Abstract
Background and objectives
Relatively smaller kidney donor to recipient size is proposed to result in higher graft loss due to nephron underdosing and hyperfiltration injury, but the potentially additive effect of sex and weight mismatch has not been explored in detail. The purpose of this study was to determine if concurrent donor and recipient absolute weight and sex mismatch was associated with graft loss in a cohort of deceased donor kidney transplant recipients.
Design, setting, participants, & measurements
The association of kidney donor and recipient absolute weight and sex difference with death-censored graft loss was explored using a cohort of United States deceased donor recipients between 2000 and 2014 through the Scientific Registry of Transplants Recipients. Donor-recipient sex pairings (male donor–male recipient; female donor–female recipient; male donor–female recipient; female donor–male recipient) were further stratified by donor and recipient absolute weight difference (>30 or 10–30 kg [donor<recipient; donor>recipient] or <10 kg [donor=recipient]) resulting in 20 weight and sex pairings. Time to death-censored graft loss for each pairing was evaluated using multivariable Cox proportional hazards models adjusting for donor, immunologic, surgical, and recipient predictors of graft loss compared with the reference group of male donor–male recipients with no weight mismatch (<10 kg difference).
Results
Of 115,124 kidney transplant recipients, 21,261 developed death-censored graft failure (median graft survival time was 3.8 years; quartile 1 to 3, 0.0 to 14.8 years). After multivariable adjustment, the highest relative hazards for graft failure were observed for female recipients of male donor kidneys and male recipients of female donor kidneys in situations where the recipient was >30 kg larger than donor (hazard ratio, 1.50; 95% confidence interval, 1.32 to 1.70; hazard ratio, 1.35; 95% confidence interval, 1.25 to 1.45, respectively).
Conclusions
A concurrent mismatch in donor-recipient weight (donor<recipient) and donor-recipient sex is associated with a higher risk of death-censored graft loss in kidney transplantation.
Keywords: kidney transplantation, graft failure, size mismatch, sex mismatch, female, graft survival, kidney, male, nephrons, proportional hazards models, registries, sex characteristics, tissue donors, transplant recipients, United States
Introduction
A number of donor and recipient factors have been identified that increase the risk of both early and late adverse outcomes after kidney transplantation. Most literature has emphasized the importance of immunologic mismatches (1,2), however, earlier studies have also demonstrated that nonimmunologic factors may influence graft and patient outcome (2–4). Hyperfiltration due to reduced renal mass or nephron underdosing resulting from kidney size mismatch between donor and recipient has been proposed as a potential nonimmunologic contributor to chronic renal graft nephropathy (2). Studies have demonstrated that a smaller donor size relative to recipient is associated with a higher risk of graft loss (5–7). Contrarily, reduced hyperfiltration injury and thereby reduced immune-mediated rejection and graft loss has been demonstrated in recipients of kidneys from relatively larger donors (8,9). A number of earlier studies have explored the association between donor/recipient size and the effect on outcomes using donor kidney weight (6), as well as body mass index (8) or body surface area (BSA) (7) as a surrogate for nephron mass. Most studies suggest that increased donor mass/size relative to recipient is protective against chronic allograft nephropathy. Interestingly, very few studies have investigated the outcomes associated with donor-recipient weight mismatching as determined by body mass in isolation.
Although findings are contradictory (10), sex mismatch has also been associated with worse graft outcomes after kidney transplant. Female kidneys are generally smaller with fewer total nephrons (11,12) and as a result, male recipients of female kidneys appear to be at highest risk of graft loss in unadjusted analyses; this finding is presumed to be due to size mismatch and nephron underdosing (11,13). In addition, female recipients of male kidneys also appear to have reduced graft survival compared with sex-matched donors and recipients, albeit to a lesser extent than in male recipients of female kidneys (14). This observation is proposed to be immunologic in nature. Mismatch between H-Y minor histocompatibility antigens between sexes (derived from the Y chromosome in male donors) is suspected to result in increased sensitization and subsequent graft rejection and loss (13,14). The potentially additive effect of size mismatch (with nephron underdosing in donor<recipient [D<R]) and sex mismatch (increased H-Y immunogenicity in male donor and female recipient) has not previously been explored.
Therefore, the purpose of this study was to determine the effect of absolute weight mismatch and sex mismatch on kidney transplant graft survival after transplantation. We hypothesized that the combination of a decreased donor-to-recipient absolute weight and sex mismatch would be associated with a higher risk of graft failure.
Materials and Methods
Design
We conducted a cohort study of all patients receiving a solitary deceased kidney transplant in the United States, identified using the Scientific Registry of Transplant Recipients (SRTR), between January 1, 2000 and December 31, 2013. We excluded living donors, patients <18 years of age, those receiving multiple organs, en bloc or sequential transplants, and patients without a documented donor or recipient weight. Lastly, donors and recipients with body weights <30 kg were excluded, as these were assumed to be miscoded values.
