Abstract
Adult neurogenesis in the hippocampal subgranular zone (SGZ) is involved in learning and memory throughout life but declines with aging. Mice lacking the CD44 transmembrane receptor for the glycosaminoglycan hyaluronan (HA) demonstrate a number of neurological disturbances including hippocampal memory deficits, implicating CD44 in the processes underlying hippocampal memory encoding, storage, or retrieval. Here, we found that HA and CD44 play important roles in regulating adult neurogenesis, and we provide evidence that HA contributes to age-related reductions in neural stem cell (NSC) expansion and differentiation in the hippocampus. CD44-expressing NSCs isolated from the mouse SGZ are self-renewing and capable of differentiating into neurons, astrocytes, and oligodendrocytes. Mice lacking CD44 demonstrate increases in NSC proliferation in the SGZ. This increased proliferation is also observed in NSCs grown in vitro, suggesting that CD44 functions to regulate NSC proliferation in a cell-autonomous manner. HA is synthesized by NSCs and increases in the SGZ with aging. Treating wild type but not CD44-null NSCs with HA inhibits NSC proliferation. HA digestion in wild type NSC cultures or in the SGZ induces increased NSC proliferation, and CD44-null as well as HA-disrupted wild type NSCs demonstrate delayed neuronal differentiation. HA therefore signals through CD44 to regulate NSC quiescence and differentiation, and HA accumulation in the SGZ may contribute to reductions in neurogenesis that are linked to age-related decline in spatial memory.
Keywords: CD44, hippocampus, hyaluronan, neural stem cell (NSC), neurogenesis
Introduction
The subgranular zone (SGZ)2 of the hippocampal dentate gyrus and the subventricular zone of the lateral ventricles are two regions in the adult brain where neural stem cells (NSCs) proliferate, migrate, and differentiate into new neurons throughout life (1, 2). In the SGZ, most NSCs differentiate into dentate granule cells that migrate into the inner granule cell layer and then functionally integrate into hippocampal neural circuits. Mice with reduced numbers of adult-born dentate granule cells have cognitive dysfunction characterized by deficiencies in forming hippocampus-dependent long term spatial memory and impaired performance in contextual fear extinction (3). Adult neurogenesis in the SGZ therefore contributes to at least some types of hippocampus-dependent learning and memory.
Adult neurogenesis in the SGZ decreases with aging, potentially contributing to age-related cognitive decline (4–9). Decreased neurogenesis in old age is at least partly linked to reduced NSC proliferation. NSCs demonstrate increased quiescence with aging that may be due to alterations in the NSC niche microenvironment (9–12). The mechanisms underlying altered NSC proliferation with aging are unknown.
The glycosaminoglycan hyaluronan (HA) accumulates in the rodent (13), non-human primate (14), and human brain with aging (15–17). HA comprises linear non-sulfated repeating units of (β,1→4)-d-glucuronic acid-(β,1→3)-N-acetyl-d-glucosamine synthesized by three transmembrane HA synthases (HAS1–3) that extrude HA into the extracellular matrix (18). Following damage in the nervous system, HA is synthesized at high levels by reactive astrocytes and inhibits proliferation of glial fibrillary acidic protein-positive cells and glial progenitor cells (19–21). Previous reports have indicated that HA is present in the SGZ and that NSCs express the transmembrane HA receptor, CD44 (22–26). Disruption of HA in the hippocampus by intrahippocampal injection of a hyaluronidase impaired hippocampus-dependent contextual fear conditioning (27). These cognitive deficits were attributed to alterations in use-dependent synaptic plasticity through HA-mediated regulation of dendritic calcium channels. However, contextual fear conditioning is also linked to adult neurogenesis in the SGZ (28). Furthermore, mice lacking CD44 have impaired hippocampus-dependent spatial memory retention in the probe trial following the first hidden platform training day in the Morris water maze (29). These findings suggest that CD44 and HA may influence hippocampus-dependent learning and memory through a mechanism involving the regulation of adult hippocampal neurogenesis.
Here, we found that CD44-expressing NSCs are self-renewing and multipotent and that they synthesize HA that accumulates with aging. CD44-null NSCs demonstrate increased proliferation in vitro and in vivo, an effect mimicked by digestion of HA in the SGZ of wild type mice. In addition, CD44-null and hyaluronidase-treated wild type NSCs demonstrate delayed neuronal maturation. These data provide novel insights into how the HA-based extracellular matrix regulates adult neurogenesis and suggest that accumulation of HA could contribute to reduced neurogenesis and impaired memory with aging.
Results
CD44-expressing NSCs Are Self-renewing Multipotent Cells
To confirm that CD44 is expressed by mouse NSCs and to assess how CD44 expression changes with aging, we examined CD44 expression in the SGZs of 3-month-old and 9-month-old wild type mice. At both ages, cell bodies and processes of cells in the SGZ expressed CD44 (Fig. 1A, arrows, and data not shown). The specificity of the immunoreactivity was confirmed by lack of staining in tissues from CD44-null mice (Fig. 1A, inset). CD44 was expressed on processes of cells expressing SOX2 (a transcription factor expressed by NSCs) within the hilus and in the SGZ (Fig. 1, B and inset in B) as well as on Ki67+ (a marker of proliferating cells) and nestin+ (an intermediate filament protein expressed by NSCs) cells in the SGZ (Fig. 1, C and D). CD44 was not expressed by MAP2+ (a neuron-specific microtubule-associated protein) cells in the granule cell layer (Fig. 1E).
FIGURE 1.
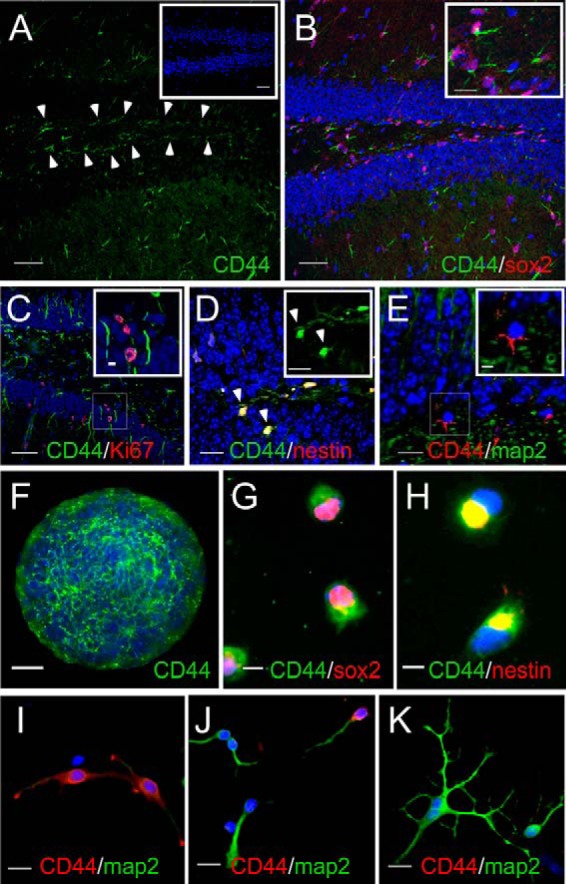
CD44 is expressed by NSCs in the SGZ. A, CD44 (green) is expressed by cells in the SGZ (arrowheads). The inset shows CD44 staining in a section of the dentate gyrus from a CD44-null mouse. B, CD44 (green) is expressed by SOX2 (red)-immunolabeled cells. The inset is a magnified image of the area outlined in the left portion of the figure. C, a section showing CD44+ processes (green) of cells with Ki67+ nuclei (red) in both the SGZ and granule cell layer. The area in the inset is an enlargement of the boxed area in the lower portion of the figure. D, a section demonstrating cell bodies that express both nestin (red) and CD44 (green). The inset shows the CD44 channel alone. E, a section showing a lack of CD44 (red) and MAP2 (green) co-localization, indicating that CD44 is not expressed by the granule cells derived from CD44+ NSCs. The inset is an enlargement of the boxed area in the lower left portion of the figure. F, whole mount immunolabeling for CD44 (green) of a neurosphere grown in vitro. G, acutely dissociated NSCs from neurosphere cultures immunolabeled for CD44 (green) and SOX2 (red). H, acutely dissociated NSCs from neurosphere cultures immunolabeled for CD44 (green) and nestin (red). The yellow signal indicates areas where CD44 and nestin co-localize. In all of the images, cells and tissues were stained with Hoechst 33342 (blue; to stain cell nuclei). I–K, CD44 expression declines as SGZ NSCs differentiate into neurons. SGZ-derived NSCs were grown in conditions that favored neuronal differentiation for 1 (I), 3 (J), and 6 days (K). Cells were stained with Hoechst 33342 (blue) to stain cell nuclei and antibodies against CD44 (red) and MAP2 (green). Note that as cells differentiate into more mature neurons they lose CD44 expression. Scale bars, 50 (A–C and inset in A), 15 (insets in B and D, D–F, and I–K), and 5 μm (G and H).
