Abstract
Selective serotonin reuptake inhibitors (SSRIs) represent the most common treatment for major depression. However, their efficacy is variable and incomplete. In order to elucidate the cause of such incomplete efficacy, we explored the hypothesis positing that SSRIs may not affect mood per se but, by enhancing neural plasticity, render the individual more susceptible to the influence of the environment. Consequently, SSRI administration in a favorable environment promotes a reduction of symptoms, whereas in a stressful environment leads to a worse prognosis. To test such hypothesis, we exposed C57BL/6 mice to chronic stress in order to induce a depression-like phenotype and, subsequently, to fluoxetine treatment (21 days), while being exposed to either an enriched or a stressful condition. We measured the most commonly investigated molecular, cellular and behavioral endophenotypes of depression and SSRI outcome, including depression-like behavior, neurogenesis, brain-derived neurotrophic factor levels, hypothalamic–pituitary–adrenal axis activity and long-term potentiation. Results showed that, in line with our hypothesis, the endophenotypes investigated were affected by the treatment according to the quality of the living environment. In particular, mice treated with fluoxetine in an enriched condition overall improved their depression-like phenotype compared with controls, whereas those treated in a stressful condition showed a distinct worsening. Our findings suggest that the effects of SSRI on the depression- like phenotype is not determined by the drug per se but is induced by the drug and driven by the environment. These findings may be helpful to explain variable effects of SSRI found in clinical practice and to device strategies aimed at enhancing their efficacy by means of controlling environmental conditions.
Introduction
Major depression constitutes an enormous medical, individual, societal and economical challenge. Depression afflicts up to 10%–15% of the population worldwide. It is the leading cause of years lost, owing to disability1 and costs over 120 billion euros in Europe and over US$83 billion in North America annually.2
Selective serotonin reuptake inhibitors (SSRIs) are the most commonly prescribed drugs for the treatment of depression. However, their efficacy is variable and incomplete: 60%–70% of patients do not experience remission and 30%–40% do not show a significant response.3 Some authors have even claimed that their effects do not differ from placebo.4 Nevertheless, most psychiatrists prescribe SSRIs.
One of the main reasons for the discordant results and views about SSRI efficacy is the poor comprehension of their action at molecular and cellular level. Studies performed on animal models of depression provide contradictory results concerning the physiological and behavioral modifications induced by SSRIs. For instance, many studies show that SSRI administration reduces depression-like behavior,5, 6 enhances neurogenesis,7 increases brain-derived neurotrophic factor (BDNF) levels,5, 8 reduces hypothalamic–pituitary–adrenal (HPA) axis activity9, 10 and heightens long-term potentiation (LTP).11 However, many others reported no or opposite effects concerning the same endpoints: behavior,12, 13, 14, 15 neurogenesis,16, 17, 18, 19, 20 BDNF levels,21, 22, 23, 24, 25, 26 HPA axis activity27, 28 and LTP.29, 30
A new hypothesis, named the undirected susceptibility to change hypothesis, posits that SSRI treatment does not drive changes in mood per se but, by increasing brain plasticity, creates a window of opportunity for a change that is driven by the quality of the environment.31 In particular, the increase in serotonin levels, induced by SSRIs, enhances neural plasticity and thus renders the individual more susceptible to the environment. The main consequence of such hypothesis is the lack of univocal outcome of SSRI administration: in a favorable environment, treatment leads to a reduction of symptoms; by contrast, in a stressful environment, it leads to a worse prognosis.
In support to the undirected susceptibility to change hypothesis, a number of evidences from both clinical and preclinical studies indicate that increased serotonin levels lead to greater brain plasticity and higher susceptibility to the environment.31, 32, 33, 34 For instance, clinical studies investigating variations of the serotonin-transporter-linked polymorphic region, 5-HTTLPR, found that individuals bearing the s/s variant, which is associated to higher brain extracellular levels of serotonin, show an enhanced susceptibility to the quality of the living environment compared with individuals bearing the l/l variant.32, 35, 36 In addition, SSRI treatment consequences on selected endpoints, such as vulnerability to obesity, have been shown to be dependent on the quality of the environment.37, 38
Here we investigated the undirected susceptibility to change hypothesis assessing the modifications in molecular and cellular processes previously shown to be affected by SSRI administration including BDNF expression, neurogenesis, LTP and behavior. In particular, we focused on the hippocampus that is a brain region deeply involved in neural plasticity, major depression and antidepressant effect. To this purpose, we exposed C57BL/6 mice first to 14 days of stress, in order to induce a depression-like phenotype, and subsequently to 21 days of either (i) an enriched condition or (ii) a stressful condition, while being administered with fluoxetine (FLX) or vehicle (VEH). Our prediction was that the trajectories of molecular, cellular and behavioral modifications induced by FLX treatment depend on the living environment. In particular, FLX mice were expected to be more sensitive to the quality of the environment than VEH mice, displaying a better recovery from a depression-like profile when exposed to an enriched condition and a faster worsening when exposed to a stressful condition.
Materials and methods
All experiments were conducted in conformity with European Directive 2010/63/EU and the Italian D.lg. 4.05.2014, n. 26. C57BL/6 male mice were housed in the Intellicage system (TSE System, NewBehavior, Zürich, Switzerland) in which they were exposed to either enriched or stressful condition.
Treatment
FLX (Fluoxetine HCl, Santa Cruz, CA, USA) was dissolved in water and saccharin solution, and delivered via drinking water for 3 weeks.
Behavioral tests
Behavioral endpoints investigated included liking- and wanting-type anhedonia, cognitive bias activity. The experimental procedures used in the present study to phenotype behavior have been selected, as they are automatically administered by the Intellicage. This allowed avoiding any bias or stress due to the experimenter.
Immunohistochemistry and volume measurements
Animals were killed, perfused transcardially and hippocampi removed and fixed. Ki67 and doublecortin-positive cells were estimated. Volume was measured in series adjacent to immuno-stained sections.
RNA extraction, RT-PCR and real-time PCR
Total RNA was obtained from the hippocampi. After RNA reverse transcription, real-time PCR was performed. Sample were normalized to the housekeeping gene GAPDH.39
Protein extraction and western blotting
Western blotting analyses were performed on total, cytoplasmic or nuclear-enriched fractions obtained from the hippocampi and medial prefrontal cortices. Data were normalized to β-tubulin.
Electrophysiological analysis
Electrophysiological measurements were performed on 350 μm transverse hippocampal slices. N/n refers to the number of slices on total number of mice analyzed.
Statistical analysis
Two different experiments aimed at comparing VEH versus FLX-treated mice, independently in the enriched and in the stressful condition, were performed. One-way analysis of variance, regression analysis, Student's t-test or, when data were non-normally distributed, non-parametric analyses were used.
For details, see Supplementary Materials.
