Abstract
Background
No nationwide epidemiological study evaluating the prevalence of subclinical and overt forms of hypothyroidism and hyperthyroidism has yet been conducted in Korea. This study aimed to evaluate the reference range of serum thyroid stimulating hormone (TSH) and the national prevalence of thyroid dysfunctions in Korea.
Methods
Nation-wide cross-sectional data were analyzed from a representative sample of the civilian, non-institutionalized Korean population (n=6,564) who underwent blood testing for thyroid function and anti-thyroid peroxidase antibody (TPOAb) as part of the Korea National Health and Nutrition Examination Survey VI (2013 to 2015).
Results
The reference interval of serum TSH in the Korean reference population was 0.62 to 6.68 mIU/L. Based on this reference interval, the prevalence of overt and subclinical hypothyroidism was 0.73% (males 0.40%, females 1.10%) and 3.10% (males 2.26%, females 4.04%), respectively. The prevalence of hypothyroidism increased with age until the age group between 50 to 59 years. Positive TPOAb were found in 7.30% of subjects (males 4.33%, females 10.62%). The prevalence of overt and subclinical hypothyroidism TPOAb-positive subjects was 5.16% and 10.88%, respectively. The prevalence of overt and subclinical hyperthyroidism was 0.54% (males 0.30%, females 0.81%) and 2.98% (males 2.43%, females, 3.59%), respectively.
Conclusion
The Serum TSH reference levels in the Korean population were higher than the corresponding levels in Western countries. Differences were found in the prevalence of hypothyroidism and hyperthyroidism according to age, sex, and TPOAb positivity. This study provides important baseline information for understanding patterns of thyroid dysfunction and diseases in Korea.
Keywords: Thyrotropin, Hypothyroidism, Hyperthyroidism, Prevalence, Thyroid gland
INTRODUCTION
The level of serum thyroid stimulating hormone (TSH) is the most sensitive diagnostic index to evaluate thyroid dysfunction in subjects without hypothalamic or pituitary dysfunction. The diagnosis of thyroid dysfunctions, especially subclinical dysfunctions, is significantly affected by the validity of the TSH reference interval. Subclinical hypothyroidism and subclinical hyperthyroidism are defined as abnormally increased or decreased serum TSH levels with a normal serum free thyroxine (fT4) level [1]. However, the optimal cutoff values of serum TSH and the clinical significance of subclinical thyroid dysfunction are still controversial [2]. The prevalence of subclinical hypothyroidism has been reported to vary from 3% to 12% and that of subclinical hyperthyroidism to vary from 1% to 6% [3,4,5,6,7,8,9,10,11,12,13].
Serum TSH levels can be affected by age, gender, thyroid autoantibodies, smoking, and iodine intake [4,5,7,8,10,11,12,13,14,15,16,17,18,19,20,21]. Therefore, the National Academy of Clinical Biochemistry suggested that the serum TSH reference range should be established in rigorously screened normal euthyroid volunteers without evidence of thyroid disease [21]. A recent Korean study using institutional routine health check-up data demonstrated that the serum TSH levels in a Korean reference population were higher than the levels that have been reported in other countries [22]. That study suggested that excess dietary iodine intake could be considered an important factor contributing to this discrepancy.
Previously, the nationwide incidence and prevalence of hypothyroidism and hyperthyroidism have only been reported based on claims data provided by the Health Insurance Review and Assessment Service [23,24]. A large nationwide epidemiological study evaluating the reference range of serum TSH and the prevalence of hypothyroidism and hyperthyroidism has never been conducted in Korea. The Centers for Disease Control and Prevention has conducted a series of Korea National Health and Nutrition Examination Surveys (KNHANES) starting in 1998. The KNHANES VI (2013 to 2015) measured serum TSH, fT4, anti-thyroid peroxidase antibody (TPOAb), and urinary iodine (UI) concentration.
This study aims to evaluate the reference range of serum TSH levels and the national prevalence of subclinical and clinical forms of thyroid dysfunction in South Korea based on the survey data obtained from the KNHANES VI (2013 to 2015).
METHODS
Study subjects
The data were collected from the KNHANES VI (2013 to 2015), a nationwide, cross-sectional survey of the Korean population that used a stratified, multistage clustered probability sampling to select a representative sample of the civilian, non-institutionalized Korean population. The KNHANES is an ongoing surveillance system that assesses the health and nutritional status of Koreans, monitors trends in health risk factors and the prevalence of major chronic diseases, and provides data for the development and evaluation of health policies and programs in Korea.
Research subjects were selected through 2-stage stratified cluster sampling using population and housing census data. Additionally, laboratory tests for serum TSH, fT4, TPOAb, and UI concentrations were performed using stratified subsampling according to gender and age in approximately 2,400 persons aged 10 years and older in each year of KNHANES VI (2013 to 2015).
Laboratory methods
For the analysis of serum TSH, fT4, and TPOAb approximately 15 mL of blood was collected. After separation of the serum within 30 minutes, the sample was transferred to the testing facility. Collected specimens were analyzed by an electrochemiluminescence immunoassay within 24 hours.