To further characterize CD44 expression by adult NSCs and granule cell layer neurons, we grew SGZ-derived neurospheres and dissociated NSCs under conditions that favored neuronal differentiation (Neurobasal medium with B27, fibroblast growth factor (FGF), and added glutamate) and immunolabeled the cells with an anti-CD44 antibody at different times in culture. Consistent with our in situ findings, CD44 was expressed by undifferentiated NSCs within neurospheres (Fig. 1F). Proliferating, acutely dissociated NSCs from these neurospheres co-expressed CD44 and SOX2 (Fig. 1G) as well as nestin (Fig. 1H). CD44 expression remained high when these cultures were switched to Neurobasal medium for 24 h (Fig. 1I). However, by 3 days in vitro, when cells demonstrated the morphology of more mature neurons and expressed MAP2, CD44 expression was diminished (Fig. 1J), and CD44 was completely absent in MAP2+ cells by 6 days in vitro (Fig. 1K). All together, these data indicate that NSCs in the SGZ express CD44 but that CD44 expression is diminished as NSCs differentiate into granule cell layer neurons.
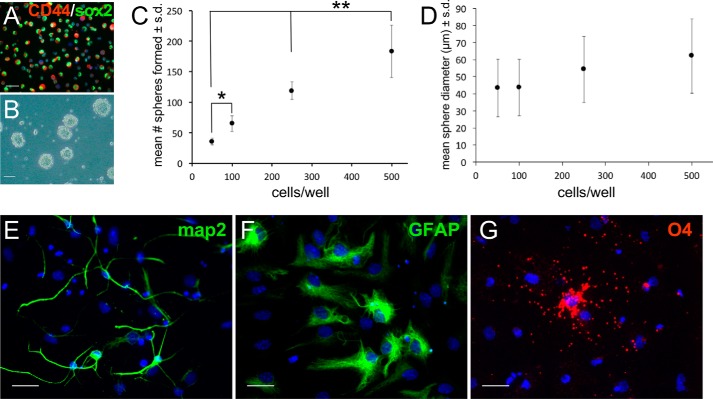
To assess the characteristics of CD44-expressing NSCs, we isolated CD44+ cells from dissociated primary SGZ neurosphere cultures by immunomagnetic cell sorting using CD44-biotin and anti-biotin microbeads. Cells were then double labeled using anti-SOX2 and anti-CD44 antibodies to confirm the efficiency of sorting. The majority of sorted cells demonstrated high CD44 immunolabeling with some cells showing less intense staining (Fig. 2A). From four separate sorts, 97.2 ± 0.9% of CD44+ cells expressed SOX2. Sorted cells were plated at clonal densities. Up to 60% of these cells formed secondary neurospheres (Fig. 2B). Plating at increasing cell densities resulted in significant increases in the numbers of neurospheres (Fig. 2C), whereas mean neurosphere densities did not significantly change with increasing plating density (Fig. 2D). These secondary neurospheres were transferred into culture media that promote their differentiation into neurons (as above), astrocytes (Dulbecco's modified Eagle's medium (DMEM) with B27 and 1% fetal bovine serum), or oligodendrocytes (DMEM/F-12 with B27, 0.1% bovine serum albumin, triiodothyronine, and N-acetyl-l-cysteine as described previously (30)). After 3 days in these differentiation media, the CD44+ cells gave rise to cells in each lineage (Fig. 2, E–G). All together, these findings indicate that CD44+ cells isolated from SGZ-derived neurospheres are self-renewing and multipotent.
FIGURE 2.
CD44-expressing NSCs are self-renewing, multipotent cells. A, image of freshly sorted, CD44+ SGZ-derived NSCs immunolabeled with antibodies against SOX2 (green) and CD44 (red) and stained with Hoechst 33342 (blue) to label cell nuclei. B, secondary neurospheres formed from sorted CD44+ NSCs. C, quantification of sphere forming assay as a function of NSC plating density. *, p < 0.001; **, p < 0.05. D, quantification of sphere diameter as a function of NSC plating density. Note there was no significant difference in mean sphere diameters with increasing plating density. E, MAP2 immunolabeling (green) in a culture of CD44+ NSCs grown in neuron differentiation medium. F, glial fibrillary acidic protein (GFAP) immunolabeling (green) in a culture of CD44+ NSCs grown in astrocyte differentiation medium. G, O4 immunolabeling (red) in a culture of CD44+ NSCs grown in oligodendrocyte differentiation medium. In each experiment (E–G), cells were stained with Hoechst 33342 (blue) to label cell nuclei. Scale bars, 25 (A and E–G) and 100 μm (B). Error bars represent S.D.
CD44 Regulates NSC Proliferation in the SGZ
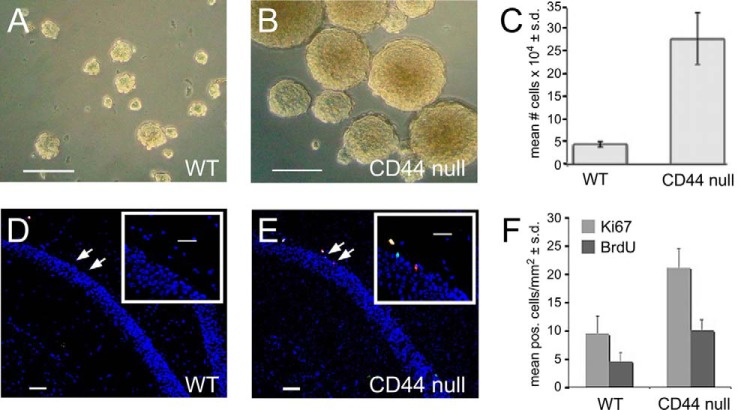
Given the hippocampal memory deficits we observed in CD44-null mice (29) and the finding that SGZ-derived NSCs are CD44-expressing cells, we assessed the role of CD44 in SGZ neurogenesis. We plated wild type and CD44-null SGZ NSCs from 2-month-old mice at equivalent cell densities and examined their growth and survival. Cells from both genotypes formed neurospheres (Fig. 3, A and B) and were each capable of differentiating into neurons, astrocytes, and oligodendrocytes although with different time courses (data not shown and see below). However, within 6 days, CD44-null neurospheres were 3–4 times larger than wild type neurospheres (Fig. 3B). The increased size of the CD44-null neurospheres was due to significant increases in cell proliferation (dividing 2–3 times faster than wild type cells) as assessed by cell counts (Fig. 3C). We did not observe any difference in the percentages of dead cells or cells expressing cleaved caspase-3 between wild type and CD44-null cultures (data not shown).
FIGURE 3.
NSCs from CD44-null mice hyperproliferate. A, SGZ neurospheres from 2-month-old WT mice grown for 6 days. B, SGZ neurospheres from 2-month-old CD44-null mice plated at the same density as the cells in A and grown for 6 days. C, quantification of total cell numbers in neurosphere cultures from WT and CD44-null mice (n = 4). *, p < 0.001. D, section through 9-month-old WT dentate gyrus following a 3-h BrdU pulse. Sections are immunolabeled with an anti-BrdU antibody (red), an anti-Ki67 antibody (green), and Hoechst 33342 (blue; to label cell nuclei). The inset is an enlarged view of the area indicated by the arrows. E, section through a 9-month-old CD44-null dentate gyrus immunolabeled as described in D. The inset is an enlarged view of the area indicated by the arrows. F, quantification of BrdU+ and Ki67+ cells in sections through the dentate gyrus of 9-month-old WT and CD44-null mice (n = 6). pos., positive. *, p < 0.01; **, p < 0.02. Scale bars, 100 (A and B), 50 (D and E), and 25 μm (insets in D and E). Error bars represent S.D.
To test whether CD44-null NSCs demonstrate increased rates of proliferation in situ, we compared 5-bromo-2-deoxyuridine (BrdU) incorporation and Ki67 expression in the SGZs of wild type and CD44-null mice. There was a significant 2-fold increase in BrdU incorporation by cells in the SGZ of 9-month-old (Fig. 3, D–F) CD44-null mice as compared with wild type animals. We also observed a slight but insignificant increase in proliferation in 3-month old animals (data not shown). These data indicate that CD44 signaling regulates NSC proliferation in the SGZ, especially in older animals. Interestingly, despite increased cell proliferation in the SGZs of CD44-null mice, the total volume of the DG was not significantly different in CD44-null animals (611,316 ± 105,778 μm3) compared with wild type mice (655,415 ± 146,968 μm3; p > 0.3).