Exposure
The primary exposure was combined donor-recipient weight and sex mismatch. Sex pairing between donor and recipient was categorized as female donor to male recipient (FDMR), female donor to female recipient (FDFR), male donor to female recipient (MDFR), and male donor to male recipient (MDMR). Absolute weight difference was categorized as >30 kg, 10–30 kg (D<R; donor>recipient [D>R]), or <10 kg (donor=recipient [D=R]). Absolute weight difference cut-points were chosen to distribute the cohort into relatively even categories. Each donor-recipient sex pairing was subcategorized by absolute weight difference resulting in 20 possible weight and sex pairings.
Outcome
The outcome of interest was death-censored graft failure. Graft failure was defined as need for chronic dialysis or repeat pre-emptive transplantation. Censoring occurred at losses to follow-up and at the date of last follow-up.
Data Collection
In addition to the primary exposure, known literature predictors of graft loss including donor and recipient age, donor and recipient height, race, donor cause of death, cold ischemia time, recipient ESRD cause, dialysis vintage, categories of human leukocyte antigen mismatch (0–6), categories of panel reactive antibody (0–<20%, 20%–80%, and >80%) (15,16), and medical comorbidities, were also collected from the SRTR. Missing data were addressed with multiple imputation using chained equations (17).
Analysis
Descriptive statistics were used to report baseline characteristics for all patients enrolled in the study. Means and SDs, and medians and interquartile ranges, were used for continuous normal and continuous non-normally distributed variables. Baseline donor and recipient characteristics were reported for all patients in each of the five weight categories: 10–30 kg (D<R), >30 kg (D<R), 10–30 kg (D>R), >30 kg (D>R), and the reference range of <10 kg absolute difference (D=R). The proportion of patients in each sex match/mismatch category (stratified by weight mismatch) was also reported.
The association between donor-recipient sex and weight mismatch and graft failure was analyzed using a multivariable Cox proportional hazards model adjusting for known predictors of graft failure highlighted above. The reference group was chosen as MDMR with <10 kg absolute weight difference. Relative hazards and 95% confidence intervals (95% CIs) were graphically displayed for each donor-recipient sex/weight pairing compared with the reference group. Proportionality was assessed using visual examination of −ln (−ln[survival]) plots.
In secondary analyses, the unadjusted and adjusted association between donor-recipient weight differences and graft loss and donor-recipient sex pairing and graft loss was analyzed using multivariable Cox proportional hazards models.
In a sensitivity analysis, we re-examined the primary exposure stratifying sex pairing instead by differences in BSA, estimated using the Mosteller formula: BSA = square root (height [centimeters] × weight [kilograms]/3600) (18). Donor/recipient absolute BSA differences were categorized as >0.03 m2 (D<R), 0.01–0.03 m2 (D<R), <0.01 m2 (D=R), 0.01–0.03 m2 (D>R), and >0.03 m2 (D>R). Cut-points for absolute BSA differences were also chosen to distribute the cohort into evenly sized categories.
All statistical analyses were performed using Stata version 13.1 (Stata Corp., College Station, TX). Graphics were created using R. For statistical comparisons, a P<0.05 was deemed the threshold for statistical significance. Institutional ethics approval to conduct this study was given through the Nova Scotia Health Authority research ethics board.
Results
Baseline Characteristics
After exclusions, our final study cohort consisted of 115,124 individuals, with 367 donor/recipient pairs dropped for missing recipient weight and 77 donor/recipient pairs excluded for weights <30 kg. No donors were missing weight measurements. Baseline characteristics are noted in Table 1. Median donor weight was 79.4 kg (quartile 1 [Q1] to quartile 3 [Q3], 68.0–92.7 kg), and median recipient weight was 79.8 kg (Q1 to Q3, 67.6–93.3 kg). The absolute weight difference between donors and recipients was normally distributed. Mean and median absolute weight difference was 0.7±27.1 kg, and 0.0 kg (Q1 to Q3, −17.1–17.6 kg), respectively. Of the donors, 59.4% were men, whereas 61.6% of recipients were men.
Table 1.
