Abstract
Study aims This study aimed to investigate the clinical usefulness of magnifying endoscopy (ME) for non-ampullary duodenal tumors.
Patients and methods We enrolled 103 consecutive patients with non-ampullary duodenal tumors that were observed by ME with narrow-band imaging (ME-NBI) and had pit pattern analysis before endoscopic resection at Hiroshima University Hospital before December 2014. ME-NBI images were classified as Type B or Type C according to the Hiroshima classification, and pit patterns were classified as regular or irregular. We studied the clinicopathological features and diagnoses with ME-NBI and pit pattern analyses according to the Vienna classification (category 3: 73 patients; category 4: 30 patients).
Results Category 4 lesions were significantly larger than category 3 lesions. According to ME-NBI images, category 4 Type C lesions (83 %) were significantly more common than category 4 Type B lesions (17 %). According to pit pattern analyses, category 4 irregular lesions 4 (77 %) were significantly more common than category 4 regular lesions (23 %). The accuracies of using Type C ME-NBI images and irregular pit patterns to diagnose category 4 lesions were 87 % and 84 %, the sensitivities were 83 % and 77 %, and the specificities were 89 % and 88 %, respectively. There was no significant difference between ME-NBI and pit pattern analyses for diagnosing the histologic grade of non-ampullary duodenal tumors.
Conclusion Our study showed that ME-NBI and pit pattern analysis had equivalent abilities to determine the histologic grade of non-ampullary duodenal tumors. ME-NBI may be more useful because it is a simple, less time-consuming procedure.
Introduction
A recent increase in the number of case reports detailing primary non-ampullary duodenal tumors has coincided with advances in the field of endoscopic examination. However, few reports have assessed preoperative diagnosis using magnifying endoscopy with narrow-band imaging (ME-NBI) and pit pattern analysis for non-ampullary duodenal tumors 1 2 3 4. Adequate knowledge of endoscopic findings for non-ampullary duodenal tumors is required for early detection and treatment.
ME-NBI provides enhanced images of surface and vessel patterns. Several reports have stated the usefulness of ME-NBI for diagnosis of small superficial esophageal cancers 5, Barrett’s esophagus 6, early oropharyngeal and hypopharyngeal carcinomas 7, early gastric cancers 8, and colorectal cancers 9 10. Pit pattern classification for colorectal lesions 11 describes the histologic characteristics of the lesion 12 13 14 15. Magnifying endoscopy (ME) is used for differential diagnosis between non-neoplastic and neoplastic lesions 16 17 18 and for assessing the depth of invasion of colorectal cancer 19 20 21 22.
Meanwhile, the endoscopic features of non-ampullary duodenal tumors and adenocarcinomas have rarely been assessed using image-enhanced endoscopy. The establishment of endoscopic diagnoses with ME-NBI and pit pattern for non-ampullary duodenal tumors is necessary. This study aimed to investigate the clinical usefulness of ME-NBI and pit pattern (via Crystal Violet) to assess non-ampullary duodenal tumors. In addition, the study focused on the clinical usefulness of magnifying endoscopy in making a differential diagnosis between category 3 and category 4 tumors according to the Vienna classification.
Methods
We enrolled 103 consecutive patients with non-ampullary duodenal tumors who were observed by ME-NBI and had pit pattern analysis before endoscopic resection (ER) at Hiroshima University Hospital before December 2014, excluding cases of familial adenomatous polyposis or ampullary duodenal tumors. Of the 103 lesions, 73 lesions were category 3, and 30 lesions were category 4, according to the Vienna classification 23 24. The indication for ER for superficial non-ampullary duodenal epithelial tumors was an endoscopically diagnosed adenoma or intramucosal adenocarcinoma (Vienna classification category 3 and 4). A tumor for which en bloc ER was regarded as technically impossible was an indication for surgical resection. Biopsies were taken from 16 tumors (16 %) before ER. We performed both ME-NBI and Crystal Violet staining for pit pattern analysis in all lesions. For the purpose of this study, data with regard to patient age and sex, lesion characteristics (location, size, color, macroscopic type, and presence of milk-white mucosa 4), ME-NBI features (Type B or Type C by the Hiroshima classification 9), and pit pattern features (regular or irregular) were collected, and we analyzed these data according to the Vienna classification (category 3 vs. category 4).