CD44-null NSCs Demonstrate Delayed Neuronal Maturation
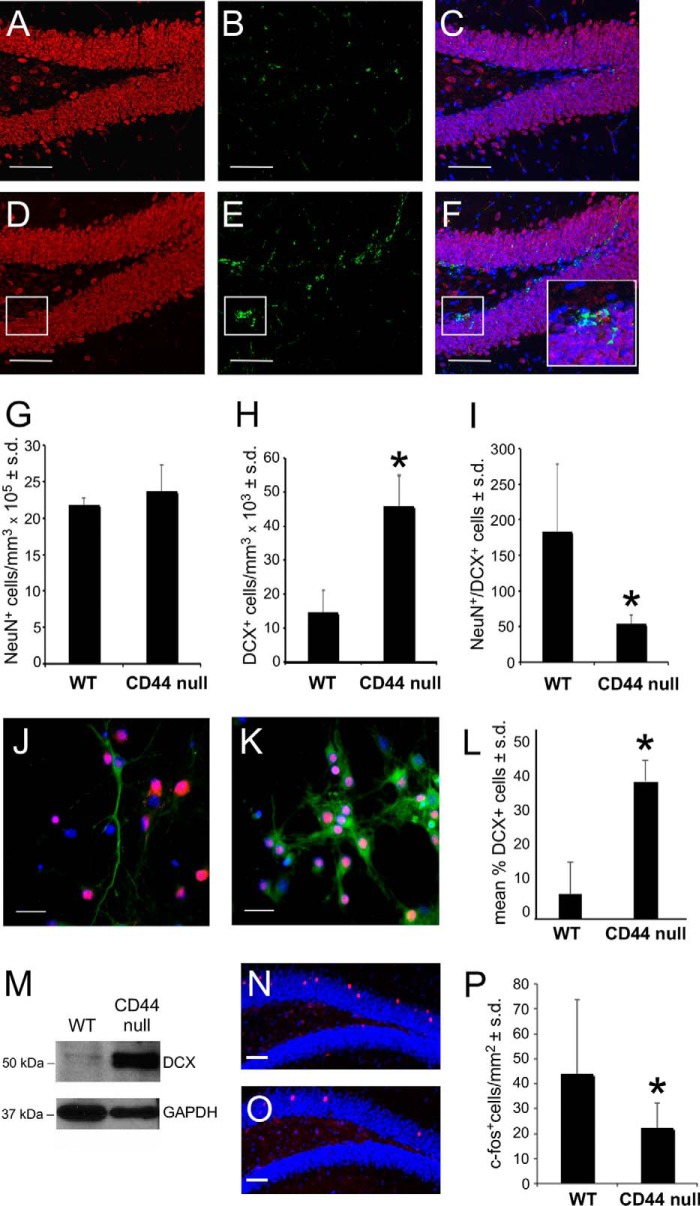
Changes in cell cycle rates can influence NSC differentiation (32). We therefore tested whether neuronal differentiation is altered in the granule cell layer of the dentate gyrus in CD44-null mice. In both 3- and 9-month-old CD44-null mice, we found no significant differences in the numbers of cells expressing the mature neuron marker NeuN (expressed predominantly in granule cell nuclei) as compared with wild type mice (Fig. 4, A, D, and G, and data not shown). However, there was a >2-fold increase in the numbers of cells expressing doublecortin (DCX), a marker of immature neurons (expressed predominantly in the processes of cells adjacent to the SGZ), in the inner granule cell layer of CD44-null as compared with wild type mice at 9 months of age (Fig. 4, B, C, E, F, and H). There was a slight increase in DCX+ cells at 3 months (data not shown). The increase in DCX+ cells in CD44-null mice led to a significant decrease in the ratio of NeuN+:DCX+ cells (Fig. 4I).
FIGURE 4.
Neuronal differentiation of CD44-null NSCs is delayed. A, NeuN staining (red) in the dentate gyrus of a 9-month-old WT mouse. B, DCX staining (green) in the same section shown in A. C, merged NeuN and DCX immunolabeling from A and B. The section was also stained with Hoechst 33342 (blue) to label cell nuclei. D, NeuN (red) immunolabeling in the dentate gyrus of a 9-month-old CD44-null mouse. E, DCX staining (green) in the same section shown in D. F, merged NeuN and DCX immunolabeling from D and E co-stained with Hoechst 33342 (blue). The insets in F are magnified areas outlined in the lower left portion of each figure. G, quantification of NeuN immunolabeling in the dentate gyrus of WT and CD44-null mice (n = 6) by unbiased stereological analysis. H, quantification of DCX immunolabeling in the dentate gyrus of WT and CD44-null mice (n = 6) by unbiased stereological analysis. *, p < 0.01. I, quantification of the ratios of NeuN:DCX immunolabeling in the dentate gyrus of WT and CD44-null mice. *, p < 0.001. J, acutely dissociated WT SGZ neurosphere cultures grown in neuronal differentiation medium and immunolabeled with NeuN (red) and DCX (green). Cells were also stained with Hoechst 33342 (blue) to label cell nuclei. K, acutely dissociated CD44-null SGZ neurosphere cultures grown in neuronal differentiation medium and immunolabeled as in J. L, quantification of DCX:NeuN ratios in WT and CD44-null NSC cultures grown in neuronal differentiation medium. *, p < 0.001. M, Western blot showing higher DCX expression in CD44-null as compared with WT SGZ-derived NSC cultures undergoing neuronal differentiation. Blots were probed for GAPDH as a loading control. N, dentate gyrus section from a 6-month-old WT mouse immunolabeled with an anti-c-Fos antibody (red) following behavioral training. The section was also stained with Hoechst 33342 (blue) to label cell nuclei. O, dentate gyrus section from a 6-month-old CD44-null mouse immunolabeled with an anti-c-Fos antibody as in N following behavioral training. P, quantification of c-Fos immunolabeling in the dentate gyrus of WT and CD44-null mice (n = 5). *, p < 0.001. Scale bars, 50 (A–F, N, and O) and 25 μm (J and K). Error bars represent S.D.
To test whether CD44 influences NSC differentiation in a cell-autonomous manner, we compared neuronal differentiation in cultures of SGZ NSCs from 2-month-old wild type and CD44-null mice. Cells were plated at identical cell densities, grown under conditions that favor neuronal differentiation as above, and then assayed for DCX and NeuN expression. Although most wild type cells became NeuN+ within 7 days, CD44-null NSCs remained DCX+ for greater than 10 days (Fig. 4, J–M).
Our findings are consistent with the hypothesis that the hippocampal memory deficits that we observed in CD44-null mice (29) are linked to delayed granule cell maturation. Following spatial learning, the transcription factor c-Fos is induced in granule cells that are recruited into spatial memory circuits (33, 34). We therefore examined c-Fos expression in the dentate gyri of 6-month-old female wild type and CD44-null mice following behavioral training as described previously (29). Compared with wild type mice, CD44-null animals displayed a significant reduction in induced c-Fos expression throughout the granule cell layer (Fig. 4, N–P). These data indicate that CD44 is required to regulate the expansion of NSCs and their differentiation into functional dentate granule layer neurons.
HA Is Expressed throughout the Dentate Gyrus and Increases with Age
HA, the major CD44 ligand, can be localized in cells and tissues using labeled HA-binding proteins. Using this approach, a previous study demonstrated strong extracellular HA staining in the adult rat SGZ (22). We therefore tested whether HA is similarly localized in the adult mouse SGZ using a biotinylated HA-binding protein (HABP) as described previously by us (19, 20). We found that HA is enriched in the SGZ in 3-month-old mice but is also found diffusely throughout the hilus and granule cell layer (Fig. 5A).
FIGURE 5.
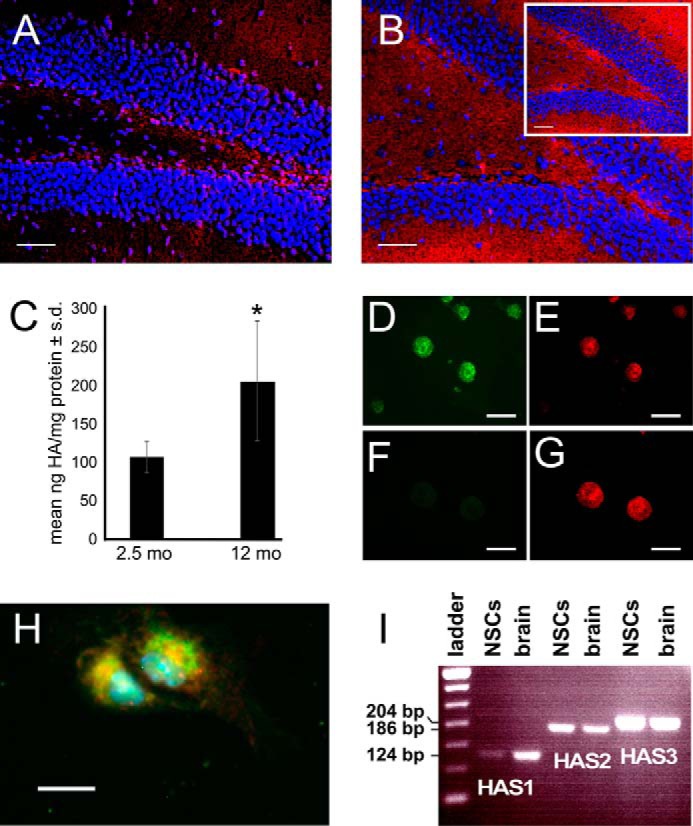
SGZ NSCs synthesize HA. A, section of dentate gyrus from a WT 3-month-old mouse labeled with a biotinylated HA-binding protein (red). The section was also stained with Hoechst 33342 (blue) to label cell nuclei. B, section of dentate gyrus from a WT 9-month-old mouse labeled with a biotinylated HA-binding protein as in A. The inset shows HA staining in a 9-month-old CD44-null mouse, demonstrating that HA accumulates in the dentate gyrus independently of CD44 expression. C, quantification of HA in young (2.5-month-old) and old (12-month-old) mouse dentate gyrus tissue lysates using an ELISA-based assay. *, p < 0.001. D, whole mount labeling for CD44 (green) of a WT neurosphere grown in vitro. E, whole mount labeling for HA (red) of the neurospheres shown in D. F, whole mount labeling for CD44 (green) of a CD44-null neurosphere grown in vitro. Note that only background signal is detectable. G, whole mount labeling for HA (red) in the neurospheres shown in F. H, WT NSCs were acutely dissociated from neurospheres and labeled with an anti-nestin antibody (green) and a biotinylated HA-binding protein (red). Cells were also stained with Hoechst 33342 (blue). I, RT-PCR for HAS1, HAS2, and HAS3 of RNA from WT mouse NSCs and WT 2-month-old mouse whole brain. Scale bars, 50 (A and B), 100 (D–G), and 15 μm (H). Error bars represent S.D.
