Abstract
Significance: Metabolic syndrome is a frequent precursor of type 2 diabetes mellitus (T2D), a disease that currently affects ∼8% of the adult population worldwide. Pancreatic beta-cell dysfunction and loss are central to the disease process, although understanding of the underlying molecular mechanisms is still fragmentary.
Recent Advances: Oversupply of nutrients, including glucose and fatty acids, and the subsequent overstimulation of beta cells, are believed to be an important contributor to insulin secretory failure in T2D. Hypoxia has also recently been implicated in beta-cell damage. Accumulating evidence points to a role for oxidative stress in both processes. Although the production of reactive oxygen species (ROS) results from enhanced mitochondrial respiration during stimulation with glucose and other fuels, the expression of antioxidant defense genes is unusually low (or disallowed) in beta cells.
Critical Issues: Not all subjects with metabolic syndrome and hyperglycemia go on to develop full-blown diabetes, implying an important role in disease risk for gene–environment interactions. Possession of common risk alleles at the SLC30A8 locus, encoding the beta-cell granule zinc transporter ZnT8, may affect cytosolic Zn2+ concentrations and thus susceptibility to hypoxia and oxidative stress.
Future Directions: Loss of normal beta-cell function, rather than total mass, is increasingly considered to be the major driver for impaired insulin secretion in diabetes. Better understanding of the role of oxidative changes, its modulation by genes involved in disease risk, and effects on beta-cell identity may facilitate the development of new therapeutic strategies to this disease. Antioxid. Redox Signal. 26, 501–518.
Keywords: : insulin secretion, hypoxia, zinc, diabetes, beta cell
Introduction
Alongside central obesity, hyperglycemia is included as a criterion in all existing definitions of the metabolic syndrome (61). Although usually accompanied by insulin resistance of peripheral tissues (notably adipose tissue, muscle, and the liver), the failure of the pancreatic beta cell to produce insulin in amounts that are sufficient to control blood glucose levels is a sine qua non for both prediabetes and full-blown type 2 diabetes mellitus (T2D) (5, 80, 168, 191, 199).
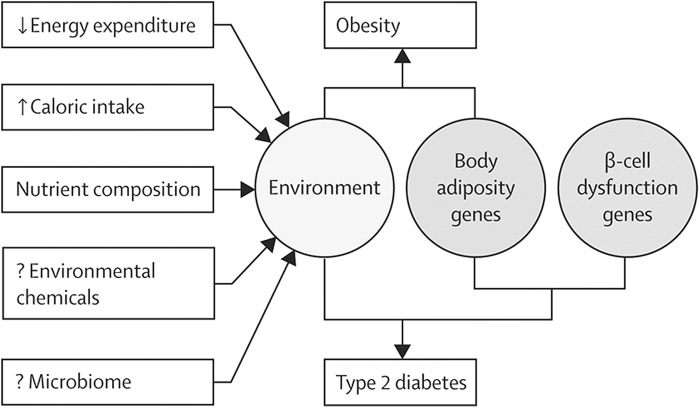
Further demonstrating the importance of disrupted beta-cell function for the development of T2D in the context of the metabolic syndrome, genome-wide association studies (38, 183) indicate that the majority of the known gene variants that increase the risk of T2D affect beta-cell function rather than insulin sensitivity (58, 164). The extent to which decreased beta-cell mass (24) and dysfunction (35) contribute to the impairments in insulin production in T2D is contested (168), although recent estimates of relatively minor changes (∼24% at diagnosis) in the former (112) have placed the onus on dysfunction as the important driver. The interaction of environment and genetic background in the development of obesity and T2D is depicted in Figure 1.
FIG. 1.
Role of genes and the environment in the development of obesity and type 2 diabetes. Interaction of genes that affect body adiposity with environmental factors results in development of obesity and associated insulin resistance. However, only when genes for abnormal beta-cell function are present along with those for body adiposity does interaction with the environment result in development of type 2 diabetes. [Reprinted from Kahn et al. (80) with permission from Elsevier].
Pancreatic beta cells are among the most metabolically active tissues within the human body, and they are highly dependent on oxidative metabolism for adenosine triphosphate (ATP) synthesis, particularly at elevated glucose concentrations (152, 176). Indeed, elevated oxygen consumption at high glucose levels is central to the stimulation of insulin secretion [(168) and see “Stimulation of the formation of ROS in beta cells by glucose”]. Accordingly, the pancreatic islet is efficiently perfused with blood (76, 107): Although islets occupy only 1–2% of the volume of the pancreas, they receive up to 15% of the pancreatic blood supply (77), and each beta cell is in direct contact with an endothelial cell (17).
Despite this high level of metabolic activity and the fact that reactive oxygen species (ROS) are an unavoidable by-product of mitochondrial respiration during glucose stimulation (and may even be required for normal glucose sensing) (100), enzymes involved in antioxidant defense are present at unusually low levels (103) or encoded by disallowed genes (152) in beta cells. As discussed below, this imbalance may render beta cells highly susceptible for damage induced by either oxidative stress or oxygen deprivation. This hypothesis will be reviewed here. We also discuss the interaction between GWAS genes, hypoxia, and oxidative stress and the possibility that in the metabolic syndrome the latter stressors may reduce functional beta-cell identity and insulin secretion without necessarily causing beta-cell destruction.
Formation of ROS in Pancreatic Beta Cells: The Role of Gluco–Lipotoxicity
Formation of ROS in pancreatic beta cells
The term “ROS” is generally used to describe reactive molecules containing oxygen. Although such molecules share some common characteristics, they also exhibit very different properties regarding their effects in biological systems, which may be either beneficial or toxic.
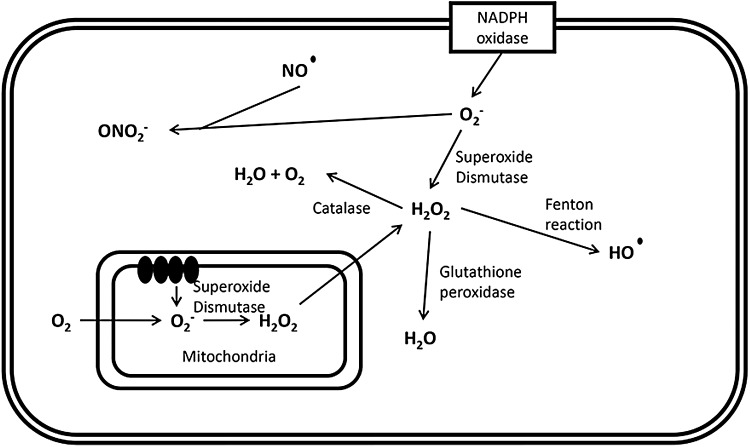
A major source of ROS within the pancreatic beta cell is the mitochondrial respiratory chain, as observed in other tissues (127, 200). Complexes I and III, located within the inner mitochondrial membrane, generate highly reactive superoxide (O2−) ions by single-electron reduction of molecular oxygen (101), which is—as a charged species—not able to freely cross biological membranes. However, it may do so via anion channels. Superoxide is converted to less active hydrogen peroxide (H2O2) by superoxide dismutase (SOD) isoenzymes. Being a less reactive, uncharged species, H2O2 can diffuse across membranes through aquaporins and be converted to highly reactive hydroxyl radicals (HO·). In addition, the formation of peroxynitrite (ONO2−) results from the reaction of superoxide with the free radical nitric oxide (NO) (135).
As well as by mitochondrial production, ROS are also generated by cytosolic and plasma membrane oxidoreductases, which oxidize NAD(P)H and directly produce ROS through the reduction of molecular oxygen (59). The general mechanisms of ROS production within the pancreatic beta cell are depicted in Figure 2.
FIG. 2.
Production of reactive oxygen species within the pancreatic beta cell. Basic mechanisms leading to the production of cellular reactive oxygen species, as described in the text, in section “Formulation of ROS in pancreatic beta cells.”
Stimulation of the formation of ROS in beta cells by glucose
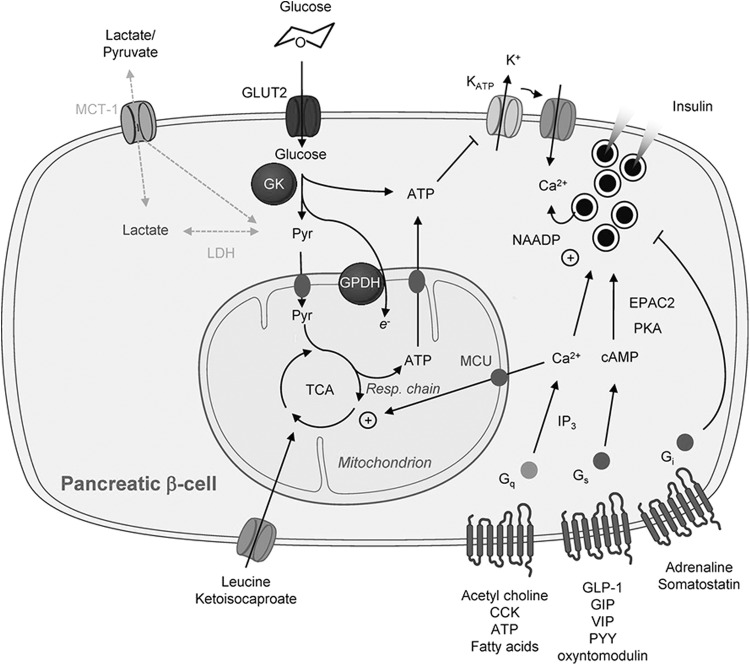
The main task assigned to the pancreatic beta cell is glucose-stimulated insulin secretion, the molecular basis of which is described below (Fig. 3). Briefly, after entering the cell via glucose transporters [with some continued debate about the role of GLUT1 and GLUT2 in human beta cells (118, 196)], glucose is phosphorylated by the high Michaelis–Menten constant (KM) hexokinase, glucokinase (102). Since the capacity for glucose transport is considerably higher than that of glucose phosphorylation, it is usually assumed that glucose concentrations quickly equilibrate across the plasma membrane, leaving glucose phosphorylation as the key step controlling glycolytic flux (168).
FIG. 3.
Overview of canonical signaling mechanisms involved in beta-cell glucose sensing and responses to secretory potentiators or inhibitors. See the text for further details, in section “Stimulation of the formation of ROS in beta cells by glucose.” This figure was originally published by G. A. Rutter in (168) (reprinted with permission).
An increase in the glycolytic flux is followed by increased tricarboxylic acid (TCA) cycle activity [as well as increased synthesis of TCA cycle intermediates via anaplerosis (174)], resulting in an increased ATP production in the mitochondria and a rise in ATP-to-ADP ratio within the cytoplasm (195). Subsequently, this promotes closure of the ATP-sensitive K+ (KATP) channels, which in turn decreases the hyperpolarizing outward K+ flux (5). As a result of these changes, depolarization of the plasma membrane occurs, followed by influx of extracellular Ca2+, a rise in intracellular Ca2+, and subsequent insulin secretion (163, 168). Other less well-defined changes, which depend on but do not lead to the closure of KATP channels, also contribute to the stimulation of secretion (67).
Compared to other mammalian cells, a rapid and proportional increase in glycolytic flux is observed in pancreatic beta cells following a rise in extracellular glucose concentrations. Uniquely in these cells, glycolytic flux is tightly coupled to increased mitochondrial oxidative activity (138, 176), with almost 100% of glucose carbons being fully oxidized to CO2 (174). This in part reflects low levels of lactate dehydrogenase (LDH) (2, 176) and plasma membrane lactate/pyruvate transporter (MCT-1/Slc16a1) (73, 213) activities, as well as the stimulation by high Ca2+ of mitochondrial glycerol phosphate dehydrogenase (167) and intramitochondrial Ca2+-sensitive dehydrogenases (36, 194) at high glucose.
The above increases in both glycolytic and TCA cycle flux are needed to assure adequate insulin secretion as a response to high blood glucose levels. However, when glucose clearance starts to be impaired due to peripheral insulin resistance, the continuous increase in glycolytic flux may also increase ROS production in the beta cell, with potential pathological consequences (Fig. 4). Notably, increases in intracellular Ca2+ are able to both stimulate the mitochondrial generation of ROS (155, 186) and activate protein kinase C (PKC), which in turn leads to NADPH oxidase-dependent generation of superoxide and other species (123, 209). In addition to these effects, there is evidence that not only hyperglycemia but also hyperinsulinemia itself, caused by peripheral insulin resistance, may contribute to the generation of hydrogen peroxide in beta cells (170). This is in contrast to other tissues, where insulin was found to reverse glucose-induced ROS generation (57).
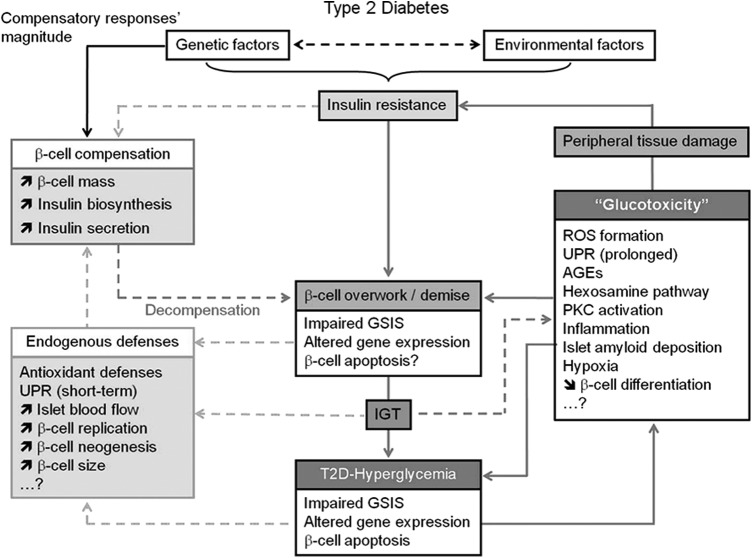
FIG. 4.