Serum TSH, fT4 and TPOAb levels were measured with an electrochemiluminescence immunoassay (Roche Diagnostics, Mannheim, Germany). TSH was measured using an E-TSH kit (Roche Diagnostics), for which the reference range was 0.35 to 5.50 mIU/L. The fT4 was measured using a E-Free T4 kit (Roche Diagnostics), the reference range was 0.89 to 1.76 ng/mL. TPOAb was measured using an E-Anti-TPO kit (Roche Diagnostics); the normal range for TPOAb in humans is <34.0 IU/mL. The reported results of TSH, fT4, and TPOAb met the specifications regarding accuracy, general chemistry, special immunology and ligand of the quality control and quality assurance program of the College of American Pathologist [25]. UI concentrations were measured with an inductively coupled plasma mass spectrometry devise (ICP-MS; Perkin Elmer ICP-MS, Waltham, MA, USA). UI concentrations were measured using an Iodine standard (Inorganic Venture, Christiansburg, VA, USA). Iodine concentrations were adjusted using creatinine concentration to correct for variable water excretion rates at the time of spot urine specimen collection.
Definition of the disease-free and reference populations
The disease-free population was defined as subjects with no prior history of thyroid disease and no history of taking medication that could influence thyroid function. The reference population was defined as subjects with no prior history of thyroid disease, no history of taking medication that could influence thyroid function, no family history of thyroid disease, negative TPOAb results, and serum fT4 levels in the reference range.
Statistical analysis
Statistical analysis was performed with SAS survey procedures version 9.4 (SAS Institute Inc., Cary, NC, USA) reflecting sample design and weight. The TSH reference range and prevalence of thyroid dysfunctions were compared after age-standardization based on the projected population in 2005. The estimated population was not calculated based on the suppression rule for proportion estimates of the United States (US) National Center for Health Statistics as the estimated prevalence of thyroid dysfunctions had a low statistical precision.
RESULTS
Baseline characteristics of the subjects
A total of 6,564 subjects underwent thyroid function testing in the KNHANES VI (2013 to 2015). The median age was 42 years (interquartile range [IQR], 28 to 56), and 3,378 males (52.8%) and 3,186 females (47.2%) were included in this study. The baseline characteristics including the distribution of each age group are summarized in Table 1. A history of thyroid disease was present in 169 subjects (2.6%), including 0.8% of males and 4.5% of females. The median UI concentration was 299.4 μg/L (IQR, 158.9 to 700.1 μg/g) and the urine iodine-to-creatinine ratio was 192.6 μg/g (IQR, 103.2 to 477.6). A positive TPOAb result was detected in 480 subjects (7.3%), including 4.3% of males and 10.5% of females. The disease-free population comprised 6,326 of the 6,564 subjects (96.4%), and the reference population comprised 5,574 subjects (84.9%).
Table 1. Baseline Characteristics of Subjects Who Underwent Thyroid Function Tests as Part of the Korea National Health and Nutrition Examination survey (2013 to 2015).
| Characteristic | Total sample | Disease-free populationa | Reference populationb | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | Male | Female | Total | Male | Female | Total | Male | Female | |
| No. of subjects | 6,564 | 3,378 (52.8) | 3,186 (47.2) | 6,326 | 3,340 (54.4) | 2,986 (45.6) | 5,574 | 3,060 (56.4) | 2,514 (43.6) |
| Age, yr | 42 (28–56) | 41 (26–54) | 44 (29–58) | 42 (27–55) | 41 (26–54) | 44 (28–57) | 41 (26–55) | 41 (26–54) | 43 (27–57) |
| Age range, yr | |||||||||
| 10–18 | 909 (13.8) | 517 (15.3) | 392 (12.3) | 908 (14.4) | 517 (15.5) | 391 (13.1) | 845 (15.2) | 485 (15.8) | 360 (14.3) |
| 19–29 | 1,106 (16.8) | 566 (16.8) | 540 (16.9) | 1,098 (17.4) | 563 (16.9) | 535 (17.9) | 981 (17.6) | 520 (17.0) | 461 (18.3) |
| 30–39 | 1,084 (16.5) | 556 (16.5) | 528 (16.6) | 1,040 (16.4) | 548 (16.4) | 492 (16.5) | 918 (16.5) | 505 (16.5) | 413 (16.4) |
| 40–49 | 1,101 (16.8) | 569 (16.8) | 532 (16.7) | 1,045 (16.5) | 562 (16.8) | 483 (16.2) | 907 (16.3) | 515 (16.8) | 392 (15.6) |
| 50–59 | 1,196 (18.2) | 584 (17.3) | 612 (19.2) | 1,133 (17.9) | 575 (17.2) | 558 (18.7) | 968 (17.4) | 524 (17.1) | 444 (17.7) |
| 60–69 | 1,058 (16.1) | 527 (15.6) | 531 (16.7) | 997 (15.8) | 518 (15.5) | 479 (16.0) | 860 (15.4) | 460 (15.0) | 400 (15.9) |
| ≥70 | 110 (1.7) | 59 (1.7) | 51 (1.6) | 105 (1.7) | 57 (1.7) | 48 (1.6) | 95 (1.7) | 51 (1.7) | 44 (1.8) |
| Urinary iodine, µg/L | 299.4 (158.9–700.1) | 295.3 (167.1–673.7) | 301.2 (150.8–747.6) | 298.4 (159.1–694.4) | 295.9 (167.3–674.6) | 300.3 (150.4–730.2) | 298.7 (160.1–692.0) | 295.3 (168.8–669.7) | 300.2 (150.8–736.1) |
| Urine iodine/Cr, µg/g | 192.6 (103.2–477.6) | 169.0 (92.2–379.4) | 228.8 (120.0–595.5) | 190.7 (102.2–466.4) | 169.0 (92.2–378.9) | 224.4 (117.7–571.8) | 191.0 (101.6–459.3) | 169.0 (92.3–374.0) | 224.8 (116.6–571.8) |
| TPOAb positivityc | 480 (7.3) | 145 (4.3) | 335 (10.5) | 388 (5.3) | 128 (3.8) | 260 (8.7) | NA | NA | NA |
| History of thyroid | 169 (2.6) | 26 (0.8) | 143 (4.5) | NA | NA | NA | NA | NA | NA |
Values are expressed as number (%) or median (interquartile range).