We previously found that HA increases with age in prefrontal cortex (14). Given that the effects of CD44 on neurogenesis are most pronounced in older animals, we tested whether the levels of HA increase in the SGZ with age. HABP staining revealed that HA levels are higher throughout the dentate gyrus in 9-month-old mice as compared with 3-month old mice (Fig. 5B). This pattern of expression is not disrupted in CD44-null mice (Fig. 5B, inset). We quantified differences in levels of HA in the dentate gyrus by dissecting and pooling dentate gyrus tissue, isolating HA, and quantifying total HA concentrations using an enzyme-linked immunosorbent assay (ELISA)-based assay. We found that by 12 months, there was nearly double the amount of HA in the dentate gyrus compared with 2.5-month-old animals (Fig. 5C). Furthermore, we found that both wild type and CD44-null SGZ-derived NSCs synthesize HA in vitro (Fig. 5, D–H) and express all three HA synthases (Fig. 5I). Collectively, these data indicate that HA is present in the SGZ, is synthesized by NSCs, and accumulates with increasing age.
HA Inhibits SGZ-derived NSC Proliferation
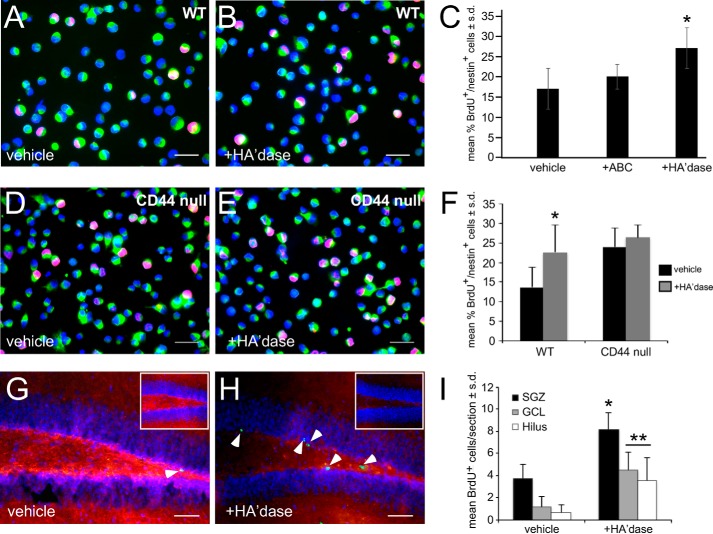
Given that elevated levels of HA are found in the SGZ of older adults and our finding that CD44, a major transmembrane HA receptor, regulates NSC proliferation in the SGZ, we tested whether adding HA to acutely dissociated NSC cultures influences NSC proliferation. Primary NSCs were isolated from wild type and CD44-null mice as described above and treated with a 100 μg of HA as described previously (20) for 48 h before the cells had synthesized substantial amounts of endogenous HA. Cells were pulsed with BrdU for the final 3 h in vitro, immunolabeled with antibodies against SOX2 and BrdU, and then stained with Hoechst 33342 (Fig. 6, A–D). Although wild type NSCs treated with HA incorporated significantly less BrdU than vehicle (PBS)-treated wild type cells (Fig. 6, A, C, and E), there was no significant difference in BrdU incorporation between CD44-null cells treated with vehicle and CD44-null cells treated with HA (Fig. 6, B, D, and E). Interestingly, HA had no effect on the proportion of DCX+ and NeuN+ cells in either wild type (Fig. 6F) or CD44-null (Fig. 6G) cultures. These data confirm that elevated HA can inhibit NSC proliferation in a CD44-dependent manner but does not, by itself, influence neuronal differentiation.
FIGURE 6.
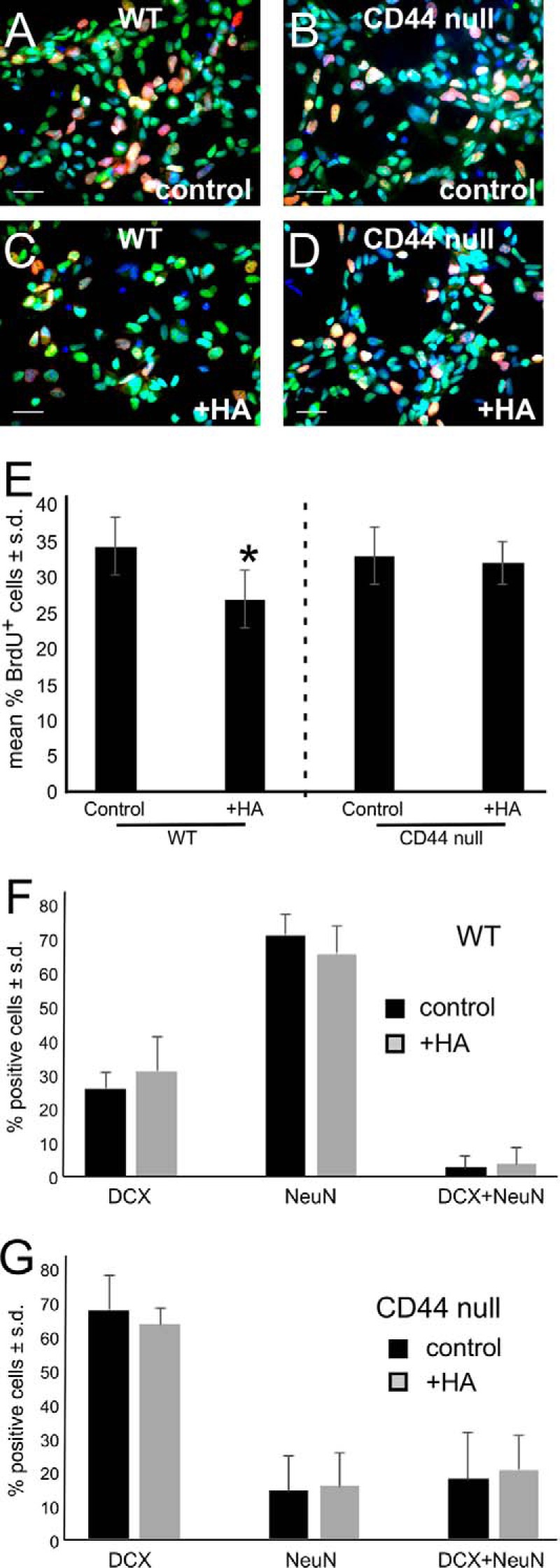
HA blocks NSC proliferation in a CD44-dependent manner. A–D, BrdU (red) and SOX2 (green) double labeling in wild type (A and C) and CD44-null (B and D) NSCs treated with vehicle (A and B) or 100 μg/ml HA (C and D). E, quantification of the percentage of BrdU+/SOX2+ HA-treated WT versus CD44-null NSCs compared with controls (cultures treated with vehicle). *, p < 0.001. F, quantification of the effects of HA on WT NSC neuronal differentiation (as assessed by DCX and NeuN immunolabeling) compared with vehicle controls. G, quantification of the effects of HA on CD44-null NSC neuronal differentiation compared with vehicle controls. Scale bars, 25 μm (A–D). Experiments were each performed three times. Error bars represent S.D.
Disruption of HA in the SGZ Induces NSC Proliferation and Delayed Neuronal Maturation
We tested whether disruption of HA in wild type NSC cultures mimics the phenotypes of CD44-null NSCs. SGZ-derived NSCs were grown in the presence of 20 units/ml recombinant PH20 (rPH20), a hyaluronidase that functions at neutral pH, or a preparation of bovine testicular hyaluronidase (BTH), the soluble activity of which is PH20. Fresh rPH20 or BTH was added to wild type cultures every 24 h. After 3 days, cells were treated with BrdU and then assayed for changes in cell proliferation. As shown in Fig. 7, A–C, the number of BrdU+/nestin+ cells significantly increased following rPH20 treatment. Similar results were obtained using BTH (data not shown). To rule out the possibility that the effects of hyaluronidase were due to activities other than HA digestion, we also tested the effects of a chondroitinase (chondroitinase ABC) on NSC proliferation using concentration ranges previously shown to digest chondroitin sulfate in cultures of NSCs (0.05–0.1 unit/ml; Ref. 35). Unlike rPH20 and BTH, chondroitinase did not influence NSC proliferation (Fig. 7C and data not shown).
FIGURE 7.
