Abstract
Natural killer (NK) cell education, a process for achieving functional maturation and self-tolerance, has been previously defined by the interaction between self-major histocompatibility complex class I (MHC-I) molecules and their specific inhibitory receptors. Over the past several years, growing evidence has highlighted the important roles of nonclassical MHC-I and non-MHC-I molecules in NK cell education. Herein, we review the current knowledge of NK cell education, with a particular focus on nonclassical MHC-I- and non-MHC-I-dependent education, and compare them with the classical MHC-I-dependent education theory. In addition, we update and extend this theory by presenting the 'Confining Model', discussing cis and trans characteristics, reassessing quantity and quality control, and elucidating the redundancy of NK cell education in tumor and virus infection.
Keywords: education, licensing, natural killer cells, nonclassical MHC, non-MHC
Introduction
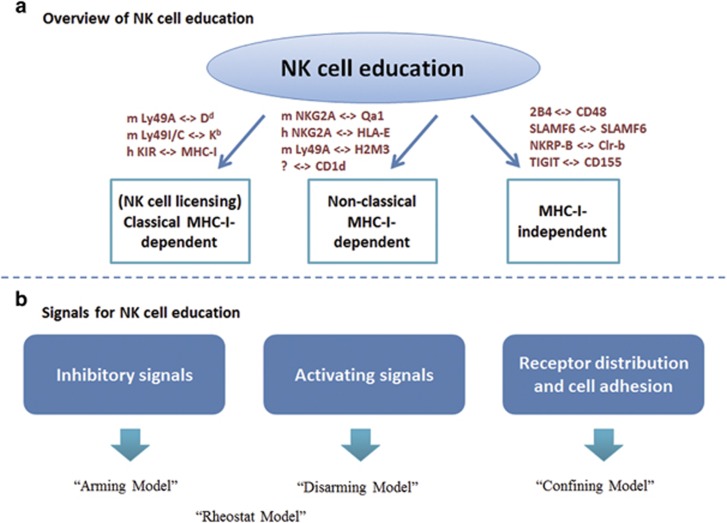
In addition to T cells and B cells, natural killer (NK) cells comprise a third major type of lymphocyte. NK cells do not express rearranged, antigen-specific receptors, and thus, NK cells are classified as an important component of the innate immune system and participate in early anti-viral and anti-tumor immune responses. Similar to T cells and B cells, NK cells possess a fine-tuning mechanism known as NK cell education, in which they acquire functional competence and adapt to the host where they develop.1, 2, 3 It is well established that the recognition of self-major histocompatibility complex class I (MHC-I) molecules via their specific inhibitory receptors is critical for the acquisition of functional effector NK cells.4, 5, 6, 7, 8 However, recent studies have identified new educational signals mediated through engagements of nonclassical MHC molecules or even non-MHC molecules (Figure 1a; Table 1). In light of these observations, we review the current data about the nonclassical MHC-dependent and MHC-independent education of NK cells; propose the 'Confining Model', which suggests an update to this signaling process; reassess the controversial 'Cis-interaction Model' discuss the plasticity of education; and finally, demonstrate relevant therapeutic implications.
Figure 1.
Outline of NK cell education. (a) Overview of NK cell education. NK cell education, a broad concept regarding the acquisition of NK cell function and self-tolerance, can be defined as classical MHC-I-dependent, nonclassical MHC-I-independent and MHC-I-independent. (b) Signals for NK cell education. Inhibitory signals, activating signals, and adhesion signals are involved in NK cell education. In the 'Arming Model', inhibitory receptors provide critical signals for education through their ITIM motifs. The 'Disarming Model' highlights the important role of activating receptors. NK cells are disarmed (hypo-responsive) if they are without inhibitory signals to balance the chronic activating stimulations. The 'Rheostat Model' proposes a quantitative perspective of NK cell education, suggesting that the increase or decrease in NK cell responsiveness is quantitatively controlled by the strength of the inhibitory input. The 'Confining Model' demonstrates that the confinement of receptor distribution is indispensable for NK cell education. ITIM, immunoreceptor tyrosine-based inhibitory motif; MHC, major histocompatibility complex; NK cells, natural killer cells.
Table 1. Animal models for NK cell education.
| Point | Animal model | Result | References |
|---|---|---|---|
| Classical-MHC-I- dependent education | B6 (H2b) mice | Ly49C+ NK cells displayed more augmented responsiveness in response to MHC-I-deficient targets and activation receptor cross-linking than Ly49C- NK cells. | 1, 9 |
| B10.D2 (Kd Dd) mice | Ly49A+ NK cells showed a higher level of cytokine production and cytotoxicity than Ly49A- NK cells. | 1 | |
| MHC-I-deficient mice (β2m−/− mice, TAP−/− mice, H2-KbDbKO mice) | Normal NK cell development and maturation; impaired NK response to MHC-I-deficient targets and activation receptor cross-linking. | 1 | |
| NKCKD mice (Ly49-deficient mice) | Normal NK cell development and maturation; defective NK cell response to MHC-I-deficient targets; strong activating signals masked the deficiency of NK cell education. | 11 | |
| H2Dd Tg mice | Ly49A+ NK cells, but not CD8αα iIELs, became responsive following the induction of H-2Dd. | 12, 14 | |
| KbDbKO-TgKIR/HLA mice | NK cells regained functional competence following KIR and HLA induction. | 13 | |
| SCT Tg β2m−/− Kb−/−Db−/− mice | Unlicensed Ly49C+ β2m−/− Kb−/−Db−/− NK cells became responsive after induced SCT-Kb expression. | 1 | |
| Nonclassical MHC-I-dependent education | B6 (H2b) mice | NKG2A+Ly49C/I+ double-positive (DP) NK cells exhibited more augmented responsiveness than the single-positive (SP) or double-negative (DN) cells. | 17, 18 |
| H2-M3-deficient mice | Normal NK cell development and MHC-I expression; attenuated NK cell ability to control B16-F10 metastases and MCA-induced fibrosarcoma; impaired NK cell response to activation receptor cross-linking. | 19, 20 | |
| CD1d1-deficient mice | Normal NK cell maturation; attenuated NK cell 'missing-self' recognition and NKG2D-dependent cytotoxicity. | 23 | |
| MHC-I-independent education | 2B4-deficient mice | Normal NK cell development; dampened NK cell lysis of CD48- targets. | 24, 27 |
| CD48-deficient mice | Impaired NK cell lysis of CD48-deficient targets. | 24, 27 | |