Recipient, donor, and kidney transplant characteristics
| Characteristics, N (%) (unless otherwise specified) | Categories | ||||
|---|---|---|---|---|---|
| >30 kg (D>R) | 10–30 kg (D>R) | <10 kg (D=R) | 10–30 kg (D<R) | >30 kg (D<R) | |
| n=15,002 (13.0) | n=24,903 (21.6) | n=35,495 (30.8) | n=26,071 (22.6) | n=13,653 (11.9) | |
| Donor factors | |||||
| Donor type | |||||
| SCD | 10,363 (69.1) | 17,727 (71.2) | 25,256 (71.2) | 18,659 (71.6) | 10,110 (74.0) |
| ECD | 2831 (18.9) | 4674 (18.8) | 6738 (19.0) | 4823 (18.5) | 2060 (15.1) |
| DCD | 1808 (12.0) | 2502 (10.0) | 3501 (9.9) | 2589 (9.9) | 1483 (10.9) |
| Mean age ±SD, yr | 42.8±12.8 | 42.2±13.7 | 41.9±14.2 | 41.7±14.4 | 40.6±14.2 |
| Sex (M) | 10,592 (70.6) | 16,811 (67.5) | 21,374 (60.2) | 13,466 (51.6) | 6153 (45.1) |
| Mean height ±SD, m | 1.77±0.1 | 1.74±0.1 | 1.72±0.1 | 1.70±0.1 | 1.68±0.1 |
| Median donor weight (Q1–Q3), kg | 108.9 (97.5–122.5) | 88.2 (79.4–99.0) | 77.2 (69.0–87.0) | 70.0 (62.0–79.4) | 64.7 (56.7–73.0) |
| Diabetes | 1905 (12.8) | 1949 (7.9) | 2177 (6.2) | 1294 (5.0) | 560 (4.1) |
| BMI (Q1–Q3), kg/m2 | 34.7 (30.6–39.7) | 28.9 (25.8–32.4) | 25.9 (23.4–29.0) | 24.2 (21.8–26.9) | 22.8 (20.4–25.4) |
| Recipient factors | |||||
| Mean age ±SD, yr | 51.5±14.1 | 52.0±13.5 | 52.9±12.9 | 53.4±12.2 | 52.1±11.6 |
| Male sex | 6811 (45.4) | 12,946 (52.0) | 21,972 (61.9) | 18,327 (70.3) | 10,837 (79.4) |
| Mean height ±SD, m | 1.65±0.1 | 1.67±0.1 | 1.70±0.1 | 1.73±0.1 | 1.78±0.1 |
| Median recipient weight (Q1–Q3), kg | 64.0 (54.9–74.4) | 69.3 (60.6–79.0) | 77.6 (68.9–87.1) | 89.3 (80.7–98.5) | 107.3 (97.9–117.9) |
| Diabetes | 4026 (26.9) | 7293 (29.4) | 11,738 (33.2) | 9981 (38.4) | 5841 (42.8) |
| Previous kidney transplant | 2433 (16.4) | 3735 (15.0) | 4531 (12.8) | 2851 (10.9) | 1281 (8.6) |
| Dialysis vintage >4 yr | 5731 (39.0) | 9369 (38.4) | 12,816 (37.0) | 8973 (35.2) | 4813 (36.0) |
| BMI (Q1–Q3), kg/m2 | 23.4 (20.8–26.6) | 24.7 (22.0–27.9) | 26.7 (24.0–30.0) | 29.8 (26.8–33.0) | 34.2 (31.0–37.4) |
| Surgical and immunologic factors | |||||
| Median cold ischemic time (IQR), h | 18.2 (12.0–23.0) | 17.9 (11.8–23.0) | 17.9 (11.6–23.0) | 17.8 (11.7–23.0) | 17.8 (11.5–22.9) |
| Peak PRA <20 | 9368 (63.6) | 15,939 (65.2) | 23,987 (69.0) | 18,397 (72.1) | 10,060 (75.3) |
| Peak PRA 20–80 | 3039 (20.6) | 4923 (20.2) | 6515 (18.8) | 4482 (17.6) | 2117 (15.9) |
| Peak PRA >80 | 2334 (15.8) | 3569 (14.6) | 4238 (12.2) | 2634 (10.3) | 1182 (8.8) |
| Induction therapy (ATG) | 6752 (45.0) | 10,849 (43.6) | 15,187 (42.8) | 10,945 (42.0) | 6010 (44.0) |
| Donor and recipient factors | |||||
| HLA mismatches | |||||
| 0MM | 1586 (10.6) | 2704 (10.9) | 3728 (10.6) | 2645 (10.2) | 1391 (10.3) |
| 1MM | 347 (2.3) | 651 (2.6) | 849 (2.4) | 667 (2.6) | 319 (2.4) |
| 2MM | 718 (4.8) | 1151 (4.7) | 1733 (4.9) | 1216 (4.7) | 652 (4.8) |
| 3MM | 2017 (13.5) | 3285 (13.3) | 4614 (13.1) | 3519 (13.6) | 1748 (12.9) |
| 4MM | 3726 (24.9) | 6265 (25.3) | 8899 (25.3) | 6341 (24.5) | 3473 (25.6) |
| 5MM | 4379 (29.3) | 7090 (28.7) | 10,222 (29.0) | 7668 (29.6) | 3964 (29.3) |
| 6MM | 2167 (14.5) | 3594 (14.5) | 5187 (14.7) | 3826 (14.8) | 2000 (14.8) |
| Absolute weight difference (Q1–Q3), kg | 41.7 (35.0–52.2) | 18.6 (14.0–23.6) | 0.0 (−5.0–4.9) | −18.6 (−23.7–−14.1) | −40 (−48.7–−34.5) |
| MDMR | 4805 (32.0) | 9057 (36.4) | 13,948 (39.3) | 9880 (37.9) | 5068 (37.1) |
| FDFR | 2404 (16.0) | 4203 (16.9) | 6097 (17.2) | 4158 (16.0) | 1731 (12.7) |
| MDFR | 5787 (38.6) | 7754 (31.1) | 7426 (20.9) | 3586 (13.8) | 1085 (8.0) |
| FDMR | 2006 (13.4) | 3889 (15.6) | 8024 (22.6) | 8447 (32.4) | 5769 (42.2) |
Proportion missing: Recipient diabetes (0.31%); donor diabetes (0.47%); DCD (0.01%); dialysis vintage (2.15%); HLA mismatch (0.68%); peak PRA (1.66%); cold ischemic time (7.77%). D>R, donor greater than recipient; D=R, donor equal to recipient; D<R, donor less than recipient; SCD, standard criteria donor; ECD, expanded criteria donor; DCD, donation by cardiac death; M, male; Q1, quarter 1; Q3, quarter 3; BMI, body mass index; IQR, interquartile range; PRA, panel reactive antibody; ATG, antithymoglobulin; HLA, human leukocyte antigen; MM, mismatch; MDMR, male donor/male recipient; FDFR, female donor/female recipient; MDFR, male donor/female recipient; FDMR, female donor/male recipient.