Digestion of HA surrounding NSCs induces NSC proliferation. A, WT NSCs were pulsed with BrdU after 3 days in vitro in the presence of vehicle (PBS). Cells were then immunolabeled with an anti-BrdU antibody (red) and an anti-nestin antibody (green). Cells were also stained with Hoechst 33342 (blue). B, WT NSCs were pulsed with BrdU after 3 days in vitro in the presence of hyaluronidase. Cells were immunolabeled for BrdU and nestin as in A. C, quantification of BrdU labeling in control and rPH20 (20 units/ml; HA'dase)- or chondroitinase ABC (0.1 units/ml)-treated SGZ NSC cultures (mean percentage of positive cells). *, p < 0.004. D, CD44-null NSCs were pulsed with BrdU after 3 days in vitro in the presence of vehicle (PBS). Cells were then immunolabeled with an anti-BrdU antibody (red) and an anti-nestin antibody (green). Cells were also stained with Hoechst 33342 (blue). E, CD44-null NSCs were pulsed with BrdU after 3 days in vitro in the presence of rPH20. Cells were immunolabeled for BrdU and nestin as in D. F, quantification of BrdU labeling in WT and CD44-null NSC cultures treated with vehicle or rPH20 (mean percentage of positive cells). *, p < 0.004. G, dentate gyrus section from a 6-month-old WT mouse stained with a biotinylated HA-binding protein (red) and an anti-BrdU antibody (green; arrowhead) 2 days following stereotactic injection of vehicle (PBS) and administration of BrdU. Sections were also stained with Hoechst 33342 (blue). Inset, dentate gyrus section from a 6-month-old WT mouse stained with a biotinylated HA-binding protein (red) 24 h following stereotactic injection of vehicle (PBS). Sections were also stained with Hoechst 33342 (blue). H, dentate gyrus section stained as in G from a 6-month-old WT mouse 2 days following stereotactic injection of rPH20. BrdU labeling was observed in the hilus and SGZ as well as in the inner face of the granule cell layer (arrowheads). Inset, dentate gyrus section stained as in G from a 6-month-old WT mouse 24 h following stereotactic injection of rPH20. I, quantification of BrdU labeling in different regions (hilus, SGZ, and granule cell layer) of dentate gyrus sections from animals treated with vehicle or rPH20 (n = 8). *, p < 0.001; **, p < 0.005. Scale bars, 25 (A, B, D, and E) and 50 μm (G and H). Experiments were each performed three times. Error bars represent S.D.
To test whether the effects of hyaluronidase treatment on NSC proliferation are dependent on CD44, we repeated the experiment shown in Fig. 7, A–C, using both wild type and CD44-null NSCs. In contrast to wild type NSCs, CD44-null NSC proliferation was not significantly altered in the presence of rPH20 (Fig. 7, D–F).
To test whether disruption of HA in the SGZ also induces increased NSC proliferation, we performed stereotactic injections of rPH20 (20 units in 1 μl) into the SGZ of adult mice (Fig. 7, G–I), pulsed animals with BrdU at 1 and 24 h postinjection, and then harvested their brains 24 h later. Control animals received injections of vehicle alone. Compared with vehicle controls (Fig. 7G, inset), HA was almost completely digested from animals that received rPH20 (Fig. 7H, inset) 24 h following injection. Some HA was returning in rPH20 sections 2 days after injection (e.g. Fig. 7, compare H with inset in H). Compared with controls, animals injected with rPH20 demonstrated significantly increased numbers of BrdU-labeled cells in the SGZ (Fig. 7, G–I).
We next tested whether disruption of HA influences neuronal differentiation by SGZ-derived NSCs. Similar to our findings in cultures of CD44-null NSCs, we found that treating wild type NSCs with rPH20 or BTH (as above) significantly delayed neuronal maturation from DCX+ to NeuN+ cells (Fig. 8, A–C, and data not shown). This effect was abolished in CD44-null NSCs (Fig. 8D). All together, these data are consistent with a model in which CD44 expressed by NSCs in the SGZ regulates neurogenesis and neuronal maturation in an HA-dependent manner.
FIGURE 8.
Hyaluronidase delays neuronal maturation of WT but not CD44-null NSCs. A, acutely dissociated WT SGZ neurosphere cultures grown in neuronal differentiation medium with vehicle and immunolabeled with NeuN (red) and DCX (green). Cells were also stained with Hoechst 33342 (blue) to label cell nuclei. B, acutely dissociated WT SGZ neurosphere cultures grown and immunolabeled as in A but treated with rPH20. C, quantification (mean percentage of positive cells) of DCX+, NeuN+, and double positive immunolabeling in WT NSC cultures undergoing neuronal differentiation and treated with vehicle (PBS) or rPH20. *, p < 0.001; **, p < 0.05. D, quantification of DCX and NeuN immunolabeling as in C in CD44-null NSC cultures undergoing neuronal differentiation and treated with vehicle (PBS) or rPH20. Scale bars, 50 μm (A and B). Error bars represent S.D.
Discussion
Growing evidence supports a role for the extracellular matrix in regulating NSC quiescence, expansion, and differentiation within NSC niches (36, 37). However, the roles of specific glycosaminoglycans and their receptors in adult neurogenesis are not well understood (38). We have identified a novel role for CD44 and HA as regulators of NSC quiescence in the SGZ. HA is enriched in the SGZ, and digestion of HA by a hyaluronidase or genetic ablation of CD44 each result in loss of NSC quiescence. Our in vitro studies of neurospheres indicate that CD44 and HA act on NSCs in a cell-autonomous manner because NSCs derived from both CD44-null neurospheres and wild type neurospheres treated with hyaluronidase demonstrated increased proliferation. Furthermore, both CD44-null NSCs and wild type NSCs treated with hyaluronidase exhibited delayed neuronal maturation. This delayed maturation could be linked to the shortened cell cycle of the NSCs. Inhibitors of cell proliferation promote neurogenesis, whereas positive regulators of cell cycle progression and cell proliferation inhibit neuronal differentiation (32).
It is also possible that HA plays a more direct role in regulating neuronal maturation. Although intact, high molecular weight HA is required to maintain NSC quiescence in the SGZ, digestion products of HA could function to block NSC differentiation or neuronal progenitor cell maturation. HA digestion products inhibit the maturation of oligodendrocyte progenitor cells in vitro and in demyelinating lesions (30, 39). HA digestion products that accumulate both in NSC cultures and in the SGZ following treatment with hyaluronidase may therefore similarly block neuronal maturation. This raises the interesting possibility that the regulated catabolism of HA in the SGZ could function to regulate neuronal maturation in situ.
CD44-null mice and wild type mice treated with hyaluronidase demonstrated greater increases in NSC proliferation in older compared with younger animals. Previous reports demonstrated that HA accumulates in the brain with aging (14–17), and we found here that HA accumulates in the SGZ of older mice. Adding excess HA to NSC cultures inhibits NSC proliferation, whereas disruption of HA in NSC cultures and in the SGZ leads to increased NSC proliferation. These data support the notion that elevated HA in the aging brain would limit NSC proliferation.
Surprisingly, despite the fact that CD44-null mice have cell-autonomous increases in cell proliferation, there was not a significant difference in dentate gyrus volumes between WT and CD44-null animals. These findings are consistent with a previous study showing that voluntary exercise in a running wheel increased the numbers of BrdU-labeled NSCs by over 200% without altering the volume of the dentate gyrus (40). However, both in our study and the exercise study, the reasons for the unchanged dentate gyrus volumes are unclear. In agreement with our findings in vitro, we did not observe any increases in apoptosis in the SGZ or granule cell layer of the dentate gyrus in CD44-null animals (data not shown). It is possible that cells are dying through a different mechanism or that apoptosis is occurring at a rate that is difficult to detect.
The delayed neuronal maturation in CD44-null animals was accompanied by reduced c-Fos activation in the hippocampal granule cell layer following behavioral training, which could explain our previous findings showing that CD44-null animals have hippocampal memory deficits (29). This reduced c-Fos activation might reflect changes in the complexity of networks that respond to behavioral training as a result of delayed granule cell maturation. In combination with the effects of HA on NSC proliferation, the data in this report are consistent with the hypothesis that excess HA in the aging SGZ contributes to the age-related declines in adult neurogenesis and neuronal function. It is intriguing to speculate that altering HA catabolism in the SGZ or lowering CD44 expression by NSCs could be a way to enhance neurogenesis in the aged brain.
Although our data support a role for HA synthesis and catabolism in adult neurogenesis, HA and CD44 may play additional roles in mature neurons that could impact cognitive function. An earlier study demonstrated impaired contextual fear memory in wild type mice following the injection of hyaluronidase into the hippocampus (27). In that study, the effects of hyaluronidase were attributed to disruption of the HA-based extracellular matrix in perineuronal nets, leading to alterations in the mobility and activity of α-amino-3-hydroxyl-5-methyl-4-isoxazolepropionate (AMPA) glutamate receptors and L-type voltage-dependent calcium channel-mediated long term potentiation (27, 41). Although contextual fear memory involves L-type voltage-dependent calcium channels (42, 43), some components of contextual fear conditioning may also involve hippocampal neurogenesis (44–46). We found that CD44-null mice demonstrated impairments in another test of hippocampal memory, the Morris water maze, which is also sensitive to alterations in adult neurogenesis (29, 47).
In another study, CD44 has been implicated in regulating synaptic plasticity through a mechanism involving regulation of the actin cytoskeleton (48), and altered synaptic plasticity in CD44-null mice may lead to a number of behavioral phenotypes including increased susceptibility to stress-induced anxiety (49). In addition, mice lacking Has3 demonstrate extensive loss of HA in the hippocampus that causes increased cell packing in the CA1 stratum pyramidale (50). These mice develop seizures associated with hippocampal epileptiform activity, but it is unclear whether neurogenesis is perturbed in these animals.
In addition to CD44, other transmembrane HA receptors may play a role in regulating NSC proliferation and differentiation. An earlier study reported that toll-like receptor 2 activation by digested HA inhibited the proliferation of NSCs from the subventricular zone (51). These studies would seem to conflict with our findings where HA digestion and presumably the accumulation of HA digestion products lead to increased NSC proliferation. It is possible that these differences are due to the fact that our study used SGZ-derived NSCs instead of subventricular zone-derived cells. Alternatively, the HA and hyaluronidase used in the earlier study may have produced different digestion products than those that arose in our experiments, raising the intriguing possibility that different sizes of HA could have distinct effects on NSCs.
