Abstract
Objectives
Exosomes are 50-90 nm extracellular membrane particles that may mediate trans-cellular communication between cells and tissues. We have reported that human urinary exosomes contain miRNA that are biomarkers for salt sensitivity and inverse salt sensitivity of blood pressure. This study examines exosomal transfer between cultured human renal proximal tubule cells (RPTCs) and from RPTCs to human distal tubule and collecting duct cells.
Design and methods
For RPTC-to-RPTC exosomal transfer, we utilized 5 RPTC lines producing exosomes that were fluorescently labeled with exosomal-specific markers CD63-EGFP or CD9-RFP. Transfer between RPTCs was demonstrated by co-culturing CD63-EGFP and CD9-RFP stable clones and performing live confocal microscopy. For RPTC-to-distal segment exosomal transfer, we utilized 5 distal tubule and 3 collecting duct immortalized cell lines.
Results
Time-lapse videos revealed unique proximal tubule cellular uptake patterns for exosomes and eventual accumulation into the multi-vesicular body. Using culture supernatant containing exosomes from 3 CD9-RFP and 2 CD63-EGFP RPTC cell lines, all 5 distal tubule cell lines and all 3 collecting duct cell lines showed exosomal uptake as measured by microplate fluorometry. Furthermore, we found that RPTCs stimulated with fenoldopam (dopamine receptor agonist) had increased production of exosomes, which upon transfer to distal tubule and collecting duct cells, reduced the basal reactive oxygen species (ROS) production rates in those recipient cells.
Conclusion
Due to the complex diversity of exosomal contents, this proximal-to-distal vesicular inter-nephron transfer may represent a previously unrecognized trans-renal communication system.
Keywords: exosomes, proximal tubule, ROS, distal tubule, collecting duct, dopamine
1.1 Introduction
During normal cellular metabolism, exosomes (50-90 nm plasma membrane particles) are secreted from specific membrane areas and contain selectively packaged mRNA, proteins, miRNA and other cellular contents.1 Apparently, exosomal signaling represents a higher order of inter-cellular communication as compared to autocrine or paracrine signaling since exosomes can change according to alterations in cellular metabolism and influence specific cellular structures and processes in the recipient cells. For example, hypoxia can increase the concentration of caveolin-1 in plasma exosomes in patients with glioblastoma.2
Exosomal signaling in the lumen of the renal nephron is unique in that plasma exosomes cannot cross the glomerular filtration apparatus and thus intra-nephron exosomes originate exclusively from the luminal epithelial cells.3 The regulation of renal function may be influenced by exosomal contents. Protein epitopes exposed on the surface of exosomes may interact with extracellular targets on cells in contact with the tubular lumen.4 Some of the targeting molecules on the exosomes and docking molecules on the recipient cells are known in certain immune cell interactions; 5 however, in the context of the human nephron, neither the targeting molecules nor the docking molecules have yet been identified. In addition, exosomes isolated from human urine contain mRNA from every segment of the renal tubule.6 Selective knockout of a collecting duct-selective marker (V-VATPase-B1) in mice eliminated this marker from exosomes in their urine, demonstrating that exosomes may contain nephron-specific contents and accurately represent the expression levels of the donor cells. One report of exosomal stimulation resulting in a change of phenotype in the kidney involved a single tubule segment and the use of desmopressin to stimulate an increase in aquaporin 2 content of secreted exosomes from cortical collecting duct cells, which then increased both aquaporin 2 expression and water transport in another batch of cortical collecting duct cells.7
Since inter-nephron segment exosomal signaling has not been demonstrated in human kidney, we examined both intra- and inter-nephron segment exosomal signaling in cells derived from proximal and distal tubules as well as collecting duct. Renal tubular reactive oxygen species (ROS) have been linked to hypertension in experimental animals.8,9 Furthermore, dopamine and angiotensin II (Ang II) oppose each other in regulating renal sodium excretion, which is a mechanism by which sodium homeostasis and hence blood pressure is maintained.8,10 Since it is known that dopamine decreases ROS10 while Ang II increases ROS in the renal proximal tubule,11 we investigated whether exosomes produced by cultured renal proximal tubule cells (RPTCs), after dopaminergic or Ang II stimulation, would modulate the local ROS production in RPTCs, distal tubule (DT) cells, or collecting duct (CD) cells.
1.2 Materials and Methods
1.2.1 Renal Proximal Tubule Cell Culture
RPTCs were obtained from normal tissue from nephrectomies in human subjects, under an institutional review board-approved protocol according to the Declaration of Helsinki, Title 45, Part 46, and U.S. Code of Federal Regulations. Cell lines were isolated and immortalized as previously described.12, 13,14 RPTCs were extensively characterized and demonstrated only proximal tubule-specific characteristics.15 We utilized i16, i22 and i25 RPTC cell lines in the current studies.
1.2.2 Distal Tubule and Collecting Duct Cell Isolation, Culture and Characterization
DT and CD cell lines were selectively isolated (Invitrogen Cellection). Human kidney tissue was chopped into 1 mm sections (McIlwain) and then digested with collagenase A (Roche Diagnostics, 2 mg/mL, 37°C, 25 minutes). Single cell suspensions were isolated (40 micron filter, Becton Dickinson) followed by lectin or immuno-magnetic affinity separation.
