Abstract
Objective
To determine if diagnostic signs of adhesive capsulitis (AC) of the shoulder at Magnetic Resonance Imaging (MRI) and arthrography (MRA) are applicable to CT arthrography (CTA).
Methods
22 shoulder CTAs with AC were retrospectively reviewed for features described in MR literature. The control group was composed of 83 shoulder CTA divided into four subgroups 1) normal (N = 20), 2) omarthrosis (N = 19), 3) labral injury (N = 23), and 4) rotator cuff tear (N = 21). Two musculoskeletal radiologists assessed the rotator interval (RI) for obliteration, increased width and thickening of coracohumeral ligament (CHL). The width and capsule thickness of the axillary recess were measured.
Results
The width of the axillary recess was significantly decreased in the AC group (4.6 ± 2.6 mm versus 9.9 ± 4.6 mm, p ≤ 0.0001; sensitivity and specificity of 84% and 80%). Thickness of the medial and lateral walls of the axillary capsule was significantly increased in the AC group (5.9 ± 1.3 mm versus 3.7 ± 1.1 mm, p ≤ 0.0001 and 5.7 ± 1 mm versus 3.5 ± 1.3 mm, p ≤ 0.0001, respectively). CHL thickness was significantly increased in the AC group (4.1 ± 1 mm (p ≤ 0.001)) in comparison to others groups. Obliteration of the RI was statistically significantly more frequent in patients with AC (72.7% (16/22) vs. 12% (10/83), p < 0.0001). Width of the RI did not differ significantly between patients and controls (p ≥ 0.428).
Conclusion
Decreased axillary width, and thickened axillary capsule are MR signs of AC applicable to CTA. Evaluation of rotator interval seems useful and reproducible only for obliteration.
Keywords: Adhesive capsulitis, Frozen shoulder, Rotator interval, Arthrography, CTA, MRA
1. Introduction
Adhesive capsulitis (also called “frozen shoulder”) is commonly encountered in general orthopaedic or rheumatology practice. It may be idiopathic, secondary to a variety of causes. The treatment is predominantly conservative, with physiotherapy and glenohumeral injections of steroids [1], [2], [3] while surgery (arthroscopic capsular release) is reserved for patients who do not respond to conservative treatment [4], [5].
The diagnosis of AC is primarily clinical based on characteristic features such as diffuse and severe shoulder pain with stiffness and limitation of both active and passive glenohumeral mobility [6]. However, the differential diagnosis of shoulder pain remains challenging [7], [8] and there is much discrepancy in the literature regarding diagnostic criteria. Imaging may be useful when the clinical presentation is less typical in order to confirm the diagnosis but also to rule out pathologic conditions that can mimic or be associated with secondary AC (i.e. rotator cuff tears, calcific tendonitis, osteoarthritis) [9], [10]. Imaging may be especially useful when invasive treatment is considered.
Arthrography has been considered as the imaging technique of reference to diagnose AC by showing the decreased joint capacity at injection as well as characteristics findings such as the obliteration of the axillary recess [11] and microlacunar humeral bone [12]. MRI and MR arthrography (MRA) of the shoulder have gained popularity, and specific signs of AC have been described and evaluated with a sensitivity and specificity varying between authors [13], [14], [15], [16], [17], [18]. Typically, the width of the RI is decreased [14], [16], [19] with presence of abnormal tissue in the RI [16], RI enhancement or abnormal MRI signal [15], [18]. Thickening of the coracohumeral ligament has also been described [18], [20]. Other findings suggestive of AC at MRI and MRA have been described in the axillary recess. These include increased thickness of capsulosynovial structures [14], [15], [16], [19], a decreased width or volume of the recess [16], [19], [20], as well as contrast enhancement and increased signal intensity of the inferior glenohumeral ligament on T2 sequences [18]. These imaging features of AC have been correlated to arthroscopic and histological findings [21], [22], [23], [24].
Arthrography may be associated with a therapeutic articular injection of corticosteroids [25]. In our institution, clinicians routinely refer patients with clinical suspicion of AC for arthrography-guided articular corticosteroid injections that may be followed by a CTA examination. CTA is used to identify the primary cause of shoulder symptoms or exclude other associated injuries (such as rotator cuff tears, chondropathy) that may affect therapeutic management [26], [27], [28]. In our institution, CTA is preferred over MRI and MRA because of its performance in the diagnosis of internal derangement of the shoulder [29], associated with its relative availability and lower cost.
To our knowledge no study has evaluated the performance of CTA for the diagnosis of AC. The purpose of our study was to determine whether MRI and MRA signs of AC described in the literature are applicable to shoulder CTA and to assess their diagnostic performance.
