Abstract
The exposure pathways of perfluoroalkyl acids (PFAAs) to humans are still not clear because of the complex living environment, and few studies have simultaneously investigated the bioaccumulative behaviour of different PFAAs in humans. In this study, serum, dust, duplicate diet, and other matrices were collected around a manufacturing plant in China, and homologous series of PFAAs were analysed. PFAA levels in dust and serum of local residents in this area were considerably higher than those in non-polluted area. Although dietary intake was the major exposure pathway in the present study, dust ingestion played an important role in this case. Serum PFAAs in local residents was significantly correlated with dust PFAAs levels in their living or working microenvironment. Serum PFAAs and dust PFAAs were significantly higher in family members of occupational workers (FM) than in ordinary residents (OR) (p < 0.01). After a careful analysis of the PFAAs exposure pathway, a potential pathway in addition to direct dust ingestion was suggested: PFAAs might transferred from occupational worker's clothes to dinners via cooking processes. The bioaccumulative potential of PFHxS and PFOS were higher than other PFAAs, which suggested a substantial difference between the bioaccumulative ability of perfluorinated sulfonic acids and perfluorinated carboxylic acids.
Perfluoroalkyl acids (PFAAs) have been extensively used as surfactants, lubricants, stain- and water-resistant coatings, and fire-resistant foams, due to their unique properties1. Perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) are the most widely distributed PFAAs in the world, and they have attracted increasing concern in the past decade2,3,4. The total historical global production of perfluorooctane sulfonyl fluoride (POSF) reached approximately 96,000 tonnes (t) and approximately 45250 t of POSF was emitted into the environment from 1970 to 20125. The primary PFOS producer, 3 M, ceased the production of POSF in 2002. In addition, the production and usage of PFOS, its salts and POSF were restricted by the Stockholm Convention in 20096. However, the production increased rapidly in China in recent years7. PFAAs with a fluorinated chain longer than seven are generally considered bioaccumulative, extremely persistent, and have been found to be ubiquitous in the environment3,8.
PFAAs have been detected in human blood worldwide, and in certain cases, their levels were even higher than those of other persistent organic pollutants9,10. Recently, the adverse effects to humans associated with PFAAs body burden have been reported, such as osteoarthritis and a later age of puberty11,12. Humans can be exposed to PFAAs through various pathways13,14,15, but the exposure pathways and bioaccumulative behaviour in humans are still not clear16,17. Previous studies considered that diet was the primary source15,18,19; however, there might be other significant exposure pathways for PFAAs beside diet20. Vestergren et al. reported that the ingestion of house dust was the second most important pathway in the high-exposure scenario21, and Beeson et al. ascribed the exceptionally high serum concentrations of some PFAAs in a Canadian family to dust ingestion and/or inhalation17.
Our previous study investigated the transportation of PFAAs from the manufacturing facility to the surrounding environment and elevated perfluorohexanesulfonate (PFHxS), PFOS, and PFOA levels in soil and biota samples were found within three kilometres of the plant22. Although PFOS production has been restricted by the Stockholm Convention6, there is still a usage exemption of PFOS for specific purposes. Thus, the production of PFOS and related products in this particular facility did not immediately halt. The purpose of the present study was to (I) evaluate PFAAs body burden of local residents around this fluorochemical manufacturing facility and to identify the exposure pathways of PFAAs in this area further and (II) to investigate bioaccumulation behaviours of PFAAs with different carbon chain lengths and functional groups in humans. Currently, this paper is the first that intensively studies the exposure pathway of PFAAs to residents near a point source.
Methods
Experimental Design and Sampling
A fluorochemical manufacturing plant located in the Hubei province of China was selected as the sampling area. The main synthesis method for PFAAs in this plant is by the electrochemical fluorination process. The sampling procedure was performed in December 2011 and 2012. Fifty-eight local residents who lived around this plant within a distance of 3 km were recruited in the present study. The gender, age, weight, occupation, dietary habits, and other information were collected by questionnaires; the demographic information of studied population is listed in Table S1. In this study, we collected blood samples of each participant to evaluate the PFAAs body burden in humans. According to whether they had family members working in the fluorochemical plant, our studied population, the local residents, were divided into two groups: family members of occupational workers (FM) and ordinary residents (OR). All the experiments (including design and execution) and sampling procedures were carried out in accordance with the guidelines approved by the Medical Research Ethics Committee, School of Medicine, Jianghan University, and all volunteers gave their written informed consent to participate in this study.
Furthermore, to investigate the exposure pathways of PFAAs to residents around the manufacturing facility, relevant exposure matrices (such as dust, Total Suspended Particle (TSP), and duplicate diet) from their living environment were also collected. Dust and TSP samples were collected from the plant and the corresponding participants' residential environment. Duplicate diet and drinking water samples were directly collected from the table and tap from the home of participants. In addition, the work clothes of workers and local residents were also collected. A total of 141 samples, including serum (n = 58), dust (n = 38), TSP (n = 15), drinking water (n = 6), duplicate diet (n = 9) and clothes (n = 15), were collected in this study. The detailed information of these samples (such as sampling sites, number of samples, and grouping of samples) is listed in Table 1, and the spatial distribution of the sampling sites and detailed sampling procedures can be found in the Supplementary Information (Figure S1).
