Abstract
Background and aims
CVD risks associated with coronary artery calcification (CAC) and aortic calcification (AC) are well known, but less is known about other calcified arteries. We aimed to assess the associations of arterial calcification in the breast, splenic, and internal and external iliac arteries with CVD risk factors and mortality.
Methods
We conducted a case-cohort study nested in a cohort of 5196 individuals who self-referred or were referred by a health care provider for whole body computed tomography (CT), including a random subcohort (n=395) and total and CVD mortality cases (n=298 and n=90), who died during a median follow-up of 9.4 years. Arterial calcification in the breast, splenic, and internal and external iliac arteries on CT was scored using a simple visual score. AC and CAC were previously measured using the Agatston technique. Logistic regression models were made to study associations of CVD risk factors with calcification in the different vascular beds. Prentice-weighted Cox proportional hazards models adjusted for CVD risk factors, and calcification in other vascular beds, were used to study associations with mortality.
Results
In the subcohort, the mean age was 56.6 years (SD 11.1) and 41.3% were female. The prevalence of calcification on CT, was 11.6% in the splenic, 47.9% in the internal iliac and 9.5% in the external iliac arteries, while 3.7% of women had breast artery calcification (BAC). Calcification in the splenic and iliac arteries was associated with calcification in the abdominal aorta but differentially associated with other CVD risk factors in logistic regression models. The prevalence of BAC was too low to fit these multivariable models. Calcification of the external iliac arteries was significantly associated with both all-cause and CVD mortality, but no longer significant when adjusted for CVD risk factors. Breast artery calcification was associated with both all-cause and CVD mortality independent of CVD risk factors and AAC and CAC (all-cause HR 5.67 [95% CI 1.50–21.41]).
Conclusions
Risk factors associated with calcification, and the association of calcification with risk of mortality differ across vascular beds, possibly reflecting different pathophysiology.
Keywords: Vascular calcification, cardiovascular risk factors, mortality
Introduction
Most research on arterial calcification has focused on intimal calcification arising within atherosclerotic plaques in the coronary arteries, the aorta, and carotid bifurcation.1–3 Intimal calcification is closely associated with traditional cardiovascular risk factors, including smoking and hyperlipidemia.1 However, other patterns of calcification are also recognized and can occur independently, namely calcification occurring in the arterial media and internal elastic lamina.4 These non-atherosclerotic types of calcification are often lumped together under the heading of “medial arterial calcification” (MAC). MAC seems to share some, but not all, risk factors of intimal calcification.5 MAC occurs frequently in patients with diabetes and kidney disease and its prevalence increases with older age6–8 but smoking is associated with a lower prevalence of MAC9–12 and hyperlipidemia does not associate with MAC.8,11 This suggests that MAC is a distinct disease process.13
Classic MAC has a circumferential and continuous “railroad” appearance on radiographs, while intimal calcification is typically spottier, but it remains difficult to distinguish MAC from intimal calcification in vivo. MAC is known to occur frequently in female breast arteries, the carotid siphon and leg arteries.6,7,14 Calcification of the splenic artery is thought to be predominantly medial.15 Only one previous study related cardiovascular risk factors to the presence of calcium in the splenic artery and found a significant unadjusted correlation of glucose levels with splenic artery calcification, but did not study a comprehensive set of risk factors.16
There are little data assessing the relationship of MAC with cardiovascular end-points. The few studies that do exist indicate higher risks.7,11,17 Breast artery calcification (BAC) on mammography has been associated with an increased risk of cardiovascular disease.18 While prior studies have examined the association of BAC on digital mammography with CT-identified coronary atherosclerosis, to our knowledge, no studies have directly examined BAC on computed tomography (CT).19 For the splenic artery, we are not aware of any studies determining associations with clinical outcomes.
Therefore, our first aim was to evaluate the risk factors associated with calcification in a number of small to medium sized arteries, namely the female breast artery, the splenic artery and the internal and external iliac arteries. Our second aim was to determine the associations of arterial calcification in the breast, splenic artery and both internal and external iliac arteries with all-cause and cardiovascular mortality.
Patients and methods
Population
The study population consisted of 5196 community-living individuals that were self-referred or referred by their primary care physician for CT scanning for preventive medicine purposes in San Diego between 2000 and 2003. The study population is described in detail elsewhere.20
For this study, we designed a nested case-cohort study. This is an efficient and frequently used design used to evaluate associations between baseline determinants and outcomes by evaluating a random sample of all participants at baseline (the subcohort) and all cases occurring from the entire cohort.21 This approach made it feasible to validly study calcification in a number of added vascular beds while only measuring them in the subcohort and cases.22 A sample of 395 (7.5%) participants was drawn randomly from all baseline participants, forming the subcohort. We previously showed that sampling fractions above 5% yield valid estimates of HR and SE, when using Prentice-weighting.22 Our cases constituted all deceased participants (n=298), resulting in a total sample size of 670, as some cases were also sampled in the randomly selected subcohort, as expected (Figure 1).
Fig. 1.
Participant flowchart.
Baseline measurement of risk factors
At baseline, all participants provided information on demographics, risk factors and past medical history. Smoking status was self-reported as former, current or never. Height and weight were measured and body mass index (BMI in kg/m2) was calculated. Trained technicians measured blood pressure after at least 5 minutes of rest. Random serum lipid and glucose measurements were obtained by finger-stick using the Cholestec LDX system (Hayward, CA, USA). Baseline measurements are described in more detail previously.20 Diabetes was defined as blood glucose level greater than 200 mg/dl or use of antiglycemic medication. Dyslipidemia was defined as a ratio of total cholesterol to high-density lipoprotein cholesterol greater than 5 or use of a cholesterol-lowering medication. Hypertension was defined as systolic blood pressure greater than 140 mm Hg or diastolic pressure greater than 90 or use of hypertensive medication for this condition.
