Abstract
Background
After a low anterior resection, creating a defunctioning stoma is vital for securing the anastomosis in low-lying rectal cancer patients receiving concurrent chemoradiotherapy. Although it decreases the complication and reoperation rates associated with anastomotic leakage, the complications that arise before and after stoma closure should be carefully evaluated and managed.
Methods
This study enrolled 95 rectal cancer patients who received neoadjuvant concurrent chemoradiotherapy and low anterior resection with anastomosis of the bowel between July 2010 and November 2012. A defunctioning stoma was created in 63 patients during low anterior resection and in another three patients after anastomotic leakage.
Results
The total complication rate from stoma creation to closure was 36.4%. Ileostomy led to greater renal insufficiency than colostomy did and significantly increased the readmission rate (all p < 0.05). The complication rate related to stoma closure was 36.0%. Patients with ileostomy had an increased risk of developing complications (p = 0.017), and early closure of the defunctioning stoma yielded a higher incidence of morbidity (p = 0.006). Multivariate analysis revealed that a time to closure of ≤109 days was an independent risk factor for developing complications (p = 0.007).
Conclusions
The optimal timing of stoma reversal is at least 109 days after stoma construction in rectal cancer patients receiving concurrent chemoradiotherapy and low anterior resection.
Keywords: Rectal cancer, Concurrent chemoradiotherapy, Defunctioning ileostomy, Defunctioning colostomy, Low anterior resection, Early closure of a stoma
Background
Fecal diversion is a common technique for managing various surgical situations including congenital diseases, acute or chronic inflammation, acute or chronic colonic obstruction, and malignancy. Two common procedures for fecal diversion are loop transverse colostomy and loop ileostomy. Temporary defunctioning stomas are often created to protect the distal anastomosis in cases where anastomotic leakage may occur. In low-lying rectal cancer patients receiving concurrent chemoradiotherapy (CCRT), the anastomosis can be protected by creating a defunctioning stoma after a low anterior resection (LAR) or coloanal anastomosis (CAA) [1]. However, the complications associated with stool diversion and the morbidity after stoma closure, which may be as high as 47.6 and 34% in reviewed literatures [2, 3], should be considered when constructing a defunctioning stoma. Although the creation of a proper loop transverse colostomy is technically challenging when the splenic flexure is mobilized to allow for a tension-free colorectal anastomosis, opinions vary regarding the choice of ileostomy or colostomy for diverging the stool stream from the perspective of complications before and after stoma reversal. Furthermore, the optimal timing of defunctioning stoma closure ranges widely and is not yet clearly defined in literatures.
Methods
In our study, data from a single medical center were retrospectively retrieved. This study enrolled 103 patients with rectal cancer with or without distant metastasis who received neoadjuvant CCRT between July 2010 and November 2012. The median distance between the lower margin of the tumor and the anal verge was 4.1 cm (SD = 2.2 cm). Locally advanced disease, metastatic disease at the time of diagnosis, and ultra-low lying early rectal cancer within 5 cm of the anal verge accounted for 55.8, 15.8, and 37.2% of all patients, respectively. In most patients, radiation was through 6- and 10-MV photons by using a three-field technique (posterior and both laterals), as described previously [4]. Pelvic radiation therapy was delivered at a dose of 45 Gy in 25 fractions over 5 weeks and was followed by a boost dose of 5.4 Gy, which was administered to the primary tumor. The CCRT chemotherapy regimens applied in our patients mainly included oral capecitabine (61.7%), oral tegafur (8.5%), FOLFOX4 (13.8%), and FOLFIRI plus bevacizumab (11.7%) for patients with distant metastasis. Patients underwent surgery at 6–10 weeks after completing CCRT. Thirty-three (34.7%) patients received laparoscopic surgery, which was considered controversial for managing low-lying rectal cancer at the time of data collection [5, 6]. The total mesorectal excision (TME) technique was performed for all patients, and extended visceral resection was performed for patients with clinical T4 cancer. Anal sphincter-sparing surgery was performed when possible. Of the 103 patients, 95 received colorectal anastomosis or CAA (Fig. 1). Seven patients who received abdominoperineal resection and one who received local excision were excluded. A temporary defunctioning stoma was intraoperatively created at the discretion of the surgeon. In 63 (66.3%) patients, a defunctioning stoma was created during LAR for low-lying rectal cancer. The mean distance from distal margin of the cancer to the anal verge was 3.6 cm (SD = 1.8) in patients with a stoma and 4.9 cm (SD = 2.6) in patients without one (p = 0.022). Among patients without intraoperative defunctioning stoma creation, three received subsequent transverse colostomy for anastomotic leakage. Finally, 66 patients with stomata were analyzed. These patients were regularly followed up in the outpatient department to monitor the constructional and functional outcome of the stoma during the defunctioning period. An antidiarrheal agent and electrolyte solution was not routinely used except in patients with profound stoma output and symptomatic dehydration. The closure of each type of temporary stoma involved the standard procedure including peristomal incision and excision of both limbs, with a hand-sewn end-to-end anastomosis using continuous locking suture and Lembert suture techniques. All operations were performed by the same surgical team. The subcutaneous placement of a drain tube and intravenous administration of prophylactic antibiotics were performed in all patients to prevent the occurrence of surgical site infections (SSIs). In total, 25 of 28 ileostomies and 25 of 38 transverse colostomies were closed after the distal anastomosis was secured and adjuvant chemotherapy (if necessary) completed. The median time from stoma construction to closure was 129 (range, 58–326) days. All demographic data were recorded, and clinical courses before and after defunctioning stoma reversal were compared.