Table 1. Overview of sampling information.
| Sampling site | Serum | Dust | TSPa | DWb | DDc | Clothes | |
|---|---|---|---|---|---|---|---|
| Plant | 8 | 7 | 5 | ||||
| (Dust-P)d | (TSP-P)e | ||||||
| Around Plant | 58 | 30 | 8 | 6 | |||
| (Dust-A)f | (TSP-A)g | ||||||
| FMh | 32 | 9 | 4 | 3 | 5 | ||
| ORi | 26 | 21 | 4 | 3 | 4 | 10 |
aTSP: Total Suspended Particle.
bDW: Drinking Water.
cDD: Duplicate Diet.
dDust-P: dust samples collected in the plant.
eTSP-P: TSP samples collected in the plant.
fDust-A: dust samples collected around the plant.
gTSP-A: TSP samples collected around the plant.
hFM: Family member of workers.
iOR: Ordinary residents.
Chemical Analysis, Quality Assurance, and Quality Control
Eleven PFAAs (perfluoro-n-pentanoic acid (PFPeA), perfluorohexanoic acid (PFHxA), perfluoroheptanoic acid (PFHpA), PFOA, perfluorononanoic acid (PFNA), perluorodecanoic acid (PFDA), perfluorododecanoic acid (PFDoDA), perfluoroundecanoic acid (PFUnDA), salts of perfluorobutanesulfonate (PFBS), PFHxS, and PFOS were determined by HPLC-ESI-MS/MS. Sample pretreatment, instrumental analysis, quality assurance, and quality control of the analytical method are based on our previous work22,23,24. The detailed pre-treatment of different types of samples, method detection limit (LOD), quality assurance, and quality control are described in the supplementary information.
Estimated Daily Intake Through Different Pathways and Methods
The daily dietary intake of PFAAs is estimated by the sum of PFAAs in the ingested diet and water. Diet and drinking water consumption are set at 2500 g and 1200 ml per day, respectively. Thus, the daily dietary intake of PFAAs is calculated as equation (1):
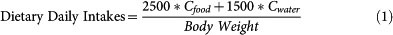 |
Cfood represents concentration of PFAAs in food (ng/g) and Cwater stands for concentration of PFAAs in drinking water (ng/mL). According to the questionnaires, the average body weight of local residents was 60 kg (Table S1). The non-dietary exposure of PFAAs was calculated as the sum of dust ingestion and TSP inhalation. The PFAAs daily intake through dust ingestion (DIdust) was calculated through the following equation: DIdust = Cdust × Adust/body weight, where Cdust is the concentration of PFAAs in house dust, Adust is the average amount of the daily dust ingestion, and the mean gastrointestinal uptake of dust is set at 50 mg/day for adults according to the US EPA exposure factors handbook25. For the calculation of TSP intake, the following equation was applied: DITSP = CTSP*Vair/body weight, where CTSP is the concentrations of PFAAs in TSP, Vair is the daily air volume inhaled by adults and was set at 16.0 m3 25. To be consistent with previous studies of PFAAs exposure assessment, we assumed 100% absorption of PFAAs intake through ingestion and inhalation15,26. Human exposure to PFAAs through dermal absorption of dust was neglected in this study because only a small fraction (0.048%) of the dose could be absorbed through the skin27.
Furthermore, based on a simplified one compartment pharmacokinetic model, we estimated the total estimated daily intake (TEDI) of humans by serum PFAA levels. The model was developed by Andersen et al. and Thompson et al.28,29, and it have been successfully used in several populations including a Chinese population30,31,32. Briefly, PFAAs levels in the serum are a result of exposure dose (E) and the change of PFAAs levels could be described as equation(2):
 |
where k is the first-order rate constant for PFAAs elimination per day (k = 0.693/t1/2, and Olsen et al. estimated that the t1/2 for PFOA and PFOS were 1257 and 1751 days, respectively, in humans33). Vd is the volume of distribution (mL/kg bw). According to previous studies, Vd of PFOA and PFOS were set at 170 and 230 mL/kg bw, respectively28,34. Cp is the PFAA concentrations in serum. If the serum PFAAs reached steady-state conditions, dCp/dt = 0. Thus, the former equation could be simplified as equation (3):
 |
Results
PFPeA, PFHxA, PFNA, PFDA, PFDoDA and PFUnDA had low detection rates in all samples, and therefore, they were not included for further data analysis and discussion. PFBS, PFHxS, PFOS and PFOA were detected in all serum samples in local residents while PFHpA had a detection frequency of 67%. The average serum PFBS, PFHxS, PFOS, PFHpA, and PFOA concentrations were 22.7, 138, 445, 2.85 and 50.6 ng/mL, respectively. The detailed characteristics of PFAAs in resident demographic subgroups by gender and age (≤45 and >45 years) were provided in Table S2 through S4. The serum PFOS were significantly higher in males than females in local residents(p < 0.05). Compared with males, females have several distinct excretion routes of PFAAs such as menstruation, bearing, and lactation. For example, menstrual clearance rate is even comparable to that renal clearance rate35, these particular elimination pathway might result in the lower serum PFOS in females. No significant differences were observed by age in all PFAAs.