Results
Opposite effects of FLX treatment on depression-like behavior in enriched and stressful conditions
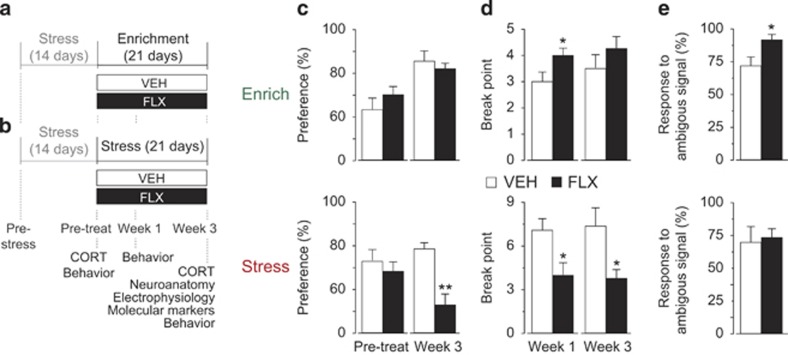
Many studies showed that SSRI administration affects depression-like behavior.7, 40 However, most of these did not consider the quality of the environment in which the experimental subjects live. To assess the relevance of the living environment in determining treatment outcome, we performed two independent experiments to investigate the effects of FLX administered in an enriched and a stressful environment. First, all mice were exposed to two weeks of chronic stress, which significantly induced anhedonic behavior (F(1,12)=17.897, P=0.0012) and increased corticosterone levels (U=5000, n1=5, n2=8, P=0.0281; Supplementary Figure S1). Afterwards, mice were treated for 3 weeks while housed in either the enriched or the stress condition. In each condition, we compared VEH vs FLX-treated mice. Our results show that FLX treatment has opposite outcomes in the two environments. In the enriched condition, it improved depression-like behavior. In particular, although no difference in liking-type anhedonia (saccharin preference) was found (Figure 1c), FLX mice showed a significant reduced wanting-type anhedonia after 1 week (F(1,14)=4.000, P=0.0486) and a trend toward reduction after 3 weeks of treatment (progressive ratio; Figure 1d) compared with VEH mice. In addition, FLX mice showed a higher optimistic bias, responding significantly more often to the ambiguous stimulus (F(1,9)=5.615, P=0.0419; Figure 1e). By contrast, in the stressful condition, FLX administration worsened depression-like phenotype. FLX mice showed a significant reduction in liking-type anhedonia at the end of the treatment period (F(1,45)=17.528, P=0.0001; Figures 1c), a significant increase in wanting-type anhedonia after both 1 and 3 weeks (respectively, Fs(1,19)=7.069, 7.094, ps=0.0155, 0.0154; Figure 1d) and no difference in cognitive bias (Figure 1e). The latter result is not in line with previous data, indicating that mice treated with SSRI show an increased preference for the sweet solution.5, 6 Nonetheless, a number of other authors found increased anhedonia following SSRI administration.12, 13, 14, 15 It is worth noting that this inconsistency could be due to the fact that several studies used sucrose instead of saccharin to assess anhedonia. This is a major limitation, because SSRIs affect metabolism leading to an increased caloric intake and thus to an increased preference for sucrose that is independent from the anhedonic state. No difference in activity was found between VEH and FLX mice in both the enriched and stressful conditions (Supplementary Figure S2). In addition, the amount of liquid (water+saccharin solution; average: 9.5 ml per day per mouse) drank by the VEH and FLX mice did not differ in both the enriched and the stressful condition. The average amount of FLX administered to each FLX mouse was 0.76 mg per day.
Figure 1.
Experimental design and effects of fluoxetine (FLX) administered either in enriched or stressful condition. (a) FLX treatment in the enriched condition. (b) FLX treatment in the stressful condition. In both cases, before treatment, mice are exposed to a 14-day period of stress to induce a depression-like phenotype. (c) Liking-type anhedonia (saccharin preference). Although no difference was found between FLX and vehicle (VEH) mice exposed to an enriched condition, a significantly increase of anhedonia (that is, reduction of saccharin preference) was found in FLX mice at the end of treatment in the stressful condition. VEH, n=23; FLX, n=24. (d) Wanting-type anhedonia (progressive ratio). When treatment was administered in the enriched condition, FLX led to a reduction of anhedonia (that is, a higher break point) at week 1, whereas in the stressful condition mice showed an increase in anhedonia (that is, lower break point) both at week 1 and 3, compared with VEH mice. Enrich: n=8 in all groups; stress: VEH, n=10, FLX, n=11. (e) Cognitive bias. In an enriched condition, FLX mice displayed a significantly ‘more optimistic' attitude than VEH by responding significantly more often to the ambiguous stimulus. No difference was found in the stressful condition. *P<0.05 vs relative VEH group. VEH, n=6, FLX, n=5. Data are means±s.e.m.
FLX treatment in a stress condition leads to reduced proliferation and decreased CA1 volume
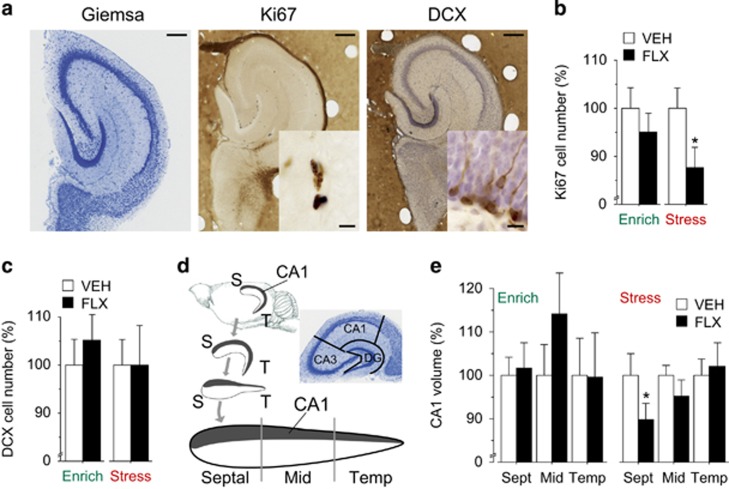
Impaired hippocampal neurogenesis has been linked to depression.41 In animal models, a reduction of hippocampal neurogenesis has been shown to be induced by stress and rescued by SSRI treatment.7, 42 However, recent data suggest that FLX may not be effective in enhancing neurogenesis16, 17 or even reduce it.16, 17, 18, 19, 43, 44 Similar contradictory results have been found for the effects of SSRI on hippocampal volumetric reduction, which is considered a further feature of depression.45 Overall, we found that FLX has no effect on neurogenesis in the enriched condition, but leads to detrimental consequences on proliferation in the stressful condition (Figure 2a and b). In particular, although doublecortin cell number is not affected, the number of proliferating cells (that is, Ki67-positive cell number) was reduced across the entire hippocampus in FLX compared with VEH mice in the stressful condition (main effect of treatment, F(1,19)=4.472, P=0.0479). With regard to volumetric changes, we found that, although the interaction volume × longitudinal axis (septal, mid and temporal) missed to reach statistical significance (F(1,38)=2.106, P=0.1357), Tukey's post-hoc analysis revealed that FLX mice show a significant reduction of CA1 volume in the septal part compared with VEH mice in the stressful condition (P<0.05, Figure 2c and d).
Figure 2.
Fluoxetine (FLX) treatment administered in stressful condition leads to a reduction of proliferation and hippocampal volume. (a) Representative sections of histological (Giemsa) and immunohistochemical (Ki67 and doublecortin (DCX) counterstained with hematoxylin) stainings in the mid region of the matrix-embedded, straightened hippocampal dentate gyrus. Scale bar: 250 μm, insert scale bar: 10 μm. (b) Ki67 cell number was not significantly affected by FLX administered in enriched condition, whereas it was significantly decreased in FLX compared with vehicle (VEH) mice when treatment was administered in the stressful condition, *P<0.05 vs VEH group. (c) DCX cell number was not significantly affected by FLX administered in both conditions. (d) Schematic drawing of the straightened hippocampus. Gray part represents CA1. The analysis has been performed independently in the septal, mid and temporal part of the hippocampus, because it has been reported that the effects of SSRIs and the environment are region specific. Boundaries of hippocampal fields are illustrated in the Giemsa-stained section of the mid region of the straightened hippocampus cut perpendicular to the septotemporal axis. S, septal; T, temporal. (e) Analysis of anatomically aligned data of volumetric measurements showed no significant differences between groups in enriched condition. However, septal CA1 volume was significantly reduced in FLX compared with VEH when treatment was administered in stressful condition. *P<0.05. n=8 in all groups. Data shown as mean±s.e.m.