| SLAMF6-deficient mice | Normal NK cell development; defective NK cell responses toward nonhematopoietic cells. | 30 | |
| NKR-P1B-deficient mice | Normal NK cell development and maturation; defective rejection of Clr-b-deficient tumors and allogenic cells. | 32, 35 | |
| Clr-b-deficient mice | Attenuated NK responsiveness of cytokine production in response to PK136 or IL-12 plus IL-18 stimulation. | 32, 35 | |
| TIGIT-deficient mice | Normal NK cell maturation but impaired NK cell functional degranulation ability; defective NK cell-mediated rejection of CD155-deficient targets. | (He, 2016, unpublished data) | |
| CD155-deficient mice | Impaired degranulation ability of TIGIT+ NK cells. | ||
| Arming model | Gene transfer of Ly49A mutant | Ly49A lacking ITIM failed to confer licensing. | 1 |
| SHP1-deficient mice (mev mice, dn SHP1 mice) | Attenuated NK cell cytotoxicity and cytokine production. | 43 | |
| SHP1-cKO mice (Ptpn6fl/fl-Ncr1icre mice) | Defective NK cell-mediated rejection of MHC-I-deficient transplants and tumors. | 42 | |
| SHIP-deficient mice | Diminished NK cell killing of tumor cells. | 44, 45 | |
| SAP-deficient mice | Compromised NK killing of hematopoietic cells; enhanced NK cell responses to a wide range of nonhematopoietic targets. | 28, 30 | |
| Disarming model | Rae1 Tg mice | Decreased NKG2D expression on NK cells; defective NK cell resistance of Rae1+ targets. | 46, 47 |
| m157 Tg mice | Decreased Ly49H expression on NK cells; attenuated NK cell response through Ly49H signaling. | 50 | |
| Quantity and quality control | B6 (H2b) mice | NK cell responses increased with the number of types of inhibitory receptors. | 32, 52, 53 |
| 1 (Db), 2 (KbDb), or 3 (KbDbDd) MHC-I-expressing mice | NK cell responsiveness increased with the number of MHC-I alleles; Kb and Dd were strong educational ligands; Ld is relatively weak. | 54 | |
| Confining model | Wild-type, KbDbKO, KbDbKO-TgKIR/HLA, and KbDbKO-TgKIR mice | Activating receptors of educated NK cells were organically confined in membrane nanodomains; receptors of uneducated NK were dispersed into the actin meshwork. | 59 |
| LFA-1 deficient mice, talin-deficient mice, and Wiskott-Aldrich Syndrome protein (WASP)-deficient mice | Dampened LFA-1 signaling was correlated with impaired reorganization of the actin cytoskeleton and polarization. | 61, 62 | |
| Cis or trans? | Ly49-72A Tg mice | Inhibitory Ly49A receptors interacted with Dd in trans but not in cis; defective NK cell response to H-2Dd -deficient targets. | 74 |
| Ly49A Δa1 Tg mice | Inhibitory Ly49A receptors interacted with Dd in cis but not in trans; defective NK cell response to activation receptor cross-linking. | 77 | |
| Ncr-cre Dd mice and CD4-Cre Dd mice (NK cell and T cell-specific H-2Dd deficient mice) | Impaired NK cell response to H-2Dd -deficient targets. | 77 | |
| Transfer of NK cells into a different SLAMF6-deficient environment | The absence of trans effects did not alter the SLAMF6-mediated NK cell education. | 30 |
Abbreviations: HLA, human leukocyte antigen; ITIM, immunoreceptor tyrosine-based inhibitory motif; KIR, killer cell immunoglobulin-like receptor; MHC, major histocompatibility complex; NK cells, natural killer cells; SCT, single-chain trimer.
Classical NK cell education (NK cell licensing)
NK cell activation is largely regulated by self MHC-I molecules. The most widely studied MHC-I-specific receptors are inhibitory Ly49 receptors in mice and KIRs (killer cell immunoglobulin-like receptors) in humans. It is worth noting that a subset of NK cells lacking the expression of inhibitory MHC-I-specific receptors are not autoreactive but have acquired a state of 'hypo-responsiveness' to MHC-I-deficient targets or cross-linked activating signals.3, 9, 10 Moreover, NK cells from either MHC-I-deficient mice (for example, β2m−/− mice, TAP−/− mice and H2-KbDbKO mice) or Ly49 receptor-deficient mice (for example, NKCKD mice) fail to reject MHC-I-deficient targets and respond poorly to many stimuli.1, 11 These findings indicate that only NK cells that have engaged their inhibitory receptors with self MHC-I molecules during development are functionally competent. This interaction between the self MHC-I molecule and its specific inhibitory receptor that allows NK cells to become functionally mature is termed NK cell licensing or classical NK cell education. The further evidence of licensing is provided by experiments utilizing MHC-I-transgenic mice. The induced expression of an MHC class I single-chain trimer consisting of ovalbumin peptide (SIINFEKL), β2m, and H2Kb heavy chain led to the licensing of Ly49C+ NK cells. Similarly, the transgenic expression of MHC ligand H2-Dd or human leukocyte antigen (HLA) rendered Ly49A+ NK cells and KIR+ NK cells, respectively, licensed and responsive.1, 12, 13, 14
Consequently, NK cell licensing results in two types of self-tolerant NK cells. The licensed cells are effective sensors of a missing MHC-I target, but are unable to attack the MHC-I sufficient hosts at the particular locations the inhibitory receptors would be ligated; in contrast, the unlicensed NK cells are hypo-responsive and therefore have a low potential to attack normal cells.
Non-classical MHC-I-Dependent NK cell education
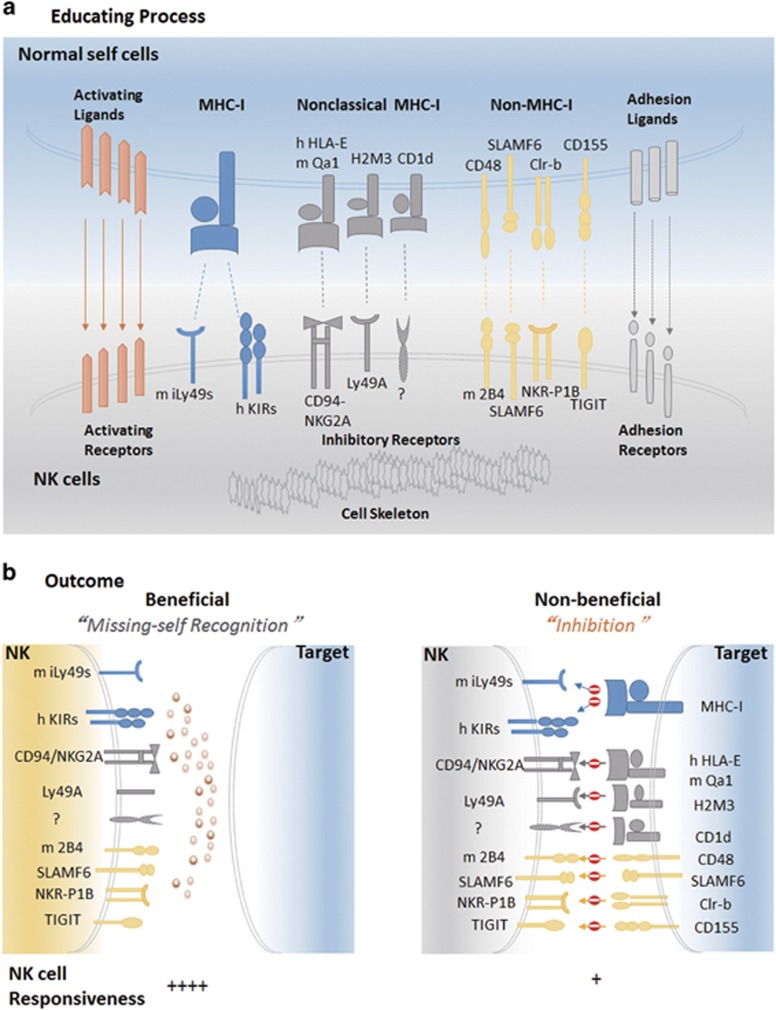
Because NK cells possess a wide range of inhibitory receptors in addition to Ly49 receptors and KIRs (Figure 2a), it is critical to determine whether non-classical MHC-I-specific inhibitory receptors can regulate NK cell education and activation.
