Abstract
Rhodoferax antarcticus is an Antarctic purple nonsulfur bacterium and the only characterized anoxygenic phototroph that grows best below 20 °C. We present here a high-quality draft genome of Rfx. antarcticus strain ANT.BRT, isolated from an Antarctic microbial mat. The circular chromosome (3.8 Mbp) of Rfx. antarcticus has a 59.1% guanine + cytosine (GC) content and contains 4036 open reading frames. In addition, the bacterium contains a sizable plasmid (198.6 kbp, 48.4% GC with 226 open reading frames) that comprises about 5% of the total genetic content. Surprisingly, genes encoding light-harvesting complexes 1 and 3 (LH1 and LH3), but not light-harvesting complex 2 (LH2), were identified in the photosynthesis gene cluster of the Rfx. antarcticus genome, a feature that is unique among purple phototrophs. Consistent with physiological studies that showed a strong capacity for nitrogen fixation in Rfx. antarcticus, a nitrogen fixation gene cluster encoding a molybdenum-type nitrogenase was present, but no alternative nitrogenases were identified despite the cold-active phenotype of this phototroph. Genes encoding two forms of ribulose 1,5-bisphosphate carboxylase/oxygenase were present in the Rfx. antarcticus genome, a feature that likely provides autotrophic flexibility under varying environmental conditions. Lastly, genes for assembly of both type IV pili and flagella are present, with the latter showing an unusual degree of clustering. This report represents the first genomic analysis of a psychrophilic anoxygenic phototroph and provides a glimpse of the genetic basis for maintaining a phototrophic lifestyle in a permanently cold, yet highly variable, environment.
Keywords: purple anoxygenic phototroph, photosynthesis gene cluster, light-harvesting complex, psychrophile, Antarctica, Rhodoferax antarcticus, nitrogen fixation, nitrogenase
1. Introduction
Anoxygenic phototrophic bacteria are widespread in nature, and within this group, the purple nonsulfur bacteria (PNB) are by far the most metabolically diverse. Species of PNB are either Alpha- or Betaproteobacteria and are noted for their capacity to grow both phototrophically (anoxic/light) and chemotrophically (oxic/dark). This broad metabolic diversity allows PNBs to adjust their metabolism to fit available conditions and resources in a wide variety of habitats [1].
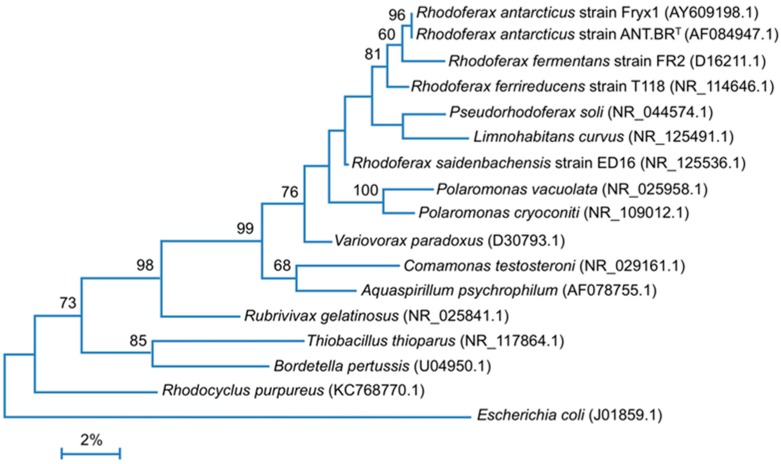
PNBs have been isolated from several extreme environments, including hot, cold, acidic, alkaline, and hypersaline [1,2]. The success of PNBs in these harsh habitats infers that they have evolved important biochemical modifications to support photosynthesis under stressful conditions. Rhodoferax antarcticus, one of four species of the genus and a member of the Betaproteobacteria (Figure 1), is the first purple bacterium isolated from a permanently cold environment, a microbial mat on Ross Island, McMurdo, Antarctica [3] (Table 1). The organism is a small curved rod, highly motile by flagellar means, and contains bacteriochlorophyll (Bchl) a. Rfx. antarcticus grows at 0 °C and optimally near 15 °C, major tenets of psychrophiles [4]. A second and phenotypically distinct strain of Rfx. antarcticus, strain Fryx1 (Figure 1), was isolated from the water column of the permanently ice-covered Lake Fryxell, McMurdo Dry Valleys [5].
Figure 1.
The 16S rRNA gene phylogenetic tree of Rfx. antarcticus strain ANT.BRT and related Betaproteobacteria with Escherichia coli (Gammaproteobacteria) as the outgroup. rRNA gene sequences were obtained from the National Center for Biotechnology Information (NCBI) GenBank database [6] and aligned with ClustalW using Mega7 [7], with a final data set of 1380 nucleotides. Phylogenetic analysis was conducted using the maximum likelihood method in conjunction with the Jukes–Cantor correction [8]. Bootstrap values above 50 (100 replicates) are shown at their respective nodes.
Table 1.
Classification and general features of Rhodoferax antarcticus str. ANT.BRT *.
| Property | Term |
|---|---|
| Classification | Domain: Bacteria |
| Phylum: Proteobacteria | |
| Class: Betaproteobacteria | |
| Order: Burkholderiales | |
| Family: Comamonadaceae | |
| Genus: Rhodoferax | |
| Species: Rhodoferax antarcticus | |
| Type strain: ANT.BRT (ATCC 700587; DSMZ 24876) | |
| Gram stain | Negative |
| Cell shape | Vibrio to spirillum |
| Motility | Highly motile |
| Endosporulation | Non-endospore forming |
| Temperature range | 0–25 °C |
| Optimum temperature | 15–18 °C |
| pH range; Optimum | 6–8; 7 |
| Carbon sources | Acetate, pyruvate, lactate, succinate, malate, fumarate, glucose, fructose, sucrose, citrate, aspartate |
| Habitat | Algal–bacterial microbial mat |
| Salinity | 0%–2% NaCl (w/v) |
| Oxygen requirement | Facultative aerobe |
| Biotic relationship | Free-living |
| Pathogenicity | Non-pathogenic |
| Geographic location | Cape Royds, Ross Island, Antarctica |
| Sample collection | December, 1993 |
| Latitude | 77.55° S |
| Longitude | 166.16° E |
| Altitude | 20 m |
* Data are adapted from Madigan et al. [3].
Genome sequences are available for several PNBs [9]. However, no genome sequence has been available for a purple bacterium that thrives in constantly cold conditions. The genetic blueprint of such an organism could begin to reveal how photocomplexes and related photosynthetic machinery are altered to function optimally in the cold. To explore these questions, we report here an analysis of the genome sequence of Rfx. antarcticus strain ANT.BRT. Our results focus on four functional gene sets where this organism shows genomic peculiarities compared with the genomes of PNBs that thrive in temperate environments, peculiarities that may have relevance for the ecology of this Antarctic phototroph.
2. Materials and Methods
Rhodoferax antarcticus strain ANT.BRT was isolated from a microbial mat in a pond near Cape Royds, Ross Island, Antarctica [3] (Table 1). A single colony was grown anaerobically and total DNA was isolated using proteinase K treatment followed by phenol extraction. Genome sequencing was performed using a random shotgun approach. Sequence reads were generated with three technologies to improve quality, aid assembly, and correct for systematic error introduced by any single method. Nearly 65 million paired-end reads were generated on the Illumina HiSeq platform resulting in an estimated 1539-fold sequence coverage of the chromosome (~3.8 million base pairs) and 2847-fold coverage of the single plasmid (198,615 base pairs) (Table 2). Additionally, 371,330 random reads representing 33-fold sequence coverage were generated by pyrosequencing on a Roche-454 GS20 sequencer (Hoffman-La Roche AG, Basel, Switzerland). Finally, 11,426 paired-end reads representing 1.5-fold sequence coverage were generated from a large insert fosmid library in the pEpiFOS-5 vector (insert sizes ranging from 28–47 kb) using dye terminator chemistry on an ABI 3730xl automated sequencer (Applied Biosystems, Waltham, MA, USA); these reads were used as a scaffold. The sequences were assembled using Velvet with default settings [10].