Interestingly, following seizures in wild type animals, there is a dramatic increase in NSC proliferation in the SGZ and an accumulation of DCX+ immature neurons, alterations that correlate with the promotion of subsequent seizures and associated cognitive difficulties (52–58). HA and levels of Has3 are altered in the hippocampus following seizures in rats (59). Furthermore, degradation of HA or treatment with CD44 function-blocking antibodies influenced seizure-induced mossy fiber sprouting (60). Cells demonstrating aberrant mossy fiber sprouting are adult-born dentate granule cells (61). Our data therefore suggest a novel mechanism for the dysregulation of neurogenesis in both the elderly nervous system and possibly following seizures and point to potential, novel therapeutic strategies to restore normal neurogenesis in aging and disease.
Experimental Procedures
Mice
Mice were housed in standard conditions. All animal experiments were performed in accordance with local, institutional, and international guidelines for the care and use of experimental animals. Wild type and CD44-null C57BL/6J;129 mice were purchased from The Jackson Laboratory, then bred, and maintained in the mouse facilities of the Oregon National Primate Research Center or the Oregon Health and Science University. Mice were subjected to behavioral testing as described previously (29) and used at ages as indicated in the text.
Cell Culture
Adult NSC cultures were prepared from 2-month-old female mice. For each animal, the dentate gyrus was minced and then digested enzymatically with 0.2% trypsin (Invitrogen) for 20 min at 37 °C. The digestion was stopped with 10% fetal bovine serum in DMEM (Invitrogen). The cells were washed with DMEM, filtered through a 70-μm cell strainer (BD Biosciences), and then resuspended in stem cell culture medium comprising DMEM/F-12 (Invitrogen) with 0.1% BSA (Sigma), 2% B27 supplement without vitamin A, 1% N2 supplement (Invitrogen), 20 ng/ml recombinant murine epidermal growth factor (EGF; R&D Systems), and 20 ng/ml recombinant human FGF-2 (Millipore). Cultures were monitored daily. Neurospheres were passaged when they were around 100 μm in diameter. For neurosphere forming assays, NSCs were either plated at single cell density or between 50 and 500 cells/200 μl in each well of a 96-well plate. The numbers and diameters of neurospheres were measured at 6 days in vitro.
For NSC monolayer cultures and for experiments involving neuronal differentiation, neurospheres were dissociated into single cells in Accutase (Sigma-Aldrich)and then plated at a density of 30,000 cells/coverslip in 24-well plates coated with poly-l-lysine (Sigma-Aldrich). NSCs were incubated in stem cell culture medium or neuron culture medium (Neurobasal with 2% B27 and 5 ng/ml FGF-2). The medium was changed every 48 h. Cells were treated with hyaluronidase (recombinant bovine PH20from R&D Systems or bovine testicular hyaluronidase type I-S from Sigma) as described previously (30), chondroitinase ABC (AMSBIO) as described in the text, or highly pure HA (106 Da; Lifecore and a gift from Paul Weigel).
Established cultures of NSCs were dissociated with Accutase and then subjected to immunomagnetic cell sorting using CD44-biotin and anti-biotin microbeads (Miltenyi Biotec Inc.) according to the manufacturer's instructions. The CD44+ cell fractions were seeded in stem cell culture medium as above on coverslips for 1 h before analysis or grown in differentiation medium as described in the text. For in vitro proliferation assays, cells were counted or pulsed with BrdU (Sigma) as described previously (19).
Stereotaxic Injection
Mice were anesthetized (2.5–5% isoflurane delivered via a vaporizer), and either PBS or hyaluronidase (total, 20 units; recombinant PH20 or bovine testicular hyaluronidase) was unilaterally injected into the dentate gyrus at 1.0 μl/site (0.1–0.2 μl/min) at the coordinates anteroposterior = −2 mm from bregma, lateral = ±1.5 mm, ventral = 2.3 mm by stereotaxic injection as described (30). All mice received an intraperitoneal injection twice at 1 and 24 h postinjection with 80 mg/kg BrdU dissolved in 0.9% NaCl. Mice were sacrificed at 48 h postinjection, and frozen coronal brain sections (30 μm thick) were prepared and processed for immunostaining.
Immunocytochemistry and Immunohistochemistry
Mouse brains were fixed in 4% paraformaldehyde in 0.1 m phosphate buffer overnight at 4 °C and then embedded in optimal cutting temperature (OCT) medium for cryosectioning or in paraffin and sectioned. Paraffin sections were deparaffinized, rehydrated, and treated with antigen retrieval buffer or with 2 n HCl for BrdU detection. Cultured cells were washed three times with PBS (pH 7.4) and fixed in 4% paraformaldehyde for 15 min. Cells were then permeabilized by incubation for 5 min in 0.1% Triton X-100 in PBS. Sections and cells were blocked with 5% donkey or goat serum for 1 h and then incubated overnight at 4 °C with primary antibodies. The next day, sections or cells were incubated with the appropriate species-specific fluoroconjugated secondary antibodies (Alexa Fluor 546 or Alexa Fluor 488; 1:1000; Molecular Probes Inc.) or FITC-conjugated anti-chicken IgY (1:500; Aves Labs) and Hoechst 33342 (1:10,000; Molecular Probes) for 1 h to label cell nuclei. Primary antibodies used were rat anti-CD44 (IM7; 1:50; a gift from Dr. Claire Isacke), chicken anti-nestin (1:500; Aves Labs), rabbit anti-SOX2 (1:500; Abcam), rabbit anti-Ki67 (1:400; Novo UK), rabbit anti-MAP2 (1:1000; Chemicon), mouse anti-NeuN (1:200; Millipore), goat anti-DCX (1:200; Santa Cruz Biotechnology), rabbit anti-c-Fos (1:10,000; Calbiochem), and mouse anti-BrdU (1:200; Calbiochem). HA was visualized using a biotinylated HABP (1:200; Seikagaku Corp., Tokyo, Japan) followed by avidin-conjugated Cy3 (1:1000; Molecular Probes Inc.) as described previously (20).
Stained sections and cells were mounted and examined by fluorescence microscopy using a Zeiss Axioskop 40 equipped with epifluorescence and an AxioCam MRc digital camera system. Images were acquired controlling for equal exposure times using AxioVision 4.5 software. For confocal microscopy, images were acquired with a Leica TCS SP confocal microscope using a 40× numerical aperture (NA) 1.25 Pl-Apo objective, pinhole 1 Airy unit. For double immunofluorescence staining, data from two channels (Alexa Fluor 488 and Alexa Fluor 546) were collected by sequential scanning. Single confocal planes were analyzed to determine co-localization of signals in tissue sections. Z-stacks of images were collected at 0.5-μm intervals to generate maximum projections.
Quantification of HA
HA was quantified from dentate gyrus lysates obtained by pooling microdissecting tissues from mice at specific ages and then lysing them in ice-cold lysis buffer containing 20 mm Tris-HCl, 150 mm NaCl, 1 mm EDTA, and 1% Triton X-100. Protein concentrations were measured using a BCA protein assay (Thermo Fisher). Samples with the same protein levels were applied to an ELISA-based HA quantification assay (Echelon Biosciences) according to the manufacturer's instructions. Lysates were diluted to a final volume of 350 μl in the kit diluent buffer. Triplicate 100-μl fractions were transferred into the ELISA plate. At the end of the assay, absorbances were read at 405 nm on a 96-well plate reader (Molecular Devices).
Stereological Analysis of Dentate Gyrus
Brains were cut on a cryostat into 20-μm-thick coronal sections. Every 10th section was mounted on a glass slide and stained with NeuN, DCX, and Hoechst 33342. Sections were anatomically matched between animals. A Marianas stereological work station with Slidebook 4.2 was used for analysis. Each section was examined, and the dentate gyrus granule cell layer was delineated on NeuN/DCX-stained sections. The work station captured multiple 20× images across the span of the dentate gyrus and built a 20× montage of the entire area. The counting points were randomly selected, and 3D images were captured for NeuN- or Hoechst 33342-positive cell counting. DCX-positive cell numbers were counted in entire sections. The cell densities were calculated and then subjected to further statistical analysis.
Western Blotting
Total cell lysates were prepared and analyzed as described (31). Blots were probed with goat anti-DCX (1:2000) and mouse anti-GAPDH (1:20,000; Sigma) followed by horseradish peroxidase-conjugated donkey anti-goat and donkey anti-mouse antibodies (1:10,000; Jackson ImmunoResearch Laboratories). Proteins were visualized using an enhanced chemiluminescence system (Amersham Biosciences).
RT-PCR
Total RNA was extracted from cell cultures and tissues by TRIzol reagent (Invitrogen) according to the manufacturer's instructions. Isolated total RNA was treated with RNase-free DNase I (Stratagene) for 30 min. The total RNA concentration was determined spectrophotometrically in RNase-free water, and 1-μg aliquots of total RNA were reverse transcribed and amplified as described previously (30). The oligonucleotide primers for RT-PCR were based on the sequences for mouse Has1, Has2, and Has3 (30). PCR products were separated by electrophoresis on 1% agarose gels containing ethidium bromide (0.5 μg/ml) in Tris borate/EDTA buffer. A molecular weight marker was run in parallel, and bands of the expected molecular size were detected under UV light.