Assessment of calcification
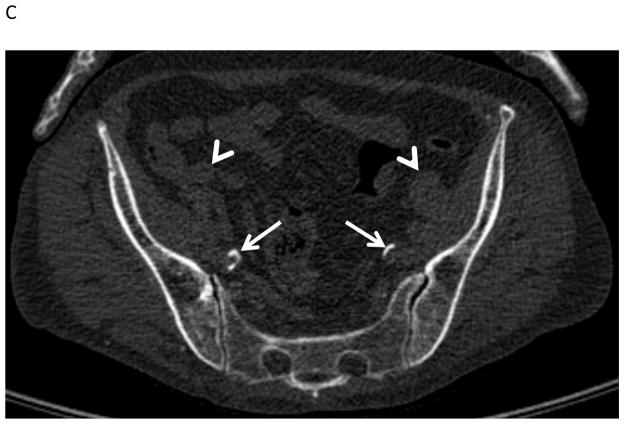
All participants underwent “whole body” CT scans i.e. scanning from the base of the skull to the pubic symphysis. Details on scanning procedures are described elsewhere.3 Within the thoracic aorta, abdominal aorta, carotid, coronary, and iliac arteries (common and external), calcium scores had been calculated previously using the method described by Agatston.23,24 For this study, we used thoracic and abdominal/pelvic CT scans with a slice thickness of 6 mm to measure breast, splenic, external and internal iliac artery calcification in the subcohort and cases only (Figure 2).
Fig. 2. CT images of calcification in the studied vascular beds.
(A) Breast artery calcification, (B) splenic artery calcification, (C) internal and external iliac arteries (calcifications in the internal iliac arteries)
For all participants in the subcohort, and all death cases, additional scoring of the breast (females), splenic, and internal and external iliac arteries was conducted by a researcher (EJEH) blinded to the patients’ clinical data. Calcification in the breast arteries was scored as absent or present. Calcification in the splenic artery and internal and external iliac arteries were scored as absent, mild, moderate, or severe using the following cut-points for artery length affected with calcification: 0, <0.5 cm, 0.5–1.5 cm, and >1.5 cm for the splenic artery and 0, <1cm, 1cm–3cm, and >3cm for the internal and external arteries, respectively. This method is an extension of previous work.16,25 The pre-specified cut-offs are arbitrary. We used the ruler tool in the OsiriX Imaging Software26, but because of the tortuosity of the arteries we evaluated, the length is considered an estimate. Two researchers scored a random sample of 30 scans independently (EJEH and PAdJ). The Cohen’s kappa’s for inter-rater reliability for presence/absence of calcification were 0.43 (95% CI 0.0 – 0.89), 0.47 (95% CI 0.14–0.80), 0.93 (95% CI 0.81–1.06), and 0.59 (95% CI 0.28–0.90) for the breast, splenic, internal iliac and external iliac arteries, respectively. The linear weighted kappa for the grades of calcification were 0.62 (95% CI 0.36–0.87) for the splenic artery, 0.70 (95% CI 0.53–0.87) for the internal iliac artery and 0.52 (95% CI 0.25–0.78) for the external iliac artery.
Cardiovascular and All-Cause Mortality
As described previously, all participants in the parent study were followed longitudinally for mortality20,27 using both the Social Security Death Index and the National Death Index through the 31st of December 2010. Patients were cross-referenced with study records to confirm identity. Death certificates were obtained for deaths ascertained through the Social Security Death Index and study physicians with experience in adjudicating CVD outcomes adjudicated the underlying and contributing causes of death. For the subset who were found to be dead by the National Death Index only, we were provide a short synopsis of cause of death and used this to classify CVD versus non-CVD related deaths. CVD-specific mortality included myocardial infarction, stroke, congestive heart failure, coronary artery disease, and other vascular diseases. Events categorized under “other vascular diseases” were rare individually; examples include rupture of an abdominal aortic aneurysm and death related to peripheral arterial disease complications. A total of 298 deaths occurred during this period, of which 90 were classified as CVD-specific mortality.
Statistical analyses
To identify unique risk factors associated with calcification in the different vascular beds, we conducted a cross-sectional study among the subcohort participants only. Characteristics of the subcohort were described by presence or absence of calcification in the different arteries, as means with standard deviations or percentages with number for continuous and categorical variables, respectively. For non-normally distributed continuous variables, median and interquartile ranges are given. Characteristics were not disaggregated by BAC status for women because the number of women with BAC in the subcohort was too small to be considered reliable (n=6).
Multiple imputation techniques were used to impute missing data in the baseline co-variables used in multivariable analysis28. Data from the full cohort was used for the imputation. In the full cohort, the mean percentage missing of the covariables that were imputed was 7.8%, with a maximum of 22.7% (for measured systolic blood pressure). Analyses were run on 15 imputed datasets and combined to obtain estimates and confidence intervals using Rubin’s rule.29
To assess the risk factors associated with calcification in the different vascular beds, we used logistic regression models with risk factors as independent variables and calcification presence or absence in the respective beds as the dependent variables. We first studied the univariable (crude) associations between each of the risk factors and calcification as the outcome (Models 1). Next, we made a multivariable model including traditional CVD risk factors (smoking status [former, current, never], dyslipidemia [present/absent], BMI [continuous], diabetes [present/absent], and hypertension [present/absent], Model 2). In model 3 the log of 1+ the quantification of the calcification of the coronary arteries and the thoracic and abdominal aorta were added.
To assess associations of calcification in each vascular bed with CVD death and all-cause mortality, we used both the subcohort and the death cases. We used modified Cox proportional hazards models that were adapted for analysis of case-cohort data as described by Prentice et al.21 In model 1, we modeled the presence/absence of calcification in each vascular bed as the independent variable and adjusted for age and sex. In model 2, we additionally adjusted for smoking status, dyslipidemia, BMI, diabetes and hypertension. In a third model, we additionally adjusted for the number of vascular beds (0–5) with evidence of calcification (Agatston score > 0): carotid arteries, thoracic aorta, abdominal aorta, common & external iliac arteries and coronary arteries. To determine whether a graded severity of calcification is associated with the mortality outcomes, we also modeled calcification in the splenic, internal and external iliac arteries by categories of severity, adjusting for variables described in model 2 above. The proportional hazards assumption was assessed by correlation tests of Schoenfeld residuals for the BAC variable and event time, using a procedure adapted to the case-cohort design.30 No violations were detected.
