Abstract
Purpose
A major problem in macular degeneration is the inability to reduce RPE and photoreceptor death. These cells die by necroptosis and apoptosis, respectively, but the upstream activator(s) of these death pathways is unknown. In this study, we use the sodium iodate (NaIO3) model of oxidative stress to test the hypothesis that activation of the Fas receptor contributes to the death of the RPE and photoreceptors.
Methods
Sodium iodate was injected in Brown-Norway rats via femoral vein injection. Both in vivo (fundus photography, optical coherence tomography, and fluorescein angiography) and ex vivo (histology, immunohistochemistry, Western blot, and RT-PCR) analyses of the RPE and retina were conducted at baseline, as well as at various times post NaIO3 injection. The ability of intravitreal injection of Met12, a small peptide inhibitor of the Fas receptor, to prevent RPE and photoreceptor cell death was assessed.
Results
Injection of NaIO3 led to Fas-mediated activation of both necroptosis and apoptosis in the RPE and photoreceptors, respectively. This was accompanied by a significant increase in the number of microglia/macrophages in the outer retina. Met12 significantly reduced the activation of the Fas-mediated death pathways, resulting in reduced RPE and photoreceptor death and a decreased immune response.
Conclusions
Our results demonstrate that NaIO3 activates Fas-mediated cell death, both in the RPE and photoreceptor, and that a small peptide antagonist of the Fas receptor, Met12, significantly reduces the extent of this cell death. These findings suggest a role for Fas inhibition to protect the RPE and photoreceptors from death due to oxidative stress.
Keywords: Fas, sodium iodate, apoptosis, necroptosis
Age-related macular degeneration is a leading cause of irreversible blindness in people over the age of 65.1 While the exact etiology of AMD is not known, numerous studies support the conclusion that the disease results from a confluence of stressors, such as age, genetic susceptibility, and environmental factors, acting on the outer retina (RPE and photoreceptors) and disrupting normal cellular homeostasis.2–5 Cellular exposure to stress initially results in the activation of compensatory pathways that act to restore homeostasis; however, if the stress is chronic or intense enough, then there is activation of death pathways such as apoptosis and necroptosis.6,7 A major mediator of RPE and photoreceptor death is the Fas/Fas ligand (FasL) system.7–9 This has been demonstrated in a variety of disease models and appears to be independent of the specific stressor acting on the cell. As such it might serve as a therapeutic target for preventing activation of the death pathways in the setting of disrupted outer retinal homeostasis.
Numerous animal models of AMD have been created in a variety of species such as mice, rats, rabbits, pigs, and non-human primates.10–13 These models have used genetic modification, alteration of diet, induction of oxidative damage, or laser-induced choroidal neovascularization. None of these models are true AMD, yet their utility lies in the fact that they recapitulate specific aspects of the disease. The sodium iodate (NaIO3) model is widely used to study the molecular mechanism of cell death in AMD as it represents the disease-associated increase in oxidative stress and induces consistent and selective damage to the RPE.14,15 Exposure to NaIO3 results in a primary death of the RPE followed by a secondary death of the overlying photoreceptors, similar to what is observed in advanced atrophic AMD.16
A major outstanding problem in AMD treatment is the inability to reduce RPE and photoreceptor death. Studies have shown that these cells die by necroptosis14,17 and apoptosis,7,9 respectively, but the upstream activator(s) of these death pathways has not been identified. In this study, we use the NaIO3 model of RPE oxidative stress to test the hypothesis that activation of the Fas receptor contributes to the death of the RPE and photoreceptors. Our results demonstrate that sodium iodate results in increased Fas-mediated cell death, both in the RPE and photoreceptor, and that administration of a small peptide antagonist of the Fas receptor, Met12,18 significantly reduces the extent of this cell death. These findings suggest a role for Fas inhibition to protect the RPE and photoreceptors from death due to oxidative stress.
Methods
Animals and Experimental Procedures
All experiments conformed to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research and the guidelines established by the University Committee on Use and Care of Animals of the University of Michigan. Male retired breeder Brown-Norway rats (300–400 g) were purchased from Charles River Laboratories (Wilmington, MA, USA). Rats were housed under standard 12-hour light/12-hour dark conditions in the University of Michigan, Kellogg Eye Center animal facility for 2 weeks before initiation of experiments. Rats were anesthetized using a mixture of ketamine (100 mg/mL; Hopira, Lake Forest, IL, USA) and xylazine (20 mg/mL; Akorn, Lake Forest, IL, USA) with a 2:1 volume ratio. Pupils were dilated with topical 2.5% phenylephrine (Paragon BioTek, Inc., Portland, OR, USA) and 0.5% tropicamide (Akorn, Lake Forest, IL, USA).
Sodium iodate (Sigma-Aldrich Corp., St. Louis, MO, USA) was dissolved in phosphate-buffered saline at a concentration of 40 mg/mL. The NaIO3 was delivered via femoral vein injection at a concentration of 40 mg/kg. Some animals received an intravitreal injection of 50 μg Met12 (HHIYLGAVNYIY, Met12) dissolved in dimethyl sulfoxide18 in their left eye 5 days before femoral injection of NaIO3 using our previously described technique for intravitreal injection. Pretreatment was performed so as to allow the Met12 to maximally diffuse across the retina and access the RPE. As a control, the right eye was injected with 50 μg of an inactive peptide designated as mutant Met12 (HHGSDHERNYIY, mMet).18
Tissue Collection
For protein and RNA analyses, retinal tissue and RPE-choroid complexes were carefully isolated as previously described19,20 at various time points. Briefly, rats were euthanized, and eyes were immediately enucleated and placed in a dish. The connective tissue and muscle were removed from the back of the eye under a dissecting microscope. The cornea and lens were removed to form an eyecup. The retina was dissected from the RPE, cut from the connection to the optic nerve head, and pulled away from the rest of the eyecup. The RPE-choroid was then carefully scraped from the sclera with a pair of flat top forceps. Retina samples were stored individually. For protein work, RPE samples from four eyes were pooled. For PCR, the RPE samples were individually analyzed.
