Abstract
Constipation is one of the most prevalent conditions in primary care settings and increases the risk of cardiovascular disease, potentially through processes mediated by altered gut microbiota. However, little is known about the association of constipation with CKD. In a nationwide cohort of 3,504,732 United States veterans with an eGFR ≥60 ml/min per 1.73 m2, we examined the association of constipation status and severity (absent, mild, or moderate/severe), defined using diagnostic codes and laxative use, with incident CKD, incident ESRD, and change in eGFR in Cox models (for time-to-event analyses) and multinomial logistic regression models (for change in eGFR). Among patients, the mean (SD) age was 60.0 (14.1) years old; 93.2% of patients were men, and 24.7% were diabetic. After multivariable adjustments, compared with patients without constipation, patients with constipation had higher incidence rates of CKD (hazard ratio, 1.13; 95% confidence interval [95% CI], 1.11 to 1.14) and ESRD (hazard ratio, 1.09; 95% CI, 1.01 to 1.18) and faster eGFR decline (multinomial odds ratios for eGFR slope <−10, −10 to <−5, and −5 to <−1 versus −1 to <0 ml/min per 1.73 m2 per year, 1.17; 95% CI, 1.14 to 1.20; 1.07; 95% CI, 1.04 to 1.09; and 1.01; 95% CI, 1.00 to 1.03, respectively). More severe constipation associated with an incrementally higher risk for each renal outcome. In conclusion, constipation status and severity associate with higher risk of incident CKD and ESRD and with progressive eGFR decline, independent of known risk factors. Further studies should elucidate the underlying mechanisms.
Keywords: chronic kidney disease, end-stage renal disease, glomerular filtration rate, Epidemiology and outcomes
CKD is a growing public health problem due to its increasing prevalence and strong association with cardiovascular disease, ESRD, and mortality.1 Although some risk factors of CKD, such as diabetes mellitus and hypertension,2 have been established, the identification of novel risk factors and interventions applicable to primary care settings may help to ameliorate the risk for subsequent adverse outcomes and reduce the global burden of CKD.
Constipation is the prototype of functional gastrointestinal disorders and one of the most prevalent conditions encountered in primary care settings. Approximately 30% of the general population experience problems with constipation during their lifetime,3 with elderly people and women being mostly affected. The chronic symptoms of constipation impair patients’ quality of life and may impose a substantial economic burden on patients and society.4,5 Furthermore, recent observational studies have shown an association of chronic constipation with increased risk of cardiovascular disease6,7 and suggested the possible involvement of chronic inflammation caused by altered gut microbiota as an underlying mechanism for the association.6,7 It is plausible that constipation may also be a risk factor for the development of CKD, potentially mediated by altered gut microbiota or other intermediate risk factors, such as diabetes, use of nonsteroidal anti–inflammatory drugs, or lack of physical exercise, which have been associated with both constipation6,8 and CKD progression2,9–11; however, to the best of our knowledge, no prior studies have examined the association between constipation and the risk of CKD.
In this study, we hypothesized that patients with constipation are at higher risk of incident kidney disease and more likely to experience rapid decline of kidney function and that patients with more severe constipation would have a greater risk of such events than those with less severe constipation. To test these hypotheses, we investigated the association of constipation status and its severity with incident CKD and ESRD along with change in eGFR using a large nationally representative cohort of United States veterans with eGFR of ≥60 ml/min per 1.73 m2.
Results
Patients’ baseline characteristics overall and those in patients categorized by constipation status are shown in Table 1. The mean age at baseline was 60.0 years old (SD=14.1); 93.2% were men, 15.3% were black, and 24.7% were diabetic. The mean eGFR was 83.8 ml/min per 1.73 m2 (SD=15.6). Compared with patients without constipation, those with constipation were older and more likely to be black, had a higher prevalence of comorbidities except HIV/AIDS and a lower per capita income, and were less likely to be married. They also had more frequent health care encounters and longer cumulative length of hospitalization during the 2-year baseline period. The use of angiotensin–converting enzyme inhibitors/angiotensin II receptor blockers, statins, antidepressants, nonopioid analgesics, and opioids and the administration of influenza vaccination(s) were more common in patients with constipation. Baseline characteristics were well balanced in the propensity-matched cohort (Table 1).
Table 1.
Baseline patient characteristics in the overall cohort and propensity-matched cohort
| Characteristics | Overall Cohort | Propensity-Matched Cohort | |||||