Table 2.
Features of the Rhodoferax antarcticus str. ANT.BRT genome.
| Attribute | Value | % of Total |
|---|---|---|
| Genome size (bp) | 4,007,881 | 100.0 |
| Chromosome size (bp) | 3,809,266 | 95.0 |
| Plasmid size (bp) | 198,615 | 5.0 |
| DNA coding (bp) | 3,564,951 | 88.9 |
| Chromosome G + C content | 59.1 | – |
| Plasmid G + C content | 48.4 | – |
| Total genes | 4324 | 100.0 |
| Protein-encoding genes | 4257 | 98.5 |
| RNA genes | 67 | 1.5 |
| Pseudogenes (putative) | 228 | 5.3 |
| Genes with function prediction | 2606 | 60.2 |
| Genes with Pfam domains | 3130 | 72.3 |
| Genes with signal peptides | 211 | 4.9 |
| Genes with transmembrane helices | 800 | 18.5 |
| CRISPR repeats | 5 | 0.1 |
Automated annotation of the genome was performed using the University of Maryland School of Medicine Institute for Genome Science’s Prokaryotic Annotation Pipeline [11] within the Analysis Engine service [12]. Pairwise alignments were generated using BLAST-extend-repraze (BER) [13], which employs a combination of BLAST and Smith–Waterman algorithms. In addition, the process includes gene identification with Glimmer, Hidden Markov Model (HMM) searches, transmembrane (Tm) HMM searches, SignalP predictions, and automatic annotations from AutoAnnotate. Additionally, the annotation tool Manatee [14] was used to manually review and confirm the annotation of every gene. Pseudogenes contained one or more mutations that would ablate expression; each inactivating mutation was subsequently checked against the original sequencing data.
In addition to Manatee, statistics in Table 2 were generated using the Pfam database (v. 30.0) [15], the SignalP database (v. 4.1) [16], the TMHMM database (v. 2.0) [17], and CRISPRFinder (v. 2.0) [18]. For the method of phylogenetic tree assembly, see the legend to Figure 1. This Whole Genome Shotgun project has been deposited at DDBJ/EMBL/GenBank under the accession MSYM00000000. The version described in this paper is version MSYM01000000.
3. Results and Discussion
3.1. Genome Properties
The genome of Rhodoferax antarcticus ANT.BRT consists of a single circular chromosome of approximately (due to the draft nature of the sequence) 3,809,266 base pairs with a G+C content of 59.1%. Rfx. antarcticus also possesses a sizeable plasmid (198,615 bp) with a significantly different G+C content than that of the chromosome (Table 2). Of the 4324 genes identified in the total genome, 4257 were protein-encoding genes, 67 were ribosomal or transfer RNAs, and 228 were putative pseudogenes. Most of the pseudogenes were putative transposases and hypothetical proteins rather than proteins having key metabolic or physiological functions (e.g., none were assigned roles relating to phototrophic energy conservation). A putative function and role category was assigned to 65.8% of protein-encoding genes, while the remaining genes were annotated as hypothetical, conserved hypothetical, or as genes of unknown function (Table 3).
Table 3.
Functional role categories of Rhodoferax antarcticus str. ANT.BRT genes.
| Characteristic | Genes | % of Genome Content * |
|---|---|---|
| Energy and central intermediary metabolism | 438 | 10.3 |
| Amino acid biosynthesis | 107 | 2.5 |
| Transport and binding proteins | 342 | 8.0 |
| Cofactor and prosthetic group biosynthesis | 206 | 4.8 |
| DNA metabolism and nucleotide synthesis | 252 | 5.9 |
| Transcription | 76 | 1.8 |
| Protein synthesis, modification, and degradation | 332 | 7.8 |
| Regulatory functions and signal transduction | 299 | 7.0 |
| Cellular processes (division, chemotaxis, motility, toxin production and resistance, detoxification) | 262 | 6.2 |
| Fatty acid and phospholipid metabolism | 71 | 1.7 |
| Mobile and extrachromosomal element functions | 159 | 3.7 |
| Cell envelope | 251 | 5.9 |
| Proteins with family/domain assignments | 407 | 9.6 |
| Hypothetical proteins | 754 | 17.7 |
| Conserved hypothetical proteins | 558 | 13.1 |
* Because some genes apply to more than one role category, this total exceeds 100%.
3.2. Major Photosynthesis Genes
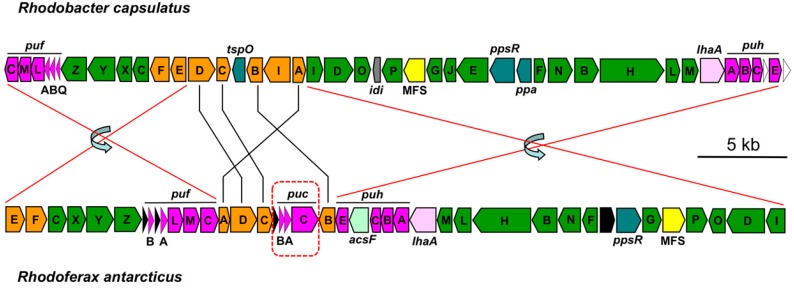
As is typical in purple bacteria, Rhodoferax antarcticus contains a photosynthesis gene cluster (PGC) in which most of the photosynthetic genes are arranged within a single superoperon on the circular chromosome (in the case of Rfx. antarcticus, at the 2.2 Mb mark). A comparison of gene synteny with that of the PGC from the well-studied species Rhodobacter (Rba.) capsulatus reveals two sizable inversions of superoperonal clusters in Rfx. antarcticus that include the bch, puf, and puh genes (Figure 2). Rfx. antarcticus contains just one copy of the pucABC operon, which encodes the peripheral light-harvesting antenna complex; interestingly, this operon is embedded within several crt (carotenoid biosynthesis) genes in the PGC. In all other purple nonsulfur bacteria, including Rba. capsulatus (Figure 2), puc genes are absent from the PGC and are instead dispersed in other regions of the chromosome, often in multiple copies.
Figure 2.
Superoperonal photosynthetic gene cluster of Rfx. antarcticus ANT.BRT. The puc genes, which are absent from Rhodobacter capsulatus, are present in the Rfx. antarcticus cluster. Lines indicate rearrangements in gene synteny and arrows indicate inversions. Color key: green, bacteriochlorophyll synthesis (bch); orange, carotenoid synthesis (crt); pink, photosynthetic reaction centers (puh); light-harvesting complexes (puf and puc); teal, regulatory proteins; white, uncharacterized proteins; black, hypothetical proteins. All other colors are unique for clarity.
As is the case for many other PNBs, the Rfx. antarcticus PGC also contains a gene encoding an unspecified major facilitator superfamily (MFS) protein (Figure 2) nested between bchG (GRFA_2400) and bchP (GRFA_2402; Figure 2), genes that encode enzymes that catalyze the final steps of bacteriochlorophyll (Bchl) synthesis. Gene products in this ubiquitous superfamily typically function in membrane transport. Due to its proximity to these genes, the MFS gene product may play a key role in the assembly of photosynthetic complexes [19], possibly by facilitating the insertion of Bchl a into membranes. It is worth noting that intracytoplasmic membranes typical of purple bacteria were not observed in electron micrographs of low-light grown cells of Rfx. antarcticus [3]. Some purple bacteria, such as Rhodocyclus species and Rubrivivax gelatinosus—purple bacteria closely related to Rfx. antarcticus (Figure 1)—have minimal intracytoplasmic membrane systems and rely on extensions of the cytoplasmic membrane to house the photosynthetic machinery [20,21,22]. It may be that Rfx. antarcticus is another such example of this.