Schematic representation of the different mechanisms underlying beta-cell pathophysiology in T2D that precede and follow the establishment of hyperglycemia and the role of glucotoxicity in the aggravation of insulin resistance and beta-cell failure. In the absence of a defect in GSIS, beta cells maintain normoglycemia at the price of hyperinsulinemia. The extent of beta-cell compensation is thought to be predetermined genetically. This adaptation involves a coordinated increase in beta-cell mass, insulin biosynthesis, and insulin secretion. However, in genetically predisposed subjects, this phase is bypassed by a second phase of decompensation due to the inability of beta cells to sustain an adequate secretory response to match the organism demand (beta-cell overwork). This phase is characterized by the alteration of glucose-induced insulin secretion, gene expression and likely beta-cell apoptosis leading to the development of IGT, and finally the establishment of hyperglycemia with reduction of functional beta-cell mass. Chronic hyperglycemia leads to the exacerbation of beta-cell overwork and the alteration of beta-cell function and survival by several not fully understood mechanisms. In addition, hyperglycemia exerts toxic effects on peripheral tissues, which contribute to the aggravation of insulin resistance. Very importantly, beta-cell endogenous defenses are triggered in response to beta-cell failure and elevation of glycemia to restore the functional beta-cell mass. The imbalance between the protective effects of endogenous defenses and the deleterious effects of glucotoxicity is at the root of T2D pathology. [Reprinted from Bensellam et al. (12) with permission from Elsevier]. GSIS, glucose-induced insulin secretion; IGT, impaired glucose tolerance; T2D, type 2 diabetes mellitus.
Although high glucose levels are clearly associated with induction of oxidative stress, there is also evidence that low glucose levels promote ROS formation in pancreatic beta cells (70, 171). Furthermore, increasing glucose concentrations suppress the generation of superoxide in these cells (113). Of importance, the suppressive effects of glucose on superoxide production occurred over a low range of glucose concentrations (0–5 mM), consistent with a requirement for basal glucose flux to allow NAD(P)H formation, which in turn is able to suppress superoxide formation by complex I.
Furthermore, recent studies confirmed that lowering of the glucose concentration from 10 to 2 mM reversibly increases, rather than decreases, ROS production in pancreatic beta cells (161). Consecutive studies of the same authors were able to demonstrate that glucose (and other nutrients) acutely reduces the glutathione oxidation ratio in pancreatic beta cells in the mitochondrial matrix but not in the cytosol/nucleus (192). These changes inversely correlated with those in NAD(P)H autofluorescence, suggesting that they indirectly resulted from increased NADPH availability rather than from changes in ROS concentration.
Stimulation of the formation of ROS in beta cells by free fatty acids
Although the relationship between circulating free fatty acid (FFA) levels and obesity, insulin resistance, and dysregulation of fat metabolism is still debated (85), many studies have shown that individuals with insulin resistance exhibit higher FFA concentrations (119), as well as increased fat content in insulin-responsive tissues, including the skeletal muscle or liver (75, 96, 137). Impaired suppression of FFA oxidation and an increased rate of FFA release into the plasma are associated with fat distribution patterns typical of the metabolic syndrome and associated with visceral obesity (78) and hepatic lipid accumulation (nonalcoholic fatty liver disease, NAFLD) (46).
High concentrations of FFA induce ROS production in a variety of different tissues, including the pancreatic beta cell (26, 104). A variety of studies have examined the mechanisms by which FFA may induce this production. In vascular smooth muscle cells, there is evidence that this involves PKC-dependent activation of NAD(P)H oxidase (72). In insulin-secreting BRIN BD11 cells, in contrast, palmitate-induced ROS production was accompanied by an increase in the expression of the p47phox component of the NADPH oxidase (123), although this was also partially sensitive to the actions of the PKC inhibitor, GF109203X. Furthermore, oleate-induced inhibition of the respiratory chain was shown to contribute to enhanced ROS induction in insulin-secreting cells by others (95).
Recent studies focused on the subcellular compartment where ROS are produced, in particular with regard to lipotoxicity. It could be demonstrated that H2O2 formation in the peroxisomes rather than in the mitochondria is responsible for toxicity induced by saturated nonesterified fatty acids (NEFAs) (45). Further studies of these authors demonstrated that this H2O2 formation in peroxisomes by saturated NEFAs could be prevented by unsaturated NEFAs (53).
Of note, and via the inhibition of complexes I and III (173), ceramides derived from NEFAs promote ROS generation in heart mitochondria (37, 62). Whether a similar mechanism pertains in beta cells remains to be tested.
Effects of Oxidative Stress and Antioxidant Defenses in the Pancreatic Beta Cell
Positive and negative effects of reactive oxygen and nitrogen species in pancreatic beta cells
The potential deleterious effects of increased reactive oxygen and nitrogen production include oxidative damage to ribonucleic acids, proteins, and lipids by the mechanisms of nitration, carbonylation, peroxidation, and nitrosylation. Consequently, ROS/RNS may impact on the function and survival of the beta cell through a variety of mechanisms, including changes in enzyme activity, ion channel transport, receptor signal transduction, dysregulated gene expression, and apoptosis (88, 128).
Disruption of normal beta-cell function in response to oxidative stress has been demonstrated in multiple studies (110, 215).
For example, oxidative stress appears to be a major contributing factor to the pathophysiology of Friedreich's ataxia (4). Affected individuals are at increased risk of developing diabetes (48), and recent studies confirmed that loss of glucose tolerance is driven by dysfunction and loss of beta cells, as demonstrated by altered glucose tolerance and decreased beta-cell mass (31, 32). Friedreich's ataxia involves lowered expression of the frataxin protein due to the expansion of a GAA repeat in the FXN gene (111). Frataxin is located at the mitochondrial matrix and directs iron–sulfur cluster assembly (125). Deletion of this protein selectively in the beta cell in mice affected oxidative energy flux, impaired glucose tolerance, and led eventually to overt diabetes mellitus (156). Diabetes followed beta-cell growth arrest and apoptosis and was paralleled by an increase in ROS production (156).
Targets for oxidative stress in the beta cell are likely to include duodenal homeobox factor 1 (PDX-1), which plays an important role in pancreas development and differentiation, as well as in maintaining normal beta-cell function (130). Thus, oxidative stress, imposed by the exposure of rat islets to H2O2, reduced the DNA binding activity of PDX-1 and consequently lowered insulin gene expression (83, 114). This effect was mediated by the c-Jun N-terminal kinase (JNK) pathway, and later studies (86) suggested that decreased nuclear accumulation of PDX-1 resulted from the JNK activation and increased nuclear uptake of forkhead box protein O1 (FOXO1) (87).
Changes in the expression or activity of MafA, a member of the basic leucine zipper family of transcription factors involved in insulin gene expression (115), are also implicated in the deleterious effects of oxidative stress on beta cells (66). Thus, beta-cell-selective overexpression of the antioxidant enzyme glutathione peroxidase preserved intranuclear MafA and reversed diabetes in db/db mice (65). p38 MAPK is a major regulator of MafA protein stability under oxidative stress (94), and the prevention of p38 MAPK-mediated MafA degradation ameliorated beta-cell dysfunction under oxidative stress (44). Recent studies in transgenic db/db mice overexpressing MafA conditionally and specifically in beta cells have corroborated these findings (116).
Might there be a positive role(s) of, or a requirement for, reactive oxygen or nitrogen species for the normal function of pancreatic beta cells? Evidence for such a role does exist. Thus, an elegant study by Penicaud and colleagues in 2009 (100) demonstrated that mitochondria-derived ROS are necessary for normal glucose-stimulated insulin stimulation. While antioxidants in this study were able to blunt insulin secretion, insulin release could be induced with mitochondrial complex blockers that generate ROS. More recent studies (109) have confirmed these observations, demonstrating that ryanodine receptor-mediated Ca2+ release induced by ROS is an essential step in glucose-induced insulin secretion.
In addition, the subcellular localization of glucokinase, the key flux-generating step responsible for the stimulation of insulin secretion by glucose (see preceding sections), is also regulated in beta cells by insulin via NO production and S-nitrosylation, the latter leading to an association of the enzyme with secretory granules (157). Similarly, insulin granule exocytosis itself is enhanced by S-nitrosylation of syntaxin 4, a key mediator of granule docking at the plasma membrane (207).
Antioxidant defense strategies of pancreatic beta cells
Given the existence of both beneficial and deleterious actions of reactive oxygen and nitrogen species in the beta cell as described above, it might be anticipated that the antioxidant properties of pancreatic beta cells differ from those of other tissues. Supporting the view that the ability of these cells to counteract oxidative stress is limited, work conducted almost 20 years ago revealed that pancreatic islets exhibit an expression level of the antioxidant enzymes SOD, glutathione peroxidase, and particularly catalase that is substantially lower than most other tissues (103). Indeed, levels of catalase are so low that this enzyme falls into the class of beta-cell disallowed genes as defined by ourselves (151, 152) and others (197). Later work demonstrated that beta cells are particularly rich in other peroxidase-based antioxidant defenses, such as glutaredoxin and thioredoxin (74).
Of interest, but in accordance with the observation that the production of ROS is both necessary and at the same time potentially hazardous for normal beta-cell function, modulation of the different antioxidant systems can elicit either beneficial or adverse effects depending on the context. For example, whereas microinjection of glutaredoxin potentiated the effects of NADPH on exocytosis, thioredoxin antagonized the action of this nucleotide (74).
The role of the uncoupling protein 2 (UCP2) (21) as a possible antioxidant mechanism in beta cells has also been assessed (98, 142, 149, 160, 212). In the report of Zhang et al. (212), UCP2-deficient mice were found to have higher insulin levels and display increased glucose-stimulated insulin secretion which was attributed to increased glucose-stimulated ATP synthesis in these mice. This observation was confirmed in later studies, where beta-cell function was similarly shown to be enhanced in UCP2-null mice after induction of hyperglycemia by multiple low-dose streptozotocin (STZ) injections (98). It was proposed in the latter report that increased chronic ROS signaling in UCP2-deficient mice enhanced beta-cell function but impaired alpha-cell function, leading to an attenuation of STZ-induced hyperglycemia.
However, other studies with UCP2-depleted mice have reached different conclusions. Thus, examined in animals backcrossed for several generations onto highly congenic background, UCP2 deletion led to a significant increase in oxidative stress, as demonstrated by increased expression of antioxidant enzymes and nitrotyrosine staining. Furthermore, on the congenic background, islets from UCP2-null mice displayed impaired glucose-stimulated insulin secretion, although no overt hyperglycemia, hypoinsulinemia, or glucose intolerance was observed in vivo (142).
Further insights into these debated mechanisms were provided by the generation of a beta-cell-specific UCP2 knockout mouse model. Islets from these mice displayed, as expected, elevated intracellular ROS levels but enhanced glucose-stimulated insulin secretion. However, UCP2BKO mice were glucose intolerant, showing greater alpha-cell area, higher islet glucagon content, and aberrant ROS-dependent glucagon secretion under high-glucose conditions. Thus, it was concluded that, in beta cells, UCP2 contributes to the regulation of intraislet ROS signals that mediate changes in alpha-cell morphology and glucagon secretion (160). It should be noted, however, that the Cre deleter strain used in the latter studies (RIP2Cre) (52) is well known to cause deletion in extrapancreatic tissues, including the brain (206), and also to express human growth hormone (23), both of which may complicate the interpretation of the results of the above study.
Transgenic mice with beta-cell-specific overexpression of the UCP2 gene exhibited no significantly modified plasma glucose and insulin levels or glucose-induced insulin secretion (149). Similar results were obtained after UCP2 induction in the pancreatic beta-cell line INS-1, where glucose-induced insulin secretion was not altered. However, increased UCP2 levels resulted in a decreased production of ROS upon cytokine exposure (149), indicating a potential protective effect. This observation was confirmed by UCP2 knockdown experiments in a similar cell line, where it was shown that UCP2 activity prevents a glucose-induced increase in mitochondrial ROS production compared to cells lacking this factor (1).
The above results illustrate how a regulator of mitochondrial coupling of oxidative phosphorylation and ATP production, as well as ROS production, may influence beta-cell function in a complex manner, acting by a variety of mechanisms.
There is some controversy regarding possible differences between human and rodent islets regarding antioxidant defenses. Thus, early studies suggested a superior antioxidant capability of human islets compared to rodent islets (42, 43, 205). However, this idea was challenged later by reports indicating low levels of the major antioxidant systems in human islets (33, 159, 198).
Interestingly, differences in antioxidant levels in beta cells between males and females have been observed in studies with mice (33) as well as humans (198), indicating reduced oxidative defense mechanisms in females. This observation is of particular interest in the context of published evidence for a better defense against oxidative stress in women in general (150).
As a possible explanation for this observation, it was proposed that during evolution, beta cells have lost some of their antioxidant defense capacity to guarantee reduced insulin action in situations of increased stress, redirecting glucose disposal toward organs that are not insulin sensitive, in particular the brain (154). The same authors hypothesized that low levels of antioxidants in females facilitate an inhibitory role of ROS on insulin secretion, allowing glucose levels during pregnancy that are sufficient to meet the increasing nutritional requirements of the growing fetus.
Figure 5 outlines the ambiguous role of ROS as well as antioxidants in beta-cell function and oxidative damage.
FIG. 5.
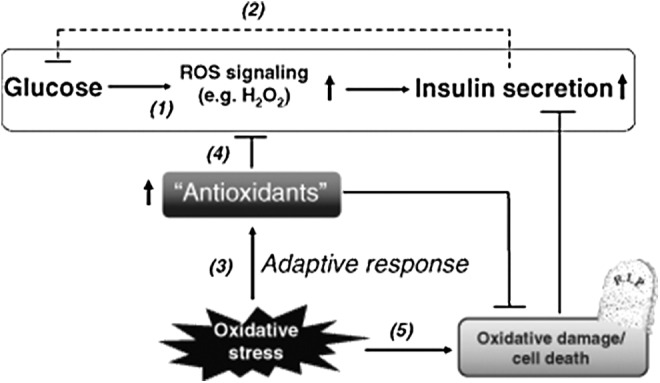
Simple scheme of how ROS could affect GSIS. 1. Glucose metabolism in beta cell produces transient increase in ROS. Together with other glucose-derived signals (e.g., ATP/ADP), insulin is secreted. 2. The action of insulin on target tissues (indicated by the dashed line) results in glucose uptake and a lowering of net plasma glucose levels. 3. Oxidative stress sets in motion the increase in enzymes and small-molecule antioxidants to scavenge these radicals. 4. This antioxidant response has the potential to also scavenge the transiently generated ROS in response to glucose in 1., thus blunting the GSIS response. 5. Exaggerated and persistent oxidative stress that is unrelieved by antioxidants can lead to cell damage and death; beta-cell destruction will also result in impaired GSIS. [Reprinted from Pi and Collins (143) with permission from Wiley]. ATP, adenosine triphosphate; ROS, reactive oxygen species.