|---|---|---|---|---|---|---|---|
| Total, n=3,504,732 | Constipation | Std. Diff. | Constipation | Std. Diff. | |||
| No, n=3,251,291 | Yes, n=253,441 | No, n=208,979 | Yes, n=208,979 | ||||
| Mean age (SD), yr | 60.0 (14.1) | 59.6 (14.1) | 64.1 (13.0) | 0.33 | 63.9 (12.3) | 63.5 (12.8) | −0.031 |
| Mean eGFR (SD), ml/min per 1.73 m2 | 83.8 (15.6) | 83.9 (15.5) | 82.8 (15.8) | −0.08 | 82.9 (15.5) | 83.1 (15.8) | 0.01 |
| Men, n (%) | 3,267,365 (93.2) | 3,030,680 (93.2) | 236,685 (93.4) | 0.028 | 195,507 (93.6) | 195,626 (93.6) | −0.002 |
| Black, n (%) | 537,191 (15.3) | 487,079 (15.0) | 50,112 (20.0) | 0.096 | 44,180 (21.1) | 43,716 (20.9) | −0.005 |
| Hypertension, n (%) | 2,079,730 (59.3) | 1,899,132 (58.4) | 180,598 (71.3) | 0.25 | 151,774 (72.6) | 150,044 (71.8) | −0.018 |
| Diabetes mellitus, n (%) | 831,043 (24.7) | 749,118 (23.0) | 81,925 (32.3) | 0.19 | 69,389 (33.2) | 68,462 (32.8) | −0.009 |
| CHD, n (%) | 399,833 (11.4) | 353,552 (10.9) | 46,281 (18.3) | 0.21 | 39,716 (19.0) | 39,293 (18.8) | −0.005 |
| CHF, n (%) | 152,126 (4.3) | 125,624 (3.9) | 26,502 (10.5) | 0.25 | 21,680 (10.4) | 22,215 (10.6) | 0.008 |
| CVD, n (%) | 212,024 (6.1) | 180,585 (5.6) | 31,439 (12.4) | 0.23 | 26,544 (12.7) | 26,203 (12.5) | −0.005 |
| PAD, n (%) | 190,548 (5.4) | 162,877 (5.0) | 27,671 (10.9) | 0.21 | 23,452 (11.2) | 23,357 (11.2) | −0.001 |
| Chronic lung disease, n (%) | 637,350 (18.2) | 558,622 (17.2) | 78,728 (31.1) | 0.32 | 67,551 (32.3) | 66,434 (31.8) | −0.011 |
| Dementia, n (%) | 28,646 (0.8) | 22,741 (0.7) | 5905 (2.3) | 0.13 | 4284 (2.1) | 4487 (2.2) | 0.007 |
| Rheumatologic disease, n (%) | 49,021 (1.4) | 43,098 (1.3) | 5923 (2.3) | 0.075 | 5167 (2.5) | 4998 (2.4) | −0.005 |
| Peptic ulcer disease, n (%) | 64,990 (1.9) | 55,294 (1.7) | 9696 (3.8) | 0.13 | 8010 (3.8) | 8224 (3.9) | 0.005 |
| Liver disease, n (%) | 14,795 (0.4) | 10,061 (0.3) | 4734 (1.9) | 0.15 | 3476 (1.7) | 4042 (1.9) | 0.02 |
| Malignancies, n (%) | 355,185 (10.1) | 307,105 (9.5) | 48,080 (19.0) | 0.28 | 40,777 (19.5) | 39,979 (19.1) | −0.01 |
| HIV/AIDS, n (%) | 21,247 (0.6) | 19,637 (0.6) | 1610 (0.6) | −0.001 | 1330 (0.6) | 1389 (0.7) | 0.004 |
| Depression, n (%) | 323,221 (9.2) | 279,955 (8.6) | 43,266 (17.1) | 0.24 | 37,499 (17.9) | 37,310 (17.9) | −0.002 |
| Bowel disorders, n (%) | 145,811 (4.2) | 122,835 (3.8) | 22,976 (9.1) | 0.21 | 19,212 (9.2) | 19,499 (9.3) | 0.005 |
| Median per capita income (IQR), $ | 22,971 (11,725–36,048) | 23,258 (11,866–37,111) | 19,061 (10,647–30,613) | −0.20 | 18,856 (10,606–30,453) | 19,119 (10,717–30,588) | 0.029 |
| Married, n (%) | 1,880,248 (53.7) | 1,758,279 (54.1) | 121,969 (48.1) | 0.096 | 107,179 (51.3) | 106,522 (51.0) | −0.006 |
| Service connected, n (%) | 1,426,273 (40.7) | 1,301,738 (40.0) | 124,535 (49.1) | 0.14 | 107,187 (51.3) | 105,723 (50.6) | −0.014 |
| Mean BMI (SD), kg/m2 | 29.2 (5.7) | 29.2 (5.7) | 29.2 (6.2) | 0.008 | 29.3 (6.1) | 29.3 (6.3) | 0.004 |
| Mean systolic BP (SD), mmHg | 135.4 (19.1) | 135.5 (19.1) | 134.1 (19.8) | −0.075 | 134.2 (19.2) | 134.2 (19.8) | −0.002 |
| Mean diastolic BP (SD), mmHg | 77.2 (11.8) | 77.3 (11.8) | 75.2 (12.0) | −0.19 | 75.3 (11.9) | 75.4 (12.0) | 0.011 |
| ACEI/ARB use, n (%) | 773,938 (22.1) | 703,531 (21.6) | 70,407 (27.8) | 0.14 | 58,796 (28.1) | 57,888 (27.7) | −0.01 |
| Statin use, n (%) | 506,980 (14.5) | 459,218 (14.1) | 47,762 (18.9) | 0.13 | 39,531 (18.9) | 39,086 (18.7) | −0.005 |
| Antidepressants use, n (%) | 622,543 (17.8) | 538,862 (16.6) | 83,681 (33.0) | 0.36 | 71,663 (34.3) | 69,854 (33.4) | −0.018 |
| Nonopioid analgesics use, n (%) | 827,856 (23.6) | 725,729 (22.3) | 102,127 (40.3) | 0.37 | 87,710 (42.0) | 85,186 (40.8) | −0.025 |
| Opioids use, n (%) | 385,838 (11.0) | 316,038 (9.7) | 69,800 (27.5) | 0.46 | 60,782 (29.1) | 59,486 (28.5) | −0.014 |
| Influenza vaccination, n (%) | 1,032,254 (29.5) | 920,070 (28.3) | 112,184 (44.3) | 0.33 | 96,629 (46.2) | 93,828 (44.9) | −0.027 |
| Living in area with high housing stress, n (%) | 1,181,986 (33.7) | 1,090,152 (33.5) | 91,834 (36.2) | 0.067 | 79,956 (38.3) | 80,040 (38.3) | 0.001 |
| Living in area with low education, n (%) | 364,485 (10.4) | 334,018 (10.3) | 30,467 (12.0) | 0.05 | 26,885 (12.9) | 26,407 (12.6) | −0.007 |
| Living in area with low employment, n (%) | 318,669 (9.1) | 292,720 (9.0) | 25,949 (10.2) | 0.035 | 22,829 (10.9) | 22,374 (10.7) | −0.007 |
| Living in area of persistent poverty, n (%) | 166,638 (4.8) | 152,459 (4.7) | 14,179 (5.6) | 0.036 | 12,603 (6.0) | 12,190 (5.8) | −0.008 |
| Health care encounters, n (IQR) | 20 (10–38) | 19 (10–35) | 51 (28–85) | 0.77 | 45 (24–81) | 54 (31–88) | 0.053 |
| Cumulative length of hospitalization (IQR), d | 0 (0–0) | 0 (0–0) | 0 (0–6) | 0.23 | 0 (0–3) | 0 (0–7) | 0.017 |
Data are presented as number (percentage), mean±SD, or median (IQR). Std. Diff., standardized difference; CHF, congestive heart failure; CVD, cerebrovascular disease; PAD, peripheral arterial disease; BMI, body mass index; ACEI/ARB, angiotensin–converting enzyme inhibitor/angiotensin II receptor blocker.