PFAAs in drinking waters were relatively low (Table S5), the main PFAAs in drinking waters were PFOS and PFOA and could only be detected in 34% and 56% of samples, respectively. The highest PFOS and PFOA level in drinking water were 3.04 and 2.08 ng/L, respectively, which were lower than that in drinking water reported in previous studies36,37,38. The result indicated that drinking water in this area was not contaminated by PFAAs. PFBS was the most prevalent PFAAs in duplicate diet samples, with an average of 7.02 ng/g ww, followed by PFOS and PFHxS (Table S6). The highest PFOS, PFHxS, and PFOA levels in diet samples were 3.47, 0.45, and 0.10 ng/g ww, with averages of 0.82, 0.19, and 0.06 ng/g ww, respectively. Compared with a study on duplicate diet monitoring in Germany, both the detected rates and levels were higher in this area39. The levels of PFAAs in TSP samples collected from the plant (TSP-P) were extremely high, and PFAAs levels decreased sharply from the point source. However, PFAAs levels in TSP samples collected around the plant (TSP-A) were much higher than that in Europe and Japan40,41, which suggested that the PFAAs might be spread to the surroundings by the TSP route (Table S7). PFOS, PFOA and PFHxS were the major compounds in dust samples, extremely high levels of PFAAs were detected in dust samples collected from the plant (Dust-P). The highest concentrations of PFOS, PFOA and PFHxS in Dust-P reached 3157, 253, and 257 μg/g of dry weight. In dust samples collected around the plant (Dust-A), PFOS, PFOA and PFHxS ranged from 2.68 to 18486 ng/g, 5.00 to 1230 ng/g, and 0.44 to 708 ng/g, with the average of 1189, 354 and 68.0 ng/g, respectively, which were almost three orders of magnitude lower than that in Dust-P.
The daily intake of PFAAs through dietary, dust ingestion, and TSP inhalation were calculated according to equation 1–3(Table 2). Daily dietary intakes of PFBS, PFHxS, PFOS, PFHpA, and PFOA for local residents were 270, 7.32, 31.2, 5.63 and 2.0 ng/day/kg bw, respectively. The DIdust for occupational workers reached 1679 ng/day/kg bw, and the DIdust of PFOS and PFOA were 604 and 759 ng/day/kg body, respectively. These values were almost three orders of magnitude higher than those estimated in previous studies18,26. The DIdust of PFOS, PFOA, and PFHxS of local residents was 0.991, 0.295, and 0.057 ng/day/kg body, respectively. PFAAs exposure via TSP inhalation was lower than dietary intake and dust ingestion in local residents (Table 2). According to Equation (3), the total estimated daily intake (TEDI) of PFOS and PFOA for local residents was 19.0 and 1.43 ng/day/kg body, respectively.
Table 2. Estimated Daily Intake (EDI) of PFAAs via Dust, TSP in this study and Total Estimated Daily Intake(TEDI) calculated by a simplified one compartment pharmacokinetic model (ng/day/kg body).
| OWa | LRb | FMc | ORd | |
|---|---|---|---|---|
| Dust ingestion (ng/day/kg body) | ||||
| PFBS | 921 | 1.64 | 4.09 | 0.593 |
| PFHxS | 554 | 0.057 | 0.145 | 0.013 |
| PFOS | 604 | 0.991 | 2.96 | 0.148 |
| PFHpA | 225 | 0.179 | 0.315 | 0.127 |
| PFOA | 759 | 0.295 | 0.581 | 0.158 |
| T- PFAAs | 1679 | 3.16 | 8.09 | 1.04 |
| TSP inhalation (ng/day/kg body) | ||||
| PFBS | 67.3 | 0.065 | 0.069 | 0.068 |
| PFHxS | 0.763 | 0.017 | 0.015 | 0.020 |
| PFOS | 4.39 | 0.061 | 0.058 | 0.069 |
| PFHpA | 33.3 | 0.120 | 0.053 | 0.210 |
| PFOA | 296 | 0.084 | 0.046 | 0.132 |
| T- PFAAs | 402 | 0.346 | 0.24 | 0.499 |
| Daily Dietary Intake (ng/day/kg body) | ||||
| PFBS | 444 | 270 | 444 | 52.1 |
| PFHxS | 10.0 | 7.32 | 10 | 3.94 |
| PFOS | 54.3 | 31.2 | 54.3 | 2.33 |
| PFHpA | 8.37 | 5.63 | 8.37 | 2.22 |
| PFOA | 2.26 | 2.01 | 2.26 | 1.70 |
| T- PFAAs | 519 | 316 | 519 | 62.3 |
| Total Estimated Daily Intake (ng/day/kg body) | ||||
| PFOS | 1000 | 19 | 32.9 | 9.34 |
| PFOA | 374 | 1.43 | 2.23 | 0.792 |
a:OW:occupational workers of the fluorochemical plant;
b:LR:local residents (include FM and OR);
c: FM: family members of occupational workers;
d: OR: ordinary residents (local residents who are not the family members of occupational workers).