Three DT cell lines were isolated using biotinylated dolichos biflorus agglutinin 16 (DBA, Vector Labs, 2.5 mg/500 mL) incubated with the cell suspension for 30 minutes at room temperature, followed by anti-biotin magnetic nanoparticle conjugated secondary antibody (Invitrogen). Alternatively 2 cell lines were established using anti-Tamm-Horsfall protein17 (THP, Santa Cruz 20631,1:200 dilution) antibody and an anti-rabbit magnetic secondary antibody (Invitrogen). Three immortalized human CD cell lines were isolated using antibody to L1CAM (1:100) and an anti-mouse magnetic secondary antibody (Invitrogen).
Both the DT and CD were cultured in DMEM-F12 medium with10% FBS and the same supplements used for growing RPTC cells.13 Cells were immortalized using human terminal transferase (hTERT)-containing lentiviral infection and selected with G418.13 The DBA and THP-isolated DT cell lines and CD cell lines were characterized (Online Supplement).
1.2.3 Exosome Purification
Exosomes were isolated from cultured cells grown in serum free media using our previously published method,18 (modified method of Gonzales et al).19 Serum free media was found to be necessary because of the high concentration of exosomes in serum competing during transfer. Details of this ultracentrifugation method and characterization of exosomes are in the Online Supplement.
1.2.4 Western Blotting for TSG101
Purified exosome pellets were re-suspended in MPER lysis buffer (Thermo Scientific) and protein concentration was determined by bicinchoninic acid (BCA) assay (Thermo Scientific). 10 μg protein per lane were analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS PAGE) and western blotting using an anti-TSG101 antibody (Sigma HPA006161, 1:1000 dilution) and Licor anti-rabbit secondary antibody (IRDye 800CW, 1:15,000 dilution) and scanning on an Odyssey infrared imaging system (Licor).
1.2.5 Proximal Tubule-derived Fluorescently Labeled Exosomes
Proximal tubule cell lines were created which stably express two different GFP fusion proteins known to fluorescently label exosomes. Two different lentiviral constructs with green or red fluorescent proteins (GFP, RFP) were purchased from System Biosciences: CYTO-tracer pCT-CD63-GFP and pCT-CD9-RFP. The plasmids were packaged into virus and three cell lines were transduced with CD63-GFP containing lentiviral particles, and two cell lines were created with CD9-RFP containing lentiviral particles. The exosomal origin of the fluorescence found in cell culture supernatant was verified by microplate fluorometry (PHERAstar FS, BMG Labtech) using purified exosomes.
An additional method, using transient transfection of RPTCs with plasmids with yellow fluorescent proteins (YFP), was utilized (LAMP1-YFP or CD82-YFP fusion protein, Addgene), was carried out under conditions previously described.15,20 The transfected RPTCs were grown on magnetic GEM™ microcarriers,13 and then were co-incubated with non-transfected RPTCs on non-magnetic GEMs. YFP fluorescence transfer was measured on the non-magnetic GEMS by fluorometry after removal of the donor fluorescent magnetic GEM™ microcarriers.
1.2.6 Exosomal Transfer
Proximal tubule-derived exosomes were isolated by ultracentrifugation of serum-free RPTC 24 hr culture supernatants and re-suspended back into the same volume of serum-free medium. The purified exosomes were incubated with RPTCs, DT cells, and CD cells for 24 hours. The recipient cells were then washed three times with HBSS and analyzed by microplate fluorometry. The amount of exosomal transfer was calculated as the fluorescence at the appropriate emission wavelength minus the background fluorescence obtained from a well containing the same number of recipient cells that received exosomes isolated from non-fluorescing RPTC cells.
1.2.7 Measurement of ROS Production
RPTC, DT and CD cells were labeled with a fluorescent total ROS detection agent (Enzo), which was detected using a PHERAstar FS microplate reader. A basal ROS production rate was measured for each well for 30 minutes using identical gain and timing for all experiments, after which fenoldopam (FEN, D1-like receptor agonist, 1 μmol/L) or angiotensin II (Ang II, 10 nmol/L) were added to each well using automated injectors. The change in ROS production rate was measured for 30 minutes, after which a ROS inhibitor (N-Acetyl-L-Cysteine, NAC, 5 mmol/L) was added and the change in ROS production rate (Relative Fluorescence Units (RFU)/min) for each treatment was measured and normalized to the negative vehicle control (VEH).
For RPTC exosome-mediated effects on the change in ROS production rate in recipient distal segment cells, fenoldopam-stimulated RPTCs were incubated for 16 hours and the exosomes purified by ultracentrifugation. The purified exosomes were then placed onto DT and CD cells for 24 hours before measuring basal NAC-inhibitable ROS production in those recipient cells. The ROS production was compared to that in cells that had received exosome-free media orexosomes from vehicle-treated donor cells.
1.2.8 Confocal microscopy of transferred exosomes
Purified exosomes from CD63-GFP transfected RPTCs were added to non-transfected RPTCs in culture and incubated overnight. Time lapse confocal microscopy was performed in an automated spinning disk confocal microscope using a 60× water immersion objective (Olympus I×81 with Disk Scanning Unit) in a temperature, CO2 and humidity controlled incubator. Static images of transferred CD9-RFP containing exosomes into CD63-GFP stable RPTCs were also taken using the same confocal microscope setup.
1.2.9 Proteomic Analysis by Mass Spectrometry
Details of the methods used to analyze the proteins in the RPTC exosomes are outlined in the Online Supplement.