Fig. 1.
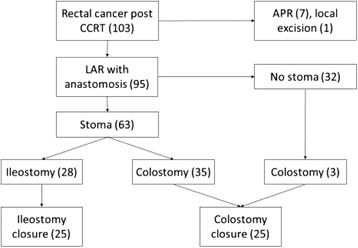
Flowchart of data collection
Statistical analysis
Qualitative variables are expressed as numbers and percentages and the quantitative variables as the median and SD. Comparisons between groups were performed using the χ2 test for categorical variables and Mann–Whitney U test for quantitative variables. The α error was set at 0.05. Logistic regression analysis was performed to identify risk factors for developing complications, and unadjusted odds ratios [and 95% confidence intervals (CIs)] were calculated for morbidity. The receiver operating curve (ROC) was used to determine the timing (days) of stoma reversal that optimally predicted the development of complications after stoma closure. Statistical analysis was performed using JMP 12.1 software (SAS Institute, Cary, North Carolina) for Windows. A p value of <0.05 was considered statistically significant.
Results
The demographic data of the patient populations who received loop ileostomies or colostomies were compared (Table 1). The total complication rate from stoma creation to closure was 36.4% (24/66 patients). Electrolyte imbalance, which occurred in 30.3% (20/66) of patients, was the leading cause of complications, followed by renal insufficiency in 21.2% (14/66 patients). Hypokalemia and hyponatremia accounted for most of the electrolyte imbalance cases. The total complication rate was significantly higher in patients with ileostomy (50.0%) than in those with colostomy (26.3%) (p = 0.048; Table 2). In addition, colostomy yielded a lower readmission rate (5.4%) than ileostomy did (35.7%; p = 0.002). Moreover, renal insufficiency developed more often before ileostomy closure than before colostomy closure (42.9 vs. 5.3%; p < 0.001). The development of electrolyte imbalance was borderline higher in patients with ileostomy than with colostomy (42.9 vs. 21.1%; p = 0.057). Stoma prolapse was noted in three of the 28 ileostomy cases, and no stoma prolapse was noted in any of the 38 colostomy cases (p = 0.021).
Table 1.
Demographic data of the loop colostomy and loop ileostomy groups
| Variables | Colostomy | Ileostomy | Total | p value |
|---|---|---|---|---|
| Number (%) | 38 (57.6) | 28 (42.4) | 66 | – |
| Age, years (SD) | 63.1 (13.1) | 63.8 (12.0) | 62.9 (12.1) | 0.834 |
| Sex (%) | 0.408 | |||
| Male/female | 22/16 (57.9/42.1) | 19/9 (67.9/32.1) | 41/25 (62.1/37.9) | |
| BMI (SD) | 23.44 (4.26) | 23.62 (2.93) | 23.34 (3.33) | 0.887 |
| Histology (%) | 0.650 | |||
| WD/MD/PD | 7/25/3 (20.0/71.4/8.6) | 3/21/22 (11.5/80.8/7.7) | 10/46/5 (16.4/75.4/8.2) | |
| ypT (%) | 0.811 | |||
| T0/T1/T2/T3/T4 | 6/3/10/18/1 (15.8/7.9/26.3/47.4/2.6) | 6/3/9/9/1 (21.4/10.7/32.1/32.1/3.6) | 12/6/19/27/2 (18.2/9.1/28.8/40.9/3.0) | |
| ypN (%) | 0.270 | |||
| N0/N1/N2 | 23/7/8 (60.5/18.4/21.1) | 20/6/2 (71.4/21.4/7.1) | 43/13/10 (65.2/19.7/15.2) | |
| Metastatic disease (%) | 0.381 | |||
| M0/M1 | 31/7 (81.6/18.4) | 25/3 (89.3/10.7) | 56/10 (84.9/15.2) | |
| Emergent operation (%) | 6 (15.8) | 1 (3.6) | 7 (10.6) | 0.091 |
| Locally advanced disease (%) | 21 (55.3) | 12 (42.9) | 33(50.0) | 0.319 |
| Distance from anal verge, cm (SD) | 3.41 (1.75) | 3.93 (1.86) | 4.06 (2.20) | 0.259 |
| CEA, ng/mL (SD) | 6.35 (10.04) | 3.16 (2.33) | 4.94 (7.32) | 0.087 |
BMI body mass index, WD well differentiated, MD moderately differentiated, PD poorly differentiated, CEA carcinoembryonic antigen
Table 2.
