Abstract
Background
Simultaneous resection of primary colorectal cancer (CRC) and synchronous liver metastases (LM) is gaining interest. We describe management and outcomes of patients undergoing simultaneous resection in the general population.
Methods
All patients with CRC who underwent surgical resection of LM between 2002 and 2009 were identified using the population-based Ontario Cancer Registry and linked electronic treatment records. Synchronous disease was defined as having resection of CRCLM within 12 weeks of surgery for the primary tumour.
Results
During the study period, 1310 patients underwent resection of CRCLM. Of these, 226 (17%) patients had synchronous disease; 100 (44%) had a simultaneous resection and 126 (56%) had a staged resection. For the simultaneous and the staged groups, the mean number of liver lesions resected was 1.6 and 2.3, respectively (p < 0.001); the mean size of the largest lesion was 3.1 and 4.8 cm, respectively (p < 0.001); and the major hepatic resection rate was 21% and 79%, respectively (p < 0.001). Postoperative mortality for simultaneous cases at 90 days was less than 5%. Five-year overall survival and cancer-specific survival for patients with simultaneous resection was 36% (95% confidence interval [CI] 26%–45%) and 37% (95% CI 25%–50%), respectively. Simultaneous resections are common in the general population. A more conservative approach is being adopted for simultaneous resections by limiting the extent of liver resection. Postoperative mortality and long-term survival in this patient population is similar to that reported in other contemporary series.
Conclusion
Compared with a staged approach, patients undergoing simultaneous resections had fewer and smaller liver metastases and underwent less aggressive resections. One-third of these patients achieved long-term survival.
Abstract
Contexte
La résection simultanée des cancers colorectaux primitifs et des métastases hépatiques synchrones suscitent de plus en plus d’intérêt. Nous décrivons la prise en charge et les résultats de patients de la population générale ayant subi une résection simultanée.
Méthodes
Tous les patients atteints d’un cancer colorectal ayant bénéficié d’une résection chirurgicale des métastases hépatiques entre 2002 et 2009 ont été identifiés au moyen du Registre des cas de cancer de l’Ontario en population générale et des dossiers électroniques associés sur le traitement. La maladie synchrone a été définie comme le fait d’avoir subi une chirurgie de résection des métastases hépatiques du cancer colorectal dans les 12 semaines de la chirurgie de la tumeur primitive.
Résultats
Pendant la période de l’étude, 1310 patients ont subi une résection des métastases hépatiques du cancer colorectal. Sur ce nombre, 226 (17 %) patients présentaient une maladie synchrone; 100 (44 %) patients ont subi une résection simultanée et 126 (56 %) patients ont subi une résection en 2 temps. Dans les groupes des résections simultanées et des résections en 2 temps, le nombre moyen de lésions hépatiques réséquées était de 1,6 et de 2,3, respectivement (p < 0,001); la taille moyenne de la lésion la plus importante était de 3,1 et de 4,8 cm, respectivement (p < 0,001) et le taux de résection hépatique majeure était de 21 % et de 79 %, respectivement (p < 0,001). La mortalité postopératoire après résection simultanée à 90 jours était inférieure à 5 %. La survie globale à 5 ans et la survie par cause des patients avec résection simultanée étaient de 36 % (intervalle de confiance [IC] de 95 %, 26 %–45 %) et de 37 % (IC 95 %, 25 %–50 %), respectivement. Les résections simultanées sont courantes au sein de la population générale. On commence à adopter une approche plus conservatrice pour les résections simultanées en limitant l’étendue de la résection hépatique. La mortalité postopératoire et la survie à long terme de cette population de patients sont semblables à celles signalées dans d’autres séries récentes.
Conclusion
Comparativement à l’approche en 2 temps, les patients avec résections simultanées présentaient moins de métastases hépatiques et des métastases de plus petite taille, et les résections pratiquées étaient moins agressives. Le tiers de ces patients ont obtenu une survie à long terme.
The management of patients with primary colorectal cancer (CRC) and synchronous liver metastases (LM) remains a challenge for the multidisciplinary team. Several studies have demonstrated inferior survival for these patients compared with those who present with metachronous disease.1–4 For patients with resectable CRC with synchronous disease, consideration must be given to surgical resection of the primary cancer, the hepatic metastases, administration of systemic chemotherapy and, in the case of those with rectal cancer, radiotherapy. Although each of these treatments is critical in the comprehensive management of these patients, the optimal timing and sequence of each modality remains controversial.