Cr, creatinine; TPOAb, anti-thyroid peroxidase antibody.
aThe disease-free population was defined as subjects with no prior history of thyroid disease and no history of taking medication that could influence thyroid function; bThe reference population was defined subjects with no prior history of thyroid disease, no history of taking medications that could influence thyroid function, no family history of thyroid disease, negative TPOAb results, and serum free thyroxine levels in the reference range (0.89 to 1.76 ng/dL); cTPOAb positivity was defined as >34.0 IU/mL.
Serum TSH and fT4 concentrations
In the total population, the median serum TSH level was 2.25 mIU/L (IQR, 1.53 to 3.30). The value of the 2.5th and 97.5th percentiles of serum TSH levels was 0.51 and 7.61 mIU/L, respectively (Table 1). In the disease-free population, the median serum TSH level was 2.26 mIU/L (IQR, 1.55 to 3.28). The median serum TSH concentration in males was 2.17 mIU/L (IQR, 1.49 to 3.07) and that of females was 2.37 mIU/L (IQR, 1.62 to 3.57). The 2.5th percentile of serum TSH levels was 0.57 mIU/L (males 0.61 mIU/L, females 0.55 mIU/L) and the 97.5th percentile of serum TSH levels was 7.49 mIU/L (males 6.83 mIU/L, females 8.18 mIU/L).
In the reference population, the median serum TSH level was 2.23 mIU/L (IQR, 1.55 to 3.18). The median values of serum TSH levels in males and females were 2.15 mIU/L (IQR, 1.49 to 3.02) and 2.31 mIU/L (IQR, 1.62 to 3.35), respectively. The 2.5th percentile of serum TSH levels was 0.62 mIU/L (males 0.63 mIU/L, females 0.60 mIU/L), and the 97.5th percentile of serum TSH levels was 6.86 mIU/L (males 6.44 mIU/L, females 7.21 mIU/L). The reference interval for serum TSH levels was considered to be between 0.62 and 6.86 mIU/L in this study.
In the total sample, the median serum fT4 level was 1.23 ng/dL (IQR, 1.12 to 1.35), as shown in Table 2. In the disease-free population, the median serum fT4 level was 1.23 ng/dL (IQR, 1.12 to 1.35). The median serum fT4 concentration in males was 1.27 ng/dL (IQR, 1.16 to 1.40) and that in females was 1.18 ng/dL (IQR, 1.08 to 1.29). In the reference population, the median serum fT4 level was 1.24 ng/dL (IQR, 1.13 to 1.35). The median values of serum fT4 levels in males and females were 1.28 ng/dL (IQR, 1.16 to 1.40) and 1.19 ng/dL (IQR, 1.09 to 1.30), respectively.
Table 2. Serum TSH and fT4 Concentrations in the Total Sample, the Disease-Free Population and the Reference Population in Korea National Health and Nutrition Examination Survey (2013 to 2015).
