Abstract
Polyneuropathy is a frequent and potentially severe side effect of clinical tumor chemotherapy. The goal of this study was to characterize paclitaxel-, cisplatin-, vincristine- and bortezomib-induced neuropathy in C57BL/6 mice with a comparative approach. The phenotype of the animals was evaluated at four time points with behavioral and electrophysiological tests, followed by histology. Treatment protocols used in this study were well tolerated and induced a sensory and predominantly axonal polyneuropathy. Behavioral testing revealed normal motor coordination, whereas all mice receiving verum treatment developed mechanical allodynia and distinct gait alterations. Electrophysiological evaluation showed a significant decrease of the caudal sensory nerve action potential amplitude for all cytostatic agents and a moderate reduction of nerve conduction velocity for cisplatin and paclitaxel. This finding was confirmed by histological analysis of the sciatic nerve which showed predominantly axonal damage: Paclitaxel and vincristine affected mostly large myelinated fibers, bortezomib small myelinated fibers and cisplatin damaged all types of myelinated fibers to a similar degree. Neuropathic symptoms developed faster in paclitaxel and vincristine treated animals compared to cisplatin and bortezomib treatment. The animal models in this study can be used to elucidate pathomechanisms underlying chemotherapy-induced polyneuropathy and for the development of novel therapeutic and preventative strategies.
Many agents used for tumor chemotherapy cause toxicity in the peripheral nervous system. Patients who develop chemotherapy-induced peripheral neuropathy (CIPN) frequently complain about loss of sensation and neuropathic pain. This condition not only increases the burden of disease, but can also be dose limiting which is detrimental to therapy (reviewed by1). Among the compounds that frequently cause severe CIPN are taxanes (paclitaxel, docetaxel), vinca alcaloids (vincristine, vinblastine, vindesine, vinflunine, vinorelbine), platinum analogs (cisplatin, carboplatin, oxaliplatin) and bortezomib. These compounds have distinct pharmacological modes of action: Taxanes, which are often used in the treatment of solid tumors, stabilize the tubulin cytoskeleton and thus prevent its disassembly2. This blocks the progression of mitosis causing a cell cycle arrest and subsequent cell death. Vinca alcaloids are frequently used in the treatment of hematologic malignancies, such as lymphomas and leukemias3. In contrast to taxanes, which promote tubulin polymerization, vinca alcaloids inhibit tubulin assembly, thus blocking the formation of a spindle apparatus during mitosis. Platinum-based antineoplastic drugs show great versatility in the treatment of malignancies including but not limited to sarcomas, carcinomas, lymphomas and germ cell tumors. Platinum compounds induce cross-linking of DNA strands, which inhibits DNA synthesis and repair (reviewed by4). Bortezomib is used in the treatment of multiple myeloma as well as mantle cell lymphoma and is currently investigated as an immunmodulator (reviewed by5). It irreversibly blocks the catalytic site of the 26S proteasome, preventing proteolytic cleavage of intracellular proteins. Several recent reviews discuss current pathophysiological concepts of neuropathy development induced by these substances6,7,8.
Despite intense research efforts, both in the clinical and in the basic sciences, CIPN and its underlying pathomechanisms are however still not fully understood. Current therapeutic options therefore only serve to alleviate the symptoms but do not prevent damage in the peripheral nervous system. One potential advantage for therapeutic studies on CIPN in comparison to other diseases of the peripheral and central nervous system is its clearly defined onset of damage. These are ideal preconditions to develop an early therapeutic or even preventative treatment. Various animal models of CIPN with different endpoints have been developed and characterized to improve our understanding of how drugs designed to target dividing cells lead to the malfunction and death of postmitotic neurons (reviewed by9,10).
In the present study we used a comparative approach and the endpoints behavior, electrophysiology and histology to characterize the development of paclitaxel-, cisplatin-, vincristine- and bortezomib-induced neuropathy in mice with the common C57BL/6 background.
Results
Unspecific effects of cytostatic drug treatment
To minimize unspecific systemic toxicity, we administered cytostatic drugs at doses that were lower or comparable to treatment of patients undergoing chemotherapy (Table 1). Calculation of the human equivalent dose was performed according to previously published pharmacological models11. We expected that neuropathies develop at different rates depending on the tested drugs. Therefore we adjusted the duration of the experiments based on previously published studies12,13,14,15,16,17. To facilitate comparison across different cytostatic agents we defined an early (between baseline and the middle of the experiment), middle (approximately half time) and late (before the end of the experiment) time point for behavioral and electrophysiological testing (Methods). Experiment duration and the experimental days corresponding with the time points outlined above are specified in table 1.
Table 1. Summary of drugs and their abbreviations, treatment schedules, cumulative doses and test time points.
| Substance & abbreviation | Single Dose | Schedule | Time | Cumulative Dose | Human equivalent dose (single) | Time points (early; middle; late) |
|---|---|---|---|---|---|---|
| Vincristine (VIN) | 200 μg/kg BW | Single i.p | 10 d | 200 μg/kg | 0.65 mg/m2 | Day: 2/3; 5/6; 9/10 |
| Paclitaxel (PTX) | 1 mg/kg BW | 4x alternating days i.p. | 14 d | 4 mg/kg | 10 mg/m2 | Day: 3/4; 7/8; 13/14 |
| Cisplatin (CIS) | 2.3 mg/kg BW | 5x −5 d rest-5x i.p. | 21 d | 23 mg/kg | 7.5 mg/m2 | Day: 5/6; 13/14; 20/21 |
| Bortezomib (BTZ) | 400 μg/kg BW | 3x/week, 4 weeks i.p. | 27 d | 4.8 mg/kg | 1.3 mg/m2 | Day: 5/6; 15/16; 26/27 |
Overall, treatment with paclitaxel, cisplatin, vincristine or bortezomib was well tolerated. Both verum- as well as control-treated animals showed normal activity, social interactions and grooming behavior. We did not observe signs of distress and mortality was very low: one mouse in the bortezomib control group and one mouse in the cisplatin verum group died.