FLX effects on antidepressant-related pathways and HPA axis activity depend on the quality of the environment
Enhancement of signaling pathways involving mitogen-activated protein kinase,46, 47 cAMP response element-binding protein (CREB)48, 49 and BDNF50 is thought to be a critical feature of antidepressant action. Our results show that FLX treatment overall potentiates these pathways in the enriched condition, while leading even to an opposite effect in the stressful condition.
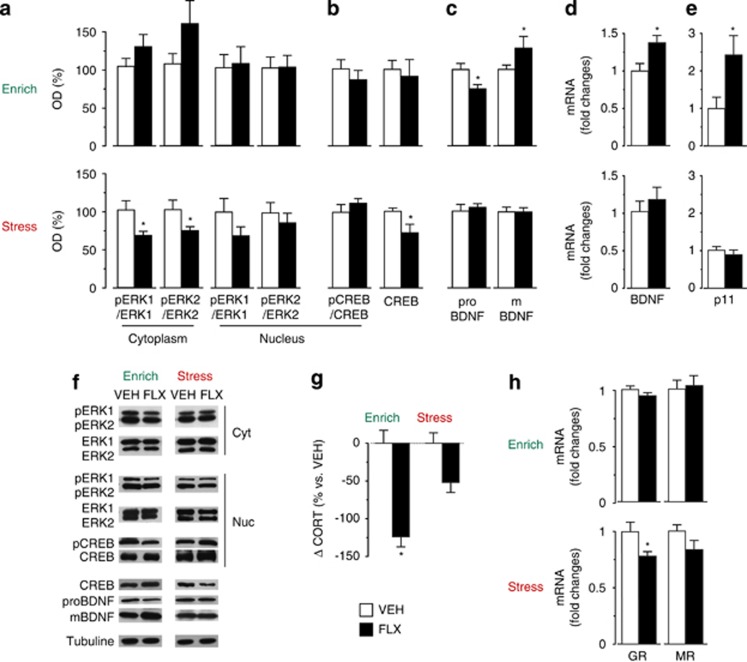
Although, in the enriched condition, the FLX-induced increase of the phosphoERK2/ERK2 ratio in the hippocampal cytoplasmic fraction missed to reach statistical significance (t=1.765, P=0.1030), in the stressful condition a reduction in ERK 1/2 phosphorylation was specifically observed in the hippocampi of FLX compared with VEH mice (t=−2.928, P=0.0118; Figure 3a). This effect was mainly due to a 30% reduction in phosho-ERK 1/2 levels and no change in ERK 1/2 levels was observed (data not shown). The nuclear phospho-CREB\CREB ratio was not affected by FLX in both environmental conditions (Figure 3b); however, a significant reduction of CREB levels was found in the total extract obtained from hippocampi of FLX mice exposed to the stressful condition (t=−2.281, P=0.0435; Figure 3b). When FLX was administered in enriched condition, we found increased hippocampal levels of mature BDNF (t=2.242, P=0.0488), accordingly associated to decreased levels of its precursor pro BDNF (t=−2.459, P=0.0276; Figure 3c). No effect was observed in the levels of both BDNF forms in the stressful condition (Figure 3c). In line with protein data, we found hippocampal BDNF expression to be increased in FLX compared with VEH mice (t=2.511, P=0.0332) in the enriched condition (Figure 3d). No significant effect was observed in the expression of the coding BDNF exon common to all the transcripts in stress FLX mice (Figure 3d). High BDNF levels induced by antidepressants have been reported to increase p11 (also known as S100a10) expression,51 which is a protein involved in depression and antidepressant response.52 Accordingly, we found increased levels of p11 mRNA (t=2.508, P=0.0290) in the hippocampus of FLX compared with VEH mice in the enriched but not in the stressful condition (Figure 3e). In order to assess whether the environment-dependent effects of FLX on antidepressant-related pathways concern specifically the hippocampus or other brain areas as well, we measured ERK, CREB and BDNF protein levels in the medial prefrontal cortex. The results obtained, although not fully overlapping with those obtained in the hippocampus, are in line with the hypothesis of the present study. In particular, in the medial prefrontal cortex, FLX treatment enhanced pERK1/2–total ERK1/2 ratio in the nuclear fraction in enrichment (t=2.466; P=0.0431), while reducing it in the stressful condition (t=−2.163; P=0.0483). In addition, in the latter condition, drug treatment reduced CREB phosphorylation in the nuclear fraction (t=−3.0761; P=0.02759) and mature BDNF protein levels in the total extract (t=−3.076; P=0.0430; Supplementary Figure S3).
Figure 3.
Fluoxetine (FLX) treatment affects hippocampal signaling pathways and HPA axis activity according to the quality of the environment. (a) No significant treatment effect was found for pERK1/ERK1 and in pERK2/ERK2 ratios in the enriched condition. Whereas both ratios were reduced in the cytoplasmic, but not in the nuclear, fraction in FLX mice in the stressful condition. (b) No treatment effect on CREB phosphorylation was observed in the nuclear enriched fraction in both environmental conditions. No difference in the total hippocampal CREB protein levels was found in the enriched condition, but it was reduced by treatment in the stressful condition. (c) In enriched conditions, reduced proBDNF and increased mBDNF levels were found in FLX mice compared with vehicle (VEH) mice. No difference in the stressful condition. (d and e) Real-time PCR analysis revealed that BDNF and p11 mRNA levels were significantly increased by FLX in the enriched condition but were not affected in the stressful condition. (f) Representative western blottings are shown. (g) Corticosterone levels were significantly reduced by treatment in FLX compared with VEH mice in the enriched but not in the stressful condition. (h) Glucocorticoid receptor (GR) mRNA expression was reduced by treatment in the stressful condition. No difference for GR expression in the enriched condition and mineralocorticoid receptor (MR) expression in both condition was found. MR, mineralocorticoid receptor. Corticosterone analysis: VEH, n= 6; FLX, n=7. For all the other analyses, n=8 in all groups. Data shown as mean±s.e.m. *P<0.05 vs respective VEH group.
Hyperactivation of the HPA axis, which leads to increased glucocorticoid levels, is a common feature in major depression.9, 53, 54 HPA axis hyperactivity and blunted feedback recover after chronic treatment with antidepressants and are associated to an improvement of depressive symptoms.55 Accordingly, our analysis of the differences in corticosterone levels before and after treatment revealed that the levels of this hormone were reduced by FLX, compared with VEH, in the enriched (F(1,11)=5.262, P=0.0425) but not in the stressful condition (Figure 3g). Glucocorticoids exert their effects on the hippocampus, including regulation of HPA axis activity and synaptic plasticity, by binding two receptor systems: the high-affinity mineralocorticoid receptor and the low-affinity glucocorticoid receptor (GR).56, 57 Activation of hippocampal GR causes inhibition of the HPA axis.58 Several groups demonstrated that a chronic FLX treatment increases GR mRNA expression in the hippocampus, possibly contributing to restore HPA axis feedback.59 Although not affecting GR and mineralocorticoid receptor mRNA expression in the enriched condition, FLX reduced GR, but not mineralocorticoid receptor, mRNA levels (t=−2.558, P=0.0228) in the stressful condition (Figure 3h). The latter result could lead to a blunted HPA feedback, which is consistent with the lack of reduction in corticosterone levels in mice exposed to stress, following FLX treatment.