Figure 2.
Schematic representation of the role of education on target recognition. (a) Educating process. During development, NK cells acquire functional maturation through an adaptation to the host. In this process, inhibitory receptors are directly involved by engaging self-ligands (either MHC-I-dependent or not) to educate NK cells to acquire effector responses. (b) Outcome. Differential roles of the education process are shown with respect to the presence of inhibitory ligands on target cells. Education is beneficial to allow NK cells with the expression of inhibitory receptors to sense missing self. However, when inhibitory ligands are sufficient on target cells, the inhibition by ligation of inhibitory receptors with their cognate ligands impedes the activation of educated NK cells. NK cells, natural killer cells.
The C-type lectin-like receptor CD94/NKG2A, another commonly studied inhibitory receptor, recognizes non-classical MHC-Ib molecules (that is, Qa-1 in mice and HLA-E in humans) and is important in educating NK cell tolerance to self and contributes to the inhibition of NK cell-mediated immunity to infections and tumors.15 NK cells expressing NKG2A are efficient killers of certain targets, especially the ligand deficient ones (specifically Qa1 or HLA-E), which might suggest an educational role via NKG2A signaling.16, 17, 18 It has been shown that the MHC-specific KIRs are not able to educate NK cells in the human fetus and instead induce hypo-responsiveness. However, NKG2A educates fetal NK cells as well as adult peripheral blood NK cells.17 The mechanism underlying the differential education via NKG2A and KIRs remains to be elucidated.
The inhibitory Ly49A receptor recognizes both the MHC-I molecule Dd and the nonclassical MHC-Ib molecule H2-M3. The Ly49A-H2M3 axis resembles the classical Ly49-H2-Dd system, which triggers the functional maturation of Ly49A+ NK cells and enables them to respond to numerous stimuli.19 Due to an impairment in NK cell education, H2-M3-deficient mice show an increased risk of tumor invasion in a Ly49A-dependent manner.20, 21 These observations extend our knowledge of the effects of H2-M3 on NK cells and provide new evidence for nonclassical MHC-I-dependent education effects.
CD1d1, a nonclassical MHC-I molecule crucial for NK T-cell maturation and activation, also binds to NK cells and inhibits NK cell cytotoxicity.19, 22 Interestingly, CD1d1-deficient mice exhibit impaired NK cell activity (due to defective 'missing-self' recognition and NKG2D-dependent cytotoxicity), despite normal NK cell number and development.23 It is possible that a trans interaction between CD1d1 and an unknown inhibitory receptor enhances NK responsiveness to provide an unknown mechanism of education.
MHC-I-Independent NK cell education
Several members of non-MHC-I families, notably signaling lymphocytic activation molecule (SLAM)-related ligand CD48 (2B4 ligand), SLAM family member 6 (SLAM6, self ligand), C-type lectin-related ligand Clr-b (NKRP1-B ligand) and poliovirus receptor CD155 (TIGIT ligand), are known to inhibit the responsiveness of NK cells. Thus, it is critical to determine whether interactions between these non-MHC molecules and their specific inhibitory receptors might have a role in MHC-I-independent education.
2B4 (CD244), a member of the SLAM family of CD2-related receptors, specifically recognizes the cognate SLAM molecule CD48. Murine 2B4 is primarily considered to be a non-MHC-I-dependent inhibitor of NK cells, especially in the absence of its adapter SAP (SLAM-associated protein), as it inhibits NK cells from killing CD48-sufficient tumor cells or allogeneic cells.24, 25, 26 Given that NK cells from either 2B4 knockout mice or CD48 knockout mice fail to reject CD48-deficient tumor cells,24, 27 this 2B4-CD48 engagement is necessary for NK cell-mediated recognition of missing self-CD48 targets and thus represents a MHC-I-independent education pathway.
SLAMF6 (CD352, also known as NTB-A and Ly108), a homotypic receptor of the SLAM family, is highly expressed on hematopoietic cells such as NK, T and B lymphocytes. Similar to 2B4, in the absence of the adapter protein SAP SLAMF6 inhibits NK cell activation when properly engaged with SLFMF6-sufficient hematopoietic target cells.28, 29 On the other hand, SLAMF6-SLAMF6 homotypic interactions enhance NK cell responses to a wide range of non-hematopoietic targets that lack the expression of SLAMF6.30 This observation suggests that the SLAMF6 receptor confers an educational effect and potentiates NK cell-mediated missing self-recognition of SLAFM6-deficient nonhematopoietic cells.
NKRP1-B, a member of the NKRP1 family, differs from the extensively studied activating receptor NKRP1-C (also called NK1.1) and inhibition of NK cells when properly ligated with its cognate C-type lectin-related ligand Clr-b.31, 32 Although NKP1-B+ NK cells are unable to kill Clr-b-sufficient targets, they can readily kill Clr-b-deficient targets and show higher responsiveness to various stimuli than NKP1-B- NK cells.33 Researchers have also observed that an NK cell subset co-expressing Ly49I/C and NKRP1-B is more responsive to stimuli, an interaction which mediates a more efficient MHC-I-independent missing self-recognition of Clr-b in both tumor environment and under CMV infections compared with those NK cell subsets expressing only one or neither of the two receptors.34, 35, 36 Furthermore, the absence of either NKRP1-B or Clr-b results in the hypo-responsiveness of NK cells, for example, the dampened rejection of Clr-b missing targets and diminished secretion of interferon-γ.32, 35 Therefore, similar to the MHC-I-specific inhibitory receptors, NKRP1-B is involved in NK cell self-tolerance and the missing self-recognition of Clr-b-deficient targets and hence contributes to NK cell education.
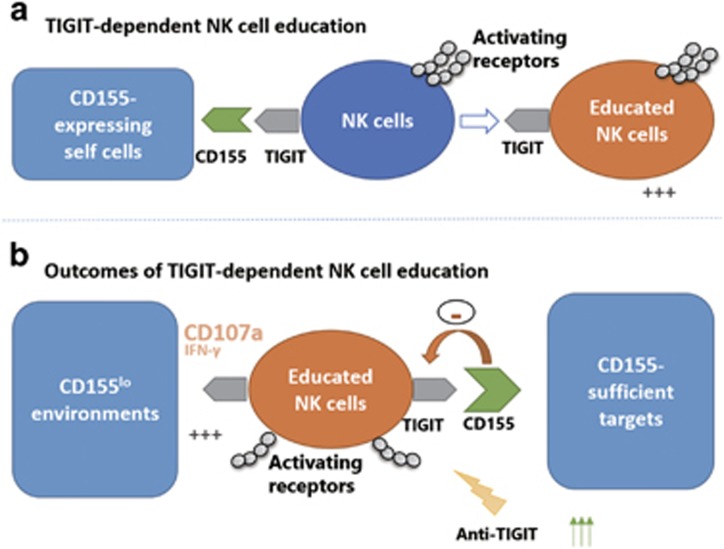
TIGIT (T cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain), a recently discovered inhibitory receptor predominantly expressed on T cells and NK cells, inhibits NK cell and T-cell responsiveness upon ligation with its immunoglobulin superfamily ligand CD155.37, 38, 39, 40 Our unpublished data suggest that TIGIT+ NK cells display an augmented responsiveness compared with TIGIT− NK cells to certain stimuli. In addition, the absence of TIGIT or self CD155 in gene knockout mice results in functional defects in NK cells, as demonstrated by impaired recognition and killing of CD155-deficient targets (He, 2016, unpublished data). This finding addresses how the functional maturation of NK cells is impacted by the TIGIT-CD155 system and reveals a certain role for non-MHC-specific receptors in regulating NK cell education (Figure 3).