Complications before defunctioning stoma closure
| Variables | Colostomy (%) | Ileostomy (%) | Total (%) | p value |
|---|---|---|---|---|
| Number | 38 (57.6) | 28 (42.4) | 66 | – |
| Complications | 10 (26.3) | 14 (50.0) | 24 (36.4) | 0.048* |
| Dermatitis | 1 (2.6) | 1 (3.6) | 2 (3.0) | 0.827 |
| Renal insufficiency | 2 (5.3) | 12 (42.9) | 14 (21.2) | <0.001* |
| Electrolyte imbalance | 8 (21.1) | 12 (42.9) | 20 (30.3) | 0.057 |
| Hypernatremia | 0 | 1 (3.6) | 1 (1.5) | 0.304 |
| Hyponatremia | 3 (7.9) | 6 (21.4) | 9 (13.6) | 0.581 |
| Hyperkalemia | 0 | 3 (10.7) | 3 (4.5) | 0.065 |
| Hypokalemia | 6 (15.8) | 7 (25.0) | 13 (19.7) | 0.439 |
| Stoma retraction | 0 | 1 (3.6) | 1 (1.5) | 0.188 |
| Stoma prolapse | 0 | 3 (10.7) | 3 (4.6) | 0.021* |
| Stoma stenosis | 0 | 0 | 0 | – |
| Parastoma hernia | 0 | 0 | 0 | – |
| Ileus | 1 (2.6) | 2 (7.1) | 3 (4.6) | 0.387 |
| Readmission | 2 (5.4) | 10 (35.7) | 12 (18.5) | 0.002* |
| Not reversal | 13 (34.2) | 3 (10.7) | 16( 24.2) | 0.061 |
*p < 0.05
Among 50 patients with defunctioning stoma closure, the median time of stool diversion was 129 days (range, 58–326 days). Patients with colostomy waited longer than patients with ileostomy did (median, 173 vs. 115 days). The median defecation time was 2.7 and 3.1 days after ileostomy and colostomy reversal, respectively (p = 0.41). No differences were observed in the median hospital stay between patients with colostomy and those with ileostomy (10.5 vs. 10.9 days; p = 0.76).
During the postoperative period after stoma closure, no mortality was recorded. The total complication rate related to stoma reversal was 36.0% (18/50 patients; Table 3). Overall complications developed more often in patients with ileostomy than in those with colostomy (52.0 vs. 20.0%; p = 0.017). Thirteen of the 25 (52%) patients with prior defunctioning ileostomy developed unfavorable events including prolonged ileus (n = 8) and superficial/deep SSI (n = 6). Most patients who suffered from prolonged ileus experienced relief after conservative treatment, and half of the patients with SSI received a subsequent reoperation and open drainage as proper management of a deep incisional infection. Among patients with colostomy, five (20.0%) developed complications after colostomy closure, including two cases of prolonged ileus, two of deep SSI, and one of anastomotic insufficiency.
Table 3.
Complication and reoperation rates after defunctioning stoma closure
| Variables | Colostomy (%) | Ileostomy (%) | Total (%) | p value |
|---|---|---|---|---|
| Number | 25 (50) | 25 (50) | 50 | – |
| Mortality | 0 | 0 | 0 | – |
| Complications | 5 (20.0) | 13 (52.0) | 18 (36.0) | 0.017* |
| Ileus | 2 (8.0) | 8 (32.0) | 10 (20.0) | 0.029* |
| Anastomotic insufficiency | 1 (4.0) | 1 (4.0) | 2 (4.0) | 1.000 |
| Wound infection | 2 (8.0) | 6 (24.0) | 8 (16.0) | 0.116 |
| Incisional hernia | 0 | 0 | 0 | – |
| Primary anastomotic site leakage | 0 | 1 (4.0) | 1 (2.0) | 0.236 |
| Re-operation | 4 (16.0) | 5 (20.0) | 9 (18.0) | 0.210 |
*p < 0.05
We identified independent risk factors for complications before stoma closure. Age, sex, body mass index (BMI), locally advanced disease, and metastatic disease all had minor effects on the complication rate. The percentage of ileostomy patients with complications during stool stream diversion was significantly higher than that of patients without complications (50 vs. 26.3%; p = 0.048; Table 4). Adjuvant chemotherapy for high-risk patients and patients with lymph node metastasis did not alter the incidence of complications (p = 0.454). A longer waiting time before stoma closure did not increase the complication rate during the stool diversion period (p = 0.406). Multivariate analysis revealed that the odds ratio for the development of complications was 3.71 times higher in patients with ileostomy than in those with colostomy, and it was the single independent risk factor for developing complications before stoma closure (OR 3.71; 95% CI 1.24–11.94; p = 0.019).