In non-neoplastic lesions, normal duodenal villi appear as a regular arc shape under white light imaging and ME-NBI. For ME-NBI images of non-ampullary duodenal tumors, we classified lesions as Type B or Type C, according to the Hiroshima classification 9, which is based on both surface and vessel patterns. Types B and C indicate neoplastic lesions. Type B was defined as fine microvessels observed around the pits with clear pits observed via the nest of microvessels. Type C was defined as irregular microvessels; heterogeneous vessel diameter or distribution; invisible pits via the microvessels; thick, irregular vessel diameter; and observation of avascular areas. Type B lesions have regular surface patterns and regular vessel patterns, which reflect tubular or villous structures ( Fig. 1a). Type C lesions have irregular surface patterns and irregular vessel patterns ( Fig. 1b). For pit pattern analyses, we classified lesions as having a regular pit pattern or an irregular pit pattern, according to pit pattern irregularity, as shown in Fig. 1c and Fig. 1d. Tanaka et al. 15 reported that Type VI was irregularly arranged and similar to Type IIIL, IIIS, or IV patterns in size. When a variety of types were present, the most common type of ME-NBI and pit pattern findings was used.
Fig. 1.
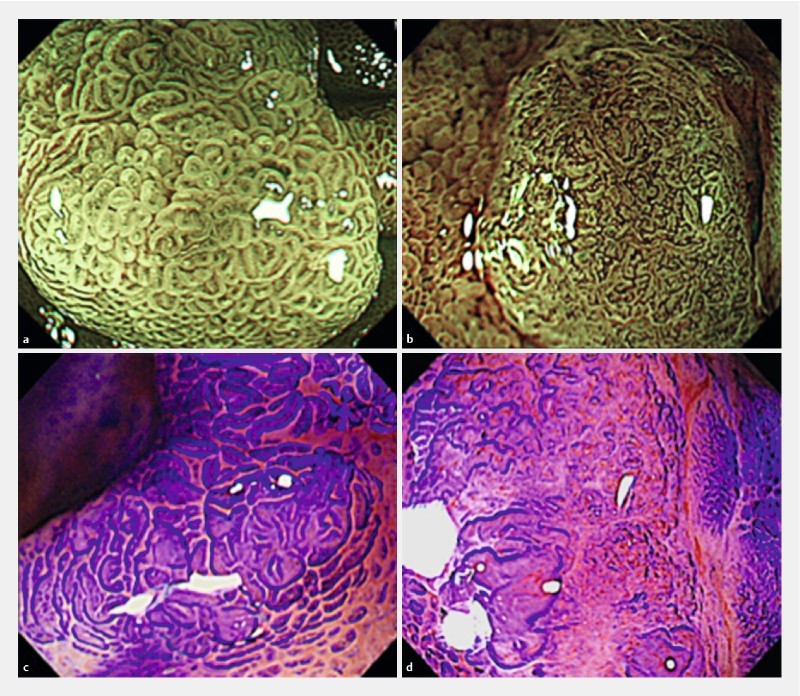
Magnifying endoscopic findings of non-ampullary duodenal tumors. a Magnifying endoscopy with narrow-band imaging (ME-NBI), Hiroshima Classification Type B. b ME-NBI, Hiroshima Classification Type C. c Regular pit pattern (Crystal Violet staining). d Irregular pit pattern (Crystal Violet staining).
The conventional and ME findings were diagnosed retrospectively. Specialists from the Japan Gastroenterological Endoscopy Society, with over 10 years of gastrointestinal endoscopic practice who had reviewed over 500 image-enhanced endoscopy cases, independently reviewed the recorded series of endoscopic images without access to any pathologic information. The pathological findings were diagnosed by endoscopic resection of the specimens. We did not perform biopsies of most of the lesions. We investigated the characteristics with ME for consecutive non-ampullary duodenal tumors resected by ER.
