Abstract
Clathrin-mediated endocytosis (CME) is a fundamental process in cell biology and has been extensively investigated over the past several decades. Every cell biologist learns about it at some point during his or her education, and the beauty of this process has led many of us to go deeper and make it the topic of our research. Great progress has been made toward elucidating the mechanisms of CME, and the field is becoming increasingly complex, with several hundred new publications every year. This makes it easy to get lost in the vast amount of literature and forget about the fundamentals of the field, which are based on the careful interpretation of simple observations made >40 years ago, as exemplified by a study performed by Anderson, Brown, and Goldstein in 1977. We examine how this seminal study was pivotal to our understanding of CME and its progression into ever-increasing complexity over the past four decades.
THE PATH FROM THE PLASMA MEMBRANE TO THE LYSOSOME
In their landmark study, Roth and Porter (1964) were the first to describe the uptake of yolk protein through small “bristle-coated pits” at the plasma membrane of mosquito oocytes and rightfully predicted their importance for the uptake of extracellular material. In March 1977, when understanding of the process of endocytosis was still in its infancy, Anderson, Brown, and Goldstein performed a study that described the uptake of the low-density-lipoprotein (LDL) receptor from coated regions on the plasma membrane into endocytic vesicles that subsequently fuse with lysosomes (Anderson et al., 1977a). In an era before the advent of fluorescently tagged proteins, relatively little was known about the uptake of cargo proteins by receptors. Goldstein and his colleagues realized the importance of specific cell surface receptor interactions from their studies following the internalization of ferritin-labeled LDL molecules. In a series of beautifully detailed electron micrographs, they observed that ∼70% of LDL was located in small coated regions that made up only ∼2% of the total surface (Figure 1). These regions would later become known as clathrin-coated pits (CCPs). By allowing LDL to bind to cells at 4°C (blocking receptor uptake) and subsequently warming the cells back to 37°C for various times before fixation, they we able to follow the uptake of LDL from these coated regions into the cell. By arranging the electron micrographs into a logical order, they concluded that LDL binds to its receptor in coated regions of the plasma membrane, which become deeply invaginated and then are pinched off to form intracellular vesicles. These vesicles are then trafficked through the cell to fuse with lysosomes, where LDL is degraded. As a control, they also visualized the uptake of horseradish peroxidase in the same manner. This protein is not recognized by specific receptors on the plasma membrane and so does not become concentrated in coated pits but is still taken up by the cell to some degree. These findings and the use of cells from a patient with hypercholesterolemia, whose fibroblasts were unable to bind LDL, led Goldstein and his colleagues to postulate the model of receptor-mediated endocytosis in a prescient review 2 years later (Goldstein et al., 1979). This basic trafficking pathway is common knowledge for many of us today, but at the time it took tremendous conceptual insight and creativity to postulate it from a series of simple observations. Moreover, this model is still valid and includes the notion of high-affinity receptors, cargo selection, and recycling pathways. Strikingly, it also suggested the existence of additional, clathrin-independent endocytic pathways, which are increasingly recognized as having key physiological roles (Mayor et al., 2014). It has become the framework for a whole field of research and has inspired scientists throughout the past 40 years.
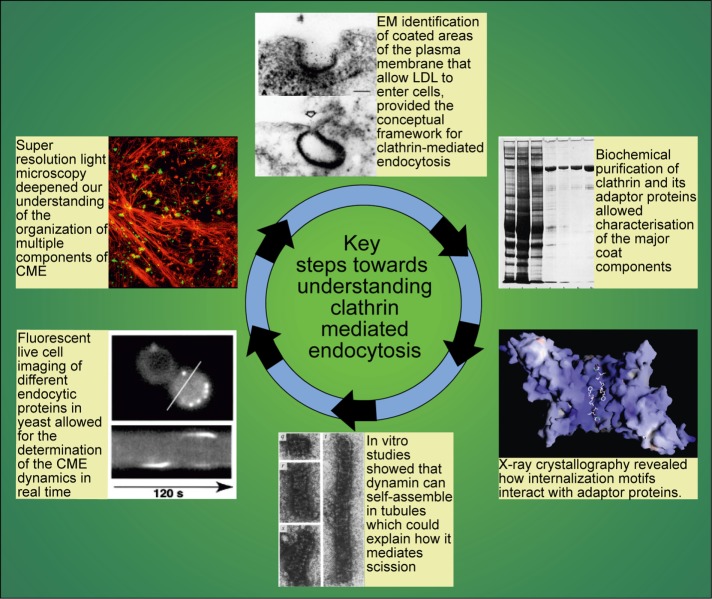
FIGURE 1:
Key steps in our understanding of the CCV cycle. Clockwise from top: original images showing LDL-ferritin being internalized through coated structures on the surface of normal fibroblasts (Anderson et al., 1977a); SDS–PAGE gel showing clathrin and adaptor proteins purified from pig brain (Pearse, 1975); x-ray crystallography studies revealing how tyrosine-based motifs interact with AP2 (Owen and Evans, 1998); in vitro studies showing that dynamin assembles into ring structures that suggest a mechanism by which dynamin might pinch off coated pits (Hinshaw and Schmid, 1995); studies in yeast pioneering live-cell imaging to understand dynamics of endocytic patch assembly (Kaksonen et al., 2003); structured illumination imaging allowing unprecedented resolution to visualize the relationship of clathrin-coated structures (green) to the actin cytoskeleton (red; Li et al., 2015).