Incident CKD
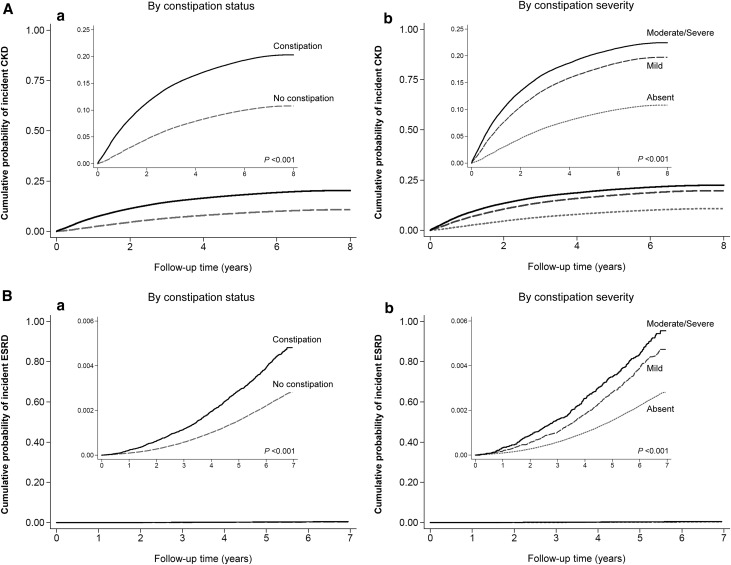
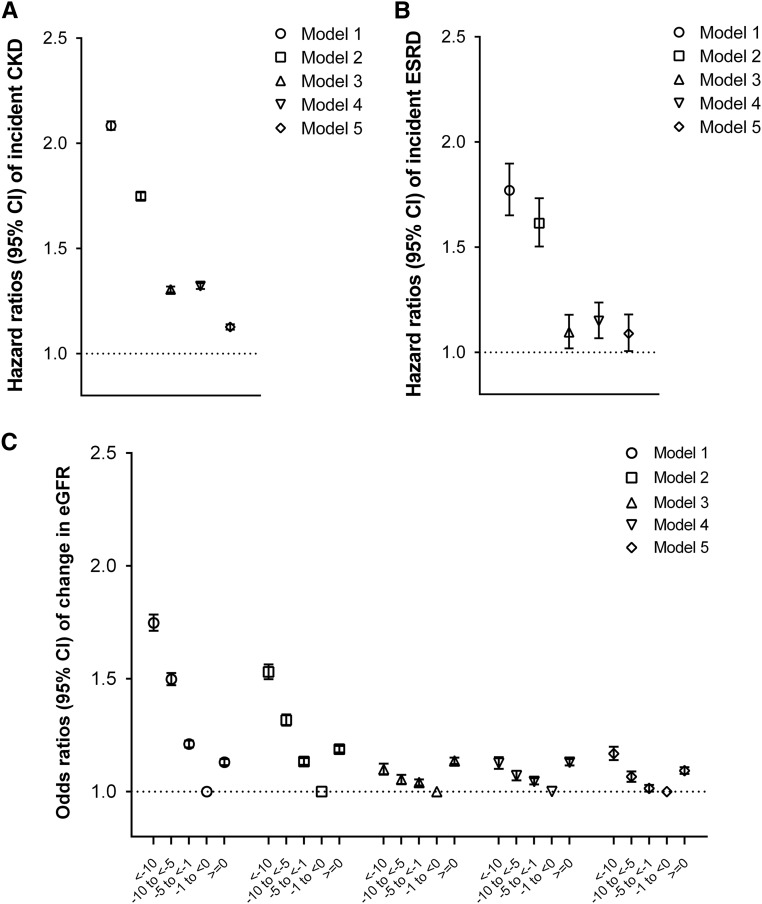
During a median follow-up of 7 years, there were a total of 360,541 events of incident CKD (crude rate, 17.2 per 1000 patient-years; 95% confidence interval [95% CI], 17.2 to 17.3), including 46,022 (crude rate, 33.9 per 1000 patient-years; 95% CI, 33.6 to 34.2) and 314,519 (crude rate, 16.1 per 1000 patient-years; 95% CI, 16.0 to 16.1) events in patients with and without constipation, respectively. As depicted in Figure 1A, patients with constipation had a higher cumulative incidence of CKD (log rank P<0.001). Figure 2A shows the association between constipation status and incident CKD in unadjusted and adjusted models. In the crude model, the presence of constipation was associated with a higher risk of incident CKD (hazard ratio [HR], 2.08; 95% CI, 2.06 to 2.10). Although adjustment for covariates resulted in the attenuation of this association, the risk of incident CKD remained significantly higher in patients with constipation (adjusted HR, 1.13; 95% CI, 1.11 to 1.14). Compared with patients with absent constipation, those with more severe constipation showed incrementally higher associations with the incidence of CKD: log rank P<0.001 (Figure 1A); fully adjusted HRs, 1.10; 95% CI, 1.09 to 1.12 and 1.16; 95% CI, 1.14 to 1.18 for mild and moderate/severe constipation, respectively (Supplemental Figure 1A).
Figure 1.
Unadjusted Kaplan–Meier cumulative event curves for renal events. (A) Incident CKD and (B) incident ESRD according to (a) constipation status and (b) its severity. Cumulative incidences of (A) CKD and (B) ESRD were higher in patients with (versus without) constipation and with more severe constipation.
Figure 2.
Association of the presence of constipation with renal events. (A) Incident CKD, (B) incident ESRD, and (C) change in eGFR. The presence of constipation was associated with higher incidence rates of CKD and ESRD and faster eGFR decline, compared with the absence of constipation. Estimates were calculated using Cox proportional models (for incident CKD and ESRD) and multinomial logistic regression models (for change in eGFR). Models represent unadjusted association (model 1) and associations after adjustment for age, sex, race, and baseline eGFR (model 2); model 2 variables plus comorbidities (diabetes mellitus, hypertension, CHD, congestive heart failure, cerebrovascular disease, peripheral arterial disease, peptic ulcer disease, rheumatic disease, malignancy, depression, liver disease, chronic lung disease, HIV/AIDS, and bowel disorders; model 3); model 3 plus baseline body mass index, systolic BP, and diastolic BP (model 4); and model 4 plus socioeconomic parameters (mean per capita income, marital status, service connectedness, housing stress, low education, low employment, and persistent poverty), number of VA health care encounters, cumulative length of hospitalization, receipt of influenza vaccination(s), and use of angiotensin–converting enzyme inhibitors/angiotensin II receptor blockers, statins, antidepressants, nonopioid analgesics, and opioids (model 5).
Incident ESRD
A total of 7677 patients developed ESRD (crude rate, 0.39 per 1000 patient-years; 95% CI, 0.38 to 0.40), including 902 (crude rate, 0.65 per 1000 patient-years; 95% CI, 0.61 to 0.70) and 6775 (crude rate, 0.37 per 1000 patient-years; 95% CI, 0.36 to 0.38) events in patients with and without constipation, respectively. Cumulative incidence of ESRD was higher in patients with (versus without) constipation (log rank P<0.001) (Figure 1B). Patients with constipation had a significantly higher risk of incident ESRD (adjusted HR, 1.09; 95% CI, 1.01 to 1.18) (Figure 2B). With increasing constipation severity, a higher cumulative incidence of ESRD (Figure 1B) as well as a greater risk of incident ESRD (Supplemental Figure 1B) were also observed.