Primary Analysis
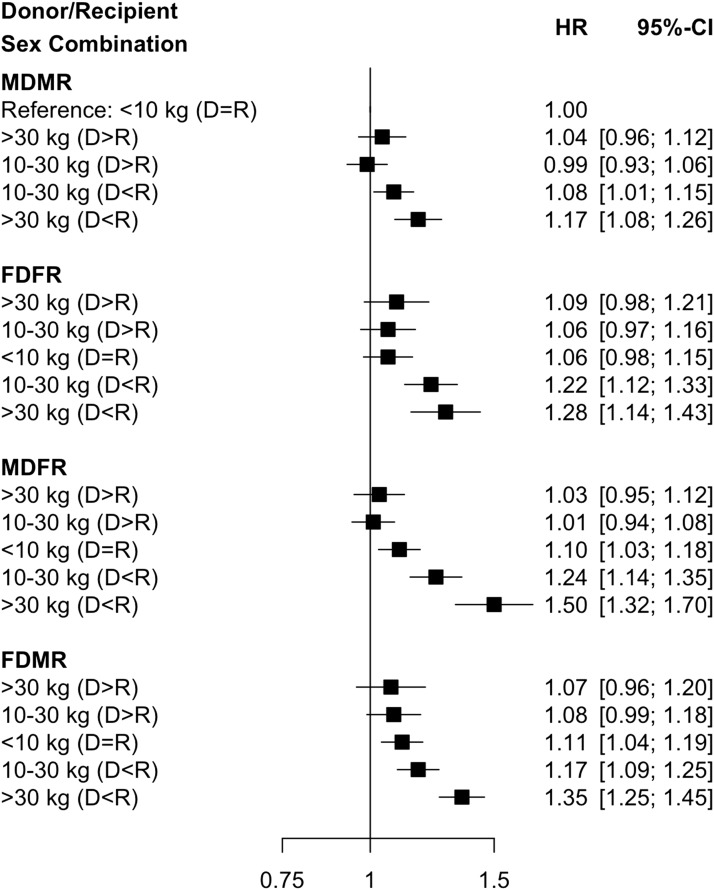
Of the 115,124 individuals included in this analysis, death-censored graft failure occurred in 21,261 patients (18.5% overall). Median follow-up time was 3.8 years (Q1 to Q3, 0.0–14.8 years). The risk of graft failure for each donor-recipient sex pairing was higher when the weight of the recipient was greater than that of the donor. In multivariable analysis, the risk of graft loss was highest among recipients with >30 kg absolute weight difference (D<R) who had concurrent sex mismatch (hazard ratio [HR], 1.50; 95% CI, 1.32 to 1.70, for MDFR; and HR, 1.35; 95% CI, 1.25 to 1.45, for FDMR), relative to MDMR with no weight mismatch. These results are depicted in Figure 1.
Figure 1.
The risk of graft failure is highest for sex mismatched donors and recipients when the recipient weight is greater than the donor. Adjusted relative hazards for graft failure using combination of absolute weight difference (>30 kg [D<R], 10–30 kg [D<R], <10 kg [D=R, reference category], 10–30 kg [D>R], and >30 kg [D>R]) and sex pairing (MDMR, male donor/male recipient; FDFR, female donor/female recipient; MDFR, male donor/female recipient; FDMR, female donor/male recipient). 95% CI, 95% confidence interval; D>R, donor greater than recipient; D=R, donor equal to recipient; D<R, donor less than recipient; HR, hazard ratio.
Secondary Analysis
In a multivariable analysis of donor-recipient weight mismatch, the adjusted relative hazard for a >30 kg absolute weight difference (D<R) relative to no weight mismatch was 1.22 (95% CI, 1.16 to 1.28; Table 2). In unadjusted analysis, recipients of kidneys from female donors were at the highest risk for graft failure (HR, 1.12; 95% CI, 1.08 to 1.16, for FDFR; and HR, 1.22; 95% CI, 1.18 to 1.27, for FDMR). However, after adjusting for donor, surgical, immunologic, and recipient factors (including donor/recipient weight), the adjusted relative hazard was similar for all sex combinations relative to MDMR (HR’s of 1.08, 1.08, and 1.10 for FDFR, MDFR, and FDMR, respectively, Table 2).