KEY STEPS TO UNDERSTANDING THE MOLECULAR MACHINERY OF CCPs
The key protein component of these coated regions was purified by Pearse (1975), who named it clathrin (from the Latin clatratus, meaning “like a lattice”). This marked the beginning of the molecular era, and the key components of clathrin-mediated endocytosis (CME) were identified. Through biochemical purifications, “assembly” or “accessory” proteins (APs) were isolated (Zaremba and Keen, 1983; Pearse and Robinson, 1984; Ahle and Ungewickell, 1986), and by reconstituting clathrin-coated vesicles (CCVs) in vitro, it was shown that they connect the clathrin coat to the membrane, acting as adaptors (Vigers et al., 1986). Progress in molecular biology made it possible to express and purify modified versions of proteins, and after years of painstaking research and contradictory results, investigators were able to identify internalization signals in cargo molecules and how they are recognized by the APs. This led to the discovery of a simple tyrosine motif as the first internalization sequence that is recognized by the adaptor molecule AP2 (Traub and Bonifacino, 2013). Elegant structural studies revealed that when AP2 is recruited to the plasma membrane, it undergoes a conformational change to enable it to bind to cargo-sorting signals and clathrin (Kelly et al., 2014; reviewed in depth by Robinson, 2015). All of these discoveries are perfectly in line with the postulated model from Goldstein and colleagues, which predicted the existence of such a sorting motif after their study in 1977.
Despite these advances, the mechanism by which CCPs are pinched off from the membrane remained unresolved. The solution to this came from understanding why a temperature-sensitive Drosophila melanogaster mutant (called shibire) is paralyzed at high temperatures (Poodry et al., 1973). Electron microscopy studies of this mutant revealed that coated pits at the neuromuscular junction were stalled at a late stage of CME as deep invaginations in the plasma membrane that could not be pinched off. After identification and cloning of the responsible gene, a defect in the GTPase dynamin was shown to be responsible for this phenotype (Chen et al., 1991; Vanderbliek and Meyerowitz, 1991). It took additional years to discover that dynamin assembles into ring-like structures (Hinshaw and Schmid, 1995) around the neck of deeply invaginated coated pits (Takei et al., 1995), and indeed the precise mechanism by which dynamin causes the final scission of CCPs is still not fully resolved and remains a major research focus (Antonny et al., 2016). Having identified the key components of the fundamental model, from the concentration of cargo into CCPs by adaptor proteins, to their scission by the GTPase dynamin, more detailed analysis led to the discovery of many new factors that influence CME. This included many more adaptors, regulatory proteins, and components that sculpt the membrane, promoting and sensing increasing curvature as the pit buds inward to form a vesicle (Merrifield and Kaksonen, 2014).