Change in eGFR
Among 3,242,681 patients in the eGFR slope analysis, 119,165 (3.7%), 189,792 (5.9%), and 905,877 (27.9%) experienced decline in eGFR of <−10, −10 to <−5, and −5 to <−1 ml/min per 1.73 m2 per year, respectively, whereas 1,378,842 patients (42.5%) had stable or increasing eGFR (≥0 ml/min per 1.73 m2 per year) (Supplemental Table 1). Figure 2C shows the association between constipation status and change in eGFR. Compared with patients without constipation, those with constipation were at a greater risk of experiencing more progressive eGFR decline, with higher risks seen in patients with faster eGFR decline (adjusted multinomial odds ratios [MORs], 1.17; 95% CI, 1.14 to 1.20 for eGFR slopes <−10; 1.07; 95% CI, 1.04 to 1.09 for −10 to <−5; and 1.01; 95% CI, 1.00 to 1.03 for −5 to <−1 [versus −1 to <0] ml/min per 1.73 m2 per year), and they also had a higher risk of increasing eGFR (adjusted odds ratio, 1.09; 95% CI, 1.08 to 1.11 for eGFR slopes ≥0 [versus −1 to <0] ml/min per 1.73 m2 per year). The risk of progressive eGFR decline was higher in patients with mild and moderate/severe constipation than those with absent constipation (adjusted MORs of eGFR slopes <−10 [versus −1 to <0] ml/min per 1.73 m2 per year, 1.18; 95% CI, 1.14 to 1.21 and 1.30; 95% CI, 1.25 to 1.35, respectively; adjusted MORs of eGFR slopes −10 to <−5, 1.07; 95% CI, 1.04 to 1.10 and 1.12; 95% CI, 1.09 to 1.16, respectively; and adjusted MORs of eGFR slopes −5 to <−1, 1.01; 95% CI, 0.99 to 1.03 and 1.04; 95% CI, 1.02 to 1.06, respectively) (Supplemental Figure 1C). Similarly, the risk of increasing eGFR was incrementally higher in those with more severe constipation (adjusted odd ratios of eGFR≥0 [versus −1 to <0] ml/min per 1.73 m2 per year, 1.08; 95% CI, 1.06 to 1.10 and 1.14; 95% CI, 1.12 to 1.17 for mild and moderate/severe constipation, respectively) (Supplemental Figure 1C).
Sensitivity Analyses
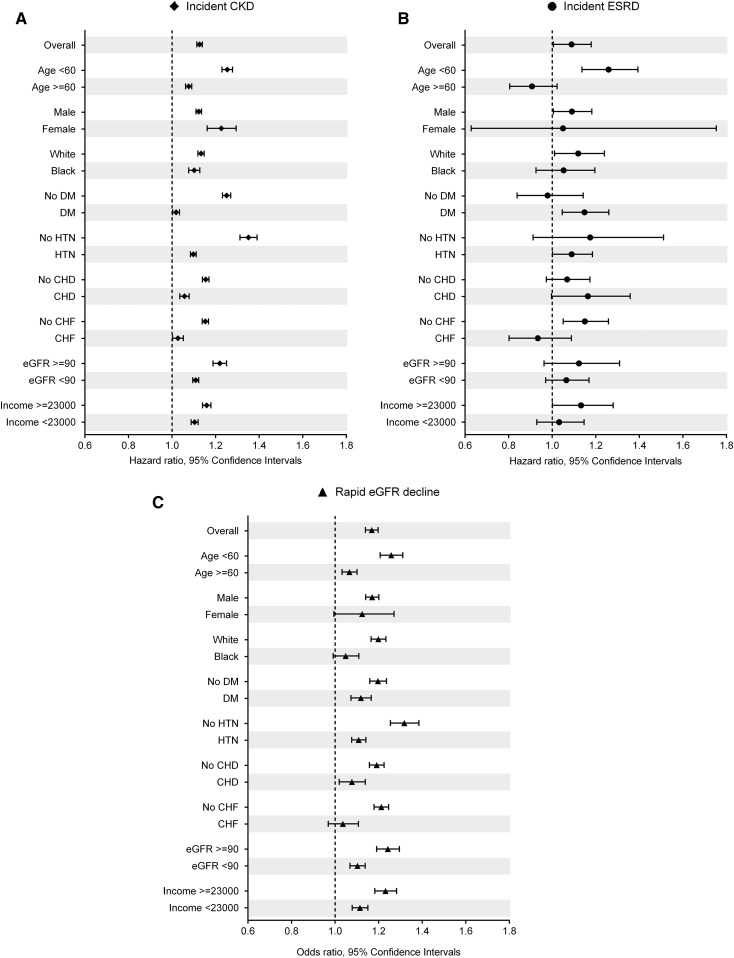
Results were similar in the various sensitivity analyses (Figure 3, Supplemental Figure 2, and Supplemental Tables 2 and 3).
Figure 3.
Association of the presence of constipation with renal events in predefined subgroups of the overall cohort. (A) Incident CKD, (B) incident ESRD, and (C) change in eGFR. The presence (versus absence) of constipation was associated with higher risk of incident CKD and ESRD and faster eGFR decline in most subgroups. Estimates were calculated using Cox proportional models (for incident CKD and ESRD) and multinomial logistic regression models (for eGFR slope [milliliter per minute per 1.73 m2 per year]; <−10 versus −1 to <0 [reference]). Data were adjusted for age, sex, race, baseline eGFR, comorbidities (diabetes mellitus [DM], hypertension, CHD, congestive heart failure [CHF], cerebrovascular disease, peripheral arterial disease, peptic ulcer disease, rheumatic disease, malignancy, depression, liver disease, chronic lung disease, HIV/AIDS, and bowel disorders), baseline body mass index, systolic BP, diastolic BP, socioeconomic parameters (mean per capita income, marital status, service connectedness, housing stress, low education, low employment, and persistent poverty), number of VA health care encounters, cumulative length of hospitalization, receipt of influenza vaccination(s), and use of angiotensin–converting enzyme inhibitors/angiotensin II receptor blockers, statins, antidepressants, nonopioid analgesics, and opioids. HTN, hypertension.
Discussion
In this large cohort of United States veterans with baseline eGFR ≥60 ml/min per 1.73 m2, we examined the association of constipation status and its severity with incident CKD, incident ESRD, and change in eGFR. We found that the presence of constipation and the severity of constipation were associated with increased risk of incident CKD, incident ESRD, and progressive eGFR decline. These findings were similarly observed in selected subgroups and robust to sensitivity analyses accounting for confounding by indication, competing risk, and missing data.