1.2.10 Statistical Analysis
The data are expressed as mean ± SE. Comparisons within and among groups were made by repeated measures or factorial ANOVA, respectively, followed by Holm-Sidak or Duncan's test. Student's t-test was used for two-group comparisons. P values of <0.05 were considered significant.
1.3 Results (Figures S1-S3 are in the Online Supplement)
1.3.1 Technical Overview
Experiments shown in Figure S1 depict our exosome uptake assays and readout by microplate fluorometry. DT and CD cells that received exosomes secreted by fluorescent CD63-GFP RPTCs showed a significant increase in fluorescent signal (*P<0.05, N=12) compared to DT and CD cells that received exosomes from non-fluorescent (NF) RPTC. When NF exosomes were mixed with the same concentration of CD63 exosomes and transferred, there was a saturable reduction in exosomal uptake. Mixing CD63-purified exosomes with NF exosomes from fetal calf serum (FCS) also caused decreased fluorescent signal. Depletion of CD63-containing exosomes using magnetic beads conjugated with the extracellular epitope-specific anti-CD63 monoclonal antibody completely blocked the fluorescent signal (5th bar set). The addition of 2 mmol/L beta-methyl-cyclodextrin (BMCD), a lipid raft disrupting and cholesterol binding molecule, produced a significant decrease in fluorescent signal in both DT and CD cells. This indicates that the binding and uptake of proximal tubule-derived exosome particles into DT and CD cells represent a combination of generalized and specific exosomal uptake processes.
1.3.2 Determination of Segment-Specific Affinity Receptors for Cell Isolation
Affinity reagents for cell isolation were chosen based on frozen section staining (Figure S2). LTA lectin stains the apical membrane of the proximal tubule, 21 DBA lectin stains the distal tubule,16 and L1CAM stains the collecting duct. 22
1.3.3 Fluorescent Exosome Transfer
We investigated whether CD9 and CD63-labeled exosomes purified from RPTC culture supernatants could transfer to RPTC, DT and CD cells (Figure 1). Exosomes were purified from 3 different CD9-RFP RPTC lines and transferred to 3 different RPTC lines, 5 DT cell lines and 3 CD cell lines and fluorescence was measured at 24 hours (N=18-36, *P<0.001 above background, Figure 1B). Similarly, CD63-GFP exosomes from 2 RPTC lines also transferred to the RPTC, DT, and CD cell lines (N=18-36, *P<0.01 above background, Figure 1C).
Figure 1.
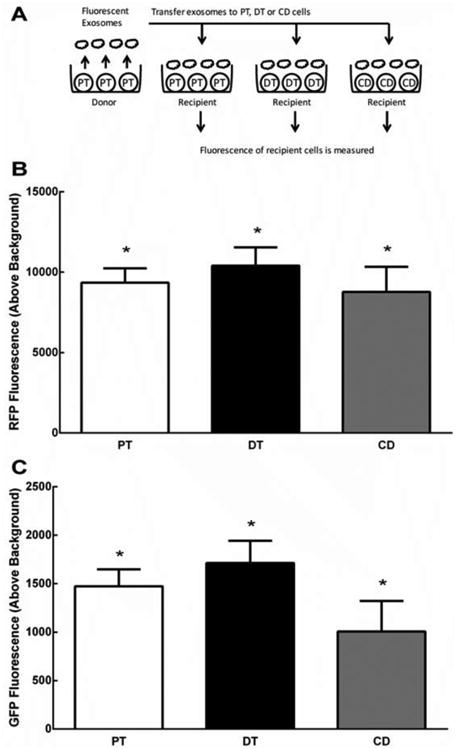
(A) Fluorescently-labeled exosomes produced from human proximal tubule cells (PT) were secreted into the supernatant. Exosomes were purified from the supernatant and transferred onto non- fluorescent recipient cells (PT, DT, or CD) for 24 hrs, and then fluorescence was measured. (B) Depicts significant fluorescence of recipient cells after receiving CD9-RFP exosomes from donor PT (N=18-36, *P<0.001 above background), indicating uptake of CD9 exosomes in all 3 cell types. (C) Depicts significant fluorescence of recipient cells after receiving CD63-GFP exosomes from donor RPTC (N=18-36, *P<0.01 above background), indicating uptake of CD63 exosomes in all 3 cell types.
1.3.4 3D Co-culture and Fluorescent Exosome Transfer
We also showed that exosomes were able to transfer between cells by utilizing a unique co-culturing system. RPTCs were electroporated while on alginate microcarriers (GEM™) with either Lamp1-YFP or CD82-YFP, previously shown to localize to the multivesicular body and be secreted in exosomes 23 (Addgene). RPTCs cultured on magnet-free GEMs were mixed with the electroporated RPTCs on magnetic GEMs and co-cultured for 24 or 48 hours. The electroporated cells on GEMs were then magnetically separated and the remaining transferred fluorescent exosomes were measured by microplate fluorometry. We found that both YFP fusion proteins were secreted into the culture medium (data not shown) and taken up by the non-transfected cells (Figure 2A). CD82-YFP exosomes transferred to RPTCs at 24 and 48 hours (N=4, *P<0.05 above background, Figure 2B). RPTC-derived LAMP1-YFP exosomes transferred to RPTCs at 24 and 48 hours (N=4, *P<0.05 above background, Figure 2C).