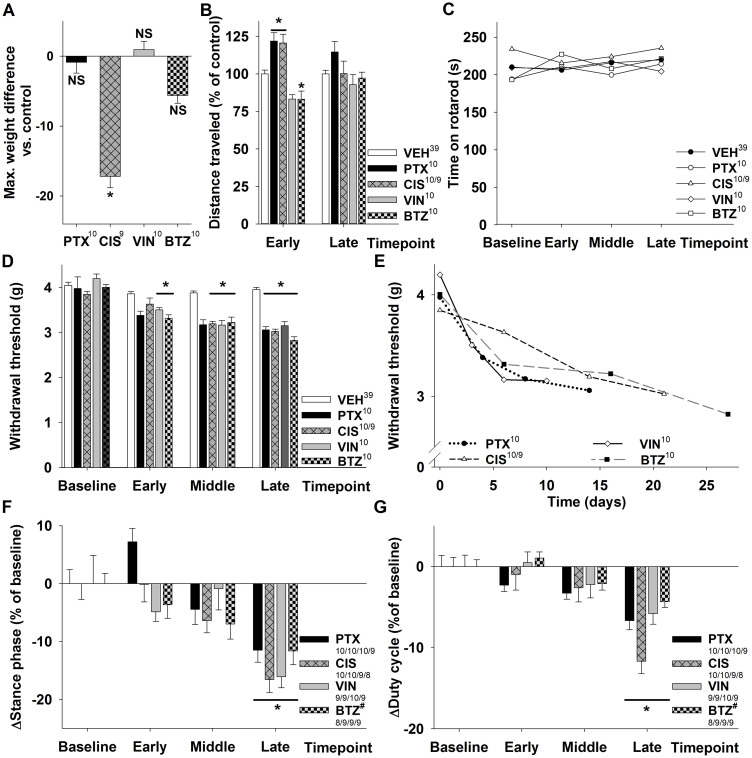
Animals receiving cisplatin significantly lost weight during the treatment, but quickly recovered after the last injection. The maximum recorded weight difference between controls and treated animals was −1.3% ± 0.3% for paclitaxel ([10], not significant), −17.2% ± 1.9% for cisplatin ([9], p < 0.001), +0.9% ± 0.6% for vincristine ([10], not significant) and −5.6% ± 0.5% for bortezomib ([10], not significant, Figure 1A). We analyzed the behavior of the animals further with the open field test. In this test the movements of the animals through a novel open field were monitored. We were interested in whether animals undergoing cytostatic therapy would be less active, as this may be a sign for general toxicity or motor neuropathy. Compared with controls, paclitaxel- and cisplatin-treated animals traveled slightly longer distances at the early time point while vincristine- and bortezomib-treated animals moved less (PTX +22% ± 6%, [10]; CIS +20% ± 6%, [10]; VIN −17% ± 3%, [10]; BTZ −17% ± 5% [10]; PTX, CIS and BTZ: p<0.05 compared to vehicle controls; Figure 1B). At the late time point no significant difference was detected for any of the substances studied. Even though animals showed small to moderate alterations in the distance they traveled at the early time point, all treatment paradigms were well tolerated.
Figure 1. Behavioral observations in animals receiving cytostatic drugs.
(A) Cisplatin-treated animals were up to 17 ± 2% lighter after the last injection than the control group, while no significant weight change was observed for bortezomib, paclitaxel or vincristine treatment. (B) The open field test revealed only moderate changes in activity patterns at the early and no significant changes at the late time point. Paclitaxel- and cisplatin-treated animals traveled longer distances, while bortezomib- and vincristine (the latter not significantly) treated animals moved less. (C) Motor performance assessed with the rotarod test was comparable in verum- and vehicle-injected animals. (D) All treatments led to mechanical allodynia with a significantly reduced mechanical withdrawal threshold in the hind paws compared to vehicle-injected animals. (E) Allodynia developed faster in PTX and VIN treated mice. Gait analysis with the catwalk method showed that animals receiving cytostatic drugs develop distinct gait alterations: The stance phase (F) and the duty cycle (G) decrease. The parameter duty cycle expresses the stance phase as a percentage of the entire step cycle (stand + swing). While paclitaxel, cisplatin and vincristine induced most prominent gait alterations of the hind paws, bortezomib affected forepaws more than hind paws. The data for bortezomib in (F + G) is presented for forepaws which is highlighted with a hash (#). Superscript numbers indicate the number of animals used in each group (One CIS treated mouse died after the early time point; in Figure F + G different time points are separated by /).* p < 0.05; compared to the substance specific vehicle control group at the same time point.
Behavioral alterations
The drugs used in this study are known to mainly affect the sensory nervous system, however in high doses they can sometimes also cause motor symptoms18. We tested for an impaired motor performance with the rotarod test and observed that performance in animals treated with cytostatic drugs was similar to that in the control group (Figure 1C).
One hallmark of murine CIPN is mechanical allodynia (reviewed by9). In line with previous reports, all treatments led to a marked reduction of the mechanical withdrawal threshold measured with the von Frey hair test. Similar observations were made for paclitaxel, cisplatin, vincristine and bortezomib with a reduction of the mechanical withdrawal threshold compared to baseline of 15% ± 3% for paclitaxel ([10], not significant), 6% ± 4% for cisplatin ([10], not significant), 17% ± 1% for vincristine ([10], p < 0.001) and 17% ± 2% for bortezomib ([10], p < 0.001) at the early time point. At the late time point mechanical withdrawal threshold was reduced by 23% ± 2% for paclitaxel ([10], p < 0.05), 21% ± 3% for cisplatin ([9], p < 0.001), 25% ± 3% for vincristine ([10], p < 0.001) and 29% ± 3% for bortezomib ([10], p < 0.001, Figure 1D). The earliest manifestations of mechanical allodynia were observed in vincristine and paclitaxel treated animals and thereafter in bortezomib-treated animals (Figure 1E). By day 10 after the first injection all animals treated with chemotherapeutic agents developed significant allodynia (Figure 1D + E).
Automated gait analysis with the catwalk technique has been used previously to detect gait alterations induced by dose-dense paclitaxel treatment19. In particular, an increase in the swing phase was observed as well as a decrease in the stance phase and duty cycle. The parameter duty cycle expresses the stance phase as a percentage of the step cycle (stance + swing phase). Given the limited experience with this technique in CIPN animal models, we were interested as to whether the drugs and treatment schedules used in this study would also lead to detectable gait alterations in mice. In light of previous publications including human studies, a symmetrical, distal and length-dependent neuropathy was to be expected19. Fore- and hindpaws were therefore analyzed separately, while parameters for the right and left side were evaluated together.