The prevalence of constipation has been shown to be higher in patients with CKD, particularly among those undergoing dialysis, than the general population,12 mostly due to their dietary restrictions, medications like phosphate binders, and high prevalence of comorbidities.13 In recent years, there has been a growing interest in the association between CKD and intestinal environment (often referred to as “CKD-Colonic Axis”), showing that CKD can cause significant quantitative and qualitative alterations of gut environment, which in turn, may contribute to the pathogenesis of CKD progression and several CKD–related complications.14 However, these studies have focused primarily on patients with advanced CKD, and hence, it remains unknown whether the altered gut environment is associated with the development of de novo kidney disease in patients with preserved kidney function. Given the fact that gastrointestinal motility and gut environment are inter-related and exert reciprocal effects on each other,15,16 it is plausible that constipation, one of the clinical forms of altered gut environment, can be a risk factor for kidney disease progression.
There are several potential mechanisms for the association between constipation and the risk of adverse renal outcomes. Recently, emerging evidence has disclosed a tight and coordinated connection between gut microbiota and host nutrition, metabolism, and immune function,17 indicating that disturbance of the gut microbiota is linked to the pathogenesis of diverse illnesses, such as metabolic syndrome18 and cardiovascular disease,19 through chronic inflammation and/or altered metabolite profiles. For example, trimethylamine-N-oxide, a gut microbiota–dependent metabolite, has been shown to play a significant role in the development and progression of atherosclerosis and adverse atherosclerotic cardiac events.20,21 Furthermore, recent metabolomics data from the Framingham Heart Study highlight that trimethylamine-N-oxide levels predict the risk of incident CKD in healthy subjects.22 Alterations in gut microbiota have also been linked to the accumulation of gut–derived uremic toxins, such as indoxyl sulfate and p-cresyl sulfate,9 which in turn, seem to accelerate kidney disease progression by causing renal fibrosis, inflammation, and oxidative stress.23 Although a large clinical trial examining the reduction of systemic toxin absorption through gastrointestinal sequestration via spherical carbon adsorbent AST-120 (Kremezin; Kureha Corporation, Tokyo, Japan) has failed to show a benefit toward slowing kidney disease progression,24 adherence to the medication in this clinical trial was poor, and hence, it remains unclear if proper administration of this or other similar products could be renoprotective. Considering the possible existence of altered gut microbiota in patient with constipation, these pathophysiologic mechanisms could serve as a potential explanation for the association between constipation and adverse renal outcomes.
Another plausible mechanism for the observed association may be through increased levels of serotonin. Serotonin is synthesized in the gut and incorporated into platelets, which when activated, release serotonin and enhance vasoconstriction and thrombus formation, resulting in the development of atherosclerotic plaques.25 Previous studies have also reported an association between elevated plasma levels of serotonin and increased risk of atherosclerotic cardiovascular disease.26 Because serotonin synthesis and release have been shown to be increased in patients with constipation27 and those using certain laxatives,28 increased serotonin levels could also explain the underlying pathogenesis of kidney disease progression. In addition to these mechanisms, the use of certain types of laxatives may directly (e.g., through drug-induced nephrotoxicity) or indirectly (e.g., through dehydration or electrolyte disturbances) cause kidney damage and could potentially contribute to the increased risk of renal events.
Interestingly, we found a significant association between constipation and the risk of increasing eGFR. As previously reported,29–31 increasing eGFR has been recognized as a predictor of adverse clinical outcomes through loss of muscle mass associated with chronic debilitating conditions, and increasing eGFR may also reflect recovery from AKI. Although we did not measure lean body mass and hence, precise mechanisms underlying the observed association remain speculative, the greater risk of increasing eGFR might reflect a higher incidence of some chronic illnesses accompanied by a decline in serum creatinine among patients with constipation. Most importantly, however, the associations of constipation with adverse renal outcomes still remained statistically significant even after accounting for various potential confounders, including comorbidities, number of Veterans Affairs (VA) health care encounters, and cumulative length of hospitalization, which highlights the biologically plausible link between the gut and kidney and provides additional insights into the pathogenesis of kidney disease progression.
Given the high prevalence of constipation and the simplicity of its assessment in primary care settings, our study may also have several clinical implications. Physicians should be aware of the risk of kidney disease progression in patients with constipation. When evaluating a patient with constipation and reduced kidney function in clinical practice, it is also important for health care providers to acknowledge the excess risk of kidney damage caused by dehydration and use (especially over the counter use) of nonsteroidal anti–inflammatory drugs. Careful observation of kidney function trajectory may, thus, be required in those patients, particularly among those with more severe constipation. Furthermore, if the relationships between constipation and renal events are causal, our findings suggest that the treatment of constipation through lifestyle modifications (e.g., exercise and high-fiber diet32,33) and/or use of probiotics34 rather than laxatives could potentially reduce the risk of adverse renal outcomes and may deserve future clinical trials.
Our study is notable for its large sample size and being representative of veterans in the entire geographic United States; however, several limitations need to be acknowledged. Because this study was observational, the results do not allow us to infer causality. It could be argued that constipation may be merely a nonspecific marker of general illness rather than a causative factor; however, the significant association that was robust to adjustment for a wide assortment of confounders combined with plausible mechanistic explanations raise the possibility of a causal link between constipation and renal outcomes. Most of our patients were men; hence, the results may not be generalizable to women or patients from other geographic areas. We defined our cohort on the basis of an eGFR≥60 ml/min per 1.73 m2, but we did not have markers of earlier stages of CKD (e.g., proteinuria). Because information about subjective symptoms of constipation was not available and constipation status and its severity were defined using the International Classification of Diseases, Ninth Revision Clinical Modification (ICD-9-CM) codes and laxative prescription records during the 2-year baseline period, we were unable to assess the effect of lifetime duration of constipation and its status over the entire follow-up period on the outcomes, and patients with constipation might have been misclassified as absent constipation or having less severe constipation. Nevertheless, such misclassification would tend to bias the true effects toward the null, and our results still showed significant associations of constipation with increased risk of renal events. Several statistical methods were applied in our sensitivity analyses to address the effect of confounders, but we cannot eliminate the possibility of unmeasured confounders. In conclusion, in this large nationwide cohort of >3 million United States veterans, we found that constipation status and its severity were associated with a higher risk of incident CKD, incident ESRD, and progressive eGFR decline, independent of known risk factors. Additional studies are needed to elucidate the underlying mechanisms and determine whether the amelioration of constipation can prevent adverse renal outcomes.