Table 4.
Univariate and multivariate analysis of the risk factors for developing complications before defunctioning stoma closure
| Variables | With complications (n = 24) (%) |
Without complications (n = 42) (%) |
Univariate analysis p value | Logistic multivariate regression analysis OR (95% CI), p value |
|---|---|---|---|---|
| Age (SD) | 65.5 (13.5) | 62.2 (12.0) | 0.319 | – |
| Male (%) | 13 (54.2) | 28 (66.7) | 0.316 | – |
| BMI | 22.56 (3.70) | 24.21 (3.30) | 0.175 | – |
| Metastatic disease (%) | 4 (16.7) | 6 (14.3) | 0.800 | – |
| Locally advanced disease (%) | 13 (54.2) | 20 (47.6) | 0.609 | – |
| Distance from anal verge, cm (SD) | 3.85 (1.85) | 3.51 (1.78) | 0.469 | – |
| Emergent operation (%) | 3 (12.5) | 4 (9.5) | 0.708 | – |
| Stoma (%) | 0.048* | |||
| Colostomy | 10 (41.7) | 28 (66.7) | ||
| Ileostomy | 14 (58.3) | 14 (33.3) | 3.71 (1.24~11.94), 0.019* | |
| Time to reversal, days (SD) | 141.8 (67.1) | 158.5 (66.0) | – | – |
| Adjuvant chemotherapy (%) | 12 (50.0) | 17 (40.5) | 0.454 |
BMI body mass index
*p < 0.05
Furthermore, risk factors for reversal-related morbidity were also analyzed. Age, sex, BMI, locally advanced disease, and metastatic disease did not alter the complication rate during the period following stoma closure. Similarly, complications developed more frequently in patients with ileostomy reversal. After defunctioning stoma closure, patients with ileostomy developed more complications than those with colostomy did (52.0% of ileostomy cases and 20.0% of colostomy cases; p = 0.017; Table 5). Adjuvant chemotherapy seemed to have no influence on the incidence of reversal-related morbidity. Moreover, a nonsignificant correlation was observed between complications before stoma closure and those after stoma closure (p = 0.483). Notably, the postoperative complication rate was significantly higher when the time to reversal was shorter. An average of 120.9 and 169.1 days passed before defunctioning stoma closure in patients with complications compared with in those without complications (p = 0.006).
Table 5.
Univariate and multivariate analysis of the risk factors for developing complications after defunctioning stoma closure
| Variables | With complications (n = 18) (%) |
Without complications (n = 32) (%) |
Univariate analysis p value | Logistic multivariate regression analysis OR (95% CI), p value |
|---|---|---|---|---|
| Age (SD) | 66.1 (14.0) | 63.5 (11.1) | 0.511 | – |
| Male (%) | 12 (66.7) | 18 (56.3) | 0.468 | – |
| Stoma (%) | 0.017* | |||
| Colostomy | 5 (20.0) | 20 (80.0) | ||
| Ileostomy | 13 (52.0) | 12 (48.0) | 2.68(0.67~10.74), 0.161 | |
| Time to reversal, days (SD) | 120.9 (45.1) | 169.1 (70.1) | 0.006* | |
| Time to reversal ≦109 days (%) | 10 (58.8) | 7 (41.2) | 0.002* | 6.31 (1.59~25.01), 0.007* |
| Prior complications (%) | 8 (44.4) | 11 (34.4) | 0.483 | – |
| Metastatic disease (%) | 0 | 4 (12.5) | 0.052 | – |
| Locally advanced disease (%) | 9 (50.0) | 12 (37.5) | 0.391 | – |
| Distance from anal verge, cm (SD) | 3.67 (1.82) | 3.09 (1.48) | 0.256 | – |
| Adjuvant Chemotherapy (%) | 5 (27.8) | 13 (40.6) | 0.359 | – |
*p < 0.05
The ROC was used to determine the timing of stoma reversal that optimally predicted the development of complications after stoma closure. The area under the curve was 0.75, and a 109-day interval from stoma construction to reversal yielded a sensitivity of 64.7% and a specificity of 87.5%. According to the univariate analysis, the closure of a defunctioning stoma within 109 days was a risk factor for developing postoperative complications related to stoma closure (p = 0.002; Table 5). In the multivariate analysis, the odds ratio for developing complications after stoma closure was 6.31 times higher in patients with a time to closure ≤109 days (95% CI 1.59–25.01; p = 0.007). However, after the multivariate analysis, ileostomy was not an independent factor for postoperative risk (OR 2.68; 95% CI 0.67–10.74; p = 0.161).