Caspase 8 Activity Assay
Dissected retina or RPE-choroid were homogenized using a membrane disruptor (sonic dismembrator; Fisher Scientific, Hercules, CA, USA) at 30% power for 15 pulses in lysis buffer containing protease inhibitor (20 mM MOPS, pH 7.0; 2 mM EGTA; 5 mM EDTA; 0.1% Triton X-100; complete protease inhibitor tablet; Roche, Indianapolis, IN, USA). The homogenates were subsequently centrifuged at 10,000 g for 15 minutes at 4°C. The protein concentration of the supernatant was then measured using an assay kit (Dc Protein Assay; Bio-Rad Laboratories, Hercules, CA, USA). Caspase 8 activity was measured using a luminescent assay kit (Caspase-Glo 8 Assay; Promega, Madison, WI, USA) according to the manufacturer's instructions. Briefly, 100 μg of cytosolic protein was incubated with substrate in a white-walled 96-well plate (Greiner Bio-One, Monroe, NC, USA) at room temperature for 1 hour. The untreated retina and RPE samples and no-tissue blank wells served as controls. Luminescence was measured in a plate reader luminometer (Turner Biosystems, Sunnyvale, CA, USA).
Western Blot Analysis
Proteins were separated by 4% to 15% SDS-PAGE (Tris-HCl Ready Gels; Bio-Rad Laboratories) and transferred to polyvinylidene fluoride membranes (Bio-Rad Laboratories). The membranes were incubated overnight with primary antibodies: cleaved caspase 8 (NB100-56116; Novus Biologicals, Littleton, CO, USA) and GAPDH (MA5-15738; Thermo Scientific, Rockford, IL, USA). Secondary polyclonal goat anti-immunoglobulin antibodies were from Dako (P0447, P0448; Glostrup, Denmark). Detection was by chemiluminescence substrate (SuperSignal West Dura Substrate; Thermo Scientific) according to the manufacturer's protocols. Quantitative densitometry of the immunoblots was performed using ImageJ software (http://rsb.info.nih.gov/ij/index.html, provided in the public domain by the National Institutes of Health, Bethesda, MD, USA) and expressed as the mean density (±SD) from replicate experimental groups. All experiments were performed a minimum of three times.
Real-Time Polymerase Chain Reaction
The rat retinas and RPE were harvested at 1 and 3 days after NaIO3 injection. For the RT-PCR experiments, RPE from two rats were pooled together as one sample, whereas retinas were collected separately. Total RNA was isolated using a purification kit (RNeasy Mini Kit; Qiagen, Germantown, MD, USA). Five hundred nanograms of total RNA was converted into cDNA with a reverse transcriptase kit (SuperScript III; Invitrogen, Carlsbad, CA, USA). The expression level of Fas, FasL, caspase 3, and protein receptor interacting serine/threonine kinase 3 (RIPK3) were evaluated using a thermal cycler (Bio-Rad). Specific primers were as follows: Fas primers 5′-ATG AGA TCG AGC ACA ACA GC-3′ (forward), 5′-TTA AAG CTT GAC ACG CAC CA-3′ (reverse); FasL 5′-TTT CTC CTG AGA CTG CAT CA-3′ (forward), 5′-CTC CCA TAA CAG AGG TCC AC-3′ (reverse); caspase 3 primers 5′-GAC AAC AAC GAA ACC TCC GT-3′ (forward), 5′-GAC TTC GTA TTT CAG GGC CA-3′ (reverse); RIPK3 primers 5′-GAG CGA GCA TCC TTC CAA AC-3′ (forward), 5′-CGC ACC ATT GAG CCA TAA CTT-3′ (reverse); HPRT 5′-GCA GAC TTT GCT TTC CTT GG-3′ (forward), 5′-CCG CTG TCT TTT AGG CTT TG-3′ (reverse). Target gene expressions were normalized to the expression of HPRT and fold change values were calculated using the comparative Ct method.
Immunohistochemistry on Whole Mount of Retina and RPE
For whole mount analyses, eyes were enucleated and fixed with 10% buffered neutral formalin fixative (Eng Scientific, Inc., Gibbstown, NJ, USA) at room temperature for 2 hours. The connective tissue, muscle, and optic nerve were removed from the back of the eye, and the cornea and lens were removed to form an eyecup. Eight radial incisions were made, and the retina was carefully dissected off the PRE/choroid under a dissecting microscope and its connection to the optic nerve severed. The retina and the RPE-choroid-sclera complexes, now as separate samples, were further fixed in round-bottom microcentrifuge tubes at room temperature for 1 hour. Samples were carefully washed with PBS three times and then incubated for 1 hour with blocking solution (10% goat serum with 0.3% Triton- X-100 in PBS) before they were incubated with the primary antibodies at 4°C overnight for RPE flat mounts and over 2 nights for retina flat mounts. Primary antibodies used were the following: Iba1, 1:100 (NB100-1028; Novus Biologicals) and ZO-1, 1:100 (61-7300; Invitrogen, Camarillo, CA, USA). After being incubated with secondary antibodies overnight at 4°C, the retina and the RPE-choroid-sclera complex were mounted separately on glass slides (Fisher Scientific, Pittsburgh, PA, USA). Confocal images were collected using a confocal microscope (Leica SP5; Leica Corp., Wetzlar, Germany).
Histology and Immunohistochemistry on Retinal Sections
Eyes were harvested at 10 hours, 7 days, 1 month, and 2 months after NaIO3 injection and fixed in 10% formalin overnight followed by paraffin embedding. The 12 o'clock position of each eye was marked before enucleation for orientation. Paraffin sections (6 μm) were obtained using a microtome (Shandon AS325; Thermo Scientific, Cheshire, England). Only sections crossing the optic nerve for each eye were used. Hematoxylin (Fisher Scientific) and eosin (Fisher Scientific) staining was performed, and images were captured with a microscope (Leica DM6000; Leica Corp.). For immunohistochemistry, sections were blocked with 10% goat serum (Sigma-Aldrich Corp.) in 0.2% Triton X-100 (Sigma-Aldrich Corp.) PBS for 1 hour after de-paraffinization and incubated with primary antibodies in a humidity chamber overnight at 4°C. Primary antibodies were as follows: Fas, 1:100 (Santa Cruz, Dallas, TX, USA); FasL, 1:100 (ab15285; Abcam, Cambridge, MA, USA); RPE65, 1:200 (a kind gift from Debra Thompson, University of Michigan, Ann Arbor, Michigan, USA); high mobility group box 1 (HMGB1), 1:200 (NB100-2322S; Novus Biologicals). After washing and incubation for 1 hour at room temperature with secondary antibodies, sections were counterstained with ProLong Gold with 4′,6-diamidino-2-phenylindole (DAPI, Invitrogen) to reveal cell nuclei. Images were obtained using a confocal microscope (Leica SP5 microscope; Leica Corp.) and were taken at the comparable area of sections. All images in each individual experiment were acquired with a fixed detection gain.
