Abstract
The incidence of myopia is constantly on the rise. Patients of high myopia and pathological myopia are young and can lose vision due to a number of degenerative changes occurring at the macula. With the emergence of new technologies such as swept-source optical coherence tomography (OCT) and OCT angiography, our understanding of macular pathology in myopia has improved significantly. New conditions such as myopic traction maculopathy have been defined. Early, noninvasive detection of myopic choroidal neovascularization and its differentiation from lacquer cracks is possible with a greater degree of certainty. We discuss the impact of these new exciting and promising technologies and management of macular pathology in myopia. Incorporation of OCT in the microscope has also improved macular surgery. New concepts such as fovea-sparing internal limiting membrane peeling have emerged. A review of literature and our experience in managing all these conditions are discussed.
Key words: Choroidal neovascular membrane, high myopia, intraoperative optical coherence tomography, lacquer crack, myopic traction maculopathy, optical coherence tomography angiography, pathological myopia, posterior staphyloma, swept-source optical coherence tomography
High myopia (HM) is an important cause of visual loss, especially in the younger population. It has been defined as a refractive error with spherical equivalent exceeding −6 diopters (D) and/or the axial length longer than 26.5 mm.[1,2] Pathological myopia (PM) or degenerative myopia refers to high axial myopia with characteristic pathological changes at the posterior pole.[3,4,5] Typical pathological features include diffuse or patchy chorioretinal atrophy, posterior staphyloma, lacquer cracks, Fuchs spots, choroidal neovascular membrane (CNV), and sometimes even foveoschisis.[6,7] These pathologic changes often lead to progressive loss of vision.[8] Literature search for this document has been performed using appropriate keywords in PubMed.
Epidemiology
PM is more prevalent in the Asian population than among other racial groups. The prevalence varies from 1% to 4% in the general population.[5,9,10] It is estimated to be 3.1% in China,[9] 1.74% in Japan,[11] and 1.2% in Australia.[10] PM is the leading cause of blindness in Japan and is the second most common cause in Denmark[12] and China.[13] In the West also, PM is among the leading causes of legal blindness.[14] There are no large scale studies in India assessing the prevalence of HM. In a recent study from our center on school-going children in North India, the prevalence of HM was found to be 1.5%.[15] Population-based studies show a higher prevalence of PM in women than in men. The Blue Mountains Eye study[10] and Hisayama study[11] reported a prevalence of 0.4% and 2.2% in women, respectively, while in men, it was 0.06% and 1.2%, respectively.
Genetics and Environmental Effects
HM seems to be caused by a complex combination of environmental and genetic factors.[16,17] There is enough evidence to suggest that the genetic loci leading to HM are heterogeneous[18,19] and these may contribute to varying degrees of myopia.[20,21] The inheritance of HM can vary from being autosomal dominant, autosomal recessive, X-linked recessive to a monogenic pattern.[21] It may also be associated with certain genetic syndromes: Stickler syndromes Type 1 and 2, Type 4 Ehlers–Danlos syndrome, Knobloch syndrome, Marfan syndrome, Noonan and Down's syndrome.
Refractive status of the parents plays an important role in the development of HM.[22,23] Twin studies report high heritability values of up to 90%.[24] Currently, the most studied environmental parameter thought to be protective against development of myopia is time spent by children outdoors.[25,26,27]
Pathophysiology
Pathological changes in high myopes start in childhood and become prominent in adulthood.[28] To begin with, there occurs excessive axial elongation.[29] Axial elongation results in chorioretinal stretching and subsequent thinning.
The mechanism behind pathological axial elongation includes an emmetropization process and involves a structural alteration of the collagen proteins.[30,31] Abnormal collagen proteins may lead to degenerative changes in the retina, choroid, and sclera.[31] Studies have hypothesized the role of choroid in ocular elongation in response to retinal defocus.[32,33,34] As a consequence of defocus, choroid thickening or thinning occurs, moving the retina toward the plane of focus.[33] There may exist a chain of molecular signals arising from the retina which modulate the changes in thickness of the choroid and the scleral growth.[34] The amacrine cells of the retina are thought to play a critical role in this chain of molecular signaling, initiated due to a retinal defocus, which leads to alteration of refractive error.[34]
Myopia-related complications such as posterior staphyloma and chorioretinal atrophy increase proportionally with increase in axial length.[4] Thinned out chorioretinal tissue is associated with poor blood circulation and may lead to CNV development by inducing vascular endothelial growth factor (VEGF) expression.[28,35] Furthermore, scleral thinning may cause deformation of the posterior pole leading to staphyloma formation with a shorter radius of curvature.
Visual acuity (VA) in HM may be subnormal even before advanced myopic maculopathy sets in. One of the reasons behind this may be the alteration in the arrangement of photoreceptors. The arrangement of the photoreceptors in high myopes is affected due to excessive stretch in the posterior pole.[36] This may lead to subnormal visual function (Stiles–Crawford effect). In high myopes, the cones in nasal hemiretina are aligned toward the optic nerve, whereas they are aligned toward the center of the exit pupil in temporal hemiretina. This discrepancy in receptor alignment is directly associated with the axial length.[36]
Features of Pathological Myopia
The early features of excess axial elongation include myopic conus, super traction, and tessellation of fundus.
Myopic conus/crescent
Peripapillary scleral expansion leads to a sharply defined concentric area of depigmentation adjacent to the optic disc where the inner surface of sclera is visible [Fig. 1a]. It occurs due to a disparity between the area of the sclera and the retinal pigment epithelium (RPE)-choriocapillaris complex and a premature termination of this complex ahead of the optic disc edge. It can either be a scleral crescent alone or choroidal crescent or both.[37] Based on the extent, it can be a temporal conus, nasal conus, inferior conus, or annular conus. Temporal conus is reported to be the most frequent type.[4]
Figure 1.