| Variable | Total sample | Disease-free populationa | Reference populationb | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | Male | Female | Total | Male | Female | Total | Male | Female | |
| TSH, mIU/L | |||||||||
| 2.5th Percentile | 0.51 | 0.59 | 0.37 | 0.57 | 0.61 | 0.55 | 0.62 | 0.63 | 0.60 |
| 25th Percentile | 1.53 | 1.48 | 1.59 | 1.55 | 1.49 | 1.62 | 1.55 | 1.49 | 1.62 |
| Median | 2.25 | 2.17 | 2.36 | 2.26 | 2.17 | 2.37 | 2.23 | 2.15 | 2.31 |
| 75th Percentile | 3.30 | 3.08 | 3.59 | 3.28 | 3.07 | 3.57 | 3.18 | 3.02 | 3.35 |
| 97.5th Percentile | 7.61 | 6.94 | 8.45 | 7.49 | 6.83 | 8.18 | 6.86 | 6.44 | 7.21 |
| fT4, ng/dL | |||||||||
| 2.5th Percentile | 0.91 | 0.96 | 0.87 | 0.92 | 0.96 | 0.88 | 0.96 | 0.99 | 0.94 |
| 25th Percentile | 1.12 | 1.16 | 1.08 | 1.12 | 1.16 | 1.08 | 1.13 | 1.16 | 1.09 |
| Median | 1.23 | 1.28 | 1.19 | 1.23 | 1.27 | 1.18 | 1.24 | 1.28 | 1.19 |
| 75th Percentile | 1.35 | 1.40 | 1.30 | 1.35 | 1.40 | 1.29 | 1.35 | 1.40 | 1.30 |
| 97.5th Percentile | 1.63 | 1.65 | 1.58 | 1.61 | 1.64 | 1.55 | 1.60 | 1.61 | 1.53 |
TSH, thyroid stimulating hormone; fT4, free thyroxine.
aThe disease-free population was defined as subjects with no prior history of thyroid disease and no history of taking medications that could influence thyroid function; bThe reference population was defined as subjects with no prior history of thyroid disease, no history of taking medications that could influence thyroid function, no family history of thyroid disease, negative anti-thyroid peroxidase antibody results, and serum fT4 levels in the reference range (0.89 to 1.76 ng/dL).
Prevalence of hypothyroidism in Korea
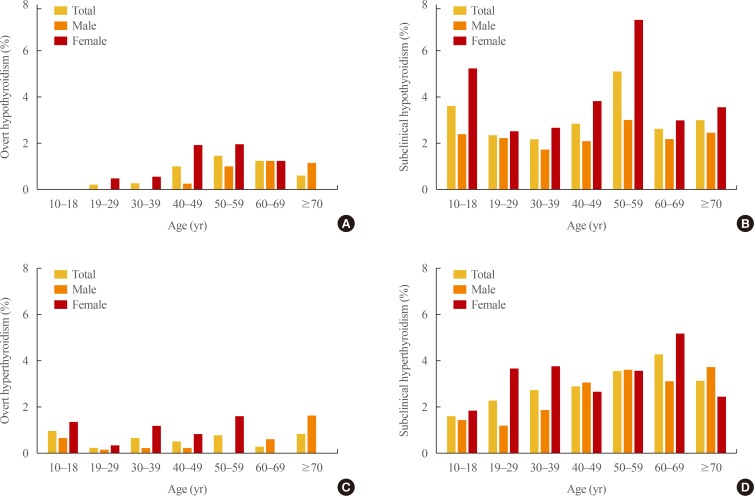
The prevalence of overt hypothyroidism and subclinical hypothyroidism in Korea was 0.73% and 3.10%, respectively (Table 3). The prevalence of overt hypothyroidism was 0.40% in males and 1.10% in females. The prevalence of subclinical hypothyroidism in males and females was 2.26% and 4.04%, respectively. The prevalence of overt and subclinical hypothyroidism according to sex and age group is illustrated in Fig. 1A, B. The prevalence of overt and subclinical hypothyroidism increased with age until the age group between 50 to 59 years (50s). The prevalence of overt hypothyroidism in 50s was 1.44% (males 0.97%, females 1.93%). The prevalence of subclinical hypothyroidism in 50s was 5.09% (males 2.98%, females 7.32%).
Table 3. Prevalence of Hypothyroidism and Hyperthyroidism in Korea according to Age and TPOAb Positivity.
| Variable | Overt hypothyroidisma | Subclinical hypothyroidismb | Overt hyperthyroidismc | Subclinical hyperthyroidismd | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | Male | Female | Total | Male | Female | Total | Male | Female | Total | Male | Female | |
| Total population, stratified by age, yr | ||||||||||||
| All ages | 0.73 | 0.40 | 1.10 | 3.10 | 2.26 | 4.04 | 0.54 | 0.30 | 0.81 | 2.98 | 2.43 | 3.59 |
| 10–18 | 0.00 | 0.00 | 0.00 | 3.60 | 2.40 | 5.23 | 0.94 | 0.63 | 1.34 | 1.60 | 1.41 | 1.85 |
| 19–29 | 0.20 | 0.00 | 0.45 | 2.34 | 2.20 | 2.52 | 0.25 | 0.19 | 0.31 | 2.26 | 1.18 | 3.65 |
| 30–39 | 0.24 | 0.00 | 0.54 | 2.15 | 1.71 | 2.68 | 0.64 | 0.20 | 1.17 | 2.73 | 1.86 | 3.78 |
| 40–49 | 0.98 | 0.23 | 1.91 | 2.86 | 2.09 | 3.81 | 0.49 | 0.22 | 0.83 | 2.88 | 3.05 | 2.66 |
| 50–59 | 1.44 | 0.97 | 1.93 | 5.09 | 2.98 | 7.32 | 0.78 | 0.00 | 1.60 | 3.57 | 3.60 | 3.53 |
| 60–69 | 1.22 | 1.21 | 1.22 | 2.62 | 2.17 | 2.98 | 0.26 | 0.60 | 0.00 | 4.30 | 3.14 | 5.20 |
| ≥70 | 0.58 | 1.13 | 0.00 | 2.97 | 2.41 | 3.55 | 0.83 | 1.63 | 0.00 | 3.13 | 3.75 | 2.48 |
| Total population, stratified by TPOAb | ||||||||||||
| Positive TPOAb | 5.16 | 5.92 | 4.81 | 10.88 | 9.10 | 11.69 | 2.92 | 2.44 | 3.13 | 3.80 | 4.06 | 3.67 |
| Negative TPOAb | 0.39 | 0.16 | 0.66 | 2.49 | 1.95 | 3.13 | 0.35 | 0.20 | 0.54 | 2.92 | 2.36 | 3.58 |
Values are expressed as percentage. TPOAb positivity was defined as >34.0 IU/mL.