Concise Methods
Cohort Definition
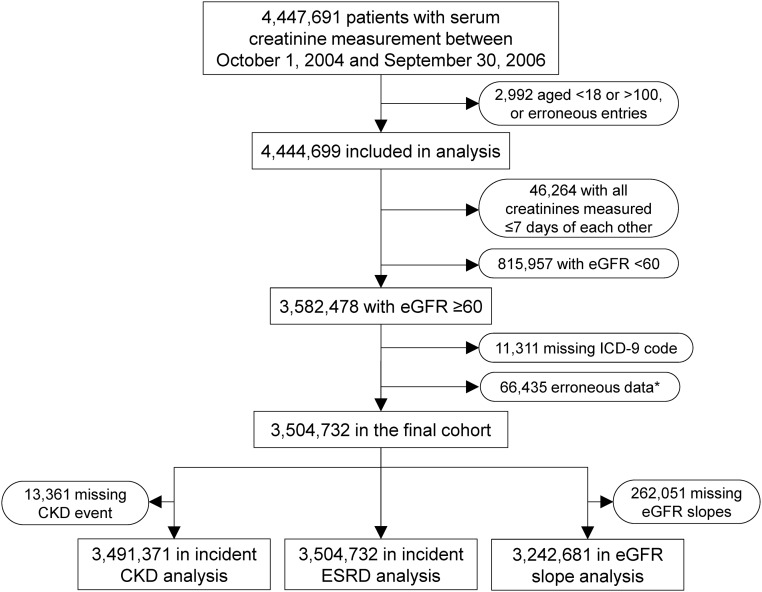
Our study used data from a retrospective cohort study examining risk factors in patients with incident CKD (the Racial and Cardiovascular Risk Anomalies in CKD Study).35 The algorithm for cohort definition is shown in Figure 4. We used the national VA Corporate Data Warehouse LabChem data files36 to extract serum creatinine measured between October 1, 2004 and September 30, 2006 (baseline period) and identified 3,582,478 patients with baseline eGFR≥60 ml/min per 1.73 m2. eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration creatinine equation.37 After exclusion of patients with missing ICD-9-CM codes (n=11,311) or erroneous data (n=66,435), 3,504,732 patients were included in our final cohort.
Figure 4.
Algorithm used to define the study cohort. *Patients with the date of cohort entry later than the date of last encounter (n=32,038), those with the date of incident ESRD later than the date of last encounter (n=2236), or those with an eGFR slope <−55 (0.5th percentile) or ≥40 (99.5th percentile) ml/min per 1.73 m2 per year (n=32,161).
Data Collection
Predictors and Covariates
Constipation was defined as having either at least two diagnoses for constipation as identified by the ICD-9-CM (Supplemental Table 4) that were >60 days apart or two or more prescriptions of laxatives of ≥30-day supply each that were 60–365 days apart during the baseline period on the basis of information obtained from VA Pharmacy dispensation records.38 Constipation severity was also quantitatively defined according to the number of different types of laxatives prescribed during the baseline period and stratified into three groups as follows: absent (no laxative), mild (one type of laxative), or moderate/severe (two or more types of laxatives).
Sociodemographic characteristics, comorbid conditions, medication use, and laboratory characteristics were obtained as previously described.39,40 Briefly, information about age, sex, race, marital status (married, single, divorced, or widowed), mean per capita income, service connectedness, body mass index, systolic and diastolic BPs, comorbid conditions, and medication use was obtained from various national VA research data files.41 Prevalent comorbidities were defined as the presence of relevant ICD-9-CM and Current Procedural Terminology codes recorded from October 1, 2004 to September 30, 2006 (Supplemental Table 4).39,40 Prevalent coronary heart disease (CHD) was defined as the presence of diagnostic codes for coronary artery disease, angina, or myocardial infarction or procedure codes for percutaneous coronary interventions or coronary artery bypass grafting. Bowel disorders were defined as the presence of diagnostic codes for inflammatory bowel disease, irritable bowel syndrome, or diarrhea. In addition to the information derived from VA sources, we included select socioeconomic indicators using 2004 county typology codes (housing stress, low education, low employment, and persistent poverty) (Supplemental Table 5).
Outcomes
The coprimary outcomes of interest were incident CKD, incident ESRD, and change in eGFR. Incident CKD was defined as two eGFR levels <60 ml/min per 1.73 m2 separated by ≥90 days and a >25% decrease from baseline eGFR.42 Incident ESRD was defined as initiation of maintenance dialysis or preemptive renal transplantation occurring between the cohort entry date and September 13, 2011, the last date of available event record provided by the United States Renal Data System (USRDS). Change in eGFR (slope) was calculated in each patient from an ordinary least squares regression model using all outpatient eGFR measurements available from the cohort entry date to October 13, 2012 (the last date of available serum creatinine measurement) and stratified into five categories as follows: <−10, −10 to <−5, −5 to <−1, −1 to 0 (reference), and ≥0 ml/min per 1.73 m2 per year. The median (interquartile range [IQR]) number of eGFR measurements used to calculate eGFR slopes was 10 (IQR, 5–17). Information about all-cause mortality was obtained from the VA Vital Status Files.43
Statistical Analyses
Data are presented as number (percentage) for categorical variables and mean±SD for continuous variables with a normal distribution or median (IQR) for those with a skewed distribution. The start of follow-up was the date of the first eGFR ≥60 ml/min per 1.73 m2 during the baseline period. Patients were followed up until death or the last date of VA contact. The associations of constipation status and its severity with outcomes were assessed with the Kaplan–Meier method and log rank tests and using Cox proportional hazards models (for time to event analyses) and multinomial logistic regression models (for change in eGFR). The proportionality assumption was tested by plotting log (−log [survival rate]) against log (survival time) and scaled Schoenfeld residuals, and it showed no violations. For the time to event analyses, patients were followed up until death or censored at the date of the last encounter or October 13, 2012 and September 13, 2011 for incident CKD and ESRD, respectively. All associations were examined in unadjusted and multivariable adjusted models. Models were incrementally adjusted for the following confounders on the basis of theoretical considerations: model 1, unadjusted; model 2, adjusted for age, sex, and baseline eGFR; model 3, model 2 variables plus prevalent comorbidities (diabetes mellitus, hypertension, CHD, congestive heart failure, cerebrovascular disease, peripheral vascular disease, peptic ulcer disease, rheumatic disease, malignancy, depression, liver disease, chronic lung disease, HIV/AIDS, and bowel disorders); model 4, model 3 variables plus baseline body mass index and systolic and diastolic BPs; and model 5, model 4 variables plus socioeconomic parameters (mean per capita income, marital status, service connectedness, housing stress, low education, low employment, and persistent poverty), indicators of sickness (number of VA health care encounters and cumulative length of hospitalization) and quality of care (receipt of influenza vaccination[s]), and use of angiotensin–converting enzyme inhibitors or angiotensin II receptor blockers, statins, antidepressants, nonopioid analgesics, and opioids at baseline.