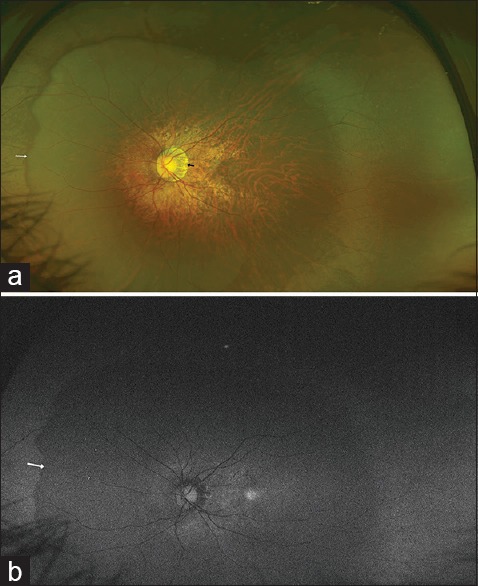
(a) A large myopic conus is seen temporal to the disc (black arrow). A large posterior staphyloma is also evident. The nasal edge of the staphyloma appears as a dark crescent (white arrow). (b) An Optos autofluorescence image of the fundus showing a difference in the autofluorescence signal between the area of the staphyloma and the rest of the peripheral fundus. The white arrow marks the boundary of the staphyloma
Super traction
It occurs due to dragging of retinochoroidal tissue over the nasal edge of optic disc with expansion of posterior pole.
Tessellation
Generalized depigmentation due to RPE atrophy leads to a tigroid appearance of the fundus [Fig. 2].
Figure 2.
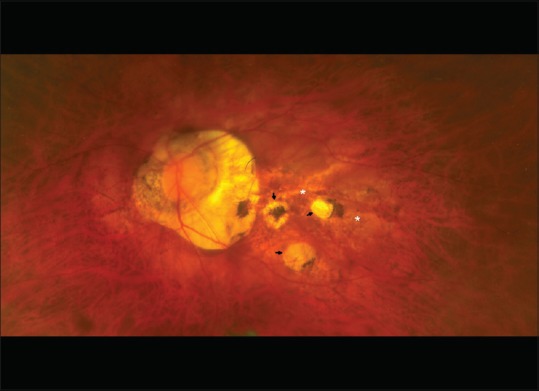
Due to the generalized depigmentation, the entire fundus appears tessellated (tigroid appearance). The areas of generalized atrophy do not have well-defined margins (white asterix). Additional focal areas of atrophy with well-defined margins are also evident (black arrows)
Posterior Staphyloma (Scarpa's Staphyloma)
It is an outward protrusion of all coats of the posterior pole and is considered pathognomonic of PM. Spaide defined a posterior staphyloma as “outpouching of the wall of the eye that has a radius of curvature less than the surrounding radii of curvature.”[38] The best way to diagnose a posterior staphyloma is by an indirect ophthalmoscope, which gives a stereoscopic view of the fundus. It is seen as a secondary depression with bending of vessels at the margin and a dark crescentic nasal reflex [Fig. 1a]. There are ten different types of staphyloma according to Curtin.[39] Its incidence increases with age, occurring mostly after the fifth decade.[40] Posterior staphyloma formation is further linked to development of myopic maculopathy.[3] The eyes with shallow staphylomas show a larger drop in VA and a greater occurrence of CNV and macular hemorrhage.[41] The normal choroidal flush is absent in a high percentage of posterior staphylomas, suggesting the possibility of ischemia which may further lead to development of a CNV in these eyes.[42] A newer classification of staphylomas based on three-dimensional magnetic resonance imaging and ultrawide field imaging has also been proposed.[43] The presence of pigment abnormalities, abnormal autofluorescence [Fig. 1b], and abnormal reflectance in infrared imaging, performed using wide field imaging systems such as Optos ™, have been proposed as markers to identify the edge of the staphyloma.[43]
Macular Changes
Clinically, there appears to be a pattern in the progression of myopic maculopathy. It ranges from an early appearance of a tessellated fundus to progressive development of diffuse atrophy and lacquer cracks, followed by progression to patchy atrophy. CNV generally develops adjacent to the area of patchy atrophy or lacquer cracks.
Recently, PM has been classified by the “META-analysis for Pathologic Myopia” study based on the presence of characteristic features of myopic maculopathy.[7] PM is diagnosed only when fundus findings are consistent with Category 2 and above [Table 1].
Table 1.
Pathological myopia as classified by “META-analysis for Pathologic Myopia” study

Macular Chorioretinal Atrophy
Chorioretinal atrophy occurs due to progressive thinning of the choroid, disappearance of choroidal vessels, and loss of RPE and photoreceptors.[28] The cause behind atrophy is probably choroidal vascular occlusion and abiotrophic degeneration. Chorioretinal atrophy is of two types – diffuse atrophy and patchy atrophy.[7] Diffuse atrophy appears as yellowish-white areas of atrophy with ill-defined borders [Fig. 2] while patchy atrophy is grayish-white, well-defined area of atrophy, and produces an absolute scotoma on visual fields [Fig. 2]. Patchy atrophy predisposes to CNV development.[44]
Choroidal thinning is an important feature of HM. Enhanced-depth imaging optical coherence tomography (OCT) shows a very thin choroid in highly myopic eyes, which progressively gets thinner with increasing age and degree of myopia.[45]
Lacquer Cracks
Lacquer cracks are breaks in the Bruch's membrane at the macula in highly myopic eyes [Fig. 3a], usually associated with a posterior staphyloma. These appear as multiple yellowish-white irregular lines, usually horizontally oriented, and coursing the posterior pole. Lacquer cracks can be linear or stellate, and sometimes show branching, crisscrossing, or both. These occur more commonly in males and decrease with aging. On fluorescein angiography, they lead to a window defect and thus appear as hyperfluorescent tracks without any leakage [Fig. 3b].[46] On fundus autofluorescent imaging, these appear hypoautofluorescent. Similarly, they appear hypofluorescent on indocyanine green angiography.[47] The same can also be identified as tracks of lack of a decorrelation signal at the level of choriocapillaris due to disruption of flow on OCT angiography (OCTA) [Fig. 3c].
Figure 3.