2. Methods
2.1. Patients and control subjects
From October 2012 to August 2013 350 shoulder CTA were performed in our institution. Medical records were analysed and 19 patients (6 men, 13 women, mean age 52.6 years) with a final diagnosis of AC and 83 control subjects were included in this retrospective IRB (institutional review board) approved study.
Inclusion criteria were based on arthrographic findings with typical clinical features documented in medical records. Arthrographic criteria were a volume of contrast medium less than 8 ml injected in the glenohumeral joint with resistance and pain during injection. Clinical features were severe pain for more than one month, limitation of passive and active ranges of glenohumeral mobility (with clinical long term follow up: at least 2 years, until August 2015). Patients without clinical sign of retractile capsulitis, previous shoulder surgery, rheumatoid arthritis, or seronegative spondyloarthropathy were excluded.
Three patients had bilateral AC, so the patient group was composed of 22 shoulders (8 right, 14 left side). One orthopaedist or one rheumatologist referred all patients for shoulder CTA to confirm the diagnosis of AC and proceed to intraarticular steroid injection during arthrography and exclude associate pathology.
The control group consisted of 83 shoulders CTA (41 right and 42 left side, 44 men, 39 woman, mean age 53.4 years) referred during the same period for painful shoulder. We excluded patients presenting clinical signs of AC, or with less than 10 ml of contrast medium injected during the arthrography. The control group was split into 4 subgroups according to the final radiologic diagnosis: 1) normal shoulder (N = 20, 13 men, 7 women, mean age 45.6 years), 2) omarthrosis (N = 19, 8 men, 11 women, mean age 66.6 years), 3) labral injury (N = 23, 8 men, 15 women, mean age 62.0 years) and 4) rotator cuff tear (N = 21, 15 men, 6 women, mean age 39.4 years).
2.2. CTA imaging protocol
All patients were examined with the same protocol: Patient was in supine position with the shoulder stabilized by a small sand pack in neutral position. Joint injection was performed under fluoroscopic guidance with a 20 Gauge needle using an anterior approach. Intraarticular position was confirmed by injection of Linisol® 2% (B. Braun, Melsungen, Germany), followed by 10 ml of the contrast media Hexabrix® (Guerbet, Villepinte, France).
MDCT data were acquired with the patient in supine position, the examined shoulder laying along the body in neutral rotation as close as possible to the centre of the gantry. The contralateral shoulder was elevated and placed above the patient head when possible in order to decrease beam attenuation and limit radiation exposure. The thyroid was excluded from the volume acquisition. Spiral CT was performed less than 40 min after joint filling with contrast media using a 64-detector helical CT scanner (SOMATOM® Definition AS Siemens Healthcare, Erlangen, Germany). After a frontal projection scout image, a 10 s scanning was performed to image the volume from the top of the acromioclavicular joint to the lower margin of the axillary recess of the glenohumeral joint. Acquisition parameters were CNR (contrast-to-noise ratio) and dose optimized kV settings (CARE kv software, Siemens Healthcare, Erlangen, Germany), 120 kV, 350 mAs, focal spot size of 0.8 × 1.2 mm, 12 mm collimation beam, 25 cm field of view, effective pitch of 0.6, effective thickness of about 0.75 mm, and 512 × 512 matrix. Images were reconstructed using a three-dimensional cone-beam back projection algorithm, a high frequency kernel, an increment of 0.6 mm (50% section overlap), and a zoom factor of 1.2. Mean DLP (Dose-Length Product) was 90 mGy.cm
2.3. Images analysis
CT images were reviewed in consensus by two musculoskeletal radiologists with 5 and 1 year of clinical experience on high-resolution monitors using a picture archiving and communication system (PACS®, Carestream, Rochester, USA) station. Examination of the AC patients and the control patients were mixed, and display as an anonymised worklist on the PACS®. Both radiologists were blinded to the clinical and demographic data. Images were analysed using multiplanar reformations (MPR) for coronal oblique and sagittal oblique planes (Fig. 1a and b).
Fig. 1.
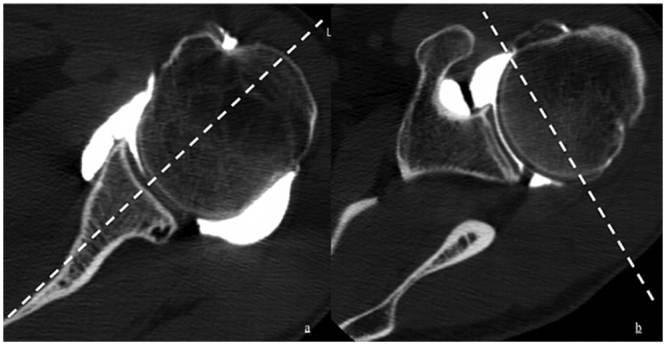
a and b. Axial view of the glenohumeral joint with demonstration of the coronal oblique (a) and sagittal oblique (b) planes at the level of the coronoid process tip.