TPOAb, anti-thyroid peroxidase antibody.
aOvert hypothyroidism was defined thyroid stimulating hormone (TSH) (mlU/L) >97.5th percentile and free thyroxine (fT4) (ng/dL) <0.9; bSubclinical hypothyroidism was defined TSH (mlU/L) >97.5th percentile and 0.9≤fT4 (ng/dL)<1.8; cOvert hyperthyroidism was defined TSH (mlU/L) <2.5th percentile and fT4 (ng/dL) ≥1.8; dOvert hyperthyroidism was defined TSH (mlU/L) <2.5th percentile and 0.9≤fT4 (ng/dL)<1.8.
Fig. 1.
Prevalence of overt and subclinical thyroid dysfunction according to age and gender in Korea. (A) Prevalence of overt hypothyroidism and (B) subclinical hypothyroidism. (C) Prevalence of overt hyperthyroidism and (D) subclinical hyperthyroidism.
The prevalence of overt and subclinical hypothyroidism in subjects with positive TPOAb results was 5.16% (males 5.92%, females 4.81%) and 10.88% (males 9.10%, females 11.69%), respectively. The corresponding rates in the subjects with negative TPOAb results were 0.39% (males 0.16%, females 0.66%) and 2.49% (males 1.95%, females 3.13%), respectively.
Prevalence of hyperthyroidism in Korea
The prevalence of overt hyperthyroidism and subclinical hyperthyroidism in Korea was 0.54% and 2.98%, respectively (Table 3). The prevalence of overt hyperthyroidism was 0.30% in males and 0.81% in females. The prevalence of subclinical hypothyroidism in males and females was 2.43% and 3.59%, respectively. The prevalence of overt and subclinical hyperthyroidism according to sex and age groups is presented in Fig. 1C, D. The prevalence of subclinical hyperthyroidism increased until the age group between 60 and 69 years (60s, 4.30%).
The prevalence of overt and subclinical hyperthyroidism in subjects with positive TPOAb results was 2.92% (males 2.44%, females 3.13%) and 3.80% (males 4.06%, female 3.67%), respectively. The corresponding rates in subjects with negative TPOAb results were 0.35% (males 0.20%, female 0.54%) and 2.92% (males 2.36%, females 3.58%), respectively.
Prevalence of TPOAb positivity in Korea
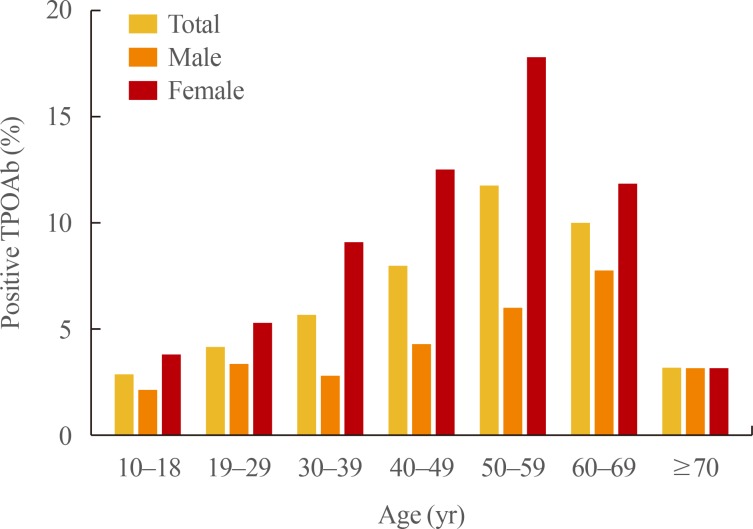
The prevalence of TPOAb positivity was 4.33% in males and 10.62% in females (Table 4). The prevalence of TPOAb positivity according to sex and age group is presented in Fig. 2. The prevalence of TPOAb positivity in the total population increased with age until 50s (11.71%). The prevalence of TPOAb positivity in 50s was 5.97% in males and 17.78% in females. The corresponding values in 60s were 7.71% in males and 11.80% in females.