Figure 2. Transfer of fluorescent exosomes from donor human proximal tubule cells (PT) grown on microcarriers to non-fluorescent recipient PT.
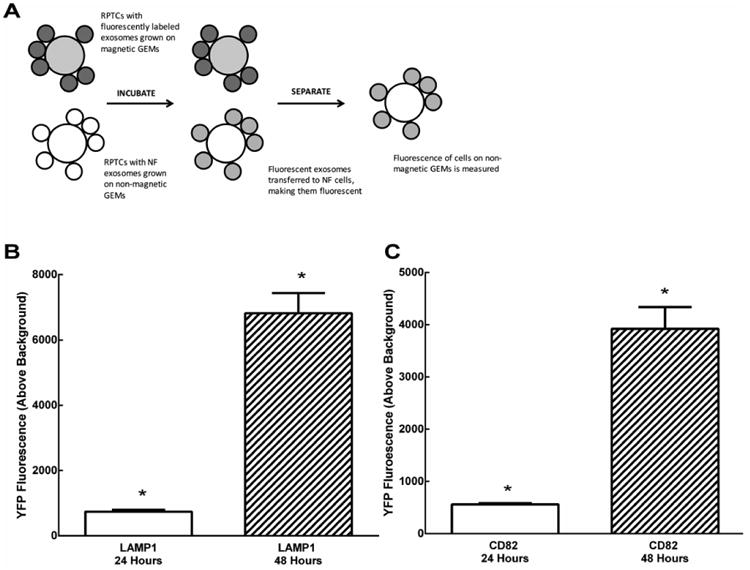
(A) PT cells transfected with LAMP1-YFP or CD82-YFP were grown on magnetic GEM™ microcarriers, while non-transfected PT cells were grown on non-magnetic GEMs. Magnetic and non-magnetic GEMs were co-cultured for 24 and 48 hours. Magnetic GEMs were then removed and fluorescence on non-magnetic GEMs was measured. (B) Depicts an increase in fluorescence of cells on non-magnetic GEMs after 24 and 48 hours (N=4, *P<0.05 above background), indicating transfer of CD82 exosomes. (C) Depicts an increase in fluorescence of cells on non-magnetic GEMs after 24 and 48 hours (N=4, *P<0.05 above background), indicating transfer of LAMP1 exosomes.
1.3.5 Stimulation of Exosomal Secretion
We next investigated whether stimulation had an effect on exosomal secretion into the culture supernatant. Stimulation of either the natriuretic dopaminergic (using D1-like agonist FEN) or antinatriuretic renin angiotensin systems (RAS) (using the AT1R agonist Ang II) stimulated exosome secretion. FEN (1 μmol/L, 24 hr) produced a 2.38±0.053 fold increase in exosome release (N=15, *P<0.05 vs VEH). Ang II stimulation (10 nmol/L, 24 hr) caused a 4.71 ± 0.35 fold increase in exosome release (N=18, *P<0.05 vs VEH) as measured by TSG101 western blotting (Figure 3). This increase in exosomal content was verified by western blotting with the exosome marker Alix (data not shown).
Figure 3. Effect of dopaminergic or renin angiotensin system (RAS) stimulation on exosomal secretion.
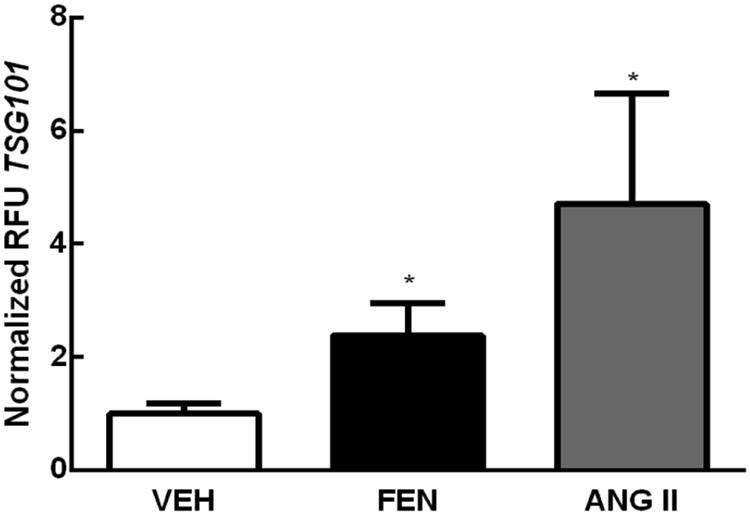
RPTCs show a 2.38±0.053 (N=15, *P<0.05) and 4.71±0.35 (N=18, *P<0.05) fold increase in TSG101 following FEN (1 μmol/L, 24 hr) and Ang II (10 nmol/L, 24 hr) treatment, respectively, indicating an increase in exosome release following treatment.
One of the common signaling pathways shared between the dopamine and angiotensin systems is an increase in intracellular calcium, previously shown to increase exosome secretion.24 Agonist-induced responses persisted after removal of extracellular calcium, indicating that RPTC exosomal release is not dependent upon a calcium influx into cells from the culture media (data not shown).
1.3.6 Localization of Transferred Exosomal Proteins
CD63 exosomes transferred to RPTCs accumulate in microvesicles and are shuttled rapidly in the receiving cells in two distinct patterns (Supplemental movie). CD63-GFP purified exosomes (of RPTC origin) transferred to CD9-RFP stable RPTCs and localized to the multivesicular body and other subcellular compartments (Figure S3).