Quantification of Extent of Retinal Damage
To quantify the extent of retinal injury seen on histologic samples we counted the number of foci of outer retinal collapse at three prespecified locations: 500 μm above and below the optic nerve and crossing the optic nerve. In addition, in the section crossing the nerve, we measured the distance of the farthest focus of collapse from the optic nerve and expressed this as a percentage of the total distance from the optic nerve to the farthest edge of the retina. Between six and eight samples were analyzed for each group, experimental and control.
Fundus Photography and Fluorescein Angiography
Rats were anesthetized and the pupils were dilated as previously described.19,20 Color fundus photography and fluorescein angiography were performed using a fundus imaging system (Micron III system; Phoenix Research Labs, Pleasanton, CA, USA). For fluorescein angiography, rats received intraperitoneal injection of 10% fluorescein sodium at a dose of 0.8 mL/kg (Akorn). The fluorescein images were taken immediately after administration and followed for 5 minutes.
Spectral-Domain Optical Coherence Tomography
Rats were anesthetized and pupils were dilated as previously described.19 Optical coherence tomography (OCT) was performed with an SD-OCT system (Bioptigen, Inc., Durham, NC, USA). Volume analyses were performed using 100 horizontal raster and consecutive B scan lines composed of 1200 A scans. The volume size was 1.6 × 1.6 mm. Both eyes of each rat were examined before treatment and at 2 weeks and 1 month post NaIO3 injection.
Statistical Analysis
Statistical analysis comparing groups was performed using 2-tailed paired Student's t-tests assuming equal variance. Nonparametric Kruskal-Wallis test was used for Western blotting analysis. Results are expressed as mean ± standard deviation. Differences were considered significant at P < 0.05.
Results
NaIO3 Results in Activation of Fas-Mediated Death Pathways
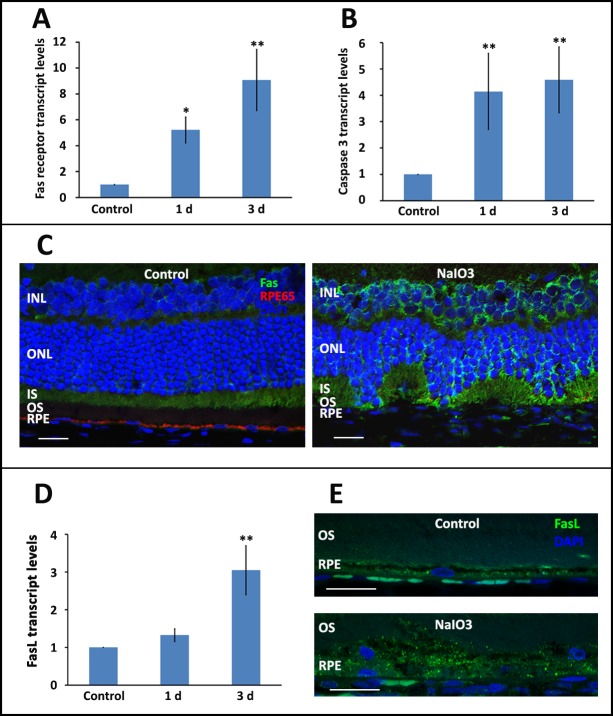
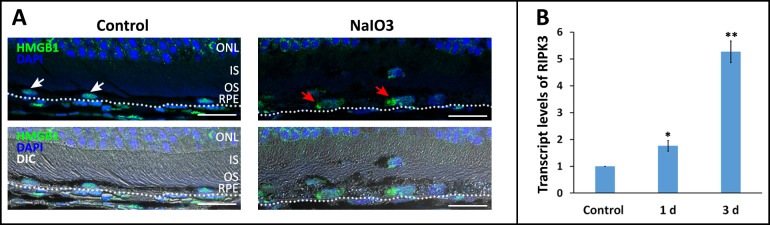
We first sought to demonstrate activation of the Fas pathway in the outer retina after NaIO3 injury. Fas transcript levels in rat the retina were increased approximately 5-fold by 1 day and up to 8-fold by 7 days after NaIO3 exposure (Fig. 1A). This was also associated with elevated transcript level of the pro-apoptotic gene encoding for caspase 3, which showed a nearly 5-fold increase after NaIO3 exposure (Fig. 1B). Immunohistochemistry confirmed this increased expression with localization to the middle and outer retina (Fig. 1C). Interestingly, we did not see an increase in Fas transcript level in the RPE after exposure to NaIO3. Rather, we saw an increase in the expression of the FasL (Figs. 1D, 1E). It has been previously demonstrated that NaIO3 results in the activation of necroptosis in the RPE,14 a process that can be induced through the activated Fas receptor.21,22 A marker of necroptosis activation in the RPE is the translocation of the alarmin protein HMGB1 from the nucleus to the cytoplasm.14,23 We recapitulate this finding (Fig. 2A). We also detect a significant increase in the transcript level of the gene encoding for the key necroptosis protein RIPK3 (Fig. 2B). A further hallmark of Fas activation is an increase in the level of activated caspase 8, as this is the first downstream target of the activated Fas receptor. We demonstrate a 30% and 70% increase in retinal and RPE caspase 8 activity levels after NaIO3 treatment, respectively. Taken together, these data are all consistent with the activation of Fas in the outer retina and RPE after exposure to NaIO3.
Figure 1.