A DYNAMIC PERSPECTIVE THROUGH LIVE-CELL IMAGING
The availability of fluorescent protein tags and the use of real-time fluorescence microscopy greatly affected how CME could be investigated. For the first time, the dynamics of the endocytic machinery and recruitment of several key components to growing CCPs could be visualized in real time. This meant that all the pieces of the model proposed by Goldstein and his colleagues could be put together to form a complete picture of CME in live cells. The ease of genetic manipulation of Saccharomyces cerevisiae made this model system extremely amenable to this new technology. Even though the process of endocytosis in yeast initially seemed to differ in some aspects from that of mammalian cells, the insights gained through these studies had a significant effect on understanding fundamental aspects of CME. In mammalian cells as well as in yeast, the sequential assembly and disassembly of protein modules during the early stages of endocytosis holds true and is similar in both systems (Goode et al., 2015). By fluorescently labeling multiple key components of the endocytic pathway, kymographs of single endocytic patches could be assembled to visualize coat invaginations in real time for the first time (Kaksonen et al., 2003). Through these early studies, the lifetime dynamics of single endocytic events were revealed and the dynamics and contribution of multiple endocytic proteins could be investigated. In particular, these approaches highlighted the importance of actin polymerization and filament organization to driving membrane invagination when the plasma membrane is under pressure (Aghamohammadzadeh and Ayscough, 2009). In mammalian cells, although the role of actin is not fully resolved, a critical need for actin when the plasma membrane is under tension has also been demonstrated (Boulant et al., 2011).
For the investigation of CME in mammalian cells, the development of total internal reflection fluorescence (TIRF) microscopy proved to be an extremely valuable tool, which allows the basal membrane of live cells to be imaged with a very low signal-to-noise ratio while keeping a high temporal resolution. This method was pioneered by Merrifield and his colleagues, who were able to detect single endocytic events and confirm that the GTPase dynamin is recruited to CCPs, peaking at the end of their lifetime and mediating their scission (Merrifield et al., 2002). Shortly thereafter, many different investigators used this experimental approach to precisely dissect the lifetime dynamics of CCPs from their nucleation at the membrane to their uncoating in the cytoplasm (Ehrlich et al., 2004; Perrais and Merrifield, 2005, Cocucci et al., 2012; Aguet et al., 2013). This allowed further refinement of the model proposed by Goldstein and colleagues and demonstrated the recruitment of different components to the growing clathrin lattice at different stages during its lifetime (Taylor et al., 2011), thus helping to elucidate their mechanisms of action. It also became apparent that different cargo molecules can regulate CME through posttranslational modifications of the endocytic machinery for their efficient uptake (Ferreira et al., 2012). This added an extra layer of intricacy to the system and resulted in the current situation in which the internalization of particular cargo molecules is sufficiently complex to warrant its own review.
FUTURE DIRECTIONS
After the use of electron microscopy to determine the morphology of CCPs and live-cell fluorescence imaging to dissect their dynamic behavior, the development of superresolution light microscopy enabled scientists to visualize CME in unforeseen detail. The resolution of conventional light microscopy is limited to ∼200 nm in the horizontal and ∼400 nm in the vertical direction. However, with a diameter of ∼150 nm, CCPs are just below this resolution limit and have been a useful benchmark for the application of many novel superresolution methods, with each technique in turn being immensely useful to investigate new details of CME (Huang et al., 2008; Miller et al., 2015). This development has been very valuable for gaining more detailed insight into the organization of different components of the endocytic machinery and has been used to investigate the interplay of clathrin with the actin cytoskeleton in beautiful detail (Li et al., 2015). Although superresolution microscopy is a powerful tool to investigate CME, the conventional methods used by Goldstein and his colleagues are still being used. One approach combining visualization of the morphology of CCPs by electron microscopy with the detection of coat components by fluorescent markers is correlative light and electron microscopy (CLEM). This method enables researchers to correlate the morphology of CCPs with the presence of different coat components and has been used with great effect to investigate the transition of flat clathrin lattices into spherical CCVs (Avinoam et al., 2015). This shows that even 40 years after the article by Goldstein and colleagues, the methods that they used are still immensely valuable. With the use of biochemical purification, x-ray crystallography, live-cell fluorescence imaging, and superresolution microscopy researchers are able investigate almost every element of CME.