Compared to baseline, the stance phase of the hindpaws at the late timepoint in paclitaxel-, cisplatin- and vincristine-treated mice was significantly (p < 0.05) reduced by −11% ± 2% for paclitaxel [9], −17% ± 2% for cisplatin [8] and −16% ± 2% for vincristine [9] (Figure 1F). Parallel to these changes, we also observed a significant (p < 0.05) reduction of the duty cycle at the late time point by −7% ± 1% ([9], PTX), −12% ± 2% ([8], CIS) and −6% ± 1% ([9], VIN, Figure 1G). Similar to our previous observations, significant results were only observed at the late time point, and thus later than the development of mechanical allodynia. Interestingly, although mice receiving bortezomib injections also showed a trend towards a reduced hindpaw stance phase and duty cycle, only forepaw parameters were significant. At the late time point, the stance phase was reduced by −12% ± 2% [9] and the duty cycle by −4% ± 1% ([9], both p < 0.05; Figure 1F–G).
Taken together, our results demonstrate the development of a sensory neuropathy with mechanical allodynia and distinct gait alterations for all tested drugs.
Electrophysiological measurements
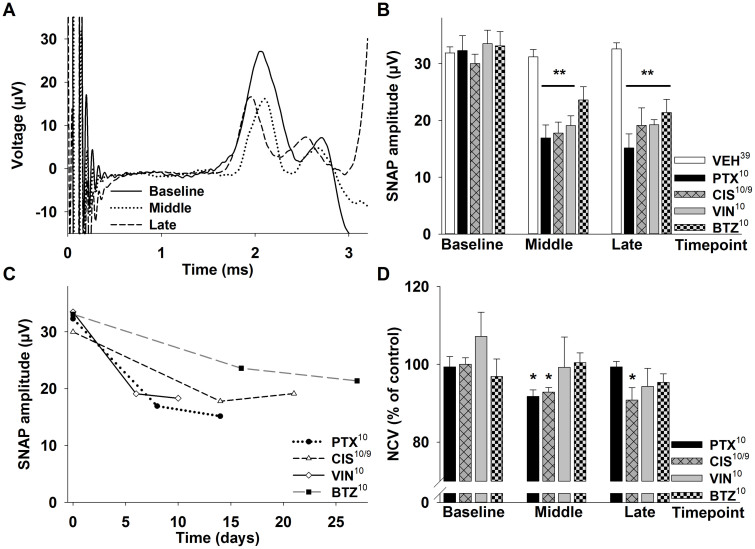
To further characterize sensory neuropathy, we measured nerve conduction velocity (NCV) and sensory nerve action potential (SNAP) in the caudal nerve. Antidromic stimulation of the caudal nerve in anesthetized mice revealed changes over time in the SNAP as well as in the NCV (Figure 2A). All animals receiving verum treatment developed a marked reduction of the caudal nerve SNAP amplitude. This was significant for paclitaxel (−39% ± 8%, [10], p < 0.01), cisplatin (−40% ± 7%, [9], p < 0.01) and vincristine (−51% ± 4%, [10] p < 0.001) at the middle time point. At the late time point, on average a week after the last injection, all injection protocols led to a significantly reduced SNAP amplitude with a reduction of −47% ± 9% for paclitaxel ([10], p < 0.001), −39% ± 10% for cisplatin ([9], p < 0.01), −39% ± 10% for vincristine ([10], p < 0.001) and −40% ± 7% in bortezomib treated animals ([10], p < 0.01; Figure 2B). Alterations of SNAP amplitudes developed faster in paclitaxel and vincristine treated animals compared to cisplatin and bortezomib therapy (Figure 2C).
Figure 2. Electrophysiological measurements in CIPN models.
(A) Representative serial electrophysiological measurements of the caudal sensory nerve in a mouse undergoing paclitaxel treatment. Compared to the baseline (solid line), the sensory nerve action potential (SNAP) amplitude is significantly reduced at the middle (dotted line) and late time point (dashed line). At the middle time point the potential is also shifted to the right, indicating a reduced nerve conduction velocity. (B) All animals receiving verum injections developed a significantly reduced SNAP amplitude of the caudal nerve which developed faster in PTX and VIN treated mice (C). (D) Nerve conduction velocity was only affected in paclitaxel- and cisplatin-treated animals. Superscript numbers indicate the number of animals used in each group (One CIS treated mouse died after the early time point). * p < 0.05; ** p < 0.01; compared to the substance specific vehicle control group at the same time point.
Previous studies noted a decreased NCV, indicative of demyelination, in animals treated with high doses of cisplatin, paclitaxel and bortezomib16. We observed a significant decrease of the NCV for paclitaxel at the middle time point (−8% ± 2% compared to baseline, [10], p < 0.05; Figure 2A + D), which had normalized 7 days later at the late time point. Cisplatin caused a more sustained reduction of the NCV which was observed at the middle (−8% ± 1%, [9], p < 0.05) and late time point (−9% ± 3%, [9], p < 0.05; Figure 2D). No significant alterations were observed for vincristine and bortezomib. In summary, all of the tested drugs led to a sustained reduction of SNAP amplitudes, suggesting a predominantly axonal pathology. Only paclitaxel and cisplatin conferred a comparatively small but significant reduction of the NCV, and in the case of paclitaxel the NCV recovered quickly after the last injection.
Analysis of sciatic nerve histology
After completion of behavioral and electrophysiological analysis, animals were killed and sciatic nerves were dissected, fixed, stained for myelin and cut as semi-thin sections (Figure 3A–E). Light microscopy was performed to capture an image of the entire nerve which was then subjected to semi-automatic software-based nerve morphometry in order to assess axon diameter and myelin thickness20. This method permits an analysis of myelinated fibers in the whole nerve which reduces possible selection bias.