Systemic administration of NaIO3 results in Fas activation in the retina and RPE. (A, B) There was a significant increase in the level of Fas receptor and caspase 3 transcript at 1 and 3 days post exposure to NaIO3. (C) The increased Fas-receptor transcript resulted in increased Fas receptor in the middle and outer retinal layers, as seen on immunohistochemistry. (D, E) There was an increased level of FasL in the RPE at both the transcript and protein levels, as detected by RT-PCR and immunohistochemistry, respectively. Of note, the isotype control did not show any nonspecific staining. *P < 0.05, **P < 0.01. Scale bars: 20 μm. INL, inner nuclear layer; ONL, outer nuclear layer; IS, inner segment; OS, outer segment; RPE65, retinal pigment epithelium protein of 65-kD molecular weight.
Figure 2.
Systemic administration of NaIO3 results in activation of necroptosis in the RPE. (A) Immunohistochemistry shows the translocation of HMGB1 protein out of the nucleus (red arrows) 3 days after systemic administration of NaIO3. By contrast, control eyes did not show any translocation of the HMGB1 (white arrows). (B) NaIO3 treatment also resulted in increased expression of RIPK3. **P < 0.01. ONL, outer nuclear layer; IS, inner segment; OS, outer segment. Scale bars: 20 μm.
Met12 Prevents NaIO3-Induced RPE and Photoreceptor Death
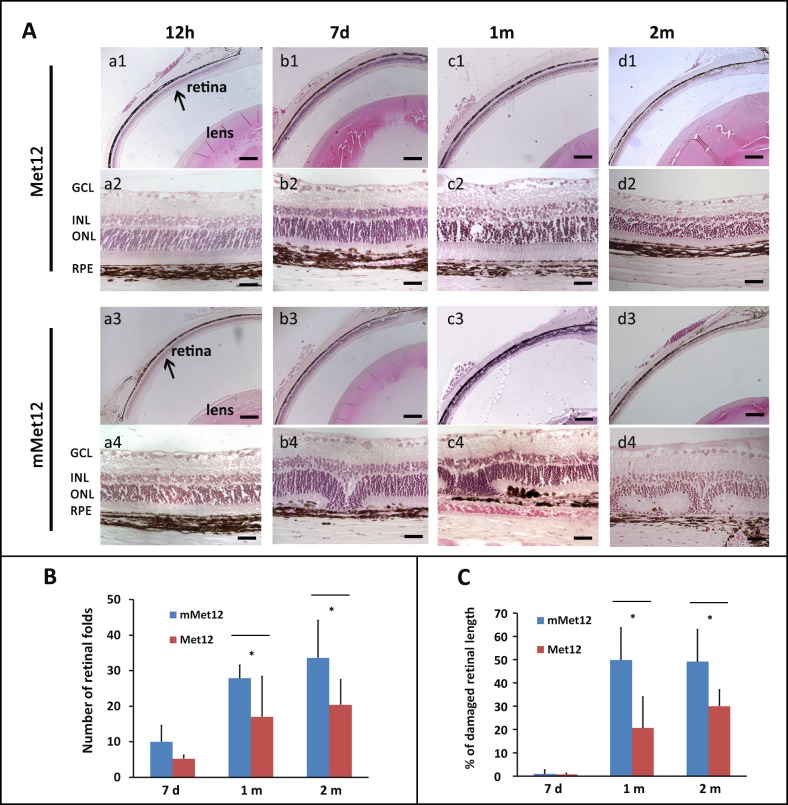
To explore the potential protective effect of Fas-receptor inhibition on the RPE and retina against NaIO3-induced injury, we pretreated the eyes of Brown-Norway rats with the small peptide antagonist of the Fas receptor, Met12, or an inactive scrambled peptide, mMet12,18 prior to systemic exposure to NaIO3. As expected, early (12 hours) after NaIO3 administration, there was minimal RPE or photoreceptor damage evident regardless of whether the eye was pretreated with either Met12 or mMet12 (Figs. 3A, a1–a4). Occasional foci of RPE disruption were detected, with no overlying disruption of the retinal contour. However, by 7 days after exposure to NaIO3, there was significant disruption of the RPE with overlying photoreceptor death and outer retinal collapse in the mMet12-treated eyes (Figs. 3A, b3–b4). In contrast, the Met12-treated eyes showed significant preservation of the RPE and retinal structure (Figs. 3A, b1–b2). There was progression of the damage by 1 month after NaIO3 exposure that appeared to stabilize by 2 months post NaIO3 exposure (Figs. 3A, c1–d4).
Figure 3.
Systemic administration of NaIO3 results in significant degeneration of the RPE and photoreceptors, which is prevented by pretreatment with the small peptide antagonist of the Fas receptor, Met12. (A) Low- and high-power photomicrographs of retinas from animals at various time points after systemic exposure to NaIO3. Eyes were pretreated with intravitreal injection of either Met12 (a1–d1, a2–d2) or an inactive, scrambled peptide, mMet12 (a3–d3, a4–d4). NaIO3 exposure resulted in significant disruption of the RPE by 7 days in the mMet12-treated eyes (b3, b4), which was prevented by Met12 treatment (b1, b2). By 1 month post exposure to the NaIO3, the overlying retina was severely degenerated in the mMet12-treated eyes (c3, c4, d3, d4) but not in the Met12-treated eyes (c1, c2, d1, d2). (B) There was a significant reduction in the number of retinal folds, or (C) extent of retina damaged as measured from the optic nerve after NaIO3 exposure in eyes that were pretreated with Met12 as compared to mMet12. *P < 0.05). Scale bars: low magnification images 200 μm, high magnification images 25 μm. GCL, ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer.
Quantification of the extent of outer retinal damage after NaIO3 exposure showed significant protection by Met12 as compared to mMet12 (Figs. 3B, 3C). Of note, in each animal, one eye was treated with Met12 and the fellow eye treated with mMet12. Analysis between Met12 versus mMet12 was done for each animal, thus ensuring that there was equal exposure to NaIO3. We found that 7 days after NaIO3 exposure there were fewer foci of outer retinal collapse in the Met12-treated eyes, but this did not reach statistical significance (Fig. 3B, P = 0.1). However, by 1 and 2 months post NaIO3 exposure, the difference in the number of retinal folds between the Met12- versus mMet12-treated groups became more apparent, with approximately a 30% to 50% reduction in the number of retinal folds in the Met12- versus mMet12-treated eyes (P = 0.047 and P = 0.026, respectively). We also compared the extent of outer retinal damage as measured by the farthest location of a retinal fold from the optic nerve (Fig. 3C). The extent (length) of damaged retina was also significantly reduced by Met12 treatment at 1 and 2 months as compared to mMet12 treatment (P = 0.036 and P = 0.017, respectively).