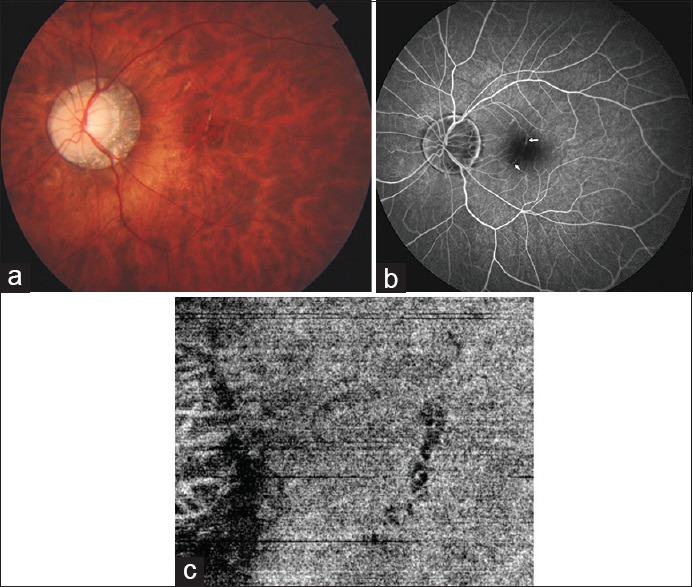
(a) Fundus image showing a well-defined curved hypopigmented line at the fovea suggestive of a lacquer crack. (b) Fundus fluorescein angiogram highlighting the lacquer crack as a hyperfluorescent line (white arrow). (c) Optical coherence tomography angiography image showing the lacquer crack as a black curved line due to lack of flow detected as an area of lack of decorrelation signal
Fresh lacquer formation may be associated with subretinal bleeding in the absence of a choroidal neovascularization.[48] Lacquer cracks are also frequently associated with formation of a choroidal neovascularization at the margins or in adjacent areas.[44]
Förster-Fuchs' Spot
Förster-Fuchs' spot is a raised, pigmented, round, or elliptical lesion that is predominantly dark but can have a gray, yellow, red, or green hue. It is named after Ernst Fuchs, who described a pigmented lesion in 1901, and Carl Förster, who described neovascularization of the retina in 1862. Forster-Fuchs' spots arise due to proliferation of RPE associated with choroidal hemorrhage.[49] These are primarily small scars formed following degeneration and neovascularization related to HM.
Myopic Choroidal Neovascular Membrane
Macular CNV is a one of the most common complications that results in reduced central vision in patients with PM.[50,51] Myopic CNV develops in 10% of high myopes and 30% myopes eventually develop CNV in the other eye as well.[44] It appears as a grayish subretinal membrane with hyperpigmented borders [Fig. 4a]. As the retina is thin, bleeding does not usually obscure these lesions and they are easily visible on clinical examination. Fluorescein angiography [Fig. 4c], indocyanine green angiography [Fig. 4d], OCT, and OCTA [Fig. 4b] are helpful in confirming the diagnosis. OCT shows a hyper-reflective-elevated lesion in the subretinal space, usually without much exudative changes such as fluid under the neurosensory retina or intraretinal edema. These are generally Type 2 CNVs. Myopic CNV are predominantly classic type on fluorescein angiography. They appear as well-defined areas of hyperfluorescence in the early phase with progressive leakage of dye in the late phases of the angiogram.[52,53]
Figure 4.
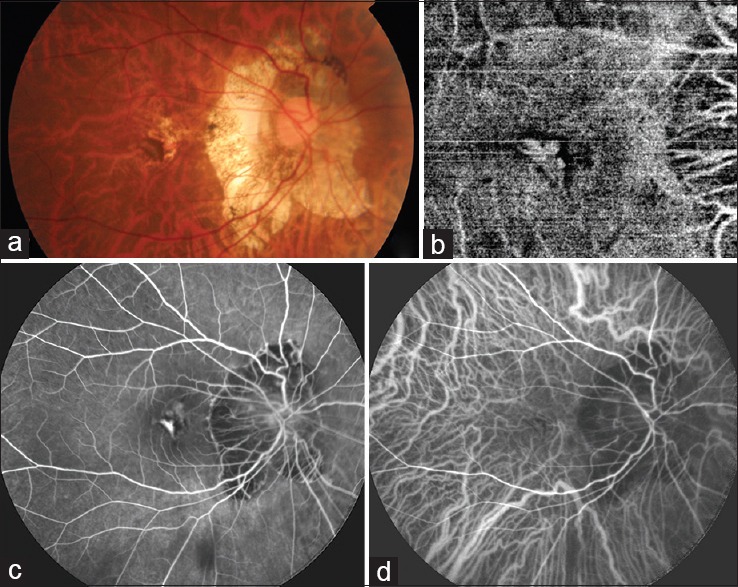
(a) Fundus image of a myopic patient showing a grayish lesion at the fovea with pigmented margins, suggestive of a choroidal neovascular membrane. (b) Optical coherence tomography angiography image of the same patient showing the branching network of vessels of the choroidal neovascular membrane with a surrounding dark halo. (c) Fluorescein angiography showing the hyperfluorescent choroidal neovascular membrane. (d) Indocyanine green angiography also showing the hyperfluorescent choroidal neovascular membrane
OCTA has a definite role in diagnosis of myopic CNV. As subretinal hemorrhage can be caused by both CNV and new lacquer crack formation in eyes with PM, OCTA can help identify CNV noninvasively. Typical lacy wheel pattern, numerous tiny capillaries, widely anastomosed network, and perilesional hypointense halo are features of active CNV on OCTA. Quiescent CNV are characterized by long filamentous linear large mature looking vessels, rare anastomosis and a dead tree appearance. OCTA has been found to successfully detect up to 94.1% of myopic CNVs [Fig. 5a–e].[54]
Figure 5.