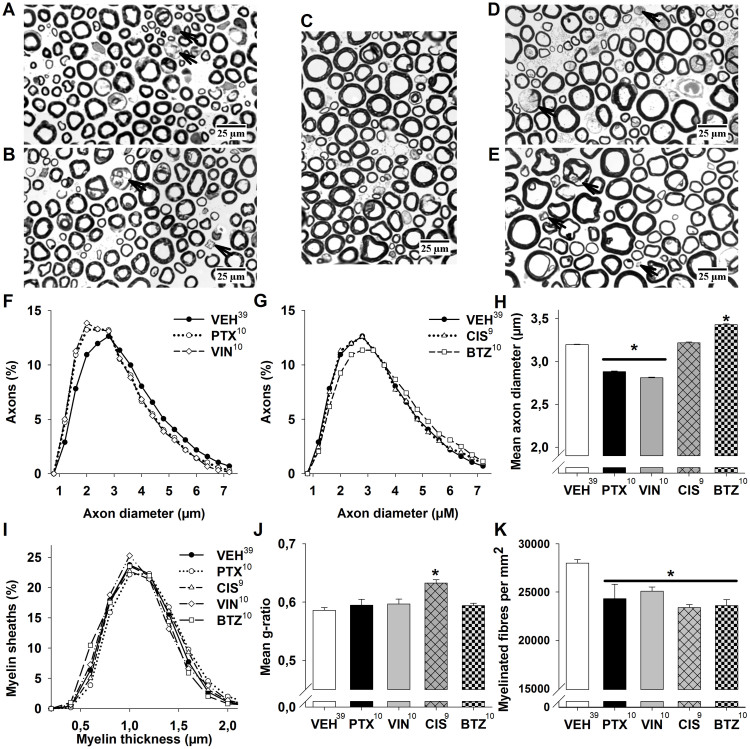
Figure 3. Histological analysis of sciatic nerves.
Representative micrographs of sciatic nerves from (A) paclitaxel-, (B) vincristine-, (C) control-, (D) cisplatin- and (E) bortezomib-treated animals. Degenerating axons are marked with an arrowhead. (F) Analysis of axon diameter distribution reveals that in paclitaxel- and vincristine-treated animals the histogram shifts to the left, suggesting a loss of bigger myelinated axons, while (G) cisplatin affected all myelinated fibers similarly and bortezomib led to a shift to the right, indicating a loss of smaller myelinated axons. (H) Mean axon diameter is reduced in paclitaxel- and vincristine-treated animals and increased in bortezomib-treated animals. (I) Distribution of myelin sheath thickness is similar in treated and control animals, underlining the predominantly axonal damage in animals suffering from chemotherapy-induced neuropathy. (J) Mean g-ratio is significantly increased in cisplatin treated animals indicating mild hypomyelination. (K) All cytostatic drugs lead to a significant reduction of myelinated fibre density. Superscript numbers indicate the number of animals used in each group.* p < 0.05; compared to the pooled control group.
Given the electrophysiological evidence for axonal damage, especially at the late time point, we were interested in whether changes in axon density or the distribution of axon diameters of myelinated fibers could be observed in animals treated with cytostatic drugs. For each substance a histogram of all axon diameters in this group was plotted and compared to the histogram of the pooled vehicles (approximately 125 000 axons). Interestingly, three distinct types of distribution were observed: In sciatic nerves from paclitaxel- or vincristine-treated animals, the histogram was shifted to the left, i.e. towards smaller axon diameters. This was also reflected by the mean axon diameter (PTX: 2.88 μm ± 0.01 μm, [10]; VIN: 2.81 μm ± 0.01 μm, [10], both p < 0.05) which was significantly smaller compared to control animals (3.20 μm ± 0.01 μm, [39], Figure 3F + H). After cisplatin treatment all types of myelinated fibers were equally affected, with an almost identical histogram compared to control [9]. Bortezomib treatment in turn led to a shift of the histogram to the right, i.e. bigger axon diameters (3.43 μm ± 0.01 μm, [10], p < 0.05; Figure 3G + H). In contrast to the distinct changes in the distribution of myelinated axons, no clear alterations of myelin thickness distribution were observed (Figure 3I). To further assess changes in myelination, we calculated the g ratio of nerve fibers (g-ratio denominates the numerical ratio between the diameter of the axon and the outer diameter of the myelinated fiber; hypomyelination would thus cause an increase of the g-ratio). G-ratio was moderately but significantly increased in cisplatin treated animals (0.63 ± 0.006 [9] vs. 0.59 ± 0.005 in controls [39], p < 0.05, Figure 3J). This finding reflects decreased NCV measured in the caudal nerve at the late time point for this substance (Figure 2D). Density of myelinated fibers was significantly (p < 0.05) reduced in all treatment groups compared to vehicle control (27997 ± 362/mm2, [39]), but strongest in cisplatin-treated animals with −17% ± 1% (23388 ± 322/mm2, [9]), −13% ± 5% in paclitaxel- (24065 ± 1402/mm2, [10]), −12% ± 2% in vincristine- (25089 ± 441/mm2, [10]) and −13% ± 2% in bortezomib-treated animals (23597 ± 622/mm2, [10], Figure 3K).
In conclusion, these findings suggest substance specific patterns of axonal damage. While paclitaxel and vincristine affect large more than small myelinated fibers, the opposite was the case with bortezomib (small > large), and cisplatin affects all types of myelinated fibers to a similar degree. Furthermore, cisplatin caused a small but significant increase of g-ratio suggesting hypomyelination.
Discussion
In this study we characterized subacute to chronic behavioral, electrophysiological and histological changes in C57Bl/6J mice with paclitaxel-, cisplatin-, vincristine- and bortezomib-induced neuropathy. Treatment schedules were chosen to induce a detectable neuropathic phenotype in all endpoints while at the same time limiting systemic side-effects to a minimum. Mortality was very low and with the exception of cisplatin-treated animals, weight loss during treatment was not significant. Activity patterns were altered only moderately and no significant changes of motor coordination or signs of paresis were observed in animals treated with cytostatic agents. All drugs led to a lower mechanical withdrawal threshold typical for the development of mechanical allodynia and distinct gait alterations. Animals suffering from CIPN minimized the duration of paw contact with the floor. Paclitaxel, cisplatin and vincristine affected the gait of hind paws more than forepaws, whereas the opposite was observed for bortezomib. Electrophysiological assessment showed that caudal nerve SNAP amplitude was markedly reduced in all treatment groups and NCV was affected in cisplatin- more than in paclitaxel-treated animals. Histological analysis of the sciatic nerve revealed substance specific patterns of damage: The strongest reduction of myelinated fiber density was seen in cisplatin-treated animals. Paclitaxel and vincristine affected large myelinated fibers more than small myelinated fibers, which was also reflected by the pronounced reduction of SNAP amplitudes in animals treated with these drugs. The opposite was observed for bortezomib, which shifted the distribution towards larger axons, while cisplatin affected all types of myelinated fibers to a similar degree. Cisplatin also led to an increase of g-ratio, a sign of hypomyelination, which is consistent with the significant decrease of NCV at the late time point. In summary, animals treated with paclitaxel, cisplatin, vincristine or bortezomib developed a predominantly sensory and axonal polyneuropathy. Development of neuropathic symptoms occurred faster in paclitaxel and vincristine treated animals compared to cisplatin and bortezomib. These findings correspond with clinical observations from patients undergoing treatment with the respective compounds (reviewed by18).