Antioxidant Therapy for Pancreatic Beta Cells
Effects of antioxidant therapy on pancreatic beta-cell dysfunction
Studies in animal models of T2D have, nonetheless, demonstrated beneficial effects of an antioxidant treatment on the course of the disease. Thus, treatment of Zucker diabetic fatty rats with the antioxidant N-acetyl-L-cysteine (NAC) or aminoguanidine prevented hyperglycemia, glucose intolerance, defective insulin secretion, and decrements in beta-cell insulin content (193). Similarly, treatment of diabetic db/db mice with NAC, vitamins C plus E, or both has been evaluated. NAC treatment resulted in retained glucose-stimulated insulin secretion and moderately decreased blood glucose levels. Vitamins C and E were not effective when used alone but slightly effective when used in combination with NAC (82).
In humans, however, beneficial effects of antioxidant treatment on glycemic control and in particular on beta-cell function in T2D have not been observed consistently or when their effects were assessed in comprehensive meta-analyses (190).
Overall, the current data do not, therefore, support the theory that oxidative stress in the pancreatic beta cell is an initial causative element in the development of the metabolic syndrome and T2D but rather a contributing factor initiated by factors, such as gluco–lipotoxicity (as stated above). Thus, any therapy that aims at targeting oxidative stress within the beta cell can only considered being an additional therapy that reduces beta-cell stress arising from a suboptimal therapy for metabolic syndrome, as insulin resistance, hyperlipidemia, and hyperglycemia (158).
Antioxidant treatment in islet transplantation
Although not a useful clinical modality in T2D, transplantation of isolated islets of Langerhans for patients with type 1 diabetes emerged as an alternative to whole-organ pancreas transplantation in 2000 after a study from Shapiro and colleagues demonstrated persistent insulin independence in recipients when a steroid-free regimen was used (180). Subsequent data have confirmed improvement of glycemic control, although with shorter durations of insulin independence (99, 181).
Oxidative stress in isolated and transplanted islets remains a concern that counteracts successful implantation and function of the islet graft (20). Thus, strategies were developed to reduce oxidative stress already before transplantation. Treatment of human islets with SOD mimics by adding these compounds to islets in culture, after isolation, allowed for the survival of a significantly higher islet cell mass. Furthermore, treated islets were superior regarding restoration of glucose control in STZ-treated mice after transplantation of a marginal islet mass (19). Similarly, induction of radical scavenging heme oxygenase-1 (HO-1) was able to protect islets from apoptosis and improve functional performance in vitro as well as in an in vivo model of marginal mass islet transplantation (144).
Further studies aimed at the introduction of antioxidant agents already very early in the process of islet transplantation by treatment of the explanted pancreas with an infusion of L-glutamine through the main pancreatic duct. This led to an increase in islet yield, and the percentage of diabetic mice rendered normoglycemic with glutamine-treated islet transplants was higher than that with control islets (7). Recent work demonstrated that a reduction of ROS in isolated pancreatic islets and improved islet transplantation outcome with increased survival of diabetic recipient mice can also be achieved by administration of exendin-4, a glucagon-like peptide-1 analog (136).
Oxygen Availability and Hypoxia in the Pancreatic Beta Cell
Hypoxia induction in pancreatic beta cells
Similar to their high susceptibility to oxidative stress caused by nutrient excess, pancreatic beta cells are also prone to the stress of oxygen deprivation, which, paradoxically, also leads to ROS production and other signs of the oxidative stress (184). Under normal conditions, the high oxidative phosphorylation rate of pancreatic islets relies on the availability of a constant oxygen supply. However, reduced oxygen content of islets is not necessarily a sign of a pathological process.
In an interesting recent study (132), it was shown that oxygenation differs widely between individual islets within the pancreas at a given time point and that these differences may reflect a mechanism to recruit only a fraction of the available islets into an active (normoxic) beta-cell mass. The remaining less well-oxygenated islets may represent a dormant subpopulation, constituting a functional reserve of endocrine cells. According to this model, the reserve islet pool may be available for recruitment upon reduction of the total islet mass, as elegantly demonstrated by experiments using pancreatectomy in a rat model (132).
In the context of therapies for T1D, oxygen deprivation is always observed in pancreatic islets upon isolation and transplantation. Separation of islets from the exocrine tissue leads to disruption of the vascular access of islets and leaves diffusion as the only way that provides oxygen to the cells. Thus, hypoxia primarily occurs within the core of isolated islets (55) because a gradient in oxygen tension exists between the surface and the center of the islet, which increases with the square of the islet diameter (6, 131). Immediately after transplantation, implanted islets still remain dependent on oxygen diffusion from the surroundings for their survival (97). It is only after 1–3 months that vascularization of engrafted islets develops, a process that depends, among other factors, on implantation location. The liver, currently the main location of human islet transplantation, was shown to be inferior compared to other sites regarding islet revascularization (133).
Of major interest is the question whether processes associated with the development of beta-cell failure in the metabolic syndrome and T2D also involve hypoxic changes in pancreatic islets. Exposure to high glucose levels has been shown to induce hypoxia and hypoxia-induced pathways in pancreatic beta-cell lines and isolated islets (11). Additional studies provided evidence that islets of animals suffering from diabetes are particularly prone to hypoxia (172, 214).
Direct comparison of the susceptibility of different islet cell types to hypoxia revealed that hypoxia results in severe functional abnormality in both beta and alpha (glucagon producing) cells, but alpha cells display a significantly lower rate of apoptosis compared to the intensive apoptotic injury of beta cells (13). Gene expression profiling in islets of prediabetic Zucker diabetic fatty rats showed increased expression of hypoxia-related genes, and further investigation revealed a severely disturbed vascular integrity of these islets as a possible reason for the development of hypoxia (106).
Roles of hypoxia-inducible factors in pancreatic beta cells
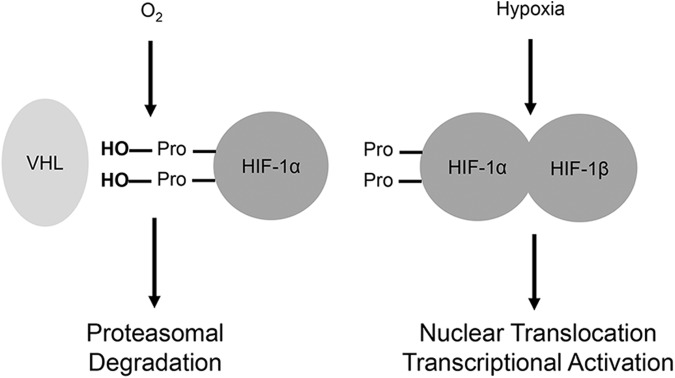
Hypoxia rapidly activates hypoxia-inducible factors (HIFs). These transcription factors, notably hypoxia-inducible factor 1alpha and 2alpha, are usually present in the cytosol. Under normoxic conditions, they are constantly hydroxylated at conserved proline residues by prolyl hydroxylases (177). This leads to their recognition and ubiquitination by the Von Hippel–Lindau (VHL) E3 ubiquitin ligase (117). In hypoxia, however, degradation of HIFs does not occur, allowing them to dimerize with the HIF-1beta subunit, be transported into the nucleus, and execute their transcriptional functions (79) (Fig. 6).
FIG. 6.
Activation of HIF-1alpha by hypoxia. Under normoxic conditions, hypoxia-inducible factors are hydroxylated at conserved proline residues by prolyl hydroxylases. This leads to their recognition and ubiquitination by the VHL E3 ubiquitin ligase. In hypoxia, hydroxylation and degradation of HIFs do not occur, allowing them to dimerize with the HIF-1beta subunit, be transported into the nucleus, and execute their transcriptional function. HIF, hypoxia-inducible factor; VHL, Von Hippel–Lindau.
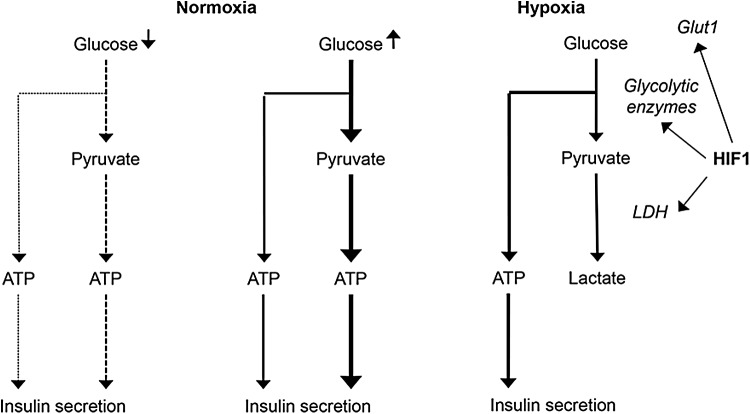
One of the main changes in cellular metabolism promoted by HIFs is a switch from aerobic to anaerobic energy production. Induction of glucose transporter 1 (GLUT1), several glycolytic enzymes, as well as LDH (178), and pyruvate dehydrogenase kinase 1 (PDK1) (91) reprograms the utilization of available glucose into anaerobic metabolism to maintain a high rate of glycolytic ATP production and at the same time downregulating mitochondrial oxygen consumption (139).
The specific effects exerted by this metabolic switch have been explored using mice deleted specifically in the beta cell for VHL. This modification leads to oxygen concentration-independent stabilization of HIFs (25, 153, 210). As might be expected, these animals exhibited stably increased rates of glycolysis, but unaffected or even increased insulin secretion in the fasting state, probably a result of a rise in ATP production. Glucose tolerance in these animals was markedly impaired, a change attributed directly to a disturbed beta-cell function by activation of the hypoxia-inducible pathway, since additional deletion of the factor HIF-1alpha in these VHL-deficient mice was able to rescue the phenotype (Fig. 7).
FIG. 7.
Insulin secretion in normoxia and hypoxia. Insulin secretion depends on the production of ATP and is, in normoxia, tightly regulated by extracellular glucose concentrations (Fig. 3). ATP is mainly not only produced by oxidative phosphorylation but also to some extent during glycolysis. In hypoxia, stabilization of hypoxia-inducible factor HIF-1alpha leads to an increase in anaerobic glycolytic flux independently of glucose levels. This may lead to higher insulin and lower glucose levels in the fasting state but clearly impairs glucose-induced insulin secretion during postprandial hyperglycemia.
Contrasting with these results, other workers (64) demonstrated that disruption, but not activation, of the hypoxia signaling pathway by knockout of HIF-1beta or HIF-1alpha (29) was also able to cause pancreatic beta-cell dysfunction with consecutive glucose intolerance in mice. The same group also observed a 90% decrease in the expression of HIF-1beta in islets from T2D patients compared to normal glucose-tolerant controls (64).
Taken together, and also considering the fact that hypoxia is probably involved in the regulation of general beta-cell activity as mentioned above, it is tempting to speculate that there might be a basal activity of the hypoxia-inducible pathway, which is necessary for normal beta-cell function but beneficial only on a low level. Disruption as well as prolonged increased activity of this pathway is followed by impaired function of beta cells.
Intermittent hypoxia
The phenomenon of intermittent hypoxia is of particular interest since it is a consequence of obstructive sleep apnea, which is a highly prevalent disorder in obese patients, characterized by repeated episodes of pharyngeal obstruction during sleep that lead to intermittent hypoxia, sleep fragmentation, and excessive daytime sleepiness (140). An association of obstructive sleep apnea with disturbed glucose tolerance and T2D could be shown by a variety of observational studies (18, 148, 162).
Experimental animal models confirmed an increase of insulin resistance in obese mice treated with intermittent hypoxia. In addition, experiments in diabetic mice demonstrated a significant drop in pancreatic tissue oxygenation and impaired insulin secretion as well as an activation of caspase-3 within islets upon treatment with intermittent hypoxia (182). Another study confirmed induction of beta-cell apoptosis by intermittent hypoxia and also demonstrated a protective effect of antioxidant treatment with N-acetylcysteine (47).
The role of hypoxia in beta-cell death
Prolonged hypoxia results in pancreatic beta-cell death, typically by necrosis (55). However, induction of apoptosis-related pathways upon hypoxia exposure has also been described. Thus, activated caspase-3 (146) colocalizes in pancreatic islets with HIF-1alpha (124), suggesting that activation of apoptosis occurs primarily in those parts of the islet where hypoxia is most profound. However, whether hypoxia is a cause or a consequence of apoptosis is still debated (60).
Although induction of proapoptotic pathways by HIFs has been described (185), HIF activation is thought to be mainly an adaptive response that allows cell survival when oxygen levels are low. This conclusion was demonstrated by comparing apoptosis rate and transplantation success between wild-type islets and islets lacking HIF-1alpha (188). The latter exhibited impaired survival and increased apoptosis. The same study presented similar results for human islets where HIF-1alpha was upregulated by chemical agents.
The Role of Zinc Ions in Oxidative Stress and Hypoxic Damage to Pancreatic Beta Cells
Role and regulation of zinc homeostasis in pancreatic beta cells
Only few years after the discovery and successful use of insulin in the treatment of patients with diabetes (8), it was also recognized that zinc ions (Zn2+) may play an important role in insulin crystallization (175). In particular, Zn2+ is present in secretory granules of beta cells, where insulin undergoes a maturation process, aggregating to form 2-Zn–hexameric complexes (14, 121, 187). This dimerization process reduces the solubility of the hexamer, causing crystallization within the granule, and the formation of a dense crystalline core. The process not only increases insulin storage capacity before secretion but also reduces the susceptibility to enzymatic degradation of insulin (40, 69). Reconversion to the monomeric form of insulin is necessary for its biological action. This happens immediately after the exposure of the granule interior to the extracellular milieu, with a concomitant substantial release of Zn2+ (49, 56).
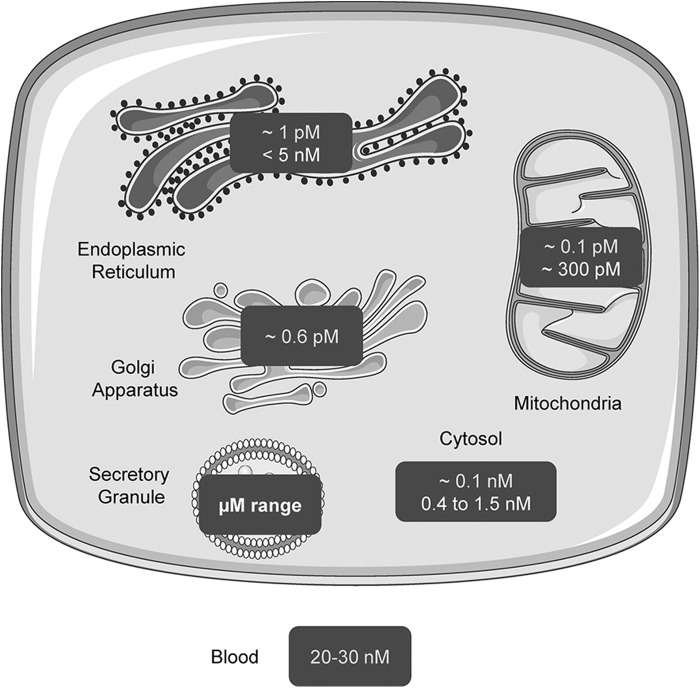
As an important structural and functional component of cells, the intracellular concentration and distribution of Zn2+ are tightly regulated (134, 201). The two main components of Zn2+ homeostasis are Zn2+ transporters and cytoplasmic Zn2+ buffers (34, 41, 81, 165). Whereas cytosolic free Zn2+ concentrations in pancreatic beta cells are comparable to those in other cells and quite low (∼1 nM) (10), they are much higher (∼120 nM) (68, 202) in the secretory granule: Corresponding total Zn2+ contents are in the mM and tens of mM range, respectively (51, 71) (Fig. 8).