Statistical analyses were performed using R, version 3.2.0 (R Foundation for Statistical Computing, Vienna, Austria.). Add-on packages ‘Amelia’ (1.7.2) and ‘survival’ (2.38-1) used for multiple imputation and Cox proportional hazards regression, respectively.
Results
Among the 395 individuals randomly sampled for inclusion in the subcohort, the mean age at baseline was 56.6 years (SD 11.1) and 41.3% were female. The prevalence of calcification in the subcohort was 11.6% (n=46) for the splenic artery, 47.9% (n=187) for the internal iliac artery and 9.5% (n=37) for the external iliac artery. Among the 163 women, 3.7% (n=6) had BAC. Characteristics of the subcohort by presence of calcium in the different vascular beds are shown in Table 1. Across the different vascular beds, those with artery calcification were older, more frequently male and former smokers, and had higher prevalence of diabetes, hypertension, and dyslipidemia. BMI was similar among those with and without artery calcification.
Table 1.
Subcohort characteristics by presence of calcium in the different vascular beds.
| Characteristic | Total subcohort | Splenic artery calcium | Internal iliac artery calcium | External iliac Artery calcium | |||
|---|---|---|---|---|---|---|---|
| Present | Absent | Present | Absent | Present | Absent | ||
| N | 395 | 46 | 349 | 187 | 203 | 37 | 353 |
| Age (years) | 56.6 (11.1) | 68.4 (9.2) | 55.0 (10.4) | 62.2 (10.0) | 51.2 (9.4) | 67.9 (9.4) | 55.3 (10.6) |
| Sex (% female) | 41.3% (n=163) | 28.3% (n=13) | 43.0% (n=150) | 27.3% (n=51) | 54.7% (n=111) | 32.4% (n=12) | 42.5% (n=150) |
| BMI (kg/m2) | 27.1 (4.5) | 27.6 (3.9) | 27.0 (4.6) | 27.6 (4.4) | 26.7 (4.6) | 26.8 (3.8) | 27.2 (4.6) |
| Current smoking | 8.6% (n=34) | 8.7% (n=4) | 8.6% (n=30) | 11.2% (n=21) | 6.4% (n=13) | 24.3% (n=9) | 7.1% (n=25) |
| Former smoking | 32.9% (n=130) | 43.5% (n=20) | 31.5% (n=110) | 40.1% (n=75) | 25.6% (n=52) | 54.1% (n=20) | 30.3% (n=107) |
| Hypertension | 35.4% (n=104) | 56.2% (n=18) | 32.8% (n=86) | 45.6% (n=67) | 26.1% (n=37) | 55.6% (n=15) | 34.0% (n=89) |
| Total/HDL cholesterol ratio | 3.8 (2.3) | 4.1 (2.9) | 3.8 (2.2) | 3.8 (2.6) | 3.7 (2.1) | 3.7 (2.7) | 3.8 (2.3) |
| Dyslipidemia | 40.9% (n=137) | 55.3% (n=21) | 39.1% (n=116) | 52.8% (n=85) | 30.2% (n=51) | 60.0% (n=18) | 39.3% (n=118) |
| Diabetes | 4.0% (n=13) | 15.4% (n=6) | 2.4% (n=7) | 5.9% (n=9) | 2.3% (n=4) | 13.8% (n=4) | 3.1% (n=9) |
| Breast artery calcium† | 3.7% (n=6) | 7.7% (n=1) | 3.3% (n=5) | 3.9% (n=2) | 3.6% (n=4) | 8.3% (n=1) | 3.3% (n=5) |
| Splenic artery calcium | 11.6% (n=46) | 100.0% (n=46) | 0.0% (n=0) | 20.9% (n=39) | 3.0% (n=6) | 51.4% (n=19) | 7.4% (n=26) |
| Internal iliac artery calcium | 47.9% (n=187) | 86.7% (n=39) | 42.9% (n=148) | 100.0% (n=187) | 0.0% (n=0) | 100.0% (n=37) | 42.5% (n=150) |
| CAC (Agatston)a | 7 (0–178) | 556 (138–1473) | 2 (0–82) | 119 (7–426) | 0 (0–8) | 607 (189–1071) | 2 (0–107) |
| Thoracic aorta calcium (Agatston)a | 0 (0–112) | 306 (110–1935) | 0 (0–46) | 78 (0–410) | 0 (0–0) | 953 (180–2864) | 0 (0–46) |
| Abdominal aorta calcium (Agatston)a | 53 (0–582) | 1700 (628–3804) | 16 (0–316) | 472 (100–1536) | 0 (0–32) | 2572 (1312–4037) | 16 (0–316) |
| Common & ext. iliac artery calcium (Agatston)a | 1 (0–400) | 1718 (189–3798) | 0 (0–242) | 406 (98–1188) | 0 (0–0) | 2910 (1474–4629) | 0 (0–233) |
For external and internal iliac artery in n=390 because 5 people did not have external and iliac artery scoring (scans partially missing). Continuous variables are described using mean (SD) or a: median (interquartile range) in case of skewed variables. Categorical variables are described as a percentage (number).
BMI, body mass index; CVD, cardiovascular disease; CAC, coronary artery calcium.
Among women (n=163).
Associations of risk factors with presence of calcium in different vascular beds
Table 2 shows the unadjusted and multivariably adjusted associations of risk factors with presence of calcium in the splenic, internal iliac and external iliac arteries. Age and diabetes were the only two factors that were strongly and independently associated with splenic artery calcification (ORs per 10 years 3.85 [95% CI 2.53–5.84] and 4.18 [95% CI 1.15–15.25], respectively, Model 2). Calcifications in the coronary and abdominal aorta were also strongly associated with splenic artery calcification. Adjustment for these variables somewhat attenuated the association of age with splenic calcification and did not meaningfully alter the strong associations of age and diabetes with splenic artery calcification.
Table 2.