Figure 3.
TIGIT-CD155 axis regulates NK cell education (He, 2016, unpublished data). (a) During NK cell development, CD155 molecules on self-cells is involved in educating TIGIT+ NK cells to obtain final functional maturation. (b) The TIGIT-CD155 system is important for NK cells in sensing missing CD155 targets. However, CD155-sufficient targets inhibit the responsiveness of functional TIGIT+ NK cells. NK cells, natural killer cells.
Signals of NK cell education
Coordinated integration of inhibitory, activating and adhesion signals regulates the process of NK cell education (Figure 1b). This next section will address several mechanisms that have been proposed to explain how NK cells become educated.
Inhibitory signals allow NK cell education
The 'Arming Model' was first proposed to address how NK cells can become educated via inhibitory receptors by suggesting that inhibitory signaling directly potentiates NK cells with functional potential.1 However, this theory is not compatible with the traditional understanding that inhibitory receptors negatively regulate cell functional ability upon ligation.41 To potentially explain this contradiction, it is important to know that a common feature of inhibitory receptors, MHC dependent or not, is the presence of an essential ITIM in the cytoplasmic tail. ITIM-mutated NK cells cannot be educated and respond poorly to stimuli,1 which reveals an indispensable role for intact ITIMs and provides strong evidence to support the Arming Model. Initial signaling by ITIM motifs involves the recruitment of several phosphatases, as follows: SH2-domain-containing protein tyrosine phosphatase 1 (SHP1), SHP2 and SH2-domain-containing inositol-5-phosphatase (SHIP). Among these proteins, SHP1 is the most abundant and best understood. NK cell-specific deletion of SHP1 alters the expression of inhibitory Ly49 receptors and impairs NK cell cytotoxicity and cytokine production,42, 43 which suggests that signaling via SHP1 is crucial for the process of NK cell education. Inhibitory signaling through non-MHC inhibitory receptors such as 2B4, TIGIT and KLRG1 is often associated with the recruitment of SHIP and not SHP1, and the absence of SHIP renders NK cells hypo-responsive and uneducated.44, 45 The adapter protein SAP, which disrupts inhibitory signaling mediated by SHP1 or SHIP, impedes NK cell education.28, 30 This phenomenon would suggest that inhibitory receptor-mediated NK cell education is an ITIM-dependent process and is mediated by either SHP1 or SHIP downstream signaling.
Sustained activation induces NK cell hypo-responsiveness
NK cell activation is coordinately regulated by both inhibitory and activating receptors. In general, activating receptors allow NK cells to detect and diminish infected, transformed host cells or tumor cells. However, the sustained engagement of activating receptors with their plate-bound ligands in vitro or with tumor ligands in vivo induces hypo-responsiveness in NK cells.46, 47, 48 Some studies performed in transgenic mice clearly demonstrate the negative effects of sustained engagement of activating receptors for NK cells. The transgenic expression of Rae-1 results in the reduced expression of the NKG2D receptor and impairment of NKG2D-mediated NK cell function.46, 49 In addition, the transduction of the m157 gene decreases the expression of activating Ly49H receptor and leads to impaired NK cell activation through Ly49H signaling.50, 51 In agreement with studies conducted in mice, the continuous engagement of the human-activating receptor KIR2DS1 impairs the responsiveness of NK cells.49 Thus, in the absence of inhibitory signals, NK cells experience sustained activation and become hypo-responsive, or 'disarmed', which is known as the Disarming Model.2
Quantity and quality control of NK cell education
The research on MHC-dependent education has demonstrated that NK cells co-expressing NKG2A and inhibitory Ly49 receptors are more responsive to stimuli than those with just one or no MHC-dependent inhibitory receptors.52, 53 Furthermore, studies utilizing MHC-deficient or transgenic mice show that the responsiveness of NK cells increases with the number of MHC-I alleles.20, 53, 54 In line with studies of mice, human NK cells with more types of KIR gene copies display augmented functional competence,55 which suggests that NK cell responsiveness varies continuously with inhibitory input.56 Recent studies have also demonstrated that NK cells expressing both Ly49I/C and NKRP1-B display augmented responsiveness,32, 35 extending the notion of inhibitory input in a MHC-I-independent manner. We have also observed that an NK cell subset co-expressing TIGIT, NKG2A and Ly49C/I exhibit notably higher responsiveness to stimuli (He, 2016, unpublished data), further broadening the concept of quantity control of NK cell education.
In addition to the number of inhibitory receptors or ligands, the education impact increases with the strength of the receptor-ligand interaction, that is, strong ligands (for example, Kb and Dd) show effective education impacts, while the role of weak ligands (for example, Ld) are compromised.54 Degranulation, as assessed by CD107a expression, and cytokine production, particularly interferon-γ secretion, are two hallmarks of NK cell responsiveness. The achievement of effector interferon-γ function requires stronger inhibitory input than achievement of degranulation ability.56 Education via nonclassical MHC or non-MHC ligands is sufficient to potentiate the ability to degranulate but is too weak to stimulate cytokine secretion. Different inhibitory receptors vary in the strength of educational input, suggesting that NK cell education is not only quantitatively but also qualitatively tuned. This notion is referred to as the Rheostat Model, in which NK cell education is not an 'on or off' procedure but is a continuously ranging process.56
Receptor distribution and cell adhesion—the 'Confining Model'
Although the proposed Arming Model and Disarming Model explains what occurs during NK cell education to a certain extent, large gaps still exist between these models and detailed mechanisms. For example, it is important to identify what determines the response capacity of educated NK cells. Cell–cell contact constitutes the first step in initiating NK cell cytotoxicity. In this type of contact, the activating and inhibitory receptors of NK cells cluster at the synapse, and then inside-out signaling through adhesion molecules is induced to form stable conjugates and to trigger the polarization of granules. Activating receptors, particularly NKG2D and NKp46, structurally form microclusters at the immune synapse, and this compartmentalization is correlated with enhanced actin remodeling and a lowered threshold for activation.57, 58 Research using dynamic fluorescence demonstrates that activating receptors of educated NK cell are organically confined in nanodomains at the membrane, whereas the receptors of hypo-responsive NK cells from MHC-I-deficient mice are dispersed into the actin meshwork.59 This physical confinement of activating receptors provides an explanation for education-mediated functional competence. Recent data have also demonstrated that NKG2A+ NK cells are more dynamic and form stronger contacts with targets.60 Our group has also found that TIGIT+ NK cells possess an enhanced ability to conjugate with targets (He, 2016, unpublished data), forming a complement in a MHC-independent manner.