All patients who underwent ER for non-ampullary duodenal tumors before December 2014 were reviewed under an Institutional Review Board (IRB)-approved protocol.
Statistical analysis
Statistical differences were evaluated using Student’s t test, the Chi-squared test, and Fisher’s exact probability test. A value of P < 0.05 was considered statistically significant.
Results
A total of 103 non-ampullary duodenal tumors were included in the study, 73 of which were Vienna classification category 3 and 30 of which were category 4. Clinicopathological characteristics of the lesions are shown in Table 1. Category 4 tumors were significantly larger than category 3 tumors (13.0 mm and 10.0 mm, respectively). There were no significant differences in location (second portion incidence: 73 % and 80 % for categories 3 and 4, respectively) or macroscopic type (depressed type incidence: 16 % and 26 % for categories 3 and 4, respectively) based on histological grade.
Table 1. Clinicopathological characteristics of non-ampullary duodenal tumors.
| Clinicopathological features | Vienna classification | P value | |
| Category 3, n = 73 | Category 4, n = 30 | ||
| Age, mean ± SD, years | 62.0 ± 11.3 | 57.0 ± 13.6 | n.s. |
| Sex, n (%) | |||
|
44 (60) | 18 (60) | n.s. |
|
29 (40) | 12 (40) | |
| Location, n (%) | |||
|
20 (27) | 6 (20) | n.s. |
|
53 (73) | 24 (80) | |
| Tumor size, mean ± SD, mm | 10.0 ± 6.0 | 13.0 ± 5.8 | 0.02 |
|
49 (67) | 17 (57) | |
|
24 (33) | 13 (43) | |
| Macroscopic type, n (%) | |||
|
12 (16) | 8 (27) | n.s. |
|
50 (68) | 14 (47) | |
|
4 (6) | 3 (10) | |
|
7 (10) | 5 (16) | |
| Color, n (%) | |||
|
16 (22) | 12 (40) | n.s. |
|
57 (78) | 18 (60) | |
| Milk-white mucosa, n (%) | |||
|
47 (64) | 20 (67) | n.s. |
|
26 (36) | 10 (33) | |
The relationships between ME-NBI/pit pattern classifications and histopathological diagnoses according to the Vienna classification for non-ampullary duodenal tumors are shown in Table 2. For ME-NBI, there were significantly more category 4 Type C lesions (83 %) than category 4 Type B lesions (17 %). For pit pattern analysis, the irregular pattern (77 %) was significantly more common than the regular pattern (23 %) in category 4 lesions.
Table 2. Relationship between the classification determined by ME-NBI and pit pattern analyses and histopathological diagnosis by Vienna classification for non-ampullary duodenal tumors.
| Magnifying endoscopy | Vienna classification | P value | |
| Category 3, n = 73 | Category 4, n = 30 | ||
| ME-NBI, n (%) | |||
|
65 (89) | 5 (17) | < 0.01 |
|
8 (11) | 25 (83) | |
| Pit pattern, n (%) | |||
|
64 (88) | 7 (23) | < 0.01 |
|
9 (12) | 23 (77) | |
ME-NBI; magnifying endoscopy with narrow-band imaging.
There were no significant differences between ME-NBI and pit pattern analysis with respect to histological grade diagnoses (Table 3). The accuracy of using Type C ME-NBI images to diagnose category 4 lesions was 87 % (90/103), the sensitivity was 83 % (25/30), the specificity was 89 % (65/73), the positive predictive value (PPV) was 76 % (25/33), and the negative predictive value (NPV) was 93 % (65/70). The accuracy of using irregular pit pattern to diagnose category 4 lesions was 84 % (87/103), the sensitivity was 77 % (23/30), the specificity was 88 % (64/73), the PPV was 72 % (23/32), and the NPV was 90 % (64/71).