The interaction between the quality of the environment and FLX determines treatment outcome on neurotransmitter receptors and LTP
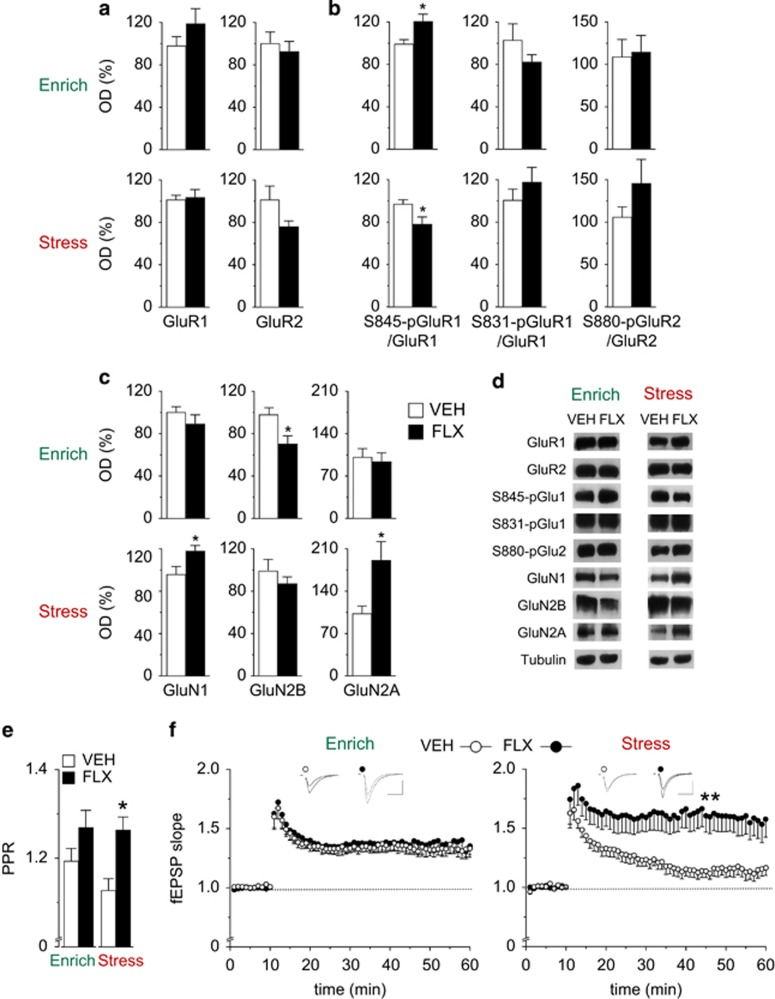
Beside its effects on intracellular pathways, FLX has been reported to affect hippocampal plasticity modulating neurotransmitter receptor number and function, and modifying synaptic LTP.11, 29, 30, 60, 61, 62, 63, 64 As AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) and NMDA (N-methyl-D-aspartate) receptor subunits are regulated during activity-dependent plasticity, we evaluated the effects of FLX on excitatory receptor subunits in both enriched and stressful conditions. Overall, we did not observe any significant FLX effect on total hippocampal GluR1 and GluR2 subunit levels in both environmental conditions (Figure 4a). As AMPA receptor functionality is regulated by phosphorylation, we measured the phosphorylation levels on the GluR1 and GluR2 subunits. Following FLX administration, Ser845 phosphorylation on GluR1 subunit was increased in the enriched (t=2.490, P=0.0259) and decreased in the stressful condition (t=−2.236, P=0.0434; Figure 4b). By contrast, phosphorylation at Ser831-GluR1 and Ser880-GluR2 were not affected in both environmental conditions (Figure 4b). Such phosphorylation profile is in line with previous findings reporting that Ser845 is a specific target of FLX.65 Although AMPA receptor phosphorylation is critical for synaptic plasticity, the functional consequences of the phosphorylation site modulation depend on the previous history of the synapse.66 This complexity makes the relationship between phosphorylation and consequences on plasticity processes in different environmental conditions difficult to predict. With regard to NMDA receptor, we found that FLX increases GluN2A and GluNR1 subunits (respectively, t=2.811, P=0.0147 and t=2.389, P=0.0342) in the stress condition, while reducing GluN2B (t=−2.830, P=0.0151) in enrichment (Figure 4c). Given that the increase in the GluNR2A/GluNR2B ratio and the insertion of the GluNR1 subunits have been associated to an enhanced LTP,67 data concerning the NMDA receptor function in the stressful condition are in line with an increase of this form of plasticity induced by FLX.
Figure 4.
Fluoxetine (FLX) modifies molecular and cellular correlates of synaptic plasticity in an environment-dependent manner. (a) Hippocampal levels of AMPA receptor subunits GluR1 and GluR2, and (b) their phosphorylation measured by western blotting. FLX induced opposite effects on GluR1 phosphorylation at Ser 845 according to the quality of the environment. (c) Hippocampal levels of NMDA receptor subunits GluN1, GluN2A and GluN2B measured by western blotting. (d) Representative western blots. n=8 for all groups in western blot analyses. Data shown as mean±s.e.m. of the control. *P<0.05 vs respective vehicle (VEH) group. (e) FLX increased paired-pulse ratio in the stressful condition but had no effect in the enriched condition. VEH: n=18/5; FLX: n=15/4. (f) FLX affect CA1 plasticity in mice exposed to stress condition. In the enriched condition, both FLX and VEH mice developed a robust LTP 45 min after stimulation (single 100 Hz burst; VEH: n=12/5; FLX: n=11/5). In the stressful condition, FLX mice showed a remarkable increase in the LTP amplitude compared with VEH (VEH: n=14/6; FLX: n=10/5). Arrows indicate time of application of HFS. fEPSP, field excitatorypostsynaptic potential; PPR, paired-pulse ratio. Data shown as mean±s.e.m. *P<0.05 and **P<0.01 vs respective VEH group.
As FLX has been reported to selectively affect excitatory neurotransmitter receptors and plasticity in specific subregions of the hippocampus,29 we decided to explore the functional implication of its administration in a selected and well-characterized activity-dependent cellular paradigm, the LTP, in the CA1 hippocampal output area. NMDA-dependent LTP processes occurring in this region are reportedly affected by FLX11, 29 and by the quality of the living environment.68, 69 We first measured the strength of CA3-CA1 connections by recording normalized input–output (I–O) curves, finding no difference between VEH and FLX groups in both environmental conditions (Supplementary Figure S4a). However, FLX treatment induced an overall increase in the absolute size of field excitatorypostsynaptic potential (1.6±0.1 in the stressful condition and 2.1±0.2 in the enriched condition). In particular, the distribution of field excitatorypostsynaptic potential data, across the entire range of the stimulus intensities, were significantly different in FLX compared with VEH mice (P=0.0024). These findings are in line with previous studies showing an FLX-mediated enhancement of the synaptic strength.11, 29, 30, 34, 64
We next analyzed the intrinsic excitability of the CA1 pyramidal cells by measuring the stimulation intensity at which a population spike is first observed. The intensity (data not shown) as well as the size of the field excitatorypostsynaptic potential slope that correlated to the occurrence of population spikes (Supplementary Figure S4b) were similar in VEH and FLX mice in both environmental conditions. Paired-pulse ratio, generally associated with changes in transmitter release probability,70 was significantly increased in FLX compared with VEH mice in the stressful (t=−3.306, P=0.002) but not in the enriched condition.