Table 2.
Relative hazards for death-censored graft loss using donor/recipient absolute weight difference and donor/recipient sex pairing
| Donor-Recipient Pairing | HR (95% CI) |
|---|---|
| Weight-unadjusted | |
| >30 kg (D>R) | 1.00 (0.96 to 1.05) |
| 10–30 kg (D>R) | 0.97 (0.93 to 1.00) |
| <10 kg (D=R) | Ref |
| 10–30 kg (D<R) | 1.11 (1.07 to 1.15) |
| >30 kg (D<R) | 1.28 (1.22 to 1.34) |
| Weight-adjusteda | |
| >30 kg (D>R) | 1.00 (0.94 to 1.04) |
| 10–30 kg (D>R) | 0.97 (0.93 to 1.01) |
| <10 kg (D=R) | Ref |
| 10–30 kg (D<R) | 1.10 (1.05 to 1.13) |
| >30 kg (D<R) | 1.22 (1.16 to 1.28) |
| Sex-unadjusted | |
| MDMR | Ref |
| FDFR | 1.12 (1.08 to 1.16) |
| MDFR | 1.00 (0.96 to 1.04) |
| FDMR | 1.22 (1.18 to 1.27) |
| Sex-adjustedb | |
| MDMR | Ref |
| FDFR | 1.08 (1.02 to 1.14) |
| MDFR | 1.08 (1.03 to 1.13) |
| FDMR | 1.10 (1.05 to 1.15) |
HR, hazard ratio; 95% CI, 95% confidence interval; D>R, donor greater than recipient; D=R, donor equal to recipient; Ref, reference; D<R, donor less than recipient; MDMR, male donor/male recipient; FDFR, female donor/female recipient; MDFR, male donor/female recipient; FDMR, female donor/male recipient.
Adjusted for: donor factors: type (expanded criteria donor versus standard criteria donor), donation by cardiac death, sex, race, height, diabetes status, hepatitis C; surgical & immunologic factors: number of human leukocyte antigen mismatches, peak panel reactive antibody, induction therapy, cold ischemia time; recipient factors: diabetes, age, cause of ESRD, sex, race, height, previous kidney transplant, dialysis vintage.
Adjusted for: donor factors: type (expanded criteria donor versus standard criteria donor), donation by cardiac death, race, height, weight, diabetes status, hepatitis C; surgical & immunologic factors: number of human leukocyte antigen mismatches, peak panel reactive antibody, induction therapy, cold ischemia time; recipient factors: diabetes, age, cause of ESRD, race, height, weight, previous kidney transplant, dialysis vintage.
Sensitivity Analysis
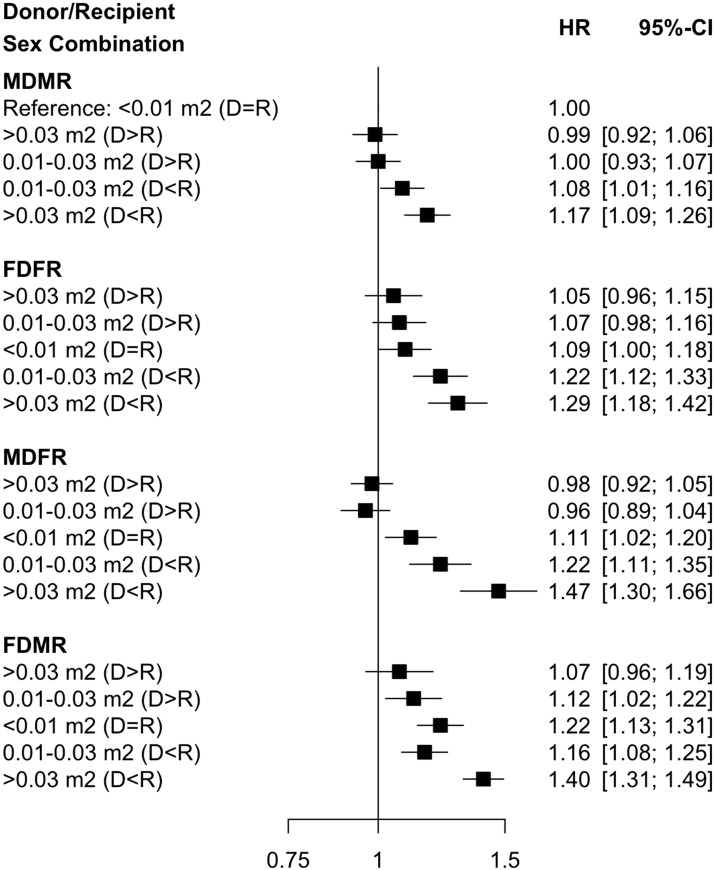
The observed trend when BSA was used instead of absolute weight difference for donor/recipient sex/weight mismatch was similar to that seen for absolute weight difference. Specifically, the highest risk of graft failure was noted among recipients with the most extreme BSA differences (>0.03 m2, D<R) who had concurrent sex mismatch (HR, 1.47; 95% CI, 1.30 to 1.66, for MDFR; and HR, 1.40; 95% CI, 1.31 to 1.49, for FDMR; Figure 2), relative to MDMR with no BSA mismatch (<0.01 m2).