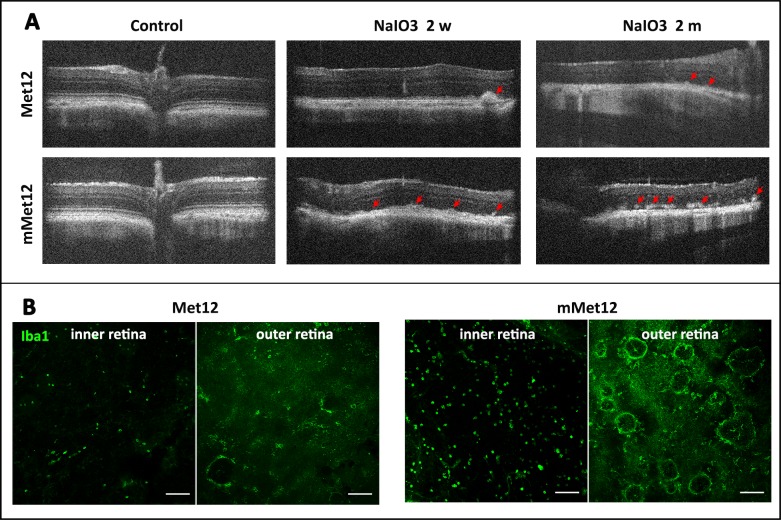
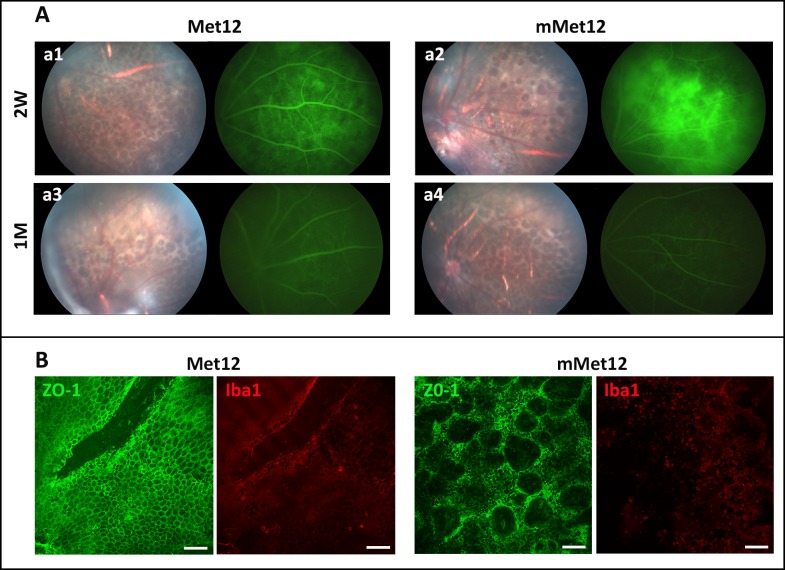
The protection of RPE and photoreceptor structure by Met12 was also detected by OCT. There was normal RPE and retinal architecture prior to NaIO3 exposure (Fig. 4A). There was significant disruption of the RPE and outer retinal architecture on OCT by 2 weeks after NaIO3 administration, which stabilized by 1 month. The OCT of NaIO3-damaged eyes showed hyperreflective foci just above the RPE layer, consistent with the foci of outer retinal collapse (Fig. 4A). There were fewer such foci in the Met12- versus the mMet12-treated eyes. We also assessed for the activation of microglia and or infiltration of macrophages, which is often associated with outer retinal injury and activation of Fas-mediated cell death.8,24,25 We observed that exposure to NaIO3 resulted in more Iba1-positive cells in retina flat mounts, and that this was significantly reduced by Met12- as compared to mMet12-treated eyes (Fig. 4B). The Iba1-positive cells appeared to concentrate in rings that corresponded to areas of photoreceptor cell death and outer retinal collapse.
Figure 4.
Met12 reduces outer retinal degeneration and immune cell infiltration induced by systemic exposure to NaIO3. (A) The protective effect of Met12 can be visualized in vivo using OCT. The red arrows show the foci of outer retinal collapse. (B) Flat mounts of the retina stained with Iba1 demonstrate that NaIO3 exposure results in marked increase in positive-staining cells that is reduced by Met12, but not mMet12, treatment. The Iba1-positive cells are located in both the inner and outer retina. The circular pattern of staining in the outer retina corresponds to areas of outer retinal collapse. Scale bars: 100 μm.
To further assess the extent of RPE damage in the Met12-treated versus mMet12-treated eyes in vivo we performed fluorescein angiography to identify areas of damaged RPE as evidenced by hyperfluorescence from fluorescein leaking through the damaged RPE from the underlying choroidal circulation. At 2 weeks after NaIO3 exposure, we detected severe and extensive hyperfluorescence in mMet12-treated eyes as compared to Met12-treated eyes (Fig. 5A). As is typical for this model,14–16 the hyperfluorescence resolved by 1 month, presumably due to scarring at the level of the RPE. Ex vivo analysis of the RPE with flat mount and ZO-1 staining showed that there was marked disruption of the normal RPE architecture after NaIO3 exposure that was not prevented with mMet12 treatment (Fig. 5B). However, in the eyes treated with Met12 there was significant preservation of the normal RPE architecture and cell density (Fig. 5B), consistent with the reduced hyperfluorescence seen on the fluorescein angiography.
Figure 5.