Discussion
As a point source, this area provided an ideal platform for us to study the exposure pathways to humans and bioaccumulative behaviour of different PFAAs. The dominance of PFHxS, PFOS, and PFOA in residents around the fluorochemical facility was consistent with the results on environmental samples from the same area22. Serum PFAAs of local residents were nearly one order of magnitude higher than that of general populations in China23,30. Elevated serum PFAAs levels were also found in the nearby community of a fluorochemical industrial zone in northern China. The predominant PFAAs in residents in that study was PFOA (378 ng/mL), which was much higher than that in our study42. However, the serum PFOS and PFHxS in their study were comparable to that in the general population and were much lower than our study (PFOS:39.4 vs. 445 ng/mL; PFHxS: 1.12 vs. 138 ng/mL)42. This is most likely due to the different production pattern between two plants. The manufacturing plants in their study mainly produced fluoropolymers such as polytetrafluoroethylene, which was known to contain PFOA as a contaminant. The manufacturing plant in the present study primarily focused on PFSA(perfluorinated sulfonic acid)-based chemicals and never produced PFOA.
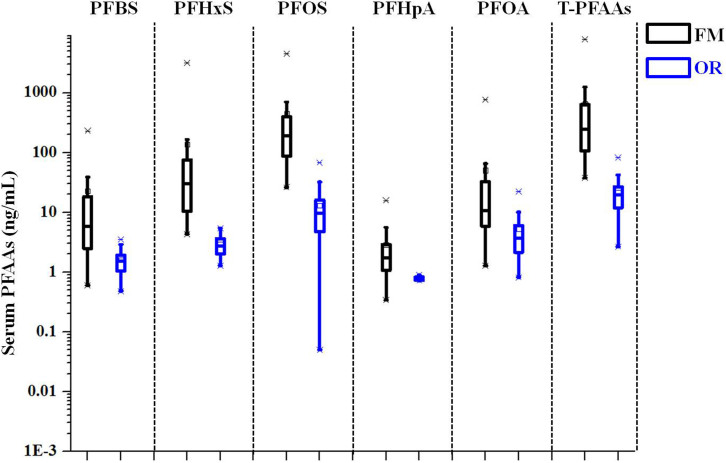
Our previous study found a decreasing gradient of PFAAs in soils and hen eggs with increasing distance from the plant22, but no such spatial trend was found in the serum of the studied population. The geometric mean serum levels of PFAAs was approximately 17 times higher in the highest quartile (Q1) than that in the lowest quartile (Q4) (1352 vs. 81 ng/mL) (Table S8). We found that serum PFAAs levels in FM group (n = 32) were statistically significantly higher than that in OR group (n = 26) (p < 0.01). The average serum PFAAs in the FM group were 1023 ng/mL, while the average serum PFAAs in the OR group were only 181 ng/mL(Table S9, Figure 1).The significantly different PFAAs body burden between FM and OR suggested that there was a special exposure pathway for PFAAs entering into FM.
Figure 1. Boxplots of PFAAs concentrations in serum samples of family members of occupational workers (FM group) and ordinary residents (OR group).
The exposure pathways of PFAAs for local residents were further examined in order to find the specific route for FM. There were no obvious difference in the PFAAs level in TSP samples between FM and OR (0.56 ng/m3 in the FM group's houses vs. 0.89 ng/m3 in the OR group's houses). In contrast to TSP, dust PFAA levels were significantly higher in the FM group (6775 ng/g) than in the OR group (429 ng/g) (p < 0.01). It was surprising that the high PFAA levels in dust seems not play a role in elevating the PFAA levels in TSP via resuspension in this study. It might because that the large noise of the TSP sampler kept human activities (such as housework, walking around the samplers and so on) away during TSP sampling process. High PFAA levels in dust samples collected from FM indicated that occupational workers elevated the PFAAs levels in the living environment. PFAAs levels in the duplicate diet were considerably higher in the FM group than in the OR group, PFOS level in duplicate diet from FM group was 1.41 ng/g ww, while it was only 0.077 ng/g ww in the duplicate diet from OR group. However, the food material of both groups was mainly purchased from the local market, and the drinking water was supplied by municipal water systems, so there was no special food source for the FM group according to questionnaires. Through our observation and further investigation, most of the occupational workers did not change their clothes after work. We collected clothes of both workers and residents, used distilled water to wash them, and then analysed PFAAs in the wash-off water (detailed information in SI). To be convenient for comparison, PFAAs concentrations in wash-off water were normalized to clothes weight according to equation (4):
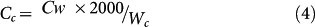 |
where Cc represents the concentration of extractable PFAAs in the clothes, Cw represents the PFAAs concentrations in wash-off water, and Wc represents the weight of the clothes. The PFAA levels in workers' clothes were significantly higher than that in residents' clothes (p < 0.05), with the GM of 218275 and 4402 ng/g, respectively (Table S10). Although not 100% of PFAAs in clothes were extracted during our washing process, however, the analysis results indicated that PFAAs concentrations in workers' clothes were almost 50 times higher than that in ordinary residents' clothes. Therefore, through the uniform manner of washing process, the determined PFAAs levels in wash-off water reflected the actual PFAAs levels in clothes. This result proved that workers unintentionally carried PFAAs to their residence via their work clothes, leading to elevated PFAAs exposures in their living environments and consequently resulting in higher PFAAs body burden of their family members. This further explained the higher PFAAs levels in the duplicate diet from FM than that in the OR group (in the case that they used the same source of food material). PFAAs probably transferred from contaminated clothes to the dinner via cooking processes, which suggested a potential PFAAs exposure pathway other than direct dusts ingestion. Contaminated clothes – diet - human is a new exposure pathway identified in our study. Such unusual route might occur in similar cases of occupational exposure, and more attention should be paid on it. Moreover, the human exposure via the new pathway could be effectively avoided by compulsory measure of changing work clothes after work.