Complete surgical removal of the primary tumour and all liver metastases is the only potentially curative treatment option. This can be performed as a single operation with combined colorectal and liver resection or as a staged approach. The traditional approach has been to resect the primary tumour, followed often by systemic chemotherapy, then liver resection in the absence of disease progression. More recently, with improvements in perioperative care and anesthesia as well as advances in imaging and liver surgery, this approach has been challenged with several studies demonstrating comparable safety and outcome with a simultaneous surgical strategy.5–12 Combing both resection of the primary malignancy and the hepatic metastases avoids the morbidity of a second major operation, theoretically lowers the risk of disease dissemination and allows for timely completion of adjuvant therapies. Although a simultaneous approach appears feasible, the literature supporting this strategy is derived largely from high-volume single centres and multi-institutional case series. Therefore, it is unclear to what extent this surgical strategy is being performed in the general population and whether comparable outcomes are being realized. Population-based studies can be useful in addressing these questions by describing practice and outcomes achieved in routine clinical practice. To our knowledge, there are only 2 population-based studies concerning simultaneous resection for synchronous CRCLM. One study13 used Medicare data to examine long-term outcomes in all patients undergoing hepatic resection for CRCLM. The analysis was limited to patients aged 65 years or older, and patients undergoing simultaneous colon and liver resection were a small subgroup of the larger cohort. The other study14 used the National Inpatient Sample and provided data on postoperative outcomes only; it did not address any long-term outcomes. We performed a population-based study to describe management of synchronous CRCLM and the short- and long-term outcomes associated with simultaneous resection in routine clinical practice.
Methods
Study design and population
This is a population-based, retrospective cohort study to describe the management and outcome of resected CRCLM in the Canadian province of Ontario, Canada. Ontario has a population of approximately 13.5 million people and a single-payer universal health insurance program. The study population included all patients with CRC who underwent liver resection between 2002 and 2009. We used the Ontario Cancer Registry (OCR) to identify all incident cases of CRC in Ontario diagnosed between 1996 and 2009. We then identified all cases of liver resection performed between 2002 and 2009. The OCR does not capture diagnoses of second CRCs. As such, patients who underwent liver resection more than 6 years after the initial CRC diagnosis were excluded, because those cases would likely represent recurrence of a second primary cancer. Patients with histology other than adenocarcinoma were excluded. To minimize misclassification of liver metastases we also excluded patients with a second primary liver, biliary or pancreatic cancer. Details on the extent of liver metastases was not available in the existing data sources; for this reason we obtained surgical pathology reports for all potentially eligible patients. Patients with evidence of metastatic CRC as per the liver resection pathology report were included. Synchronous disease was defined as having resection of CRCLM within 12 weeks of surgery for the primary tumour. The research ethics board of Queen’s University approved our study protocol.
Data sources and linkage
The OCR is a passive, population-based cancer registry that captures diagnostic and demographic information on at least 98% of all incident cases of cancer in the province of Ontario.15 It also provides information about vital status and cause of death. Records of hospitalization from the Canadian Institute for Health Information (CIHI) provided information about surgical interventions; these records are known to be complete.16 Provincial physician billing records from the Ontario Health Insurance Plan (OHIP), treatment records (Activity Level Reporting [ALR]) from regional cancer centres and provincial records of chemotherapy delivery (New Drug Funding Program [NDFP] and Ontario Drug Benefits [ODB]) were used to identify chemotherapy use. Incident cases of CRC identified from the OCR were linked to other electronic administrative health databases at the Institute of Clinical and Evaluative Sciences (ICES). We obtained surgical pathology reports from the OCR. A team of trained data abstractors reviewed the pathology reports and entered information about extent of disease and surgical procedure into an electronic database.
Measures and outcomes
We classified comorbidity using the Charlson Index, modified for administrative data based on all noncancer diagnoses recorded during any hospital admission within the 5 years before surgery.17 Preoperative chemotherapy was defined as chemotherapy given within 16 weeks before resection of CRCLM; postoperative chemotherapy was defined as treatment initiated within 16 weeks after surgery for CRCLM. Postoperative mortality for staged cases was determined from the date of liver resection. Length of stay for staged group was based on the sum of colon and liver resection. Overall (OS) and cancer-specific survival (CSS) were determined from the time of liver resection. To account for possible cause of death miscoding, CSS included death from any cancer. Complete information about vital status in the OCR was available up to Dec. 31, 2012; cause of death was available up to Dec. 31, 2010.