Figure 2.
The risk of graft failure is highest for sex mismatched donors and recipients when the recipient body surface area is greater than the donor. Adjusted relative hazards for graft failure using combination of absolute body surface area difference (>0.03 m2 [D<R], 0.01–0.03 m2 [D<R], <0.01 m2 [D=R, reference category], 0.01–0.03 m2 [D>R], and >0.03 m2 [D>R]) and sex pairing (MDMR, male donor/male recipient; FDFR, female donor/female recipient; MDFR, male donor/female recipient; FDMR, female donor/male recipient). 95% CI, 95% confidence interval; D>R, donor greater than recipient; D=R, donor equal to recipient; D<R, donor less than recipient; HR, hazard ratio.
Discussion
In this study we identified an association between the combined exposure of donor-recipient weight/sex mismatch and kidney graft loss. The highest risk of graft failure was observed in the setting of a smaller donor weight relative to recipient. Specifically, female recipients of male kidneys (MDFR) with concurrent weight mismatch (>30 kg, D<R) were at the highest overall risk for graft loss. In isolation, donor-recipient weight mismatch (D<R) and specific donor/recipient sex pairings (MDFR, FDFR, FDMR) were associated with a higher risk of graft failure. Importantly, the risk of graft loss in isolated donor-recipient weight or sex mismatch was much less pronounced than in the setting of concurrent weight and sex mismatch.
Earlier studies, using different size metrics, have also demonstrated a higher risk of kidney graft failure in the setting of small kidney donors relative to recipients (8,19). It is hypothesized that reduced renal mass in the donor results in relative nephron underdosing with hyperfiltration of the remaining nephrons (2). This precipitates glomerular hypertension and, over time, chronic allograft nephropathy and potentially graft loss (20,21). Conversely, larger donor kidney mass relative to recipient provides an increased number of glomeruli with less metabolic demand on the transplanted kidney and thus less hyperfiltration and subsequent related injury over the long term (8,20). The theory of nephron underdosing may also be partially responsible for the worse graft outcomes observed in donor-recipient sex-mismatched kidney transplantation (12). A number of studies have demonstrated a reduction in graft survival in male recipients of female kidneys (8,12,13). Female kidneys are generally smaller with an average of 12%–17% fewer total nephrons than male kidneys (11,12,22); this finding could conceivably lead to nephron underdosing in an equally weighted female donor and male recipient (11,13).
Reduced nephron mass may explain the higher risk of graft failure we demonstrated for female donors regardless of recipient sex in unadjusted sex analyses. Conversely, MDFR has also been shown to have worse graft outcomes than sex-matched controls, an observation which is proposed to be immunologic in nature (13). Mismatch between H and Y minor histocompatibility antigens (on the Y chromosome in male donors) is suspected to result in increased sensitization, allograft rejection, and subsequent graft failure in this population (11,13,14,23). This may account for the relative increase in graft failure for MDFR after adjusting for factors including donor/recipient weight. The potential synergistic increase in graft loss we demonstrated in the setting of combined weight and sex mismatch (particularly in MDFR) may be due to an added immunologic strain imposed on a reduced number of nephrons with exaggerated compensatory hyperfiltration and subsequent injury (11,13,14,23).
Literature regarding the association between donor and recipient size discrepancy and kidney transplant outcomes has been conflicting (5–9,24,25). It is possible that this may relate to variability in study design (short- versus long-term outcomes) and exposure definitions. Although isolated kidney mass is a validated predictor of nephron number (22), this involves measuring the organ at the time of retrieval in the operating room, which is likely too late to inform recipient selection processes. Additionally, donor-recipient height and BSA comparisons have been studied in this regard previously, however, these measures are also less practical for point-of-care decisions at the time of organ allocation. Interestingly, there are very few studies assessing the association between donor and recipient weight directly, which is a standardized measure and the easiest parameter to determine for rapid bedside decision-making. In addition, early physiology studies have demonstrated that the process of glomerular hyperfiltration is driven by body mass and the degree of expected hyperfiltration is a product of the weight perfused by the kidney, with increased weight resulting in increased metabolic demands on the kidney (2). Although we focused on absolute weight difference, a repeated analysis using combined donor-recipient sex and BSA difference (instead of weight difference) showed similar results with increased graft loss with both sex mismatch (MDFR/FDMR) and maximal BSA difference (D<R; Figure 2).