All genes necessary for Bchl a biosynthesis were identified in the Rfx. antarcticus genome, although not all are located in the PGC (Figure 2). Apart from the PGC, there are two other regions of the chromosome that contain Bchl biosynthesis genes. The genes bchJ (GRFA_3894) and bchE (GRFA_3895) are found contiguously at approximately 3.6 Mb, and a second copy of the genes bchI (GRFA_1518) and bchD (GRFA_1519) are found at approximately 1.4 Mb. BLASTp analyses show moderate to low protein sequence identity (bchI- 91% coverage, 45% identity; bchD- 68% coverage, 36% identity) and disparate lineages (exclusively Betaproteobacteria at 1.4 Mb versus Beta-, Alpha-, and Gammaproteobacteria at 3.6 Mb) between the two sets of bchID. The purpose of maintaining two phylogenetically distinct sets of genes that encode the enzyme magnesium chelatase, which adds Mg2+ to the tetrapyrrole ring structure of Bchl a, in separate regions of the Rfx. antarcticus genome is unclear; however, it is possible that they are regulated differently by temperature or some other environmental variable.
Also noteworthy was the presence of acsF (GRFA_2386), located within the PGC, and bchE (GRFA_3895), which was located on a separate region of the chromosome outside of the PGC. These two genes encode versions of magnesium-protoporphyrin IX monomethyl ester cyclase (EC 1.14.13.81) that should enable Rfx. antarcticus to synthesize Bchl a under both anaerobic and aerobic conditions, respectively [23,24]. This flexibility emphasizes the metabolic versatility of Rfx. antarcticus, an organism that may need to oscillate regularly between aerobic and anaerobic (and chemotrophic or phototrophic) metabolisms, depending on the prevailing physicochemical conditions in its microbial mat habitat. Especially during the winter-to-spring and fall-to-winter transitions in Antarctica, light and O2 levels may fluctuate rapidly in such a habitat.
Carotenoids are important accessory pigments that assist phototrophs in collecting light energy and protect the cell from the damaging effects of reactive oxygen species [25,26]. Annotation of the Rfx. antarcticus genome revealed the presence of crt genes that encode enzymes needed for the complete spheroidene and spirilloxanthin pathways. Whereas most crt genes are located in the PGC (Figure 2), crtI (GRFA_3896) is located at approximately 3.6 Mb within the same operon as bchJE, and a second copy of crtA (GRFA_2303) is located at approximately 2.1 Mb. These findings are in agreement with carotenoid analyses of Rfx. antarcticus, which showed high levels of hydroxyspheroidene and other spheroidene derivatives and traces of spirilloxanthin and other spirilloxanthin derivatives in this organism [4,19].
3.3. Light-Harvesting Complexes
From genomic sequence data, it is evident that Rfx. antarcticus can biosynthesize both a core and a peripheral light-harvesting complex. The puf operon, which encodes the alpha and beta subunits of the LH1 complex, and the puc operon, which encodes the alpha and beta subunits of the peripheral LH2/LH3 complexes, are both located in the PGC of Rfx. antarcticus (Figure 2). The presence of genes encoding the LH1 complex is consistent with the absorption spectrum of intact cells given by Madigan et al. [3], which showed an absorbance peak at 866 nm. However, the same spectra failed to yield evidence for an LH2 complex, as no maxima were observed between 850 and 855 nm. However, the presence of absorbance maxima at 819 nm instead suggests that Rfx. antarcticus synthesizes an LH3 complex [3]. At least three species of purple bacteria that produce both LH2 and LH3 (Rhodobacter azotoformans, Phaeospirillum molischianum, and Rhodoblastus acidophilus) have been shown to exhibit a change in the ratio of peripheral complexes when light intensity or temperature changes [27,28,29,30]. In these bacteria, low temperatures and/or low light intensity elicit an increase in the expression of LH3 complexes and a decrease in the expression of LH2 complexes. In addition, Mascle-Allemand et al. [28] showed that the transition between spectral forms of peripheral complexes can be complete, and therefore LH2 is not necessary as a mediator between LH3 and LH1.
Spectroscopic data presented by Madigan et al. [3] were gathered from cells grown at 18 °C and low incandescent light. Considering that Rfx. antarcticus is capable of growth between 0 and 25 °C, it is reasonable to assume that incubation at 18 °C would have been warm enough to trigger preferential expression of LH2 complexes over LH3 if LH2 complexes could indeed be synthesized. However, no spectral evidence for an LH2 complex was observed, and this suggests that the sole puc operon identified in the genome encodes the LH3 complex, responsible for producing absorbance at 819 nm.
The spectrum produced by peripheral light-harvesting complexes depends upon the interactions of the C-3 acetyl group of Bchl a with the peripheral complex. If key residues at positions 44 and 45 of the LH2 alpha subunit (PucA) are able to hydrogen bond with the C-3 acetyl group, the spectrum shows a peak near 850 nm, whereas the absence of hydrogen bonding shifts the peak to 820 nm [29,31]. Examination of the amino acid sequence of PucA in Rfx. antarcticus revealed two phenylalanine residues at positions 44 and 45, not tyrosine and tryptophan, respectively, which are needed to form hydrogen bonds [29,32]. In addition, protein sequence comparison of the PucA and PucB subunits from Rfx. antarcticus to corresponding LH3 sequences from Phaeospirillum molischianum [33] revealed conserved motifs, with 38% and 36% sequence identity, respectively.
These data lead to the hypothesis that, in addition to LH1, Rfx. antarcticus produces LH3 (B800/820) complexes to the exclusion of LH2 under all growth conditions, presumably with the arrangement shown in Figure 3. If true, this phenomenon would be unique among purple bacteria. Such an atypical physiological response makes sense considering the habitat of Rfx. antarcticus. In the aquatic microbial mat from which strain ANT.BRT was isolated (Table 1), the maximum water temperature during the summer is 8 °C [19,34]. In addition, light levels approach zero during the austral winter, and therefore, in combination with permanently cold temperatures, preferential selection for the ability to synthesize LH3 over LH2 may have occurred in Rfx. antarcticus. The production of an LH3 complex to the exclusion of an LH2 complex may be an adaptation that gives Rfx. antarcticus a selective advantage in its cold and light-limiting microbial mat habitat.
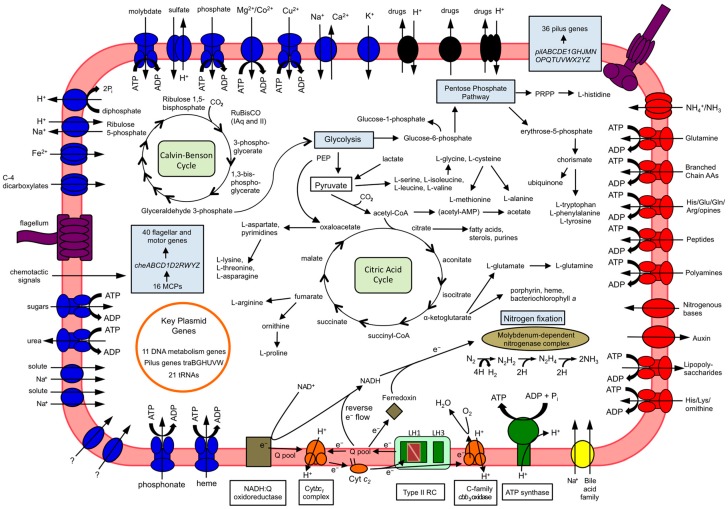
Figure 3.