Table 4. Prevalence of TPOAb Positivity in Korea according to Age.
| Variable | Positive TPOAb results, % | ||
|---|---|---|---|
| Total | Male | Female | |
| Total population, stratified by age, yr | |||
| All ages | 7.30 | 4.33 | 10.62 |
| 10–18 | 2.81 | 2.12 | 3.73 |
| 19–29 | 4.13 | 3.27 | 5.23 |
| 30–39 | 5.63 | 2.76 | 9.08 |
| 40–49 | 7.94 | 4.22 | 12.54 |
| 50–59 | 11.71 | 5.97 | 17.78 |
| 60–69 | 10.01 | 7.71 | 11.80 |
| ≥70 | 3.13 | 3.13 | 3.14 |
TPOAb positivity was defined as >34.0 IU/mL.
TPOAb, anti-thyroid peroxidase antibody.
Fig. 2.
Prevalence of subjects with positive anti-thyroid peroxidase antibody (TPOAb) results according to age and gender in Korea.
DISCUSSION
This nation-wide cross-sectional study was the first to evaluate the distribution of serum TSH levels in the Korean population. We evaluated the levels of serum TSH in the total population, the disease-free population, and the reference population to compare our data with the results of the National Health and Nutrition Examination Surveys (NHANES) in the US. In the US NHANES III data (1988 to 1994), the 2.5th and 97.5th percentiles of serum TSH in the reference population were 0.45 and 4.12 mIU/L, respectively [8]. However, those corresponding values in the reference population in our study were 0.62 and 6.86 mIU/L. This finding is also compatible with those of a recent single-institutional study in Korea that indicated the 2.5th and 97.5th percentiles of serum TSH were 0.73 and 7.06 mIU/L, respectively [22]. The median serum TSH level in the reference population was also higher in our study (2.23 mIU/L) than that in the US NHANES III data (1.39 mIU/L). This difference in serum TSH levels can be explained by ethnic differences. In the US NHANES III data, the 2.5th and 97.5th percentiles of serum TSH in the reference groups were 0.49 and 4.18 mIU/L in whites, 0.35 and 3.63 mIU/L in blacks, 0.43 and 3.91 mIU/L in Maxican-Americans, and 0.40 and 4.19 mIU/L in the remaining races [8]. The discrepancy with the distribution of serum TSH levels in Koreans was greater than expected. These findings suggest that other factors might have contributed to the higher levels of serum TSH in the Korean population.
Iodine intake could be an important factor contributing to the distribution of serum TSH in the Korean population. The median value of UI was 299.4 μg/L in our study, whereas the corresponding values found in the US data are 145 μg/L (1988 to 1994) and 160 μg/L (2003 to 2004) [26,27]. Previously, several studies have suggested that iodine intake affects the reference interval of serum TSH and the prevalence of thyroid disease [11,19,20]. A Chinese study observed a UI-related increase of serum TSH levels in areas with different iodine intake levels. In that study, the 2.5th and 97.5th percentiles of serum TSH levels in the reference group were 0.59 and 5.98 mIU/L, respectively, in areas with excess iodine consumption, and 0.33 and 3.42 mIU/L in iodine-deficient areas [17]. In the US NHANES III, significantly higher TSH concentrations were found in persons with high UI-to-creatinine ratios, but not in people with low iodine-to-creatinine ratios [8]. These findings supported the possibility that iodine consumption is an important factor associated with TSH concentrations areas such as Korea where large amounts of iodine are consumed.
The prevalence of subclinical hypothyroidism and hyperthyroidism can vary according to the reference value of serum TSH because these conditions are defined in terms of elevated or depressed levels of serum TSH with a normal serum fT4 level. The prevalence of subclinical hypothyroidism in our study was 3.1%. In the US NHANES III, the prevalence of subclinical hypothyroidism was 4.3%, with a cutoff value of serum TSH greater than 4.5 mIU/L [8]. However, the prevalence of subclinical hypothyroidism in the Ansung cohort and Korean Longitudinal Study on Health and Aging study was 11.7% and 17.3%, respectively, with a cutoff value of serum TSH greater than 4.1 mIU/L [28]. That study did not evaluate the distribution of serum TSH in the study population and the reference population. If a lower reference value of serum TSH is used in areas with excess iodine consumption, the prevalence of subclinical hypothyroidism would be overestimated and that of subclinical hyperthyroidism would be underestimated.
In our study, we included 6,564 subjects who were older than 10 years representing the Korean population. The age distribution of subjects in each decade was between 13.8% to 18.2% except those with older than 70 years (1.7%). Men and women were accounted for 52.8% and 47.2% of the total study population, respectively. In US NHANES III, thyroid function testing was done in a sample of 17,353 people (aged between 12 and 74 years) representing the geographic and ethnic distribution of the US population were included [8]. The age distribution of the study subjects in each decade and the proportion of each gender were similar with our study except individuals older than 70 years. We included relatively small portion of subjects who were older than 70 years. The US NHANES III did not include subjects who were older than 75 years old. Therefore, it needs to be cautious when apply our results directly to the older populations.