Finally, we explored CA1 plasticity inducing LTP by weak stimulation (single 100 Hz burst) of Shaffer Collateral inputs.69 In the enriched condition, the experimental groups did not differ, both exhibiting a robust LTP (Figure 4f), in line with the literature reporting that enrichment enhances LTP amplitude evoked by a weak stimulation.69 In the stressful condition, FLX mice showed a significantly increased LTP amplitude compared with VEH mice (t=−3.813, P=0.002) that displayed a slight potentiation following the stimulation (Figure 4f). Overall, these results demonstrate that also the effects of FLX on short- and long-term plasticity in the CA1 region are dependent on the quality of the environment.
In order to study FLX outcome without the influence of enrichment, we investigated the effects of the drug in the standard condition. This consisted in exposing mice to chronic stress for 2 weeks and, afterwards, administering them with either VEH or FLX for 3 weeks, while housed in a standard laboratory condition (Supplementary Figure S5a). Overall, the results obtained are in line with the notion that the shift from an uncertain situation (that is, the stress) to a neutral one (that is, the standard condition) leads to an increase in the ability of the organism to cope and control the environment and can thus have beneficial effects (see Lazarus and Folkman71). Indeed, anhedonic responses and LTP levels overlap in the standard and the enriched condition. However, in the standard condition, FLX did not increase BDNF levels, suggesting that enrichment drives the FLX effects more powerfully than the improvement associated to the standard condition following chronic stress (Supplementary Figure S5).
Discussion
The main finding of the present study is that the effects of the SSRI FLX are highly dependent on the quality of the living environment. FLX administration in an enriched condition led to a significant recovery from the depression-like phenotype compared with VEH, which is in line with the expected effects of antidepressants.72 However, these effects cannot be ascribed to drug action per se, but arise from the interaction of the drug with the quality of the environment, as FLX treatment under stress led to a worsening of the depression-like phenotype (Table 1). Such environment-dependent effect is pervasive, as it concerns most of the phenotypic features analyzed, from molecular pathways to behavior. It is worth noting that, in line with the undirected susceptibility to change hypothesis, our results show that FLX treatment made the experimental subjects more susceptible to the quality of the environment, be it either enrichment or stress.
Table 1. Effects of fluoxetine, compared with vehicle, in the enriched and the stressful conditions.
| Reduction in liking-type anhedonia | Reduction in wanting-type anhedonia | Cognitive bias | Neurogenesis (Ki67) | ERK signaling | CREB signaling | BDNF levels | Reduction in CORT levels | LTP | |
|---|---|---|---|---|---|---|---|---|---|
| Enriched condition | – | ↑ | ↑ | – | – | – | ↑ | ↑ | – |
| Stressful condition | ↓ | ↓ | – | ↓ | ↓ | ↓ | – | – | ↓ |
Taking into account the quality of the environment as a moderator of SSRI treatment outcome may explain the inconsistency of the findings obtained in previous studies. Indeed, overlooking the environment as key factor allowing to identify subgroups of individuals differently responding to treatment73 might have led to the reported contradictory results in pivotal endpoints of depression-like phenotype, including BDNF levels,8, 21 LTP,11, 29 HPA axis activity,9, 27 neurogenesis7, 16 and behavior.5, 12
Although we found that FLX effects are driven by the quality of the environment, we did not observe an overlap between neural and behavioral endpoints modified by FLX in the enriched and stressful conditions, suggesting that the mechanisms mediating the improvement and the worsening of the depressive status may differ (Table 1). For instance, BDNF levels were increased in the enriched, but not affected in the stressful condition. This is in line with previous studies31 showing that the levels of this neurotrophin are modified by FLX treatment in an enriched but not in a constantly stressful environment. By contrast, other parameters, such as neurogenesis, were affected only when the drug was administered in the stressful environment. Although a number of papers have reported that antidepressant treatment enhances neurogenesis,42 our results are concordant with an increasing number of recent studies indicating no effect16, 17 or even a reduction of neurogenesis following antidepressant administration.18 Overall, while suggesting a novel conceptual framework to understand SSRI action, the present findings confirm the involvement in the depression-like phenotype of molecular pathways or mechanisms already demonstrated to be implicated in major depression, including BDNF expression, HPA axis activity and neurogenesis. The modifications in these endpoints here reported accompany the improvement or worsening of the depression-like behavioral phenotype in a manner coherent with previous studies on the neurobiological substrates of depression.74, 75
With regard to LTP, a primary form of plasticity, we expected that, in line with our hypothesis, it would have been increased by FLX treatment. Indeed, our results show that, in the stressful condition, FLX significantly enhances plasticity in the CA1 region in comparison with VEH. However, in the enriched condition, both groups exhibited a robust LTP amplitude, in line with the literature reporting increased LTP levels following enrichment.69 Such potentiation might have masked the effect of FLX because of a ceiling effect due to the saturation of LTP magnitude. Our results on NMDA receptor subunit expression profile, showing an increase in the GluNR2A/GluNR2B ratio, are concordant with an increase in neural plasticity induced by FLX administration. Such modifications in neuronal functionality may represent the mechanisms underlying the effects of the drug treatment. Accordingly, it has been shown that chronic FLX induces a de-differentiation of mature dentate granule neurons in the adult hippocampus that reinstates high levels of synaptic plasticity similar to those of the early postnatal phase,76 which is characterized by a high susceptibility to environmental inputs.77 Further research is warranted to shed light on the molecular pathways and pathophysiological mechanisms underlying the different consequences of the FLX–environment interactions. It is worth noting that the effects of FLX described in the present paper may represent only part of the action of SSRIs on brain and behavior, as the serotonergic system has a high molecular complexity and is involved in a wide range of functions. It should also be mentioned that the effects of chronic stress are strain dependent.78 For instance, FLX treatment during chronic stress in BALB/c mice, a strain widely used to study major depression, counteracts stress effects, restoring food consumption in the Cookie test, reverting deterioration of the coat state and reinstating HPA axis negative feedback and neurogenesis in the dentate gyrus.79, 80
Few clinical studies have investigated the influence of the environment on antidepressant action in patients and the findings show that living conditions, for example, socioeconomic status and lifestyle, modulate the effects of antidepressants.81 In particular, in line with the present results, these studies suggest that antidepressants are more effective in patients having a better quality of life. Findings from the Sequenced Treatment Alternatives to Relieve Depression study, which investigated the response to the SSRI citalopram in over 4000 depressed patients, showed that participants having better living conditions, because they are employed or are with higher levels of education or income, had higher remission rates. By contrast, longer index episodes, more concurrent psychiatric disorders, more general medical disorders and lower baseline function and quality of life were associated with lower remission rates.3, 82 In addition, antidepressant treatment has been reported to lead more often to an unfavorable outcome in people living in worst conditions. In particular, patients in the poor-income census tract were significantly more likely to report suicidal ideation than those in the middle- and high- income census tracts.82, 83 Overall, these findings show that the quality of the living environment, such as adverse economic conditions, affects the effectiveness of antidepressant treatment. Therefore, the quality of the living environment acts as moderator of treatment response. Taking into account this moderator in the clinical practice may specify for whom or under what conditions the treatment works and may suggest to clinicians which of their patients might be most responsive to the treatment and for which patients other, more appropriate, treatments might be sought.73
The conceptual shift in considering the modification in serotonin levels from being the cause of depression to acting as a permissive factor in the onset of and the recovery from the psychopathology allows to reconcile experimental and clinical data that apparently do not fit together.31 For instance, the currently available theoretical framework leads to the paradox that the same mechanism of action has two opposite outcomes. In particular, high extracellular serotonin levels are beneficial when induced by SSRI administration, but confer a high risk to develop psychopathology when associated to the s variant of 5-HTTLPR. This discordant picture can be coherently interpreted in light of our results and the undirected susceptibility to change hypothesis, positing that high serotonin levels lead to increased plasticity and thus to high susceptibility to change, which may promote either an improvement or a worsening according to the quality of the environment.