Notwithstanding the great advances that have been made in recent years, many challenging questions about CCV assembly and disassembly remain unanswered. One of these challenges is to understand precisely how the membrane is bent and curvature is generated to deform a flat membrane into a CCV. The delicate design and complex geometry of these vesicles has fascinated researchers since the first electron microscopy images were published. It was soon realized that the conversion of hexagonal arrangements into pentagons is essential for the transition of a flat clathrin lattice into a spherical vesicle (Kanaseki and Kadota, 1969). However, how this rearrangement takes place is still unclear, and understanding the generation of curvature during CME has proven to be a complex challenge. Two models have been proposed that aim to explain this process. In the model of “constant curvature,” the CCP grows through the continuous polymerization of coat components and steadily increases in size with constant curvature, whereas in the “constant-area” model, the CCP starts as flat lattice that is continuously deformed into a vesicle (Lampe et al. 2016). In vitro, the stepwise polymerization of clathrin is sufficient to drive curvature in reconstituted membranes into vesicles (Dannhauser and Ungewickell, 2012; Kirchhausen, 2012), whereas in vivo, the clathrin lattice seems to start as a flat coat that is continuously deformed into a CCV (Avinoam et al., 2015). Among the main differences between these systems is that the membrane composition of live cells differs in some aspects to that of in vitro model membranes and many components of the membrane cannot be effectively modeled in these systems. It is therefore possible that the different properties of the plasma membrane and the presence of different cargo molecules could influence the way it can be deformed. Answering these challenging questions will greatly improve our understanding of CME and will require the collaborative effort of biologists, physicists, and mathematicians.
IMPORTANCE OF CME IN DISEASE
Brown and Goldstein were motivated to understand the reasons why their patients suffered from hypercholesterolemia. The use of patient fibroblasts suggested the importance of high-affinity receptors (Anderson et al., 1977a). Some patients with this disorder have a mutation in the cargo recognition sequence of the LDL receptor such that the protein is unable to bind to adaptor proteins and undergo endocytosis. This inability to take up LDL leads to an excessive amount of cholesterol in the bloodstream, which can cause coronary heart disease (Anderson et al., 1977b; Marks et al., 2003). It is therefore no surprise that the regulation of CME became crucial in understanding and battling some major healthcare challenges.
One of many examples of the importance of CME is in the development of Alzheimer’s disease (AD; Nordstedt et al., 1993; Cossec et al., 2010). Differential processing of the amyloid precursor protein (APP) is crucial for the development of AD and the disposition of amyloid plaques that are found in the brains of AD patients. The subcellular localization of APP is an important factor for its processing and determines whether it is cleaved into its amyloidogenic (pathogenic) or nonamyloidogenic (nonpathogenic) product. Whereas amyloidogenic processing takes place after uptake through CME, the nonamyloidogenic cleavage occurs at the plasma membrane (Haass et al., 2012). Inhibition of CME shifts the processing of APP toward the nonamyloidogenic pathway, and endocytic factors have been identified as risk factors for development of AD (Harold et al., 2009; Kanatsu et al., 2014). In the future, it will be interesting to see whether targeting endocytic uptake of APP could be a promising way to interfere with the development of AD (Schreiber et al., 2012). In addition, CME is involved in many other diseases, such as cancer (Mellman and Yarden, 2013), viral uptake, and bacterial infections (Cossart and Helenius, 2014). This makes it clear that the model of receptor-mediated endocytosis proposed by Goldstein and his colleagues is still at the core of developments that have the potential to affect the lives of many people.
CONCLUDING REMARKS
Anderson, Brown, and Goldstein observed the uptake of LDL though coated regions on the plasma membrane into the cytoplasm. From these simple observations, they were able to postulate a model of receptor-mediated endocytosis that has provided the conceptual framework for this pathway for researchers ever since. Understanding the pathway is of interest not only to those involved in elucidating fundamental endocytic processes but also to many other researchers. Owing to its central role in cell biology, CME is intimately connected to key advances in fields such as neuroscience, cell signaling, and immunology, as well as for mechanisms of development and adult homeostasis. Gaining a better understanding of it will benefit all of these fields. A challenge for the future is to understand how this key cellular pathway is regulated in cell- and tissue-specific contexts. Tremendous progress has already been made, and, with the development of new methods to investigate CME, the field is sure to progress even further. However, the findings from Goldstein and his colleagues will always be the foundations upon which this progress is being made. Therefore, when we feel overwhelmed by the ever-increasing complexity of the field, it is good to remind ourselves that its foundation is the clever interpretation of simple observations made >40 years ago.
Acknowledgments
We apologize to colleagues whose work we were unable to cite due to the concise nature of this short review. H.M. is supported by the Biopol EU ITN network.