Statistical Analyses
Comparisons of different positive cell numbers between CD44 wild type and CD44-null dentate gyrus tissues or cultured cells were performed using a Student's t test with significance at p < 0.05. For all analyses, the null hypothesis was rejected at the 0.05 level.
Author Contributions
L. S. S. conceived and coordinated the study and wrote the paper. W. S. designed, performed, and analyzed all of the in vitro experiments and performed and analyzed all of the immunohistochemical analyses. S. C. F., R. X., and K. F. assisted with the in vitro and in vivo experiments including stereotaxic injections, tissue harvesting, and cell and tissue preparation. R. H. J. O., S. F. A., and J. R. performed all of the behavioral testing. All authors reviewed the results, provided feedback on the manuscript, and approved the final version of the manuscript.
Acknowledgment
We thank Dr. Steven Matsumoto for helpful comments. The Oregon National Primate Research Center was supported by Core Grant RR00163 from the National Institutes of Health. The imaging core at the Oregon National Primate Research Center was supported by Grant P30-NS061800 from the National Institutes of Health.
This work was supported by National Institutes of Health Grant NS056234 (to L. S. S.) and Training Grant T32 DA07262 from the National Institute on Drug Abuse (NIDA), and a grant from Vertex Pharmaceuticals. The authors declare that they have no conflicts of interest with the contents of this article. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
- SGZ
- subgranular zone
- HA
- hyaluronan
- NSC
- neural stem cell
- HAS
- HA synthase
- DCX
- doublecortin
- HABP
- HA-binding protein
- rPH20
- recombinant PH20
- BTH
- bovine testicular hyaluronidase.
References
- 1. Spalding K. L., Bergmann O., Alkass K., Bernard S., Salehpour M., Huttner H. B., Boström E., Westerlund I., Vial C., Buchholz B. A., Possnert G., Mash D. C., Druid H., and Frisén J. (2013) Dynamics of hippocampal neurogenesis in adult humans. Cell 153, 1219–1227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ming G. L., and Song H. (2011) Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 70, 687–702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Deng W., Saxe M. D., Gallina I. S., and Gage F. H. (2009) Adult-born hippocampal dentate granule cells undergoing maturation modulate learning and memory in the brain. J. Neurosci. 29, 13532–13542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Kuhn H. G., Dickinson-Anson H., and Gage F. H. (1996) Neurogenesis in the dentate gyrus of the adult rat: age-related decrease of neuronal progenitor proliferation. J. Neurosci. 16, 2027–2033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Bondolfi L., Ermini F., Long J. M., Ingram D. K., and Jucker M. (2004) Impact of age and caloric restriction on neurogenesis in the dentate gyrus of C57BL/6 mice. Neurobiol. Aging 25, 333–340 [DOI] [PubMed] [Google Scholar]
- 6. Bizon J. L., Lee H. J., and Gallagher M. (2004) Neurogenesis in a rat model of age-related cognitive decline. Aging Cell 3, 227–234 [DOI] [PubMed] [Google Scholar]
- 7. Ben Abdallah N. M., Slomianka L., Vyssotski A. L., and Lipp H. P. (2010) Early age-related changes in adult hippocampal neurogenesis in C57 mice. Neurobiol. Aging 31, 151–161 [DOI] [PubMed] [Google Scholar]
- 8. Encinas J. M., Michurina T. V., Peunova N., Park J. H., Tordo J., Peterson D. A., Fishell G., Koulakov A., and Enikolopov G. (2011) Division-coupled astrocytic differentiation and age-related depletion of neural stem cells in the adult hippocampus. Cell Stem Cell 8, 566–579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Miranda C. J., Braun L., Jiang Y., Hester M. E., Zhang L., Riolo M., Wang H., Rao M., Altura R. A., and Kaspar B. K. (2012) Aging brain microenvironment decreases hippocampal neurogenesis through Wnt-mediated survivin signaling. Aging Cell 11, 542–552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Hattiangady B., and Shetty A. K. (2008) Aging does not alter the number or phenotype of putative stem/progenitor cells in the neurogenic region of the hippocampus. Neurobiol. Aging 29, 129–147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Ahlenius H., Visan V., Kokaia M., Lindvall O., and Kokaia Z. (2009) Neural stem and progenitor cells retain their potential for proliferation and differentiation into functional neurons despite lower number in aged brain. J. Neurosci. 29, 4408–4419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Villeda S. A., Luo J., Mosher K. I., Zou B., Britschgi M., Bieri G., Stan T. M., Fainberg N., Ding Z., Eggel A., Lucin K. M., Czirr E., Park J. S., Couillard-Després S., Aigner L., et al. (2011) The ageing systemic milieu negatively regulates neurogenesis and cognitive function. Nature 477, 90–94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Jenkins H. G., and Bachelard H. S. (1988) Developmental and age-related changes in rat brain glycosaminoglycans. J. Neurochem. 51, 1634–1640 [DOI] [PubMed] [Google Scholar]
- 14. Cargill R., Kohama S. G., Struve J., Su W., Banine F., Witkowski E., Back S. A., and Sherman L. S. (2012) Astrocytes in aged nonhuman primate brain gray matter synthesize excess hyaluronan. Neurobiol. Aging 33, 830.e13–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Suzuki K., Katzman R., and Korey S. R. (1965) Chemical studies on Alzheimer's disease. J. Neuropathol. Exp. Neurol. 24, 211–224 [DOI] [PubMed] [Google Scholar]
- 16. Jenkins H. G., and Bachelard H. S. (1988) Glycosaminoglycans in cortical autopsy samples from Alzheimer brain. J. Neurochem. 51, 1641–1645 [DOI] [PubMed] [Google Scholar]
- 17. Back S. A., Kroenke C. D., Sherman L. S., Lawrence G., Gong X., Taber E. N., Sonnen J. A., Larson E. B., and Montine T. J. (2011) White matter lesions defined by diffusion tensor imaging in older adults. Ann. Neurol. 70, 465–476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Toole B. P. (2001) Hyaluronan in morphogenesis. Semin. Cell Dev. Biol. 12, 79–87 [DOI] [PubMed] [Google Scholar]
- 19. Struve J., Maher P. C., Li Y. Q., Kinney S., Fehlings M. G., Kuntz C. 4th, and Sherman L. S. (2005) Disruption of the hyaluronan-based extracellular matrix in spinal cord promotes astrocyte proliferation. Glia 52, 16–24 [DOI] [PubMed] [Google Scholar]
- 20. Back S. A., Tuohy T. M., Chen H., Wallingford N., Craig A., Struve J., Luo N. L., Banine F., Liu Y., Chang A., Trapp B. D., Bebo B. F. Jr., Rao M. S., and Sherman L. S. (2005) Hyaluronan accumulates in demyelinated lesions and inhibits oligodendrocyte progenitor maturation. Nat. Med. 11, 966–972 [DOI] [PubMed] [Google Scholar]
- 21. Lin C. M., Lin J. W., Chen Y. C., Shen H. H., Wei L., Yeh Y. S., Chiang Y. H., Shih R., Chiu P. L., Hung K. S., Yang L. Y., and Chiu W. T. (2009) Hyaluronic acid inhibits the glial scar formation after brain damage with tissue loss in rats. Surg. Neurol. 72, Suppl. 2, 50–54 [DOI] [PubMed] [Google Scholar]
- 22. Fuxe K., Tinner B., Chadi G., Härfstrand A., and Agnati L. F. (1994) Evidence for a regional distribution of hyaluronic acid in the rat brain using a highly specific hyaluronic acid recognizing protein. Neurosci. Lett. 169, 25–30 [DOI] [PubMed] [Google Scholar]
- 23. Schwartz P. H., Bryant P. J., Fuja T. J., Su H., O'Dowd D. K., and Klassen H. (2003) Isolation and characterization of neural progenitor cells from post-mortem human cortex. J. Neurosci. Res. 74, 838–851 [DOI] [PubMed] [Google Scholar]
- 24. Oishi K., and Ito-Dufros Y. (2006) Angiogenic potential of CD44+ CD90+ multipotent CNS stem cells in vitro. Biochem. Biophys. Res. Commun. 349, 1065–1072 [DOI] [PubMed] [Google Scholar]
- 25. Pollard S. M., Wallbank R., Tomlinson S., Grotewold L., and Smith A. (2008) Fibroblast growth factor induces a neural stem cell phenotype in foetal forebrain progenitors and during embryonic stem cell differentiation. Mol. Cell. Neurosci. 38, 393–403 [DOI] [PubMed] [Google Scholar]
- 26. Naruse M., Shibasaki K., Yokoyama S., Kurachi M., and Ishizaki Y. (2013) Dynamic changes of CD44 expression from progenitors to subpopulations of astrocytes and neurons in developing cerebellum. PLoS One 8, e53109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kochlamazashvili G., Henneberger C., Bukalo O., Dvoretskova E., Senkov O., Lievens P. M., Westenbroek R., Engel A. K., Catterall W. A., Rusakov D. A., Schachner M., and Dityatev A. (2010) The extracellular matrix molecule hyaluronic acid regulates hippocampal synaptic plasticity by modulating postsynaptic L-type Ca2+ channels. Neuron 67, 116–128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Pan Y. W., Storm D. R., and Xia Z. (2013) Role of adult neurogenesis in hippocampus-dependent memory, contextual fear extinction and remote contextual memory: new insights from ERK5 MAP kinase. Neurobiol. Learn. Mem. 105, 81–92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Raber J., Olsen R. H., Su W., Foster S., Xing R., Acevedo S. F., and Sherman L. S. (2014) CD44 is required for spatial memory retention and sensorimotor functions. Behav. Brain Res. 275, 146–149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Preston M., Gong X., Su W., Matsumoto S. G., Banine F., Winkler C., Foster S., Xing R., Struve J., Dean J., Baggenstoss B., Weigel P. H., Montine T. J., Back S. A., and Sherman L. S. (2013) Digestion products of the PH20 hyaluronidase inhibit remyelination. Ann. Neurol. 73, 266–280 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Su W., Gutmann D. H., Perry A., Abounader R., Laterra J., and Sherman L. S. (2004) CD44-independent hepatocyte growth factor/c-Met autocrine loop promotes malignant peripheral nerve sheath tumor cell invasion in vitro. Glia 45, 297–306 [DOI] [PubMed] [Google Scholar]