Statistical analysis
We used the χ2 test to compare proportions between study groups. We determined OS and CSS using the Kaplan–Meier method. Factors associated with OS/CSS were evaluated using the Cox proportional hazards regression model. We considered results to be significant at p < 0.05. All analyses were performed using SAS software version 9.3 (SAS Institute).
Results
Study population
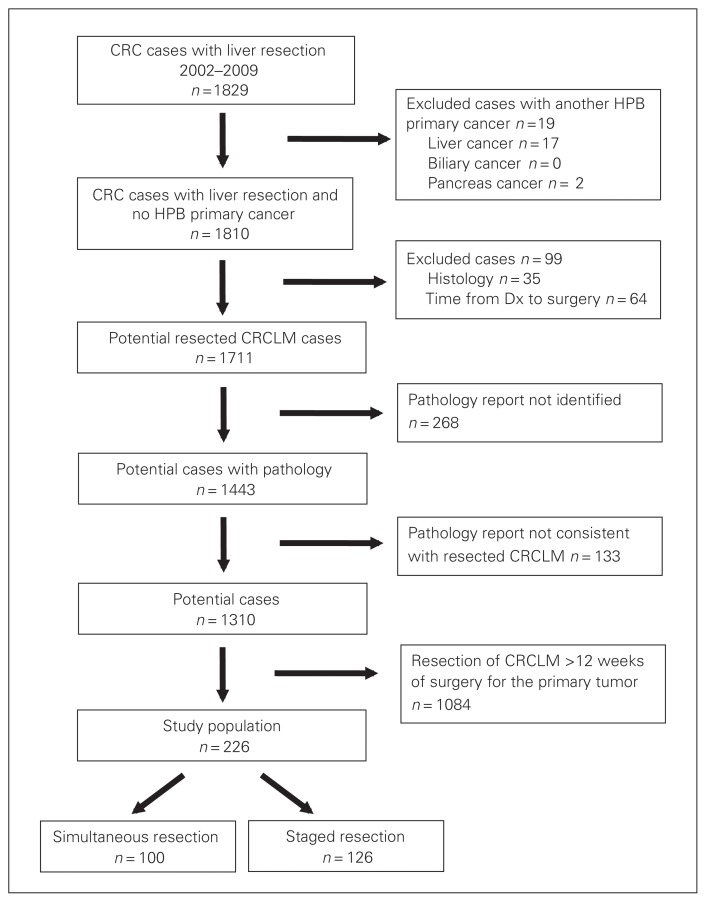
Using linked administrative data sets, we identified 1711 potentially eligible patients who underwent surgical resection of CRCLM during the study period (Fig. 1). Surgical pathology reports were available for 1443 (84%) patients; 133 (9%) of these reports indicated that the procedure did not include resection of CRCLM. Of the remaining 1310 potential cases, 226 (17%) patients were identified as having synchronous disease; 100 (44%) underwent a simultaneous resection, whereas 126 (56%) had a staged resection. Characteristics of patients with simultaneous and staged resections of CRCLM are shown in Table 1. Age, sex and comorbidity were comparable between the groups. Patients undergoing simultaneous resections received more neoadjuvant chemotherapy (p = 0.017), but both groups had similar rates of adjuvant chemotherapy (p = 0.09).
Fig. 1.
Identification of patients with colorectal cancer (CRC) who underwent resection of liver metastases (LM) in Ontario between 2002 and 2009. Dx = diagnosis; HPB = hepatopancreatobiliary.
Table 1.
Characteristics of patients with synchronous colorectal cancer liver metastases who underwent hepatic resection in Ontario between 2002 and 2009 (n = 226)
| Characteristic | Group; no. (%)* | ||
|---|---|---|---|
| All cases n = 226 |
Simultaneous n = 100 |
Staged n = 126 |
|
| Age, yr | |||
| 20–49 | 37 (16) | 15 (15) | 22 (17) |
| 50–59 | 61 (27) | 24 (24) | 37 (29) |
| 60–69 | 80 (35) | 36 (36) | 44 (35) |
| 70–79 | 35 (15) | 16 (16) | < 20 (< 20) |
| ≥ 80 | 13 (6) | 9 (9) | < = 5 (< 5) |
| Age, mean [range], yr | 61 [20–87] | 62 [22–87] | 60 [20–83] |
| Sex | |||
| Male | 124 (55) | 54 (54) | 70 (56) |
| Female | 102 (45) | 46 (46) | 56 (44) |
| Chemotherapy | |||
| Neoadjuvant | 47 (21) | 28 (28) | 19 (15) |
| Between colon and liver | — | — | 7 (6) |
| Adjuvant | 145 (64) | 58 (58) | 87 (69) |
| Charlson Comorbidity Index score | |||
| 0 | 195 (86) | 87 (87) | 108 (86) |
| 1 | 23 (10) | < 10 (< 10)† | < 15 (< 15)† |
| ≥ 2 | 8 (4) | < 6 (< 5)† | < 6 (< 5)† |
Unless indicated otherwise.