First, qualitative analysis was performed. Humeral bone was assessed and considered either as normal or pathologic if subchondral resorption was present. The RI was evaluated in an oblique sagittal plan, perpendicular to the glenohumeral joint at the level of the tip of the coracoid process (Fig. 1b). RI was defined as the space between the subscapularis tendon anteriorly, the supraspinatous tendon supero-posteriorly, the coracohumeral ligament sitting above and the glenoid bone lying inferiorly. RI was considered as free if some fat contained in the RI had the same density as subcutaneous fat (Fig. 2), or obliterated if fat was no more visible.
Fig. 2.
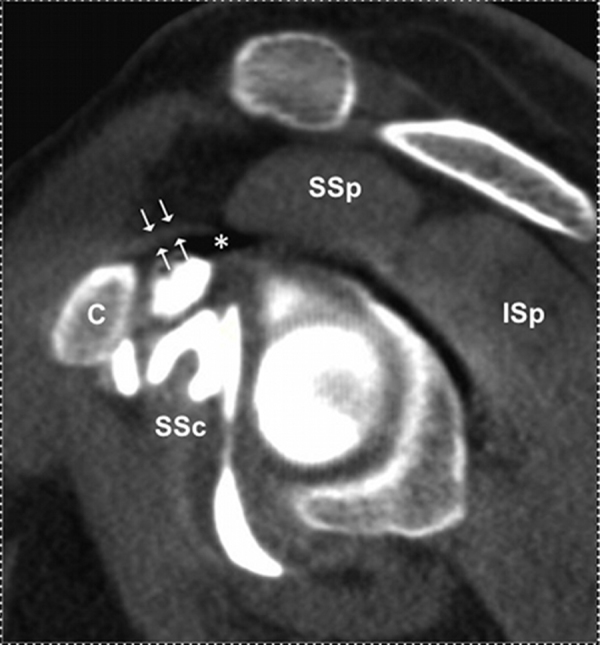
55-year-old patient addressed for suspicion of cuff rupture, with a normal CT arthrography. Sagittal oblique reconstruction at the level of the coracoid tip (c). The rotator interval (*) is free with the coracohumeral ligament lying over (white arrow).
* = rotator interval; c = coracoid tip; SSp = supraspinatus muscle; SSc = subscapularis muscle.
Second, quantitative measurements were realised. The RI width was measured in the same oblique sagittal plane: width was defined as the shortest distance between the anterior border of the subscapularis and the superior border of the supraspinatus muscles respectively. Coracohumeral ligament thickness was measured on a sagittal oblique plane close to his coracoid insertion (Fig. 2). The thicknesses of the lateral and medial wall of the axillary recess were measured on a coronal oblique plane perpendicular to the glenoid (Fig. 1a), and defined as the distance between the fat plane and the articular contrast at the widest portion of the axillary recess [14], [16]. The axillary recess width was defined as the greatest distance between the inner aspect of his lateral and medial walls on the coronal oblique plane (Fig. 3). Subcoracoid space was not analysed because of its poor visibility.
Fig. 3.
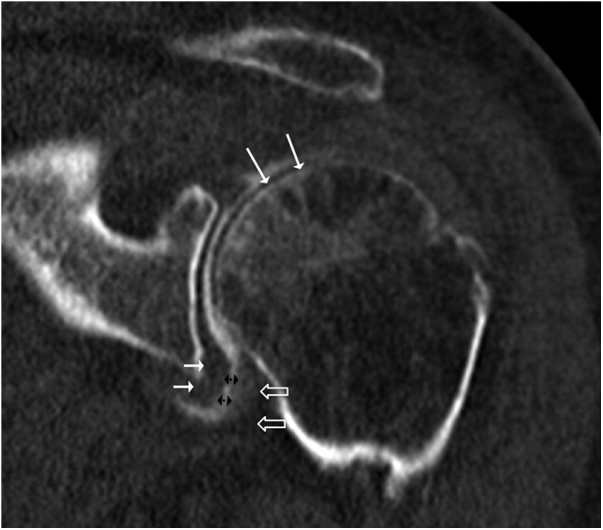
CTA coronal oblique reconstruction of a 62-year-old patient with adhesive capsulitis reveals the subchondral bone resorption of the humeral head (long white arrow→), the thickened capsule (empty arrow〉) and synovium (short white arrow→), and the narrow axillary recess (double black arrows).