1.3.7 Dopaminergic and RAS Stimulates ROS production in Different Cell Types Along the Nephron
We tested if RPTC, DT and CD cells all robustly respond to dopaminergic D1-like stimulation with a decreased ROS production. FEN stimulation (1 μmol/L) of RPTC, DT, and CD cells reduced ROS production by 42.1 ± 1.4%, N=24; 54.3 ± 3.4%, N= 25; 40.2 ± 4.4%, N=15, respectively, *P<0.0001 vs VEH. Stimulation of cells with Ang II (10 nmol/L) increased ROS production 22.4 ± 2.4%, N=24; 20.2 ± 8.3%, N= 25; 27.3 ± 5.2%, N=15, respectively, *P<0.01 vs VEH (Figure 4).
Figure 4.
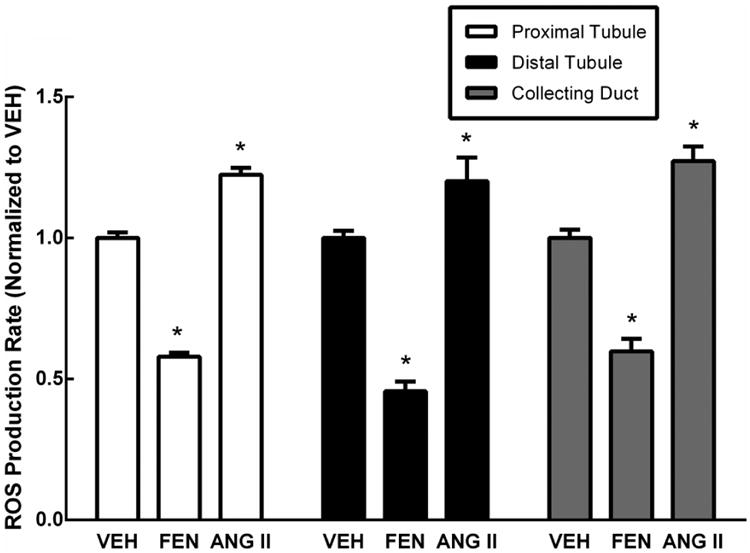
ROS production following treatment with fenoldopam (FEN) and angiotensin II (ANG II). Proximal tubule, distal tubule, and collecting duct cells show decreased ROS (42.1±1.4%, N=24; 54.3±3.4%, N= 25; 40.2±4.4%, N=15, respectively, *P<0.0001 vs VEH) following FEN (1 μmol/L) treatment. Treatment with ANG II (10 nmol/L) increased ROS in the RPTC, DT and CD cells (22.4±2.4%, N=24; 20.2±8.3%, N= 25; 27.3±5.2%, N=15, respectively, *P<0.01 vs VEH). The vehicle control (VEH) is serum-free media.
1.3.8 Dopaminergic Stimulation Mediates Phenotypic Transfer via Exosomes
We examined whether a phenotypic transfer of ROS production rate from FEN or Ang II-stimulated RPTCs to DT or CD cells could be mediated by washed exosomes isolated from the stimulated RPTCs and then transferred to DT and CD cells (Figure 5). When FEN stimulated cell exosomes were transferred, the ROS production rate was found to decrease in both DT cells (17.5 ± 2.2% decrease, N=12, *P<0.001) and CD cells (15.7 ± 3.4% decrease, N=12, *P<0.001). No change in ROS production from RPTC to DT and CD was seen when exosomes isolated from Ang II-stimulated RPTCs were transferred to DT and CD cells.
Figure 5.
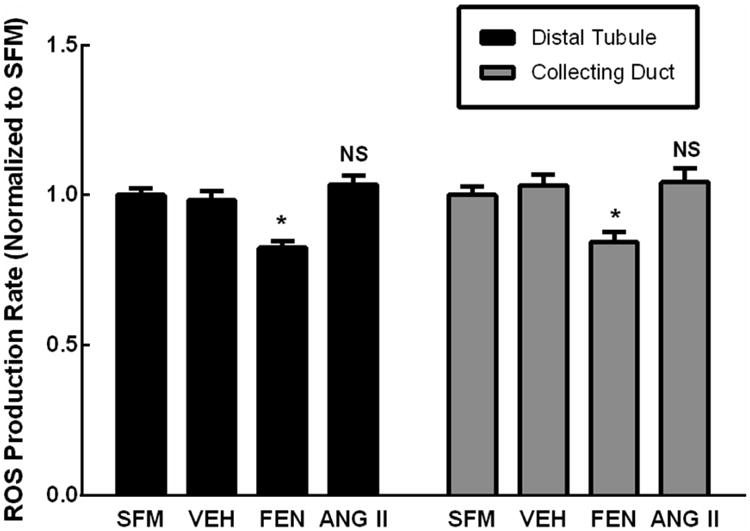
Exosomes isolated from RPTCs treated with fenoldopam (FEN) or angiotensin II (ANG II) were purified and transferred to untreated recipient cells (distal tubule, collecting duct). Incubation with exosomes purified from FEN-stimulated donor PT led to a 17.5±2.2% decrease (N=12, *P<0.001) in ROS production rate in DT cells, and a 15.7±3.4% decrease (N=12, *P<0.001) in ROS production rate in CD cells. However, incubation with exosomes isolated from ANG II-stimulated donor RPTCs did not change ROS production. Data was normalized to serum-free media (SFM) that was not incubated with cells. Another vehicle control (VEH) was SFM with exosomes derived from non-stimulated donor RPTCs.