FIG. 8.
Measurement of free Zn2+ concentrations in subcellular compartments in mammalian cells using genetically encoded sensors. Measurements with different genetically encoded zinc sensors using Förster Resonance Energy Transfer as the sensing modality and consisting of a donor and acceptor fluorescent protein linked by a zinc-binding peptide sequence. Dark numbers: Measurements with the sensor ZapCY1/2 (secretory granule: eZinCh-1); bright numbers: Measurements with the sensor eCALWY-4. While similar values were returned for all probes when located in the cytosol, much more variation exists for the mitochondria and endoplasmic reticulum. The reasons for these variations are unclear and may involve differences in intracellular pH on which the probes are steeply dependent. This figure was originally published by G.A. Rutter in (165) (reprinted with permission).
One of the most important regulatory transporters in beta cells, expressed almost uniquely in these cells and pancreatic alpha cells, is the zinc transporter ZnT8, responsible for zinc transport from the cytosol into the secretory granules (30). Paradoxically, however, deletion of ZnT8 from beta cells lowers both cytosolic and granular free Zn2+ (54, 122, 129). This zinc transporter became of major interest when genome-wide association studies (GWAS) revealed that a nonsynonymous single-nucleotide polymorphism (rs13266634) in the SLC30A8 gene, which encodes ZnT8, is associated with an ∼20% increase in T2D risk per allele (16, 92, 183). However, it is noteworthy that recent studies show that rare loss-of-function mutations in the SLC30A8 gene are associated with protection against T2D. The potential reasons for this apparent discrepancy are discussed elsewhere (166).
The role of Zn2+ in oxidative stress and hypoxia
Besides its specific role in insulin storage and secretion in the pancreatic beta cell, zinc is known for its antioxidative properties (22, 27, 147) and has been studied extensively as a possible treatment option in the context of diabetes in animal models (3, 208, 211) as well as human patients (50, 63). However, the results have been inconclusive possibly because changes in zinc intake are often too small to alter intracellular levels of Zn2+ to an extent that allows a measurable effect on oxidative stress. Nonetheless, zinc supplementation was found to lower fasting glucose levels in carriers of the common T2D risk allele at rs11558471 (84), and studies in rodent models of diabetes have indicated an action to improve insulin sensitivity (9).
A further challenge in the use of zinc supplementation as a therapeutic or prophylactic strategy in the metabolic syndrome is that the range of intracellular concentrations offering beneficial effects may be limited: From studies in neuronal tissues, it is well known that high levels of intracellular Zn2+ are able to induce production of ROS and that this effect can be attenuated by antioxidant treatment (145, 169). The dose dependency of the antioxidant versus oxidative stress-inducing effects of zinc was also demonstrated using isolated brain mitochondria (179).
Similarly, Zn2+ has “Janus-faced” properties regarding its effects in hypoxia. As described above, hypoxia induces apoptosis in pancreatic islets. Zinc is known to be a potent inhibitor of the apoptotic protease, caspase-3 (141), improves cell survival after hypoxia exposure (15), and protects tissues from ischemia-induced damage (203). In contrast, based on experiments performed with neuronal tissues, it is believed that rising zinc levels increase hypoxia-mediated cell death occurring during tissue ischemia (120, 189). Correspondingly, this effect can be reduced by the Zn2+ chelator N,N,N',N'-tetrakis(2-pyridylmethyl)ethane-1,2-diamine (TPEN) (108, 204).
In pancreatic beta cells, the effect of zinc on hypoxia-mediated effects has not been studied in detail. In general, it is known that high levels of zinc are cytotoxic and enhance pancreatic islet cell death (28, 89). In contrast, zinc increases the expression of metallothioneins (39, 93, 126), which are protective factors in pancreatic islets and other tissues vis-à-vis the deleterious effects of hypoxia and oxidative stress (90, 105).
Our own studies (54) have revealed that hypoxia has profound effects on cytosolic Zn2+ in pancreatic beta cells, downregulating zinc transporter ZnT8 and decreasing cytosolic zinc levels. When comparing the response to hypoxia of islets lacking ZnT8 completely, we detected a reduced rate of cell death in subpopulations of these islets in older mice. Thus, we concluded that reduced expression of ZnT8 and reduced cytosolic Zn2+ levels induced by hypoxia may reflect an adaptive response of beta cells to permit survival under hypoxia by reducing possible deleterious effects of high levels of zinc in this situation of acute stress (Fig. 9).
FIG. 9.
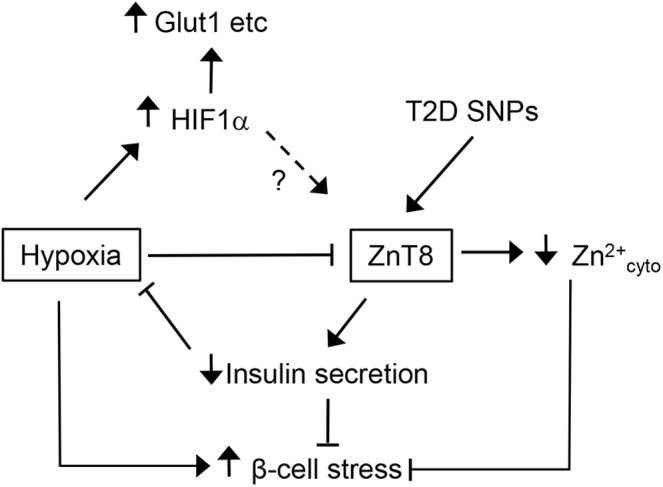
Possible role of ZnT8 in beta-cell adaptation to hypoxia. Hypoxia induces downregulation of the zinc transporter ZnT8. This may compromise insulin secretion but thereby also reduce further aggravation of hypoxia. Furthermore, the consecutive reduction of cytosolic Zn2+ concentrations possibly prevents deleterious effects of zinc in the situation of hypoxia-induced stress.
Concluding Remarks
Availability of oxygen, aerobic and anaerobic glycolysis, oxidative phosphorylation, and the production of ROS are closely linked to the normal function of the pancreatic beta cell since these processes are directly involved in glucose sensing and insulin release. Although hypoxia and oxidative stress are potentially harmful, and even lethal, processes for these metabolically highly active cells, evidence gathered during the past two decades indicates that both ROS and hypoxia-induced pathways have an important regulatory function in pancreatic beta cells. It became evident that these factors belong to the regulatory network of the beta cell and that changes in their availability and activation may lead to beta-cell failure and death.
There are still many open questions regarding the precise role of these processes in the development of disturbed glucose tolerance and T2D in the context of the metabolic syndrome, and further research is necessary to understand their contribution to the pathophysiology as well as possibilities to modulate them in a therapeutic way. To what extent, for example, does oxidative stress affect beta-cell identity—and consequently function—independently of cell survival (168)? Do T2D-associated genes in addition to SLC30A8 also modulate beta-cell responses to hypoxia or oxidative stress? Future studies will be needed to explore this and other questions and may lead to the identification of new ways to modulate the impact of oxidative stress for therapeutic benefit.
Abbreviations Used
- ATP
adenosine triphosphate
- FFA
free fatty acid
- FOXO1
forkhead box protein O1
- GLUT1
glucose transporter 1
- GSIS
glucose-induced insulin secretion
- H2O2
hydrogen peroxide
- HIF
hypoxia-inducible factor
- HO·
hydroxyl radical
- HO-1
heme oxygenase-1
- IGT
impaired glucose tolerance
- JNK
c-Jun N-terminal kinase
- Ko
knockout
- LDH
lactate dehydrogenase
- MAPK
mitogen-activated protein kinases
- NAC
N-acetyl-L-cysteine
- NADP
nicotinamide adenine dinucleotide phosphate
- NAFLD
nonalcoholic fatty liver disease
- NEFA
nonesterified fatty acid
- NO
nitric oxide
- NOS
nitric oxide synthase
- PDK1
pyruvate dehydrogenase lipoamide kinase isozyme 1
- PDX-1
pancreatic and duodenal homeobox 1
- PKC
protein kinase C
- RNS
reactive nitrogen species
- ROS
reactive oxygen species
- SOD
superoxide dismutase
- STZ
streptozotocin
- T1D
type 1 diabetes mellitus
- T2D
type 2 diabetes mellitus
- TCA
tricarboxylic acid
- TPEN
N,N,N',N'-tetrakis(2-pyridylmethyl)ethane-1,2-diamine
- UCP2
uncoupling protein 2
- VHL
Von Hippel–Lindau
- ZnT8
zinc transporter 8
Acknowledgments
GAR is supported by grants from the Wellcome Trust (Senior Investigator Award, WT098424AIA), an MRC Programme Grant (MR/J0003042/1), Biotechnology and Biological Sciences Research Council (BB/J015873/1) and Diabetes UK (11/0004210) project grants, an Imperial Confidence in Concept (ICiC) grant, and a Royal Society Wolfson Research Merit Award. PG was supported by the Foundation of Diabetes Research at the University Hospital Zurich. This work was also supported by the Innovative Medicines Initiative Joint Undertaking under grant agreement 155005 (IMIDIA), resources of which are composed of financial contribution from the European Union's Seventh Framework Programme (FP7/2007–2013), and European Federation of Pharmaceutical Industries and Associations (EFPIA) companies.
References
- 1.Affourtit C, Jastroch M, and Brand MD. Uncoupling protein-2 attenuates glucose-stimulated insulin secretion in INS-1E insulinoma cells by lowering mitochondrial reactive oxygen species. Free Radic Biol Med 50: 609–616, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alcazar O, Tiedge M, and Lenzen S. Importance of lactate dehydrogenase for the regulation of glycolytic flux and insulin secretion in insulin-producing cells. Biochem J 352: 373–380, 2000 [PMC free article] [PubMed] [Google Scholar]
- 3.Aly HF. and Mantawy MM. Comparative effects of zinc, selenium and vitamin E or their combination on carbohydrate metabolizing enzymes and oxidative stress in streptozotocin induced-diabetic rats. Eur Rev Med Pharmacol Sci 16: 66–78, 2012 [PubMed] [Google Scholar]
- 4.Armstrong JS, Khdour O, and Hecht SM. Does oxidative stress contribute to the pathology of Friedreich's ataxia? A radical question. FASEB J 24: 2152–2163, 2010 [DOI] [PubMed] [Google Scholar]
- 5.Ashcroft FM. and Rorsman P. Diabetes mellitus and the beta cell: the last ten years. Cell 148: 1160–1171, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Avgoustiniatos ES. and Colton CK. Effect of external oxygen mass transfer resistances on viability of immunoisolated tissue. Ann N Y Acad Sci 831: 145–167, 1997 [DOI] [PubMed] [Google Scholar]
- 7.Avila J, Barbaro B, Gangemi A, Romagnoli T, Kuechle J, Hansen M, Shapiro J, Testa G, Sankary H, Benedetti E, Lakey J, and Oberholzer J. Intra-ductal glutamine administration reduces oxidative injury during human pancreatic islet isolation. Am J Transplant 5: 2830–2837, 2005 [DOI] [PubMed] [Google Scholar]
- 8.Banting FG. and Best CH. The internal secretion of the pancreas. 1922. Indian J Med Res 125: 251–266, 2007 [PubMed] [Google Scholar]
- 9.Begin-Heick N, Dalpe-Scott M, Rowe J, and Heick HM. Zinc supplementation attenuates insulin secretory activity in pancreatic islets of the ob/ob mouse. Diabetes 34: 179–184, 1985 [DOI] [PubMed] [Google Scholar]
- 10.Bellomo EA, Meur G, and Rutter GA. Glucose regulates free cytosolic Zn(2)(+) concentration, Slc39 (ZiP), and metallothionein gene expression in primary pancreatic islet beta-cells. J Biol Chem 286: 25778–25789, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bensellam M, Duvillie B, Rybachuk G, Laybutt DR, Magnan C, Guiot Y, Pouyssegur J, and Jonas JC. Glucose-induced O(2) consumption activates hypoxia inducible factors 1 and 2 in rat insulin-secreting pancreatic beta-cells. PLoS One 7: e29807, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bensellam M, Laybutt DR, and Jonas JC. The molecular mechanisms of pancreatic beta-cell glucotoxicity: recent findings and future research directions. Mol Cell Endocrinol 364: 1–27, 2012 [DOI] [PubMed] [Google Scholar]
- 13.Bloch K, Vennang J, Lazard D, and Vardi P. Different susceptibility of rat pancreatic alpha and beta cells to hypoxia. Histochem Cell Biol 137: 801–810, 2012 [DOI] [PubMed] [Google Scholar]
- 14.Blundell TL, Cutfield JF, Cutfield SM, Dodson EJ, Dodson GG, Hodgkin DC, and Mercola DA. Three-dimensional atomic structure of insulin and its relationship to activity. Diabetes 21: 492–505, 1972 [DOI] [PubMed] [Google Scholar]
- 15.Bodiga VL, Thokala S, Vemuri PK, and Bodiga S. Zinc pyrithione inhibits caspase-3 activity, promotes ErbB1-ErbB2 heterodimerization and suppresses ErbB2 downregulation in cardiomyocytes subjected to ischemia/reperfusion. J Inorg Biochem 153: 49–59, 2015 [DOI] [PubMed] [Google Scholar]
- 16.Boesgaard TW, Zilinskaite J, Vanttinen M, Laakso M, Jansson PA, Hammarstedt A, Smith U, Stefan N, Fritsche A, Haring H, Hribal M, Sesti G, Zobel DP, Pedersen O, Hansen T, and Consortium E. The common SLC30A8 Arg325Trp variant is associated with reduced first-phase insulin release in 846 non-diabetic offspring of type 2 diabetes patients—the EUGENE2 study. Diabetologia 51: 816–820, 2008 [DOI] [PubMed] [Google Scholar]