Adhesion molecules, notably LFA-1, are critical for NK cells to control the compartmentalization of receptors and enable stable conjugate formation.61, 62 It is worth noting that the weak NK cell contacts of uneducated NK cells can be partially explained by a lack of inside-out signaling mediated by LFA-1.63 Another well-studied costimulatory adhesion molecule, CD226 (DNAM-1), which is involved in synapse formation and NK-mediated cytotoxicity of certain tumors, co-localizes with LFA-1 and facilitates LFA-1 mediated contacts.64 The synergy between CD226 and LFA-1 in educated NK cells constitutes an important regulator in cell conjugation formation and promotion of NK cell reactivity.65 Moreover, the expression pattern of CD226 is correlated with that of MHC-I-specific inhibitory receptors, suggesting that CD226 acts as a sensor of classical NK cell education.65, 66
The cell receptors associated with adhesion molecules and the cytoskeleton at the plasma membrane form a well-confined distribution to ensure NK cell responsiveness. This idea represents a new Confining Model, in which the education-induced confinement of receptor compartmentalization prompts efficient NK cell contacts and sequential NK cell activation. Overall, NK cell education is a process determined by the coordination of inhibitory, activating and adhesion signals by which adhesion signaling functions as a link through which inhibitory and activating signaling is functionally connected.
Uneducated or immature?
Because MHC-I-specific inhibitory receptors, such as Ly49C/I and KIRs, are highly expressed on terminally mature CD11b+CD27− NK cells,67, 68 a debate exists regarding whether the hypo-responsive, uneducated NK cells are actually at an immature stage of development. Recent studies demonstrate that either MHC-I- or Ly49-deficient mice contain a normal number of terminally mature NK cells.1, 11 In addition, hypo-responsive NK cells from mice deficient in the expression of H2M3, CD1d1, 2B4, CD48, SLAMF6, NKRP1-B, Clr-b or even TIGIT,26, 32, 35, 69, 70, 71 which are either nonclassical MHC-dependent or non-MHC-dependent receptors, likewise undergo normal development and exhibit regular expression of activating receptors, for example, NKG2D and activating Ly49 receptors. Thus, although functionally impaired, uneducated NK cells develop normally and exhibit the characteristics of a mature NK cell.
Cis, trans or both?
Inhibitory Ly49 receptor family members recognize MHC-I in both a cis (receptor and ligand expressed on the same cell) and trans (receptor and ligand expressed on different cells) manner.72, 73 To address which manner of recognition is relevant to NK cell education, two animal models of Ly49A variants were generated. The Ly49-72A variant, which recognizes Dd in trans but not in cis, inhibits the reactivity of NK cells upon ligation but fails to educate NK cells.74 Although this outcome highlights the importance of the cis interaction, it is contrary to results obtained from adoptive transfer experiments in which uneducated NK cells gain functional competence after transfer into MHC-I-sufficient hosts13, 75 and educated NK cells become functionally defective after transfer into an MHC-I-deficient environment.13, 76 More work is required to clarify this somewhat undefined cis model. The studies of another Ly49A variant, Ly49A Δa1, which interacts with Dd in cis but not in trans, suggests a favorable effect of trans interaction in NK cell education, as NK cells are rendered hypo-responsive in this variant.77 The recent finding that NK cells from both NK cell-specific and T cell-specific H-2Dd-deficient mice show defective functional competence77 suggests that both cis and trans interactions might contribute to the process of NK cell education. Cis interactions might provide a relatively short sequestration of unengaged inhibitory receptors to lower the activation threshold,78 while trans interactions might transmit a relatively durable signal to balance the chronic disarming signals during NK education.
Studies using in vivo adoptive transfer assays and in vitro mixing experiments have shown that trans interactions are insufficient for SLAM receptors to confer an education effect.30 However, due to the lack of animal models of receptor variants, no additional details have been revealed regarding the impacts of cis or trans interactions on MHC-independent education. Nevertheless, the expression patterns of self-inhibitory ligands provide some clues to as which mode of interaction is important. Because some ligands, for example, CD48 and Clr-b, are expressed on both NK cells and non-NK cells, education via these specific ligands probably involves the cooperation of both cis and trans interactions. Other self-ligands that are hardly expressed on NK cells, for example, H2-M3, Qa1 and CD1d, would only allow interactions in trans to initiate an educational procedure.
Reversal of NK cell education in tumor and virus infection
Resetting responsiveness of uneducated NK cells
Uneducated NK cells, which are presumed to be hypo-responsive, restore functional competence in inflammatory microenvironments, like viral and bacterial infections and certain tumor invasions.1, 5, 10, 79 During infection of murine cytomegalovirus (MCMV), the uneducated NK cells, and not the educated ones, dominantly control the infection.80 One possible explanation for this observation is that cytokine stimulation increases the expression of inhibitory receptors and therefore a large population of NK cells are re-educated.81 Another explanation is that education deficiency can be masked by a strong activation. To illustrate this phenomenon, a resetting response is triggered by strong signals following NKG2D ligation in which uneducated Ly49-deficient NK cells lyse RMA-S-Rae-1 tumor cells as efficiently as educated wild-type NK cells.11 Overall, NK cell education is a plastic process that adapts to environmental changes, and this adaptation may explain the existence of responsive NK cell subsets lacking inhibitory receptors specific for self MHC-I molecules.82
Ligation of inhibitory receptors offsets education benefits
To further elucidate the effects of education on NK cell reactivity, it is necessary to determine whether target cells express ligands specific for inhibitory receptors. Inhibitory receptors that drive the education of NK cells are not always helpful, especially when self-specific ligands are sufficient on target cells. Licensed NK cells expressing Ly49C/I fail to respond to MHC-sufficient RMA tumor cells.80 Moreover, in certain viral infections such as MCMV and influenza, the educating receptor Ly49C/I does not potentiate strong responses but instead inhibits the proliferation and activation of NK cells.80, 83, 84 Similarly, when potential targets, for example, autoreactive T cells or tumor cells, maintain a high expression of nonclassical MHC Qa-1 or HLA-E, NKG2A-mediated education is not beneficial for NK cell effector function and conversely inhibits NK cell responses.85, 86, 87 Furthermore, the high expression of ligands specific for non-MHC inhibitory receptors, once engaged, triggers dominant negative signals to inhibit NK cell reactivity. The Clr-b-NKRP1-B axis, as an example, is critical for the immune escape of spontaneous myc-induced B lymphomas.32 In light of these observations, functional benefits obtained from education are apparent only when potential targets lack the expression of self-educating ligands.83, 88 Thus, two roles for inhibitory receptors are shown. First, inhibitory receptors trigger beneficial educational impacts to enable NK cells to sense a missing self. Second, inhibitory receptors serve as a negative regulator to limit the reactivity of NK cells upon ligation (Figure 2b). Accordingly, NK cell therapies based on the blockade of inhibitory receptors have attracted increasing attention in recent years. The anti-KIR blocking antibody, for example, relieves educated NK cells from MHC-I-mediated inhibition and provides a promising therapeutic approach for tumor treatment.88, 89, 90
Concluding remarks
Increasing evidence has demonstrated that both MHC-I molecules and the nonclassical MHC-I, or even non-MHC-I, molecules are involved in NK cell education. Interestingly, inhibitory receptors mediate positive signals for NK cells to achieve functional competence, whereas continuous activating signals induce NK cell hypo-responsiveness. Moreover, adhesion molecules link inhibitory and activating receptors together to form confined compartmentalization that ensures NK cell reactivity. However, distinct signals to illuminate MHC-I-independent education theory require further investigation. Another significant finding is that both cis-interactions and trans interactions contribute to NK cell education. Nevertheless, cell type specific education needs to be further clarified. In addition, elucidating how the education of NK cells adapts to its environment, particularly in tumor invasion and virus infection, can provide valuable therapeutic implications.