Table 3. Diagnostic measurements for category 4 lesions with ME-NBI and pit pattern analyses.
| Magnifying endoscopy | Accuracy | Sensitivity | Specificity | PPV | NPV |
| ME-NBI, % (n) | |||||
|
87 (90/103) | 83 (25/30) | 89 (65/73) | 76 (25/33) | 93 (65/70) |
| Pit pattern, % (n) | |||||
|
84 (87/103) | 77 (23/30) | 88 (64/73) | 72 (23/32) | 90 (64/71) |
ME-NBI, magnifying endoscopy with narrow-band imaging; PPV, positive predictive value; NPV, negative predictive value.
There was no significant difference in histological grade diagnoses based on use of ME-NBI or pit pattern analysis, regardless of microscopic type (Table 4). The respective accuracies, sensitivities, and specificities of using Type C images to diagnose category 4 lesions were 89 % (75/84), 82 % (18/22), and 92 % (57/62) for elevated lesions, and 79 % (15/19), 88 % (7/8), and 73 % (8/11) for depressed lesions. On the other hand, the respective accuracies, sensitivities, and specificities of using irregular pit patterns to diagnose category 4 lesions were 87 % (53/84), 82 % (18/22), and 89 % (55/62) in elevated lesions, and 74 % (14/19), 63 % (5/8), and 82 % (9/11) in depressed lesions.
Table 4. Diagnostic measurements for category 4 lesions with ME-NBI and pit pattern analyses according to microscopic type.
| Magnifying endoscopy | Accuracy | Sensitivity | Specificity | |||
| Elevated | Depressed | Elevated | Depressed | Elevated | Depressed | |
| ME-NBI, % (n) | ||||||
|
89 (75/84) | 79 (15/19) | 82 (18/22) | 88 (7/8) | 92 (57/62) | 73 (8/11) |
| Pit pattern, % (n) | ||||||
|
87 (53/84) | 74 (14/19) | 82 (18/22) | 63 (5/8) | 89 (55/62) | 82 (9/11) |
ME-NBI, magnifying endoscopy with narrow-band imaging.
Discussion
Our study showed that ME-NBI and pit pattern images had equivalent abilities to diagnose histological grade (category 3 and category 4) of non-ampullary duodenal tumors. There are currently no guidelines about the indications for ER for non-ampullary duodenal tumors. Making a differential diagnosis before ER is important, because the malignant potential is quite different between category 3 and category 4 tumors, and en bloc ER should be performed for category 4 tumors. This study had a low selection bias, because we investigated the characteristics with ME for consecutive non-ampullary duodenal tumors resected by ER. Kikuchi et al. 25 reported that patients with category 3 non-ampullary duodenal tumors were significantly younger and had relatively smaller tumor diameters than those with category 4/5 tumors. In our study, compared to category 4 tumors, category 3 tumors had significantly smaller tumor diameters, but no significant differences in patient age or other findings were observed.
Milk-white mucosa is defined as a glossy and opalescent appearance. This change in villi is the suspected result of lipid droplets in the absorptive epithelium that cause abnormal secretion of chylomicron. Yoshimura et al. 4 classified the distribution pattern for milk-white mucosa as entire or marginal for non-ampullary duodenal tumors. They reported that 92 % of lesions had a milk-white mucosa, which was generally marginal in category 4 lesions (86 %) and entire for ~60 % of category 3 lesions, indicating a significant intergroup difference. A white opaque substance (WOS) visualized on ME-NBI was first described in 2008 as a substance in the superficial area of gastric neoplasia that masked the subepithelial microvascular architecture. Yao and colleagues 26 27 reported that the gastric lesion WOS originated from lipid droplets that accumulate in the superficial part of epithelial neoplasms. The literature contains some descriptions with regard to correlations between histologic findings and the WOS in gastric neoplastic lesions. In colorectal lesions, Kawasaki et al. 28 reported that the incidence of high grade dysplasia or carcinoma was significantly higher in WOS-positive lesions (62 %) than in WOS-negative lesions (29 %). Milk-white mucosa was present in 65 % (67/103) of the tumors in our study (Table 1). However, there was no significant difference in the presence of milk-white mucosa between category 3 and category 4 lesions.