↔ axillary pouch width; white arrow = medial wall of the axillary pouch, black empty arrow = lateral wall of the axillary pouch.
2.4. Statistical analysis
The data were processed using a statistical software package (SAS ® software, version 9.2; Cary, NC HQ). P-values < 0.05 were considered to be statistically significant. The Wilcoxon rank-sum test was used to compare groups in the quantitative analysis, while the Fisher test was used in the qualitative analyses.
Using ROC curves (supplementary material), we determined cut-off values for qualitative and quantitative criteria of AC. In the absence of a reference standard, we considered the control group with a normal CTA (subgroup “normal”) as our reference standard, calculating the sensitivity and specificity of each qualitative and quantitative criteria of AC.
3. Results
3.1. Qualitative criteria
Results of qualitative criteria for patient and control groups are shown in Table 1. Humeral bone was pathological in 7/22 patients with AC (31.8% vs. none in the control groups, p < 0.0001) with a sensitivity of 31.8% and a specificity of 100%. RI obliteration was statistically significantly more frequent in patients with AC (72.7%; 16/22) vs. 12% (10/83) in the control groups, p < 0.0001). Among those patients with RI obliteration (12%; 10/83) in the control group, 5 had a normal shoulder, 3 had omarthrosis, none had labral injury and 2 had a rotator cuff injury. Sensitivity for RI obliteration in AC was 72.2% and specificity 75%.
Table 1.
Qualitative values for patient and control groups.
| Humeral bone |
RI obliteration |
|||
|---|---|---|---|---|
| Normal | Pathologic | No | Yes | |
| Group | ||||
| Patient (AC) | 15 (68.2%) | 7 (31.8%)* | 6 (27,3%) | 16 (72,7%)* |
| Control | 83 (100%) | 0 (0%)* | 73 (87.6%) | 10 (12.4%)* |
| Normal | 20 (100%) | 0 (0%) | 15 (75%) | 5 (25%) |
| Omarthrosis | 19 (100%) | 0 (0%) | 16 (84.2%) | 3 (15.8%) |
| Labral injury | 21 (100%) | 0 (0%) | 21 (100%) | 0 (0%) |
| Rotator cuff cuRotator ff tear | 23 (100%) | 0 (0%) | 21 (91.3%) | 2 (8.7%) |
AC = adhesive capsulitis.
p-value < 0.0001.
3.2. Quantitative criteria
Results of quantitative criteria with mean values ± standard deviation for patient and control groups are shown in Table 2. The width of the RI was not statistically significantly increased in the AC group in comparison with the control groups (mean value ± SD, 16.5 ± 2.4 mm vs. 15.9 ± 3.8 mm, p ≥ 0.428). Coracohumeral ligament thickness was statistically significantly increased in the AC group in comparison with the control groups (mean value ± SD, 4.1 ± 1 mm vs. 2.5 ± 0.6 mm, p ≤ 0.0001).
Table 2.
Quantitative measures for patient and control groups.
| N |
Criteria |
|||||
|---|---|---|---|---|---|---|
| Group | Axillary recess width (mm) Mean ± SD |
Lateral wall thickness (mm) Mean ± SD | Medial wall thickness (mm) Mean ± SD | RI width (mm) Mean ± SD |
CHL thickness (mm) Mean ± SD |
|
| AC | 22 | 4.6 ± 2.6* | 5.7 ± 1.0* | 5.9 ± 1.3* | 16.5 ± 2.4 | 4.1 ± 1.0* |
| Control | 83 | 9.9 (4,6) | 3.5 (1.3) | 3.7 (1.1) | 15.9 (3.8) | 2.5 (0.6) |
| Normal | 20 | 9.8 ± 3.8 | 4.2 ± 1.0 | 3.8 ± 1.1 | 15.3 ± 2.9 | 2.7 ± 0.6 |
| Omarthrosis | 19 | 10.3 ± 4.6 | 4.0 ± 1.6 | 4.0 ± 1.0 | 17.4 ± 5.9 | 2.5 ± 0.6 |
| Labral injury | 20 | 10.6 ± 4.1 | 3.3 ± 0.8 | 3.8 ± 1.0 | 14.8 ± 3.1 | 2.6 ± 0.5 |
| Rotator cuff tear | 23 | 8.9 ± 5.6 | 2.7 ± 1.1 | 3.1 ± 0.9 | 16.3 ± 2.2 | 2.3 ± 0.6 |
AC = adhesive capsulitis; RI = rotator interval; CHL = coracohumeral ligament.
p-value < 0.0001.