In conclusion, the identification of the living environment as a moderator of treatment response represents a critical step in developing a personalized medicine approach aimed at better matching patients with treatment through selective enhancement of treatment efficacy and avoiding potential harmful consequences. The control of the patients' living environment could be achieved by training patients to cope with harsh conditions, for instance, through cognitive behavioral therapy,62 as it is unlikely that people can rapidly and effectively change their living milieu. The cost of this approach is limited, as no new psychoactive molecules need to be developed, while the benefits for the patients could be substantial.
Acknowledgments
We thank Shih-hui Huang for her help with figures and drawings, and Stefanie Engler and Stella Falsini for technical support. Funding for this study was provided by the Italian Ministry of Health, Ricerca Finalizzata RF-2009-1498890 to FC, RF-2011-02349921 to IB, the Italian Ministry of University and Research, FIRB RBFR12QW4I to MT, Olga Mayenfisch foundation to IA and Sapienza University to CL. IB was partially supported by EU, Marie Curie IEF fellowship for Experienced Researcher n: 302881–Acronym: MODELMOOD. DPW is a member of the Neuroscience Center Zurich (ZNZ) and the Zurich Center for Integrative Human Physiology (ZIHP).
Footnotes
Supplementary Information accompanies the paper on the Molecular Psychiatry website (http://www.nature.com/mp)
The authors declare no conflict of interest.
Supplementary Material
References
- WHOThe Global Burden of Disease: 2004 Update. World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Balak N, Elmaci I. Costs of disorders of the brain in Europe. Eur J Neurol 2007; 14: e9. [DOI] [PubMed] [Google Scholar]
- Trivedi MH, Rush AJ, Wisniewski SR, Nierenberg AA, Warden D, Ritz L et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry 2006; 163: 28–40. [DOI] [PubMed] [Google Scholar]
- Kirsch I, Deacon BJ, Huedo-Medina TB, Scoboria A, Moore TJ, Johnson BT. Initial severity and antidepressant benefits: a meta-analysis of data submitted to the Food and Drug Administration. PLoS Med 2008; 5: e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bessa JM, Ferreira D, Melo I, Marques F, Cerqueira JJ, Palha JA et al. The mood-improving actions of antidepressants do not depend on neurogenesis but are associated with neuronal remodeling. Mol Psychiatry 2009; 14: 764–773, 739. [DOI] [PubMed] [Google Scholar]
- Rygula R, Abumaria N, Flugge G, Hiemke C, Fuchs E, Ruther E et al. Citalopram counteracts depressive-like symptoms evoked by chronic social stress in rats. Behav Pharmacol 2006; 17: 19–29. [DOI] [PubMed] [Google Scholar]
- Malberg JE, Eisch AJ, Nestler EJ, Duman RS. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J Neurosci 2000; 20: 9104–9110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nibuya M, Nestler EJ, Duman RS. Chronic antidepressant administration increases the expression of cAMP response element binding protein (CREB) in rat hippocampus. J Neurosci 1996; 16: 2365–2372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pariante CM. Glucocorticoid receptor function in vitro in patients with major depression. Stress 2004; 7: 209–219. [DOI] [PubMed] [Google Scholar]
- Uys JD, Muller CJ, Marais L, Harvey BH, Stein DJ, Daniels WM. Early life trauma decreases glucocorticoid receptors in rat dentate gyrus upon adult re-stress: reversal by escitalopram. Neuroscience 2006; 137: 619–625. [DOI] [PubMed] [Google Scholar]
- Bath KG, Jing DQ, Dincheva I, Neeb CC, Pattwell SS, Chao MV et al. BDNF Val66Met impairs fluoxetine-induced enhancement of adult hippocampus plasticity. Neuropsychopharmacology 2012; 37: 1297–1304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenes JC, Fornaguera J. The effect of chronic fluoxetine on social isolation-induced changes on sucrose consumption, immobility behavior, and on serotonin and dopamine function in hippocampus and ventral striatum. Behav Brain Res 2009; 198: 199–205. [DOI] [PubMed] [Google Scholar]
- Prendergast MA, Yells DP, Balogh SE, Paige SR, Hendricks SE. Fluoxetine differentially suppresses sucrose solution consumption in free-fed and food-deprived rats—reversal by amantadine. Med Sci Monit 2002; 8: BR385–BR390. [PubMed] [Google Scholar]
- Sammut S, Bethus I, Goodall G, Muscat R. Antidepressant reversal of interferon-alpha-induced anhedonia. Physiol Behav 2002; 75: 765–772. [DOI] [PubMed] [Google Scholar]
- Tonissaar M, Mallo T, Eller M, Haidkind R, Koiv K, Harro J. Rat behavior after chronic variable stress and partial lesioning of 5-HT-ergic neurotransmission: effects of citalopram. Prog Neuropsychopharmacol Biol Psychiatry 2008; 32: 164–177. [DOI] [PubMed] [Google Scholar]
- David DJ, Samuels BA, Rainer Q, Wang JW, Marsteller D, Mendez I et al. Neurogenesis- dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron 2009; 62: 479–493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marlatt MW, Potter MC, Bayer TA, van Praag H, Lucassen PJ. Prolonged running, not fluoxetine treatment, increases neurogenesis, but does not alter neuropathology, in the 3xTg mouse model of Alzheimer's disease. Curr Topics Behavi Neurosci 2013; 15: 313–340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu MV, Shamy JL, Bedi G, Choi CW, Wall MM, Arango V et al. Impact of social status and antidepressant treatment on neurogenesis in the baboon hippocampus. Neuropsychopharmacology 2014; 39: 1861–1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klomp A, Vaclavu L, Meerhoff GF, Reneman L, Lucassen PJ. Effects of chronic fluoxetine treatment on neurogenesis and tryptophan hydroxylase expression in adolescent and adult rats. PLoS One 2014; 9: e97603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Possamai F, dos Santos J, Walber T, Marcon JC, dos Santos TS, Lino de Oliveira C. Influence of enrichment on behavioral and neurogenic effects of antidepressants in Wistar rats submitted to repeated forced swim test. Prog Neuropsychopharmacol Biol Psychiatry 2015; 58: 15–21. [DOI] [PubMed] [Google Scholar]
- Jacobsen JP, Mork A. The effect of escitalopram, desipramine, electroconvulsive seizures and lithium on brain-derived neurotrophic factor mRNA and protein expression in the rat brain and the correlation to 5-HT and 5-HIAA levels. Brain Res 2004; 1024: 183–192. [DOI] [PubMed] [Google Scholar]
- Kozisek ME, Middlemas D, Bylund DB. Brain-derived neurotrophic factor and its receptor tropomyosin-related kinase B in the mechanism of action of antidepressant therapies. Pharmacol Ther 2008; 117: 30–51. [DOI] [PubMed] [Google Scholar]