As per institutional policy cells < 6 cases are suppressed.
Surgical and pathological characteristics
Details regarding the surgical procedures and pathological findings are shown in Table 2. Patients undergoing simultaneous resections were less likely to undergo a major liver resection (21% v. 79%, p < 0.001). The most frequent liver procedure performed in the simultaneous cohort was a wedge resection; among staged cases the most common procedure was a lobectomy. The size of the largest lesion (3.1 cm v. 4.8 cm, p < 0.001) was significantly smaller and the number of metastases resected (1.6 v. 2.3, p < 0.001) was significantly lower in the simultaneous cohort.
Table 2.
Surgical procedure and pathological characteristics of patients with synchronous colorectal cancer liver metastases who underwent hepatic resection in Ontario between 2002 and 2009 (n = 226)
| Characteristic | Group; no. (%)* | |
|---|---|---|
| Simultaneous n = 100 |
Staged n = 126 |
|
| Extent of liver resection | ||
| Major (≥ 3 segments) | 21 (21) | 99 (79) |
| Minor (< 3 segments) | 78 (79) | 26 (21) |
| Type of liver resection† | ||
| Wedge | 77 (77) | 43 (34) |
| Bisegmentectomy | 20 (17) | < 12 (< 10) |
| Lobectomy | 18 (15) | 77 (49) |
| Extended lobectomy | < 6 (< 6)§§ | 22 (14) |
| Unstated | < 6 (< 6)§§ | < 6 (< 6)§§ |
| Liver metastases | ||
| No. of lesions resected‡ | ||
| Mean [range] | 1.6 [1–12] | 2.3 [1–13] |
| Median [IQR] | 1 [1–2] | 2 [1–3] |
| Largest liver lesion resected, cm§ | ||
| Mean [range] | 3.1 [0.3–22] | 4.8 [0.8–22] |
| Median [IQR] | 2 [2–4] | 4 [3–6] |
| Margin status¶ | ||
| R0 | 75 (89) | 112 (91) |
| R1 | < 10 (< 10)§§ | 11 (9) |
| R2 | < 6 (< 5)§§ | 0 (0) |
| Primary colorectal cancer** | ||
| T stage†† | ||
| T1–T2 | < 6 (5) | 11 (9) |
| T3–T4 | 60 (60) | 63 (50) |
| N stage | ||
| N+ | 56 (56) | 61 (48) |
| N− | < 25 (< 25)§§ | 21 (17) |
| NX | < 6 (< 3)§§ | 0 (0) |
| Laterality‡‡ | ||
| Right | 29 (29) | 11 (9) |
| Left | 51 (51) | 70 (56) |
IQR = interquartile range.
Unless otherwise indicated.
A patient may have had more than 1 resection; the denominator is the total number of resections.
Nine patients excluded owing to missing data.
Eight patients excluded owing to missing data.
Twenty-three patients excluded owing to missing data.
Fewer than 6 patients excluded owing to missing data.
Margin status for the liver resection was available for 84/100 (84%) of the
simultaneous cases and 123/126 (98%) of the staged cases.
Pathological data for the primary tumour were available for 80/100 (80%) of the simultaneous cases and 82 /126 (65%) of the staged cases.
As per institutional policy cells (< 6 cases) are suppressed.
Surgical and pathological data for the primary tumour were available for 80 (80%) patients in the simultaneous group and 81 (64%) patients in the staged group. The simultaneous cohort was more likely to have right-sided primary tumours (29% v. 9%, p = 0.001), had more advanced primary tumours than staged patients with higher T3/4-stage (60% v. 50%, p = 0.19) and had more node-positive tumours (56% v. 48%, p = 0.039).
Table 3 describes the type of hepatic resection and the location of the primary CRC tumour for patients in the simultaneous group. The most frequent procedure performed was a wedge resection regardless of the location of the primary malignancy. Major liver resections were performed more commonly for right-sided than left-sided primary cancers, and none were completed for rectal primary cancers.
Table 3.