The width of the axillary recess of patients with AC was statistically significantly inferior in comparison with the control groups (mean value ± SD, 4.6 ± 2.6 mm vs. 9.9 ± 4.6 mm, p ≤ 0.001). The width of the axillary recess of the control group “normal” was 9.8 mm ± 3.8 mm (mean value ± SD). The lateral and the medial wall thicknesses of the axillary recess were significantly increased in the patients with AC in comparison with the control groups (mean value ± SD, 5.7 ± 1 mm and 5.9 ± 1.3 mm respectively, p ≤ 0.001). Lateral and medial wall thicknesses of the axillary recess of the control group “normal” were 4.2 mm ± 1 mm and 3.8 ± 1.1 mm (mean value ± SD).
Sensitivity and specificity results of cut-off values for quantitative criteria are shown in Table 3. We obtained a cut-off value of 7.1 mm for the axillary recess width, with a sensitivity of 84% and a sensitivity of 80%. For the lateral and the medial wall thicknesses of the axillary recess, respective cut-off values were 4.9 mm and 4 mm, with sensitivities and specificities of 81% and 85%, and 100% and 75% respectively. Coracohumeral ligament thickness cut-off value was 3.2 mm with a sensitivity of 90% and a specificity of 75%. Cut-off value for RI width was 15.7 cm with sensitivity and specificity both of 68%.
Table 3.
Cut-off values of quantitative criteria for CTA.
| Criteria | Cut-off value (mm) | Sensitivity (%) | Specificity (%) |
|---|---|---|---|
| Axillary width | 7.1 | 84% | 80% |
| Lateral wall thickness | 4.9 | 81% | 85% |
| Medial wall thickness | 4 | 100% | 75% |
| RI width | 15.7 | 68% | 68% |
| CHL thickness | 3.2 | 90% | 75% |
4. Discussion
The present study demonstrates that some MRI and MRA signs of AC, those concerning changes of the axillary recess, are applicable to CTA. The width of the axillary recess is significantly decreased and the thicknesses of the lateral and to a lesser extent the medial wall of the axillary recess are significantly increased in AC patients in comparison with the control group. The humeral bone is pathological only in patients with AC. Evaluation of rotator interval seems useful and reproducible only for obliteration, as decrease of his width is not significantly different in the patient group in comparison with the control group, and therefore are not applicable to CTA.
Narrow axillary recess is a well-recognized feature of AC in arthrography that have been later describe for MRA and MRI [16], [19], [20]. We found a significant decrease in axillary width in comparison to all the controls groups. Measuring in a same plane coronal oblique on MRA, Jung and al [16] found a value for axillary width of 4.3 mm ± 3.2 mm in the patient group and 9.3 mm ± 2.5mmin the control group. Lee et al. showed that diminished filling ratio of the axillary recess to posterior joint cavity was useful to differentiate AC patients and controls (filling ratio of 0.51 vs. 0.82 respectively) [19]. Mengiardi and al [20] measured in preoperative MRA the articular recess in three dimensions, calculating a volume that was also significantly smaller in AC (mean, 0.53 vs. 0.88 ml, p = 0.03).
We found that the thickness of lateral and to a lesser extent the medial wall of the axillary recess were significantly increased in AC. Our findings confirmed previous work with MRA, showing a thickening of both aspect of the joint capsule of 3 mm: Jung and al [16] obtained a sensitivity of 79%–93% and a specificity of 86%-100% on oblique coronal T2. Emig and al [14] and Connell and al [15] found a thickening axillary pouch of >4 mm on MR, with a sensitivity of 70% and a specificity of 98% in Emig study. Lefevre-Colau and al [30] showed a good interobserver agreement for the thickness of the articular capsule at the axillary recess. According to Watson and al [22], arthroscopic inflammatory changes occur in the posterior and anterior recesses with obliteration secondary to scar tissue deposition [24], [31]. However, in the literature the association of a narrow axillary recess to a thickened joint capsule is not always recognized. Indeed, Mengiardi and al [20] demonstrated that although the volume of the axillary recess was significantly smaller in patients than in control subjects; neither the lateral nor the medial wall of the articular capsule at the axillary recess was increased. Moreover, Manton et al. [13] reported in a study including only 9 patients, that not only axillary recess volume but also the thickness of his walls on both side were inconclusive MRA signs for distinguishing shoulders with AC from those without.