1.3.9 Proteomic Analysis Validates Microvesicle Transfer Components
Proteomic analysis of proximal tubule cell line-derived exosomes identified 155 unique proteins (see Online Supplement Tables S1 and S2), 68 of which were identified previously as urinary exosomal proteins.25 There is a link in Table S1 to the Human Protein Atlas kidney immunohistochemistry and RNA sequencing section which can be used to verify human kidney expression.
1.4 Discussion
A compelling hypothesis is that the proximal nephron signals the distal nephron via exosomes shed into the tubular lumen. Alternatively, one might consider exosomal uptake by the distal nephron as a means by which the distal nephron understands proximal tubular actions that are reflected by the contents of their membrane and cytoplasm. We isolated, immortalized, and characterized DT and CD cells from human kidney since there are none commercially available. Using these new cell lines, we demonstrated that both DT and CD cells take up RPTC exosomes. Our definition of exosomes assumes that our preparation does not consist of apoptotic bodies and other forms of microvesicles, which are larger than exosomes and would not be found using our ultracentrifugation method. A subset of ectosomes (particles shed directly from the cell membrane and not from intracellular sorting particles) are the same size as exosomes and with similar extracellular epitope orientation. However, ectosomes are defined as CD63 negative in immune cells,26 so by this definition the particles used in this study would not be considered ectosomes.
When CD63-GFP-containing exosomes were mixed with non-fluorescent exosomes isolated from FCS, the fluorescent signal in the recipient DT or CD cells was greatly diminished. This finding suggests a docking and uptake system that is able to be saturated and indicates that macropinocytosis is not a major component of exosome uptake in this model system. Exosomal endocytosis was non-clathrin lipid raft dependent since it was inhibited by BCMD, similar to that found by Svensson. 27
Uptake of exosomes may be concentration dependent. To rule out the possibility that we were using excessively diluted exosomes, we developed a liquid phase co-culture experiment. We mixed two populations of RPTCs, one growing on magnetic 3D microcarriers and another growing on non-magnetic microcarriers.13 Exosomal uptake by the recipient cell line (isolated magnetically) appeared highly efficient since in this culture system the exosome concentration in the media was a closer approximation of what one would expect in the nanoliters of fluid in the human nephron.
Exosome production increases in response to cell stimulation in various models.4 In renal cells, we found that both fenoldopam (dopamine receptor agonist) and Ang II stimulate exosome production. Normally, Ang II and fenoldopam have opposing effects in RPTC on sodium uptake. The secretion of exosomes is calcium (Ca++) dependent 24 and both fenoldopam and Ang II increase intracellular Ca++. 28, 29 It would be of interest to determine if there is a difference in exosome contents between these two stimulatory pathways. We did determine that there was a response of both DT and CD only to the transferred fenoldopam-stimulated exosomes. The component in the exosomes mediating these effects could be altered genetic material transfer (mRNA, miRNA) 30 that changed expression in the recipient cells, second messengers including cAMP, or direct transfer of proteins like cell surface receptors or enzymes. The reduction in ROS production is less than direct stimulation of the receptor but still highly significant. We ruled out the transfer of fenoldopam trapped in the exosomal-like particles since a vanishingly small amount of fenoldopam would be trapped in the exosomes. Furthermore, fenoldopam is known to oxidize completely in cell culture media during the time frame of our experiments.
To further validate our cell culture model and exosome isolation, a small-scale exosomal proteomics study was performed. The Human Protein Atlas kidney link on the proteomics spreadsheet allows us to see that a vast majority of proteins identified by exosomal proteomics show abundant protein and corresponding RNA expression in proximal tubule cells. Additionally, many show expression patterns consistent with exosomes expressed from proximal tubule cells since there is immunoreactive tubular lumen staining. The list of proteins identified was analyzed for common pathways by an online WEB-based GEne SeT AnaLysis Toolkit called WebGestalt (see Gestalt Analysis supplemental file).31,32 Many of the protein signalling groups identified were phagosomes, endosomes, focal adhesions, gap junctions, ubiquitin proteasomes, integrins, and syndecans; all of which are necessary for exosome function.
The lack of dependence on extracellular calcium for exosome secretion does not rule out release form intracellular stores as found in neurons and cancer cells.33 Once taken up by the recipient cell, exosomes were found in at least two subcellular compartments, suggesting that several pathways are involved in exosomal uptake in the kidney including lipid rafts.27 It is not known if various pathways are at play in the same cell or if uptake mechanisms are specific to cell type. The association of exosomal miRNA content with salt sensitivity of blood pressure18 suggest that miRNAs may be the mediators of the phenotypic change observed in this study. Our model system will help us determine if specific miRNA from human RPTCs change in response to dietary salt and cause resulting changes in DT and CD cells.