The total PFAAs daily intake (sum of dietary intake and DIdust) of FM was higher than that of OR, which was consistent with the PFAAs body burden. Dietary intake was the major exposure pathway in the present study, which was consistent with previous studies15,18,19,21. DIdust of PFOS and PFOA accounted for 5.2% and 20.0% of the total daily intake (TDI) in the FM group, and 6.0% and 8.0% in OR group, respectively. Compared to that DIdust accounted for less than 2% of daily intake in the Chinese population18, our result further suggested that dust ingestion was an important exposure pathway to residents around the fluorochemical plant. Moreover, the house dust exposure pathway could contribute to a much higher exposure to toddlers due to the more frequent hand-to-mouth behaviour. Recently studies suggested that some PFAAs were associated with adverse health effects in children12,26, the health effects of PFAAs exposure to local children need to be further investigated.
In this study, a steady state condition of PFAAs accumulation/elimination in residents was assumed. The TEDI of PFOS and PFOA was slightly lower than TDI; serum PFAAs could be underestimated if the exposure time was not enough to achieve the steady state, possibly resulting in a low TEDI value. Furthermore, the TEDI estimated through this model referred to a daily absorption dose of humans, and in reality, humans would not absorb all PFAAs ingested into the body. In this study, the ratio of TEDI and TDI for PFOS and PFOA were 60.9% and 72.5%, respectively, which might indicate the absorption rate of PFAAs.
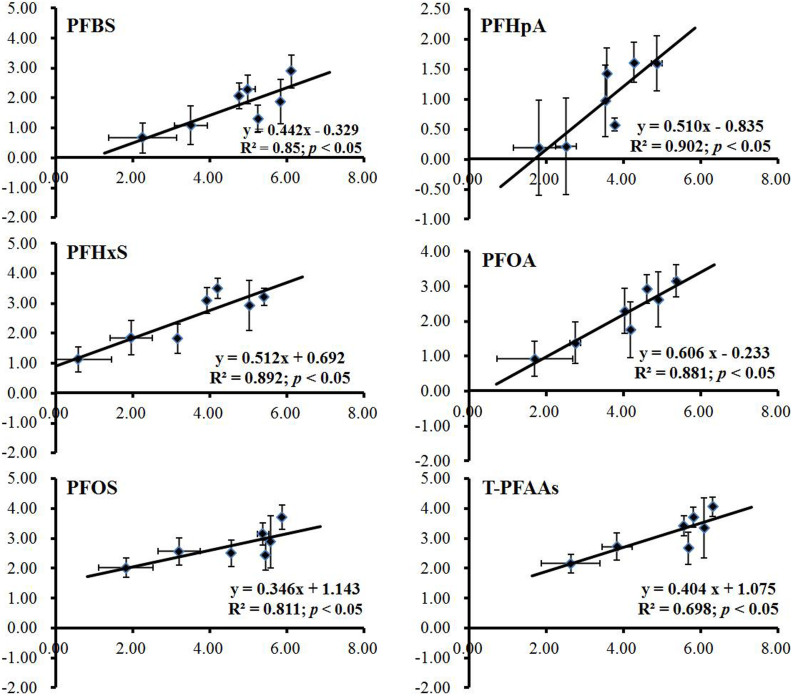
The above discussion has illustrated that dust played an important role in this case. The correlation between dust PFAAs levels and serum PFAAs levels were further investigated. There were seven groups of dust data (dust samples from FM, dust samples from OR, and dust samples from the plant were divided into five sub-groups: the sulphonation department, the fabric finishing agent department, the electrolytic department, the research building, and the management office) and two groups of serum data (FM and OR group) in this study. Thus, we cited a set of unpublished data (data only for the reviewer), the serum PFAAs levels of occupational workers from different departments in this plant in 2011 (including workers from management office, electrolytic department, fabric finishing agent department, research building, and sulphonation department), to get more paired data of dust and serum samples and make a better significance of fit. The result indicated that logarithm serum PFAA concentrations were significantly correlated with the corresponding logarithm dust PFAA concentrations (p < 0.05), which suggested that PFAAs body burden was closely related to dust ingestion in this setting (Figure 2).
Figure 2. Linear correlations between logarithm PFAAs of serum and dust samples.
The X and Y axis represent logarithm serum and dust concentrations of PFAAs, respectively.