The rotator cuff interval is known to be important in the motion of the glenohumeral joint, and has been implicated in the pathogenesis of AC [21], [23], [32]. Abnormal signal and enhancement of the RI have been described with MRI [13], [15], [20] whereas Jung and al [16] described the presence of abnormal tissue in RI. In our study, RI obliteration was statistically significantly more frequent in patients with AC than in the control groups with sensitivity of 72.2% and a specificity of 75%.
The RI width is also considered as decreased in AC at MRA [16], [7] and MRI [14]. Jung and al [16] found a significant decrease with a mean RI width at the coracoids tip in sagittal-oblique T1-weight images of 10.2 mm in the patient group compared to 13.4 mm in the control group (P = 0.012). Using the same measure, Kim and al [17] had a significant decrease in AC with 12.75 mm compared to 15.44 mm in control group, based on standardised values established from patient features, as volume of the joint capsule vary widely from patient to patient depending on the clinical circumstances. In contrast, Lee and al [19] did not find a statistically significant difference in the mean width of the RI between patients with AC and controls (7.45 vs. 8.48 mm). In our series, RI tended to be similar or larger than the mean RI width of all CTA (AC and control groups together). The smaller values for RI width were found in the control group with rotator cuff tears.
Concerning the coracohumeral ligament, we found a significantly thicker CHL in the AC group. Mengiardi et al. have shown that CHL was thicker in the AC patients than in control patient (cut-off value of >4.1 mm vs. 2.7 mm, p > 0.001) with a specificity of 95% and sensitivity of 59% [20]. However this structure is not always well identified on MRI. In the study by Blum et al. [18], CHL thickness was not measurable in 6 and 8 (reader 1 and 2 respectively) out of 68 unenhanced MRI. In our study, the contrast resolution of CT arthrography did not allow the accurate measurement of the CHL thickness. This fine tubular structure is difficult to individualize when the surrounding fat plane is infiltrated and can be confounded with the articular capsule or the coracoacromial ligament.
This study has a few limitations. First, we excluded patients who were diagnosed with AC based on a clinical basis only in whom arthrography showed a normal joint volume, leading to a small number of patients in our pathological group. However, despite limited statistical power, we showed statistically significant difference between patients and controls, for some of the imaging signs of AC previously described, applied to CTA. Second, we calculated cut-off values with sensitivity and specificity to evaluate the diagnostic performance of CTA, based on a clinico-arthrographic gold standard. However, we did not obtain a clinical follow-up or an arthroscopic correlation to confirm the diagnosis. Third, our control group was not composed of asymptomatic volunteers but of subjects referred for shoulder pain who did not fulfil any diagnostic criteria for AC. Our population may however be representative of patients in whom CTA of the shoulder is indicated, and from which AC need to be distinguished.
Imaging can play a role in the diagnostic work-up of AC for challenging cases, and it may affect therapeutic decisions. In our experience, CTA represents a valuable and more accessible imaging modality to MRI and MRA, with short examination time and patient irradiation kept as low as possible to provide sufficient diagnostic quality imaging [29].
In conclusion, previously described signs of AC including decreased width of the axillary recess, thickening of the lateral wall and to a lesser extent thickening of the medial wall of the joint capsule at the axillary recess can serve as diagnostic signs of AC at CTA. On the contrary, the assessment of the rotator interval seems useful and reproducible only for obliteration.
Conflicts of interest
None of the authors has any conflicts of interest to declare.