Yoshimura et al. 4 classified mucosal patterns in ME-NBI as heterogeneous or obscure. They reported that obscure mucosal patterns are observed at significantly higher rates in category 4 lesions. They also classified abnormal microvascular patterns as network microvascular patterns or intravillous-structure irregular microvascular patterns. The frequencies of the network microvascular pattern in the category 4 groups were significantly higher than those in the category 3 groups. Kikuchi et al. 25 reported that superficial non-ampullary duodenal epithelial tumors displayed either a single or multiple surface patterns, known as monotype and mixed type, respectively. They reported that 57 % of monotype lesions were category 4 lesions whereas all mixed-type lesions were category 4/5, indicating that mixed-type lesions had a significantly higher probability of being category 4/5 lesions than monotype lesions. Endo et al. 29 divided classified non-ampullary duodenal tumors by convoluted, leaf, reticular/sulciolar, or colon-like pattern. In all cases, there were areas of the tumor with generalized or localized whitening. Goda et al. 30 reported that the sensitivities, specificities, and accuracies of preoperative diagnosis for endoscopy and biopsy when diagnosing high grade dysplasia or superficial adenocarcinoma were 77 %/72 %/75 % and 58 %/93 %/68 %, respectively.
Our study showed that the rate of accuracy for ME-NBI Type C diagnosis of category 4 lesions was 87 %, the sensitivity was 83 %, and the specificity was 89 %. Our results were similar but higher than those previously reported. We observed both the surface and vessel patterns of non-ampullary duodenal tumors using ME-NBI, which may have contributed to our higher numbers. Usually, biopsy specimens are obtained using forceps for preoperative diagnosis of non-ampullary duodenal tumors, but it can be difficult to remove the tumors by ER after the biopsy because biopsy can cause severe fibrosis of the submucosal layer. Nonaka et al. 2 reported that biopsy accuracy was 71 %, and the PPVs of biopsy for adenocarcinoma/high grade neoplasia and adenoma/low grade neoplasia were 75 % and 70 %, respectively. These results indicated that endoscopic diagnosis might be more useful than biopsy for non-ampullary duodenal tumors. In addition, poor mucosal lifting by preoperative biopsy might result in a higher incidence of piecemeal resection and perforation as well as an increased risk of other complications, such as delayed bleeding and delayed perforation. Therefore, optical biopsy with ME-NBI and pit pattern analyses might be useful to determine the treatment strategy for patients with non-ampullary duodenal tumors.
This study was limited by its retrospective nature as well as the single-center location and small patient number. Further prospective and multicenter studies are needed to establish endoscopic diagnostic criteria for non-ampullary duodenal tumors.
In conclusion, our study suggests that both ME-NBI and pit pattern analyses have clinical usefulness for non-ampullary duodenal tumor diagnosis. ME-NBI and pit pattern analysis might provide a more reliable diagnosis of histological grade of non-ampullary duodenal tumors than biopsy. Overall, ME-NBI may be the more useful method because it is a simple, less time-consuming procedure.