Behavioral assessment in our study comprised both general measures like locomotor activity in the open field test and gait alterations, as well as more specific parameters such as motor performance on the rotarod and the mechanical withdrawal threshold. Further behavioral testing, for instance to detect hot/cold allodynia, could have been included. However, it has to be kept in mind that exposing animals to repeated painful stimuli can result in distress and therefore an altered reaction to pain. In addition, anxiety and/or agitation which may be caused by overhandling and repeated testing are likely to impact open field and rotarod results. Therefore it was important to prevent stress by limiting the number of tests performed. Although it is known that CIPN is a mainly sensory neuropathy18,21 motor assessment should be included in the examination of CIPN models for two reasons: First, behavioral tests of the sensory nervous system frequently rely on a motor response in the animal, so the absence of motor deficits is important in validating these tests. Secondly, in addition to sensory alterations higher doses of certain drugs, like paclitaxel, have been shown to impair motor performance22. Apart from the predictable development of mechanical allodynia, we were interested in whether spontaneous pain-related behavior could be detected in CIPN models. As observed before19, gait alterations appeared later than electrophysiological changes or mechanical allodynia. Given the fact that gait analysis does not rely on behavior evoked by an artificial stimulus and is less influenced by the investigator as well as the methodological simplicity, these results may be of interest when preventative or therapeutic strategies are evaluated. Furthermore, the development of detectable gait alterations in several different models of CIPN underlines the usefulness of this technique in the evaluation of murine sensory polyneuropathy.
Neurophysiological changes were monitored in the caudal nerve, as previous studies demonstrated that it has a high sensitivity for toxins16,23 and alterations of the caudal nerve SNAP amplitude showed a strong positive correlation with the mechanical withdrawal threshold19. In the present study, alterations in caudal nerve SNAP amplitude developed in parallel to behavioral alterations. Electrophysiology of the caudal nerve is therefore a useful additional outcome parameter in animals developing CIPN. However, it is important to bear in mind that SNAP amplitudes mainly reflect the integrity of large myelinated fibers and thus are not suitable for assessing small myelinated and unmyelinated fibers. This was observed in bortezomib-treated animals, which only at the late time point developed a significant decrease of caudal nerve SNAP amplitude and histological analysis consistently showed that small and medium sized myelinated axons were more affected than large myelinated axons. One caveat of our experimental approach is that electrophysiology was conducted in the caudal nerve, while the sciatic nerve was subjected to histological analysis. Histological and electrophysiological data therefore can only be indirectly correlated given the distal-symmetrical nature of CIPN. Further neurophysiological evaluations such as sciatic and digital nerve neurophysiology could have been included, however this approach is challenging as insertion of electrodes in the hindlimbs may interfere with gait analysis.
In the assessment of preclinical CIPN models histology with semi-automatic nerve morphometry should be interpreted together with behavioral and electrophysiological measurements to obtain a comprehensive picture. Depending on the research focus, this approach can be complemented with histology of the spinal cord, the ventral/dorsal roots, dorsal root ganglia and intraepidermal nerve fibers. The observed histological changes in our study generally agree with previously reported nerve biopsy findings from patients undergoing cytostatic therapy: A significant loss of large myelinated fibers was observed in sural nerve biopsy after treatment with paclitaxel24. For vincristine both a predominant loss of large myelinated fibers as well as a more uniform reduction of all myelinated fibers in the sural nerve has been described25. Following cisplatin-treatment a significant reduction of myelinated fibers as well as abnormal myelination with an increased g-ratio was detected in sural nerve biopsies26,27. In contrast to our observations in mice, patient data shows a more pronounced loss of large myelinated fibers with a reduction of the mean axonal diameter after cisplatin-therapy27. Histological data from bortezomib treated patients is less abundant. However, published studies report marked axonal loss and degeneration in the sural nerve28 as well as a significant loss of intraepidermal nerve fibers29. In addition, development of a pure small fiber neuropathy without electrophysiological abnormalities seems to be frequent in patients undergoing bortezomib treatment29,30.
The results of our study introduce a comparative dataset for CIPN induced by different toxins in C57BL/6 mice which is summarized in Table 2. Some findings such as the development of mechanical allodynia and decreased caudal nerve SNAP amplitude are in line with previously published results and models of CIPN (reviewed by9) while others such as typical gait alterations and substance specific patterns of damage in myelinated fibers were not evaluated across several substances in previous studies. In comparison with comparative studies assessing neuropathy-inducing effects of paclitaxel, cisplatin, bortezomib and epothilon B16 as well as paclitaxel, eribulin mesylate and ixabepilone31 we report less severe alterations of sciatic nerve histology. This may be due to lower single and cumulative doses as well as to strain differences as discussed below.
Table 2. Semiquantitative summary of observed treatment effects in the course of the experiment. Statistically significant changes are marked with an asterisk.
| Endpoint | Paclitaxel | Cisplatin | Vincristine | Bortezomib |
|---|---|---|---|---|
| Weight | = | ↓↓* | = | ↓ |
| Behavior | ||||
| • Rotarod | = | = | = | = |
| • Von Frey | ↓↓↓* | ↓↓↓* | ↓↓↓* | ↓↓↓* |
| • Catwalk | ↓↓* | ↓↓* | ↓↓* | ↓↓* (forepaws) |
| Electrophysiology | ||||
| • NCV | ↓↓* | ↓↓↓* | ↓ | ↓ |
| • SNAP amplitude | ↓↓↓* | ↓↓↓* | ↓↓↓* | ↓↓↓* |
| Histology | ||||
| • Mean axon diameter | ↓↓* | = | ↓↓* | ↑↑* |
| • Axon density | ↓↓* | ↓↓↓* | ↓↓* | ↓↓* |
| • G-ratio | ↑ | ↑↑* | ↑ | ↑ |
Differences between species and strains can be quite distinct when preclinical pain research and specifically chemotherapy-induced neuropathy is concerned. In a study by Smith and coworkers12, DBA/2J mice were particularly sensitive towards paclitaxel, while C57BL/6 mice were more resistant and developed only a moderate decrease of the mechanical withdrawal threshold. To conclude that all studies should hence be conducted in sensitive mouse species may however be shortsighted. A multitude of available transgenic mice with C57BL/6 as genetic background, the broad availability of animals with this genotype as well as its thorough genetic and phenotypic characterization make this strain suitable for preclinical investigations of the pathomechanisms underlying CIPN and the development of preventative strategies. As C57BL/6 mice have a high susceptibility to diet-induced obesity and type 2 diabetes32, they present a suitable framework for studying the relevance of a pre-existing metabolic neuropathy in the development of CIPN. Another advantage of this strain is that despite its low susceptibility for tumor development, it is frequently used in different tumor models (reviewed by33), including models used in breast cancer research (e.g.34,35). The ability to evaluate antineoplastic activity and neurotoxic side effects in the same organism increases the possibility of finding prevention strategies for CIPN which do not impair antineoplastic activity. It also facilitates early screening for neurotoxic side effects in the development of novel antineoplastic drugs. The recent refusal of the European Medicines Agency (EMEA) to market the microtubule-stabilizing drug ixabepilone on the ground of safety concerns related to the frequency and magnitude of CIPN underlines the relevance of this approach36.