References
- 1.Carette S., Moffet H., Tardif J., Bessette L., Morin F., Fremont P. Intraarticular corticosteroids, supervised physiotherapy, or a combination of the two in the treatment of adhesive capsulitis of the shoulder: a placebo-controlled trial. Arthritis Rheum. 2003;48(3):829–838. doi: 10.1002/art.10954. [DOI] [PubMed] [Google Scholar]
- 2.Neviaser A.S., Hannafin J.A. Adhesive capsulitis: a review of current treatment. Am. J. Sports Med. 2010;38(11):2346–2356. doi: 10.1177/0363546509348048. [DOI] [PubMed] [Google Scholar]
- 3.Marx R.G., Malizia R.W., Kenter K., Wickiewicz T.L., Hannafin J.A. Intra-articular corticosteroid injection for the treatment of idiopathic adhesive capsulitis of the shoulder. HSS J. 2007;3(2):202–207. doi: 10.1007/s11420-007-9044-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Le Lievre H.M., Murrell G.A. Long-term outcomes after arthroscopic capsular release for idiopathic adhesive capsulitis. J. Bone Joint Surg. Am. 2012;94(13):1208–1216. doi: 10.2106/JBJS.J.00952. [DOI] [PubMed] [Google Scholar]
- 5.Dattani R., Ramasamy V., Parker R., Patel V.R. Improvement in quality of life after arthroscopic capsular release for contracture of the shoulder. Bone Joint J. 2013;95-B(7):942–946. doi: 10.1302/0301-620X.95B7.31197. [DOI] [PubMed] [Google Scholar]
- 6.Brue S., Valentin A., Forssblad M., Werner S., Mikkelsen C., Cerulli G. Idiopathic adhesive capsulitis of the shoulder: a review. Knee Surg. Sports Traumatol. Arthroscopy. 2007;15(8):1048–1054. doi: 10.1007/s00167-007-0291-2. [DOI] [PubMed] [Google Scholar]
- 7.Bamji A.N., Erhardt C.C., Price T.R., Williams P.L. The painful shoulder: can consultants agree? Br. J. Rheumatol. 1996;35(11):1172–1174. doi: 10.1093/rheumatology/35.11.1172. [DOI] [PubMed] [Google Scholar]
- 8.de Winter A.F., Jans M.P., Scholten R.J., Deville W., van Schaardenburg D., Bouter L.M. Diagnostic classification of shoulder disorders: interobserver agreement and determinants of disagreement. Ann. Rheum. Dis. 1999;58(5):272–277. doi: 10.1136/ard.58.5.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Robinson D., Halperin N., Agar G., Alk D., Rami K. Shoulder girdle neoplasms mimicking frozen shoulder syndrome. J. Shoulder Elbow Surg./Am. Shoulder Elbow Surgeons. 2003;12(5):451–455. doi: 10.1016/s1058-2746(03)00092-2. [DOI] [PubMed] [Google Scholar]
- 10.Walmsley S., Osmotherly P.G., Rivett D.A. Clinical identifiers for early-stage primary/idiopathic adhesive capsulitis: are we seeing the real picture? Phys. Ther. 2016;94(7):968–976. doi: 10.2522/ptj.20130398. [DOI] [PubMed] [Google Scholar]
- 11.Neviaser J.S. Arthrography of the shoulder joint: study of the findings in adhesive capsulitis of the shoulder. Study of the findings in adhesive capsulitis of the shoulder. J. Bone Joint Surg. Am. 1962;44-A:1321–1359. [PubMed] [Google Scholar]
- 12.Leppälä J., Kannus P., Sievänen H., Järvinen M., Vuori I. Adhesive capsulitis of the shoulder (frozen shoulder) produces bone loss in the affected humerus, but long term bony recovery is good. Bonne. 1998;22(6):691–694. doi: 10.1016/s8756-3282(98)00049-0. [DOI] [PubMed] [Google Scholar]
- 13.Manton G.L., Schweitzer M.E., Weishaupt D., Karasick D. Utility of MR arthrography in the diagnosis of adhesive capsulitis. Skeletal Radiol. 2001;30(6):326–330. doi: 10.1007/s002560100326. [DOI] [PubMed] [Google Scholar]
- 14.Emig E.W., Schweitzer M.E., Karasick D., Lubowitz J. Adhesive capsulitis of the shoulder: MR diagnosis. AJR Am. J. Roentgenol. 1995;164(6):1457–1459. doi: 10.2214/ajr.164.6.7754892. [DOI] [PubMed] [Google Scholar]
- 15.Connell D., Padmanabhan R., Buchbinder R. Adhesive capsulitis: role of MR imaging in differential diagnosis. Eur. Radiol. 2002;12(8):2100–2106. doi: 10.1007/s00330-002-1349-7. [DOI] [PubMed] [Google Scholar]
- 16.Jung J.Y., Jee W.H., Chun H.J., Kim Y.S., Chung Y.G., Kim J.M. Adhesive capsulitis of the shoulder: evaluation with MR arthrography. Eur. Radiol. 2006;16(4):791–796. doi: 10.1007/s00330-005-0020-5. [DOI] [PubMed] [Google Scholar]