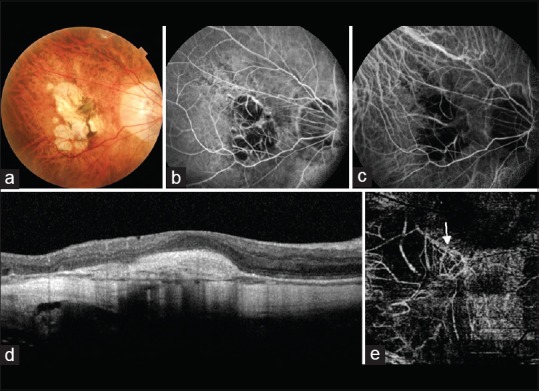
(a) Fundus image of a myopic patient showing a grayish membrane at the fovea clinically suggestive of a choroidal neovascular membrane. (b) The gray membrane appears hyperfluorescent of fundus fluorescein angiography. However, the hyperfluorescence is not as marked as is seen in classic choroidal neovascular membranes. (c) Indocyanine green angiography fails to detect the choroidal neovascular membrane. (d) Optical coherence tomography shows a spindle-shaped hyper-reflective area above the retinal pigment epithelium suggestive of a choroidal neovascular membrane. There is absence of associated subretinal or intraretinal fluid. (e) An abnormal branching network of vessels (white arrow) is visible on the optical coherence tomography angiography image segmented at the deep retinal level. This is suggestive of a Type 2 choroidal neovascular membrane
Focal chorioretinal atrophy, steeper posterior staphyloma, and lacquer cracks are thought to be the risk factors for development of myopic CNV.[3,51] Myopic CNV progresses through three main stages. In the initial phase, there is direct damage to the photoreceptors, causing central visual loss. Then, as the CNV regresses, a fibrous pigmented scar forms, referred to as Forster–Fuch's spot. Finally, chorioretinal atrophy develops around the regressed CNV, which results in a poor long-term visual outcome.[55]
Photodynamic therapy (PDT), anti-VEGF therapy, and a combination of these have been tried for treatment of myopic CNVm's. The ranibizumab and PDT (verteporfin) evaluation in myopic choroidal neovascularization study,[56] a randomized controlled trial comparing ranibizumab against verteporfin PDT for the treatment of myopic CNV, reported superior VA gains with ranibizumab as compared to PDT. The suspicion of CNV formation should be high in cases of myopia with a history of recent onset of metamorphopsia. The CNV may be detected as an area of leakage on fundus fluorescein angiography or visualized on OCTA. The number and frequency of intravitreal injections required for myopic CNVm's are generally less than those for CNVm's related to age-related macular degeneration. However, these patients do need to be kept under regular monthly follow-up and need to be retreated in case of activity.
Myopic Macular Retinoschisis
Myopic macular retinoschisis or myopic foveoschisis describes a schisis-like thickening of neurosensory retina into a thicker inner layer and a thinner outer layer at the macula in highly myopic eyes with a posterior staphyloma [Fig. 6].[57] Although myopic macular retinoschisis has been alternatively described as myopic traction maculopathy (MTM), MTM in addition includes other conditions such as vitreomacular traction, retinal thickening, lamellar or full-thickness macular hole formation, and foveal detachment.[58] MTM always occurs within a posterior staphyloma. Myopic macular retinoschisis is reported to occur in 9% of highly myopic eyes with posterior staphyloma.[59]
Figure 6.
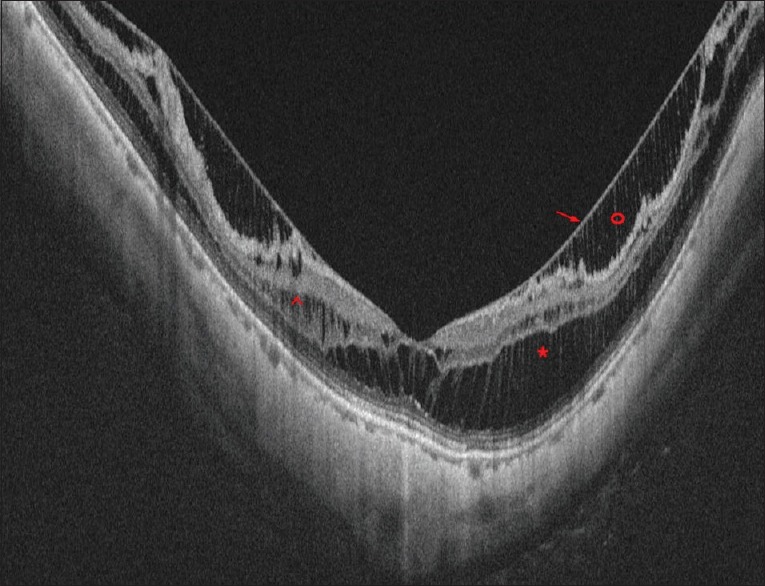
Optical coherence tomography image of a case of myopic maculoschisis with posterior staphyloma. Red * shows the schisis in the outer plexiform layer. Red ^ shows the schisis in the inner nuclear layer. Red o shows the schisis in the nerve fibre layer and the red arrow shows the overlying stretched internal limiting membrane
The pathogenesis behind MTM is probably splitting of retina over time due to relative tautness and noncompliance of the inner retina compared with outer retina within the posterior staphyloma. The split occurs at the level of the external limiting membrane.[60] The tractional mechanisms responsible for inner retinal noncompliance are diverse and include vitreomacular traction from incomplete posterior vitreous detachment (PVD), remnant preretinal cortical vitreous layer after PVD, epiretinal membranes, taut internal limiting membrane (ILM), and shortened and stiff retinal arterioles (vascular microfolds).[58,61,62,63,64,65,66,67]
Visual complaints are minimal and progress gradually. Patients may complain of blurring of vision or distortion of vision (metamorphopsia).[57] Vision loss occurs with outer lamellar hole formation or foveal detachment. Early stages may be easily underestimated by biomicroscopic examination. The diagnosis is confirmed on OCT, which shows the typical splitting of neurosensory retina, bridging columns, and intraretinal cysts [Fig. 6].[57,62] Other OCT findings include epiretinal membrane, retinal microfolds, ILM detachments, and macular hole.[60]
OCT-based progression of myopic foveal retinoschisis to retinal detachment has been described in detail by Shimada et al.[68] Stage 1 shows focal irregularity of the thickness of external retinal layer. In Stage 2, an outer lamellar hole develops within the thickened area with a small retinal detachment. Vertical enlargement of the outer lamellar hole occurs in Stage 3. Finally, in Stage 4, elevation of the upper edge of the external retinal layer occurs accompanied by an increase in the height of the retinal detachment and resolution of schisis. Similar progression has been suggested by Fang et al.[62] Macular buckling with or without vitrectomy has been recommended by few authors for MTM.[69]
Retinal detachment from a paravascular microhole associated with posterior major vessels can also develop in highly myopic eyes. The vitreoretinal adhesion is quite strong at the paravascular region and vitreous traction at this site leads to formation of retinal cysts and breaks.[70]
Myopic Macular Hole
Macular hole formation tends to occur at a younger age as compared to idiopathic age-related macular holes. The degree of myopia and axial length has been shown to have an inverse correlation to the age of onset of the macular hole.[71] Macular hole formation in myopic eyes may be related to the early onset of vitreous degeneration with development of tangential traction at the level of the premacular cortex.[71] Macular holes may also form as an end stage of MTM. These eyes may further progress to a retinal detachment which may be limited to the posterior pole or could even be a total rhegmatogenous retinal detachment. Additional factors which lead to a retinal detachment in myopic eyes with a macular hole include the presence of a posterior staphyloma causing centrifugal traction, chorioretinal atrophy leading to a weak adhesion between the neurosensory retina and RPE, anteroposterior tractional forces leading to lifting of the hole edge, and inelasticity of stretched retinal vessels.[72]
Dome-shaped Macula
Convex elevation of macula within the concavity of a posterior staphyloma has been described as a dome-shaped macula on OCT and ultrasonography in pathologically myopic eyes. Probable pathological causes include tangential vitreomacular traction, localized choroidal or scleral thickening, hypotony, and retinal resistance to scleral deformation.[73] It may be the cause of unexplained visual loss in such eyes.