Summary of metabolism and transporters of Rfx. antarcticus ANT.BRT. Noteworthy features include light-harvesting via a type II (quinone-type) reaction center [25] in association with LH1 and LH3, an intact Calvin–Benson cycle with two forms of RuBisCO, the ability to synthesize 20 amino acids, and antibiotic resistance properties conferred by drug exporters. In addition, both a flagellum and a type IV pilus are shown, providing two means of motility. Interestingly, although biotin is required for growth, no biotin transporter was identified in the genome. However, it is possible that a novel biotin transporter is present among several transporters of undetermined substrates.
The LH complexes of Rfx. antarcticus are also intriguing considering the spectral differences between the two strains of Rfx. antarcticus. Strain Fryx1, which conducts a planktonic lifestyle in the water column of Lake Fryxell, shows an absorbance peak at 836 nm [5]. Fowler et al. [35] demonstrated that a single amino acid substitution (Tyr44 → Phe) in the LH2 alpha subunit primary structure blue-shifted the spectrum to 839 nm, essentially forming a light-harvesting complex having spectral qualities intermediate of LH2 and LH3. Since Fryx1 and ANT.BRT are two strains of the same species, it is possible that, with respect to the alpha44 and 45 positions, Fryx1 has one hydrogen-bond forming residue and one non-hydrogen-bond forming residue, while ANT.BRT maintains two non-hydrogen-bond forming residues. Differences in light quality and availability between the water column of the permanently ice-covered Lake Fryxell and microbial mats of Ross Island ponds could have selected for these differential adaptations in order for these two strains to achieve maximal fitness in their respective environments.
3.4. Carbon Metabolism
Due to their metabolic versatility, purple bacteria often inhabit fluctuating environments, which would include the Antarctic microbial mat from which strain ANT.BRT was isolated [2]. Beside the long periods of darkness or light during the respective winter and summer seasons, Antarctic microbial mat communities experience turbulent anoxic/oxic cycles during the seasonal transition periods, stressors for which multiple energy generation pathways and the ability to easily switch between them are beneficial. Like most other purple bacteria, Rfx. antarcticus is metabolically versatile, as confirmed by genomic analysis, which revealed genes for both phototrophic (anoxic/light) and heterotrophic (oxic/dark) growth.
Genes encoding all enzymes of the Calvin–Benson cycle are present in the Rfx. antarcticus genome (Figure 3). One notable feature is the presence of two forms (IAq and II) of the enzyme ribulose 1,5-bisphosphate carboxylase/oxygenase (RuBisCO), which carboxylates ribulose 1,5-bisphosphate in the first step of the cycle to produce two molecules of 3-phosphoglycerate [25,36]. The presence of multiple RuBisCO forms is thought to confer metabolic versatility since the unique enzymatic properties of each form suit different environmental conditions, such as variations in [CO2]/[O2] ratios [36]. Presumably, possessing both form IAq (kcat 3.7 s−1), which operates best with medium-to-low CO2 availability [37], and form II (kcat 5.7 s−1), which functions best in high CO2 and low O2 concentrations, would enable Rfx. antarcticus to continue carbon fixation under transitional periods of fluctuating dissolved O2 concentrations [36]. In addition, it is possible that the two Rfx. antarcticus RuBisCO enzymes differ in their temperature optima for activity.
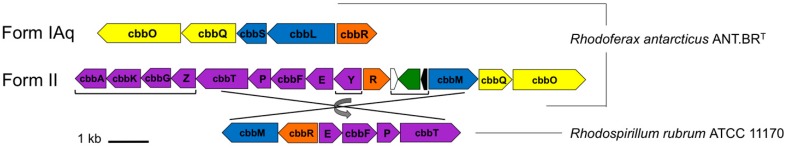
The form IAq gene cluster arrangement is typical of other purple nonsulfur bacteria, sharing the same synteny as Rhodospirillum centenum str. SW (ATCC 51521) [37]. However, the form II gene arrangement in Rfx. antarcticus is unusual in that it seems to be a combination of two typical form II gene cluster arrangements. While the Rfx. antarcticus form II RuBisCO is directly transcribed with the cbbQO genes (GRFA_112 and GRFA_113, respectively), which are linked to post-translational regulation of RuBisCO, the metabolic gene cluster for the remaining components of the Calvin–Benson cycle lies immediately adjacent to and is transcribed in the opposite direction of the transcriptional regulator cbbR (GRFA_107; Figure 4). This proximity is unusual considering that Calvin–Benson cycle metabolic genes are normally located elsewhere in the genome when form II RuBisCO and cbbQO are transcribed simultaneously [36]. However, bacteria that switch between Calvin–Benson cycle-mediated autotrophy and heterotrophic growth typically have genes for the remaining components of the Calvin–Benson cycle adjacent to the form II RuBisCO [36], suggesting that Rfx. antarcticus uses the combination of arrangements to tightly regulate its method of growth depending upon conditions and resources in its environment. Synteny comparisons to the Rhodospirillum rubrum form II RuBisCO gene cluster reveals that a segment of the Rfx. antarcticus form II cluster has been inverted and includes additional genes (Figure 4). Especially unusual is the presence of three unrelated genes [cbiA (GRFA_109), which encodes cobyrinic acid a,c-diamide synthetase, and two unidentified genes] between cbbR and the rest of the gene cluster; their function at this locus is unknown.
Figure 4.
RuBisCO gene cluster arrangements in the purple nonsulfur bacteria Rfx. antarcticus and Rhodospirillum rubrum. Rfx. antarcticus possesses two forms of RuBisCO, forms IAq and II. Brackets indicate the presence of extra genes. Color key: blue, RuBisCO subunits; orange, transcriptional regulators; yellow, RuBisCO activation proteins; purple, CBB cycle metabolic genes; green, cobyrinic acid a,c-diamide synthetase; white, uncharacterized proteins; black, hypothetical proteins.
The Rfx. antarcticus genome also contains complete sets of genes for glycolysis and the citric acid cycle (Figure 3). Genes for the Entner–Doudoroff and pentose phosphate pathways were also identified. Complete catabolic pathways for carbon source utilization were confirmed for all substrates previously shown to support growth of strain ANT.BRT, except for fructose. No genes encoding fructokinase, hexokinase, or other enzymes that would facilitate entry of fructose into glycolysis via conversion to another sugar were identified in the genome, despite the fact that fructose supported strong growth of Rfx. antarcticus [3]. Also of interest is the presence of genes encoding phosphoenolpyruvate (PEP) carboxylase (EC 4.1.1.31), which adds CO2 to PEP to produce oxaloacetate, typically the first step of the C-4 dicarboxylic acid cycle. Since the cycle is presumably incomplete in Rfx. antarcticus due to the absence of a gene encoding pyruvate-phosphate dikinase (EC 2.7.9.1), it is postulated that PEP carboxylase and other enzymes of this partial cycle instead facilitate anaplerotic assimilation of CO2 [37], thereby replenishing intermediates of the citric acid cycle that have been depleted during amino acid biosynthesis [38]. Rfx. antarcticus is also capable of importing C-4-dicarboxylic acids—prime growth substrates for this organism [3]—via a C-4 dicarboxylate ABC transporter; this enables replenishment of citric acid cycle intermediates via a second route and diminishes the requirement for obligatory C-4 dicarboxylate synthesis. A C-family heme-copper cbb3 oxidase was identified in the Rfx. antarcticus genome and presumably serves as the terminal electron acceptor during growth supported by aerobic respiration (Figure 3).