Abbreviations used:
- AD
Alzheimer’s disease
- AP
adaptor protein
- CCP
clathrin-coated pit
- CME
clathrin-mediated endocytosis
- LDL
low density lipoprotein
- TIRF
total internal reflection fluorescence.
Footnotes
REFERENCES
- Aghamohammadzadeh S, Ayscough KR. Differential requirements for actin during yeast and mammalian endocytosis. Nat Cell Biol. 2009;11:1039–1042. doi: 10.1038/ncb1918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguet F, Antonescu CN, Mettlen M, Schmid SL, Danuser G. Advances in analysis of low signal-to-noise images link dynamin and ap2 to the functions of an endocytic checkpoint. Dev Cell. 2013;26:279–291. doi: 10.1016/j.devcel.2013.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahle S, Ungewickell E. Purification and properties of a new clathrin assembly protein. EMBO J. 1986;5:3143–3149. doi: 10.1002/j.1460-2075.1986.tb04621.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson RGW, Brown MS, Goldstein JL. Role of the coated endocytic vesicle in the uptake of receptor-bound low density lipoprotein in human fibroblasts. Cell. 1977a;10:351–364. doi: 10.1016/0092-8674(77)90022-8. [DOI] [PubMed] [Google Scholar]
- Anderson RGW, Goldstein JL, Brown MS. Mutation that impairs ability of lipoprotein receptors to localize in coated pits on cell-surface of human fibroblasts. Nature. 1977b;270:695–699. doi: 10.1038/270695a0. [DOI] [PubMed] [Google Scholar]
- Antonny B, Burd C, De Camilli P, Chen E, Daumke O, Faelber K, Ford M, Frolov VA, Frost A, Hinshaw JE, et al. Membrane fission by dynamin: What we know and what we need to know. EMBO J. 2016;35:2270–2284. doi: 10.15252/embj.201694613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avinoam O, Schorb M, Beese CJ, Briggs JAG, Kaksonen M. Endocytic sites mature by continuous bending and remodeling of the clathrin coat. Science. 2015;348:1369–1372. doi: 10.1126/science.aaa9555. [DOI] [PubMed] [Google Scholar]
- Boulant S, Kural C, Zeeh JC, Ubelmann F, Kirchhausen T. Actin dynamics counteract membrane tension during clathrin-mediated endocytosis. Nat Cell Biol. 2011;13:U1124–U1158. doi: 10.1038/ncb2307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen MS, Obar RA, Schroeder CC, Austin TW, Poodry CA, Wadsworth SC, Vallee RB. Multiple forms of dynamin are encoded by shibire, a drosophila gene involved in endocytosis. Nature. 1991;351:583–586. doi: 10.1038/351583a0. [DOI] [PubMed] [Google Scholar]
- Cocucci E, Aguet F, Boulant S, Kirchhausen T. The first five seconds in the life of a clathrin-coated pit. Cell. 2012;150:495–507. doi: 10.1016/j.cell.2012.05.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cossart P, Helenius A. Endocytosis of viruses and bacteria. Cold Spring Harb Perspect Biol. 2014;6:a016972. doi: 10.1101/cshperspect.a016972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cossec JC, Simon A, Marquer C, Moldrich RX, Leterrier C, Rossier J, Duyckaerts C, Lenkei Z, Potier MC. Clathrin-dependent APP endocytosis and A beta secretion are highly sensitive to the level of plasma membrane cholesterol. Biochim Biophys Acta. 2010;1801:846–852. doi: 10.1016/j.bbalip.2010.05.010. [DOI] [PubMed] [Google Scholar]
- Dannhauser PN, Ungewickell EJ. Reconstitution of clathrin-coated bud and vesicle formation with minimal components. Nat Cell Biol. 2012;14:634–639. doi: 10.1038/ncb2478. [DOI] [PubMed] [Google Scholar]
- Ehrlich M, Boll W, van Oijen A, Hariharan R, Chandran K, Nibert ML, Kirchhausen T. Endocytosis by random initiation and stabilization of clathrin-coated pits. Cell. 2004;118:591–605. doi: 10.1016/j.cell.2004.08.017. [DOI] [PubMed] [Google Scholar]
- Ferreira F, Foley M, Cooke A, Cunningham M, Smith G, Woolley R, Henderson G, Kelly E, Mundell S, Smythe E. Endocytosis of G protein-coupled receptors is regulated by clathrin light chain phosphorylation. Curr Biol. 2012;22:1361–1370. doi: 10.1016/j.cub.2012.05.034. [DOI] [PubMed] [Google Scholar]