Associations of risk factors with presence of calcium in the different vascular beds
| Splenic artery calcium | Models 1 | Model 2 | Model 3 | |||
|---|---|---|---|---|---|---|
| Variable | OR (CI) | p | OR (CI) | p | OR (CI) | p |
| Age (10 years) | 3.31 (2.33–4.70) | <0.001 | 3.85 (2.53–5.84) | <0.001 | 1.55 (0.90–2.66) | 0.115 |
| Sex (male) | 1.91 (0.97–3.76) | 0.060 | 1.88 (0.82–4.35) | 0.138 | 1.38 (0.53–3.58) | 0.513 |
| BMI | 1.03 (0.96–1.10) | 0.448 | 1.05 (0.96–1.15) | 0.249 | 0.99 (0.90–1.09) | 0.869 |
| Smoking: current vs never | 1.27 (0.41–3.93) | 0.682 | 1.95 (0.54–7.10) | 0.310 | 0.61 (0.14–2.65) | 0.506 |
| former vs never | 1.73 (0.90–3.30) | 0.098 | 1.10 (0.51–2.37) | 0.802 | 0.77 (0.34–1.76) | 0.532 |
| Hypertension | 2.08 (1.02–4.24) | 0.045 | 1.20 (0.50–2.89) | 0.682 | 1.08 (0.40–2.88) | 0.885 |
| Dyslipidemia | 1.92 (0.99–3.71) | 0.052 | 1.57 (0.70–3.51) | 0.275 | 1.25 (0.54–2.91) | 0.604 |
| Diabetes | 7.26 (2.33–22.68) | 0.001 | 4.18 (1.15–15.25) | 0.030 | 4.49 (1.00–20.19) | 0.050 |
| Thoracic aorta calcium | 1.57 (1.38–1.78) | <0.001 | - | - | 1.10 (0.91–1.33) | 0.314 |
| Abdominal aorta calcium | 1.82 (1.50–2.22) | <0.001 | - | - | 1.31 (1.02–1.68) | 0.034 |
| CAC score | 1.81 (1.52–2.16) | <0.001 | - | - | 1.34 (1.08–1.66) | 0.007 |
|
| ||||||
| Internal iliac artery calcium | Models 1 | Model 2 | Model 3 | |||
| Variable | OR (CI) | p | OR (CI) | p | OR (CI) | p |
|
| ||||||
| Age (10 years) | 3.20 (2.46–4.18) | <0.001 | 3.74 (2.74–5.12) | <0.001 | 1.50 (0.99–2.27) | 0.059 |
| Sex (male) | 3.22 (2.11–4.92) | <0.001 | 4.04 (2.28–7.16) | <0.001 | 3.95 (2.04–7.65) | <0.001 |
| BMI | 1.05 (1.00–1.10) | 0.037 | 1.03 (0.97–1.10) | 0.299 | 1.01 (0.94–1.08) | 0.880 |
| Smoking: current vs. never | 2.45 (1.17–5.14) | 0.018 | 4.21 (1.61–10.96) | 0.003 | 2.62 (0.86–7.96) | 0.089 |
| former vs. never | 2.19 (1.41–3.40) | 0.001 | 1.51 (0.87–2.64) | 0.144 | 1.23 (0.65–2.30) | 0.524 |
| Hypertension | 2.13 (1.34–3.38) | 0.001 | 1.10 (0.59–2.06) | 0.765 | 1.04 (0.51–2.13) | 0.904 |
| Dyslipidemia | 2.52 (1.62–3.91) | <0.001 | 1.67 (0.94–2.96) | 0.079 | 1.11 (0.58–2.14) | 0.752 |
| Diabetes | 2.53 (0.77–8.37) | 0.127 | 0.69 (0.18–2.66) | 0.592 | 0.77 (0.15–4.04) | 0.753 |
| Thoracic aorta calcium | 1.49 (1.36–1.63) | <0.001 | 1.08 (0.94–1.25) | 0.281 | ||
| Abdominal aorta calcium | 1.64 (1.50–1.79) | <0.001 | 1.41 (1.24–1.59) | <0.001 | ||
| CAC score | 1.59 (1.44–1.75) | <0.001 | 1.21 (1.06–1.37) | 0.004 | ||
|
| ||||||
| External iliac artery calcium | Models 1 | Model 2 | Model 3 | |||
| Variable | OR (CI) | p | OR (CI) | p | OR (CI) | p |
|
| ||||||
| Age (10 years) | 2.96 (2.07–4.24) | <0.001 | 3.47 (2.21–5.45) | <0.001 | 0.92 (0.47–1.78) | 0.800 |
| Sex (male) | 1.54 (0.75–3.16) | 0.240 | 1.22 (0.48–3.08) | 0.679 | 0.93 (0.29–3.03) | 0.905 |
| BMI | 0.98 (0.91–1.06) | 0.692 | 1.01 (0.92–1.12) | 0.790 | 0.94 (0.84–1.06) | 0.315 |
| Smoking: current vs. never | 9.95 (3.52–28.09) | <0.001 | 25.23 (6.75–94.33) | <0.001 | 9.05 (1.88–43.54) | 0.006 |
| former vs. never | 5.16 (2.20–12.10) | <0.001 | 4.13 (1.54–11.04) | 0.005 | 2.91 (0.99–8.50) | 0.051 |
| Hypertension | 2.26 (1.05–4.83) | 0.037 | 1.40 (0.55–3.54) | 0.481 | 1.04 (0.35–3.10) | 0.945 |
| Dyslipidemia | 2.06 (0.98–4.34) | 0.057 | 1.83 (0.70–4.78) | 0.214 | 1.80 (0.62–5.25) | 0.282 |
| Diabetes | 4.60 (1.34–15.75) | 0.015 | 6.07 (1.32–27.86) | 0.021 | 6.14 (0.91–41.36) | 0.062 |
| Thoracic aorta calcium | 1.85 (1.54–2.22) | <0.001 | 1.24 (0.95–1.61) | 0.110 | ||
| Abdominal aorta calcium | 3.19 (2.15–4.75) | <0.001 | 1.96 (1.22–3.17) | 0.006 | ||
| CAC score | 1.80 (1.48–2.18) | <0.001 | 1.25 (0.97–1.61) | 0.091 | ||
Models 1: Univariable association of each risk factor with calcification. Model 2: a multivariable model including traditional CVD risk factors. Model 3: the log of 1+ the quantification of the calcification of the coronary arteries and the thoracic and abdominal aorta were added to Model 2. Analyses done in subcohort (n=395). For external and internal iliac artery in n=390 because 5 people did not have external and iliac artery scoring (scans partially missing).