- 32. Salomoni P., and Calegari F. (2010) Cell cycle control of mammalian neural stem cells: putting a speed limit on G1. Trends Cell Biol. 20, 233–243 [DOI] [PubMed] [Google Scholar]
- 33. Guzowski J. F., Timlin J. A., Roysam B., McNaughton B. L., Worley P. F., and Barnes C. A. (2005) Mapping behaviorally relevant neural circuits with immediate-early gene expression. Curr. Opin. Neurobiol. 15, 599–606 [DOI] [PubMed] [Google Scholar]
- 34. Kee N., Teixeira C. M., Wang A. H., and Frankland P. W. (2007) Preferential incorporation of adult-generated granule cells into spatial memory networks in the dentate gyrus. Nat. Neurosci. 10, 355–362 [DOI] [PubMed] [Google Scholar]
- 35. Sirko S., von Holst A., Wizenmann A., Götz M., and Faissner A. (2007) Chondroitin sulfate glycosaminoglycans control proliferation, radial glia cell differentiation and neurogenesis in neural stem/progenitor cells. Development 134, 2727–2738 [DOI] [PubMed] [Google Scholar]
- 36. Kazanis I., and ffrench-Constant C. (2011) Extracellular matrix and the neural stem cell niche. Dev. Neurobiol. 71, 1006–1017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Steindler D. A. (2012) Neurogenic astrocytes and their glycoconjugates: not just “glue” anymore. Methods Mol. Biol. 814, 9–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Preston M., and Sherman L. S. (2011) Neural stem cell niches: roles for the hyaluronan-based extracellular matrix. Front. Biosci. 3, 1165–1179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Sloane J. A., Batt C., Ma Y., Harris Z. M., Trapp B., and Vartanian T. (2010) Hyaluronan blocks oligodendrocyte progenitor maturation and remyelination through TLR2. Proc. Natl. Acad. Sci. U.S.A. 107, 11555–11560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. van Praag H., Kempermann G., and Gage F. H. (1999) Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat. Neurosci. 2, 266–270 [DOI] [PubMed] [Google Scholar]
- 41. Frischknecht R., Heine M., Perrais D., Seidenbecher C. I., Choquet D., and Gundelfinger E. D. (2009) Brain extracellular matrix affects AMPA receptor lateral mobility and short-term synaptic plasticity. Nat. Neurosci. 12, 897–904 [DOI] [PubMed] [Google Scholar]
- 42. Bauer E. P., Schafe G. E., and LeDoux J. E. (2002) NMDA receptors and L-type voltage-gated calcium channels contribute to long-term potentiation and different components of fear memory formation in the lateral amygdala. J. Neurosci. 22, 5239–5249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Suzuki A., Josselyn S. A., Frankland P. W., Masushige S., Silva A. J., and Kida S. (2004) Memory reconsolidation and extinction have distinct temporal and biochemical signatures. J. Neurosci. 24, 4787–4795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Pan Y. W., Storm D. R., and Xia Z. (2012) The maintenance of established remote contextual fear memory requires ERK5 MAP kinase and ongoing adult neurogenesis in the hippocampus. PLoS One 7, e50455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Fitzsimons C. P., van Hooijdonk L. W., Schouten M., Zalachoras I., Brinks V., Zheng T., Schouten T. G., Saaltink D. J., Dijkmans T., Steindler D. A., Verhaagen J., Verbeek F. J., Lucassen P. J., de Kloet E. R., Meijer O. C., et al. (2013) Knockdown of the glucocorticoid receptor alters functional integration of newborn neurons in the adult hippocampus and impairs fear-motivated behavior. Mol. Psychiatry 18, 993–1005 [DOI] [PubMed] [Google Scholar]
- 46. Kheirbek M. A., Tannenholz L., and Hen R. (2012) NR2B-dependent plasticity of adult-born granule cells is necessary for context discrimination. J. Neurosci. 32, 8696–8702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Jessberger S., Clark R. E., Broadbent N. J., Clemenson G. D. Jr., Consiglio A., Lie D. C., Squire L. R., and Gage F. H. (2009) Dentate gyrus-specific knockdown of adult neurogenesis impairs spatial and object recognition memory in adult rats. Learn. Mem. 16, 147–154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Roszkowska M., Skupien A., Wójtowicz T., Konopka A., Gorlewicz A., Kisiel M., Bekisz M., Ruszczycki B., Dolezyczek H., Rejmak E., Knapska E., Mozrzymas J. W., Wlodarczyk J., Wilczynski G. M., and Dzwonek J. (2016) CD44—a novel synaptic cell adhesion molecule regulating structural and functional plasticity of dendritic spines. Mol. Biol. Cell. 27, 4055–4066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Barzilay R., Ventorp F., Segal-Gavish H., Aharony I., Bieber A., Dar S., Vescan M., Globus R., Weizman A., Naor D., Lipton J., Janelidze S., Brundin L., and Offen D. (2016) CD44 deficiency is associated with increased susceptibility to stress-induced anxiety-like behavior in mice. J. Mol. Neurosci. 60, 548–558 [DOI] [PubMed] [Google Scholar]
- 50. Arranz A. M., Perkins K. L., Irie F., Lewis D. P., Hrabe J., Xiao F., Itano N., Kimata K., Hrabetova S., and Yamaguchi Y. (2014) Hyaluronan deficiency due to Has3 knock-out causes altered neuronal activity and seizures via reduction in brain extracellular space. J. Neurosci. 34, 6164–6176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Okun E., Griffioen K. J., Son T. G., Lee J. H., Roberts N. J., Mughal M. R., Hutchison E., Cheng A., Arumugam T. V., Lathia J. D., van Praag H., and Mattson M. P. (2010) TLR2 activation inhibits embryonic neural progenitor cell proliferation. J. Neurochem. 114, 462–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Bengzon J., Kokaia Z., Elmér E., Nanobashvili A., Kokaia M., and Lindvall O. (1997) Apoptosis and proliferation of dentate gyrus neurons after single and intermittent limbic seizures. Proc. Natl. Acad. Sci. U.S.A. 94, 10432–10437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Parent J. M., Yu T. W., Leibowitz R. T., Geschwind D. H., Sloviter R. S., and Lowenstein D. H. (1997) Dentate granule cell neurogenesis is increased by seizures and contributes to aberrant network reorganization in the adult rat hippocampus. J. Neurosci. 17, 3727–3738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Parent J. M. (2007) Adult neurogenesis in the intact and epileptic dentate gyrus. Prog. Brain Res. 163, 529–540 [DOI] [PubMed] [Google Scholar]
- 55. Gray W. P., and Sundstrom L. E. (1998) Kainic acid increases the proliferation of granule cell progenitors in the dentate gyrus of the adult rat. Brain Res. 790, 52–59 [DOI] [PubMed] [Google Scholar]
- 56. Laurén H. B., Ruohonen S., Kukko-Lukjanov T. K., Virta J. E., Grönman M., Lopez-Picon F. R., Järvelä J. T., and Holopainen I. E. (2013) Status epilepticus alters neurogenesis and decreases the number of GABAergic neurons in the septal dentate gyrus of 9-day-old rats at the early phase of epileptogenesis. Brain Res. 1516, 33–44 [DOI] [PubMed] [Google Scholar]
- 57. Jiruska P., Shtaya A. B., Bodansky D. M., Chang W. C., Gray W. P., and Jefferys J. G. (2013) Dentate gyrus progenitor cell proliferation after the onset of spontaneous seizures in the tetanus toxin model of temporal lobe epilepsy. Neurobiol. Dis. 54, 492–498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Rotheneichner P., Marschallinger J., Couillard-Despres S., and Aigner L. (2013) Neurogenesis and neuronal regeneration in status epilepticus. Epilepsia 54, Suppl. 6, 40–42 [DOI] [PubMed] [Google Scholar]
- 59. McRae P. A., Baranov E., Rogers S. L., and Porter B. E. (2012) Persistent decrease in multiple components of the perineuronal net following status epilepticus. Eur. J. Neurosci. 36, 3471–3482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Bausch S. B. (2006) Potential roles for hyaluronan and CD44 in kainic acid-induced mossy fiber sprouting in organotypic hippocampal slice cultures. Neuroscience 143, 339–350 [DOI] [PubMed] [Google Scholar]
- 61. Hester M. S., and Danzer S. C. (2013) Accumulation of abnormal adult-generated hippocampal granule cells predicts seizure frequency and severity. J. Neurosci. 33, 8926–8936 [DOI] [PMC free article] [PubMed] [Google Scholar]