We show for the first time in human renal cells that each major segment of the nephron (RPTC, DT, and CD) demonstrated a robust inhibition of ROS in response to dopamine stimulation and an increase in ROS with Ang II stimulation. Interestingly, when we collected secreted exosomes from RPTC cells exposed to either fenoldopam or Ang II and incubated them with both DT and CD cells, we found that only the exosomes secreted by fenoldopam-stimulated cells could modulate ROS activity. Possible explanations for this differential effect is that a reduction in ROS by the dopaminergic system may be a physiologic event that needs to be transmitted downstream in the nephron while the Ang II stimulation of ROS is an event that is mediated locally. Alternatively, Ang II may have to be stimulated at a different concentration in order to achieve a particular threshold. Ang II can stimulate natriuresis or antinatriuresis depending on the dose employed.34
Future work will focus on Identifying the molecule within the exosome mediating this effect, including determining whether specific exosomal miRNAs are causing changes in distal nephron expression related to ROS production.
In summary, exosomes have emerged as an exciting new area of cell signaling research. Unlike classic G protein coupled receptor pathways, exosomes involve a more complex level of signaling in that a multitude of biomolecules are captured from the cell cytoplasm as well as membrane proteins during the formation of exosomes. Thus, this donor cell “snapshot” of cell components is transferred to the recipient cell by a process involving complex exosome docking and fusion initiated by cell surface molecules. Model systems are being developed to study the nature of the exosome and the responses they elicit in recipient cells under varying physiologic conditions and pathological states. Exosomes may also provide a novel therapeutic approach to controlling complex multifactorial chronic diseases such as hypertension.
Supplementary Material
Highlights.
Exosomes have emerged as an exciting new area of cell signaling research
Exosomal capture of biomolecules from the donor cell provides a cytoplasmic “snapshot” that is transferred to the recipient cell
Model systems such as ours study the nature of the responses exosomes elicit in recipient cells
Exosomal contents contain biomarkers that may provide novel clinical diagnostic opportunities
Acknowledgments
The authors thank Helen E. McGrath for her assistance in the preparation and editing of this paper and Charles E. Lyons in his assistance during the acquisition of the mass spectral data.
Sources of Funding This work was funded by National Institutes of Health grants HL074940 and DK039308.
Footnotes
Disclosures There are no COI to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Wen H, Frokiaer J, Kwon TH, et al. Urinary excretion of aquaporin-2 in rat is mediated by a vasopressin-dependent apical pathway. J Am Soc Nephrol. 1999;10:1416–1429. doi: 10.1681/ASN.V1071416. [DOI] [PubMed] [Google Scholar]
- 2.Kucharzewska P, Christianson HC, Welch JE, et al. Exosomes reflect the hypoxic status of glioma cells and mediate hypoxia-dependent activation of vascular cells during tumor development. Proc Natl Acad Sci U S A. 2013;110:7312–7317. doi: 10.1073/pnas.1220998110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pisitkun T, Shen RF, Knepper MA. Identification and proteomic profiling of exosomes in human urine. Proc Natl Acad Sci U S A. 2004;101:13368–13373. doi: 10.1073/pnas.0403453101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van Balkom BW, Pisitkun T, Verhaar MC, et al. Exosomes and the kidney: prospects for diagnosis and therapy of renal diseases. Kidney Int. 2011;80:1138–1145. doi: 10.1038/ki.2011.292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Morelli AE, Larregina AT, Shufesky WJ, et al. Endocytosis, intracellular sorting, and processing of exosomes by dendritic cells. Blood. 2004;104:3257–3266. doi: 10.1182/blood-2004-03-0824. [DOI] [PubMed] [Google Scholar]
- 6.Miranda KC, Bond DT, McKee M, et al. Nucleic acids within urinary exosomes/microvesicles are potential biomarkers for renal disease. Kidney Int. 2010;78:191–199. doi: 10.1038/ki.2010.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Street JM, Birkhoff W, Menzies RI, et al. Exosomal transmission of functional aquaporin 2 in kidney cortical collecting duct cells. J Physiol. 2011;589:6119–6127. doi: 10.1113/jphysiol.2011.220277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gildea JJ. Dopamine and angiotensin as renal counterregulatory systems controlling sodium balance. Curr Opin Nephrol Hypertens. 2009;18:28–32. doi: 10.1097/MNH.0b013e32831a9e0b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chugh G, Lokhandwala MF, Asghar M. Oxidative stress alters renal D1 and AT1 receptor functions and increases blood pressure in old rats. Am J Physiol Renal Physiol. 2011;300:F133–138. doi: 10.1152/ajprenal.00465.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zeng C, Villar VA, Yu P, et al. Reactive oxygen species and dopamine receptor function in essential hypertension. Clin Exp Hypertens. 2009;31:156–178. doi: 10.1080/10641960802621283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jennings BL, Anderson LJ, Estes AM, et al. Involvement of cytochrome P-450 1B1 in renal dysfunction, injury, and inflammation associated with angiotensin II-induced hypertension in rats. Am J Physiol Renal Physiol. 2012;302:F408–420. doi: 10.1152/ajprenal.00542.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Felder RA, Sanada H, Xu J, et al. G protein-coupled receptor kinase 4 gene variants in human essential hypertension. Proc Natl Acad Sci USA. 2002;99:3872–3877. doi: 10.1073/pnas.062694599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gildea JJ, McGrath HE, Van Sciver RE, et al. Isolation, growth, and characterization of human renal epithelial cells using traditional and 3D methods. Methods Mol Biol. 2013;945:329–345. doi: 10.1007/978-1-62703-125-7_20. [DOI] [PubMed] [Google Scholar]