3.5. Nitrogen Fixation
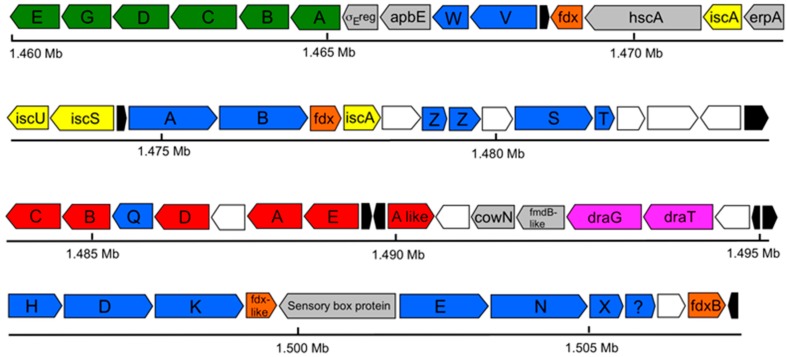
Previous observations that Rfx. antarcticus can fix N2 [3] were supported in the genome, which revealed genes encoding a molybdenum (Mo) nitrogenase. One set of nif genes, which encodes Mo-nitrogenase and assembly proteins, as well as one set of mod genes, which encodes an ABC transporter for molybdenum, were present in the nitrogen fixation gene cluster (Figure 5). Notably, however, neither vnf nor anf genes, which encode vanadium (V) and iron (Fe)-only nitrogenases, respectively [39], nor a vanadium transporter, were identified, indicating that the Mo nitrogenase is the only such enzyme present in Rfx. antarcticus. Mo-nitrogenases are present in all known diazotrophic Bacteria [40], so finding the genes for this enzyme in the Rfx. antarcticus genome is not surprising. However, it is surprising that Rfx. antarcticus does not have an alternative V- or Fe-only nitrogenase, as V nitrogenases have been shown to have a greater specific activity than Mo nitrogenases at lower temperatures [41], and several other diazotrophic Proteobacteria possess alternative nitrogenases [42].
Figure 5.
Nitrogen fixation gene cluster of Rhodoferax antarcticus ANT.BRT. Color key: green, rnf genes; blue, nif genes; red, mod genes; yellow, isc genes; pink, dra genes; orange, ferredoxin; gray, other nitrogen fixation-associated proteins; white, uncharacterized proteins; black, hypothetical proteins.
V nitrogenases have been postulated to allow for nitrogen fixation in cold environments [43]. In the absence of such an enzyme, the Rfx. antarcticus Mo nitrogenase must be able to function at low temperatures, an uncommon feature among characterized nitrogenases. Acetylene reduction assays by Madigan et al. [3] showed nitrogenase activity down to 2 °C, but no further experiments were done to describe the nitrogenase system of Rfx. antarcticus. It might thus be of interest to better characterize the Rfx. antarcticus nif gene products to determine what structural features enable this Mo nitrogenase to function under cold conditions.
All nif genes absolutely required for nitrogen fixation [44] were present in the Rfx. antarcticus genome, but there were some notable absences from the canonical nitrogen fixation cluster. Although nifJ, which encodes pyruvate flavodoxin/ferredoxin oxidoreductase, is missing from the Rfx. antarcticus nitrogen fixation gene cluster, a gene was identified that is predicted to encode a pyruvate flavodoxin/ferredoxin oxidoreductase (GRFA_1003; EC 1.2.7.1) at approximately 0.9 Mb, adjacent to other electron transfer genes. This suggests that this gene product could serve the function of NifJ. Additionally, while no strong matches for flavodoxin were found in the Rfx. antarcticus genome, four different ferredoxin genes are located in the nitrogen fixation cluster, suggesting that ferredoxin, not flavodoxin (nifF-encoded or otherwise), may be used by this phototroph to shuttle electrons from the electron transport chain to the nifH-encoded Fe protein of the nitrogenase complex [45]. Other nif genes missing from the Rfx. antarcticus genome include nifU (scaffold protein associated with nifS activity), nifM (Fe protein processing), nifY (insertion of FeMo-cofactor into dinitrogenase), and nifL (negative regulator of the nif genes) [45,46,47]. The composition of nif gene clusters varies greatly among species, depending upon the presence or absence of other proteins that can substitute for nif gene products [47,48]. For example, both iscU (GRFA_1626), a homolog of nifU, and two copies of iscA (GRFA_1624 and GRFA_2967), which have been postulated to be an alternate scaffold to NifU for the construction of Fe-S clusters [49,50], are present in the Rfx. antarcticus nitrogen fixation cluster. The significance of these would require further study.
3.6. Motility
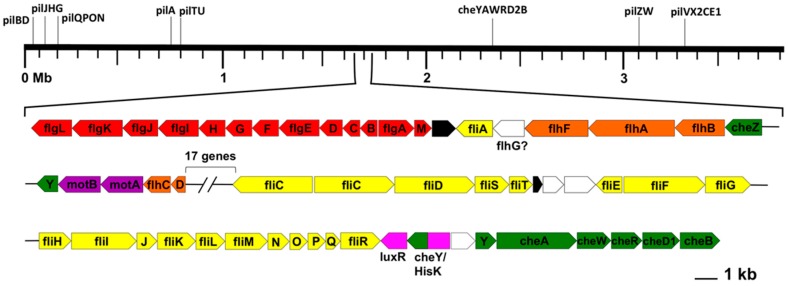
The Rfx. antarcticus genome contains genes for motility via flagella and type IV pili, as well as genes for chemotaxis. This genetic evidence supports previous observations by Madigan et al. [3] that cells of Rfx. antarcticus are highly motile and possess at least one polar flagellum. However, the arrangement of flagellar genes is unusual in that the genes are located in only two clusters, separated by 17 unrelated genes (Figure 6). Typically, flagellar genes are dispersed over the length of the chromosome, such as in the case of the purple bacterium Rhodospirillum centenum, whose five flagellar gene clusters are found at five different loci on the chromosome [37]. In contrast, type IV pilus genes in Rfx. antarcticus are scattered in a more typical fashion throughout the chromosome.
Figure 6.
Linear representation of the Rfx. antarcticus chromosome, loci of motility genes, and expanded flagellar gene cluster. Type IV pilus genes and a second set of chemotaxis genes (black lines) are distributed down the length of the chromosome, but the flagellar genes are all located in one superoperonal gene cluster. Color key: red, flg gene cluster; yellow, fli gene cluster; orange, flh gene cluster; green, che gene cluster; purple, mot gene cluster; pink, unrelated characterized proteins; white, uncharacterized proteins; black, hypothetical proteins.
Although the components of the flagellar gene cluster of Rfx. antarcticus are typical of those of other flagellated bacteria, there are some peculiarities. To date, genomic analyses of cells that synthesize polar flagella have shown that all genes in the fliEFGHIJKLMNPQR section of the flagellar gene cluster are transcribed together in the same direction [51]. However, in the Rfx. antarcticus flagellar cluster, fliE (GRFA_1877) is transcribed in the opposite direction of all other genes in the cluster, which may be the result of an inversion, although the consequence of this change is unclear. In addition, a presumed duplication event has produced a second copy of fliC, which encodes flagellin, the protein that forms the filament of the flagellum. The two copies of fliC (GRFA_1870 and GRFA_1871), which are adjacent to each other and transcribed in opposite directions, are 90% identical to each other, as determined by BLASTp analysis. In the highly motile, predatory deltaproteobacterium Bdellovibrio bacteriovorus, whose genome also encodes multiple copies of flagellin, it was shown that the distinct copies of flagellin form different regions of the flagellum [52]. It is therefore possible that the protein products of the duplicated fliC genes also compose different regions of the Rfx. antarcticus flagellum.
Although no pili of any type have been observed in cultured Rfx. antarcticus cells, it is possible that pili serve an important function for survival in an Antarctic microbial mat. Besides microbial cells, microbial mats also contain debris that provides the required solid surface for twitching motility. This form of motility is pilus-facilitated and involves anchoring the pilus to a solid surface in a grappling hook-type manner and then jerkily reeling the cell toward or across the surface. Within a microbial mat where flagellum-powered swimming may not be practical or possible, twitching motility may be the more efficient form of locomotion.