According to our data, the time to defunctioning stoma closure is related to several factors. The overall development of complications during the defunctioning period had no impact on the waiting time (p = 0.406). However, patients with renal insufficiency had earlier defunctioning stoma closure than those without renal insufficiency did (113.8 vs. 163.5 days; p = 0.003; Table 6). In addition, patients with readmissions for complications during the stool diversion period tended to have earlier stoma closure (122.5 vs. 161.0 days; p = 0.032). By contrast, patients with ileus waited 36.6 days longer (187.5 vs. 150.9 days; p < 0.001), and patients with initial ultra-low lying cancer (anastomotic level ≤5 cm) had stoma reversal 42.6 days later (157.6 vs. 115.0 days; p = 0.011) than their respective counterparts.
Table 6.
Factors associated with the time to reversal of defunctioning stoma
| Time to reversal, days (SD) | p value | |
|---|---|---|
| Age | 0.479 | |
| <70 | 157.5 (67.1) | |
| ≧70 | 143.5 (65.6) | |
| Sex | 0.384 | |
| Male | 145.1 (55.6) | |
| Female | 163.8 (80.6) | |
| Metastatic disease | 0.653 | |
| Yes | 162.5 (41.2) | |
| No | 151.5 (68.3) | |
| Locally advanced disease | 0.071 | |
| Yes | 173.8 (79.6) | |
| No | 136.5 (50.0) | |
| Distance from anal verge | 0.011* | |
| ≦5 cm | 157.6 (68.6) | |
| >5 cm | 115.0 (25.7) | |
| Complications during defuntioning period | 0.406 | |
| Yes | 141.8 (67.1) | |
| No | 158.5 (66.0) | |
| Renal insufficiency during defunctioning period | 0.003* | |
| Yes | 113.8 (36.3) | |
| No | 163.5 (69.0) | |
| Electrolyte imbalance during defunctioning period | 0.562 | |
| Yes | 163.6 (73.4) | |
| No | 149.1 (64.7) | |
| Ileus during defunctioning period | <0.001* | |
| Yes | 187.5 (3.5) | |
| No | 150.9 (67.2) | |
| Readmission during defunctioning period | 0.032* | |
| Yes | 122.5 (42.1) | |
| No | 161.0 (69.8) |
*p < 0.05
Discussion
Defunctioning stomata play a crucial role in low-lying rectal cancer surgery. According to previous research [7], they reduce not only symptomatic anastomotic leakage but also the need for urgent reoperation when the anastomosis level is ≤7 cm. A defunctioning stoma diverges the stool stream and protects the distal anastomosis in patients who undergo low-lying rectal cancer surgery, particularly those with locally advanced rectal cancer after CCRT. With the creation of a protective defunctioning stoma, the pooled rate of anastomotic leakage requiring relaparotomy is significantly lower in LAR and ultra-LAR than in anterior resection [8]. Neoadjuvant CCRT followed by TME combined with routine defunctioning stoma construction provides both surgical quality and effective local disease control for low-lying rectal cancers [9].
The type of defunctioning stoma created is mostly determined by the surgeon. When the splenic flexure is appropriately mobilized to allow for a tension-free colorectal anastomosis, the creation of a proper loop transverse colostomy is technically challenging and results in low-quality ostomies of tenuous blood supply. When a complication occurs in the pelvis or when a tumor recurs (leading to a repeat resection), a loop transverse colostomy limits the surgical options for the remaining colonic conduit. However, from the perspective of complications before and after stoma reversal, opinions regarding the choice of ileostomy or colostomy for diverging the stool stream remain divided. Our data show that colostomies were performed more often than ileostomies (57.6 vs. 42.4%) for fecal diversion, possibly because of the higher complication rate associated with ileostomies.
The occurrence of dermatitis and renal insufficiency is significantly higher in patients with loop ileostomy than in those with loop transverse colostomy, and after stoma closure, wound infection occurs significantly more often in loop transverse colostomy [10]. The Cochrane database supports the choice of loop ileostomy for fecal diversion, according to results obtained for the occurrence of postoperative stoma prolapse [11]. Another meta-analysis concluded that loop ileostomy reduces the risk of complications, including prolapse and sepsis, during the construction of the stoma but is associated with a high risk of dehydration and occlusion after stoma closure [12]. Defunctioning ileostomy-related complications during stool diversion include electrolyte and acid–base imbalance caused by an increased ileostomy output [13]. The readmission rate in patients with ileostomy is twice that in patients with colostomy after colorectal surgery [14]. The most common etiologies for readmission include postoperative infection, renal failure, and dehydration. The readmission rate for dehydration or renal failure within 30 days following ileostomy creation ranges from 17 to 36%, and an age of >50 years is an independent predictor of readmission for renal failure [15]. Consistent with our findings, the aforementioned observations indicate that ileostomy more frequently leads to renal insufficiency than colostomy does and significantly increases the readmission rate. A higher incidence of stoma prolapse was also observed in our ileostomy patients.