Intravitreal injection of Met12 prevented the loss of RPE induced by the systemic exposure to NaIO3. (A) Fundus photography and fluorescein angiography 2 weeks after NaIO3-exposure RPE loss and hyperfluorescence that is prevented by Met12 treatment (a1) but not mMet12 (a2). By 1 month after NaIO3 exposure, the hyperfluorescence on fluorescein angiography is no longer seen in the mMet12-treated eyes (a4), presumably due to scarring. (B) Flat mounts of the RPE taken 1 month after systemic exposure to NaIO3 stained with ZO-1 show that Met12 preserves the normal hexagonal architecture of the cells, while mMet12 treatment does not. Staining of the flat mounts with Iba1 shows many more microglia/macrophage cells in the mMet12-treated eyes versus Met12-treated eyes. Scale bars: 100 μm.
Met12 Prevents the NaIO3-Induced Activation of Fas
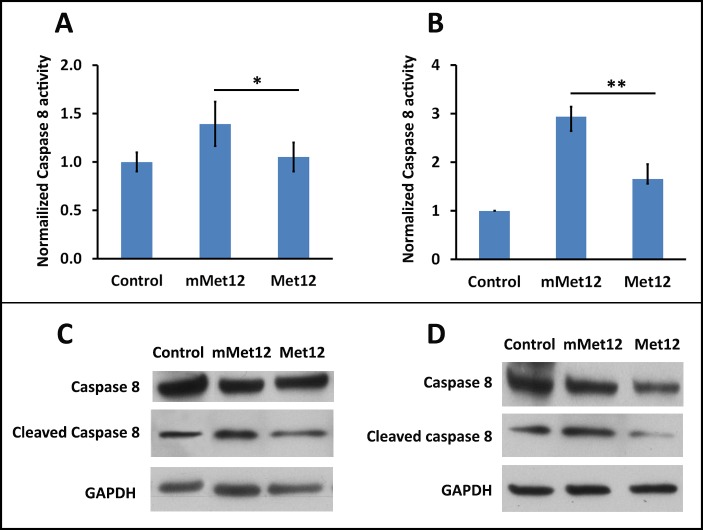
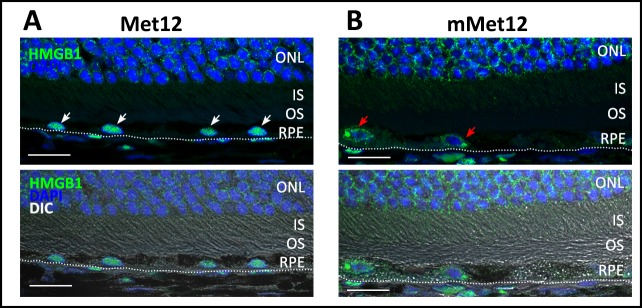
Met12 exerts a protective effect via the inhibition of Fas-receptor activation, and prevention of the resultant activation of death pathways.18 As such, we wanted to demonstrate that the protective effect of Met12 in the NaIO3 model of oxidative stress is associated with a reduction in the extent of Fas-pathway activation. To validate the hypothesis that Met12 was exerting its protective effect on the RPE and photoreceptors via inhibition of the Fas receptor, we tested the prediction that Met12 treatment would result in decreased caspase 8 activity levels. Figure 6 shows the caspase 8 activity levels in the RPE and retina after NaIO3 exposure and treatment with either Met12 or mMet12 (Figs. 6A, 6B, respectively). As can be seen, there is significant increase in the caspase 8 activity level after exposure to NaIO3 that is not reduced by mMet12 treatment. In contrast, the caspase 8 activity was reduced to near baseline levels by Met12. The effect of Met12 on caspase 8 is also demonstrated by Western blot analysis, which shows marked reduction in the level of cleaved (active) caspase 8 in the Met12- versus mMet12-treated eyes (Figs. 6C, 6D). Furthermore, Met12 treatment prevented the NaIO3-induced translocation of HMBG1 from the nuclei of RPE cells, consistent with Met12 preventing the Fas-induced activation of necroptosis (Fig. 7).
Figure 6.
Intravitreal injection of Met12 significantly reduced the NaIO3-induced activation of the Fas receptor. Caspase 8 cleavage is the first downstream effect of the activated Fas receptor. Intravitreal injection of Met12 prevented the cleavage and activation of caspase 8, as detected by caspase 8 activity assay and Western blot in the RPE (A, C) and the retina (B, D), whereas mMet12 did not.
Figure 7.
Met12 treatment prevented the NaIO3-induced translocation of HMGB1 protein from the nucleus of the RPE cells (A) (white arrows), whereas mMet12 did not (B) (red arrows). Scale bars: 20 μm.
Discussion
In this study we use the NaIO3 model of RPE injury to study the effect of oxidative stress on the RPE and photoreceptors. This model recapitulates a key aspect of the AMD phenotype, which is primary damage and death of the RPE followed by a secondary death of the overlying photoreceptors. We demonstrate that after systemic administration of NaIO3, there is activation of Fas-mediated cell death in both the RPE and retina, and that blocking this activation with the small peptide, Met12, significantly protects these two cell types against damage. Collectively, the data presented in this study confirm that inhibition of Fas-pathway activation results in significant protection against NaIO3-induced damage of the RPE and photoreceptors.
In AMD there are multiple stressors acting upon the RPE and retina, but the ultimate mechanism by which the RPE and photoreceptors die is still unclear. Studies have shown the activation of both apoptosis and necroptosis in these cells,9,14 but the upstream activator(s) of these death pathways has not been identified. Previous work from various laboratories has shown that Fas induces cell death under a variety of disease conditions.8,18,26 In many cases, this death occurs through the induction of the receptor-mediated apoptosis cascade. However, recent work has also shown that necroptosis can be activated in both the RPE and photoreceptors, which can also be induced through the activation of the Fas receptor.21,22
Our findings support the hypothesis that oxidative stress leads to the death of the RPE and photoreceptors via activation of the Fas receptor and subsequent activation of the necroptosis and apoptosis cascades. Oxidative stress, including that created by NaIO3, has been shown to induce RPE death via necroptosis,14,17 and we postulated that this process is Fas-mediated. Our finding of reduced HMGB1 translocation in the RPE with Met12 treatment strongly supports the hypothesis that RPE necroptosis is regulated by Fas. In AMD, death of the photoreceptor is often considered as a secondary effect of RPE loss. Our data show that inhibiting Fas receptor prevents the NaIO3-induced death of the RPE and photoreceptors. Preservation of the photoreceptors in this model could be attributed to preservation of the RPE, as well as to direct inhibition of Fas-mediated photoreceptor cell death.