Unlike the planktonic Rfx. antarcticus strain Fryx1, which clearly possesses gas vesicles [5], neither gas vesicles nor the genes that encode them were found in Rfx. antarcticus strain ANT.BRT. Gas vesicles provide buoyancy and allow cells to change positions in a water column. Although beneficial for a planktonic existence, they would not be useful within the confines of a stratified microbial mat. The mechanisms of motility in these two phylogenetically identical strains therefore seem well suited to their respective environments.
3.7. Plasmid Features
The 198.6 kbp plasmid contains 226 open reading frames (ORFs), most of which are annotated as hypothetical or uncharacterized proteins. The G+C content of the plasmid (48.4%) is significantly lower than that of the chromosome (59.1%), which suggests horizontal gene transfer and supports the idea that this replicon is indeed a plasmid rather than a secondary chromosome, as has been described for some other Beta- and Gammaproteobacteria [53,54]. Extensive analysis of plasmid contents did not identify any unique genes essential to the survival of Rfx. antarcticus. However, since the majority of the plasmid genes have undetermined functions, further molecular studies are needed to confirm the accessory nature of this replicon.
Of the plasmid-encoded proteins that have a putative assigned function, 11 participate in DNA metabolism, consistent with analyses by Dziewit and Bartosik [53] that show the highest number of proteins encoded by plasmids from cold-adapted bacteria function in replication, recombination, and repair of DNA. No antibiotic or heavy metal resistance genes were found on the plasmid, although genes for resistance to beta-lactam antibiotics were identified on the chromosome. The plasmid also contains six genes, traBGHUVW, associated with conjugation. However, not all genes required for conjugation pilus assembly are present [55], rendering this operon incomplete and leaving Rfx. antarcticus likely unable to transfer genes through this mechanism. In addition, no gvp gene cluster for gas vesicle formation was identified on the plasmid. Other polar gas-vesiculate bacteria, such as Octadecabacter arcticus, contain plasmids having complete gvp gene clusters [56]. It is likely that Rfx. antarcticus strain Fryx1 [5] gained the capacity to synthesize gas vesicles through lateral transfer of such a gvp-containing plasmid.
A final notable feature of the Rfx. antarcticus plasmid is the unexpected presence of 21 tRNA genes, constituting about one-third of the tRNA genes in the genome. tRNAscan-SE (v.1.21) [57] analysis reveals that the average Cove score, which indicates the probability that the predicted secondary structure of a tRNA matches the statistical model [58], for the plasmid-encoded tRNAs is significantly lower at 60.6 bits than the average Cove score for the chromosomal tRNAs (79.88 bits). An increasingly higher Cove score indicates an increasingly higher probability that a tRNA is functional once it has been transcribed and assumes its folded conformation [58]. This result brings into question the functionality of the plasmid-encoded tRNAs and indicates that the tRNA genes on this extraneous replicon may be degenerating. Moreover, all tRNA genes on the plasmid duplicate anticodons already present on the chromosome, so it is unclear what function (if any) the plasmid tRNAs serve in translation processes of Rfx. antarcticus.
It is possible that the extensive assemblage of potentially degenerate tRNA genes on the plasmid plays a greater role in Rfx. antarcticus as a cold-adaptation strategy than as a tool for translation processes. The accumulation of tRNAs for the purpose of cryoprotection has been reported in other psychrophiles [59,60], and it may also be important for Rfx. antarcticus. We were unable to identify genes that encode well-known cold-adaptation proteins, including the cold shock proteins CspA or CspB; the cold-adaptive proteins CapA or CapB; ice-nucleating proteins InaQ, InaK, or InaZ; and antifreeze protein AfpA. However, a gene encoding tRNA dihydrouridine synthase A (GRFA_2724), an enzyme that incorporates dihydrouridine into tRNA molecules to increase their conformational flexibility [61], was present in the Rfx. antarcticus genome. We also identified genes encoding an ABC-type transporter for glycine betaine, which has been shown to function as a cryoprotective compatible solute in a variety of psychrophilic bacteria [62]. Together, these features may be essential for Rfx. antarcticus to maintain viability and sustain growth in permanently cold Antarctic mats.
4. Conclusions
As Rfx. antarcticus strain ANT.BRT is the first psychrophilic anoxygenic phototroph to have its genome sequenced, its genetic blueprint broadens our understanding of photosynthesis in extreme environments and fills in one of the significant gaps in genomic databases of anoxygenic phototrophic bacteria. Insights from this genome and from future genomic studies of phototrophic extremophiles should help clarify the origin and evolution of photosynthesis and the transition from anoxygenic to oxygenic phototrophy.
Acknowledgments
This work was supported by the U.S. National Science Foundation Phototrophic Prokaryotes Sequencing Project as an award to J.W.T., R.E.B., and M.T.M. (Evolutionary Diversification of Photosynthesis and the Anoxygenic to Oxygenic Transition; NSF Grant #0950550). Additional support for participation of J.M.B., C.J.R., B.M.S., and W.M.S. in this research was provided by Hodson Summer Research Institute (Indiana Wesleyan University) grants to W.M.S. We thank Dr. Yih-Kuang Lu and Renxia Huang for excellent technical assistance and discussion during the genome sequencing, assembly, and annotation efforts. M.T.M. acknowledges Richard W. Castenholz for the sample of the Ross Island microbial mat from which Rfx. antarcticus strain ANT.BRT was isolated. W.M.S. and J.M.B. acknowledge Michelle Giglio, Suvarna Nadendla, James Matsumura, and Jain George at the Institute for Genomic Sciences, School of Medicine, University of Maryland for their invaluable assistance during the genome annotation and submission process.
Author Contributions
J.W.T., R.E.B., M.T.M., J.M.B., and W.M.S. conceived and designed the experiments; all authors performed the experiments and analyzed the data, with the bulk of the analysis done by J.M.B.; M.T.M., R.E.B., and J.W.T. contributed reagents/materials/analysis tools; J.M.B., W.M.S., M.T.M., and J.W.T. wrote the paper, with other authors providing reviews and edits during the draft stage.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Madigan M.T., Jung D.O. An overview of purple bacteria: Systematics, physiology, and habitats. In: Hunter C.N., Daldal F., Thurnauer C., Beatty J.T., editors. The Purple Phototrophic Bacteria. Volume 28. Springer; Dordrecht, the Netherlands: 2009. pp. 1–15. [Google Scholar]
- 2.Madigan M.T. Anoxygenic phototrophic bacteria from extreme environments. Photosynth. Res. 2003;76:157–171. doi: 10.1023/A:1024998212684. [DOI] [PubMed] [Google Scholar]
- 3.Madigan M.T., Jung D.O., Woese C.R., Achenbach L.A. Rhodoferax antarcticus sp. nov., a moderately psychrophilic purple nonsulfur bacterium isolated from an Antarctic microbial mat. Arch. Microbiol. 2000;173:269–277. doi: 10.1007/s002030000140. [DOI] [PubMed] [Google Scholar]
- 4.Morita R.Y. Psychrophilic bacteria. Bacteriol. Rev. 1975;39:144–167. doi: 10.1128/br.39.2.144-167.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jung D.O., Achenbach L.A., Karr E.A., Takaichi S., Madigan M.T. A gas vesiculate strain of the purple non-sulfur bacterium Rhodoferax antarcticus isolated from Lake Fryxell, Dry Valleys, Antarctica. Arch. Microbiol. 2004;182:236–243. doi: 10.1007/s00203-004-0719-8. [DOI] [PubMed] [Google Scholar]
- 6.Clark K., Karsch-Mizrachi I., Lipman D.J., Ostell J., Sayers E.W. GenBank. Nucleic Acids Res. 2016;44:D67–D72. doi: 10.1093/nar/gkv1276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kumar S., Stetcher G., Tamura K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016;30:1870–1874. doi: 10.1093/molbev/msw054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jukes T.H., Cantor C.R. Evolution of protein molecules. In: Munro H.N., editor. Mammalian Protein Metabolism. Academic Press; New York, NY, USA: 1969. pp. 21–132. [Google Scholar]
- 9.Choudhary M., Mackenzie C., Donohue T.J., Kaplan S. Purple bacterial genomics. In: Hunter C.N., Daldal F., Thurnauer C., Beatty J.T., editors. The Purple Phototrophic Bacteria. Volume 28. Springer; Dordrecht, The Netherlands: 2009. pp. 691–706. [Google Scholar]
- 10.Zerbino D.R. Using the Velvet de novo assembler for short-read sequencing technologies. Curr. Protoc. Bioinform. 2010;31:11.5.1–11.5.12. doi: 10.1002/0471250953.bi1105s31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Galens K., Orvis J., Daugherty S., Creasy H.H., Angiuoli S., White O., Wortman J., Mahurkar A., Giglio M.G. The IGS Standard Operating Procedure for Automated Prokaryotic Annotation. Stand. Genom. Sci. 2011;4:244–251. doi: 10.4056/sigs.1223234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.University of Maryland School of Medicine Institute for Genome Sciences. [(accessed on 21 February 2017)]. Available online: http://www.igs.umaryland.edu/research/bioinformatics/analysis/
- 13.BLAST-Extend-Repraze. [(accessed on 21 February 2017)]. Available online: http://ber.sourceforge.net/
- 14.Manatee. [(accessed on 21 February 2017)]. Available online: http://manatee.sourceforge.net.