Another concern is that the consequences of stoma reversal are often underestimated [16]. Defunctioning ileostomy closure is associated with a morbidity rate of 18–40% and complications such as bowel obstruction, wound infection, peritonitis, intra-abdominal abscess, anastomotic leakage, enterocutaneous fistula, and bleeding [17–19]. Among these complications, bowel obstruction and SSI are the most common. These complications are associated with a reoperation rate of 3%–8% [20, 21]. Male sex and SSI after primary surgery are independent risk factors for developing wound infections [22]. One recent study suggested that purse-string skin closure is an effective technique for reducing the incidence of superficial SSI [23]. The higher wound infection and total complication rate after ileostomy closure in our study was due to the inclusion of not only deep but also superficial SSIs. The deep incisional infection rate was similar after two types of stoma reversal (3/25 patients for ileostomy and 2/25 patients for colostomy). The nonclosure of stomata is another concern to be addressed. Anastomotic leakage, fistula, advanced primary disease, local recurrence, and comorbidity have been identified as risk factors for nonreversal ileostomy after sphincter-preserving surgery for rectal cancer [24].
The interval between defunctioning stoma construction and stoma closure after LAR for rectal cancer treatment varies, and the median delay in stoma closure ranges from 5.6 to 10.3 months for ileostomy [18, 19, 25, 26]. Increased postoperative complications are associated with a delay in closure. The optimal period is suggested to be approximately 3–6 months [19]. Waterland et al. reported that a delay of >6 months in the reversal of defunctioning ileostomy increases complications and the length of the postoperative hospital stay [25]. The main causes of the delay are adjuvant chemotherapy, medical illness, and anastomotic leakage. In our study, the median time before stoma closure was 129 days. Differences in the interval did not alter the incidence of complications before stoma reversal, but the early closure of a defunctioning stoma was associated with a higher number of postoperative complications after stoma reversal. It is reasonable for a surgeon to close a defunctioning stoma earlier if the patient has renal insufficiency and is frequently readmitted. However, this might be harmful from the perspective of complications after stoma reversal. In the current study, a time to closure of ≤109 days was an independent risk factor for developing complications, including ileus, anastomotic insufficiency, and SSI, during the postoperative period. The precise cause of this phenomenon was not extensively analyzed, but this phenomenon might be attributable to the intraoperative difficulties in manipulation caused by prior unsolved intraperitoneal adhesions, subsequent excessive tissue trauma, and developing complications.
This study has some limitations, including the retrospective design and relatively small sample. Prospective studies with larger samples must be conducted to verify whether a colostomy is the ideal stoma creation procedure and to provide guidance regarding the effective utilization of defunctioning stomata in managing low-lying rectal cancer after CCRT and LAR.
Conclusions
We believe that the optimal timing of stoma reversal is at least 109 days after stoma construction in rectal cancer patients receiving neoadjuvant CCRT and LAR. Surgeons should especially show more patience in managing patients who experience impaired renal function and are readmitted for complications during the stool diversion period. The difference in the interval between stoma construction and reversal does not alter the incidence of complications before stoma reversal, but the early closure of a defunctioning stoma increases the complications associated with stoma closure.
Acknowledgements
The authors acknowledge the Colorectal Cancer Group from the Cancer Center of Kaohsiung Medical University Hospital for their contribution in data collection. This work was supported by grants from the Excellence for Cancer Research Center Grant through funding by the Ministry of Science and Technology (MOST105-2325-B-037-001); Ministry of Health and Welfare (MOHW105-TDU-B-212-134007); and Health and welfare surcharge of tobacco products, Taiwan, Republic of China, as well as grants from Kaohsiung Medical University Hospital (KMUHS10522, KMUHS10505, KMUHS10418, KMUHGCRC2016002) and the Center for Biomarkers and Biotech Drugs, Kaohsiung Medical University (KMU-TP105C01, KMU-TP105C11, KMU-TP106005, KMU-DK106005, SH000113(Give2Asia)). This work was partly supported by a grant from the Research Center for Environmental Medicine, Kaohsiung Medical University (KMU-TP105A11, KMU-TP105A12), and the grant of Biosignature in Colorectal Cancers, Academia Sinica, Taiwan.