- Goldstein JL, Anderson RGW, Brown MS. Coated pits, coated vesicles, and receptor-mediated endocytosis. Nature. 1979;279:679–685. doi: 10.1038/279679a0. [DOI] [PubMed] [Google Scholar]
- Goode BL, Eskin JA, Wendland B. Actin and endocytosis in budding yeast. Genetics. 2015;199:315–358. doi: 10.1534/genetics.112.145540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haass C, Kaether C, Thinakaran G, Sisodia S. Trafficking and proteolytic processing of APP. Cold Spring Harb Perspect Med. 2012;2:a006270. doi: 10.1101/cshperspect.a006270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harold D, Abraham R, Hollingworth P, Sims R, Gerrish A, Hamshere ML, Pahwa JS, Moskvina V, Dowzell K, Williams A, et al. Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer’s disease. Nat Genet. 2009;41:1088–1093. doi: 10.1038/ng.440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinshaw JE, Schmid SL. Dynamin self-assembles into rings suggesting a mechanism for coated vesicle budding. Nature. 1995;374:190–192. doi: 10.1038/374190a0. [DOI] [PubMed] [Google Scholar]
- Huang B, Wang WQ, Bates M, Zhuang XW. Three-dimensional super-resolution imaging by stochastic optical reconstruction microscopy. Science. 2008;319:810–813. doi: 10.1126/science.1153529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaksonen M, Sun Y, Drubin DG. A pathway for association of receptors, adaptors, and actin during endocytic internalization. Cell. 2003;115:475–487. doi: 10.1016/s0092-8674(03)00883-3. [DOI] [PubMed] [Google Scholar]
- Kanaseki T, Kadota K. Vesicle in a basket—a morphological study of coated vesicle isolated from nerve endings of guinea pig brain, with special reference to mechanism of membrane movements. J Cell Biol. 1969;42:202–220. doi: 10.1083/jcb.42.1.202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanatsu K, Morohashi Y, Suzuki M, Kuroda H, Watanabe T, Tomita T, Iwatsubo T. Decreased CALM expression reduces A beta 42 to total A beta ratio through clathrin-mediated endocytosis of gamma-secretase. Nat Commun. 2014;5:3386. doi: 10.1038/ncomms4386. [DOI] [PubMed] [Google Scholar]
- Kelly BT, Graham SC, Liska N, Dannhauser PN, Honing S, Ungewickell EJ, Owen DJ. AP2 controls clathrin polymerization with a membrane-activated switch. Science. 2014;345:459–463. doi: 10.1126/science.1254836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirchhausen T. Bending membranes. Nat Cell Biol. 2012;14:906–908. doi: 10.1038/ncb2570. [DOI] [PubMed] [Google Scholar]
- Lampe M, Vassilopoulos S, Merrifield C. Clathrin coated pits, plaques and adhesion. J Struct Biol. 2016;196:48–56. doi: 10.1016/j.jsb.2016.07.009. [DOI] [PubMed] [Google Scholar]
- Li D, Shao L, Chen BC, Zhang X, Zhang MS, Moses B, Milkie DE, Beach JR, Hammer JA, Pasham M, et al. Extended-resolution structured illumination imaging of endocytic and cytoskeletal dynamics. Science. 2015;349:aab3500. doi: 10.1126/science.aab3500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marks D, Thorogood M, Neil HAW, Humphries SE. A review on the diagnosis, natural history, and treatment of familial hypercholesterolaemia. Atherosclerosis. 2003;168:1–14. doi: 10.1016/s0021-9150(02)00330-1. [DOI] [PubMed] [Google Scholar]
- Mayor S, Parton RG, Donaldson JG. Clathrin-independent pathways of endocytosis. Cold Spring Harb Perspect Biol. 2014;6:a016758. doi: 10.1101/cshperspect.a016758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellman I, Yarden Y. Endocytosis and cancer. Cold Spring Harb Perspect Biol. 2013;5:a016949. doi: 10.1101/cshperspect.a016949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrifield CJ, Feldman ME, Wan L, Almers W. Imaging actin and dynamin recruitment during invagination of single clathrin-coated pits. Nat Cell Biol. 2002;4:691–698. doi: 10.1038/ncb837. [DOI] [PubMed] [Google Scholar]