- 15.Finn R.D., Coggill P., Eberhardt R.Y., Eddy S.R., Mistry J., Mitchell A.L., Potter S.C., Punta M., Qureshi M., Sangrador-Vegas A., et al. The Pfam protein families database: Towards a more sustainable future. Nucleic Acids Res. 2016;44:D279–D285. doi: 10.1093/nar/gkv1344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Petersen T.N., Brunak S., von Heijne G., Nielsen H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods. 2011;8:785–786. doi: 10.1038/nmeth.1701. [DOI] [PubMed] [Google Scholar]
- 17.Krogh A., Larrson B., von Heijne G., Sonnhammer E.L. Predicting transmembrane protein topology with a hidden Markov model: Application to complete genomes. J. Mol. Biol., 2001;305:567–580. doi: 10.1006/jmbi.2000.4315. [DOI] [PubMed] [Google Scholar]
- 18.Grissa I., Vergnaud G., Pourcel C. CRISPRFinder: A web tool to identify clustered regularly interspaced short palindromic repeats. Nucleic Acids Res. 2007;35:W52–W57. doi: 10.1093/nar/gkm360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Addlesee H.A., Fiedor L., Hunter C.N. Physical mapping of bchG, orf427, and orf177 in the photosynthesis gene cluster of Rhodobacter. sphaeroides: Functional assignment of the bacteriochlorophyll synthetase gene. J. Bacteriol. 2000;182:3175–3182. doi: 10.1128/JB.182.11.3175-3182.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhao T. Master’s Thesis. Arizona State University; Tempe, AZ, USA: Jul, 2011. Genome sequencing and analysis of the psychrophilic anoxygenic phototrophic bacterium Rhodoferax antarcticus sp. ANT.BR. [Google Scholar]
- 21.Weckesser J., Drews G., Tauschel H.-D. Zur Feinstruktur und Taxonomie von Rhodopseudomonas. gelatinosa. Arch. Mikrobiol. 1969;65:346–358. doi: 10.1007/BF00412213. [DOI] [PubMed] [Google Scholar]
- 22.Drews G., Golecki J. Structure, molecular organization, and biosynthesis of membranes of purple bacteria. In: Blankenship R.E., Madigan M.T., Bauer C.E., editors. Anoxygenic Photosynthetic Bacteria. Volume 2. Kluwer Academic; Dordrecht, The Netherlands: 1995. pp. 231–257. [Google Scholar]
- 23.Ochane S., Steunou A.-S., Picaud M., Astier C. Aerobic and anaerobic Mg-protoporphyrin monomethyl ester cyclases in purple bacteria: A strategy adopted to bypass the repressive oxygen control system. J. Biol. Chem. 2004;279:6385–6394. doi: 10.1074/jbc.M309851200. [DOI] [PubMed] [Google Scholar]
- 24.Tang K.-H., Wen J., Li X., Blankenship R.E. Role of the AcsF protein in Chloroflexus. aurantiacus. J. Bacteriol. 2009;191:3580–3587. doi: 10.1128/JB.00110-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Blankenship R.E. Molecular Mechanisms of Photosynthesis. 2nd ed. Wiley Blackwell; Oxford, UK: 2014. [Google Scholar]
- 26.Tuveson R.W., Larson R.A., Kagan J. Role of cloned carotenoid genes expressed in Escherichia coli in protecting against inactivation by near-UV light and specific phototoxic molecules. J. Bacteriol. 1988;170:4675–4680. doi: 10.1128/jb.170.10.4675-4680.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li K., Zhao C., Yue H., Yang S. A unique low light adaptation mechanism in Rhodobacter. azotoformans. J. Basic Microbiol. 2014;54:1350–1357. doi: 10.1002/jobm.201400422. [DOI] [PubMed] [Google Scholar]
- 28.Mascle-Allemand C., Duquesne K., Lebrun R., Scheuring S., Sturgis J.N. Antenna mixing in photosynthetic membranes from Phaeospirillum. molischianum. Proc. Natl. Acad. Sci. USA. 2010;107:5357–5362. doi: 10.1073/pnas.0914854107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McLuskey K., Prince S.M., Cogdell R.J., Isaacs N.W. The crystallographic structure of the B800-820 LH3 light-harvesting complex from the purple bacteria Rhodopseudomonas. acidophila strain 7050. Biochemistry. 2001;40:8783–8789. doi: 10.1021/bi010309a. [DOI] [PubMed] [Google Scholar]
- 30.Gardiner A.T., Cogdell R.J., Takaichi S. The effect of growth conditions on the light-harvesting apparatus in Rhodopseudomonas acidophila. Photosynth. Res. 1993;38:159–167. doi: 10.1007/BF00146415. [DOI] [PubMed] [Google Scholar]
- 31.Cogdell R.J. (University of Glasgow, Glasgow, Scotland, UK). Personal communication. 2016.