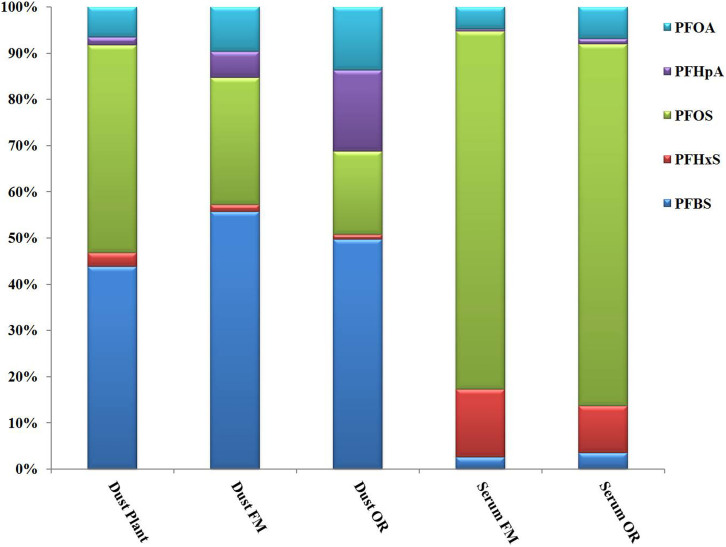
The PFAAs profiles changed largely between the dust and serum samples of local residents. PFSAs accounted for 68.2% of the total PFAAs in dust samples, which then increased to 87.4% in serum samples in the present study (Figure 3). The results suggested that PFSAs are more preferentially accumulated in humans than perfluorocarboxylic acids (PFCAs), which is also consistent with previous research in animals. As for individual PFAAs, PFBS, PFOS and PFOA accounted for 50.5%, 20.1%, and 15.9% of the total PFAAs in dust samples, respectively. However, PFOS became the predominant PFAAs in serum samples, which accounted for 78.3%, followed by PFHxS (12.6%), whereas the proportion of PFBS sharply decreased from 50.5% in dust to 2.9% in serum.
Figure 3. PFAAs composition profiles in different groups of sample matrices.
The ratio of the proportion of PFAASerum to the proportion of PFAADust was calculated to illustrate the different accumulation potential of the studied PFAAs, and this value was defined as the potential bioaccumulative index (PBI) in later discussions. The PBI of five PFAAs were ranked as follows: PFHxS (7.41) > PFOS (3.90) > PFOA (0.359) > PFBS (0.057) > PFHpA (0.051). The PBIPFHpA and PBIPFBS exhibited a low bioaccumulation potential in the present study, and it was consistent with Conder et al.'s report that PFCAs with less than seven fluorinated carbons might not accumulate in biota8. The PBIPFHxS and PBIPFOS were nearly 130 and 68.4 times higher than PBIPFBS, respectively. Olsen et al. reported that half-lives of PFHxS and PFOS were 107 and 70 times of PFBS, respectively, which were highly consistent with the PBI results in this study33,43. The PBI of PFHxS suggested the high accumulative capacity in humans, which partly supported the longer half-life of PFHxS than PFOS in human33. The bioaccumulation potential of PFOA is still a controversial issue8,33,44. Kowalczyk et al. reported that the kinetics of PFOA were similar to those of PFBS and substantially differed from those of PFHxS and PFOS in dairy cows45. We can conclude that both the length of fluorinated carbon chain and the function group influence the bioaccumulation potentital of PFAAs. Long chain PFAAs, PFOS and PFHxS, are more bioaccumulative than PFBS in our case. Although PFHxS and PFOA has six and seven perfluorinated carbons, respectively, the PBI value of PFHxS is much higher than that of PFOA in our study, suggesting that PFSAs are much more bioaccumulative than PFCAs in humans.
Supplementary Material
Supplementary information
Acknowledgments
This study was jointly supported by the Chinese Academy of Sciences (KZCX2-YW-BR-25 and YSW2013B01), the Strategic Priority Research Program of the Chinese Academy of Sciences,Grant No. XDB14030500, and the National Natural Science Foundation (21107128, 21222702, 21277164, and 21477154). The authors would also like to thank all of the blood donors and other people for their great help during the sampling process.
Footnotes
The authors declare no competing financial interests.
Author Contributions J.J.F. conceived the idea and wrote the manuscript text; J.J.F., Y.G., T.W. and Y.L. collected the samples and analysed PFAAs in samples, J.J.F., Y.W.W. and A.Q.Z. analyzed data; A.Q.Z. and G.B.J. gave conceptual advice and revised the manuscript.
References
- Key B. D., Howell R. D. & Criddle C. S. Fluorinated organics in the biosphere. Environ. Sci. Technol. 31, 2445–2454 (1997). [Google Scholar]
- Benskin J. P. et al. Perfluoroalkyl acids in the Atlantic and Canadian Arctic oceans. Environ. Sci. Technol. 46, 5815–5823 (2012). [DOI] [PubMed] [Google Scholar]
- Houde M., De Silva A. O., Muir D. C. G. & Letcher R. J. Monitoring of perfluorinated compounds in aquatic biota: An updated review PFCs in aquatic biota. Environ. Sci. Technol. 45, 7962–7973 (2011). [DOI] [PubMed] [Google Scholar]
- Shi Y. L., Pan Y. Y., Yang R. Q., Wang Y. W. & Cai Y. Q. Occurrence of perfluorinated compounds in fish from Qinghai-Tibetan Plateau. Environ. Int. 36, 46–50 (2010). [DOI] [PubMed] [Google Scholar]
- Paul A. G., Jones K. C. & Sweetman A. J. A first global production, emission, and environmental inventory for perfluorooctane sulfonate. Environ. Sci. Technol. 43, 386–392 (2009). [DOI] [PubMed] [Google Scholar]
- UNEP, The new POPs under the Stockholm Convention. (2009) Available at: http://chm.pops.int/TheConvention/ThePOPs/TheNewPOPs/tabid/2511/Default.aspx. (Accessed: 8th November 2014).
