [Google Scholar]
- 26.Scaramuzzi M, Querques G, Spina CL, et al. Repeated intravitreal dexamethasone implant (Ozurdex) for diabetic macular edema. Retina. 2015;35:1216–1222. doi: 10.1097/IAE.0000000000000443. [DOI] [PubMed] [Google Scholar]
- 27.Dutra Medeiros M, Postorino M, Navarro R, et al. Dexamethasone intravitreal implant for treatment of patients with persistent diabetic macular edema. Ophthalmologica. 2014;231:141–146. doi: 10.1159/000356413. [DOI] [PubMed] [Google Scholar]
- 28.Callanan DG, Gupta S, Boyer DS, et al. Dexamethasone intravitreal implant in combination with laser photocoagulation for the treatment of diffuse diabetic macular edema. Ophthalmology. 2013;120:1843–1851. doi: 10.1016/j.ophtha.2013.02.018. [DOI] [PubMed] [Google Scholar]
- 29.Haller JA, Kuppermann BD, Blumenkranz MS, et al. Randomized controlled trial of an intravitreous dexamethasone drug delivery system in patients with diabetic macular edema. Arch Ophthalmol. 2010;128:289–296. doi: 10.1001/archophthalmol.2010.21. [DOI] [PubMed] [Google Scholar]
- 30.Rishi P, Rishi E, Kuniyal L, Mathur G. Short-term results of intravitreal dexamethasone implant (OZURDEX(®)) in treatment of recalcitrant diabetic macular edema: a case series. Oman J Ophthalmol. 2012;5:79–82. doi: 10.4103/0974-620X.99368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Badala F. The treatment of branch retinal vein occlusion with bevacizumab. Curr Opin Ophthalmol. 2008;19:234–238. doi: 10.1097/ICU.0b013e3282fb7fb0. [DOI] [PubMed] [Google Scholar]