Our study is the first large-scale analysis to demonstrate that worse kidney transplant outcomes associated with donor and recipient weight mismatch (as determined by absolute differences in body weight [D<R]), and donor and recipient sex mismatch are additive. The hazard ratio for this combined exposure of >30 kg weight mismatch (D<R) and sex mismatch was 1.50 (95% CI, 1.32 to 1.70) in MDFR and 1.35 (95% CI, 1.25 to 1.45) in FDMR. This higher risk is comparable to other immunologic and nonimmunologic risk factors for graft loss which historically influence organ allocation and were found to be associated with a higher risk of graft failure in our cohort, including donor diabetes (HR, 1.55; 95% CI, 1.47 to 1.63), dialysis vintage >4 years (HR, 1.49; 95% CI, 1.41 to 1.59), and expanded versus standard criteria donors (HR, 1.81; 95% CI, 1.75 to 1.88). These results may eventually inform recipient selection processes directly, or be incorporated into decisions around donor and recipient matching. For example, in the case of expanded criteria donors, studies have shown that younger, healthier patients benefit from waiting for a more ideal organ rather than accepting expanded criteria donor organs that are predicted to have a higher risk of graft loss (26,27). Conversely, given the higher risk of death on the transplant weight list, older patients can benefit from accepting the first available kidney match, even if the organ is from an expanded criteria donor (26,27). This may also be the case in the setting of absolute weight mismatch between donor and recipient especially if there is concurrent sex mismatch. Any benefit derived from weight and sex matching would have to offset the potential risk of longer wait times in heavier recipients.
Our study was conducted using the SRTR, which is a large-scale, nationally representative cohort of United States kidney transplant recipients. This database has been used in numerous earlier studies and is felt to be a robust record of national transplant outcomes (28,29). However, there are limitations to our study. Although this is a large population, donor and recipient data (including weight) may have been miscoded or misclassified. However, any errors in weight coding would be expected to be distributed evenly between weight difference categories and thus would be unlikely to significantly bias results. Additionally, timing of donor and recipient weight measures relative to kidney transplantation was not documented, and thus the weights included in this analysis were not necessarily representative of the most current weight for donor or recipient.
The combination of size and sex mismatch is a clinically relevant exposure which is easily identifiable and appears to have a significant effect on graft outcome. This study provides some suggestion that these factors could be collectively considered in organ allocation programs with avoidance of the combination of large weight discrepancy (D<R) and sex mismatch. Future studies should be targeted at identifying the measure of size mismatch between donor and recipient that has the highest predictive value for post-transplant outcomes.
Disclosures
None.
Acknowledgments
The data reported here have been supplied by the Minneapolis Medical Research Foundation as the contractor for the Scientific Registry of Transplant Recipients (SRTR).
There was no funding source for this study.
The interpretation and reporting of these data are the responsibility of the authors and in no way should be seen as an official policy of or interpretation by the SRTR or the US government.
All authors reviewed and approved the final manuscript.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
See related editorial, “Donor Quality in the Eye of the Beholder: Interactions between Nonimmunologic Recipient and Donor Factors as Determinants of Graft Survival,” on pages 565–567.
References
- 1.Almond PS, Matas A, Gillingham K, Dunn DL, Payne WD, Gores P, Gruessner R, Najarian JS: Risk factors for chronic rejection in renal allograft recipients. Transplantation 55: 752–756, discussion 756–757, 1993 [DOI] [PubMed] [Google Scholar]
- 2.Brenner BM, Cohen RA, Milford EL: In renal transplantation, one size may not fit all. J Am Soc Nephrol 3: 162–169, 1992 [DOI] [PubMed] [Google Scholar]
- 3.Guedes AM, Malheiro J, Fonseca I, Martins LS, Pedroso S, Almeida M, Dias L, Castro Henriques A, Cabrita A: Over ten-year kidney graft survival determinants. Int J Nephrol 2012: 302974, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Swanson SJ, Hypolite IO, Agodoa LY, Batty DS Jr, Hshieh PB, Cruess D, Kirk AD, Peters TG, Abbott KC: Effect of donor factors on early graft survival in adult cadaveric renal transplantation. Am J Transplant 2: 68–75, 2002 [DOI] [PubMed] [Google Scholar]
- 5.el-Agroudy AE, Hassan NA, Bakr MA, Foda MA, Shokeir AA, Shehab el-Dein AB: Effect of donor/recipient body weight mismatch on patient and graft outcome in living-donor kidney transplantation. Am J Nephrol 23: 294–299, 2003 [DOI] [PubMed] [Google Scholar]