Risk factors associated with internal iliac calcification differed from those associated with splenic artery calcification. Here, age (OR per 10 years 3.74 [95% CI 2.74–5.12]), male sex (OR [95% CI 4.04 2.28–7.16]), and current smoking (OR 4.21 [95% CI 1.61–10.96]) were independently associated with iliac calcification, whereas diabetes was not (model 2). Calcification in the coronary arteries and abdominal aorta were also strongly associated with internal iliac artery calcification. Inclusion of these calcification variables weakened the association of age and smoking with internal iliac calcification, but the association of male sex with internal iliac calcification persisted despite this adjustment, and age and current smoking were rendered not significant but borderline (p=0.059 and p= 0.089, respectively, Model 3).
For external iliac artery calcification, age (OR per 10 years 3.47 [95% CI 2.21–5.45]), current smoking (25.23 [95% CI 6.75–94.33]), and diabetes (6.07 [95% CI 1.32–27.86]) were found to be independent risk factors (Model 2). Again, calcification in the abdominal aorta was strongly associated with external iliac calcification. The effect size estimate of the association of CAC with external iliac artery calcification was similar as with internal iliac artery calcification, but not significant. After adjustment for calcification in these vascular beds, only smoking persisted (OR 9.05 [95% CI 1.88–43.54]) as an independent risk factor for external iliac calcification, and diabetes closely approached statistical significance (OR 6.14 (95% CI 0.91–41.36]) in Model 3.
Associations of presence of calcium in different vascular beds risk with mortality
During 9.4 years median follow-up, 298 participants died (115 deaths among female), of which 90 (37 among females) were classified as CVD deaths. Table 3 depicts the associations of calcification prevalence with CVD mortality and all-cause mortality. Presence of BAC was significantly and strongly associated with both CVD mortality (HR 12.30 [95% CI 2.84–53.37]) and all-cause mortality (HR 4.67 [95% CI 1.57–13.88]), adjusted for age. After multivariable adjustment for traditional CVD risk factors, BAC remained significantly associated with total mortality (HR 5.67 [95% CI 1.50–21.41]). While the hazard ratio for CVD mortality remained very high based on the point estimate, the association was no longer statistically significant, perhaps because of limited statistical power (HR 19.05 [95% CI 0.76–480.64]).
Table 3.
Associations of presence of medium artery calcification with all-cause and cardiovascular mortality
| CVD mortality (n cases=37) | All-cause mortality (n cases=115) | |||
|---|---|---|---|---|
| Breast artery calcium (present/absent)a | ||||
| Model | HR (95% CI) | p | HR (95% CI) | p |
| Model 1a | 12.30 (2.84–53.37) | 0.001 | 4.67 (1.57–13.88) | 0.006 |
| Model 2a | 29.59 (1.86–471.38) | 0.017 | 7.33 (2.03–26.45) | 0.002 |
| Model 3a | 19.05 (0.76–480.64) | 0.074 | 5.67 (1.50–21.41) | 0.011 |
| CVD mortality (n cases = 90) | All-cause mortality (n cases = 298) | |||
| Splenic artery calcium (present/absent) | ||||
| Model | HR (95% CI) | p | HR (95% CI) | p |
| Model 1 | 1.69 (0.83–3.42) | 0.146 | 1.65 (0.97–2.78) | 0.063 |
| Model 2 | 1.46 (0.71–2.97) | 0.302 | 1.51 (0.89–2.57) | 0.129 |
| Model 3 | 1.37 (0.68–2.79) | 0.380 | 1.43 (0.84–2.42) | 0.191 |
| Internal iliac artery calcium (present/absent) | ||||
| Model | HR (95% CI) | p | HR (95% CI) | p |
| Model 1 | 1.24 (0.60–2.58) | 0.563 | 1.37 (0.86–2.17) | 0.185 |
| Model 2 | 0.91 (0.41–2.04) | 0.826 | 1.15 (0.70–1.87) | 0.586 |
| Model 3 | 0.75 (0.31–1.79) | 0.514 | 0.97 (0.56–1.68) | 0.901 |
| External iliac artery calcium (present/absent) | ||||
| Model | HR (95% CI) | p | HR (95% CI) | p |
| Model 1 | 2.15 (1.06–4.38) | 0.034 | 1.86 (1.08–3.20) | 0.025 |
| Model 2 | 1.47 (0.63–3.42) | 0.375 | 1.34 (0.73–2.47) | 0.348 |
| Model 3 | 1.33 (0.56–3.12) | 0.517 | 1.22 (0.66–2.27) | 0.526 |
Model 1: adjusted for age and sex. Model 2: adjusted for sex, age, smoking status, dyslipidemia, BMI, diabetes and hypertension. Model 3: additionally adjusted a 0–5 score of the number of calcified vascular beds out of the carotids, coronaries, abdominal and thoracic aorta and common iliac arteries.
HR, hazard ratio; 95% CI, 95% confidence interval.
Analysis in women only. Models 1 to 3 adjusted as described above except for sex.
Although point estimates were greater than 1, we did not find significant associations of splenic artery or internal iliac artery calcification with CVD mortality or total mortality, neither when adjusting for age and sex (HRs ranging from 1.24 to 1.69) nor when additionally adjusting for other CVD risk factors (HRs ranging from 0.91 to 1.51). Conversely, external iliac artery calcification was associated with both CVD mortality and all-cause mortality when adjusting for age and sex (HRs 2.15 [95% CI 1.06–4.38] and 1.86 [95% CI 1.08–3.20], respectively), but the associations attenuated to non-significant when additionally adjusted for CVD risk factors (HRs 1.47 [95% CI 0.63–3.42] and 1.34 [95% CI 0.73–2.47]). Adding the sum score of the number of calcified vascular beds (Agatston score > 0) further lowered estimates. The results of the analysis relating calcification by graded severity to mortality outcomes are shown in supplemental Table 1. The associations were largely non-significant across the different vascular beds.