- 32.Prince S.M., Papiz M.Z., Freer A.A., McDermott G., Hawthornthwaite-Lawless A.M., Cogdell R.J., Isaacs N.W. Apoprotein structure in the LH2 complex from Rhodopseudomonas. acidophila strain 10050: Modular assembly and protein pigment interactions. J. Mol. Biol. 1997;268:412–423. doi: 10.1006/jmbi.1997.0966. [DOI] [PubMed] [Google Scholar]
- 33.Horibe T., Qian P., Hunter C.N., Hashimoto H. Stark absorption spectroscopy on the carotenoids bound to B800-820 and B800-850 type LH2 complexes from a purple photosynthetic bacterium, Phaeospirillum. molischianum strain DSM120. Arch. Biochem. Biophys. 2014;572:158–166. doi: 10.1016/j.abb.2014.12.015. [DOI] [PubMed] [Google Scholar]
- 34.Vincent W.F., Downes M.T., Castenholz R.W., Howard-Williams C. Community structure and pigment organization of cyanobacteria-dominated microbial mats in Antarctica. Eur. J. Phycol. 1993;l28:213–221. doi: 10.1080/09670269300650321. [DOI] [Google Scholar]
- 35.Fowler G.J.S., Visschers R.W., Grief G.G., van Grondelle R., Hunter C.N. Genetically modified photosynthetic antenna complexes with blueshifted absorbance bands. Nature. 1992;355:848–850. doi: 10.1038/355848a0. [DOI] [PubMed] [Google Scholar]
- 36.Badger M.R., Bek E.J. Multiple Rubisco forms in proteobacteria: Their functional significance in relation to CO2 acquisition by the CBB cycle. J. Exp. Bot. 2008;59:1525–1541. doi: 10.1093/jxb/erm297. [DOI] [PubMed] [Google Scholar]
- 37.Lu Y.-K., Marden J., Han M., Swingley W.D., Mastrian S.D., Chowdhury S.R., Hao J., Helmy T., Kim S., Kurdoglu A.A., et al. Metabolic flexibility revealed in the genome of the cyst-forming α-1 proteobacterium Rhodospirillum. centenum. BMC Genom. 2010;11:325–336. doi: 10.1186/1471-2164-11-325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sauer U., Eikmanns B.J. The PEP-pyruvate-oxaloacetate node as the switch point for carbon flux distribution in bacteria. FEMS Microbiol. Rev. 2005;29:765–794. doi: 10.1016/j.femsre.2004.11.002. [DOI] [PubMed] [Google Scholar]
- 39.Hu Y., Ribbe M.W. Nitrogenase and homologs. J. Biol. Inorg. Chem. 2015;20:436–445. doi: 10.1007/s00775-014-1225-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bellenger J.P., Xu Y., Zhang X., Morel F.M.M., Kraepiel A.M.L. Possible contribution of alternative nitrogenases to nitrogen fixation by asymbiotic N2-fixing bacteria in soils. Soil Biol. Biochem. 2013;69:413–420. doi: 10.1016/j.soilbio.2013.11.015. [DOI] [Google Scholar]
- 41.Miller R.W., Eady R.R. Molybdenum and vanadium nitrogenases of Azotobacter. chroococcum: Low temperature favors N2 reduction by vanadium nitrogenase. Biochem. J. 1988;256:429–432. doi: 10.1042/bj2560429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Raymond J., Siefert J.L., Staples C.R., Blankenship R.E. The natural history of nitrogen fixation. Mol. Biol. Evol. 2004;21:541–554. doi: 10.1093/molbev/msh047. [DOI] [PubMed] [Google Scholar]
- 43.Walmsley J., Kennedy C. Temperature-dependent regulation by molybdenum and vanadium of expression of the structural genes encoding three nitrogenases in Azotobacter. vinelandii. Appl. Environ. Microbiol. 1991;57:622–624. doi: 10.1128/aem.57.2.622-624.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dos Santos P.C., Fang Z., Mason S.W., Setubal J., Dixon R. Distribution of nitrogen fixation and nitrogenase-like sequences amongst microbial genomes. BMC Genom. 2012;13:162. doi: 10.1186/1471-2164-13-162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cheng Q. Perspectives in biological nitrogen fixation research. J. Integr. Plant. Biol. 2008;50:786–798. doi: 10.1111/j.1744-7909.2008.00700.x. [DOI] [PubMed] [Google Scholar]
- 46.Dos Santos P.C., Smith A.D., Frazzon J., Cash V.L., Johnson M.K., Dean D.R. Iron-sulfur cluster assembly: NifU-directed activation of the nitrogenase Fe protein. J. Biol. Chem. 2004;279:19705–19711. doi: 10.1074/jbc.M400278200. [DOI] [PubMed] [Google Scholar]
- 47.Boyd E.S., Costas A.M.G., Hamilton T.L., Mus F., Peters J.W. Evolution of molybdenum nitrogenase during the transition from anaerobic to aerobic metabolism. J. Bacteriol. 2015;197:1690–1699. doi: 10.1128/JB.02611-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang L., Zhang L., Liu Z., Zhao D., Liu X., Zhang B., Xie J., Hong Y., Li P., Chen S., Dixon R., Li J. A minimal nitrogen fixation cluster from Paenibacillus. sp. WLY78 enables expression of active nitrogenase in Escherichia coli. PLoS Genet. 2013;9:e1003865. doi: 10.1371/annotation/1e9bcb70-265a-4383-abf4-3466d144d56e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Krebs C., Agar J.N., Smith A.D., Frazzon J., Dean D.R., Huynh B.H., Johnson M.K. IscA, an alternate scaffold for Fe-S cluster biosynthesis. Biochemistry. 2001;40:14069–14080. doi: 10.1021/bi015656z. [DOI] [PubMed] [Google Scholar]
- 50.Blanc B., Gerez C., de Choudens S.O. Assembly of Fe/S proteins in bacterial systems: Biochemistry of the bacterial ISC system. Biochim. Biophys. Acta. 2014;1853:1436–1447. doi: 10.1016/j.bbamcr.2014.12.009. [DOI] [PubMed] [Google Scholar]
- 51.Liu R., Ochman H. Origins of flagellar gene operons and secondary flagellar systems. J. Bacteriol. 2007;189:7098–7104. doi: 10.1128/JB.00643-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Iida Y., Hobley L., Lambert C., Fenton A.K., Sockett R.E., Aizawa S.-I. Roles of multiple flagellins in flagellar formation and flagellar growth post bdelloplast lysis in Bdellovibrio. bacteriovorus. J. Mol. Biol. 2009;394:1011–1021. doi: 10.1016/j.jmb.2009.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Dziewit L., Bartosik D. Plasmids of psychrophilic and psychrotolerant bacteria and their role in adaptation to cold environments. Front. Microbiol. 2014;5:596. doi: 10.3389/fmicb.2014.00596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cooper V.S., Vohr S.H., Wrocklage S.C., Hatcher P.J. Why Genes Evolve Faster on Secondary Chromosomes in Bacteria. PLoS Comput. Biol. 2010;6:e1000732. doi: 10.1371/journal.pcbi.1000732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Arutyunov D., Frost L.S. F conjugation: Back to the beginning. Plasmid. 2013;70:18–32. doi: 10.1016/j.plasmid.2013.03.010. [DOI] [PubMed] [Google Scholar]
- 56.Vollmers J., Voget S., Dietrich S., Gollnow K., Smits M., Meyer K., Brinkhoff T., Simon M., Daniel R. Poles apart: Arctic and Antarctic Octadecabacter. strains share high genome plasticity and a new type of xanthorhodopsin. PLoS ONE. 2013;8:e63422. doi: 10.1371/journal.pone.0063422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lowe T.M., Eddy S.R. tRNAscan-SE: A program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res. 1997;25:955–964. doi: 10.1093/nar/25.5.0955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Eddy S.R., Durbin R. RNA sequence analysis using covariance models. Nucleic Acids Res. 1994;22:2079–2088. doi: 10.1093/nar/22.11.2079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Math R.K., Jin H.M., Kim J.M., Hahn Y., Park W., Madsen E.L., Jeon C.O. Comparative genomics reveals adaptation by Alteromonas. sp. SN2 to marine tidal-flat conditions: Cold tolerance and aromatic hydrocarbon metabolism. PLoS ONE. 2012;7:e35784. doi: 10.1371/journal.pone.0035784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Riley M., Staley J.T., Danchin A., Wang T.Z., Brettin T.S., Hauser L.J., Land M.L., Thompson L.S. Genomics of an extreme psychrophile, Psychromonas. ingrahamii. BMC Genom. 2008;9:210. doi: 10.1186/1471-2164-9-210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dalluge J.J., Hamamoto T., Horikoshi K., Morita R.Y., Stetter K.O., McCloskey J.A. Posttranscriptional modification of tRNA in psychrophilic bacteria. J. Bacteriol. 1997;79:1918–1923. doi: 10.1128/jb.179.6.1918-1923.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Qin Q.-L., Xie B.-B., Yu Y., Shu Y.-L., Rong J.-C., Zhang Y.-J., Zhao D.-L., Chen X.-L., Zhang X.-Y., Chen B., et al. Comparative genomics of the marine bacterial genus Glaciecola. reveals the high degree of genomic diversity and genomic characteristic for cold adaptation. Environ. Microbiol. 2014;16:1642–1653. doi: 10.1111/1462-2920.12318. [DOI] [PubMed] [Google Scholar]