Type of hepatic resection and location of primary colorectal tumour of patients with simultaneous resection of primary colorectal cancer and liver metastases in Ontario between 2002 and 2009, % (n = 100)*
| Resection | Right colon | Left colon | Rectal |
|---|---|---|---|
| Major resection | 38 | 14 | 0 |
| Minor resection | 24 | 14 | 18 |
| Wedge resection | 38 | 71 | 82 |
| Total | 100 | 100 | 100 |
Data available for 79 cases
Outcomes
Short- and long-term outcomes of the study population are shown in Table 4. For illustrative purposes, outcomes are also shown for patients who underwent staged resection. Patients undergoing a single simultaneous resection had a significantly shorter mean length of stay than patients requiring 2 separate procedures in the staged group (13 v. 16 d, p < 0.001). Median OS and CSS for the simultaneous patients was 40 and 43 months, respectively; 5-year OS and CSS was 36% (95% confidence interval [CI] 26%–45%) and 37% (95% CI 25%–50%), respectively. Factors associated with survival for patients treated with a simultaneous approach to CRCLM are shown in Table 5. Age and extent of surgical resection were associated with OS and CSS.
Table 4.
Short- and long-term outcomes of patients with synchronous colorectal cancer liver metastases who underwent hepatic resection in Ontario between 2002 and 2009 (n = 226)
| Outcome | Group; no. (%)* | |
|---|---|---|
| Simultaneous n = 100 |
Staged n = 126 |
|
| 30-d mortality† | < 6 (< 2)§ | < 6 (< 3)§ |
| 90-d mortality† | < 6 (< 5)§ | < 6 (< 3)§ |
| Length of stay, mean/median, d‡ | 13/9 | 16/14 |
| OS, median [IQR], mo | 40 [32–48] | 61 [46–85] |
| CSS, median [IQR], mo | 43 [35–53] | 53 [45–73] |
| 5-year OS (95% CI) | 36% (26%–45%) | 51% (41%–60%) |
| 5-year CSS (95% CI) | 37% (25%–50%) | 46% (35%–57%) |
CI = confidence interval; CSS = cancer-specific survival; IQR = interquartile range; OS = overall survival.
Unless indicated otherwise.
Postoperative mortality was calculated based on the date of liver resection for the synchronous and staged groups.
Length of stay for the staged group was calculated based on length of stay for colon resection and liver resection.
As per institutional policy cells (< 6 cases) are suppressed.
Table 5.
Factors associated with CSS and OS among patients with synchronous colorectal cancer liver metastases treated with simultaneous resection in Ontario between 2002 and 2009 (n = 100*)
| Covariate | Multivariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
|
|
|
|||||
| 5-year CSS | HR (95%CI) | p value | 5-year OS | HR (95%CI) | p value | |
| Patient-related | ||||||
|
| ||||||
| Age, yr | 0.035 | 0.005 | ||||
|
| ||||||
| < 65 | 55% | Ref | 50% | Ref | ||
|
| ||||||
| 65–74 | 21% | 2.18 (1.04–4.56) | 28% | 1.95 (1.04–3.64) | ||
|
| ||||||
| ≥ 75 | 0% | 2.79 (1.12–6.96) | 0% | 3.37 (1.56–7.29) | ||
|
| ||||||
| Charlson Comorbidity Index score | 0.33 | 0.49 | ||||
|
| ||||||
| 0 | 41% | Ref | 39% | Ref | ||
|
| ||||||
| ≥ 1 | 0% | 1.60 (0.63–4.08) | 0% | 1.31 (0.60–2.86) | ||
|
| ||||||
| Disease-related | ||||||
|
| ||||||
| Mean no. of lesions | 0.58 | 0.26 | ||||
|
| ||||||
| 1 | 40% | Ref | 40% | Ref | ||
|
| ||||||
| > 1 | 36% | 1.23 (0.59–2.54) | 29% | 1.38 (0.79–2.42) | ||
|
| ||||||
| Mean size largest lesion, cm | 0.13 | 0.15 | ||||
|
| ||||||
| < 5 | 36% | Ref | 36% | Ref | ||
|
| ||||||
| ≥ 5 | 37% | 2.03 (0.81–5.10) | 29% | 1.74 (0.82–3.67) | ||
|
| ||||||
| Treatment-related | ||||||
|
| ||||||
| Extent of surgical resection | 0.038 | 0.027 | ||||
|
| ||||||
| Minor (< 3 segments) | 32% | Ref | 30% | Ref | ||
|
| ||||||
| Major ( > 3 segments) | 57% | 0.31 (0.10–0.94) | 57% | 0.38 (0.16–0.90) | ||
CI = confidence interval; CSS = cancer-specific survival; HR = hazard ratio; OS = overall survival.