Discussion
In this nested case-cohort study, we determined the association of various risk factors with calcification in the splenic, internal and external iliac arteries, and the association of calcification in these arteries with mortality. We found that the presence of calcium in the abdominal aorta was strongly associated with calcium in the splenic, internal, and external iliac arteries. However, the associations of traditional CVD risk factors with calcification differed across artery beds. Calcification in the splenic, internal or external iliac arteries was not associated with mortality after multivariable adjustment. In contrast, BAC was uncommon, but when present, was strongly and independently associated with mortality risk.
Our first aim was to evaluate the risk factors associated with calcification in a number of small to medium sized arteries and to see if differences inform potential underlying pathophysiological mechanisms (i.e. intimal/atherosclerotic versus MAC/non-atherosclerotic calcification). In the literature, older age7–9,11, diabetes,7,8,11,17 and kidney disease 17 are consistently associated with higher prevalence of MAC. In contrast, smoking is related to lower prevalence of MAC or studies report a lack of association.9–12,17,31 Although there is some heterogeneity, most studies report no relationship of hypertension or dyslipidemia with MAC.8,11,17 For atherosclerotic calcification, such as predominant in the coronary arteries, traditional cardiovascular risk factors, including smoking and hyperlipidemia are associated with calcification.1,11,17
Splenic artery calcification has been described as predominantly medial in prior studies.15 In our study, only age and diabetes were significantly associated with splenic artery calcification, which fits a MAC profile. To our knowledge, only one previous study related cardiovascular risk factors to the presence of calcium in the splenic artery.16 This study showed a small (r=0.17) but significant correlation between blood glucose level and splenic calcification in 276 patients at high CVD risk, but no significant correlations with BMI, cholesterol or systolic blood pressure. These findings are consistent with the association of diabetes with splenic artery calcification observed in our study. In the previous study, strong correlations were found between splenic artery calcification and calcification in other vascular beds, similar to our study. However, this prior study evaluated individual risk factors without multivariable adjustment. Thus, one important contribution of our study is that it demonstrates the independent relationship of risk factors with splenic artery calcification for the first time. We find that diabetes and age are strong and independent risk factors for splenic calcification.
The risk factors for calcification of the internal and external iliac arteries were generally similar in our study, although age and male sex were significant risk factors for the internal iliac artery but not for the external iliac artery. Importantly, current smoking was a risk factor associated with both internal and external iliac artery calcification. As smoking is an important risk factor for atherosclerosis but associated with a lower prevalence of MAC in prior studies, it is possible that calcification of the internal and external iliac artery reflects predominantly atherosclerotic, intimal calcification rather than MAC. Prior studies have generally grouped parts of the iliac vasculature (internal, external, common) together and report associations with age, male gender, hypertension, diabetes and smoking.32 A prior study from this cohort also reported on the associations with calcium in the combined common and external iliac arteries, and found independent associations with age, hypercholesterolemia and cigarette smoking.3 In contrast, we did not find associations with hypertension or hypercholesterolemia in this study, but we also did not incorporate the slightly larger common iliac artery. We hypothesize that hypercholesterolemia, hypertension, and smoking are important risk factors for intimal calcification but not necessarily for MAC and that their role in calcification increases as the contribution of atherosclerotic calcifications to the total calcium burden increases.
A secondary objective of this study was to determine whether calcification in medium sized arteries is associated with mortality, and whether such associations differed across artery beds. For calcification in the splenic, internal and external iliac arteries we did not find associations with mortality in adjusted models. Hazard ratios were consistently greater than one across models, albeit not significant. Prior studies have reported similarly sized, but statistically significant, associations of common iliac artery calcification with mortality, yet to our knowledge, no prior study has differentiated calcification in the common, external, or internal iliac arteries.20,33 Thus, future studies are required to determine if associations of calcification in these arteries are attenuated and explained by age and other CVD risk factors, or if our results were rendered not significant due to insufficient statistical power.
We found that presence of BAC on CT in females was strongly and significantly associated with increased risk of both CVD and all-cause mortality after adjusting for cardiovascular risk factors. These results should be interpreted with caution since we had a limited sample size for women, resulting in wide confidence intervals. Prior studies have demonstrated that BAC on mammography is associated with cardiovascular mortality, but associations were less strong than observed in this study.9 In similarly aged populations, the prevalence of mammographic BAC has been reported to be higher than that observed here by CT. CT may be less sensitive, thus simultaneously resulting in lower prevalence estimates, and stronger associations with end-points. Consistent with this hypothesis, a prior study on the association of mammographic BAC with cardiovascular outcomes showed stronger associations for severe BAC compared to any BAC.18 Although our findings do not have direct clinical implications, our results support the relevance of MAC as a clinically significant entity. Further research is needed to establish to what extent it contributes to the total cardiovascular disease burden and whether it could form a potential treatment target.
This study has multiple strengths. To our knowledge, this is the first study to evaluate associations of splenic and BAC on CT with mortality risk. The community-based setting, availability of imaging of multiple vascular beds, and traditional CVD risk factors allow us to determine and compare strengths of association with risk factors and outcomes across medium sized artery beds. Despite this and other strengths, the study also has important limitations. The study sample was not originally collected for research purposes, resulting in missing values for some variables. The study sample consists of patients self-referred or referred by a provider, which somewhat limits generalizability of the study’s findings to the population as a whole. As a consequence of using dichotomized variables for dyslipidemia and hypertension, there is a possibility for some residual confounding. For grading calcification severity, we used a semi-quantitative scoring method with some degree of subjectivity, reflected in moderate inter-rater agreement for some vascular beds. Variation between observers can be explained by differences in detections; small calcifications that are missed by one of both observers, and by different judgments; high density voxels judged to be noise related or judged to be outside the arterial wall. Future studies using quantitative assessment and semi-automated centerline length for tortuous vessels may provide more accurate and reproducible calcification measures. Some of the confidence intervals were quite wide, indicating low statistical power. When positive and significant, we are confident in these associations, but have less confidence in the precision of the estimate. Associations should be interpreted within the confines of the reported 95% confidence intervals. This was especially true for the analysis of BAC, which was limited to women and had low prevalence, and the analysis by grades of severity. Unfortunately, due to the limited number of women with BAC in the subcohort we could not assess the associations of risk factors with the presence of BAC in our study. We lack pathological data, which would clarify whether calcification in a certain bed is predominantly medial, intimal or both and could provide further insight into the consequences associated with the different types of calcification.