- Miro X, Perez-Torres S, Artigas F, Puigdomenech P, Palacios JM, Mengod G. Regulation of cAMP phosphodiesterase mRNAs expression in rat brain by acute and chronic fluoxetine treatment. An in situ hybridization study. Neuropharmacology 2002; 43: 1148–1157. [DOI] [PubMed] [Google Scholar]
- Zetterstrom TS, Pei Q, Madhav TR, Coppell AL, Lewis L, Grahame-Smith DG. Manipulations of brain 5-HT levels affect gene expression for BDNF in rat brain. Neuropharmacology 1999; 38: 1063–1073. [DOI] [PubMed] [Google Scholar]
- Alboni S, Benatti C, Capone G, Corsini D, Caggia F, Tascedda F et al. Time-dependent effects of escitalopram on brain derived neurotrophic factor (BDNF) and neuroplasticity related targets in the central nervous system of rats. Eur J Pharmacol 2010; 643: 180–187. [DOI] [PubMed] [Google Scholar]
- Goekint M, Roelands B, Heyman E, Njemini R, Meeusen R. Influence of citalopram and environmental temperature on exercise-induced changes in BDNF. Neurosci Lett 2011; 494: 150–154. [DOI] [PubMed] [Google Scholar]
- Shen Q, Lal R, Luellen BA, Earnheart JC, Andrews AM, Luscher B. gamma-Aminobutyric acid- type A receptor deficits cause hypothalamic-pituitary-adrenal axis hyperactivity and antidepressant drug sensitivity reminiscent of melancholic forms of depression. Biol Psychiatry 2010; 68: 512–520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber CC, Eckert GP, Muller WE. Effects of antidepressants on the brain/plasma distribution of corticosterone. Neuropsychopharmacology 2006; 31: 2443–2448. [DOI] [PubMed] [Google Scholar]
- Rubio FJ, Ampuero E, Sandoval R, Toledo J, Pancetti F, Wyneken U. Long-term fluoxetine treatment induces input-specific LTP and LTD impairment and structural plasticity in the CA1 hippocampal subfield. Front Cell Neurosci 2013; 7: 66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart CA, Reid IC. Repeated ECS and fluoxetine administration have equivalent effects on hippocampal synaptic plasticity. Psychopharmacology 2000; 148: 217–223. [DOI] [PubMed] [Google Scholar]
- Branchi I. The double edged sword of neural plasticity: increasing serotonin levels leads to both greater vulnerability to depression and improved capacity to recover. Psychoneuroendocrinology 2011; 36: 339–351. [DOI] [PubMed] [Google Scholar]
- Belsky J, Jonassaint C, Pluess M, Stanton M, Brummett B, Williams R. Vulnerability genes or plasticity genes? Mol Psychiatry 2009; 14: 746–754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maya Vetencourt JF, Sale A, Viegi A, Baroncelli L, De Pasquale R, O'Leary OF et al. The antidepressant fluoxetine restores plasticity in the adult visual cortex. Science 2008; 320: 385–388. [DOI] [PubMed] [Google Scholar]
- Karpova NN, Pickenhagen A, Lindholm J, Tiraboschi E, Kulesskaya N, Agustsdottir A et al. Fear erasure in mice requires synergy between antidepressant drugs and extinction training. Science 2011; 334: 1731–1734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brummett BH, Boyle SH, Siegler IC, Kuhn CM, Ashley-Koch A, Jonassaint CR et al. Effects of environmental stress and gender on associations among symptoms of depression and the serotonin transporter gene linked polymorphic region (5-HTTLPR). Behav Genet 2008; 38: 34–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eley TC, Sugden K, Corsico A, Gregory AM, Sham P, McGuffin P et al. Gene-environment interaction analysis of serotonin system markers with adolescent depression. Mol Psychiatry 2004; 9: 908–915. [DOI] [PubMed] [Google Scholar]
- Mastronardi C, Paz-Filho GJ, Valdez E, Maestre-Mesa J, Licinio J, Wong ML. Long-term body weight outcomes of antidepressant-environment interactions. Mol Psychiatry 2011; 16: 265–272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong ML, Licinio J. Research and treatment approaches to depression. Nat Rev Neurosci 2001; 2: 343–351. [DOI] [PubMed] [Google Scholar]
- Benatti C, Alboni S, Montanari C, Caggia F, Tascedda F, Brunello N et al. Central effects of a local inflammation in three commonly used mouse strains with a different anxious phenotype. Behav Brain Res 2011; 224: 23–34. [DOI] [PubMed] [Google Scholar]
- Tsankova NM, Berton O, Renthal W, Kumar A, Neve RL, Nestler EJ. Sustained hippocampal chromatin regulation in a mouse model of depression and antidepressant action. Nat Neurosci 2006; 9: 519–525. [DOI] [PubMed] [Google Scholar]
- Small SA, Schobel SA, Buxton RB, Witter MP, Barnes CA. A pathophysiological framework of hippocampal dysfunction in ageing and disease. Nat Rev Neurosci 2011; 12: 585–601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santarelli L, Saxe M, Gross C, Surget A, Battaglia F, Dulawa S et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 2003; 301: 805–809. [DOI] [PubMed] [Google Scholar]
- Navailles S, Hof PR, Schmauss C. Antidepressant drug-induced stimulation of mouse hippocampal neurogenesis is age-dependent and altered by early life stress. J Comp Neurol 2008; 509: 372–381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowen DS, Takase LF, Fornal CA, Jacobs BL. Age-dependent decline in hippocampal neurogenesis is not altered by chronic treatment with fluoxetine. Brain Res 2008; 1228: 14–19. [DOI] [PubMed] [Google Scholar]
- Bremner JD, Narayan M, Anderson ER, Staib LH, Miller HL, Charney DS. Hippocampal volume reduction in major depression. Am J Psychiatry 2000; 157: 115–118. [DOI] [PubMed] [Google Scholar]
- Gourley SL, Wu FJ, Kiraly DD, Ploski JE, Kedves AT, Duman RS et al. Regionally specific regulation of ERK MAP kinase in a model of antidepressant-sensitive chronic depression. Biol Psychiatry 2008; 63: 353–359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duric V, Banasr M, Licznerski P, Schmidt HD, Stockmeier CA, Simen AA et al. A negative regulator of MAP kinase causes depressive behavior. Nat Med 2010; 16: 1328–1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen AC, Shirayama Y, Shin KH, Neve RL, Duman RS. Expression of the cAMP response element binding protein (CREB) in hippocampus produces an antidepressant effect. Biol Psychiatry 2001; 49: 753–762. [DOI] [PubMed] [Google Scholar]
- Thome J, Sakai N, Shin K, Steffen C, Zhang YJ, Impey S et al. cAMP response element-mediated gene transcription is upregulated by chronic antidepressant treatment. J Neurosci 2000; 20: 4030–4036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirayama Y, Chen AC, Nakagawa S, Russell DS, Duman RS. Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J Neurosci 2002; 22: 3251–3261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warner-Schmidt JL, Chen EY, Zhang X, Marshall JJ, Morozov A, Svenningsson P et al. A role for p11 in the antidepressant action of brain-derived neurotrophic factor. Biol Psychiatry 2010; 68: 528–535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svenningsson P, Kim Y, Warner-Schmidt J, Oh YS, Greengard P. p11 and its role in depression and therapeutic responses to antidepressants. Nat Rev Neurosci 2013; 14: 673–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alboni S, Tascedda F, Corsini D, Benatti C, Caggia F, Capone G et al. Stress induces altered CRE/CREB pathway activity and BDNF expression in the hippocampus of glucocorticoid receptor-impaired mice. Neuropharmacology 2011; 60: 1337–1346. [DOI] [PubMed] [Google Scholar]