- 17.Kim K.C., Rhee K.J., Shin H.D. Adhesive capsulitis of the shoulder: dimensions of the rotator interval measured with magnetic resonance arthrography. J. Shoulder Elbow Surg./Am. Shoulder Elbow Surgeons. 2009;18(3):437–442. doi: 10.1016/j.jse.2008.10.018. [DOI] [PubMed] [Google Scholar]
- 18.Gondim Teixeira P.A., Balaj C., Chanson A., Lecocq S., Louis M., Blum A. Adhesive capsulitis of the shoulder: value of inferior glenohumeral ligament signal changes on T2-weighted fat-saturated images. AJR Am. J. Roentgenol. 2012;198(6):W589–96. doi: 10.2214/AJR.11.7453. [DOI] [PubMed] [Google Scholar]
- 19.Lee M.H., Ahn J.M., Muhle C., Kim S.H., Park J.S., Kim S.H. Adhesive capsulitis of the shoulder: diagnosis using magnetic resonance arthrography, with arthroscopic findings as the standard. J. Comput. Assist. Tomogr. 2003;27(6):901–906. doi: 10.1097/00004728-200311000-00012. [DOI] [PubMed] [Google Scholar]
- 20.Mengiardi B., Pfirrmann C.W., Gerber C., Hodler J., Zanetti M. Frozen shoulder: MR arthrographic findings. Radiology. 2004;233(2):486–492. doi: 10.1148/radiol.2332031219. [DOI] [PubMed] [Google Scholar]
- 21.Ozaki J., Nakagawa Y., Sakurai G., Tamai S. Recalcitrant chronic adhesive capsulitis of the shoulder: role of contracture of the coracohumeral ligament and rotator interval in pathogenesis and treatment. J. Bone Joint Surg. Am. 1989;71(10):1511–1515. [PubMed] [Google Scholar]
- 22.Watson L., Dalziel R., Story I. Frozen shoulder: a 12-month clinical outcome trial. J. Shoulder Elbow Surg./Am. Shoulder Elbow Surgeons. 2000;9(1):16–22. doi: 10.1016/s1058-2746(00)90004-1. [DOI] [PubMed] [Google Scholar]
- 23.Wiley A.M. Arthroscopic appearance of frozen shoulder. Arthroscopy. 1991;7(2):138–143. doi: 10.1016/0749-8063(91)90098-i. [DOI] [PubMed] [Google Scholar]
- 24.Tamai K., Akutsu M., Yano Y. Primary frozen shoulder: brief review of pathology and imaging abnormalities. J. Orthop. Sci. 2014;19(1):1–5. doi: 10.1007/s00776-013-0495-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Loyd J.A., Loyd H.M. Adhesive capsulitis of the shoulder: arthrographic diagnosis and treatment. South. Med. J. 1983;76(7):879–883. doi: 10.1097/00007611-198307000-00016. [DOI] [PubMed] [Google Scholar]
- 26.Omoumi P., Bafort A.C., Dubuc J.E., Malghem J., Vande Berg B.C., Lecouvet F.E. Evaluation of rotator cuff tendon tears: comparison of multidetector CT arthrography and 1.5-T MR arthrography. Radiology. 2012;264(3):812–822. doi: 10.1148/radiol.12112062. [DOI] [PubMed] [Google Scholar]
- 27.Omoumi P., Mercier G.A., Lecouvet F., Simoni P., Vande Berg B.C. CT arthrography, MR, arthrography, PET, and scintigraphy in osteoarthritis. Radiol. Clin. North Am. 2009;47(4):595–615. doi: 10.1016/j.rcl.2009.04.005. [DOI] [PubMed] [Google Scholar]
- 28.Lecouvet F.E., Dorzee B., Dubuc J.E., Vande Berg B.C., Jamart J., Malghem J. Cartilage lesions of the glenohumeral joint: diagnostic effectiveness of multidetector spiral CT arthrography and comparison with arthroscopy. Eur. Radiol. 2007;17(7):1763–1771. doi: 10.1007/s00330-006-0523-8. [DOI] [PubMed] [Google Scholar]
- 29.Lecouvet F.E., Simoni P., Koutaissoff S., Vande Berg B.C., Malghem J., Dubuc J.E. Multidetector spiral CT arthrography of the shoulder Clinical applications and limits, with MR arthrography and arthroscopic correlations. Eur. J. Radiol. 2008;68(1):120–136. doi: 10.1016/j.ejrad.2008.02.025. [DOI] [PubMed] [Google Scholar]
- 30.Lefevre-Colau M.M., Drape J.L., Fayad F., Rannou F., Diche T., Minvielle F. Magnetic resonance imaging of shoulders with idiopathic adhesive capsulitis: reliability of measures. Eur. Radiol. 2005;15(12):2415–2422. doi: 10.1007/s00330-005-2830-x. [DOI] [PubMed] [Google Scholar]
- 31.Carrillon Y., Noel E., Fantino O., Perrin-Fayolle O., Tran-Minh V.A. Magnetic resonance imaging findings in idiopathic adhesive capsulitis of the shoulder. Rev. Rhum. Engl. Ed. 1999;66(4):201–206. [PubMed] [Google Scholar]
- 32.Ha'eri G.B., Maitland A. Arthroscopic findings in the frozen shoulder. J. Rheumatol. 1981;8(1):149–152. [PubMed] [Google Scholar]