We additionally performed several sensitivity analyses. All outcomes were examined in subgroups of patients categorized by baseline age, sex, race, prevalent diabetes mellitus, hypertension, CHD, congestive heart failure, eGFR, and income level. Analyses were repeated in a propensity score–matched cohort to account for baseline differences arising from dissimilarities in clinical and demographic characteristics of patients with and without constipation. Propensity scores for the likelihood of presence versus absence of constipation were calculated by logistic regression using all variables included in multivariable models and performing a 1:1 nearest neighbor matching without replacement. Because death and incident CKD/ESRD are competing events, competing risk regressions were also performed using unadjusted models in the overall cohort as well as the propensity-matched cohort. Of the variables included in multivariable adjusted models, data points were missing for race (9.7%), body mass index (4.7%), BP (1.4%), per capita income (6.9%), and socioeconomic indicators (4.0%). Missing values were not imputed in primary analyses but were substituted by multiple imputation procedures using the Stata (Stata Corporation, College Station, TX) “mi” set of commands in sensitivity analyses.
Because of the large sample size, the significance of differences in the main cohort was established on the basis of considerations of biologically or clinically meaningful differences. Differences between variables in the overall and propensity-matched cohorts were examined by calculating standardized differences, and values >0.1 were considered significant. All of the analyses were conducted using Stata/MP version 14 (Stata Corporation). The study was approved by the institutional review boards at the Memphis and Long Beach VA Medical Centers.
Disclosures
K.K.-Z. and C.P.K. are employees of the US Department of Veterans Affairs. None of the authors have relevant conflicts of interest.
Supplementary Material
Acknowledgments
This study was supported by grant R01DK096920 (to K.K.-Z. and C.P.K.) from the National Institute of Diabetes and Digestive and Kidney Diseases and is the result of work supported with resources and the use of facilities at the Memphis Veterans Affairs Medical Center and the Long Beach Veterans Affairs Medical Center. Support for Veterans Affairs/Centers for Medicare and Medicaid Services (CMS) data is provided by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Health Services Research and Development, Veterans Affairs Information Resource Center projects SDR 02-237 and 98-004.
The sponsors had no role in the design and conduct of the study; the collection, analysis, and interpretation of the data; or the preparation, review, or approval of the manuscript. Opinions expressed in this paper are those of the authors and do not necessarily represent the opinion of the Department of Veterans Affairs.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2016060656/-/DCSupplemental.
References
- 1.Eckardt KU, Coresh J, Devuyst O, Johnson RJ, Köttgen A, Levey AS, Levin A: Evolving importance of kidney disease: From subspecialty to global health burden. Lancet 382: 158–169, 2013 [DOI] [PubMed] [Google Scholar]
- 2.Levey AS, Coresh J: Chronic kidney disease. Lancet 379: 165–180, 2012 [DOI] [PubMed] [Google Scholar]
- 3.Bharucha AE, Pemberton JH, Locke GR 3rd : American Gastroenterological Association technical review on constipation. Gastroenterology 144: 218–238, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sun SX, Dibonaventura M, Purayidathil FW, Wagner JS, Dabbous O, Mody R: Impact of chronic constipation on health-related quality of life, work productivity, and healthcare resource use: An analysis of the National Health and Wellness Survey. Dig Dis Sci 56: 2688–2695, 2011 [DOI] [PubMed] [Google Scholar]
- 5.Guerin A, Carson RT, Lewis B, Yin D, Kaminsky M, Wu E: The economic burden of treatment failure amongst patients with irritable bowel syndrome with constipation or chronic constipation: A retrospective analysis of a Medicaid population. J Med Econ 17: 577–586, 2014 [DOI] [PubMed] [Google Scholar]
- 6.Salmoirago-Blotcher E, Crawford S, Jackson E, Ockene J, Ockene I: Constipation and risk of cardiovascular disease among postmenopausal women. Am J Med 124: 714–723, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kubota Y, Iso H, Tamakoshi A: Bowel movement frequency, laxative use, and mortality from coronary heart disease and stroke among Japanese men and women: The Japan Collaborative Cohort (JACC) study. J Epidemiol 26: 242–248, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Talley NJ, Jones M, Nuyts G, Dubois D: Risk factors for chronic constipation based on a general practice sample. Am J Gastroenterol 98: 1107–1111, 2003 [DOI] [PubMed] [Google Scholar]
- 9.Ramezani A, Raj DS: The gut microbiome, kidney disease, and targeted interventions. J Am Soc Nephrol 25: 657–670, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ibáñez L, Morlans M, Vidal X, Martínez MJ, Laporte JR: Case-control study of regular analgesic and nonsteroidal anti-inflammatory use and end-stage renal disease. Kidney Int 67: 2393–2398, 2005 [DOI] [PubMed] [Google Scholar]
- 11.Michishita R, Matsuda T, Kawakami S, Kiyonaga A, Tanaka H, Morito N, Higaki Y: The association between unhealthy lifestyle behaviors and the prevalence of Chronic Kidney Disease (CKD) in middle-aged and older men. J Epidemiol 26: 378–385, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shirazian S, Radhakrishnan J: Gastrointestinal disorders and renal failure: Exploring the connection. Nat Rev Nephrol 6: 480–492, 2010 [DOI] [PubMed] [Google Scholar]
- 13.Cano AE, Neil AK, Kang JY, Barnabas A, Eastwood JB, Nelson SR, Hartley I, Maxwell D: Gastrointestinal symptoms in patients with end-stage renal disease undergoing treatment by hemodialysis or peritoneal dialysis. Am J Gastroenterol 102: 1990–1997, 2007 [DOI] [PubMed] [Google Scholar]
- 14.Pahl MV, Vaziri ND: The chronic kidney disease - colonic axis. Semin Dial 28: 459–463, 2015 [DOI] [PubMed] [Google Scholar]
- 15.Attaluri A, Jackson M, Valestin J, Rao SS: Methanogenic flora is associated with altered colonic transit but not stool characteristics in constipation without IBS. Am J Gastroenterol 105: 1407–1411, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Quigley EM: The enteric microbiota in the pathogenesis and management of constipation. Best Pract Res Clin Gastroenterol 25: 119–126, 2011 [DOI] [PubMed] [Google Scholar]