Footnotes
Competing interests None
References
- 1.Oka S, Tanaka S, Nagata S et al. Clinicopathologic features and endoscopic resection of early primary nonampullary duodenal carcinoma. J Clin Gastroenterol. 2003;37:381–386. doi: 10.1097/00004836-200311000-00006. [DOI] [PubMed] [Google Scholar]
- 2.Nonaka S, Oda I, Tada K et al. Clinical outcome of endoscopic resection for nonampullary duodenal tumors. Endoscopy. 2014;47:129–135. doi: 10.1055/s-0034-1390774. [DOI] [PubMed] [Google Scholar]
- 3.Yamamoto Y, Yoshizawa N, Tomida H et al. Therapeutic outcome of endoscopic resection for superficial non-ampullary duodenal tumor. Dig Endosc. 2014;26:50–56. doi: 10.1111/den.12273. [DOI] [PubMed] [Google Scholar]
- 4.Yoshimura N, Goda K, Tajiri H et al. Endoscopic features of nonampullary duodenal tumors with NBI. Hepatogastroenterology. 2010;57:462–467. [PubMed] [Google Scholar]
- 5.Yoshida T, Inoue H, Usui S et al. Narrow-band imaging system with magnifying endoscopy for superficial esophageal lesions. Gastrointest Endosc. 2004;59:288–295. doi: 10.1016/s0016-5107(03)02532-x. [DOI] [PubMed] [Google Scholar]
- 6.Hamamoto Y, Endo T, Nosho K et al. Usefulness of NBI endoscopy for diagnosis of Barrett’s esophagus. J Gastroenterol. 2004;39:14–20. doi: 10.1007/s00535-003-1239-z. [DOI] [PubMed] [Google Scholar]
- 7.Muto M, Katada C, Sano Y et al. NBI: a new diagnostic approach to visualize angiogenesis in superficial neoplasia. Clin Gastroenterol Hepatol. 2005;3:16–20. doi: 10.1016/s1542-3565(05)00262-4. [DOI] [PubMed] [Google Scholar]
- 8.Sumiyama K, Kaise M, Nakayoshi T et al. Combined use of a magnifying endoscope with a narrow band imaging system and a multibending endoscope for en bloc EMR of early stage gastric cancer. Gastrointest Endosc. 2004;60:79–84. doi: 10.1016/s0016-5107(04)01285-4. [DOI] [PubMed] [Google Scholar]
- 9.Kanao H, Tanaka S, Oka S et al. Narrow-band imaging magnification predicts the histology and invasion depth of colorectal tumors. Gastrointest Endosc. 2009;69:631–636. doi: 10.1016/j.gie.2008.08.028. [DOI] [PubMed] [Google Scholar]
- 10.Hirata M, Tanaka S, Oka S et al. Magnifying endoscopy with narrow band imaging for diagnosis of colorectal tumors. Gastrointest Endosc. 2007;65:988–995. doi: 10.1016/j.gie.2006.07.046. [DOI] [PubMed] [Google Scholar]
- 11.Kudo S, Hirota S, Nakajima T et al. Colorectal tumours and pit pattern. J Clin Pathol. 1994;47:880–885. doi: 10.1136/jcp.47.10.880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nagata S, Tanaka S, Haruma K et al. Pit pattern diagnosis of early colorectal carcinoma by magnifying colonoscopy: clinical and histological implications. Int J Oncol. 2000;16:927–934. doi: 10.3892/ijo.16.5.927. [DOI] [PubMed] [Google Scholar]
- 13.Kudo S, Tamura S, Nakajima T et al. Diagnosis of colorectal tumorous lesions by magnifying endoscopy. Gastrointest Endosc. 1996;44:8–14. doi: 10.1016/s0016-5107(96)70222-5. [DOI] [PubMed] [Google Scholar]
- 14.Tanaka S, Haruma K, Ito M et al. Detailed colonoscopy for detecting early superficial carcinoma: recent developments. J Gastroenterol. 2000;35 12:121–125. [PubMed] [Google Scholar]
- 15.Tanaka S, Kaltenbach T, Chayama K et al. High- magnification colonoscopy (with videos) Gastrointest Endosc. 2006;64:604–613. doi: 10.1016/j.gie.2006.06.007. [DOI] [PubMed] [Google Scholar]