Prevention of Myopia Progression
Under-correction increases myopia progression and optimal correction is necessary.[74] Progressive or bifocal lenses may slow the progression of myopia by limiting ocular accommodation.[75] Increased time spent outdoors are also a protective factor. The best results so far have been observed with atropine eye drops with a dose-effect relationship.[76,77] Scleral reinforcement surgeries have also been described as a measure to slow down progression but are usually reserved for severe progressive myopia.[78] Yet, till date, we do not have any definitive measure to retard the progression of PM.
Conclusion
High and PM can affect the macula in various ways and can lead to a dramatic fall in VA. Ophthalmologists need to know these varied macular changes associated with HM. OCT and now OCTA aid in an early diagnosis and are important imaging tools required for a serial follow-up of such eyes. An educated patient who can recognize the symptoms of macular disease early and a vigilant ophthalmologist would help in reducing ocular morbidity due to the macular complications of HM.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Morgan IG, Ohno-Matsui K, Saw SM. Myopia. Lancet. 2012;379:1739–48. doi: 10.1016/S0140-6736(12)60272-4. [DOI] [PubMed] [Google Scholar]
- 2.Miller DG, Singerman LJ. Natural history of choroidal neovascularization in high myopia. Curr Opin Ophthalmol. 2001;12:222–4. doi: 10.1097/00055735-200106000-00014. [DOI] [PubMed] [Google Scholar]
- 3.Hayashi K, Ohno-Matsui K, Shimada N, Moriyama M, Kojima A, Hayashi W, et al. Long-term pattern of progression of myopic maculopathy: A natural history study. Ophthalmology. 2010;117:1595–611. 1611.e1–4. doi: 10.1016/j.ophtha.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 4.Curtin BJ, Karlin DB. Axial length measurements and fundus changes of the myopic eye. Am J Ophthalmol. 1971;71(1 Pt 1):42–53. doi: 10.1016/0002-9394(71)91092-0. [DOI] [PubMed] [Google Scholar]
- 5.Wong TY, Ferreira A, Hughes R, Carter G, Mitchell P. Epidemiology and disease burden of pathologic myopia and myopic choroidal neovascularization: An evidence-based systematic review. Am J Ophthalmol. 2014;157:9–25.e12. doi: 10.1016/j.ajo.2013.08.010. [DOI] [PubMed] [Google Scholar]
- 6.Grossniklaus HE, Green WR. Pathologic findings in pathologic myopia. Retina. 1992;12:127–33. doi: 10.1097/00006982-199212020-00009. [DOI] [PubMed] [Google Scholar]
- 7.Ohno-Matsui K, Kawasaki R, Jonas JB, Cheung CM, Saw SM, Verhoeven VJ, et al. International photographic classification and grading system for myopic maculopathy. Am J Ophthalmol. 2015;159:877–83.e7. doi: 10.1016/j.ajo.2015.01.022. [DOI] [PubMed] [Google Scholar]
- 8.Saw SM. How blinding is pathological myopia? Br J Ophthalmol. 2006;90:525–6. doi: 10.1136/bjo.2005.087999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Liu HH, Xu L, Wang YX, Wang S, You QS, Jonas JB. Prevalence and progression of myopic retinopathy in Chinese adults: The Beijing eye study. Ophthalmology. 2010;117:1763–8. doi: 10.1016/j.ophtha.2010.01.020. [DOI] [PubMed] [Google Scholar]
- 10.Vongphanit J, Mitchell P, Wang JJ. Prevalence and progression of myopic retinopathy in an older population. Ophthalmology. 2002;109:704–11. doi: 10.1016/s0161-6420(01)01024-7. [DOI] [PubMed] [Google Scholar]
- 11.Asakuma T, Yasuda M, Ninomiya T, Noda Y, Arakawa S, Hashimoto S, et al. Prevalence and risk factors for myopic retinopathy in a Japanese population: The Hisayama Study. Ophthalmology. 2012;119:1760–5. doi: 10.1016/j.ophtha.2012.02.034. [DOI] [PubMed] [Google Scholar]
- 12.Buch H, Vinding T, La Cour M, Appleyard M, Jensen GB, Nielsen NV. Prevalence and causes of visual impairment and blindness among 9980 Scandinavian adults: The Copenhagen City Eye Study. Ophthalmology. 2004;111:53–61. doi: 10.1016/j.ophtha.2003.05.010. [DOI] [PubMed] [Google Scholar]
- 13.Xu L, Wang Y, Li Y, Wang Y, Cui T, Li J, et al. Causes of blindness and visual impairment in urban and rural areas in Beijing: The Beijing Eye Study. Ophthalmology. 2006;113:1134.e1–11. doi: 10.1016/j.ophtha.2006.01.035. [DOI] [PubMed] [Google Scholar]
- 14.Cotter SA, Varma R, Ying-Lai M, Azen SP, Klein R. Los Angeles Latino Eye Study Group. Causes of low vision and blindness in adult Latinos: The Los Angeles Latino Eye Study. Ophthalmology. 2006;113:1574–82. doi: 10.1016/j.ophtha.2006.05.002. [DOI] [PubMed] [Google Scholar]
- 15.Saxena R, Vashist P, Tandon R, Pandey RM, Bhardawaj A, Menon V, et al. Prevalence of myopia and its risk factors in urban school children in Delhi: The North India Myopia Study (NIM study) PLoS One. 2015;10:e0117349. doi: 10.1371/journal.pone.0117349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jacobsen N, Jensen H, Goldschmidt E. Does the level of physical activity in university students influence development and progression of myopia? A 2-year prospective cohort study. Invest Ophthalmol Vis Sci. 2008;49:1322–7. doi: 10.1167/iovs.07-1144. [DOI] [PubMed] [Google Scholar]