- 17.Musso G, Gambino R, Cassader M: Interactions between gut microbiota and host metabolism predisposing to obesity and diabetes. Annu Rev Med 62: 361–380, 2011 [DOI] [PubMed] [Google Scholar]
- 18.Ley RE, Turnbaugh PJ, Klein S, Gordon JI: Microbial ecology: Human gut microbes associated with obesity. Nature 444: 1022–1023, 2006 [DOI] [PubMed] [Google Scholar]
- 19.Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, Wu Y, Schauer P, Smith JD, Allayee H, Tang WH, DiDonato JA, Lusis AJ, Hazen SL: Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 472: 57–63, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tang WH, Hazen SL: The contributory role of gut microbiota in cardiovascular disease. J Clin Invest 124: 4204–4211, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tang WH, Wang Z, Levison BS, Koeth RA, Britt EB, Fu X, Wu Y, Hazen SL: Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N Engl J Med 368: 1575–1584, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rhee EP, Clish CB, Ghorbani A, Larson MG, Elmariah S, McCabe E, Yang Q, Cheng S, Pierce K, Deik A, Souza AL, Farrell L, Domos C, Yeh RW, Palacios I, Rosenfield K, Vasan RS, Florez JC, Wang TJ, Fox CS, Gerszten RE: A combined epidemiologic and metabolomic approach improves CKD prediction. J Am Soc Nephrol 24: 1330–1338, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lekawanvijit S, Kompa AR, Wang BH, Kelly DJ, Krum H: Cardiorenal syndrome: The emerging role of protein-bound uremic toxins. Circ Res 111: 1470–1483, 2012 [DOI] [PubMed] [Google Scholar]
- 24.Schulman G, Berl T, Beck GJ, Remuzzi G, Ritz E, Arita K, Kato A, Shimizu M: Randomized placebo-controlled EPPIC trials of AST-120 in CKD. J Am Soc Nephrol 26: 1732–1746, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hara K, Hirowatari Y, Yoshika M, Komiyama Y, Tsuka Y, Takahashi H: The ratio of plasma to whole-blood serotonin may be a novel marker of atherosclerotic cardiovascular disease. J Lab Clin Med 144: 31–37, 2004 [DOI] [PubMed] [Google Scholar]
- 26.Vikenes K, Farstad M, Nordrehaug JE: Serotonin is associated with coronary artery disease and cardiac events. Circulation 100: 483–489, 1999 [DOI] [PubMed] [Google Scholar]
- 27.Costedio MM, Coates MD, Brooks EM, Glass LM, Ganguly EK, Blaszyk H, Ciolino AL, Wood MJ, Strader D, Hyman NH, Moses PL, Mawe GM: Mucosal serotonin signaling is altered in chronic constipation but not in opiate-induced constipation. Am J Gastroenterol 105: 1173–1180, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Capasso F, Mascolo N, Autore G, Romano V: Laxatives and the production of autacoids by rat colon. J Pharm Pharmacol 38: 627–629, 1986 [DOI] [PubMed] [Google Scholar]
- 29.Coresh J, Turin TC, Matsushita K, Sang Y, Ballew SH, Appel LJ, Arima H, Chadban SJ, Cirillo M, Djurdjev O, Green JA, Heine GH, Inker LA, Irie F, Ishani A, Ix JH, Kovesdy CP, Marks A, Ohkubo T, Shalev V, Shankar A, Wen CP, de Jong PE, Iseki K, Stengel B, Gansevoort RT, Levey AS; CKD Prognosis Consortium : Decline in estimated glomerular filtration rate and subsequent risk of end-stage renal disease and mortality. JAMA 311: 2518–2531, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kovesdy CP, Coresh J, Ballew SH, Woodward M, Levin A, Naimark DM, Nally J, Rothenbacher D, Stengel B, Iseki K, Matsushita K, Levey AS; CKD Prognosis Consortium : Past decline versus current eGFR and subsequent ESRD risk. J Am Soc Nephrol 27: 2447–2455, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sumida K, Molnar MZ, Potukuchi PK, Thomas F, Lu JL, Jing J, Ravel VA, Soohoo M, Rhee CM, Streja E, Kalantar-Zadeh K, Kovesdy CP: Association of slopes of estimated glomerular filtration rate with post-end-stage renal disease mortality in patients with advanced chronic kidney disease transitioning to dialysis. Mayo Clin Proc 91: 196–207, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dukas L, Willett WC, Giovannucci EL: Association between physical activity, fiber intake, and other lifestyle variables and constipation in a study of women. Am J Gastroenterol 98: 1790–1796, 2003 [DOI] [PubMed] [Google Scholar]
- 33.Hsieh C: Treatment of constipation in older adults. Am Fam Physician 72: 2277–2284, 2005 [PubMed] [Google Scholar]
- 34.Dimidi E, Christodoulides S, Fragkos KC, Scott SM, Whelan K: The effect of probiotics on functional constipation in adults: A systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr 100: 1075–1084, 2014 [DOI] [PubMed] [Google Scholar]
- 35.Kovesdy CP, Norris KC, Boulware LE, Lu JL, Ma JZ, Streja E, Molnar MZ, Kalantar-Zadeh K: Association of race with mortality and cardiovascular events in a large cohort of US veterans. Circulation 132: 1538–1548, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gosmanova EO, Lu JL, Streja E, Cushman WC, Kalantar-Zadeh K, Kovesdy CP: Association of medical treatment nonadherence with all-cause mortality in newly treated hypertensive US veterans. Hypertension 64: 951–957, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF III , Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J; CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) : A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.VA Information Resource Center : VIReC Research User Guide: VHA Pharmacy Prescription Data, 2nd Ed., Hines, IL, US Department of Veterans Affairs, Health Services Research and Development Service, VA Information Resource Center, 2008 [Google Scholar]
- 39.Kovesdy CP, Bleyer AJ, Molnar MZ, Ma JZ, Sim JJ, Cushman WC, Quarles LD, Kalantar-Zadeh K: Blood pressure and mortality in U.S. veterans with chronic kidney disease: A cohort study. Ann Intern Med 159: 233–242, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kovesdy CP, Lu JL, Molnar MZ, Ma JZ, Canada RB, Streja E, Kalantar-Zadeh K, Bleyer AJ: Observational modeling of strict vs conventional blood pressure control in patients with chronic kidney disease. JAMA Intern Med 174: 1442–1449, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.US Department of Veterans Affairs: VA Information Resource Center. Available at: http://www.virec.research.va.gov/Resources/Info-About-VA-Data.asp. Accessed December 15, 2014
- 42.Levin A, Stevens PE: Summary of KDIGO 2012 CKD Guideline: Behind the scenes, need for guidance, and a framework for moving forward. Kidney Int 85: 49–61, 2014 [DOI] [PubMed] [Google Scholar]
- 43.Sohn MW, Arnold N, Maynard C, Hynes DM: Accuracy and completeness of mortality data in the Department of Veterans Affairs. Popul Health Metr 4: 2, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.