In conclusion, the present study describes the subacute to chronic course of CIPN onset in C57BL/6 mice for paclitaxel, vincristine, cisplatin and bortezomib. All substances induced a predominantly sensory and axonal polyneuropathy. The phenotype of the animals was assessed with behavioral, electrophysiological and histological tests. This study thus introduces a platform which allows further elucidation of pathomechanisms underlying CIPN development with genetic models on the common C57BL/6 background. In addition, the effectiveness of novel therapeutic and especially preventative strategies can be evaluated based on a combined analysis with the endpoints behavior, electrophysiology and histology.
Methods
Animals
A total of 80 nine-week-old male C57BL/6J mice from Charles River (Sulzfeld, Germany) were used for this study. Animals were assigned to cages with the help of randomly generated numbers to reduce possible litter effects. Mice were housed in groups of five and allowed food and water ad libitum. A 12:12 hour light/dark cycle (7 am–7 pm) was maintained and behavioral testing was conducted between 10 am and 6 pm. Injections administered on the same day as behavior tests, were given only after all testing had been completed. The general well-being of the mice was assessed each day and weight was recorded before and 24h after drug administration as well as before the Catwalk test. For each drug a corresponding control group treated with the appropriate vehicle was included. Baseline weights were comparable: 22.7 g ± 0.4 g (paclitaxel group, [10]), 22.4 g ± 0.4 g (paclitaxel control group, [10]), 22 g ± 0.2 g (cisplatin group, [10]), 22.7 g ± 0.2 g (cisplatin control group, [10]), 22.5 g ± 0.2 g (vincristine group, [10]), 21.8 g ± 0.2 g (vincristine control group, [10]), 21.6 g ± 0.2 g (bortezomib group, [10]), 22 g ± 0.3 g (bortezomib control group [9]). This study was approved by the official animal ethics committee of Berlin (Landesamt fuer Gesundheit und Soziales, Berlin, Germany) prior to the execution of the experiments. All experiments were conducted in accordance with government and institutional animal welfare guidelines.
Drug Injection protocol
Solutions of paclitaxel (Sigma-Aldrich, Taufkirchen, Germany), cisplatin (Sigma-Aldrich, Taufkirchen, Germany), vincristine sulfate (Tocris, Bristol, UK) and bortezomib (Selleck Chemicals, Houston, Texas, USA) were prepared before each treatment. All substances were injected intraperitoneally (i.p.); animals in the control groups received an injection of the corresponding vehicle. Treatment schedules, doses and time points are summarized in Table 1. We determined group sizes based on the effect size observed in experiments conducted previously with paclitaxel19, with a desired power of 0.8 and an alpha level of 0.05 using SigmaPlot software (SigmaPlot, Systat, Richmond, CA). The calculated sample size with two different groups (verum/vehicle) per substance was 10 animals per group.
Paclitaxel was dissolved in Cremophor EL:Ethanol (1:1) with a concentration of 2 mg/ml and diluted in sterile 0.9% NaCl solution to a final concentration of 0.1 mg/ml, a dose previously used in rat and mouse studies12,13. Cisplatin was dissolved in sterile 0.9% NaCl solution with a concentration of 0.23 mg/ml and administered according to the protocol published by14. Vincristine sulfate was dissolved in sterile 0.9% NaCl solution with a concentration of 20 μg/ml; the treatment protocol and dose were adapted from15. Bortezomib was dissolved in dimethylsulfoxide (DMSO) with a concentration of 1 mg/ml and diluted in sterile 0.9% NaCl solution to a final concentration of 40 μg/ml; the protocol was modified from16,17.
Behavior analysis
Behavior analysis was conducted on consecutive days: On day one mice were tested with the catwalk apparatus, allowed to rest and then tested on the rotarod. On day two, the mechanical withdrawal threshold was measured with the von Frey method, followed by electrophysiological tests in anesthesia. Animals were also tested in the open field twice: Testing was done one day prior to the tests described above and only at the early and late time point. Animals as well as cages were randomly selected for testing. A laboratory with soundproof chambers was used for all behavior tests.
Open Field
To determine general wellbeing of mice after injection of cytostatic agents, locomotion was evaluated in the open field test. Mice were placed in a box (120 cm × 120 cm × 40 cm) and monitored for 10 min at the early and late time points listed in table 1. An automated video tracking system (TSE Systems GmbH, Bad Homburg, Germany) collected information on seven different parameters. We analyzed the total distance traveled.
Rotarod
Motor coordination was assessed using the rotarod performance test as described before19. A baseline value was recorded on the last day of training by measuring the initial latency to fall off the rod. Subsequently testing was done at three time points to determine if motor coordination was affected (early, middle, and late (Table 1)). For each time point the results from three trials were averaged.
Von Frey Hair test
Mechanical allodynia was measured with an electronic von Frey hair test as described previously19,37. We recorded four time points (baseline, early, middle, and late (Table 1)) and per time point the results from five trials were averaged.