- 14.Gildea JJ, Israel JA, Johnson AK, et al. Caveolin-1 and dopamine-mediated internalization of NaKATPase in human renal proximal tubule cells. Hypertension. 2009;54:1070–1076. doi: 10.1161/HYPERTENSIONAHA.109.134338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gildea JJ, Shah I, Weiss R, et al. HK-2 human renal proximal tubule cells as a model for G protein-coupled receptor kinase type 4-mediated dopamine 1 receptor uncoupling. Hypertension. 2010;56:505–511. doi: 10.1161/HYPERTENSIONAHA.110.152256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nauli SM, Alenghat FJ, Luo Y, et al. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet. 2003;33:129–137. doi: 10.1038/ng1076. [DOI] [PubMed] [Google Scholar]
- 17.Baer PC, Geiger H. Human renal cells from the thick ascending limb and early distal tubule: characterization of primary isolated and cultured cells by reverse transcription polymerase chain reaction. Nephrology (Carlton) 2008;13:316–321. doi: 10.1111/j.1440-1797.2008.00927.x. [DOI] [PubMed] [Google Scholar]
- 18.Gildea JJ, Carlson JM, Schoeffel CD, et al. Urinary exosome miRNome analysis and its applications to salt sensitivity of blood pressure. Clin Biochem. 2013;46:1131–1134. doi: 10.1016/j.clinbiochem.2013.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gonzales PA, Zhou H, Pisitkun T, et al. Isolation and purification of exosomes in urine. Methods in Molecular Biology. 2010;641:89–99. doi: 10.1007/978-1-60761-711-2_6. [DOI] [PubMed] [Google Scholar]
- 20.Gildea JJ, Wang X, Shah N, et al. Dopamine and Angiotensin type 2 receptors cooperatively inhibit sodium transport in human renal proximal tubule cells. Hypertension. 2012;60:396–403. doi: 10.1161/HYPERTENSIONAHA.112.194175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Faraggiana T, Malchiodi F, Prado A, et al. Lectin-peroxidase conjugate reactivity in normal human kidney. J Histochem Cytochem. 1982;30:451–458. doi: 10.1177/30.5.7077075. [DOI] [PubMed] [Google Scholar]
- 22.Allory Y, Audard V, Fontanges P, et al. The L1 cell adhesion molecule is a potential biomarker of human distal nephron injury in acute tubular necrosis. Kidney Int. 2008;73:751–758. doi: 10.1038/sj.ki.5002640. [DOI] [PubMed] [Google Scholar]
- 23.Sherer NM, Lehmann MJ, Jimenez-Soto LF, et al. Visualization of retroviral replication in living cells reveals budding into multivesicular bodies. Traffic. 2003;4:785–801. doi: 10.1034/j.1600-0854.2003.00135.x. [DOI] [PubMed] [Google Scholar]
- 24.Savina A, Furlán M, Vidal M, et al. Exosome release is regulated by a calcium-dependent mechanism in K562 cells. J Biol Chem. 2003;278:20083–20090. doi: 10.1074/jbc.M301642200. [DOI] [PubMed] [Google Scholar]
- 25.Wang Z, Hill S, Luther JM, et al. Proteomic analysis of urine exosomes by multidimensional protein identification technology (MudPIT) Proteomics. 2012;12:329–338. doi: 10.1002/pmic.201100477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sadallah S, Eken C, Schifferli JA. Ectosomes as modulators of inflammation and immunity. Clin Exp Immunol. 2011;163:26–32. doi: 10.1111/j.1365-2249.2010.04271.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Svensson KJ, Christianson HC, Wittrup A, et al. Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid Raft-mediated endocytosis negatively regulated by caveolin-1. J Biol Chem. 2013;288:17713–17724. doi: 10.1074/jbc.M112.445403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gildea JJ, Lahiff DT, Van Sciver RE, et al. A linear relationship between the ex-vivo sodium mediated expression of two sodium regulatory pathways as a surrogate marker of salt sensitivity of blood pressure in exfoliated human renal proximal tubule cells: the virtual renal biopsy. Clin Chim Acta. 2013;421:236–242. doi: 10.1016/j.cca.2013.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gildea JJ, Shah IT, Van Sciver RE, et al. The cooperative roles of the dopamine receptors, D1R and D5R, on the regulation of renal sodium transport. Kidney Int. 2014 doi: 10.1038/ki.2014.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Valadi H, Ekström K, Bossios A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9:654–659. doi: 10.1038/ncb1596. [DOI] [PubMed] [Google Scholar]
- 31.Wang J, Duncan D, Shi Z, et al. WEB-based GEne SeT AnaLysis Toolkit (WebGestalt): update 2013. Nucleic Acids Res. 2013;41:W77–83. doi: 10.1093/nar/gkt439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang B, Kirov S, Snoddy J. WebGestalt: an integrated system for exploring gene sets in various biological contexts. Nucleic Acids Res. 2005;33:W741–748. doi: 10.1093/nar/gki475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Savina A, Fader CM, Damiani MT, et al. Rab11 promotes docking and fusion of multivesicular bodies in a calcium-dependent manner. Traffic. 2005;6:131–143. doi: 10.1111/j.1600-0854.2004.00257.x. [DOI] [PubMed] [Google Scholar]
- 34.Olsen ME, Hall JE, Montaini JP, et al. Angiotensin II natriuresis and anti-natriuresis: role of renal artery pressure in anaesthetized dogs. J Hypertens Suppl. 1984;2:S347–350. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