This study has several limitations. This is a cross-sectional study and it was not possible to evaluate the changes in thyroid function in each individual. The study only included noninstitutionalized subjects and might have excluded individuals at a higher risk for thyroid disease. Twenty-four-hour urine samples were not available for the improved assessment of iodine intake. The presence of thyroid disease, the use of medicines that could influence thyroid function, and the presence of a family history of thyroid disease were self-reported. More detailed histories and physical examination findings were not available. Thyroid function tests were not performed in children under the age of 10 years. Nonetheless, this is the first nation-wide epidemiological study to evaluate the prevalence of hypothyroidism and hyperthyroidism in Korea by researches from both the Korean Thyroid Association and the Centers for Disease Control and Prevention.
In conclusion, the reference interval of TSH in the Korean population was found to be between 0.62 and 6.68 mIU/L, which is higher than that of Western countries. Based on this reference interval, the prevalence of overt and subclinical hypothyroidism was 0.73% and 3.10%, respectively. Overt and subclinical hyperthyroidism was present in 0.54% and 2.98% of the Korean population, respectively. Differences were found in the prevalence of hypothyroidism and hyperthyroidism according to age, sex, and TPOAb positivity. This study provides important baseline information for understanding patterns of thyroid dysfunction and disease in Korea.
Footnotes
CONFLICT OF INTEREST: No potential conflict of interest relevant to this article was reported.
References
- 1.Cooper DS. Subclinical hypothyroidism. JAMA. 1987;258:246–247. [PubMed] [Google Scholar]
- 2.Biondi B, Cooper DS. The clinical significance of subclinical thyroid dysfunction. Endocr Rev. 2008;29:76–131. doi: 10.1210/er.2006-0043. [DOI] [PubMed] [Google Scholar]
- 3.Kim YA, Park YJ. Prevalence and risk factors of subclinical thyroid disease. Endocrinol Metab (Seoul) 2014;29:20–29. doi: 10.3803/EnM.2014.29.1.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aghini-Lombardi F, Antonangeli L, Martino E, Vitti P, Maccherini D, Leoli F, et al. The spectrum of thyroid disorders in an iodine-deficient community: the Pescopagano survey. J Clin Endocrinol Metab. 1999;84:561–566. doi: 10.1210/jcem.84.2.5508. [DOI] [PubMed] [Google Scholar]
- 5.Laurberg P, Pedersen KM, Hreidarsson A, Sigfusson N, Iversen E, Knudsen PR. Iodine intake and the pattern of thyroid disorders: a comparative epidemiological study of thyroid abnormalities in the elderly in Iceland and in Jutland, Denmark. J Clin Endocrinol Metab. 1998;83:765–769. doi: 10.1210/jcem.83.3.4624. [DOI] [PubMed] [Google Scholar]
- 6.O'Leary PC, Feddema PH, Michelangeli VP, Leedman PJ, Chew GT, Knuiman M, et al. Investigations of thyroid hormones and antibodies based on a community health survey: the Busselton thyroid study. Clin Endocrinol (Oxf) 2006;64:97–104. doi: 10.1111/j.1365-2265.2005.02424.x. [DOI] [PubMed] [Google Scholar]
- 7.Sawin CT, Castelli WP, Hershman JM, McNamara P, Bacharach P. The aging thyroid. Thyroid deficiency in the Framingham Study. Arch Intern Med. 1985;145:1386–1388. [PubMed] [Google Scholar]
- 8.Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, Spencer CA, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III) J Clin Endocrinol Metab. 2002;87:489–499. doi: 10.1210/jcem.87.2.8182. [DOI] [PubMed] [Google Scholar]
- 9.Bagchi N, Brown TR, Parish RF. Thyroid dysfunction in adults over age 55 years. A study in an urban US community. Arch Intern Med. 1990;150:785–787. [PubMed] [Google Scholar]
- 10.Canaris GJ, Manowitz NR, Mayor G, Ridgway EC. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000;160:526–534. doi: 10.1001/archinte.160.4.526. [DOI] [PubMed] [Google Scholar]
- 11.Teng W, Shan Z, Teng X, Guan H, Li Y, Teng D, et al. Effect of iodine intake on thyroid diseases in China. N Engl J Med. 2006;354:2783–2793. doi: 10.1056/NEJMoa054022. [DOI] [PubMed] [Google Scholar]
- 12.Parle JV, Franklyn JA, Cross KW, Jones SC, Sheppard MC. Prevalence and follow-up of abnormal thyrotrophin (TSH) concentrations in the elderly in the United Kingdom. Clin Endocrinol (Oxf) 1991;34:77–83. doi: 10.1111/j.1365-2265.1991.tb01739.x. [DOI] [PubMed] [Google Scholar]