- 17.Bonner-Weir S. and Orci L. New perspectives on the microvasculature of the islets of Langerhans in the rat. Diabetes 31: 883–889, 1982 [DOI] [PubMed] [Google Scholar]
- 18.Botros N, Concato J, Mohsenin V, Selim B, Doctor K, and Yaggi HK. Obstructive sleep apnea as a risk factor for type 2 diabetes. Am J Med 122: 1122–1127, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bottino R, Balamurugan AN, Bertera S, Pietropaolo M, Trucco M, and Piganelli JD. Preservation of human islet cell functional mass by anti-oxidative action of a novel SOD mimic compound. Diabetes 51: 2561–2567, 2002 [DOI] [PubMed] [Google Scholar]
- 20.Bottino R, Balamurugan AN, Tse H, Thirunavukkarasu C, Ge X, Profozich J, Milton M, Ziegenfuss A, Trucco M, and Piganelli JD. Response of human islets to isolation stress and the effect of antioxidant treatment. Diabetes 53: 2559–2568, 2004 [DOI] [PubMed] [Google Scholar]
- 21.Brand MD. and Esteves TC. Physiological functions of the mitochondrial uncoupling proteins UCP2 and UCP3. Cell Metab 2: 85–93, 2005 [DOI] [PubMed] [Google Scholar]
- 22.Bray TM. and Bettger WJ. The physiological role of zinc as an antioxidant. Free Radic Biol Med 8: 281–291, 1990 [DOI] [PubMed] [Google Scholar]
- 23.Brouwers B, de Faudeur G, Osipovich AB, Goyvaerts L, Lemaire K, Boesmans L, Cauwelier EJ, Granvik M, Pruniau VP, Van Lommel L, Van Schoors J, Stancill JS, Smolders I, Goffin V, Binart N, in't Veld P, Declercq J, Magnuson MA, Creemers JW, Schuit F, and Schraenen A. Impaired islet function in commonly used transgenic mouse lines due to human growth hormone minigene expression. Cell Metab 20: 979–990, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, and Butler PC. Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 52: 102–110, 2003 [DOI] [PubMed] [Google Scholar]
- 25.Cantley J, Selman C, Shukla D, Abramov AY, Forstreuter F, Esteban MA, Claret M, Lingard SJ, Clements M, Harten SK, Asare-Anane H, Batterham RL, Herrera PL, Persaud SJ, Duchen MR, Maxwell PH, and Withers DJ. Deletion of the von Hippel-Lindau gene in pancreatic beta cells impairs glucose homeostasis in mice. J Clin Invest 119: 125–135, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Carlsson C, Borg LA, and Welsh N. Sodium palmitate induces partial mitochondrial uncoupling and reactive oxygen species in rat pancreatic islets in vitro. Endocrinology 140: 3422–3428, 1999 [DOI] [PubMed] [Google Scholar]
- 27.Carocho M. and Ferreira IC. A review on antioxidants, prooxidants and related controversy: natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem Toxicol 51: 15–25, 2013 [DOI] [PubMed] [Google Scholar]
- 28.Chang I, Cho N, Koh JY, and Lee MS. Pyruvate inhibits zinc-mediated pancreatic islet cell death and diabetes. Diabetologia 46: 1220–1227, 2003 [DOI] [PubMed] [Google Scholar]
- 29.Cheng K, Ho K, Stokes R, Scott C, Lau SM, Hawthorne WJ, O'Connell PJ, Loudovaris T, Kay TW, Kulkarni RN, Okada T, Wang XL, Yim SH, Shah Y, Grey ST, Biankin AV, Kench JG, Laybutt DR, Gonzalez FJ, Kahn CR, and Gunton JE. Hypoxia-inducible factor-1alpha regulates beta cell function in mouse and human islets. J Clin Invest 120: 2171–2183, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chimienti F, Favier A, and Seve M. ZnT-8, a pancreatic beta-cell-specific zinc transporter. Biometals 18: 313–317, 2005 [DOI] [PubMed] [Google Scholar]
- 31.Cnop M, Igoillo-Esteve M, Rai M, Begu A, Serroukh Y, Depondt C, Musuaya AE, Marhfour I, Ladriere L, Moles Lopez X, Lefkaditis D, Moore F, Brion JP, Cooper JM, Schapira AH, Clark A, Koeppen AH, Marchetti P, Pandolfo M, Eizirik DL, and Fery F. Central role and mechanisms of beta-cell dysfunction and death in friedreich ataxia-associated diabetes. Ann Neurol 72: 971–982, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cnop M, Mulder H, and Igoillo-Esteve M. Diabetes in Friedreich ataxia. J Neurochem 126 Suppl 1: 94–102, 2013 [DOI] [PubMed] [Google Scholar]
- 33.Cornelius JG, Luttge BG, and Peck AB. Antioxidant enzyme activities in IDD-prone and IDD-resistant mice: a comparative study. Free Radic Biol Med 14: 409–420, 1993 [DOI] [PubMed] [Google Scholar]
- 34.Cousins RJ, Liuzzi JP, and Lichten LA. Mammalian zinc transport, trafficking, and signals. J Biol Chem 281: 24085–24089, 2006 [DOI] [PubMed] [Google Scholar]
- 35.Del Guerra S, Lupi R, Marselli L, Masini M, Bugliani M, Sbrana S, Torri S, Pollera M, Boggi U, Mosca F, Del Prato S, and Marchetti P. Functional and molecular defects of pancreatic islets in human type 2 diabetes. Diabetes 54: 727–735, 2005 [DOI] [PubMed] [Google Scholar]
- 36.Denton RM, Rutter GA, Midgley PJ, and McCormack JG. Effects of Ca2+ on the activities of the calcium-sensitive dehydrogenases within the mitochondria of mammalian tissues. J Cardiovasc Pharmacol 12 Suppl 5: S69–S72, 1988 [PubMed] [Google Scholar]
- 37.Di Paola M, Cocco T, and Lorusso M. Ceramide interaction with the respiratory chain of heart mitochondria. Biochemistry 39: 6660–6668, 2000 [DOI] [PubMed] [Google Scholar]
- 38.Diabetes Genetics Initiative of Broad Institute of H, Mit LU, Novartis Institutes of BioMedical R, Saxena R, Voight BF, Lyssenko V, Burtt NP, de Bakker PI, Chen H, Roix JJ, Kathiresan S, Hirschhorn JN, Daly MJ, Hughes TE, Groop L, Altshuler D, Almgren P, Florez JC, Meyer J, Ardlie K, Bengtsson Bostrom K, Isomaa B, Lettre G, Lindblad U, Lyon HN, Melander O, Newton-Cheh C, Nilsson P, Orho-Melander M, Rastam L, Speliotes EK, Taskinen MR, Tuomi T, Guiducci C, Berglund A, Carlson J, Gianniny L, Hackett R, Hall L, Holmkvist J, Laurila E, Sjogren M, Sterner M, Surti A, Svensson M, Svensson M, Tewhey R, Blumenstiel B, Parkin M, Defelice M, Barry R, Brodeur W, Camarata J, Chia N, Fava M, Gibbons J, Handsaker B, Healy C, Nguyen K, Gates C, Sougnez C, Gage D, Nizzari M, Gabriel SB, Chirn GW, Ma Q, Parikh H, Richardson D, Ricke D, and Purcell S. Genome-wide association analysis identifies loci for type 2 diabetes and triglyceride levels. Science 316: 1331–1336, 2007 [DOI] [PubMed] [Google Scholar]
- 39.Dube A, Harrisson JF, Saint-Gelais G, and Seguin C. Hypoxia acts through multiple signaling pathways to induce metallothionein transactivation by the metal-responsive transcription factor-1 (MTF-1). Biochem Cell Biol 89: 562–577, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dunn MF. Zinc-ligand interactions modulate assembly and stability of the insulin hexamer—a review. Biometals 18: 295–303, 2005 [DOI] [PubMed] [Google Scholar]
- 41.Eide DJ. Zinc transporters and the cellular trafficking of zinc. Biochim Biophys Acta 1763: 711–722, 2006 [DOI] [PubMed] [Google Scholar]
- 42.Eizirik DL. Beta-cell defence and repair mechanisms in human pancreatic islets. Horm Metab Res 28: 302–305, 1996 [DOI] [PubMed] [Google Scholar]
- 43.Eizirik DL, Pipeleers DG, Ling Z, Welsh N, Hellerstrom C, and Andersson A. Major species differences between humans and rodents in the susceptibility to pancreatic beta-cell injury. Proc Natl Acad Sci U S A 91: 9253–9256, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.El Khattabi I. and Sharma A. Preventing p38 MAPK-mediated MafA degradation ameliorates beta-cell dysfunction under oxidative stress. Mol Endocrinol 27: 1078–1090, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Elsner M, Gehrmann W, and Lenzen S. Peroxisome-generated hydrogen peroxide as important mediator of lipotoxicity in insulin-producing cells. Diabetes 60: 200–208, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fabbrini E, deHaseth D, Deivanayagam S, Mohammed BS, Vitola BE, and Klein S. Alterations in fatty acid kinetics in obese adolescents with increased intrahepatic triglyceride content. Obesity (Silver Spring) 17: 25–29, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fang Y, Zhang Q, Tan J, Li L, An X, and Lei P. Intermittent hypoxia-induced rat pancreatic beta-cell apoptosis and protective effects of antioxidant intervention. Nutr Diabetes 4: e131, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Finocchiaro G, Baio G, Micossi P, Pozza G, and di Donato S. Glucose metabolism alterations in Friedreich's ataxia. Neurology 38: 1292–1296, 1988 [DOI] [PubMed] [Google Scholar]
- 49.Formby B, Schmid-Formby F, and Grodsky GM. Relationship between insulin release and 65zinc efflux from rat pancreatic islets maintained in tissue culture. Diabetes 33: 229–234, 1984 [DOI] [PubMed] [Google Scholar]
- 50.Foster M, Chu A, Petocz P, and Samman S. Zinc transporter gene expression and glycemic control in post-menopausal women with Type 2 diabetes mellitus. J Trace Elem Med Biol 28: 448–452, 2014 [DOI] [PubMed] [Google Scholar]
- 51.Foster MC, Leapman RD, Li MX, and Atwater I. Elemental composition of secretory granules in pancreatic islets of Langerhans. Biophys J 64: 525–532, 1993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gannon M, Shiota C, Postic C, Wright CV, and Magnuson M. Analysis of the Cre-mediated recombination driven by rat insulin promoter in embryonic and adult mouse pancreas. Genesis 26: 139–142, 2000 [DOI] [PubMed] [Google Scholar]
- 53.Gehrmann W, Wurdemann W, Plotz T, Jorns A, Lenzen S, and Elsner M. Antagonism between saturated and unsaturated fatty acids in ROS mediated lipotoxicity in rat insulin-producing cells. Cell Physiol Biochem 36: 852–865, 2015 [DOI] [PubMed] [Google Scholar]
- 54.Gerber PA, Bellomo EA, Hodson DJ, Meur G, Solomou A, Mitchell RK, Hollinshead M, Chimienti F, Bosco D, Hughes SJ, Johnson PR, and Rutter GA. Hypoxia lowers SLC30A8/ZnT8 expression and free cytosolic Zn2+ in pancreatic beta cells. Diabetologia 57: 1635–1644, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Giuliani M, Moritz W, Bodmer E, Dindo D, Kugelmeier P, Lehmann R, Gassmann M, Groscurth P, and Weber M. Central necrosis in isolated hypoxic human pancreatic islets: evidence for postisolation ischemia. Cell Transplant 14: 67–76, 2005 [DOI] [PubMed] [Google Scholar]
- 56.Gold G. and Grodsky GM. Kinetic aspects of compartmental storage and secretion of insulin and zinc. Experientia 40: 1105–1114, 1984 [DOI] [PubMed] [Google Scholar]
- 57.Gonzalez M, Rojas S, Avila P, Cabrera L, Villalobos R, Palma C, Aguayo C, Pena E, Gallardo V, Guzman-Gutierrez E, Saez T, Salsoso R, Sanhueza C, Pardo F, Leiva A, and Sobrevia L. Insulin reverses D-glucose-increased nitric oxide and reactive oxygen species generation in human umbilical vein endothelial cells. PLoS One 10: e0122398, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Grarup N, Sandholt CH, Hansen T, and Pedersen O. Genetic susceptibility to type 2 diabetes and obesity: from genome-wide association studies to rare variants and beyond. Diabetologia 57: 1528–1541, 2014 [DOI] [PubMed] [Google Scholar]
- 59.Gray JP. and Heart E. Usurping the mitochondrial supremacy: extramitochondrial sources of reactive oxygen intermediates and their role in beta cell metabolism and insulin secretion. Toxicol Mech Methods 20: 167–174, 2010 [DOI] [PubMed] [Google Scholar]
- 60.Greijer AE. and van der Wall E. The role of hypoxia inducible factor 1 (HIF-1) in hypoxia induced apoptosis. J Clin Pathol 57: 1009–1014, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Gordon DJ, Krauss RM, Savage PJ, Smith SC, Jr., Spertus JA, Costa F, American Heart A, National Heart L, and Blood I. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 112: 2735–2752, 2005 [DOI] [PubMed] [Google Scholar]
- 62.Gudz TI, Tserng KY, and Hoppel CL. Direct inhibition of mitochondrial respiratory chain complex III by cell-permeable ceramide. J Biol Chem 272: 24154–24158, 1997 [DOI] [PubMed] [Google Scholar]
- 63.Gunasekara P, Hettiarachchi M, Liyanage C, and Lekamwasam S. Effects of zinc and multimineral vitamin supplementation on glycemic and lipid control in adult diabetes. Diabetes Metab Syndr Obes 4: 53–60, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Gunton JE, Kulkarni RN, Yim S, Okada T, Hawthorne WJ, Tseng YH, Roberson RS, Ricordi C, O'Connell PJ, Gonzalez FJ, and Kahn CR. Loss of ARNT/HIF1beta mediates altered gene expression and pancreatic-islet dysfunction in human type 2 diabetes. Cell 122: 337–349, 2005 [DOI] [PubMed] [Google Scholar]
- 65.Harmon JS, Bogdani M, Parazzoli SD, Mak SS, Oseid EA, Berghmans M, Leboeuf RC, and Robertson RP. beta-Cell-specific overexpression of glutathione peroxidase preserves intranuclear MafA and reverses diabetes in db/db mice. Endocrinology 150: 4855–4862, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Harmon JS, Stein R, and Robertson RP. Oxidative stress-mediated, post-translational loss of MafA protein as a contributing mechanism to loss of insulin gene expression in glucotoxic beta cells. J Biol Chem 280: 11107–11113, 2005 [DOI] [PubMed] [Google Scholar]