- 17.Lyhne N, Sjølie AK, Kyvik KO, Green A. The importance of genes and environment for ocular refraction and its determiners: A population based study among 20-45 year old twins. Br J Ophthalmol. 2001;85:1470–6. doi: 10.1136/bjo.85.12.1470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Paluru P, Ronan SM, Heon E, Devoto M, Wildenberg SC, Scavello G, et al. New locus for autosomal dominant high myopia maps to the long arm of chromosome 17. Invest Ophthalmol Vis Sci. 2003;44:1830–6. doi: 10.1167/iovs.02-0697. [DOI] [PubMed] [Google Scholar]
- 19.Young TL, Ronan SM, Alvear AB, Wildenberg SC, Oetting WS, Atwood LD, et al. A second locus for familial high myopia maps to chromosome 12q. Am J Hum Genet. 1998;63:1419–24. doi: 10.1086/302111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ibay G, Doan B, Reider L, Dana D, Schlifka M, Hu H, et al. Candidate high myopia loci on chromosomes 18p and 12q do not play a major role in susceptibility to common myopia. BMC Med Genet. 2004;5:20. doi: 10.1186/1471-2350-5-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mutti DO, Semina E, Marazita M, Cooper M, Murray JC, Zadnik K. Genetic loci for pathological myopia are not associated with juvenile myopia. Am J Med Genet. 2002;112:355–60. doi: 10.1002/ajmg.10683. [DOI] [PubMed] [Google Scholar]
- 22.Ashton GC. Segregation analysis of ocular refraction and myopia. Hum Hered. 1985;35:232–9. doi: 10.1159/000153551. [DOI] [PubMed] [Google Scholar]
- 23.Yap M, Wu M, Liu ZM, Lee FL, Wang SH. Role of heredity in the genesis of myopia. Ophthalmic Physiol Opt. 1993;13:316–9. doi: 10.1111/j.1475-1313.1993.tb00479.x. [DOI] [PubMed] [Google Scholar]
- 24.Dirani M, Chamberlain M, Shekar SN, Islam AF, Garoufalis P, Chen CY, et al. Heritability of refractive error and ocular biometrics: The Genes in Myopia (GEM) twin study. Invest Ophthalmol Vis Sci. 2006;47:4756–61. doi: 10.1167/iovs.06-0270. [DOI] [PubMed] [Google Scholar]
- 25.Jones LA, Sinnott LT, Mutti DO, Mitchell GL, Moeschberger ML, Zadnik K. Parental history of myopia, sports and outdoor activities, and future myopia. Invest Ophthalmol Vis Sci. 2007;48:3524–32. doi: 10.1167/iovs.06-1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sherwin JC, Reacher MH, Keogh RH, Khawaja AP, Mackey DA, Foster PJ. The association between time spent outdoors and myopia in children and adolescents: A systematic review and meta-analysis. Ophthalmology. 2012;119:2141–51. doi: 10.1016/j.ophtha.2012.04.020. [DOI] [PubMed] [Google Scholar]
- 27.Rose KA, Morgan IG, Ip J, Kifley A, Huynh S, Smith W, et al. Outdoor activity reduces the prevalence of myopia in children. Ophthalmology. 2008;115:1279–85. doi: 10.1016/j.ophtha.2007.12.019. [DOI] [PubMed] [Google Scholar]
- 28.Silva R. Myopic maculopathy: A review. Ophthalmologica. 2012;228:197–213. doi: 10.1159/000339893. [DOI] [PubMed] [Google Scholar]
- 29.Curtin BJ. The Myopias: Basic Science and Clinical Management. Philadelphia: Harper and Row; 1985. [Google Scholar]
- 30.Siegwart JT, Jr, Norton TT. Perspective: How might emmetropization and genetic factors produce myopia in normal eyes? Optom Vis Sci. 2011;88:E365–72. doi: 10.1097/OPX.0b013e31820b053d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hornbeak DM, Young TL. Myopia genetics: A review of current research and emerging trends. Curr Opin Ophthalmol. 2009;20:356–62. doi: 10.1097/ICU.0b013e32832f8040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wallman J, Turkel J, Trachtman J. Extreme myopia produced by modest change in early visual experience. Science. 1978;201:1249–51. doi: 10.1126/science.694514. [DOI] [PubMed] [Google Scholar]
- 33.Wallman J, Wildsoet C, Xu A, Gottlieb MD, Nickla DL, Marran L, et al. Moving the retina: Choroidal modulation of refractive state. Vision Res. 1995;35:37–50. doi: 10.1016/0042-6989(94)e0049-q. [DOI] [PubMed] [Google Scholar]
- 34.Wallman J, Winawer J. Homeostasis of eye growth and the question of myopia. Neuron. 2004;43:447–68. doi: 10.1016/j.neuron.2004.08.008. [DOI] [PubMed] [Google Scholar]
- 35.Wakabayashi T, Ikuno Y. Choroidal filling delay in choroidal neovascularisation due to pathological myopia. Br J Ophthalmol. 2010;94:611–5. doi: 10.1136/bjo.2009.163535. [DOI] [PubMed] [Google Scholar]
- 36.Choi SS, Garner LF, Enoch JM. The relationship between the Stiles-Crawford effect of the first kind (SCE-I) and myopia. Ophthalmic Physiol Opt. 2003;23:465–72. doi: 10.1046/j.1475-1313.2003.00142.x. [DOI] [PubMed] [Google Scholar]
- 37.Kobayashi K, Ohno-Matsui K, Kojima A, Shimada N, Yasuzumi K, Yoshida T, et al. Fundus characteristics of high myopia in children. Jpn J Ophthalmol. 2005;49:306–11. doi: 10.1007/s10384-004-0204-6. [DOI] [PubMed] [Google Scholar]