In conclusion, we found that calcification of the splenic, internal and external iliac arteries is strongly associated with calcification in the abdominal aorta, but differentially associated with other CVD risk factors. Age, male gender, and diabetes were predominant risk factors for splenic artery calcification, while other CVD risk factors were not, suggesting that splenic artery calcification may be predominantly MAC. After adjustment for CVD risk factors, calcifications in the splenic, internal and external arteries were not associated with mortality. BAC is a rare finding on CT scan in community living females, but when present, was strongly and independently associated with mortality. This finding supports our hypothesis that, beyond intimal calcification, MAC may also be associated with increased risk of mortality in community living individuals and merits further investigation as a potential treatment target.
Supplementary Material
Highlights.
Calcification in the splenic, internal and external iliac arteries is strongly associated with calcification in the abdominal aorta but differentially associated with other CVD risk factors
Different associations with risk factors may indicate a different underlying pathophysiology
Breast artery calcification is a rare finding on CT scans in community living females, but when present, may be a marker of increased risk for mortality
Besides intimal/atherosclerotic calcification, medial arterial calcification may also be associated with increased mortality risks
Acknowledgments
Financial support
Funding for this project comes from the National Institutes of Health, National Heart, Lung, and Blood Institute R01HL116395 (MHC), American Heart Association Established Investigator Award EIA18560026 (JHI), American Heart Association Fellow to Faculty Award 0475029N (MAA).
The authors thank the participants of this study, the clinic staff who received and examined these participants, and Julie Denenberg for her assistance and organization.
Abbreviations
- MAC
medial arterial calcification
- BAC
breast artery calcification
- CT
computed tomography
- CVD
cardiovascular disease
Footnotes
Conflict of interest
The authors declared they do not have anything to disclose regarding conflict of interest with respect to this manuscript.
Author contributions
Authorship contributions were as follows:
Conception and design: MCW, EJH, MAA, JHI.
Analysis and interpretation of data: PAJ, JWB, YTvdS, WNS, EJH, MAA, JHI.
Drafting of the manuscript: EJH, JHI.
Revising manuscript critically for important intellectual content: PAJ, JWJ, YTvdS, WNS, MCW, MHC, MAA, JIH.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Madhavan MV, Tarigopula M, Mintz GS, Maehara A, Stone GW, Généreux P. Coronary artery calcification: pathogenesis and prognostic implications. J Am Coll Cardiol. 2014;63(17):1703–1714. doi: 10.1016/j.jacc.2014.01.017. [DOI] [PubMed] [Google Scholar]
- 2.Criqui MH, Denenberg JO, McClelland R, et al. Abdominal Aortic Calcium, Coronary Artery Calcium, and Cardiovascular Morbidity and Mortality in the Multi-Ethnic Study of Atherosclerosis. Arterioscler Thromb Vasc Biol. 2014 Jun; doi: 10.1161/ATVBAHA.114.303268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Allison MA, Criqui MH, Wright CM. Patterns and Risk Factors for Systemic Calcified Atherosclerosis. Arterioscler Thromb Vasc Biol. 2004;24(2):331–336. doi: 10.1161/01.ATV.0000110786.02097.0c. [DOI] [PubMed] [Google Scholar]
- 4.Fishbein MC, Fishbein Ga. Arteriosclerosis: facts and fancy. Cardiovasc Pathol. 2015 doi: 10.1016/j.carpath.2015.07.007. [DOI] [PubMed] [Google Scholar]
- 5.Kalra SS, Shanahan CM. Vascular calcification and hypertension: cause and effect. Ann Med. 2012;44(Suppl 1):S85–92. doi: 10.3109/07853890.2012.660498. [DOI] [PubMed] [Google Scholar]
- 6.Duhn V, D’Orsi ET, Johnson S, D’Orsi CJ, Adams AL, O’Neill WC. Breast arterial calcification: a marker of medial vascular calcification in chronic kidney disease. Clin J Am Soc Nephrol. 2011;6(2):377–382. doi: 10.2215/CJN.07190810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Everhart JE, Pettitt DJ, Knowler WC, Rose FA, Bennett PH. Medial arterial calcification and its association with mortality and complications of diabetes. Diabetologia. 1988;31(1):16–23. doi: 10.1007/BF00279127. [DOI] [PubMed] [Google Scholar]
- 8.Moon JCVM. A controlled study of medial arterial calcification of legs: Implications for diabetic polyneuropathy. Arch Neurol. 2011;68(10):1290–1294. doi: 10.1001/archneurol.2011.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hendriks EJE, de Jong PA, van der Graaf Y, Mali WPTM, van der Schouw YT, Beulens JWJ. Breast arterial calcifications: a systematic review and meta-analysis of their determinants and their association with cardiovascular events. Atherosclerosis. 2015;239(1):11–20. doi: 10.1016/j.atherosclerosis.2014.12.035. [DOI] [PubMed] [Google Scholar]
- 10.Iribarren C, Go AS, Tolstykh I, Sidney S, Johnston SC, Spring DB. Breast Vascular Calcification and Risk of Coronary Heart Disease, Stroke, and Heart Failure. J Women’s Heal. 2004;13(4):381–389. doi: 10.1089/154099904323087060. [DOI] [PubMed] [Google Scholar]
- 11.Lehto S, Niskanen L, Suhonen M, Rönnemaa T, Laakso M. Medial artery calcification. A neglected harbinger of cardiovascular complications in non-insulin-dependent diabetes mellitus. Arterioscler Thromb Vasc Biol. 1996;16(8):978–983. doi: 10.1161/01.atv.16.8.978. [DOI] [PubMed] [Google Scholar]