- 67.Henquin JC. Triggering and amplifying pathways of regulation of insulin secretion by glucose. Diabetes 49: 1751–1760, 2000 [DOI] [PubMed] [Google Scholar]
- 68.Hessels AM, Chabosseau P, Bakker MH, Engelen W, Rutter GA, Taylor KM, and Merkx M. eZinCh-2: A Versatile, Genetically Encoded FRET Sensor for Cytosolic and Intraorganelle Zn(2+) Imaging. ACS Chem Biol 10: 2126–2134, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hill CP, Dauter Z, Dodson EJ, Dodson GG, and Dunn MF. X-ray structure of an unusual Ca2+ site and the roles of Zn2+ and Ca2+ in the assembly, stability, and storage of the insulin hexamer. Biochemistry 30: 917–924, 1991 [DOI] [PubMed] [Google Scholar]
- 70.Hou N, Torii S, Saito N, Hosaka M, and Takeuchi T. Reactive oxygen species-mediated pancreatic beta-cell death is regulated by interactions between stress-activated protein kinases, p38 and c-Jun N-terminal kinase, and mitogen-activated protein kinase phosphatases. Endocrinology 149: 1654–1665, 2008 [DOI] [PubMed] [Google Scholar]
- 71.Hutton JC, Penn EJ, and Peshavaria M. Low-molecular-weight constituents of isolated insulin-secretory granules. Bivalent cations, adenine nucleotides and inorganic phosphate. Biochem J 210: 297–305, 1983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Inoguchi T, Li P, Umeda F, Yu HY, Kakimoto M, Imamura M, Aoki T, Etoh T, Hashimoto T, Naruse M, Sano H, Utsumi H, and Nawata H. High glucose level and free fatty acid stimulate reactive oxygen species production through protein kinase C—dependent activation of NAD(P)H oxidase in cultured vascular cells. Diabetes 49: 1939–1945, 2000 [DOI] [PubMed] [Google Scholar]
- 73.Ishihara H, Wang H, Drewes LR, and Wollheim CB. Overexpression of monocarboxylate transporter and lactate dehydrogenase alters insulin secretory responses to pyruvate and lactate in beta cells. J Clin Invest 104: 1621–1629, 1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ivarsson R, Quintens R, Dejonghe S, Tsukamoto K, in 't Veld P, Renstrom E, and Schuit FC. Redox control of exocytosis: regulatory role of NADPH, thioredoxin, and glutaredoxin. Diabetes 54: 2132–2142, 2005 [DOI] [PubMed] [Google Scholar]
- 75.Jacob S, Machann J, Rett K, Brechtel K, Volk A, Renn W, Maerker E, Matthaei S, Schick F, Claussen CD, and Haring HU. Association of increased intramyocellular lipid content with insulin resistance in lean nondiabetic offspring of type 2 diabetic subjects. Diabetes 48: 1113–1119, 1999 [DOI] [PubMed] [Google Scholar]
- 76.Jansson L. The regulation of pancreatic islet blood flow. Diabetes Metab Rev 10: 407–416, 1994 [DOI] [PubMed] [Google Scholar]
- 77.Jansson L. and Carlsson PO. Graft vascular function after transplantation of pancreatic islets. Diabetologia 45: 749–763, 2002 [DOI] [PubMed] [Google Scholar]
- 78.Jensen MD, Haymond MW, Rizza RA, Cryer PE, and Miles JM. Influence of body fat distribution on free fatty acid metabolism in obesity. J Clin Invest 83: 1168–1173, 1989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Jiang BH, Rue E, Wang GL, Roe R, and Semenza GL. Dimerization, DNA binding, and transactivation properties of hypoxia-inducible factor 1. J Biol Chem 271: 17771–17778, 1996 [DOI] [PubMed] [Google Scholar]
- 80.Kahn SE, Cooper ME, and Del Prato S. Pathophysiology and treatment of type 2 diabetes: perspectives on the past, present, and future. Lancet 383: 1068–1083, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Kambe T, Hashimoto A, and Fujimoto S. Current understanding of ZIP and ZnT zinc transporters in human health and diseases. Cell Mol Life Sci 71: 3281–3295, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Kaneto H, Kajimoto Y, Miyagawa J, Matsuoka T, Fujitani Y, Umayahara Y, Hanafusa T, Matsuzawa Y, Yamasaki Y, and Hori M. Beneficial effects of antioxidants in diabetes: possible protection of pancreatic beta-cells against glucose toxicity. Diabetes 48: 2398–2406, 1999 [DOI] [PubMed] [Google Scholar]
- 83.Kaneto H, Xu G, Fujii N, Kim S, Bonner-Weir S, and Weir GC. Involvement of c-Jun N-terminal kinase in oxidative stress-mediated suppression of insulin gene expression. J Biol Chem 277: 30010–30018, 2002 [DOI] [PubMed] [Google Scholar]
- 84.Kanoni S, Nettleton JA, Hivert MF, Ye Z, van Rooij FJ, Shungin D, Sonestedt E, Ngwa JS, Wojczynski MK, Lemaitre RN, Gustafsson S, Anderson JS, Tanaka T, Hindy G, Saylor G, Renstrom F, Bennett AJ, van Duijn CM, Florez JC, Fox CS, Hofman A, Hoogeveen RC, Houston DK, Hu FB, Jacques PF, Johansson I, Lind L, Liu Y, McKeown N, Ordovas J, Pankow JS, Sijbrands EJ, Syvanen AC, Uitterlinden AG, Yannakoulia M, Zillikens MC, Investigators M, Wareham NJ, Prokopenko I, Bandinelli S, Forouhi NG, Cupples LA, Loos RJ, Hallmans G, Dupuis J, Langenberg C, Ferrucci L, Kritchevsky SB, McCarthy MI, Ingelsson E, Borecki IB, Witteman JC, Orho-Melander M, Siscovick DS, Meigs JB, Franks PW, and Dedoussis GV. Total zinc intake may modify the glucose-raising effect of a zinc transporter (SLC30A8) variant: a 14-cohort meta-analysis. Diabetes 60: 2407–2416, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Karpe F, Dickmann JR, and Frayn KN. Fatty acids, obesity, and insulin resistance: time for a reevaluation. Diabetes 60: 2441–2449, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kawamori D, Kajimoto Y, Kaneto H, Umayahara Y, Fujitani Y, Miyatsuka T, Watada H, Leibiger IB, Yamasaki Y, and Hori M. Oxidative stress induces nucleo-cytoplasmic translocation of pancreatic transcription factor PDX-1 through activation of c-Jun NH(2)-terminal kinase. Diabetes 52: 2896–2904, 2003 [DOI] [PubMed] [Google Scholar]
- 87.Kawamori D, Kaneto H, Nakatani Y, Matsuoka TA, Matsuhisa M, Hori M, and Yamasaki Y. The forkhead transcription factor Foxo1 bridges the JNK pathway and the transcription factor PDX-1 through its intracellular translocation. J Biol Chem 281: 1091–1098, 2006 [DOI] [PubMed] [Google Scholar]
- 88.Keane KN, Cruzat VF, Carlessi R, de Bittencourt PI, Jr., and Newsholme P. Molecular events linking oxidative stress and inflammation to insulin resistance and beta-cell dysfunction. Oxid Med Cell Longev 2015: 181643, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kim BJ, Kim YH, Kim S, Kim JW, Koh JY, Oh SH, Lee MK, Kim KW, and Lee MS. Zinc as a paracrine effector in pancreatic islet cell death. Diabetes 49: 367–372, 2000 [DOI] [PubMed] [Google Scholar]
- 90.Kim HG, Hwang YP, Han EH, Choi CY, Yeo CY, Kim JY, Lee KY, and Jeong HG. Metallothionein-III provides neuronal protection through activation of nuclear factor-kappaB via the TrkA/phosphatidylinositol-3 kinase/Akt signaling pathway. Toxicol Sci 112: 435–449, 2009 [DOI] [PubMed] [Google Scholar]
- 91.Kim JW, Tchernyshyov I, Semenza GL, and Dang CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab 3: 177–185, 2006 [DOI] [PubMed] [Google Scholar]
- 92.Kirchhoff K, Machicao F, Haupt A, Schafer SA, Tschritter O, Staiger H, Stefan N, Haring HU, and Fritsche A. Polymorphisms in the TCF7L2, CDKAL1 and SLC30A8 genes are associated with impaired proinsulin conversion. Diabetologia 51: 597–601, 2008 [DOI] [PubMed] [Google Scholar]
- 93.Kojima I, Tanaka T, Inagi R, Nishi H, Aburatani H, Kato H, Miyata T, Fujita T, and Nangaku M. Metallothionein is upregulated by hypoxia and stabilizes hypoxia-inducible factor in the kidney. Kidney Int 75: 268–277, 2009 [DOI] [PubMed] [Google Scholar]
- 94.Kondo T, El Khattabi I, Nishimura W, Laybutt DR, Geraldes P, Shah S, King G, Bonner-Weir S, Weir G, and Sharma A. p38 MAPK is a major regulator of MafA protein stability under oxidative stress. Mol Endocrinol 23: 1281–1290, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Koshkin V, Wang X, Scherer PE, Chan CB, and Wheeler MB. Mitochondrial functional state in clonal pancreatic beta-cells exposed to free fatty acids. J Biol Chem 278: 19709–19715, 2003 [DOI] [PubMed] [Google Scholar]
- 96.Krssak M, Falk Petersen K, Dresner A, DiPietro L, Vogel SM, Rothman DL, Roden M, and Shulman GI. Intramyocellular lipid concentrations are correlated with insulin sensitivity in humans: a 1H NMR spectroscopy study. Diabetologia 42: 113–116, 1999 [DOI] [PubMed] [Google Scholar]
- 97.Lau J, Henriksnas J, Svensson J, and Carlsson PO. Oxygenation of islets and its role in transplantation. Curr Opin Organ Transplant 14: 688–693, 2009 [DOI] [PubMed] [Google Scholar]
- 98.Lee SC, Robson-Doucette CA, and Wheeler MB. Uncoupling protein 2 regulates reactive oxygen species formation in islets and influences susceptibility to diabetogenic action of streptozotocin. J Endocrinol 203: 33–43, 2009 [DOI] [PubMed] [Google Scholar]
- 99.Lehmann R, Graziano J, Brockmann J, Pfammatter T, Kron P, de Rougemont O, Mueller T, Zuellig RA, Spinas GA, and Gerber PA. Glycemic control in simultaneous islet-kidney versus pancreas-kidney transplantation in type 1 diabetes: a prospective 13-year follow-up. Diabetes Care 38: 752–759, 2015 [DOI] [PubMed] [Google Scholar]
- 100.Leloup C, Tourrel-Cuzin C, Magnan C, Karaca M, Castel J, Carneiro L, Colombani AL, Ktorza A, Casteilla L, and Penicaud L. Mitochondrial reactive oxygen species are obligatory signals for glucose-induced insulin secretion. Diabetes 58: 673–681, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lenaz G. The mitochondrial production of reactive oxygen species: mechanisms and implications in human pathology. IUBMB Life 52: 159–164, 2001 [DOI] [PubMed] [Google Scholar]
- 102.Lenzen S. A fresh view of glycolysis and glucokinase regulation: history and current status. J Biol Chem 289: 12189–12194, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lenzen S, Drinkgern J, and Tiedge M. Low antioxidant enzyme gene expression in pancreatic islets compared with various other mouse tissues. Free Radic Biol Med 20: 463–466, 1996 [DOI] [PubMed] [Google Scholar]
- 104.Li N, Frigerio F, and Maechler P. The sensitivity of pancreatic beta-cells to mitochondrial injuries triggered by lipotoxicity and oxidative stress. Biochem Soc Trans 36: 930–934, 2008 [DOI] [PubMed] [Google Scholar]
- 105.Li X, Chen H, and Epstein PN. Metallothionein protects islets from hypoxia and extends islet graft survival by scavenging most kinds of reactive oxygen species. J Biol Chem 279: 765–771, 2004 [DOI] [PubMed] [Google Scholar]
- 106.Li X, Zhang L, Meshinchi S, Dias-Leme C, Raffin D, Johnson JD, Treutelaar MK, and Burant CF. Islet microvasculature in islet hyperplasia and failure in a model of type 2 diabetes. Diabetes 55: 2965–2973, 2006 [DOI] [PubMed] [Google Scholar]
- 107.Lifson N, Lassa CV, and Dixit PK. Relation between blood flow and morphology in islet organ of rat pancreas. Am J Physiol 249: E43–E48, 1985 [DOI] [PubMed] [Google Scholar]
- 108.Liu Z, Huang YY, Wang YX, Wang HG, Deng F, Heng B, Xie LH, and Liu YQ. Prevention of cell death by the zinc ion chelating agent TPEN in cultured PC12 cells exposed to Oxygen-Glucose Deprivation (OGD). J Trace Elem Med Biol 31: 45–52, 2015 [DOI] [PubMed] [Google Scholar]
- 109.Llanos P, Contreras-Ferrat A, Barrientos G, Valencia M, Mears D, and Hidalgo C. Glucose-dependent insulin secretion in pancreatic beta-cell islets from male rats requires Ca2+ release via ROS-stimulated ryanodine receptors. PLoS One 10: e0129238, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Maechler P, Jornot L, and Wollheim CB. Hydrogen peroxide alters mitochondrial activation and insulin secretion in pancreatic beta cells. J Biol Chem 274: 27905–27913, 1999 [DOI] [PubMed] [Google Scholar]
- 111.Marmolino D. Friedreich's ataxia: past, present and future. Brain Res Rev 67: 311–330, 2011 [DOI] [PubMed] [Google Scholar]
- 112.Marselli L, Suleiman M, Masini M, Campani D, Bugliani M, Syed F, Martino L, Focosi D, Scatena F, Olimpico F, Filipponi F, Masiello P, Boggi U, and Marchetti P. Are we overestimating the loss of beta cells in type 2 diabetes? Diabetologia 57: 362–365, 2014 [DOI] [PubMed] [Google Scholar]
- 113.Martens GA, Cai Y, Hinke S, Stange G, Van de Casteele M, and Pipeleers D. Glucose suppresses superoxide generation in metabolically responsive pancreatic beta cells. J Biol Chem 280: 20389–20396, 2005 [DOI] [PubMed] [Google Scholar]