- 38.Spaide RF. Staphyloma. In: Spaide RF, Ohno-Matsui K, Yanuuuzzi LA, editors. Pathologic Myopia. New York: Springer; 2013. [Google Scholar]
- 39.Curtin BJ. The posterior staphyloma of pathologic myopia. Trans Am Ophthalmol Soc. 1977;75:67–86. [PMC free article] [PubMed] [Google Scholar]
- 40.Hsiang HW, Ohno-Matsui K, Shimada N, Hayashi K, Moriyama M, Yoshida T, et al. Clinical characteristics of posterior staphyloma in eyes with pathologic myopia. Am J Ophthalmol. 2008;146:102–10. doi: 10.1016/j.ajo.2008.03.010. [DOI] [PubMed] [Google Scholar]
- 41.Steidl SM, Pruett RC. Macular complications associated with posterior staphyloma. Am J Ophthalmol. 1997;123:181–7. doi: 10.1016/s0002-9394(14)71034-7. [DOI] [PubMed] [Google Scholar]
- 42.Moriyama M, Ohno-Matsui K, Futagami S, Yoshida T, Hayashi K, Shimada N, et al. Morphology and long-term changes of choroidal vascular structure in highly myopic eyes with and without posterior staphyloma. Ophthalmology. 2007;114:1755–62. doi: 10.1016/j.ophtha.2006.11.034. [DOI] [PubMed] [Google Scholar]
- 43.Ohno-Matsui K. Proposed classification of posterior staphylomas based on analyses of eye shape by three-dimensional magnetic resonance imaging and wide-field fundus imaging. Ophthalmology. 2014;121:1798–809. doi: 10.1016/j.ophtha.2014.03.035. [DOI] [PubMed] [Google Scholar]
- 44.Ohno-Matsui K, Yoshida T, Futagami S, Yasuzumi K, Shimada N, Kojima A, et al. Patchy atrophy and lacquer cracks predispose to the development of choroidal neovascularisation in pathological myopia. Br J Ophthalmol. 2003;87:570–3. doi: 10.1136/bjo.87.5.570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fujiwara T, Imamura Y, Margolis R, Slakter JS, Spaide RF. Enhanced depth imaging optical coherence tomography of the choroid in highly myopic eyes. Am J Ophthalmol. 2009;148:445–50. doi: 10.1016/j.ajo.2009.04.029. [DOI] [PubMed] [Google Scholar]
- 46.Ikuno Y, Sayanagi K, Soga K, Sawa M, Gomi F, Tsujikawa M, et al. Lacquer crack formation and choroidal neovascularization in pathologic myopia. Retina. 2008;28:1124–31. doi: 10.1097/IAE.0b013e318174417a. [DOI] [PubMed] [Google Scholar]
- 47.Ohno-Matsui K, Morishima N, Ito M, Tokoro T. Indocyanine green angiographic findings of lacquer cracks in pathologic myopia. Jpn J Ophthalmol. 1998;42:293–9. doi: 10.1016/s0021-5155(98)00008-2. [DOI] [PubMed] [Google Scholar]
- 48.Ohno-Matsui K, Ito M, Tokoro T. Subretinal bleeding without choroidal neovascularization in pathologic myopia. A sign of new lacquer crack formation. Retina. 1996;16:196–202. doi: 10.1097/00006982-199616030-00003. [DOI] [PubMed] [Google Scholar]
- 49.Levy JH, Pollock HM, Curtin BJ. The Fuchs' spot: An ophthalmoscopic and fluorescein angiographic study. Ann Ophthalmol. 1977;9:1433–43. [PubMed] [Google Scholar]
- 50.Leveziel N, Yu Y, Reynolds R, Tai A, Meng W, Caillaux V, et al. Genetic factors for choroidal neovascularization associated with high myopia. Invest Ophthalmol Vis Sci. 2012;53:5004–9. doi: 10.1167/iovs.12-9538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ikuno Y, Jo Y, Hamasaki T, Tano Y. Ocular risk factors for choroidal neovascularization in pathologic myopia. Invest Ophthalmol Vis Sci. 2010;51:3721–5. doi: 10.1167/iovs.09-3493. [DOI] [PubMed] [Google Scholar]
- 52.Neelam K, Cheung CM, Ohno-Matsui K, Lai TY, Wong TY. Choroidal neovascularization in pathological myopia. Prog Retin Eye Res. 2012;31:495–525. doi: 10.1016/j.preteyeres.2012.04.001. [DOI] [PubMed] [Google Scholar]
- 53.Chan WM, Ohji M, Lai TY, Liu DT, Tano Y, Lam DS. Choroidal neovascularisation in pathological myopia: An update in management. Br J Ophthalmol. 2005;89:1522–8. doi: 10.1136/bjo.2005.074716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Miyata M, Ooto S, Hata M, Yamashiro K, Tamura H, Akagi-Kurashige Y, et al. Detection of myopic choroidal neovascularization using optical coherence tomography angiography. Am J Ophthalmol. 2016;165:108–14. doi: 10.1016/j.ajo.2016.03.009. [DOI] [PubMed] [Google Scholar]
- 55.Yoshida T, Ohno-Matsui K, Yasuzumi K, Kojima A, Shimada N, Futagami S, et al. Myopic choroidal neovascularization: A 10-year follow-up. Ophthalmology. 2003;110:1297–305. doi: 10.1016/S0161-6420(03)00461-5. [DOI] [PubMed] [Google Scholar]
- 56.Wolf S, Balciuniene VJ, Laganovska G, Menchini U, Ohno-Matsui K, Sharma T, et al. RADIANCE: A randomized controlled study of ranibizumab in patients with choroidal neovascularization secondary to pathologic myopia. Ophthalmology. 2014;121:682–92.e2. doi: 10.1016/j.ophtha.2013.10.023. [DOI] [PubMed] [Google Scholar]
- 57.Gohil R, Sivaprasad S, Han LT, Mathew R, Kiousis G, Yang Y. Myopic foveoschisis: A clinical review. Eye (Lond) 2015;29:593–601. doi: 10.1038/eye.2014.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Panozzo G, Mercanti A. Optical coherence tomography findings in myopic traction maculopathy. Arch Ophthalmol. 2004;122:1455–60. doi: 10.1001/archopht.122.10.1455. [DOI] [PubMed] [Google Scholar]