- Xie S. W. et al. Industrial source identification and emission estimation of perfluorooctanesulfonate in China. Environ. Int. 52, 1–8 (2013). [DOI] [PubMed] [Google Scholar]
- Conder J. M., Hoke R. A., De Wolf W., Russell M. H. & Buck R. C. Are PFCAs bioaccumulative? A critical review and comparison with regulatory lipophilic compounds. Environ. Sci. Technol. 42, 995–1003 (2008). [DOI] [PubMed] [Google Scholar]
- Dallaire R. et al. Determinants of plasma concentrations of perfluorooctanesulfonate and brominated organic compounds in nunavik inuit adults (Canada). Environ. Sci. Technol. 43, 5130–5136 (2009). [DOI] [PubMed] [Google Scholar]
- Karrman A., van Bavel B., Jarnberg U., Hardell L. & Lindstrom G. Perfluorinated chemicals in relation to other persistent organic pollutants in human blood. Chemosphere 64, 1582–1591 (2006). [DOI] [PubMed] [Google Scholar]
- Uhl S. A., James-Todd T. & Bell M. L. Association of osteoarthritis with perfluorooctanoate and perfluorooctane sulfonate in NHANES 2003-2008. Environ. Health Perspect. 121, 447–452 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Espinosa M. J. et al. Association of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) with age of puberty among children living near a chemical plant. Environ. Sci. Technol. 45, 8160–8166 (2011). [DOI] [PubMed] [Google Scholar]
- Domingo J. L. et al. Human exposure to perfluorinated compounds in Catalonia, Spain: Contribution of drinking water and fish and shellfish. J. Agric. Food. Chem. 60, 4408–4415 (2012). [DOI] [PubMed] [Google Scholar]
- Vestergren R. & Cousins I. T. Tracking the pathways of human exposure to perfluorocarboxylates. Environ. Sci. Technol. 43, 5565–5575 (2009). [DOI] [PubMed] [Google Scholar]
- Jogsten I. E., Nadal M., van Bavel B., Lindstrom G. & Domingo J. L. Per- and polyfluorinated compounds (PFCs) in house dust and indoor air in Catalonia, Spain: Implications for human exposure. Environ. Int. 39, 172–180 (2012). [DOI] [PubMed] [Google Scholar]
- Zhang T. et al. Perfluorinated compounds in human blood, water, edible freshwater fish, and seafood in China: Daily intake and regional differences in human exposures. J. Agric. Food. Chem. 59, 11168–11176 (2011). [DOI] [PubMed] [Google Scholar]
- Beesoon S., Genuis S. J., Benskin J. P. & Martin J. W. Exceptionally high serum concentrations of perfluorohexanesulfonate in a Canadian family are linked to home carpet treatment applications. Environ. Sci. Technol. 46, 12960–12967 (2012). [DOI] [PubMed] [Google Scholar]
- Zhang T. et al. Perfluorochemicals in meat, eggs and indoor dust in China: Assessment of sources and pathways of human exposure to perfluorochemicals. Environ. Sci. Technol. 44, 3572–3579 (2010). [DOI] [PubMed] [Google Scholar]
- Rylander C., Sandanger T. M., Froyland L. & Lund E. Dietary patterns and plasma concentrations of perfluorinated compounds in 315 Norwegian women: The NOWAC postgenome study. Environ. Sci. Technol. 44, 5225–5232 (2010). [DOI] [PubMed] [Google Scholar]
- Karrman A. et al. Relationship between dietary exposure and serum perfluorochemical (PFC) levels-A case study. Environ. Int. 35, 712–717 (2009). [DOI] [PubMed] [Google Scholar]
- Vestergren R., Cousins I. T., Trudel D., Wormuth M. & Scheringer M. Estimating the contribution of precursor compounds in consumer exposure to PFOS and PFOA. Chemosphere 73, 1617–1624 (2008). [DOI] [PubMed] [Google Scholar]
- Wang Y. W. et al. Distribution of perfluorooctane sulfonate and other perfluorochemicals in the ambient environment around a manufacturing facility in China. Environ. Sci. Technol. 44, 8062–8067 (2010). [DOI] [PubMed] [Google Scholar]
- Pan Y., Shi Y., Wang J., Cai Y. & Wu Y. Concentrations of perfluorinated compounds in human blood from twelve cities in China. Environ. Toxicol. Chem. 29, 2695–2701 (2010). [DOI] [PubMed] [Google Scholar]
- Pan Y. Y., Shi Y. L. & Cai Y. Q. Determination of perfluorinated compounds in human blood samples by high performance liquid chromatography-electrospray tandem mass spectrometry. Chinese J. Anal. Chem. 36, 1321–1326 (2008). (In Chinese). [Google Scholar]
- USEPA, Exposure factors handbook. (2011). Available at: http://www.epa.gov/ncea/efh/pdfs/efh-frontmatter.pdf. (Accessed: 1st April 2014).