- 114.Matsuoka T, Kajimoto Y, Watada H, Kaneto H, Kishimoto M, Umayahara Y, Fujitani Y, Kamada T, Kawamori R, and Yamasaki Y. Glycation-dependent, reactive oxygen species-mediated suppression of the insulin gene promoter activity in HIT cells. J Clin Invest 99: 144–150, 1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Matsuoka TA, Artner I, Henderson E, Means A, Sander M, and Stein R. The MafA transcription factor appears to be responsible for tissue-specific expression of insulin. Proc Natl Acad Sci U S A 101: 2930–2933, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Matsuoka TA, Kaneto H, Kawashima S, Miyatsuka T, Tochino Y, Yoshikawa A, Imagawa A, Miyazaki J, Gannon M, Stein R, and Shimomura I. Preserving Mafa expression in diabetic islet beta-cells improves glycemic control in vivo. J Biol Chem 290: 7647–7657, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Maxwell PH, Wiesener MS, Chang GW, Clifford SC, Vaux EC, Cockman ME, Wykoff CC, Pugh CW, Maher ER, and Ratcliffe PJ. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399: 271–275, 1999 [DOI] [PubMed] [Google Scholar]
- 118.McCulloch LJ, van de Bunt M, Braun M, Frayn KN, Clark A, and Gloyn AL. GLUT2 (SLC2A2) is not the principal glucose transporter in human pancreatic beta cells: implications for understanding genetic association signals at this locus. Mol Genet Metab 104: 648–653, 2011 [DOI] [PubMed] [Google Scholar]
- 119.McGarry JD. Banting lecture 2001: dysregulation of fatty acid metabolism in the etiology of type 2 diabetes. Diabetes 51: 7–18, 2002 [DOI] [PubMed] [Google Scholar]
- 120.Medvedeva YV, Lin B, Shuttleworth CW, and Weiss JH. Intracellular Zn2+ accumulation contributes to synaptic failure, mitochondrial depolarization, and cell death in an acute slice oxygen-glucose deprivation model of ischemia. J Neurosci 29: 1105–1114, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Milthorpe BK, Nichol LW, and Jeffrey PD. The polymerization pattern of zinc(II)-insulin at pH 7.0. Biochim Biophys Acta 495: 195–202, 1977 [DOI] [PubMed] [Google Scholar]
- 122.Mitchell RK, Hu M, Chabosseau PL, Cane MC, Meur G, Bellomo EA, Carzaniga R, Collinson LM, Li WH, Hodson DJ, and Rutter GA. Molecular genetic regulation of Slc30a8/ZnT8 reveals a positive association with glucose tolerance. Mol Endocrinol 77–91, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Morgan D, Oliveira-Emilio HR, Keane D, Hirata AE, Santos da Rocha M, Bordin S, Curi R, Newsholme P, and Carpinelli AR. Glucose, palmitate and pro-inflammatory cytokines modulate production and activity of a phagocyte-like NADPH oxidase in rat pancreatic islets and a clonal beta cell line. Diabetologia 50: 359–369, 2007 [DOI] [PubMed] [Google Scholar]
- 124.Moritz W, Meier F, Stroka DM, Giuliani M, Kugelmeier P, Nett PC, Lehmann R, Candinas D, Gassmann M, and Weber M. Apoptosis in hypoxic human pancreatic islets correlates with HIF-1alpha expression. FASEB J 16: 745–747, 2002 [DOI] [PubMed] [Google Scholar]
- 125.Muhlenhoff U, Richhardt N, Ristow M, Kispal G, and Lill R. The yeast frataxin homolog Yfh1p plays a specific role in the maturation of cellular Fe/S proteins. Hum Mol Genet 11: 2025–2036, 2002 [DOI] [PubMed] [Google Scholar]
- 126.Murphy BJ, Kimura T, Sato BG, Shi Y, and Andrews GK. Metallothionein induction by hypoxia involves cooperative interactions between metal-responsive transcription factor-1 and hypoxia-inducible transcription factor-1alpha. Mol Cancer Res 6: 483–490, 2008 [DOI] [PubMed] [Google Scholar]
- 127.Newsholme P, Haber EP, Hirabara SM, Rebelato EL, Procopio J, Morgan D, Oliveira-Emilio HC, Carpinelli AR, and Curi R. Diabetes associated cell stress and dysfunction: role of mitochondrial and non-mitochondrial ROS production and activity. J Physiol 583: 9–24, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Newsholme P, Rebelato E, Abdulkader F, Krause M, Carpinelli A, and Curi R. Reactive oxygen and nitrogen species generation, antioxidant defenses, and beta-cell function: a critical role for amino acids. J Endocrinol 214: 11–20, 2012 [DOI] [PubMed] [Google Scholar]
- 129.Nicolson TJ, Bellomo EA, Wijesekara N, Loder MK, Baldwin JM, Gyulkhandanyan AV, Koshkin V, Tarasov AI, Carzaniga R, Kronenberger K, Taneja TK, da Silva Xavier G, Libert S, Froguel P, Scharfmann R, Stetsyuk V, Ravassard P, Parker H, Gribble FM, Reimann F, Sladek R, Hughes SJ, Johnson PR, Masseboeuf M, Burcelin R, Baldwin SA, Liu M, Lara-Lemus R, Arvan P, Schuit FC, Wheeler MB, Chimienti F, and Rutter GA. Insulin storage and glucose homeostasis in mice null for the granule zinc transporter ZnT8 and studies of the type 2 diabetes-associated variants. Diabetes 58: 2070–2083, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Ohlsson H, Karlsson K, and Edlund T. IPF1, a homeodomain-containing transactivator of the insulin gene. EMBO J 12: 4251–4259, 1993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Olsson R. and Carlsson PO. Oxygenation of cultured pancreatic islets. Adv Exp Med Biol 578: 263–268, 2006 [DOI] [PubMed] [Google Scholar]
- 132.Olsson R. and Carlsson PO. A low-oxygenated subpopulation of pancreatic islets constitutes a functional reserve of endocrine cells. Diabetes 60: 2068–2075, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Olsson R, Olerud J, Pettersson U, and Carlsson PO. Increased numbers of low-oxygenated pancreatic islets after intraportal islet transplantation. Diabetes 60: 2350–2353, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Outten CE. and O'Halloran TV. Femtomolar sensitivity of metalloregulatory proteins controlling zinc homeostasis. Science 292: 2488–2492, 2001 [DOI] [PubMed] [Google Scholar]
- 135.Pacher P, Beckman JS, and Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol Rev 87: 315–424, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Padmasekar M, Lingwal N, Samikannu B, Chen C, Sauer H, and Linn T. Exendin-4 protects hypoxic islets from oxidative stress and improves islet transplantation outcome. Endocrinology 154: 1424–1433, 2013 [DOI] [PubMed] [Google Scholar]
- 137.Pan DA, Lillioja S, Kriketos AD, Milner MR, Baur LA, Bogardus C, Jenkins AB, and Storlien LH. Skeletal muscle triglyceride levels are inversely related to insulin action. Diabetes 46: 983–988, 1997 [DOI] [PubMed] [Google Scholar]
- 138.Panten U. and Klein H. O2 consumption by isolated pancreatic islets, as measured in a microincubation system with a Clark-type electrode. Endocrinology 111: 1595–1600, 1982 [DOI] [PubMed] [Google Scholar]
- 139.Papandreou I, Cairns RA, Fontana L, Lim AL, and Denko NC. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab 3: 187–197, 2006 [DOI] [PubMed] [Google Scholar]
- 140.Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, and Hla KM. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol 177: 1006–1014, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Perry DK, Smyth MJ, Stennicke HR, Salvesen GS, Duriez P, Poirier GG, and Hannun YA. Zinc is a potent inhibitor of the apoptotic protease, caspase-3. A novel target for zinc in the inhibition of apoptosis. J Biol Chem 272: 18530–18533, 1997 [DOI] [PubMed] [Google Scholar]
- 142.Pi J, Bai Y, Daniel KW, Liu D, Lyght O, Edelstein D, Brownlee M, Corkey BE, and Collins S. Persistent oxidative stress due to absence of uncoupling protein 2 associated with impaired pancreatic beta-cell function. Endocrinology 150: 3040–3048, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Pi J. and Collins S. Reactive oxygen species and uncoupling protein 2 in pancreatic beta-cell function. Diabetes Obes Metab 12 Suppl 2: 141–148, 2010 [DOI] [PubMed] [Google Scholar]
- 144.Pileggi A, Molano RD, Berney T, Cattan P, Vizzardelli C, Oliver R, Fraker C, Ricordi C, Pastori RL, Bach FH, and Inverardi L. Heme oxygenase-1 induction in islet cells results in protection from apoptosis and improved in vivo function after transplantation. Diabetes 50: 1983–1991, 2001 [DOI] [PubMed] [Google Scholar]
- 145.Pong K, Rong Y, Doctrow SR, and Baudry M. Attenuation of zinc-induced intracellular dysfunction and neurotoxicity by a synthetic superoxide dismutase/catalase mimetic, in cultured cortical neurons. Brain Res 950: 218–230, 2002 [DOI] [PubMed] [Google Scholar]
- 146.Porter AG. and Janicke RU. Emerging roles of caspase-3 in apoptosis. Cell Death Differ 6: 99–104, 1999 [DOI] [PubMed] [Google Scholar]
- 147.Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med 14: 353–357, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Priou P, Le Vaillant M, Meslier N, Chollet S, Masson P, Humeau MP, Pigeanne T, Bizieux-Thaminy A, Goupil F, Gagnadoux F, and Group ISC. Independent association between obstructive sleep apnea severity and glycated hemoglobin in adults without diabetes. Diabetes Care 35: 1902–1906, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Produit-Zengaffinen N, Davis-Lameloise N, Perreten H, Becard D, Gjinovci A, Keller PA, Wollheim CB, Herrera P, Muzzin P, and Assimacopoulos-Jeannet F. Increasing uncoupling protein-2 in pancreatic beta cells does not alter glucose-induced insulin secretion but decreases production of reactive oxygen species. Diabetologia 50: 84–93, 2007 [DOI] [PubMed] [Google Scholar]
- 150.Proteggente AR, England TG, Rehman A, Rice-Evans CA, and Halliwell B. Gender differences in steady-state levels of oxidative damage to DNA in healthy individuals. Free Radic Res 36: 157–162, 2002 [DOI] [PubMed] [Google Scholar]
- 151.Pullen TJ, Khan AM, Barton G, Butcher SA, Sun G, and Rutter GA. Identification of genes selectively disallowed in the pancreatic islet. Islets 2: 89–95, 2010 [DOI] [PubMed] [Google Scholar]
- 152.Pullen TJ. and Rutter GA. When less is more: the forbidden fruits of gene repression in the adult beta-cell. Diabetes Obes Metab 15: 503–512, 2013 [DOI] [PubMed] [Google Scholar]
- 153.Puri S, Cano DA, and Hebrok M. A role for von Hippel-Lindau protein in pancreatic beta-cell function. Diabetes 58: 433–441, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Rashidi A, Kirkwood TB, and Shanley DP. Metabolic evolution suggests an explanation for the weakness of antioxidant defences in beta-cells. Mech Ageing Dev 130: 216–221, 2009 [DOI] [PubMed] [Google Scholar]
- 155.Rharass T, Lemcke H, Lantow M, Kuznetsov SA, Weiss DG, and Panakova D. Ca2+-mediated mitochondrial reactive oxygen species metabolism augments Wnt/beta-catenin pathway activation to facilitate cell differentiation. J Biol Chem 289: 27937–27951, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Ristow M, Mulder H, Pomplun D, Schulz TJ, Muller-Schmehl K, Krause A, Fex M, Puccio H, Muller J, Isken F, Spranger J, Muller-Wieland D, Magnuson MA, Mohlig M, Koenig M, and Pfeiffer AF. Frataxin deficiency in pancreatic islets causes diabetes due to loss of beta cell mass. J Clin Invest 112: 527–534, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Rizzo MA. and Piston DW. Regulation of beta cell glucokinase by S-nitrosylation and association with nitric oxide synthase. J Cell Biol 161: 243–248, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Robertson RP. Oxidative stress and impaired insulin secretion in type 2 diabetes. Curr Opin Pharmacol 6: 615–619, 2006 [DOI] [PubMed] [Google Scholar]
- 159.Robertson RP. and Harmon JS. Pancreatic islet beta-cell and oxidative stress: the importance of glutathione peroxidase. FEBS Lett 581: 3743–3748, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Robson-Doucette CA, Sultan S, Allister EM, Wikstrom JD, Koshkin V, Bhattacharjee A, Prentice KJ, Sereda SB, Shirihai OS, and Wheeler MB. Beta-cell uncoupling protein 2 regulates reactive oxygen species production, which influences both insulin and glucagon secretion. Diabetes 60: 2710–2719, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Roma LP, Duprez J, Takahashi HK, Gilon P, Wiederkehr A, and Jonas JC. Dynamic measurements of mitochondrial hydrogen peroxide concentration and glutathione redox state in rat pancreatic beta-cells using ratiometric fluorescent proteins: confounding effects of pH with HyPer but not roGFP1. Biochem J 441: 971–978, 2012 [DOI] [PubMed] [Google Scholar]
- 162.Ronksley PE, Hemmelgarn BR, Heitman SJ, Hanly PJ, Faris PD, Quan H, and Tsai WH. Obstructive sleep apnoea is associated with diabetes in sleepy subjects. Thorax 64: 834–839, 2009 [DOI] [PubMed] [Google Scholar]
- 163.Rutter GA. Visualising insulin secretion. The Minkowski Lecture 2004. Diabetologia 47: 1861–1872, 2004 [DOI] [PubMed] [Google Scholar]
- 164.Rutter GA. Dorothy Hodgkin Lecture 2014. Understanding genes identified by genome-wide association studies for type 2 diabetes. Diabet Med 31: 1480–1487, 2014 [DOI] [PubMed] [Google Scholar]
- 165.Rutter GA, Chabosseau P, Bellomo EA, Maret W, Mitchell RK, Hodson DJ, Solomou A, and Hu M. Intracellular zinc in insulin secretion and action: a determinant of diabetes risk? Proc Nutr Soc 61–72, 2016 [DOI] [PubMed] [Google Scholar]
- 166.Rutter GA. and Chimienti F. SLC30A8 mutations in type 2 diabetes. Diabetologia 58: 31–36, 2015 [DOI] [PubMed] [Google Scholar]
- 167.Rutter GA, Pralong WF, and Wollheim CB. Regulation of mitochondrial glycerol-phosphate dehydrogenase by Ca2+ within electropermeabilized insulin-secreting cells (INS-1). Biochim Biophys Acta 1175: 107–113, 1992 [DOI] [PubMed] [Google Scholar]