- 16.Togashi K, Konishi F, Ishizuka T et al. Efficacy of ME in the differential diagnosis of neoplastic and non-neoplastic polyps of the large bowel. Dis Colon Rectum. 1999;42:1602–1608. doi: 10.1007/BF02236215. [DOI] [PubMed] [Google Scholar]
- 17.Konishi K, Kaneko K, Kurahashi T et al. A comparison of magnifying and nonmagnifying colonoscopy for diagnosis of colorectal polyps: A prospective study. Gastrointest Endosc. 2003;57:48–53. doi: 10.1067/mge.2003.31. [DOI] [PubMed] [Google Scholar]
- 18.Hurlstone D P, Cross S S, Adam I et al. Efficacy of high magnification chromoscopic colonoscopy for the diagnosis of neoplasia in flat and depressed lesions of the colorectum: a prospective analysis. Gut. 2004;53:284–290. doi: 10.1136/gut.2003.027623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nagata S, Tanaka S, Haruma K et al. Pit pattern diagnosis of early colorectal carcinoma by magnifying colonoscopy: clinical and histological implications. Int J Oncol. 2000;16:927–934. doi: 10.3892/ijo.16.5.927. [DOI] [PubMed] [Google Scholar]
- 20.Tanaka S, Haruma K, Oh-E H et al. Conditions of curability after endoscopic resection for colorectal carcinoma with submucosally massive invasion. Oncol Rep. 2000;7:783–788. doi: 10.3892/or.7.4.783. [DOI] [PubMed] [Google Scholar]
- 21.Tanaka S, Haruma K, Nagata S et al. Diagnosis of invasion depth in early colorectal carcinoma by pit pattern analysis with magnifying endoscopy. Dig Endosc. 2001;13:2–5. [Google Scholar]
- 22.Tanaka S, Nagata S, Oka S et al. Determining depth of invasion by VN pit pattern analysis in submucosal colorectal carcinoma. Oncol Rep. 2002;9:1005–1008. [PubMed] [Google Scholar]
- 23.Dixon M F. Gastrointestinal epithelial neoplasia: Vienna revisited. Gut. 2002;51:130–131. doi: 10.1136/gut.51.1.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stolte M. The new Vienna classification of epithelial neoplasia of the gastrointestinal tract: advantages and disadvantages. Virchows Arch. 2003;442:99–106. doi: 10.1007/s00428-002-0680-3. [DOI] [PubMed] [Google Scholar]
- 25.Kikuchi D, Hoteya S, Iizuka T et al. Diagnostic algorithm of magnifying endoscopy with NBI for superficial non-ampullary duodenal epithelial tumors. Dig Endosc. 2014;26:16–22. doi: 10.1111/den.12282. [DOI] [PubMed] [Google Scholar]
- 26.Yao K, Iwashita A, Tanabe H et al. White opaque substance within superficial elevated gastric neoplasia as visualized by magnification endoscopy with NBI: a new optical sign for differentiating between adenoma and carcinoma. Gastrointest Endosc. 2008;68:574–580. doi: 10.1016/j.gie.2008.04.011. [DOI] [PubMed] [Google Scholar]
- 27.Yao K, Iwashita A, Nambu M et al. Nature of white opaque substance in gastric epithelial neoplasia as visualized by ME-NBI. Dig Endosc. 2012;24:419–425. doi: 10.1111/j.1443-1661.2012.01314.x. [DOI] [PubMed] [Google Scholar]
- 28.Kawasaki K, Kurahara K, Yanai S et al. Significance of a white opaque substance under magnifying NBI colonoscopy for the diagnosis of colorectal epithelial neoplasms. Gastrointest Endosc. 2015;82:1097–1104. doi: 10.1016/j.gie.2015.06.023. [DOI] [PubMed] [Google Scholar]
- 29.Endo M, Abiko Y, Oana S et al. Usefulness of endoscopic treatment for duodenal adenoma. Dig Endosc. 2010;22:360–365. doi: 10.1111/j.1443-1661.2010.01014.x. [DOI] [PubMed] [Google Scholar]
- 30.Goda K, Kikuchi D, Yamamoto Y et al. Endoscopic diagnosis of superficial non-ampullary duodenal epithelial tumors in Japan: Multicenter case series. Dig Endosc. 2014;26:23–29. doi: 10.1111/den.12277. [DOI] [PubMed] [Google Scholar]