Catwalk
The Catwalk test was performed as reported previously19. In short: The catwalk apparatus (Noldus Information Technology, Netherlands) consists of a 1.3 m long black tunnel with a glass platform illuminated from within by total internal reflection of fluorescent light. Light is reflected and illuminates the stimulus, once downward pressure is applied. Walking patterns are captured with a high-speed camera mounted underneath the glass. Mice walked freely across the runway into their home cage. The experiment was performed sheltered from noise in a darkened room with red light. The Catwalk XT 8.1 software package was used for data analysis. Prior to the first injection, each animal was trained over four days to familiarize it to the task and the baseline was recorded on the last day of training. A trial was regarded as successful if the animal did not show a maximum speed variation greater than 60%, did not exceed a walking speed of 400 mm/s, and did not stop on the runway. Six compliant trials were recorded for each animal and time point (baseline, early, middle, and late (Table 1)). As CIPN-induced alterations develop in a symmetrical fashion, we analyzed parameters for the left and right side together.
Electrophysiology
NCV and SNAP amplitudes of the caudal nerve were recorded with a Dantec Keypoint electromyography system (Natus Medical Inc., Planegg, Germany) in animals anesthetized with isoflurane (1.3% to 1.5% in O2). The protocol was adapted from38. At the base of the tail stimulation electrodes were placed with the recording electrodes five cm distal and a ground electrode in-between the stimulation and recording electrodes. 50 stimuli (0.1 ms) with supramaximal stimulation intensity and a frequency of 1 Hz were averaged to measure SNAP and NCV at three timepoints (baseline, middle, and late).
Histology
Semi-thin sections of the sciatic nerve
Following behavior analysis at the late time point, animals were injected with a lethal amount of phenobarbital (100 mg/kg BW) and decapitated. Sciatic nerve samples from the mid-thigh were obtained from all animals, fixed in 2.5% glutaraldehyde and stained with osmium tetroxide prior to embedding in solvent-free, modified bisphenol A epoxy resin. 0.5 μm sections stained with toluidine blue were examined with light microscopy. Images of semi-thin sections were captured on a Leica DMRA microscope (Leica, Wetzlar, Germany) equipped with a PL-APO 100× oil immersion objective using a Retiga 2000 CCD camera (Qimaging, Surrey, BC, Canada), with a PC running MCID Core (InterFocus Imaging Ltd, Cambridge, England). Sub-images were automatically combined to one image comprising the entire sciatic nerve.
Semi-automatic nerve morphometry
Morphometrical analysis was performed as described previously20: semi-automatic software based evaluation was used to examine images of semi-thin sections of the sciatic nerve. After misidentified axons were manually corrected an analysis of the myelin sheath area was performed. The myelin as well as the axon area was exported to a text file. To facilitate comparison, these area measurements were transformed to axon diameters of an ideal (round) axon. Changes in myelination were assessed with G-ratios which are determined by calculating the ratio of (ideal) axon diameter to (ideal) total fiber diameter.
Statistical analysis
Data is expressed as mean ± sem and the manuscript was written in accordance with ARRIVE guidelines39. [N] depicts the number of mice used in each group. Data analysis was completed before the researcher was unblinded. Statistical analysis of the differences between treated versus control groups was performed at multiple time points by using a one-way ANOVA with the Holm-Sidak post-hoc test in case of normal distribution, and Dunn's method for samples which failed a Shapiro-Wilk normality test (SigmaStat, Systat, Richmond, CA). Data points which deviated by more than two standard deviations from the sample mean were treated as outliers in the catwalk analysis and the respective parameter pair (right/left) was excluded from analysis; this affected per experimental group between zero and (maximum) two animals. To obtain comparable results parameters were normalized to the respective control group and expressed as percent of baseline if statistical significance was detected between the treated and the control group in otherwise untransformed data. Vehicle groups in graphs depict pooled data from four different experiments; significance of behavioral and electrophysiological tests was however calculated against the substance-specific vehicle group only. p < 0.05 was considered statistically significant.
Acknowledgments
We would like to thank Catherine Aubel for thoughtful text editing, Heather L. More for the Matlab routine used to perform semi-automatic nerve morphometry and Petra Loge for excellent technical assistance. The research leading to these results has received funding from the federal ministry of education and research via the grant center for stroke research Berlin (01 EO 0801), the Volkswagen foundation (Lichtenberg program to Matthias Endres), and Deutsche Forschungsgemeinschaft DFG (NeuroCure). W. Boehmerle is participant in the Charité Clinical Scientist Program funded by the Charité Universitätsmedizin Berlin and the Berlin Institute of Health.
Footnotes
The authors declare no competing financial interests.
Author Contributions W.B., P.H., S.P. and M.B. designed and conducted experiment. W.B., P.H., S.P. and M.B. analyzed the data. W.B., P.H. and M.E. wrote the main text. W.B. made the figures 1–3. All authors reviewed the manuscript.