Acknowledgments
This work was supported by the Ministry of Science & Technology of China (973 Basic Science Project) #2013CB944902 (ZT), Natural Science Foundation of China #81361120388 and #31390433 (ZT), and Key Program of the Chinese Academy of Sciences (KJZD-EW-L10 -003) (RS, ZT).
Footnotes
The authors declare no conflict of interest.
References
- Kim S, Poursine-Laurent J, Truscott S, Lybarger L, Song Y-J, Yang L et al. Licensing of natural killer cells by host major histocompatibility complex class I molecules. Nature 2005; 436: 709–713. [DOI] [PubMed] [Google Scholar]
- Raulet DH, Vance RE. Self-tolerance of natural killer cells. Nat Rev Immunol 2006; 6: 520–531. [DOI] [PubMed] [Google Scholar]
- Anfossi N, André P, Guia S, Falk CS, Roetynck S, Stewart CA et al. Human NK cell education by inhibitory receptors for MHC Class I. Immunity 2006; 25: 331–342. [DOI] [PubMed] [Google Scholar]
- Yokoyama WM, Kim S. Licensing of natural killer cells by self-major histocompatibility complex class I. Immunol Rev 2006; 214: 143–154. [DOI] [PubMed] [Google Scholar]
- Elliott JM, Yokoyama WM. Unifying concepts of MHC-dependent natural killer cell education. Trends Immunol 2011; 32: 364–372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Höglund P, Brodin P. Current perspectives of natural killer cell education by MHC class I molecules. Nat Rev Immunol 2010; 10: 724–734. [DOI] [PubMed] [Google Scholar]
- Kadri N, Luu Thanh T, Höglund P. Selection, tuning, and adaptation in mouse NK cell education. Immunol Rev 2015; 267: 167–177. [DOI] [PubMed] [Google Scholar]
- Goodridge JP, Önfelt B, Malmberg K-JJ. Newtonian cell interactions shape natural killer cell education. Immunol Rev 2015; 267: 197–213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez NC, Treiner E, Vance RE, Jamieson AM, Lemieux S, Raulet DH. A subset of natural killer cells achieves self-tolerance without expressing inhibitory receptors specific for self-MHC molecules. Blood 2005; 105: 4416–4423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez NC. A subset of natural killer cells achieves self-tolerance without expressing inhibitory receptors specific for self-MHC molecules. Blood 2005; 105: 4416–4423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bélanger S, Tu M, Rahim M, Mahmoud A, Patel R, Tai L-H et al. Impaired natural killer cell self-education and "missing-self" responses in Ly49-deficient mice. Blood 2012; 120: 592–602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi T, Ferris S, Matsumoto N, Poursine-Laurent J, Yokoyama W. Ly49-dependent NK cell licensing and effector inhibition involve the same interaction site on MHC ligands. J Immunol 2011; 186: 3911–3917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebihara T, Jonsson A, Yokoyama W. Natural killer cell licensing in mice with inducible expression of MHC class I. Proc Natl Acad Sci USA 2013; 110: E4232–E4237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taveirne S, Filtjens J, Van Ammel E, De Colvenaer V, Kerre T, Taghon T et al. Inhibitory receptors specific for MHC class I educate murine NK cells but not CD8αα intestinal intraepithelial T lymphocytes. Blood 2011; 118: 339–347. [DOI] [PubMed] [Google Scholar]
- Braud VM, Allan DSJ, O'Callaghan CA, Söderström K. HLA-E binds to natural killer cell receptors CD94/NKG2A, B and C. Nature 1998; 391: 795–799. [DOI] [PubMed] [Google Scholar]
- Yawata M, Yawata N, Draghi M, Partheniou F, Little A-M, Parham P. MHC class I-specific inhibitory receptors and their ligands structure diverse human NK-cell repertoires toward a balance of missing self-response. Blood 2008; 112: 2369–2380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivarsson M, Loh L, Marquardt N, Kekäläinen E, Berglin L, Björkström N et al. Differentiation and functional regulation of human fetal NK cells. J Clin Invest 2013; 123: 3889–3901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lisovsky I, Isitman G, Song R, DaFonseca S, Tremblay-McLean A, Lebouché B et al. A higher frequency of NKG2A+ than NKG2A- NK cells respond to autologous HIV-infected CD4 cells irrespective of whether they co-express KIR3DL1. J Virol 2015; 89: 9909–9919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews D, Sullivan L, Baschuk N, Chan C, Berry R, Cotterell C et al. Recognition of the nonclassical MHC class I molecule H2-M3 by the receptor Ly49A regulates the licensing and activation of NK cells. Nat Immunol 2012; 13: 1171–1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Held W, Nonclassical NK. cell education. Nat Immunol 2012; 13: 1135–1137. [DOI] [PubMed] [Google Scholar]
- Chang C, Brossay L, Kronenberg M, Kane K. The murine nonclassical class I major histocompatibility complex-like CD1.1 molecule protects target cells from lymphokine-activated killer cell cytolysis. J Exp Med 1999; 189: 483–491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang M, Borszcz P, Sidobre S, Kronenberg M, Kane K. CD1d1 displayed on cell size beads identifies and enriches an NK cell population negatively regulated by CD1d1. J Immunol 2004; 172: 5304–5312. [DOI] [PubMed] [Google Scholar]
- Wickström S, Oberg L, Kärre K, Johansson M. A genetic defect in mice that impairs missing self recognition despite evidence for normal maturation and MHC class I-dependent education of NK cells. J Immunol 2014; 192: 1577–1586. [DOI] [PubMed] [Google Scholar]
- Lee K-MM, McNerney ME, Stepp SE, Mathew PA, Schatzle JD, Bennett M et al. 2B4 acts as a non-major histocompatibility complex binding inhibitory receptor on mouse natural killer cells. J Exp Med 2004; 199: 1245–1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waggoner SN, Taniguchi RT, Mathew PA, Kumar V, Welsh RM. Absence of mouse 2B4 promotes NK cell-mediated killing of activated CD8+ T cells, leading to prolonged viral persistence and altered pathogenesis. J Clin Invest 2010; 120: 1925–1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JE, Lim SA, Kim T-JJ, Kim K, Ng J, Kim YH et al. NKG2D ligation relieves 2B4-mediated NK-cell self-tolerance in mice. Eur J Immunol 2014; 44: 1802–1813. [DOI] [PubMed] [Google Scholar]
- Lee K-MM, Forman JP, McNerney ME, Stepp S, Kuppireddi S, Guzior D et al. Requirement of homotypic NK-cell interactions through 2B4(CD244)/CD48 in the generation of NK effector functions. Blood 2006; 107: 3181–3188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong Z, Davidson D, Perez-Quintero LA, Kurosaki T, Swat W, Veillette A. The adaptor SAP controls NK cell activation by regulating the enzymes Vav-1 and SHIP-1 and by enhancing conjugates with target cells. Immunity 2012; 36: 974–985. [DOI] [PubMed] [Google Scholar]