- 168.Rutter GA, Pullen TJ, Hodson DJ, and Martinez-Sanchez A. Pancreatic beta-cell identity, glucose sensing and the control of insulin secretion. Biochem J 466: 203–218, 2015 [DOI] [PubMed] [Google Scholar]
- 169.Ryu R, Shin Y, Choi JW, Min W, Ryu H, Choi CR, and Ko H. Depletion of intracellular glutathione mediates zinc-induced cell death in rat primary astrocytes. Exp Brain Res 143: 257–263, 2002 [DOI] [PubMed] [Google Scholar]
- 170.Sampson SR, Bucris E, Horovitz-Fried M, Parnas A, Kahana S, Abitbol G, Chetboun M, Rosenzweig T, Brodie C, and Frankel S. Insulin increases H2O2-induced pancreatic beta cell death. Apoptosis 15: 1165–1176, 2010 [DOI] [PubMed] [Google Scholar]
- 171.Sarre A, Gabrielli J, Vial G, Leverve XM, and Assimacopoulos-Jeannet F. Reactive oxygen species are produced at low glucose and contribute to the activation of AMPK in insulin-secreting cells. Free Radic Biol Med 52: 142–150, 2012 [DOI] [PubMed] [Google Scholar]
- 172.Sato Y, Endo H, Okuyama H, Takeda T, Iwahashi H, Imagawa A, Yamagata K, Shimomura I, and Inoue M. Cellular hypoxia of pancreatic beta-cells due to high levels of oxygen consumption for insulin secretion in vitro. J Biol Chem 286: 12524–12532, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Schonfeld P. and Wojtczak L. Fatty acids decrease mitochondrial generation of reactive oxygen species at the reverse electron transport but increase it at the forward transport. Biochim Biophys Acta 1767: 1032–1040, 2007 [DOI] [PubMed] [Google Scholar]
- 174.Schuit F, De Vos A, Farfari S, Moens K, Pipeleers D, Brun T, and Prentki M. Metabolic fate of glucose in purified islet cells. Glucose-regulated anaplerosis in beta cells. J Biol Chem 272: 18572–18579, 1997 [DOI] [PubMed] [Google Scholar]
- 175.Scott DA. and Fisher AM. The Insulin and the Zinc Content of Normal and Diabetic Pancreas. J Clin Invest 17: 725–728, 1938 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Sekine N, Cirulli V, Regazzi R, Brown LJ, Gine E, Tamarit-Rodriguez J, Girotti M, Marie S, MacDonald MJ, Wollheim CB, et al. . Low lactate dehydrogenase and high mitochondrial glycerol phosphate dehydrogenase in pancreatic beta-cells. Potential role in nutrient sensing. J Biol Chem 269: 4895–4902, 1994 [PubMed] [Google Scholar]
- 177.Semenza GL. Hydroxylation of HIF-1: oxygen sensing at the molecular level. Physiology (Bethesda) 19: 176–182, 2004 [DOI] [PubMed] [Google Scholar]
- 178.Semenza GL, Jiang BH, Leung SW, Passantino R, Concordet JP, Maire P, and Giallongo A. Hypoxia response elements in the aldolase A, enolase 1, and lactate dehydrogenase A gene promoters contain essential binding sites for hypoxia-inducible factor 1. J Biol Chem 271: 32529–32537, 1996 [DOI] [PubMed] [Google Scholar]
- 179.Sensi SL, Ton-That D, Sullivan PG, Jonas EA, Gee KR, Kaczmarek LK, and Weiss JH. Modulation of mitochondrial function by endogenous Zn2+ pools. Proc Natl Acad Sci U S A 100: 6157–6162, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Shapiro AM, Lakey JR, Ryan EA, Korbutt GS, Toth E, Warnock GL, Kneteman NM, and Rajotte RV. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med 343: 230–238, 2000 [DOI] [PubMed] [Google Scholar]
- 181.Shapiro AM, Ricordi C, Hering BJ, Auchincloss H, Lindblad R, Robertson RP, Secchi A, Brendel MD, Berney T, Brennan DC, Cagliero E, Alejandro R, Ryan EA, DiMercurio B, Morel P, Polonsky KS, Reems JA, Bretzel RG, Bertuzzi F, Froud T, Kandaswamy R, Sutherland DE, Eisenbarth G, Segal M, Preiksaitis J, Korbutt GS, Barton FB, Viviano L, Seyfert-Margolis V, Bluestone J, and Lakey JR. International trial of the Edmonton protocol for islet transplantation. N Engl J Med 355: 1318–1330, 2006 [DOI] [PubMed] [Google Scholar]
- 182.Sherwani SI, Aldana C, Usmani S, Adin C, Kotha S, Khan M, Eubank T, Scherer PE, Parinandi N, and Magalang UJ. Intermittent hypoxia exacerbates pancreatic beta-cell dysfunction in a mouse model of diabetes mellitus. Sleep 36: 1849–1858, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Sladek R, Rocheleau G, Rung J, Dina C, Shen L, Serre D, Boutin P, Vincent D, Belisle A, Hadjadj S, Balkau B, Heude B, Charpentier G, Hudson TJ, Montpetit A, Pshezhetsky AV, Prentki M, Posner BI, Balding DJ, Meyre D, Polychronakos C, and Froguel P. A genome-wide association study identifies novel risk loci for type 2 diabetes. Nature 445: 881–885, 2007 [DOI] [PubMed] [Google Scholar]
- 184.Solaini G, Baracca A, Lenaz G, and Sgarbi G. Hypoxia and mitochondrial oxidative metabolism. Biochim Biophys Acta 1797: 1171–1177, 2010 [DOI] [PubMed] [Google Scholar]
- 185.Sowter HM, Ratcliffe PJ, Watson P, Greenberg AH, and Harris AL. HIF-1-dependent regulation of hypoxic induction of the cell death factors BNIP3 and NIX in human tumors. Cancer Res 61: 6669–6673, 2001 [PubMed] [Google Scholar]
- 186.Starkov AA, Polster BM, and Fiskum G. Regulation of hydrogen peroxide production by brain mitochondria by calcium and Bax. J Neurochem 83: 220–228, 2002 [DOI] [PubMed] [Google Scholar]
- 187.Steiner DF. Adventures with insulin in the islets of Langerhans. J Biol Chem 286: 17399–17421, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Stokes RA, Cheng K, Deters N, Lau SM, Hawthorne WJ, O'Connell PJ, Stolp J, Grey S, Loudovaris T, Kay TW, Thomas HE, Gonzalez FJ, and Gunton JE. Hypoxia-inducible factor-1alpha (HIF-1alpha) potentiates beta-cell survival after islet transplantation of human and mouse islets. Cell Transplant 22: 253–266, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Stork CJ. and Li YV. Rising zinc: a significant cause of ischemic neuronal death in the CA1 region of rat hippocampus. J Cereb Blood Flow Metab 29: 1399–1408, 2009 [DOI] [PubMed] [Google Scholar]
- 190.Suksomboon N, Poolsup N, and Sinprasert S. Effects of vitamin E supplementation on glycaemic control in type 2 diabetes: systematic review of randomized controlled trials. J Clin Pharm Ther 36: 53–63, 2011 [DOI] [PubMed] [Google Scholar]
- 191.Tabak AG, Jokela M, Akbaraly TN, Brunner EJ, Kivimaki M, and Witte DR. Trajectories of glycaemia, insulin sensitivity, and insulin secretion before diagnosis of type 2 diabetes: an analysis from the Whitehall II study. Lancet 373: 2215–2221, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Takahashi HK, Santos LR, Roma LP, Duprez J, Broca C, Wojtusciszyn A, and Jonas JC. Acute nutrient regulation of the mitochondrial glutathione redox state in pancreatic beta-cells. Biochem J 460: 411–423, 2014 [DOI] [PubMed] [Google Scholar]
- 193.Tanaka Y, Gleason CE, Tran PO, Harmon JS, and Robertson RP. Prevention of glucose toxicity in HIT-T15 cells and Zucker diabetic fatty rats by antioxidants. Proc Natl Acad Sci U S A 96: 10857–10862, 1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Tarasov AI, Griffiths EJ, and Rutter GA. Regulation of ATP production by mitochondrial Ca(2+). Cell Calcium 52: 28–35, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Tarasov AI, Semplici F, Li D, Rizzuto R, Ravier MA, Gilon P, and Rutter GA. Frequency-dependent mitochondrial Ca(2+) accumulation regulates ATP synthesis in pancreatic beta cells. Pflugers Arch 465: 543–554, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Thorens B. GLUT2, glucose sensing and glucose homeostasis. Diabetologia 58: 221–232, 2015 [DOI] [PubMed] [Google Scholar]
- 197.Thorrez L, Laudadio I, Van Deun K, Quintens R, Hendrickx N, Granvik M, Lemaire K, Schraenen A, Van Lommel L, Lehnert S, Aguayo-Mazzucato C, Cheng-Xue R, Gilon P, Van Mechelen I, Bonner-Weir S, Lemaigre F, and Schuit F. Tissue-specific disallowance of housekeeping genes: the other face of cell differentiation. Genome Res 21: 95–105, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Tonooka N, Oseid E, Zhou H, Harmon JS, and Robertson RP. Glutathione peroxidase protein expression and activity in human islets isolated for transplantation. Clin Transplant 21: 767–772, 2007 [DOI] [PubMed] [Google Scholar]
- 199.Turner RC. The U.K. Prospective Diabetes Study. A review. Diabetes Care 21 Suppl 3: C35–C38, 1998 [DOI] [PubMed] [Google Scholar]
- 200.Turrens JF. Mitochondrial formation of reactive oxygen species. J Physiol 552: 335–344, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Vallee BL. and Falchuk KH. The biochemical basis of zinc physiology. Physiol Rev 73: 79–118, 1993 [DOI] [PubMed] [Google Scholar]
- 202.Vinkenborg JL, Nicolson TJ, Bellomo EA, Koay MS, Rutter GA, and Merkx M. Genetically encoded FRET sensors to monitor intracellular Zn2+ homeostasis. Nat Methods 6: 737–740, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Viswanath K, Bodiga S, Balogun V, Zhang A, and Bodiga VL. Cardioprotective effect of zinc requires ErbB2 and Akt during hypoxia/reoxygenation. Biometals 24: 171–180, 2011 [DOI] [PubMed] [Google Scholar]
- 204.Wang WM, Liu Z, Liu AJ, Wang YX, Wang HG, An D, Heng B, Xie LH, Duan JL, and Liu YQ. The Zinc Ion Chelating Agent TPEN Attenuates Neuronal Death/apoptosis Caused by Hypoxia/ischemia Via Mediating the Pathophysiological Cascade Including Excitotoxicity, Oxidative Stress, and Inflammation. CNS Neurosci Ther 21: 708–717, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Welsh N, Margulis B, Borg LA, Wiklund HJ, Saldeen J, Flodstrom M, Mello MA, Andersson A, Pipeleers DG, Hellerstrom C, et al. . Differences in the expression of heat-shock proteins and antioxidant enzymes between human and rodent pancreatic islets: implications for the pathogenesis of insulin-dependent diabetes mellitus. Mol Med 1: 806–820, 1995 [PMC free article] [PubMed] [Google Scholar]
- 206.Wicksteed B, Brissova M, Yan W, Opland DM, Plank JL, Reinert RB, Dickson LM, Tamarina NA, Philipson LH, Shostak A, Bernal-Mizrachi E, Elghazi L, Roe MW, Labosky PA, Myers MG, Jr., Gannon M, Powers AC, and Dempsey PJ. Conditional gene targeting in mouse pancreatic ss-Cells: analysis of ectopic Cre transgene expression in the brain. Diabetes 59: 3090–3098, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Wiseman DA, Kalwat MA, and Thurmond DC. Stimulus-induced S-nitrosylation of Syntaxin 4 impacts insulin granule exocytosis. J Biol Chem 286: 16344–16354, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Yoshikawa Y, Adachi Y, Yasui H, Hattori M, and Sakurai H. Oral administration of Bis(aspirinato)zinc(II) complex ameliorates hyperglycemia and metabolic syndrome-like disorders in spontaneously diabetic KK-A(y) mice: structure-activity relationship on zinc-salicylate complexes. Chem Pharm Bull (Tokyo) 59: 972–977, 2011 [DOI] [PubMed] [Google Scholar]
- 209.Yu JH, Kim KH, and Kim H. Role of NADPH oxidase and calcium in cerulein-induced apoptosis: involvement of apoptosis-inducing factor. Ann N Y Acad Sci 1090: 292–297, 2006 [DOI] [PubMed] [Google Scholar]
- 210.Zehetner J, Danzer C, Collins S, Eckhardt K, Gerber PA, Ballschmieter P, Galvanovskis J, Shimomura K, Ashcroft FM, Thorens B, Rorsman P, and Krek W. PVHL is a regulator of glucose metabolism and insulin secretion in pancreatic beta cells. Genes Dev 22: 3135–3146, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Zhang C, Lu X, Tan Y, Li B, Miao X, Jin L, Shi X, Zhang X, Miao L, Li X, and Cai L. Diabetes-induced hepatic pathogenic damage, inflammation, oxidative stress, and insulin resistance was exacerbated in zinc deficient mouse model. PLoS One 7: e49257, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Zhang CY, Baffy G, Perret P, Krauss S, Peroni O, Grujic D, Hagen T, Vidal-Puig AJ, Boss O, Kim YB, Zheng XX, Wheeler MB, Shulman GI, Chan CB, and Lowell BB. Uncoupling protein-2 negatively regulates insulin secretion and is a major link between obesity, beta cell dysfunction, and type 2 diabetes. Cell 105: 745–755, 2001 [DOI] [PubMed] [Google Scholar]
- 213.Zhao C, Wilson MC, Schuit F, Halestrap AP, and Rutter GA. Expression and distribution of lactate/monocarboxylate transporter isoforms in pancreatic islets and the exocrine pancreas. Diabetes 50: 361–366, 2001 [DOI] [PubMed] [Google Scholar]
- 214.Zheng X, Zheng X, Wang X, Ma Z, Gupta Sunkari V, Botusan I, Takeda T, Bjorklund A, Inoue M, Catrina SB, Brismar K, Poellinger L, and Pereira TS. Acute hypoxia induces apoptosis of pancreatic beta-cell by activation of the unfolded protein response and upregulation of CHOP. Cell Death Dis 3: e322, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Zraika S, Aston-Mourney K, Laybutt DR, Kebede M, Dunlop ME, Proietto J, and Andrikopoulos S. The influence of genetic background on the induction of oxidative stress and impaired insulin secretion in mouse islets. Diabetologia 49: 1254–1263, 2006 [DOI] [PubMed] [Google Scholar]