Our previous work has shown the utility of Met12 in preventing photoreceptor cell death after retinal detachment.18 In this work, we extend the demonstration of the protective effect of Met12 beyond just the photoreceptors during retinal detachment, and show preservation of both the RPE and the photoreceptors in the context of severe oxidative stress. These findings strongly support targeting the Fas receptor as a novel therapeutic point of intervention for disease-related oxidative damage to the RPE. Met12 is a small peptide antagonist of the Fas receptor derived from the alpha chain of the Met oncogene, which encodes for a tyrosine kinase receptor composed of an extracellular chain and a transmembrane chain.27 The extracellular chain contains an amino terminal sequence motif, TyrLeuGlyAla, which has high homology with FasL. The oncogenic capacity of Met derives, in part, from its ability to bind with Fas directly, thus preventing receptor activation by FasL and inhibiting activation of both necroptosis and apoptosis.
Although we achieved significant protection by Met12, the effect was not total. One potential explanation is that the delivery of Met12 was not ideal. We administered Met12 via intravitreal injection at a dose previously found to be protective of photoreceptors during retinal detachment. This may not have resulted in optimal drug levels in the RPE and photoreceptor layers for protection against NaIO3-induced oxidative stress. Future work would be to analyze and optimize the concentration of drug and timing of delivery required to achieve maximal protection. Another potential contributor to the cell death may be the induction of pathways that are not Fas-receptor mediated, such as the intrinsic apoptosis pathway. Sodium iodate has been found to be directly toxic to photoreceptors28 and may be acting through activation of these non-Fas-dependent pathways. Additional validation of Fas-receptors' role in mediating RPE and photoreceptor death would be to test the effect of NaIO3 administration in the lpr and gld mouse strains, which contain defective Fas receptor and FasL, respectively.
In the experiments described in this report, the Met12 was administered prior to the induction of the oxidative stress. It remains to be determined whether administration of the drug after the induction of oxidative stress will also protect the RPE and photoreceptors and, if so, to what extent. We would expect that Met12 would not reverse death that has already occurred but would prevent progression of the RPE loss and secondary photoreceptor degeneration.
Our data demonstrate that there is activation of Fas-mediated cell death pathways in the RPE and photoreceptors after NaIO3 administration. An unresolved question, though, is the source of the ligand activating the Fas receptor. We find that the transcript level for FasL increased in the RPE, suggesting these cells as a potential source of the ligand. Expression of FasL by the RPE has been previously described29 and is thought to contribute to the regulation of the immune surveillance under homeostatic and stress conditions. Increases in serum FasL has also been demonstrated in patients with AMD,30 suggesting that this might serve as a biomarker for RPE cells under stress. In addition, we see a significant increase in the amount of Iba1-positive cells in the intra- and sub-retinal space after NaIO3 exposure. This suggests a strong activation of retinal microglia and or infiltration of macrophages, both of which could be a potential source of the Fas ligand. Intrinsic retinal and infiltrating immune cells and Fas-mediated cell death has been implicated in the progression of RPE death in AMD.31–33 Our results support this conclusion, particularly our ability to preserve RPE using a Fas-receptor antagonist. Interestingly, we see an increase in the transcript for FasL in the RPE cells. This has been reported to occur in the apolipoprotein E model of AMD and has been postulated to represent a protective mechanism by the RPE cells to induce apoptosis in infiltrating immune cells that would otherwise accelerate RPE loss.29 This raises the concern that Fas inhibition could potentially exacerbate the RPE death by preventing apoptosis of the infiltrating macrophages. Against this argument, however, is the fact that we observe fewer Iba-positive cells and less RPE loss in Met12- as compared to mMet12-treated eyes. This raises the possibility that Fas inhibition might not only work to directly antagonize the Fas-receptor activation of death pathways, but that Met12 also prevents activation of inflammatory signaling that stimulates the microglial/macrophage response in the first place. This has been suggested to be the case in a model of autoimmune uveitis, in which the inflammation was greatly reduced by Met12 treatment.34
In summary, our results strongly support the hypothesis that Fas contributes to RPE and photoreceptor cell death during periods of oxidative stress and that this death can be pharmacologically prevented using a competitive inhibitor to the Fas receptor. We suggest that Fas/FasL should be considered as potential therapeutic targets for preventing progression of RPE and photoreceptor cell death in diseases with a large component of oxidative stress.
Acknowledgments
Supported by the National Eye Institute Grant EY-020823 (DNZ) and Core Center EY007003 (University of Michigan Kellogg Eye Center). This study was also supported (in part) by a Sybil B. Harrington Physician Scientist Award From Research to Prevent Blindness (New York, NY, USA).