Seven cases excluded from analysis owing to unavailable data (number lesions, lesion size, extent of surgical resection).
Discussion
Our study provides insight into the management and outcomes of patients in the general population with synchronous CRCLM who undergo simultaneous resection. We found that simultaneous resections are common in routine clinical practice. Compared with those who underwent a staged approach, patients who underwent simultaneous resections had fewer and smaller liver metastases, and received less aggressive resections. Postoperative mortality in the simultaneous resection group was in an acceptable range, and one-third of patients achieved long-term survival.
To our knowledge, there is only 1 other population-based report specific to patients undergoing simultaneous resection of synchronous CRCLM. Abbott and colleagues14 used the National Inpatient Sample to evaluate short-term outcomes in a contemporary cohort to determine if simultaneous resection is a safe approach. Mortality for simultaneous resections was 3.5%, and average length of stay was 10.9 days, which is comparable to our findings (< 5% and 13 d, respectively). Consistent with our findings, Abbott and colleagues found that the majority of patients who underwent a simultaneous procedure underwent a right colectomy and liver wedge resection. These data suggest that a more conservative approach is being adopted for simultaneous resections in the general population by limiting the extent of liver resection.
The rationale for performing colorectal and liver resections separately relates to the perceived increased perioperative risk with simultaneous resections. Indeed, earlier studies reported higher mortality with combined colon and liver resections.11,18,19 However, these reports are based on procedures performed in the 1990s, when liver resections were not as safe as they are now. In fact, more recent studies consistently report comparable mortality between simultaneous and staged resections7–9,12,20–24 ranging from 0% to 3.5%. In the present study, 30- and 90-day mortality from simultaneous resection was < 2% and < 5%, respectively, which is similar to that reported in most contemporary series.
A simultaneous approach has the advantage of shorter overall length of stay in hospital. This has been shown consistently in the literature7,9,12,20,22,24–26 and in our study. Even in studies where morbidity was significantly higher in the simultaneous group, the overall length of stay in this group was still shorter.27 The magnitude of this advantage may not be as marked nowadays given the more recent implementation of “fast-track surgery” and “enhanced recovery after surgery” in colorectal surgery.
Several reports have described long-term survival of patients who undergo simultaneous resection of primary CRC and synchronous liver metastases.5,6,8,12,20,21,25,26,28 In these series, 5-year OS for simultaneous resections ranged from 29% to 55%; our results suggest that comparable outcomes are achieved in routine practice. We report outcomes of patients who underwent staged resections for illustrative purposes only. We purposefully did not undertake comparative analyses of outcome for a simultaneous versus staged approach, because such an analysis would be fraught with several methodological limitations, which could bias the results in both directions. For instance, a staged approach offers the advantage of time between the 2 operations to allow for sub-clinical metastases to become detectable, either within the liver or extrahepatically. Theoretically, this would result in better tumour clearance at the time of hepatic resection and would allow for better patient selection by identifying patients who would not benefit from hepatic resection. Based on this concept, comparative analyses might suggest a survival benefit to a staged approach. Conversely, as seen in our data, patients undergoing a staged resection may have more advanced liver metastases, precluding them from consideration of a simultaneous approach. This selection bias may result in comparative outcomes that favour a simultaneous approach. Another possibility is that patients who underwent simultaneous resections actually had more advanced liver disease and that the initial simultaneous resection represented the first of 2 planned liver resections for complete tumour clearance. For these reasons we feel that comparative survival analyses of simultaneous versus staged resection of CRCLM would not provide meaningful or accurate information.
Limitations
Although our study provides data regarding the management and outcome of simultaneous compared with staged resections in a contemporary population-based cohort, certain methodological limitations require mention. Although the electronic data sources used in this study describe general aspects of disease, treatment and outcome for all patients in the province, detailed information related to postoperative complications and various factors that influenced surgeons to select a simultaneous approach versus a staged approach for individual patients is not available. In addition, given the time-based definition of synchronous disease, some patients who underwent a staged resection would have been inadvertently excluded from this analysis because their liver resection was performed 12 weeks after resection of the primary tumour. We also do not have detailed information regarding the burden of metastatic disease, and were were only able to describe the extent of disease based on what was resected. Despite these limitations a major strength of this study is the large, unselected study population without the single-institution biases based on referral patterns and surgical volume. As a result, the outcomes more accurately reflect what is being achieved in routine clinical practice.