Funding
No funding was received.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study
Authors’ contributions
TCY, HLT, and MYH contributed to the conception and design. CJM, CWH, and CMH contributed to the data analysis and interpretation. PFY and WCS contributed to the collection and assembly of patient data. All authors contributed to the manuscript writing and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable
Ethics approval and consent to participate
Not applicable
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- BMI
Body mass index
- CAA
Coloanal anastomosis
- CCRT
Concurrent chemoradiotherapy
- LAR
Low anterior resection
- ROC
Receiver operating curve
- SSI
Surgical site infection
- TME
Total mesorectal excision
Contributor Information
Tzu-Chieh Yin, Email: ajaxyin@gmail.com.
Hsiang-Lin Tsai, Email: chunpin870132@yahoo.com.tw.
Ping-Fu Yang, Email: adiemus.0615@gmail.com.
Wei-Chih Su, Email: lake0126@yahoo.com.tw.
Cheng-Jen Ma, Email: aisa.hsieh@msa.hinet.net.
Ching-Wen Huang, Email: baseball5824@yahoo.com.tw.
Ming-Yii Huang, Email: miyihu@kmu.edu.tw.
Chun-Ming Huang, Email: 930321@ms.kmuh.org.tw.
Jaw-Yuan Wang, Phone: +886-7-3122805, Email: cy614112@ms14.hinet.net, Email: jayuwa@cc.kmu.edu.tw.
References
- 1.Yin T, Kao L, Yang P, et al. Defunctioning stoma in locally advanced rectal cancer receiving concurrent chemoradiotherapy and low anterior resection. J Soc Colon Rectal Surgeon (Taiwan) 2014;25:8–15. [Google Scholar]
- 2.Rullier E, Le Toux N, Laurent C, Garrelon JL, Parneix M, Saric J. Loop ileostomy versus loop colostomy for defunctioning low anastomoses during rectal cancer surgery. World J Surg. 2001;25:274–7. doi: 10.1007/s002680020091. [DOI] [PubMed] [Google Scholar]
- 3.Sakai Y, Nelson H, Larson D, Maidl L, Young-Fadok T, Ilstrup D. Temporary transverse colostomy vs loop ileostomy in diversion: a case-matched study. Arch Surg. 2001;136:338–42. doi: 10.1001/archsurg.136.3.338. [DOI] [PubMed] [Google Scholar]
- 4.Chen CF, Huang MY, Huang CJ, et al. A observational study of the efficacy and safety of capecitabine versus bolus infusional 5-fluorouracil in pre-operative chemoradiotherapy for locally advanced rectal cancer. Int J Colorectal Dis. 2012;27:727–36. doi: 10.1007/s00384-011-1377-3. [DOI] [PubMed] [Google Scholar]
- 5.Martinez-Perez A, Carra MC, Brunetti F, de'Angelis N. Pathologic Outcomes of Laparoscopic vs Open Mesorectal Excision for Rectal Cancer: A Systematic Review and Meta-analysis. JAMA Surg. 2017:e165665. [DOI] [PubMed]
- 6.Fleshman J, Branda M, Sargent DJ, Boller AM, George V, Abbas M, Peters WR, Jr, Maun D, Chang G, Herline A, et al. Effect of laparoscopic-assisted resection vs open resection of stage II or III rectal cancer on pathologic outcomes: the ACOSOG Z6051 randomized clinical trial. JAMA. 2015;314:1346–55. doi: 10.1001/jama.2015.10529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Matthiessen P, Hallbook O, Rutegard J, Simert G, Sjodahl R. Defunctioning stoma reduces symptomatic anastomotic leakage after low anterior resection of the rectum for cancer: a randomized multicenter trial. Ann Surg. 2007;246:207–14. doi: 10.1097/SLA.0b013e3180603024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cong ZJ, Hu LH, Bian ZQ, et al. Systematic review of anastomotic leakage rate according to an international grading system following anterior resection for rectal cancer. PLoS One. 2013;8:e75519. doi: 10.1371/journal.pone.0075519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lin SC, Chen PC, Lee CT, et al. Routine defunctioning stoma after chemoradiation and total mesorectal excision: a single-surgeon experience. World journal of gastroenterology : WJG. 2013;19:1797–804. doi: 10.3748/wjg.v19.i11.1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Klink CD, Lioupis K, Binnebosel M, et al. Diversion stoma after colorectal surgery: loop colostomy or ileostomy? Int J Colorectal Dis. 2011;26:431–6. doi: 10.1007/s00384-010-1123-2. [DOI] [PubMed] [Google Scholar]
- 11.Guenaga KF, Lustosa SA, Saad SS, Saconato H and Matos D. Ileostomy or colostomy for temporary decompression of colorectal anastomosis. Cochrane Database Syst Rev. 2007: CD004647. [DOI] [PMC free article] [PubMed]