- Meinke S, Watzl C. NK cell cytotoxicity mediated by 2B4 and NTB-A is dependent on SAP acting downstream of receptor phosphorylation. Front Immunol 2013; 4: 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu N, Zhong MC, Roncagalli R, Perez-Quintero LA, Guo H, Zhang Z et al. A hematopoietic cell-driven mechanism involving SLAMF6 receptor, SAP adaptors and SHP-1 phosphatase regulates NK cell education. Nat Immunol 2016; 17: 387–396. [DOI] [PubMed] [Google Scholar]
- Carlyle JR, Martin A, Mehra A, Attisano L, Tsui FW, Zúñiga-Pflücker JC. Mouse NKR-P1B, cnovel NK1.1 antigen with inhibitory function. J Immunol 1999; 162: 5917–5923. [PubMed] [Google Scholar]
- Rahim MM, Chen P, Mottashed AN, Mahmoud AB, Thomas MJ, Zhu Q et al. The mouse NKR-P1B:Clr-b recognition system is a negative regulator of innate immune responses. Blood 2015; 125: 2217–2227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aust JG, Gays F, Mickiewicz KM, Buchanan E, Brooks CG. The expression and function of the NKRP1 receptor family in C57BL/6 mice. J Immunol 2009; 183: 106–116. [DOI] [PubMed] [Google Scholar]
- Voigt S, Mesci A, Ettinger J, Fine JH, Chen P, Chou W et al. Cytomegalovirus evasion of innate immunity by subversion of the NKR-P1B:Clr-b missing-self axis. Immunity 2007; 26: 617–627. [DOI] [PubMed] [Google Scholar]
- Chen P, Aguilar OA, Rahim MM, Allan DS, Fine JH, Kirkham CL et al. Genetic investigation of MHC-independent missing-self recognition by mouse NK cells using an in vivo bone marrow transplantation model. J Immunol 2015; 194: 2909–2918. [DOI] [PubMed] [Google Scholar]
- Carlyle JR, Jamieson AM, Gasser S, Clingan CS, Arase H, Raulet DH. Missing self-recognition of Ocil/Clr-b by inhibitory NKR-P1 natural killer cell receptors. Proc Natl Acad Sci USA 2004; 101: 3527–3532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X, Harden K, Gonzalez LC, Francesco M, Chiang E, Irving B et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol 2009; 10: 48–57. [DOI] [PubMed] [Google Scholar]
- Stanietsky N, Rovis TL, Glasner A, Seidel E, Tsukerman P, Yamin R et al. Mouse TIGIT inhibits NK-cell cytotoxicity upon interaction with PVR. Eur J Immunol 2013; 43: 2138–2150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bi J, Zheng X, Chen Y, Wei H, Sun R, Tian Z. TIGIT safeguards liver regeneration through regulating natural killer cell-hepatocyte crosstalk. Hepatology 2014; 60: 1389–1398. [DOI] [PubMed] [Google Scholar]
- Bi J, Zhang Q, Liang D, Xiong L, Wei H, Sun R et al. T-cell Ig and ITIM domain regulates natural killer cell activation in murine acute viral hepatitis. Hepatology 2014; 59: 1715–1725. [DOI] [PubMed] [Google Scholar]
- Abeyweera T, Kaissar M, Huse M. Inhibitory receptor signaling destabilizes immunological synapse formation in primary NK cells. Front Immunol 2013; 4: 410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viant C, Fenis A, Chicanne G, Payrastre B, Ugolini S, Vivier E. SHP-1-mediated inhibitory signals promote responsiveness and anti-tumour functions of natural killer cells. Nat Commun 2014; 5: 5108. [DOI] [PubMed] [Google Scholar]
- Lowin-Kropf B, Kunz B, Beermann F, Held W. Impaired natural killing of MHC class I-deficient targets by NK cells expressing a catalytically inactive form of SHP-1. J Immunol 2000; 165: 1314–1321. [DOI] [PubMed] [Google Scholar]
- Wahle JA, Paraiso KH, Kendig RD, Lawrence HR, Chen L, Wu J et al. Inappropriate recruitment and activity by the Src homology region 2 domain-containing phosphatase 1 (SHP1) is responsible for receptor dominance in the SHIP-deficient NK cell. J Immunol 2007; 179: 8009–8015. [DOI] [PubMed] [Google Scholar]
- Wang J-WW, Howson JM, Ghansah T, Desponts C, Ninos JM, May SL et al. Influence of SHIP on the NK repertoire and allogeneic bone marrow transplantation. Science 2002; 295: 2094–2097. [DOI] [PubMed] [Google Scholar]
- Oppenheim D, Roberts S, Clarke S, Filler R, Lewis J, Tigelaar R et al. Sustained localized expression of ligand for the activating NKG2D receptor impairs natural cytotoxicity in vivo and reduces tumor immunosurveillance. Nat Immunol 2005; 6: 928–937. [DOI] [PubMed] [Google Scholar]
- Coudert J, Scarpellino L, Gros F, Vivier E, Held W. Sustained NKG2D engagement induces cross-tolerance of multiple distinct NK cell activation pathways. Blood 2008; 111: 3571–3578. [DOI] [PubMed] [Google Scholar]
- Tripathy SK, Keyel PA, Yang L, Pingel JT, Cheng TP, Schneeberger A et al. Continuous engagement of a self-specific activation receptor induces NK cell tolerance. J Exp Med 2008; 205: 1829–1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fauriat C, Ivarsson M, Ljunggren H-G, Malmberg K-J, Michaëlsson J. Education of human natural killer cells by activating killer cell immunoglobulin-like receptors. Blood 2010; 115: 1166–1174. [DOI] [PubMed] [Google Scholar]
- Sun J, Lanier L. Tolerance of NK cells encountering their viral ligand during development. J Exp Med 2008; 205: 1819–1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazumdar B, Bolanos FD, Tripathy SK. Viral infection transiently reverses activation receptor-mediated NK cell hyporesponsiveness in an MHC class I-independent mechanism. Eur J Immunol 2013; 43: 1345–1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joncker N, Fernandez N, Treiner E, Vivier E, Raulet D. NK cell responsiveness is tuned commensurate with the number of inhibitory receptors for self-MHC class I: the rheostat model. J Immunol 2009; 182: 4572–4580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brodin P, Lakshmikanth T, Johansson S, Kärre K, Höglund P. The strength of inhibitory input during education quantitatively tunes the functional responsiveness of individual natural killer cells. Blood 2009; 113: 2434–2441. [DOI] [PubMed] [Google Scholar]
- Johansson S, Johansson M, Rosmaraki E, Vahlne G, Mehr R, Salmon-Divon M et al. Natural killer cell education in mice with single or multiple major histocompatibility complex class I molecules. J Exp Med 2005; 201: 1145–1155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Béziat V, Traherne J, Liu L, Jayaraman J, Enqvist M, Larsson S et al. Influence of KIR gene copy number on natural killer cell education. Blood 2013; 121: 4703–4707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brodin P, Kärre K, Höglund P. NK cell education: not an on-off switch but a tunable rheostat. Trends Immunol 2009; 30: 143–149. [DOI] [PubMed] [Google Scholar]
- Hadad U, Thauland TJ, Martinez OM, Butte MJ, Porgador A, Krams SM. NKp46 Clusters at the Immune Synapse and Regulates NK Cell Polarization. Front Immunol 2015; 6: 495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagrue K, Carisey A, Morgan DJ, Chopra R, Davis DM. Lenalidomide augments actin remodeling and lowers NK-cell activation thresholds. Blood 2015; 126: 50–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guia S, Jaeger BN, Piatek S, Mailfert S, Trombik T, Fenis A et al. Confinement of activating receptors at the plasma membrane controls natural killer cell tolerance. Sci Signal 2011; 4: ra21. [DOI] [PubMed] [Google Scholar]