- 12.Janssen T, Bannas P, Herrmann J, et al. Association of linear 18F-sodium fluoride accumulation in femoral arteries as a measure of diffuse calcification with cardiovascular risk factors: a PET/CT study. J Nucl Cardiol Off Publ Am Soc Nucl Cardiol. 2013;20(4):569–577. doi: 10.1007/s12350-013-9680-8. [DOI] [PubMed] [Google Scholar]
- 13.Lanzer P, Boehm M, Sorribas V, et al. Medial vascular calcification revisited: review and perspectives. Eur Heart J. 2014;35(23):1515–1525. doi: 10.1093/eurheartj/ehu163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vos A, Van Hecke W, Spliet WGMM, et al. Predominance of Nonatherosclerotic Internal Elastic Lamina Calcification in the Intracranial Internal Carotid Artery. Stroke. 2016;47(1):221–223. doi: 10.1161/STROKEAHA.115.011196. [DOI] [PubMed] [Google Scholar]
- 15.Golder WA. Tortuosity and calcification of the splenic artery. More than an additional finding. Radiologe. 2008;48(11):1066–1067. 1074. doi: 10.1007/s00117-008-1631-z. [DOI] [PubMed] [Google Scholar]
- 16.Takx RAP, Zanen P, Leiner T, van der Graaf Y, de Jong PA. The interdependence between cardiovascular calcifications in different arterial beds and vascular risk factors in patients at high cardiovascular risk. Atherosclerosis. 2015;238(1):140–146. doi: 10.1016/j.atherosclerosis.2014.11.024. [DOI] [PubMed] [Google Scholar]
- 17.London GM, Guérin AP, Marchais SJ, Métivier F, Pannier B, Adda H. Arterial media calcification in end-stage renal disease: impact on all-cause and cardiovascular mortality. Nephrol Dial Transplant. 2003;18(9):1731–1740. doi: 10.1093/ndt/gfg414. [DOI] [PubMed] [Google Scholar]
- 18.Hendriks EJE, Beulens JWJ, Mali WPTM, et al. Breast Arterial Calcifications and Their Association With Incident Cardiovascular Disease and Diabetes. J Am Coll Cardiol. 2015;65(8):859–860. doi: 10.1016/j.jacc.2014.12.015. [DOI] [PubMed] [Google Scholar]
- 19.Margolies L, Salvatore M, Hecht HS, et al. Digital Mammography and Screening for Coronary Artery Disease. JACC Cardiovasc Imaging. 2016;9(4):350–360. doi: 10.1016/j.jcmg.2015.10.022. [DOI] [PubMed] [Google Scholar]
- 20.Allison MA, Hsi S, Wassel CL, et al. Calcified Atherosclerosis in Different Vascular Beds and the Risk of Mortality. Arterioscler Thromb Vasc Biol. 2012;32(1):140–146. doi: 10.1161/ATVBAHA.111.235234. [DOI] [PubMed] [Google Scholar]
- 21.Prentice RL. A case-cohort design for epidemiologic cohort studies and disease prevention trials. Biometrika. 1986;73(1):1–11. doi: 10.1093/biomet/73.1.1. [DOI] [Google Scholar]
- 22.Onland-Moret NC, van der ADL, van der Schouw YT, et al. Analysis of case-cohort data: A comparison of different methods. J Clin Epidemiol. 2007;60(4):350–355. doi: 10.1016/j.jclinepi.2006.06.022. [DOI] [PubMed] [Google Scholar]
- 23.Agatston A, Janowitz W, Hildner F, Zusmer N, Viamonte M, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 1990;15(4):827–832. doi: 10.1016/0735-1097(90)90282-t. [DOI] [PubMed] [Google Scholar]
- 24.Jensky NE, Criqui MH, Wright CM, Wassel CL, Alcaraz JE, Allison MA. The Association Between Abdominal Body Composition and Vascular Calcification. Obesity. 2011;19(12):2418–2424. doi: 10.1038/oby.2011.70. [DOI] [PubMed] [Google Scholar]
- 25.Jairam PM, de Jong PA, Mali WPThM, et al. Impact of cardiovascular calcifications on the detrimental effect of continued smoking on cardiovascular risk in male lung cancer screening participants. PLoS One. 2013;8(6) doi: 10.1371/journal.pone.0066484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rosset A, Spadola L, Ratib O. OsiriX: an open-source software for navigating in multidimensional DICOM images. J Digit Imaging. 2004;17(3):205–216. doi: 10.1007/s10278-004-1014-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Blair KJ, Allison MA, Morgan C, et al. Comparison of ordinal versus Agatston coronary calcification scoring for cardiovascular disease mortality in community-living individuals. Int J Cardiovasc Imaging. 2014;30(4):813–818. doi: 10.1007/s10554-014-0392-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Honaker J, King G. What to Do about Missing Values in Time-Series Cross-Section Data. Am J Pol Sci. 2010;54(2):561–581. doi: 10.1111/j.1540-5907.2010.00447.x. [DOI] [Google Scholar]
- 29.Rubin DB. In: Multiple Imputation for Nonresponse in Surveys. Rubin DB, editor. Hoboken, NJ, USA: John Wiley & Sons, Inc; 1987. [DOI] [Google Scholar]
- 30.Xue X, Xie X, Gunter M, et al. Testing the proportional hazards assumption in case-cohort analysis. BMC Med Res Methodol. 2013;13(1) doi: 10.1186/1471-2288-13-88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hendriks EJE, Westerink J, de Jong PA, et al. Association of High Ankle Brachial Index With Incident Cardiovascular Disease and Mortality in a High-Risk Population. Arterioscler Thromb Vasc Biol. 2016;36(2):412–417. doi: 10.1161/ATVBAHA.115.306657. [DOI] [PubMed] [Google Scholar]
- 32.Strobl FF, Rominger A, Wolpers S, et al. Impact of cardiovascular risk factors on vessel wall inflammation and calcified plaque burden differs across vascular beds: a PET-CT study. Int J Cardiovasc Imaging. 2013;29(8):1899–1908. doi: 10.1007/s10554-013-0277-8. [DOI] [PubMed] [Google Scholar]
- 33.Huang C-L, Wu I-H, Wu Y-W, et al. Association of lower extremity arterial calcification with amputation and mortality in patients with symptomatic peripheral artery disease. PLoS One. 2014;9(2):e90201. doi: 10.1371/journal.pone.0090201. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.