Disclosure: J. Xiao, None; J. Yao, None; L. Jia, None; C. Lin, None; D.N. Zacks, ONL Therapeutics (C, I, S), P
References
- 1. Jager RD,, Mieler WF,, Miller JW. Age-related macular degeneration. N Engl J Med. 2008; 358: 2606–2617. [DOI] [PubMed] [Google Scholar]
- 2. Lambert NG,, ElShelmani H,, Singh MK,, et al. Risk factors and biomarkers of age-related macular degeneration. Prog Retin Eye Res. 2016; 54: 64–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Tan PL,, Bowes Rickman C,, Katsanis N. AMD and the alternative complement pathway: genetics and functional implications. Hum Genomics. 2016; 10: 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Schwartz SG,, Hampton BM,, Kovach JL,, Brantley MA., Jr. Genetics and age-related macular degeneration: a practical review for the clinician. Clin Ophthalmol. 2016; 10: 1229–1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Shaw PX,, Stiles T,, Douglas C,, et al. Oxidative stress, innate immunity, and age-related macular degeneration. AIMS Mol Sci. 2016; 3: 196–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wubben TJ,, Besirli CG,, Zacks DN. Pharmacotherapies for retinal detachment. Ophthalmology. 2016; 123: 1553–1562. [DOI] [PubMed] [Google Scholar]
- 7. Chinskey ND,, Besirli CG,, Zacks DN. Retinal cell death and current strategies in neuroprotection. Curr Opin Ophthalmol. 2014; 25: 228–233. [DOI] [PubMed] [Google Scholar]
- 8. Matsumoto H,, Murakami Y,, Kataoka K,, et al. Membrane-bound and soluble Fas ligands have opposite functions in photoreceptor cell death following separation from the retinal pigment epithelium. Cell Death Dis. 2015; 6: e1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Murakami Y,, Notomi S,, Hisatomi T,, et al. Photoreceptor cell death and rescue in retinal detachment and degenerations. Prog Retin Eye Res. 2013; 37: 114–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Pennesi ME,, Neuringer M,, Courtney RJ. Animal models of age related macular degeneration. Mol Aspects Med. 2012; 33: 487–509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Penn J,, Mihai DM,, Washington I. Morphological and physiological retinal degeneration induced by intravenous delivery of vitamin A dimers in rabbits. Dis Model Mech. 2015; 8: 131–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Obata R,, Yanagi Y,, Tamaki Y,, Hozumi K,, Mutoh M,, Tanaka Y. Retinal degeneration is delayed by tissue factor pathway inhibitor-2 in RCS rats and a sodium-iodate-induced model in rabbits. Eye (Lond). 2005; 19: 464–468. [DOI] [PubMed] [Google Scholar]
- 13. Monés J,, Leiva M,, Peña T,, et al. Swine model of selective geographic atrophy of outer retinal layers mimicking atrophic AMD: a phase I escalating dose of subretinal sodium iodate. Invest Ophthalmol Vis Sci. 2016; 57: 3974–3983. [DOI] [PubMed] [Google Scholar]
- 14. Hanus J,, Anderson C,, Sarraf D,, Ma J,, Wang S. Retinal pigment epithelial cell necroptosis in response to sodium iodate. Cell Death Discov. 2016; 2: 16054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kannan R,, Hinton DR. Sodium iodate induces retinal degeneration: new insights from an old model. Neural Regen Res. 2014; 9: 2044–2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kiuchi K,, Yoshizawa K,, Shikata N,, Moriguchi K,, Tsubura A. Morphologic characteristics of retinal degeneration induced by sodium iodate in mice. Curr Eye Res. 2002; 25: 373–379. [DOI] [PubMed] [Google Scholar]
- 17. Hanus J,, Anderson C,, Wang S. RPE necroptosis in response to oxidative stress and in AMD. Ageing Res Rev. 2015; 24 Pt B: 286–298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Besirli CG,, Chinskey ND,, Zheng QD,, Zacks DN. Inhibition of retinal detachment-induced apoptosis in photoreceptors by a small peptide inhibitor of the Fas receptor. Invest Ophthalmol Vis Sci. 2010; 51: 2177–2184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Yao J,, Jia L,, Feathers K,, et al. Autophagy-mediated catabolism of visual transduction proteins prevents retinal degeneration. Autophagy. 2016; 12: 2439–2450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Yao J,, Jia L,, Shelby SJ,, et al. Circadian and noncircadian modulation of autophagy in photoreceptors and retinal pigment epithelium. Invest Ophthalmol Vis Sci. 2014; 55: 3237–3246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Smith CC,, Yellon DM. Necroptosis, necrostatins and tissue injury. J Cell Mol Med. 2011; 15: 1797–1806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Degterev A,, Huang Z,, Boyce M,, et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol. 2005; 1: 112–119. [DOI] [PubMed] [Google Scholar]
- 23. Murakami Y,, Matsumoto H,, Roh M,, et al. Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsRNA-induced retinal degeneration. Cell Death Differ. 2014; 21: 270–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Scholz R,, Sobotka M,, Caramoy A,, Stempfl T,, Moehle C,, Langmann T. Minocycline counter-regulates pro-inflammatory microglia responses in the retina and protects from degeneration. J Neuroinflammation. 2015; 12: 209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Zhang M,, Xu G,, Liu W,, Ni Y,, Zhou W. Role of fractalkine/CX3CR1 interaction in light-induced photoreceptor degeneration through regulating retinal microglial activation and migration. PLoS One. 2012; 7: e35446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chang Q,, Peter ME,, Grassi MA. Fas ligand-Fas signaling participates in light-induced apoptotic death in photoreceptor cells. Invest Ophthalmol Vis Sci. 2012; 53: 3703–3716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Zou C,, Ma J,, Wang X,, et al. Lack of Fas antagonism by Met in human fatty liver disease. Nat Med. 2007; 13: 1078–1085. [DOI] [PubMed] [Google Scholar]
- 28. Wang J,, Iacovelli J,, Spencer C,, Saint-Geniez M. Direct effect of sodium iodate on neurosensory retina. Invest Ophthalmol Vis Sci. 2014; 55: 1941–1952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Levy O,, Calippe B,, Lavalette S,, et al. Apolipoprotein E promotes subretinal mononuclear phagocyte survival and chronic inflammation in age-related macular degeneration. EMBO Mol Med. 2015; 7: 211–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Jiang S,, Moriarty-Craige SE,, Li C,, et al. Associations of plasma-soluble Fas ligand with aging and age-related macular degeneration. Invest Ophthalmol Vis Sci. 2008; 49: 1345–1349. [DOI] [PubMed] [Google Scholar]
- 31. Zhao H,, Roychoudhury J,, Doggett TA,, Apte RS,, Ferguson TA. Age-dependent changes in FasL (CD95L) modulate macrophage function in a model of age-related macular degeneration. Invest Ophthalmol Vis Sci. 2013; 54: 5321–5331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Kim Y,, Tarallo V,, Kerur N,, et al. DICER1/Alu RNA dysmetabolism induces Caspase-8-mediated cell death in age-related macular degeneration. Proc Natl Acad Sci U S A. 2014; 111: 16082–16087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wang Y,, Shen D,, Wang VM,, et al. Enhanced apoptosis in retinal pigment epithelium under inflammatory stimuli and oxidative stress. Apoptosis. 2012; 17: 1144–1155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Jiang G,, Wang Y,, Yun J,, et al. HMGB1 release triggered by the interaction of live retinal cells and uveitogenic T cells is Fas/FasL activation-dependent. J Neuroinflammation. 2015; 12: 179. [DOI] [PMC free article] [PubMed] [Google Scholar]