Conclusion
Simultaneous resection of synchronous CRCLM is common in routine clinical practice. Compared with patients who underwent a staged approach, patients who underwent simultaneous resections had fewer and smaller liver metastases and received less aggressive resections. Simultaneous resection of the primary tumour and CRCLM in the general population appears to be safe, and a substantial proportion of patients will achieve long-term survival.
Acknowledgements
Parts of this material are based on data and information provided by Cancer Care Ontario. However, the analysis, conclusions, opinions and statements expressed herein are those of the authors and not necessarily those of Cancer Care Ontario. This study was supported by the Institute for Clinical Evaluative Sciences (ICES), which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred. C. Booth had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Previous communication of this manuscript was presented as a podium presentation at the 2014 Canadian Surgery Forum in Vancouver, BC. The abstract was awarded the Editor’s Choice Award.
Competing interests: None declared.
Contributors: S. Nanji, W. Mackillop and C. Booth designed the study. All authors acquired and analyzed the data. S. Nanji and C. Booth wrote the article, which all authors reviewed and approved for publication.
Funding: Dr. Booth is supported as a Canada Research Chair in Population Cancer Care. This work was also supported by the Canada Foundation for Innovation and Queen’s University Department of Surgery.
References
- 1.Fong Y, Fortner J, Sun RL, et al. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg. 1999;230:309–18. doi: 10.1097/00000658-199909000-00004. 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schlag P, Hohenberger P, Herfarth C. Resection of liver metastases in colorectal cancer–competitive analysis of treatment results in synchronous versus metachronous metastases. Eur J Surg Oncol. 1990;16:360–5. [PubMed] [Google Scholar]
- 3.Nordlinger B, Guiguet M, Vaillant JC, et al. Surgical resection of colorectal carcinoma metastases to the liver. A prognostic scoring system to improve case selection, based on 1568 patients. Association Francaise de Chirurgie. Cancer. 1996;77:1254–62. [PubMed] [Google Scholar]
- 4.Tsai MS, Su YH, Ho MC, et al. Clinicopathological features and prognosis in resectable synchronous and metachronous colorectal liver metastasis. Ann Surg Oncol. 2007;14:786–94. doi: 10.1245/s10434-006-9215-5. [DOI] [PubMed] [Google Scholar]
- 5.Yin Z, Liu C, Chen Y, et al. Timing of hepatectomy in resectable synchronous colorectal liver metastases (SCRLM): Simultaneous or delayed? Hepatology. 2013;57:2346–57. doi: 10.1002/hep.26283. [DOI] [PubMed] [Google Scholar]
- 6.Brouquet A, Mortenson MM, Vauthey JN, et al. Surgical strategies for synchronous colorectal liver metastases in 156 consecutive patients: classic, combined or reverse strategy? J Am Coll Surg. 2010;210:934–41. doi: 10.1016/j.jamcollsurg.2010.02.039. [DOI] [PubMed] [Google Scholar]
- 7.Martin RC, II, Augenstein V, Reuter NP, et al. Simultaneous versus staged resection for synchronous colorectal cancer liver metastases. J Am Coll Surg. 2009;208:842–50. doi: 10.1016/j.jamcollsurg.2009.01.031. 2. [DOI] [PubMed] [Google Scholar]
- 8.Lyass S, Zamir G, Matot I, et al. Combined colon and hepatic resection for synchronous colorectal liver metastases. J Surg Oncol. 2001;78:17–21. doi: 10.1002/jso.1117. [DOI] [PubMed] [Google Scholar]
- 9.Martin R, Paty P, Fong Y, et al. Simultaneous liver and colorectal resections are safe for synchronous colorectal liver metastasis. J Am Coll Surg. 2003;197:233–41. doi: 10.1016/S1072-7515(03)00390-9. 2. [DOI] [PubMed] [Google Scholar]
- 10.Weber JC, Bachellier P, Oussoultzoglou E, et al. Simultaneous resection of colorectal primary tumour and synchronous liver metastases. Br J Surg. 2003;90:956–62. doi: 10.1002/bjs.4132. [DOI] [PubMed] [Google Scholar]