- Forslund E, Sohlberg E, Enqvist M, Olofsson PE, Malmberg K-JJ, Önfelt B. Microchip-based single-cell imaging reveals that CD56dimCD57-KIR-NKG2A+ NK cells have more dynamic migration associated with increased target cell conjugation and probability of killing compared to CD56dimCD57-KIR-NKG2A- NK cells. J Immunol 2015; 195: 3374–3381. [DOI] [PubMed] [Google Scholar]
- Barber DF, Faure M, Long EO. LFA-1 contributes an early signal for NK cell cytotoxicity. J Immunol 2004; 173: 3653–3659. [DOI] [PubMed] [Google Scholar]
- Mace EM, Zhang J, Siminovitch KA, Takei F. Elucidation of the integrin LFA-1-mediated signaling pathway of actin polarization in natural killer cells. Blood 2010; 116: 1272–1279. [DOI] [PubMed] [Google Scholar]
- Thomas LM, Peterson ME, Long EO. Cutting edge: NK cell licensing modulates adhesion to target cells. J Immunol 2013; 191: 3981–3985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hou S, Ge K, Zheng X, Wei H, Sun R, Tian Z. CD226 protein is involved in immune synapse formation and triggers Natural Killer (NK) cell activation via its first extracellular domain. J Biol Chem 2014; 289: 6969–6977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enqvist M, Ask EH, Forslund E, Carlsten M, Abrahamsen G, Béziat V et al. Coordinated expression of DNAM-1 and LFA-1 in educated NK cells. J Immunol 2015; 194: 4518–4527. [DOI] [PubMed] [Google Scholar]
- Martinet L, Ferrari De Andrade L, Guillerey C, Lee JS, Liu J, Souza-Fonseca-Guimaraes F et al. DNAM-1 expression marks an alternative program of NK cell maturation. Cell Rep 2015; 11: 85–97. [DOI] [PubMed] [Google Scholar]
- Hayakawa Y, Smyth MJ. CD27 dissects mature NK cells into two subsets with distinct responsiveness and migratory capacity. J Immunol 2006; 176: 1517–1524. [DOI] [PubMed] [Google Scholar]
- Chiossone L, Chaix J, Fuseri N, Roth C, Vivier E, Walzer T. Maturation of mouse NK cells is a 4-stage developmental program. Blood 2009; 113: 5488–5496. [DOI] [PubMed] [Google Scholar]
- Andrews DM, Sullivan LC, Baschuk N, Chan CJ, Berry R, Cotterell CL et al. Recognition of the nonclassical MHC class I molecule H2-M3 by the receptor Ly49A regulates the licensing and activation of NK cells. Nat Immunol 2012; 13: 1171–1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickström SL, Öberg L, Kärre K, Johansson MH. A genetic defect in mice that impairs missing self recognition despite evidence for normal maturation and MHC class I-dependent education of NK cells. J Immunol 2014; 192: 1577–1586. [DOI] [PubMed] [Google Scholar]
- Assarsson E, Kambayashi T, Persson CM, Chambers BJ, Ljunggren H-GG. 2B4/CD48-mediated regulation of lymphocyte activation and function. J Immunol 2005; 175: 2045–2049. [DOI] [PubMed] [Google Scholar]
- Scarpellino L, Oeschger F, Guillaume P, Coudert J, Lévy F, Leclercq G et al. Interactions of Ly49 family receptors with MHC class I ligands in trans and cis. J Immunol 2007; 178: 1277–1284. [DOI] [PubMed] [Google Scholar]
- Back J, Malchiodi E, Cho S, Scarpellino L, Schneider P, Kerzic M et al. Distinct conformations of Ly49 natural killer cell receptors mediate MHC class I recognition in trans and cis. Immunity 2009; 31: 598–608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chalifour A, Scarpellino L, Back J, Brodin P, Devevre E, Gros F et al. A Role for cis Interaction between the Inhibitory Ly49A receptor and MHC class I for natural killer cell education. Immunity 2009; 30: 337–347. [DOI] [PubMed] [Google Scholar]
- Elliott J, Wahle J, Yokoyama W. MHC class I-deficient natural killer cells acquire a licensed phenotype after transfer into an MHC class I-sufficient environment. J Exp Med 2010; 207: 2073–2079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joncker N, Shifrin N, Delebecque F, Raulet D. Mature natural killer cells reset their responsiveness when exposed to an altered MHC environment. J Exp Med 2010; 207: 2065–2072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bessoles S, Angelov G, Back J, Leclercq G, Vivier E, Held W. Education of murine NK cells requires both cis and trans 'recognition of MHC class I molecules. J Immunol 2013; 191: 5044–5051. [DOI] [PubMed] [Google Scholar]
- Back J, Chalifour A, Scarpellino L, Held W. Stable masking by H-2Dd cis ligand limits Ly49A relocalization to the site of NK cell/target cell contact. Proc Natl Acad Sci USA 2007; 104: 3978–3983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orr MT, Lanier LL. Natural killer cell licensing during viral infection. Adv Exp Med Biol 2011; 780: 37–44. [DOI] [PubMed] [Google Scholar]
- Orr M, Murphy W, Lanier L. 'Unlicensed' natural killer cells dominate the response to cytomegalovirus infection. Nat Immunol 2010; 11: 321–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juelke K, Killig M, Thiel A, Dong J, Romagnani C. Education of hyporesponsive NK cells by cytokines. Eur J Immunol 2009; 39: 2548–2555. [DOI] [PubMed] [Google Scholar]
- Bessoles S, Grandclement C, Alari-Pahissa E, Gehrig J, Jeevan-Raj B, Held W. Adaptations of natural killer cells to self-MHC class I. Front Immunol 2014; 5: 349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mario-Ernesto C-M, André V, Do NK. cells always need a license to kill? Nat Immunol 2010; 11: 279–280. [DOI] [PubMed] [Google Scholar]
- Mahmoud AB, Tu MM, Wight A, Zein HS, Rahim MM, Lee SH et al. Influenza virus targets class I MHC-educated NK cells for immunoevasion. PLoS Pathog 2016; 12: e1005446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu L, Ikizawa K, Hu D, Werneck M, Wucherpfennig KW, Cantor H. Regulation of activated CD4+ T cells by NK cells via the Qa-1–NKG2A inhibitory pathway. Immunity 2007; 26: 593–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li F, Wei H, Wei H, Gao Y, Xu L, Yin W et al. Blocking the natural killer cell inhibitory receptor NKG2A increases activity of human natural killer cells and clears hepatitis B virus infection in mice. Gastroenterology 2013; 144: 392–401. [DOI] [PubMed] [Google Scholar]
- Peng H, Wisse E, Tian Z. Liver natural killer cells: subsets and roles in liver immunity. Cell Mol Immunol 2016; 13: 328–336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baptiste NJ, Eric V. When NK cells overcome their lack of education. J Clin Invest 2012; 122: 3053–3056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sola C, Andre P, Lemmers C, Fuseri N, Bonnafous C, Blery M et al. Genetic and antibody-mediated reprogramming of natural killer cell missing-self recognition in vivo. Proc Natl Acad Sci USA 2009; 106: 12879–12884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun C, Sun H, Zhang C, Tian Z. NK cell receptor imbalance and NK cell dysfunction in HBV infection and hepatocellular carcinoma. Cell Mol Immunol 2015; 12: 292–302. [DOI] [PMC free article] [PubMed] [Google Scholar]