- Holsboer F, Ising M. Stress hormone regulation: biological role and translation into therapy. Ann Rev Psychol 2010; 61: 81–109, C101-C111. [DOI] [PubMed] [Google Scholar]
- Ising M, Horstmann S, Kloiber S, Lucae S, Binder EB, Kern N et al. Combined dexamethasone/corticotropin releasing hormone test predicts treatment response in major depression - a potential biomarker? Biol Psychiatry 2007; 62: 47–54. [DOI] [PubMed] [Google Scholar]
- de Kloet ER, Fitzsimons CP, Datson NA, Meijer OC, Vreugdenhil E. Glucocorticoid signaling and stress-related limbic susceptibility pathway: about receptors, transcription machinery and microRNA. Brain Res 2009; 1293: 129–141. [DOI] [PubMed] [Google Scholar]
- Datson NA, Speksnijder N, Mayer JL, Steenbergen PJ, Korobko O, Goeman J et al. The transcriptional response to chronic stress and glucocorticoid receptor blockade in the hippocampal dentate gyrus. Hippocampus 2012; 22: 359–371. [DOI] [PubMed] [Google Scholar]
- Furay AR, Bruestle AE, Herman JP. The role of the forebrain glucocorticoid receptor in acute and chronic stress. Endocrinology 2008; 149: 5482–5490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anacker C, Zunszain PA, Carvalho LA, Pariante CM. The glucocorticoid receptor: pivot of depression and of antidepressant treatment? Psychoneuroendocrinology 2011; 36: 415–425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alme MN, Wibrand K, Dagestad G, Bramham CR. Chronic fluoxetine treatment induces brain region-specific upregulation of genes associated with BDNF-induced long-term potentiation. Neural Plasticity 2007; 2007: 26496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi K, Ikeda Y, Asada M, Inagaki H, Kawada T, Suzuki H. Corticosterone facilitates fluoxetine-induced neuronal plasticity in the hippocampus. PLoS One 2013; 8: e63662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiles N, Thomas L, Abel A, Ridgway N, Turner N, Campbell J et al. Cognitive behavioural therapy as an adjunct to pharmacotherapy for primary care based patients with treatment resistant depression: results of the CoBalT randomised controlled trial. Lancet 2012; 381: 375–384. [DOI] [PubMed] [Google Scholar]
- Marsden WN. Synaptic plasticity in depression: molecular, cellular and functional correlates. Prog Neuropsychopharmacol Biol Psychiatry 2013; 43: 168–184. [DOI] [PubMed] [Google Scholar]
- Wang JW, David DJ, Monckton JE, Battaglia F, Hen R. Chronic fluoxetine stimulates maturation and synaptic plasticity of adult-born hippocampal granule cells. J Neurosci 2008; 28: 1374–1384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svenningsson P, Tzavara ET, Witkin JM, Fienberg AA, Nomikos GG, Greengard P. Involvement of striatal and extrastriatal DARPP-32 in biochemical and behavioral effects of fluoxetine (Prozac). Proc NatlAcad Sci USA 2002; 99: 3182–3187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee HK, Barbarosie M, Kameyama K, Bear MF, Huganir RL. Regulation of distinct AMPA receptor phosphorylation sites during bidirectional synaptic plasticity. Nature 2000; 405: 955–959. [DOI] [PubMed] [Google Scholar]
- Peng Y, Zhao J, Gu QH, Chen RQ, Xu Z, Yan JZ et al. Distinct trafficking and expression mechanisms underlie LTP and LTD of NMDA receptor-mediated synaptic responses. Hippocampus 2010; 20: 646–658. [DOI] [PubMed] [Google Scholar]
- Joels M, Krugers HJ. LTP after stress: up or down? Neural Plasticity 2007; 2007: 93202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duffy SN, Craddock KJ, Abel T, Nguyen PV. Environmental enrichment modifies the PKA- dependence of hippocampal LTP and improves hippocampus-dependent memory. Learn Memory 2001; 8: 26–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zucker RS. Short-term synaptic plasticity. Ann Rev Neurosci 1989; 12: 13–31. [DOI] [PubMed] [Google Scholar]
- Lazarus RS, Folkman S. Stress, Appraisal, and Coping. Springer: New York, 1984. [Google Scholar]
- Kramer PD. Listening to Prozac: Psychiatrist Explores Antidepressant Drugs and the Remaking of the Self. Fourth Estate Ltd: London, 1994. [Google Scholar]
- Kraemer HC, Frank E, Kupfer DJ. Moderators of treatment outcomes: clinical, research, and policy importance. JAMA 2006; 296: 1286–1289. [DOI] [PubMed] [Google Scholar]
- Belmaker RH, Agam G. Major depressive disorder. N Engl J Med 2008; 358: 55–68. [DOI] [PubMed] [Google Scholar]
- Krishnan V, Nestler EJ. Linking molecules to mood: new insight into the biology of depression. Am J Psychiatry 2010; 167: 1305–1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi K, Ikeda Y, Sakai A, Yamasaki N, Haneda E, Miyakawa T et al. Reversal of hippocampal neuronal maturation by serotonergic antidepressants. Proc Natl Acad Sci USA 2010; 107: 8434–8439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baroncelli L, Braschi C, Spolidoro M, Begenisic T, Sale A, Maffei L. Nurturing brain plasticity: impact of environmental enrichment. Cell Death Differ 2010; 17: 1092–1103. [DOI] [PubMed] [Google Scholar]
- Ibarguen-Vargas Y, Surget A, Touma C, Palme R, Belzung C. Multifaceted strain-specific effects in a mouse model of depression and of antidepressant reversal. Psychoneuroendocrinology 2008; 33: 1357–1368. [DOI] [PubMed] [Google Scholar]
- Surget A, Tanti A, Leonardo ED, Laugeray A, Rainer Q, Touma C et al. Antidepressants recruit new neurons to improve stress response regulation. Mol Psychiatry 2011; 16: 1177–1188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanti A, Westphal WP, Girault V, Brizard B, Devers S, Leguisquet AM et al. Region-dependent and stage-specific effects of stress, environmental enrichment, and antidepressant treatment on hippocampal neurogenesis. Hippocampus 2013; 23: 797–811. [DOI] [PubMed] [Google Scholar]
- Jain FA, Hunter AM, Brooks JO 3rd, Leuchter AF. Predictive socioeconomic and clinical profiles of antidepressant response and remission. Depress Anxiety 2013; 30: 624–630. [DOI] [PubMed] [Google Scholar]
- Cohen A, Houck PR, Szanto K, Dew MA, Gilman SE, Reynolds CF 3rd. Social inequalities in response to antidepressant treatment in older adults. Arch Gen psychiatry 2006; 63: 50–56. [DOI] [PubMed] [Google Scholar]
- Cohen A, Gilman SE, Houck PR, Szanto K, Reynolds CF 3rd. Socioeconomic status and anxiety as predictors of antidepressant treatment response and suicidal ideation in older adults. Soc Psychiatry Psychiatr Epidemiol 2009; 44: 272–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.