- 59.Baba T, Ohno-Matsui K, Futagami S, Yoshida T, Yasuzumi K, Kojima A, et al. Prevalence and characteristics of foveal retinal detachment without macular hole in high myopia. Am J Ophthalmol. 2003;135:338–42. doi: 10.1016/s0002-9394(02)01937-2. [DOI] [PubMed] [Google Scholar]
- 60.Sayanagi K, Morimoto Y, Ikuno Y, Tano Y. Spectral-domain optical coherence tomographic findings in myopic foveoschisis. Retina. 2010;30:623–8. doi: 10.1097/iae.0b013e3181ca4e7c. [DOI] [PubMed] [Google Scholar]
- 61.Wu PC, Chen YJ, Chen YH, Chen CH, Shin SJ, Tsai CL, et al. Factors associated with foveoschisis and foveal detachment without macular hole in high myopia. Eye (Lond) 2009;23:356–61. doi: 10.1038/sj.eye.6703038. [DOI] [PubMed] [Google Scholar]
- 62.Fang X, Weng Y, Xu S, Chen Z, Liu J, Chen B, et al. Optical coherence tomographic characteristics and surgical outcome of eyes with myopic foveoschisis. Eye (Lond) 2009;23:1336–42. doi: 10.1038/eye.2008.291. [DOI] [PubMed] [Google Scholar]
- 63.Gaucher D, Haouchine B, Tadayoni R, Massin P, Erginay A, Benhamou N, et al. Long-term follow-up of high myopic foveoschisis: Natural course and surgical outcome. Am J Ophthalmol. 2007;143:455–62. doi: 10.1016/j.ajo.2006.10.053. [DOI] [PubMed] [Google Scholar]
- 64.Benhamou N, Massin P, Haouchine B, Erginay A, Gaudric A. Macular retinoschisis in highly myopic eyes. Am J Ophthalmol. 2002;133:794–800. doi: 10.1016/s0002-9394(02)01394-6. [DOI] [PubMed] [Google Scholar]
- 65.Sayanagi K, Ikuno Y, Tano Y. Tractional internal limiting membrane detachment in highly myopic eyes. Am J Ophthalmol. 2006;142:850–2. doi: 10.1016/j.ajo.2006.05.031. [DOI] [PubMed] [Google Scholar]
- 66.Ikuno Y, Gomi F, Tano Y. Potent retinal arteriolar traction as a possible cause of myopic foveoschisis. Am J Ophthalmol. 2005;139:462–7. doi: 10.1016/j.ajo.2004.09.078. [DOI] [PubMed] [Google Scholar]
- 67.Sayanagi K, Ikuno Y, Gomi F, Tano Y. Retinal vascular microfolds in highly myopic eyes. Am J Ophthalmol. 2005;139:658–63. doi: 10.1016/j.ajo.2004.11.025. [DOI] [PubMed] [Google Scholar]
- 68.Shimada N, Ohno-Matsui K, Yoshida T, Sugamoto Y, Tokoro T, Mochizuki M. Progression from macular retinoschisis to retinal detachment in highly myopic eyes is associated with outer lamellar hole formation. Br J Ophthalmol. 2008;92:762–4. doi: 10.1136/bjo.2007.131359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Mateo C, Gómez-Resa MV, Burés-Jelstrup A, Alkabes M. Surgical outcomes of macular buckling techniques for macular retinoschisis in highly myopic eyes. Saudi J Ophthalmol. 2013;27:235–9. doi: 10.1016/j.sjopt.2013.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Spencer LM, Foos RY. Paravascular vitreoretinal attachments. Role in retinal tears. Arch Ophthalmol. 1970;84:557–64. doi: 10.1001/archopht.1970.00990040559001. [DOI] [PubMed] [Google Scholar]
- 71.Kobayashi H, Kobayashi K, Okinami S. Macular hole and myopic refraction. Br J Ophthalmol. 2002;86:1269–73. doi: 10.1136/bjo.86.11.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Morita H, Ideta H, Ito K, Yonemoto J, Sasaki K, Tanaka S. Causative factors of retinal detachment in macular holes. Retina. 1991;11:281–4. doi: 10.1097/00006982-199111030-00002. [DOI] [PubMed] [Google Scholar]
- 73.Ellabban AA, Tsujikawa A, Muraoka Y, Yamashiro K, Oishi A, Ooto S, et al. Dome-shaped macular configuration: Longitudinal changes in the sclera and choroid by swept-source optical coherence tomography over two years. Am J Ophthalmol. 2014;158:1062–70. doi: 10.1016/j.ajo.2014.08.006. [DOI] [PubMed] [Google Scholar]
- 74.Chung K, Mohidin N, O'Leary DJ. Undercorrection of myopia enhances rather than inhibits myopia progression. Vision Res. 2002;42:2555–9. doi: 10.1016/s0042-6989(02)00258-4. [DOI] [PubMed] [Google Scholar]
- 75.Cheng D, Woo GC, Drobe B, Schmid KL. Effect of bifocal and prismatic bifocal spectacles on myopia progression in children: Three-year results of a randomized clinical trial. JAMA Ophthalmol. 2014;132:258–64. doi: 10.1001/jamaophthalmol.2013.7623. [DOI] [PubMed] [Google Scholar]
- 76.Chia A, Chua WH, Cheung YB, Wong WL, Lingham A, Fong A, et al. Atropine for the treatment of childhood myopia: Safety and efficacy of 0.5%, 0.1%, and 0.01% doses (atropine for the treatment of myopia 2) Ophthalmology. 2012;119:347–54. doi: 10.1016/j.ophtha.2011.07.031. [DOI] [PubMed] [Google Scholar]
- 77.Li SM, Wu SS, Kang MT, Liu Y, Jia SM, Li SY, et al. Atropine slows myopia progression more in Asian than white children by meta-analysis. Optom Vis Sci. 2014;91:342–50. doi: 10.1097/OPX.0000000000000178. [DOI] [PubMed] [Google Scholar]
- 78.Avetisov ES, Tarutta EP, Iomdina EN, Vinetskaya MI, Andreyeva LD. Nonsurgical and surgical methods of sclera reinforcement in progressive myopia. Acta Ophthalmol Scand. 1997;75:618–23. doi: 10.1111/j.1600-0420.1997.tb00617.x. [DOI] [PubMed] [Google Scholar]