References
- Windebank A. J. & Grisold W. Chemotherapy-induced neuropathy. J Peripher Nerv Syst. 13, 27–46 (2008). [DOI] [PubMed] [Google Scholar]
- Schiff P. B., Fant J. & Horwitz S. B. Promotion of microtubule assembly in vitro by taxol. Nature 277, 665–667 (1979). [DOI] [PubMed] [Google Scholar]
- Johnson I. S., Armstrong J. G., Gorman M. & Burnett J. P. Jr The Vinca Alkaloids: A New Class of Oncolytic Agents. Cancer Res 23, 1390–1427 (1963). [PubMed] [Google Scholar]
- Kelland L. The resurgence of platinum-based cancer chemotherapy. Nat Rev Cancer 7, 573–584 (2007). [DOI] [PubMed] [Google Scholar]
- Mohty M., Brissot E., Savani B. N. & Gaugler B. Effects of bortezomib on the immune system: a focus on immune regulation. BBMT 19, 1416–1420 (2013). [DOI] [PubMed] [Google Scholar]
- Grisold W., Cavaletti G. & Windebank A. J. Peripheral neuropathies from chemotherapeutics and targeted agents: diagnosis, treatment, and prevention. Neuro-oncology 14 Suppl 4, iv45–54 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Argyriou A. A., Kyritsis A. P., Makatsoris T. & Kalofonos H. P. Chemotherapy-induced peripheral neuropathy in adults: a comprehensive update of the literature. Cancer Manag Res 6, 135–147 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Y. & Smith M. T. Pathobiology of cancer chemotherapy-induced peripheral neuropathy (CIPN). Front Pharmacol 4, 156 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Authier N. et al. Animal models of chemotherapy-evoked painful peripheral neuropathies. Neurotherapeutics 6, 620–629 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoke A. & Ray M. Rodent models of chemotherapy-induced peripheral neuropathy. ILAR J 54, 273–281 (2014). [DOI] [PubMed] [Google Scholar]
- Reagan-Shaw S., Nihal M. & Ahmad N. Dose translation from animal to human studies revisited. FASEB J 22, 659–661 (2008). [DOI] [PubMed] [Google Scholar]
- Smith S. B., Crager S. E. & Mogil J. S. Paclitaxel-induced neuropathic hypersensitivity in mice: responses in 10 inbred mouse strains. Life sciences 74, 2593–2604 (2004). [DOI] [PubMed] [Google Scholar]
- Polomano R. C., Mannes A. J., Clark U. S. & Bennett G. J. A painful peripheral neuropathy in the rat produced by the chemotherapeutic drug, paclitaxel. Pain 94, 293–304 (2001). [DOI] [PubMed] [Google Scholar]
- Ta L. E., Low P. A. & Windebank A. J. Mice with cisplatin and oxaliplatin-induced painful neuropathy develop distinct early responses to thermal stimuli. Mol Pain 5, 9 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alessandri-Haber N., Dina O. A., Joseph E. K., Reichling D. B. & Levine J. D. Interaction of transient receptor potential vanilloid 4, integrin, and SRC tyrosine kinase in mechanical hyperalgesia. J Neruosci 28, 1046–1057 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carozzi V. A. et al. Neurophysiological and neuropathological characterization of new murine models of chemotherapy-induced chronic peripheral neuropathies. Exp Neurol 226, 301–309 (2010). [DOI] [PubMed] [Google Scholar]
- Bruna J. et al. Neurophysiological, histological and immunohistochemical characterization of bortezomib-induced neuropathy in mice. Exp Neurol 223, 599–608 (2010). [DOI] [PubMed] [Google Scholar]
- Quasthoff S. & Hartung H. P. Chemotherapy-induced peripheral neuropathy. J Neurol 249, 9–17 (2002). [DOI] [PubMed] [Google Scholar]
- Huehnchen P., Boehmerle W. & Endres M. Assessment of Paclitaxel induced sensory polyneuropathy with “catwalk” automated gait analysis in mice. PloS one 8, e76772 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- More H. L., Chen J., Gibson E., Donelan J. M. & Beg M. F. A semi-automated method for identifying and measuring myelinated nerve fibers in scanning electron microscope images. J Neurosci Methods 201, 149–158 (2011). [DOI] [PubMed] [Google Scholar]
- Cavaletti G. & Marmiroli P. Chemotherapy-induced peripheral neurotoxicity. Nat Rev Neurol 6, 657–666 (2010). [DOI] [PubMed] [Google Scholar]
- Wang M. S. et al. Calpain inhibition protects against Taxol-induced sensory neuropathy. Brain. 127, 671–679 (2004). [DOI] [PubMed] [Google Scholar]
- Schaumburg H. H., Zotova E., Raine C. S., Tar M. & Arezzo J. The rat caudal nerves: a model for experimental neuropathies. J Peripher Nerv Syst 15, 128–139 (2010). [DOI] [PubMed] [Google Scholar]
- Sahenk Z., Barohn R., New P. & Mendell J. R. Taxol neuropathy. Electrodiagnostic and sural nerve biopsy findings. Arch Neurol 51, 726–729 (1994). [DOI] [PubMed] [Google Scholar]
- McLeod J. G. & Penny R. Vincristine neuropathy: an electrophysiological and histological study. J Neurol Neurosurg Psychiatry 32, 297–304 (1969). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krarup-Hansen A. et al. Examination of distal involvement in cisplatin-induced neuropathy in man. An electrophysiological and histological study with particular reference to touch receptor function. Brain 116 (Pt 5), 1017–1041 (1993). [DOI] [PubMed] [Google Scholar]
- Krarup-Hansen A. et al. Histology and platinum content of sensory ganglia and sural nerves in patients treated with cisplatin and carboplatin: an autopsy study. Neuropathol Appl Neurobiol 25, 29–40 (1999). [DOI] [PubMed] [Google Scholar]
- Stubblefield M. D. et al. An electrodiagnostic evaluation of the effect of pre-existing peripheral nervous system disorders in patients treated with the novel proteasome inhibitor bortezomib. Clin Oncol (R Coll Radiol) 18, 410–418 (2006). [DOI] [PubMed] [Google Scholar]
- Richardson P. G. et al. Single-agent bortezomib in previously untreated multiple myeloma: efficacy, characterization of peripheral neuropathy, and molecular correlations with response and neuropathy. J Clin Oncol 27, 3518–3525 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson P. G. et al. Frequency, characteristics, and reversibility of peripheral neuropathy during treatment of advanced multiple myeloma with bortezomib. J Clin Oncol 24, 3113–3120 (2006). [DOI] [PubMed] [Google Scholar]
- Wozniak K. M. et al. Comparison of neuropathy-inducing effects of eribulin mesylate, paclitaxel, and ixabepilone in mice. Cancer Res 71, 3952–3962 (2011). [DOI] [PubMed] [Google Scholar]
- Surwit R. S., Kuhn C. M., Cochrane C., McCubbin J. A. & Feinglos M. N. Diet-induced type II diabetes in C57BL/6J mice. Diabetes 37, 1163–1167 (1988). [DOI] [PubMed] [Google Scholar]
- Hunter K. W. Mouse models of cancer: does the strain matter? Nat Rev Cancer 12, 144–149 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson J. G. et al. p53-mediated senescence impairs the apoptotic response to chemotherapy and clinical outcome in breast cancer. Cancer cell 21, 793–806 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta P. B. et al. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 138, 645–659 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmonson T. & Ersboll J. Withdrawal assessment report for Ixempra. Report No. EMEA/594033/2008, 1–59 (European Medicines Agency, 2008). [Google Scholar]
- Cunha T. M. et al. An electronic pressure-meter nociception paw test for mice. Braz J Med Biol Res 37, 401–407 (2004). [DOI] [PubMed] [Google Scholar]
- Wang M. S., Davis A. A., Culver D. G. & Glass J. D. WldS mice are resistant to paclitaxel (taxol) neuropathy. Ann Neurol 52, 442–447 (2002). [DOI] [PubMed] [Google Scholar]
- Kilkenny C., Browne W. J., Cuthill I. C., Emerson M. & Altman D. G. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. J Pharmacol Pharmacother 1, 94–99 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]