- Merrifield CJ, Kaksonen M. Endocytic accessory factors and regulation of clathrin-mediated endocytosis. Cold Spring Harb Perspect Biol. 2014;6:a016733. doi: 10.1101/cshperspect.a016733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller SE, Mathiasen S, Bright NA, Pierre F, Kelly BT, Kladt N, Schauss A, Merrifield CJ, Stamou D, Honing S, Owen DJ. CALM regulates clathrin-coated vesicle size and maturation by directly sensing and driving membrane curvature. Dev Cell. 2015;33:163–175. doi: 10.1016/j.devcel.2015.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nordstedt C, Caporaso GL, Thyberg J, Gandy SE, Greengard P. Identification of the alzheimer beta/A4 amyloid precursor protein in clathrin-coated vesicles purified from Pc12 cells. J Biol Chem. 1993;268:608–612. [PubMed] [Google Scholar]
- Owen DJ, Evans PR. A structural explanation for the recognition of tyrosine-based endocytotic signals. Science. 1998;282:1327–1332. doi: 10.1126/science.282.5392.1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearse BMF. Coated vesicles from pig brain-purification and biochemical characterization. J Mol Biol. 1975;97:93–98. doi: 10.1016/s0022-2836(75)80024-6. [DOI] [PubMed] [Google Scholar]
- Pearse BMF, Robinson MS. Purification and properties of 100-Kd proteins from coated vesicles and their reconstitution with clathrin. EMBO J. 1984;3:1951–1957. doi: 10.1002/j.1460-2075.1984.tb02075.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perrais D, Merrifield CJ. Dynamics of endocytic vesicle creation. Dev Cell. 2005;9:581–592. doi: 10.1016/j.devcel.2005.10.002. [DOI] [PubMed] [Google Scholar]
- Poodry CA, Hall L, Suzuki DT. Temperature-sensitive mutations in drosophila-melanogaster 14. Developmental properties of shibire—pleiotropic mutation affecting larval and adult locomotion and development. Dev Biol. 1973;32:373–386. doi: 10.1016/0012-1606(73)90248-0. [DOI] [PubMed] [Google Scholar]
- Robinson MS. Forty years of clathrin-coated vesicles. Traffic. 2015;16:1210–1238. doi: 10.1111/tra.12335. [DOI] [PubMed] [Google Scholar]
- Roth TF, Porter RF. Yolk protein uptake in the oocyte of the mosquito Aedes aegypti. L. J Cell Biol. 1964;20:313–332. doi: 10.1083/jcb.20.2.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber A, Fischer S, Lang T. The amyloid precursor protein forms plasmalemmal clusters via its pathogenic amyloid-beta domain. Biophys J. 2012;102:1411–1417. doi: 10.1016/j.bpj.2012.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takei K, Mcpherson PS, Schmid SL, Decamilli P. Tubular membrane invaginations coated by dynamin rings are induced by Gtp-gamma-S in nerve-terminals. Nature. 1995;374:186–190. doi: 10.1038/374186a0. [DOI] [PubMed] [Google Scholar]
- Taylor MJ, Perrais D, Merrifield CJ. A high precision survey of the molecular dynamics of mammalian clathrin-mediated endocytosis. PLoS Biol. 2011;9:e1000604. doi: 10.1371/journal.pbio.1000604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traub LM, Bonifacino JS. Cargo recognition in clathrin-mediated endocytosis. Cold Spring Harb Perspect Biol. 2013;5:a016790. doi: 10.1101/cshperspect.a016790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanderbliek AM, Meyerowitz EM. Dynamin-Like protein encoded by the drosophila-shibire gene associated with vesicular traffic. Nature. 1991;351:411–414. doi: 10.1038/351411a0. [DOI] [PubMed] [Google Scholar]
- Vigers GPA, Crowther RA, Pearse BMF. Location of the 100 Kd-50 Kd accessory proteins in clathrin coats. EMBO J. 1986;5:2079–2085. doi: 10.1002/j.1460-2075.1986.tb04469.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaremba S, Keen JH. Assembly polypeptides from coated vesicles mediate reassembly of unique clathrin coats. J Cell Biol. 1983;97:1339–1347. doi: 10.1083/jcb.97.5.1339. [DOI] [PMC free article] [PubMed] [Google Scholar]