- 13.Boekholdt SM, Titan SM, Wiersinga WM, Chatterjee K, Basart DC, Luben R, et al. Initial thyroid status and cardiovascular risk factors: the EPIC-Norfolk prospective population study. Clin Endocrinol (Oxf) 2010;72:404–410. doi: 10.1111/j.1365-2265.2009.03640.x. [DOI] [PubMed] [Google Scholar]
- 14.Surks MI, Hollowell JG. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab. 2007;92:4575–4582. doi: 10.1210/jc.2007-1499. [DOI] [PubMed] [Google Scholar]
- 15.Spencer CA, Hollowell JG, Kazarosyan M, Braverman LE. National Health and Nutrition Examination Survey III thyroid-stimulating hormone (TSH)-thyroperoxidase antibody relationships demonstrate that TSH upper reference limits may be skewed by occult thyroid dysfunction. J Clin Endocrinol Metab. 2007;92:4236–4240. doi: 10.1210/jc.2007-0287. [DOI] [PubMed] [Google Scholar]
- 16.Cho NH, Choi HS, Kim KW, Kim HL, Lee SY, Choi SH, et al. Interaction between cigarette smoking and iodine intake and their impact on thyroid function. Clin Endocrinol (Oxf) 2010;73:264–270. doi: 10.1111/j.1365-2265.2010.03790.x. [DOI] [PubMed] [Google Scholar]
- 17.Guan H, Shan Z, Teng X, Li Y, Teng D, Jin Y, et al. Influence of iodine on the reference interval of TSH and the optimal interval of TSH: results of a follow-up study in areas with different iodine intakes. Clin Endocrinol (Oxf) 2008;69:136–141. doi: 10.1111/j.1365-2265.2007.03150.x. [DOI] [PubMed] [Google Scholar]
- 18.Surks MI, Boucai L. Age- and race-based serum thyrotropin reference limits. J Clin Endocrinol Metab. 2010;95:496–502. doi: 10.1210/jc.2009-1845. [DOI] [PubMed] [Google Scholar]
- 19.Knudsen N, Bulow I, Jorgensen T, Laurberg P, Ovesen L, Perrild H. Comparative study of thyroid function and types of thyroid dysfunction in two areas in Denmark with slightly different iodine status. Eur J Endocrinol. 2000;143:485–491. doi: 10.1530/eje.0.1430485. [DOI] [PubMed] [Google Scholar]
- 20.Bulow Pedersen I, Knudsen N, Jorgensen T, Perrild H, Ovesen L, Laurberg P. Large differences in incidences of overt hyper- and hypothyroidism associated with a small difference in iodine intake: a prospective comparative register-based population survey. J Clin Endocrinol Metab. 2002;87:4462–4469. doi: 10.1210/jc.2002-020750. [DOI] [PubMed] [Google Scholar]
- 21.Baloch Z, Carayon P, Conte-Devolx B, Demers LM, Feldt-Rasmussen U, Henry JF, et al. Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid. 2003;13:3–126. doi: 10.1089/105072503321086962. [DOI] [PubMed] [Google Scholar]
- 22.Kim M, Kim TY, Kim SH, Lee Y, Park SY, Kim HD, et al. Reference interval for thyrotropin in a ultrasonography screened Korean population. Korean J Intern Med. 2015;30:335–344. doi: 10.3904/kjim.2015.30.3.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Seo GH, Chung JH. Incidence and prevalence of overt hypothyroidism and causative diseases in Korea as determined using claims data provided by the Health Insurance Review and Assessment Service. Endocrinol Metab (Seoul) 2015;30:288–296. doi: 10.3803/EnM.2015.30.3.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Seo GH, Kim SW, Chung JH. Incidence & prevalence of hyperthyroidism and preference for therapeutic modalities in Korea. J Korean Thyroid Assoc. 2013;6:56–63. [Google Scholar]
- 25.Ministry of the Interior. Seegene Medical Foundation, Laboratory Medicine of the Korea National Health and Nutrition Examination Survey VI (2013–2015) [Internet] Seoul: Centers for Disease Control and Prevention; 2015. [updated 2015 Dec 18]. [cited 2017 Jan 23]. Available from: http://www.prism.go.kr/homepage/entire/retrieveEntireDetail.do?pageIndex=1&research_id=1351000-201500293&leftMenuLevel=160&cond_research_name=%EC%A7%84%EB%8B%A8%EC%9D%98%ED%95%99%EA%B2%80%EC%82%AC&cond_research_start_date=&cond_research_end_date=&pageUnit=10&cond_order=3. [Google Scholar]
- 26.Hollowell JG, Staehling NW, Hannon WH, Flanders DW, Gunter EW, Maberly GF, et al. Iodine nutrition in the United States. Trends and public health implications: iodine excretion data from National Health and Nutrition Examination Surveys I and III (1971-1974 and 1988-1994) J Clin Endocrinol Metab. 1998;83:3401–3408. doi: 10.1210/jcem.83.10.5168. [DOI] [PubMed] [Google Scholar]
- 27.Caldwell KL, Miller GA, Wang RY, Jain RB, Jones RL. Iodine status of the U.S. population, National Health and Nutrition Examination Survey 2003-2004. Thyroid. 2008;18:1207–1214. doi: 10.1089/thy.2008.0161. [DOI] [PubMed] [Google Scholar]
- 28.Choi HS, Park YJ, Kim HK, Choi SH, Lim S, Park DJ, et al. Prevalence of subclinical hypothyroidism in two population based-cohort: Ansung and KLoSHA cohort in Korea. J Korean Thyroid Assoc. 2010;3:32–40. [Google Scholar]