- 6.Giral M, Foucher Y, Karam G, Labrune Y, Kessler M, Hurault de Ligny B, Büchler M, Bayle F, Meyer C, Trehet N, Daguin P, Renaudin K, Moreau A, Soulillou JP: Kidney and recipient weight incompatibility reduces long-term graft survival. J Am Soc Nephrol 21: 1022–1029, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kasiske BL, Snyder JJ, Gilbertson D: Inadequate donor size in cadaver kidney transplantation. J Am Soc Nephrol 13: 2152–2159, 2002 [DOI] [PubMed] [Google Scholar]
- 8.McGee J, Magnus JH, Islam TM, Jaffe BM, Zhang R, Florman SS, Hamm LL, Mruthinti N, Sullivan K, Slakey DP: Donor-recipient gender and size mismatch affects graft success after kidney transplantation. J Am Coll Surg 210: 718–725.e1, 725–726, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hugen CM, Polcari AJ, Farooq AV, Fitzgerald MP, Holt DR, Milner JE: Size does matter: Donor renal volume predicts recipient function following live donor renal transplantation. J Urol 185: 605–609, 2011 [DOI] [PubMed] [Google Scholar]
- 10.Jindal RM, Ryan JJ, Sajjad I, Murthy MH, Baines LS: Kidney transplantation and gender disparity. Am J Nephrol 25: 474–483, 2005 [DOI] [PubMed] [Google Scholar]
- 11.Kolonko A, Chudek J, Wiecek A: Nephron underdosing as a risk factor for impaired early kidney graft function and increased graft loss during the long-term follow-up period. Transplant Proc 45: 1639–1643, 2013 [DOI] [PubMed] [Google Scholar]
- 12.Nyengaard JR, Bendtsen TF: Glomerular number and size in relation to age, kidney weight, and body surface in normal man. Anat Rec 232: 194–201, 1992 [DOI] [PubMed] [Google Scholar]
- 13.Gratwohl A, Döhler B, Stern M, Opelz G: H-Y as a minor histocompatibility antigen in kidney transplantation: A retrospective cohort study. Lancet 372: 49–53, 2008 [DOI] [PubMed] [Google Scholar]
- 14.Tan JC, Kim JP, Chertow GM, Grumet FC, Desai M: Donor-recipient sex mismatch in kidney transplantation. Gend Med 9: 335–347.e2, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sapir-Pichhadze R, Tinckam KJ, Laupacis A, Logan AG, Beyene J, Kim SJ: Immune sensitization and mortality in wait-listed kidney transplant candidates. J Am Soc Nephrol 27: 570–578, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tennankore KK, Kim SJ, Alwayn IPJ, Kiberd BA: Prolonged warm ischemia time is associated with graft failure and mortality after kidney transplantation. Kidney Int 89: 648–658, 2016 [DOI] [PubMed] [Google Scholar]
- 17.Azur MJ, Stuart EA, Frangakis C, Leaf PJ: Multiple imputation by chained equations: What is it and how does it work? Int J Methods Psychiatr Res 20: 40–49, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mosteller RD: Simplified calculation of body-surface area. N Engl J Med 317: 1098, 1987 [DOI] [PubMed] [Google Scholar]
- 19.Goldberg RJ, Smits G, Wiseman AC: Long-term impact of donor-recipient size mismatching in deceased donor kidney transplantation and in expanded criteria donor recipients. Transplantation 90: 867–874, 2010 [DOI] [PubMed] [Google Scholar]
- 20.Brenner BM, Milford EL: Nephron underdosing: A programmed cause of chronic renal allograft failure. Am J Kidney Dis 21[Suppl 2]: 66–72, 1993 [DOI] [PubMed] [Google Scholar]
- 21.Bertoni E, Rosati A, Zanazzi M, Di Maria L, Moscarelli L, Colonna FM, Gallo M, Romagnani P, Biagini M, Piperno R, Salvadori M: Functional reserve and hyperfiltration after cadaveric renal transplantation. Transplant Proc 33: 3363–3364, 2001 [DOI] [PubMed] [Google Scholar]
- 22.Luyckx VA, Shukha K, Brenner BM: Low nephron number and its clinical consequences. Rambam Maimonides Med J 2: e0061, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Popli R, Sahaf B, Nakasone H, Lee JY, Miklos DB: Clinical impact of H-Y alloimmunity. Immunol Res 58: 249–258, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gaston RS, Hudson SL, Julian BA, Laskow DA, Deierhoi MH, Sanders CE, Phillips MG, Diethelm AG, Curtis JJ: Impact of donor/recipient size matching on outcomes in renal transplantation. Transplantation 61: 383–388, 1996 [DOI] [PubMed] [Google Scholar]
- 25.Ghafari A, Etemadi J, Ardalan M: Impact of donor/recipient body weight mismatch on allograft outcome in renal transplant recipients. Transplant Proc 40: 135–136, 2008 [DOI] [PubMed] [Google Scholar]
- 26.Schnitzler MA, Whiting JF, Brennan DC, Lin G, Chapman W, Lowell J, Boxerman S, Hardinger KL, Kalo Z: The expanded criteria donor dilemma in cadaveric renal transplantation. Transplantation 75: 1940–1945, 2003 [DOI] [PubMed] [Google Scholar]
- 27.Merion RM, Ashby VB, Wolfe RA, Distant DA, Hulbert-Shearon TE, Metzger RA, Ojo AO, Port FK: Deceased-donor characteristics and the survival benefit of kidney transplantation. JAMA 294: 2726–2733, 2005 [DOI] [PubMed] [Google Scholar]
- 28.Leppke S, Leighton T, Zaun D, Chen SC, Skeans M, Israni AK, Snyder JJ, Kasiske BL: Scientific registry of transplant recipients: Collecting, analyzing, and reporting data on transplantation in the United States. Transplant Rev (Orlando) 27: 50–56, 2013 [DOI] [PubMed] [Google Scholar]
- 29.Massie AB, Kucirka LM, Segev DL: Big data in organ transplantation: Registries and administrative claims. Am J Transplant 14: 1723–1730, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]