- 11.Tanaka K, Shimada H, Matsuo K, et al. Outcome after simultaneous colorectal and hepatic resection for colorectal cancer with synchronous metastases. Surgery. 2004;136:650–9. doi: 10.1016/j.surg.2004.02.012. [DOI] [PubMed] [Google Scholar]
- 12.Chua HK, Sondenaa K, Tsiotos GG, et al. Concurrent vs. staged colectomy and hepatectomy for primary colorectal cancer with synchronous hepatic metastases. Dis Colon Rectum. 2004;47:1310–6. doi: 10.1007/s10350-004-0586-z. [DOI] [PubMed] [Google Scholar]
- 13.Robertson DJ, Stukel TA, Gottlieb DJ, et al. Survival after hepatic resection of colorectal cancer metastases: a national experience. Cancer. 2009;115:752–9. doi: 10.1002/cncr.24081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Abbott AM, Parsons HM, Tuttle TM, et al. Short-term outcomes after combined colon and liver resection for synchronous colon cancer liver metastases: a population study. Ann Surg Oncol. 2013;20:139–47. doi: 10.1245/s10434-012-2515-z. [DOI] [PubMed] [Google Scholar]
- 15.Clarke EAML, Krieger N. Cancer registration in Ontario: a computer approach. Lyon (France): IARC; 1991. [PubMed] [Google Scholar]
- 16.Williams JYW. A summary of studies on the quality of health care administrative databases in Canada The ICES Practice Atlas. ed 2. Ottawa (ON): ICES; 1996. [Google Scholar]
- 17.Deyo RACD, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45:613–9. doi: 10.1016/0895-4356(92)90133-8. [DOI] [PubMed] [Google Scholar]
- 18.Bolton JS, Fuhrman GM. Survival after resection of multiple bilobar hepatic metastases from colorectal carcinoma. Ann Surg. 2000;231:743–51. doi: 10.1097/00000658-200005000-00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Scheele J. Hepatectomy for liver metastases. Br J Surg. 1993;80:274–6. doi: 10.1002/bjs.1800800302. [DOI] [PubMed] [Google Scholar]
- 20.Yan TD, Chu F, Black D, et al. Synchronous resection of colorectal primary cancer and liver metastases. World J Surg. 2007;31:1496–501. doi: 10.1007/s00268-007-9085-4. [DOI] [PubMed] [Google Scholar]
- 21.Turrini O, Viret F, Guiramand J, et al. Strategies for the treatment of synchronous liver metastasis. Eur J Surg Oncol. 2007;33:735–40. doi: 10.1016/j.ejso.2007.02.025. [DOI] [PubMed] [Google Scholar]
- 22.Capussotti L, Ferrero A, Vigano L, et al. Major liver resections synchronous with colorectal surgery. Ann Surg Oncol. 2007;14:195–201. doi: 10.1245/s10434-006-9055-3. [DOI] [PubMed] [Google Scholar]
- 23.de Haas RJ, Adam R, Wicherts DA, et al. Comparison of simultaneous or delayed liver surgery for limited synchronous colorectal metastases. Br J Surg. 2010;97:1279–89. doi: 10.1002/bjs.7106. [DOI] [PubMed] [Google Scholar]
- 24.Luo Y, Wang L, Chen C, et al. Simultaneous liver and colorectal resections are safe for synchronous colorectal liver metastases. J Gastrointest Surg. 2010;14:1974–80. doi: 10.1007/s11605-010-1284-x. [DOI] [PubMed] [Google Scholar]
- 25.Hillingsø JG, Wille-Jorgensen P. Staged or simultaneous resection of synchronous liver metastases from colorectal cancer — a systematic review. Colorectal Dis. 2009;11:3–10. doi: 10.1111/j.1463-1318.2008.01625.x. [DOI] [PubMed] [Google Scholar]
- 26.Chen J, Li Q, Wang C, et al. Simultaneous vs. staged resection for synchronous colorectal liver metastases: a metaanalysis. Int J Colorectal Dis. 2011;26:191–9. doi: 10.1007/s00384-010-1018-2. [DOI] [PubMed] [Google Scholar]
- 27.Reddy SK, Pawlik TM, Zorzi D, et al. Simultaneous resections of colorectal cancer and synchronous liver metastases: a multi-institutional analysis. Ann Surg Oncol. 2007;14:3481–91. doi: 10.1245/s10434-007-9522-5. [DOI] [PubMed] [Google Scholar]
- 28.Thelen A, Jonas S, Benckert C, et al. Simultaneous versus staged liver resection of synchronous liver metastases from colorectal cancer. Int J Colorectal Dis. 2007;22:1269–76. doi: 10.1007/s00384-007-0286-y. [DOI] [PubMed] [Google Scholar]