- 12.Rondelli F, Reboldi P, Rulli A, et al. Loop ileostomy versus loop colostomy for fecal diversion after colorectal or coloanal anastomosis: a meta-analysis. Int J Colorectal Dis. 2009;24:479–88. doi: 10.1007/s00384-009-0662-x. [DOI] [PubMed] [Google Scholar]
- 13.Weise WJ, Serrano FA, Fought J, Gennari FJ. Acute electrolyte and acid-base disorders in patients with ileostomies: a case series. Am J Kidney Dis. 2008;52:494–500. doi: 10.1053/j.ajkd.2008.04.015. [DOI] [PubMed] [Google Scholar]
- 14.Tyler JA, Fox JP, Dharmarajan S, et al. Acute health care resource utilization for ileostomy patients is higher than expected. Dis Colon Rectum. 2014;57:1412–20. doi: 10.1097/DCR.0000000000000246. [DOI] [PubMed] [Google Scholar]
- 15.Paquette IM, Solan P, Rafferty JF, Ferguson MA, Davis BR. Readmission for dehydration or renal failure after ileostomy creation. Dis Colon Rectum. 2013;56:974–9. doi: 10.1097/DCR.0b013e31828d02ba. [DOI] [PubMed] [Google Scholar]
- 16.Chow A, Tilney HS, Paraskeva P, Jeyarajah S, Zacharakis E, Purkayastha S. The morbidity surrounding reversal of defunctioning ileostomies: a systematic review of 48 studies including 6,107 cases. Int J Colorectal Dis. 2009;24:711–23. doi: 10.1007/s00384-009-0660-z. [DOI] [PubMed] [Google Scholar]
- 17.Poskus E, Kildusis E, Smolskas E, Ambrazevicius M, Strupas K. Complications after loop ileostomy closure: a retrospective analysis of 132 patients. Viszeralmedizin. 2014;30:276–80. doi: 10.1159/000366218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sier MF, van Gelder L, Ubbink DT, Bemelman WA, Oostenbroek RJ. Factors affecting timing of closure and non-reversal of temporary ileostomies. Int J Colorectal Dis. 2015;30:1185–92. doi: 10.1007/s00384-015-2253-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rubio-Perez I, Leon M, Pastor D, Diaz Dominguez J, Cantero R. Increased postoperative complications after protective ileostomy closure delay: an institutional study. World J Gastrointest Surg. 2014;6:169–74. doi: 10.4240/wjgs.v6.i9.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.van Westreenen HL, Visser A, Tanis PJ, Bemelman WA. Morbidity related to defunctioning ileostomy closure after ileal pouch-anal anastomosis and low colonic anastomosis. Int J Colorectal Dis. 2012;27:49–54. doi: 10.1007/s00384-011-1276-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mennigen R, Sewald W, Senninger N, Rijcken E. Morbidity of loop ileostomy closure after restorative proctocolectomy for ulcerative colitis and familial adenomatous polyposis: a systematic review. J Gastrointest Surg. 2014;18:2192–200. doi: 10.1007/s11605-014-2660-8. [DOI] [PubMed] [Google Scholar]
- 22.Akiyoshi T, Fujimoto Y, Konishi T, et al. Complications of loop ileostomy closure in patients with rectal tumor. World J Surg. 2010;34:1937–42. doi: 10.1007/s00268-010-0547-8. [DOI] [PubMed] [Google Scholar]
- 23.Wada Y, Miyoshi N, Ohue M, et al. Comparison of surgical techniques for stoma closure: a retrospective study of purse-string skin closure versus conventional skin closure following ileostomy and colostomy reversal. Mol Clin Oncol. 2015;3:619–22. doi: 10.3892/mco.2015.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kim YA, Lee GJ, Park SW, Lee WS, Baek JH. Multivariate analysis of risk factors associated with the nonreversal ileostomy following sphincter-preserving surgery for rectal cancer. Ann Coloproctol. 2015;31:98–102. doi: 10.3393/ac.2015.31.3.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Waterland P, Goonetilleke K, Naumann DN, Sutcliff M, Soliman F. Defunctioning ileostomy reversal rates and reasons for delayed reversal: does delay impact on complications of ileostomy reversal? A study of 170 defunctioning ileostomies. J Clin Med Res. 2015;7:685–9. doi: 10.14740/jocmr2150w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Phatak UR, Kao LS, You YN, et al. Impact of ileostomy-related complications on the multidisciplinary treatment of rectal cancer. Ann Surg Oncol. 2014;21:507–12. doi: 10.1245/s10434-013-3287-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study