- Bjorklund J. A., Thuresson K. & De Wit C. A. Perfluoroalkyl compounds (PFCs) in indoor dust: Concentrations, human exposure estimates, and sources. Environ. Sci. Technol. 43, 2276–2281 (2009). [DOI] [PubMed] [Google Scholar]
- Fasano W. J. et al. Penetration of ammonium perfluorooctanoate through rat and human skin in vitro. Drug Chem. Toxicol. 28, 79–90 (2005). [DOI] [PubMed] [Google Scholar]
- Thompson J. et al. Use of simple pharmacokinetic modeling to characterize exposure of Australians to perfluorooctanoic acid and perfluorooctane sulfonic acid. Environ. Int. 36, 390–397 (2010). [DOI] [PubMed] [Google Scholar]
- Andersen M. E., Clewell H. J., Tan Y. M., Butenhoff J. L. & Olsen G. W. Pharmacokinetic modeling of saturable, renal resorption of perfluoroalkylacids in monkeys - Probing the determinants of long plasma half-lives. Toxicology 227, 156–164 (2006). [DOI] [PubMed] [Google Scholar]
- Zhang T. et al. Perfluorinated compounds in whole blood samples from infants, children, and adults in China. Environ. Sci. Technol. 44, 4341–4347 (2010). [DOI] [PubMed] [Google Scholar]
- Zhang Y., Beesoon S., Zhu L. & Martin J. W. Isomers of perfluorooctanesulfonate and perfluorooctanoate and total perfluoroalkyl acids in human serum from two cities in North China. Environ. Int. 53, 9–17 (2013). [DOI] [PubMed] [Google Scholar]
- Thompson J., Eaglesham G. & Mueller J. Concentrations of PFOS, PFOA and other perfluorinated alkyl acids in Australian drinking water. Chemosphere 83, 1320–1325 (2011). [DOI] [PubMed] [Google Scholar]
- Olsen G. W. et al. Half-life of serum elimination of perfluorooctanesulfonate, perfluorohexanesulfonate, and perfluorooctanoate in retired fluorochemical production workers. Environ. Health Perspect. 115, 1298–1305 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen G. W. et al. Decline in perfluorooctanesulfonate and other polyfluoroalkyl chemicals in American Red Cross adult blood donors, 2000-2006. Environ. Sci. Technol. 42, 4989–4995 (2008). [DOI] [PubMed] [Google Scholar]
- Harada K. et al. Renal clearance of perfluorooctane sulfonate and perfluorooctanoate in humans and their species-specific excretion. Environ. Res. 99, 253–261 (2005). [DOI] [PubMed] [Google Scholar]
- Boiteux V., Dauchy X., Rosin C. & Munoz J. F. National screening study on 10 perfluorinated compounds in raw and treated tap water in France. Arch. Environ. Con. Tox. 63, 1–12 (2012). [DOI] [PubMed] [Google Scholar]
- Ericson I. et al. Levels of perfluorinated chemicals in municipal drinking water from Catalonia, Spain: Public health implications. Arch. Environ. Con. Tox. 57, 631–638 (2009). [DOI] [PubMed] [Google Scholar]
- Wang B. B. et al. Distribution of perfluorinated compounds in surface water from Hanjiang River in Wuhan, China. Chemosphere 93, 468–473 (2013). [DOI] [PubMed] [Google Scholar]
- Fromme H. et al. Exposure of an adult population to perfluorinated substances using duplicate diet portions and biomonitoring data. Environ. Sci. Technol. 41, 7928–7933 (2007). [DOI] [PubMed] [Google Scholar]
- Barber J. L. et al. Analysis of per- and polyfluorinated alkyl substances in air samples from Northwest Europe. J. Environ. Monit. 9, 530–541 (2007). [DOI] [PubMed] [Google Scholar]
- Harada K. et al. Particle size distribution and respiratory deposition estimates of airborne perfluorooctanoate and perfluorooctanesulfonate in Kyoto area, Japan. B. Environ. Contam. Tox. 76, 306–310 (2006). [DOI] [PubMed] [Google Scholar]
- Wang J., Zhang Y., Zhang W., Jin Y. & Dai J. Association of perfluorooctanoic acid with HDL cholesterol and circulating miR-26b and miR-199–3p in workers of a fluorochemical plant and nearby residents. Environ. Sci. Technol. 46, 9274–9281 (2012). [DOI] [PubMed] [Google Scholar]
- Olsen G. W. et al. A comparison of the pharmacokinetics of perfluorobutanesulfonate (PFBS) in rats, monkeys, and humans. Toxicology 256, 65–74 (2009). [DOI] [PubMed] [Google Scholar]
- Kelly B. C. et al. Perfluoroalkyl contaminants in an Arctic marine food web: trophic magnification and wildlife exposure. Environ. Sci. Technol. 43, 4037–4043 (2009). [DOI] [PubMed] [Google Scholar]
- Kowalczyk J. et al. Absorption, distribution, and milk secretion of the perfluoroalkyl acids PFBS, PFHxS, PFOS, and PFOA by dairy cows fed naturally contaminated feed. J. Agric. Food. Chem. 61, 2903–2912 (2013). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary information