Abstract
Background
Hepatitis C infection is a major public health concern in low- and middle-income countries where an estimated 71.1 million individuals are living with chronic infection. The World Health Organization (WHO) has recently released new guidance for hepatitis C virus (HCV) treatment programs, which include improving the access to new direct-acting antiviral agents. In Vietnam, a highly populated middle-income country, the seroprevalence of HCV infection is approximately 4% and multiple genotypes co-circulate in the general population. Here we review what is currently known regarding the epidemiology of HCV in Vietnam and outline options for reducing the significant burden of morbidity and mortality in our setting.
Methods
We performed a systematic review of the currently available literature to evaluate what has been achieved to date with efforts to control HCV infection in Vietnam.
Results
This search retrieved few publications specific to Vietnam indicating a significant gap in baseline epidemiological and public health data. Key knowledge gaps identified included an understanding of the prevalence in specific high-risk groups, characterization of circulating HCV genotypes in the population and likely response to treatment, and the extent to which HCV treatment is available, accessed and utilized.
Conclusions
We conclude that there is an urgent need to perform up to date assessments of HCV disease burden in Vietnam, especially in high-risk groups, in whom incidence is high and cross infection with multiple genotypes is likely to be frequent. Coordinating renewed surveillance measures with forthcoming HCV treatment studies should initiate the traction required to achieve the WHO goal of eliminating HCV as a public health threat by 2030, at least in this region.
Keywords: Hepatitis C virus, Treatment access, Low and middle-income countries, Epidemiology, Vietnam
Background
Approximately 71.1 million people worldwide have chronic hepatitis C virus infection, resulting in over 500,000 deaths each year and creating a substantial burden on healthcare services [1–4]. The prevalence of hepatitis C infection is estimated to be 1%, although the highest burden (85%) of chronic infection is in low and middle income countries (LMICs) [1–4].
Hepatitis C virus (HCV), a single-stranded positive RNA virus, can cause individual infection with multiple subtypes, and therefore knowledge of viral genotype is particularly important for predicting virological and clinical treatment response. Genotype distribution varies geographically and is known to be particularly diverse in Southeast Asia [1].
Recently, the treatment options available for chronic hepatitis C infection have undergone a revolution with development of the direct-acting antivirals (DAAs) [5]. Treatment regimes containing drugs from this new class have demonstrated significantly higher rates of sustained virological response (SVR) compared to standard interferon- (IFN) and ribavirin-focused regimes [6, 7]. Additional benefits of this class include shorter treatment durations, oral administration and fewer side-effects. It has been estimated that, with unrestricted access to these costly DAA-containing regimes, more than 90% of HCV infected patients could attain SVR and thus achieve a definitive cure [8].
In May 2016, the World Health Organisation (WHO) launched a new global strategy to eliminate viral hepatitis as public health threat by 2030 [9]. A key priority in these recommendations is to ensure open access to the DAAs [10]. However, individuals living in LMICs, are unlikely to be able to afford regimes containing these new costly treatments [10]. Ongoing efforts to ensure equity of access include the development of generic drug formulations [10, 11].
The Socialist Republic of Vietnam has a population exceeding 93 million people. Since the mid-1980s there have been dramatic improvements in the key development indicators of this Southeast Asian country, including increases in life expectancy, gross-domestic product and expanding state and private healthcare provision [12]. This includes a new national strategy to improve access to viral hepatitis diagnostic and treatment services [12]. To achieve the WHO stated goal of HCV elimination as a public health problem, understanding how the control of HCV in countries such as Vietnam could be improved, is vital. This article aims to review the currently available published literature regarding the epidemiology, virological features and the current treatment options available for hepatitis C infection in Vietnam. We use these data to propose measures which could help in reducing the burden of HCV infections in Vietnam.
Methods
Search strategy and selection criteria
We searched PubMed-, MEDLINE- and Embase®-indexed articles for English-Vietnamese languages articles in June 2016, according to the PRISMA guidelines (Fig. 1) [13].
Fig. 1.

Flow chart of studies included in data synthesis. * Other sources from which records were obtained include Clinicaltrials.gov (n = 9, see Table 2) and Standard Operating Procedures for the identification, diagnosis and management of Hepatitis C infection published in Vietnamese and identified through web searches (n = 5)
Databases were searched using a combination of the following MeSH terms and keywords, including (Hepatitis C virus OR HCV) AND Vietnam AND (Prevalence OR Epidemiology OR Surve* OR Seroprevalence) (OR hepatitis C). No date restriction was used although included articles/data were restricted to original research articles relating to hepatitis C infection and HCV in Vietnam only, and on those published in either English or Vietnamese. We further searched the clinical trials registry http:/ClinicalTrial.gov for treatment trials currently underway or completed in Vietnam using the search term “Hepatitis C” AND “Vietnam”.
After performing the literature searches, one author (AB) extracted and screened the titles and abstracts before selecting and reviewing the full-text articles. The following data were extracted from each article: first author’s name, publication year, country of origin, date of study, type of patient group, and additional details of epidemiology, seroprevalence, HCV RNA positivity figures, treatment availability etc.
Results
Search results
A total of 99 abstracts for manuscripts published between September 1990 and June 2016 were identified. Of these, 42 contained information relevant for our review and full texts were retrieved [11, 14–60] (Table 1 and Fig. 1). The majority of publications (73.8%, 31/42) assessed the prevalence of HCV infection specifically in high-risk groups, including HIV infected patients, injecting drug users (IDU), community sex workers (CSW), men that have sex with men (MSM) and renal haemodialysis patients [11, 17, 21–23, 25, 26, 28, 29, 31, 32, 34, 36, 37, 39, 41–47, 51–53, 55–60] (Table 1). Online search for Vietnamese articles retrieved one research publication and four Standard Operating Procedures (SOPs); only the Vietnamese research paper retrieved from a Vietnamese journal was included in this analysis [38].
Table 1.
Characteristics population-based studies reporting prevalence of HCV in Vietnam
| Year | First Author | Prevalence | Group Type | Sample Size | Reference |
|---|---|---|---|---|---|
| 1998 | Kakumu | 1% | General Population | 1179 | [47] |
| 2007 | Nguyen | 1% | General Population | 837 | [45] |
| 2015 | Quesada | 4.7% | General Population | 27/571 | [27] |
| 2015 | Do | 3.3% 1.3%* |
General population | 509 | [30] |
| 2003 | Tran | 2% | General population | 334 | [15] |
| 1996 | Corwin | 2% | General population | 188 | [49] |
| 2012 | Sereno | 0.38–4.3% | General population | NAb | [21] |
| 2016 | Martinello | 42.5% | HIV | 89.452a | [24] |
| 2015 | Nadol | 53.3% | HIV/IDU | 3010 | [32] |
| 2015 | Zang | 88% | HIV | 1434 | [29] |
| 1999 | Follézou | >80% | HIV | 280 | [18] |
| 2012 | Durier | 22.9% | HIV | 110 | [20] |
| 2016 | Nguyen | 89% | HIV | 104 | [26] |
| 2016 | Hser | 74% | HIV/IDU | NAb, c | [11] |
| 2012 | Sereno | 95.8% | HIV/IDU | NAb | [21] |
| 2012 | Gish | 87% | IDU | NAb | [36] |
| 1998 | Kakumu | 47% | Liver Disease | 1179 | [47] |
| 2003 | Tran | 10% | Liver disease | 334 | [15] |
| 1996 | Corwin | 10% | Liver Disease | 188 | [49] |
| 2004 | Buchy | 9% | Liver disease | 45 | [46] |
| 2012 | Gish | 23% | Liver disease | NAb | [36] |
| 1993 | Cordier | 2% | Hepatocarcinoma | 152 | [54] |
| 2010 | Bjoerkvoll | 12.7% | Blood donors | 1305 | [38] |
| 2012 | Viet | 76.4% | Blood Donors | 1200a | [40] |
| 1994 | Song | 20.6% | Blood Donors | 491 | [52] |
| 2012 | Dunford | 26.6% | Dialysis | 8652a | [37] |
| 2016 | Duong | 8% | Dialysis | 142 | [22] |
| 2015 | Duong | 6% | Dialysis | 113 | [31] |
| 2012 | Gish | 54% | Hemodialysis | NAb | [36] |
| 2016 | Nadol | 28.4% | MSM | 1588 | [25] |
| 2012 | Dunford | 8.7% | CSW | 8652a | [37] |
*Presence of HCV RNA
aMulti countries study
b NA Not applicable
cEstimate
Twenty one percent (9/42) of the publications reported data from Ho Chi Minh City (HCMC) or from the southern Vietnam region (Table 1), while 52.3% (22/42) reported studies conducted in the north of the country, principally in Hanoi and surrounding provinces [18, 22, 27, 32, 36, 47, 49, 52, 53] (Table 1). Eight of forty two retrieved publications characterized the diversity of HCV genotypes in Vietnam [14, 16, 21, 33, 34, 48, 50, 51] (Table 1). Two manuscripts were reviews assessing risk factors associated with HCV acquisition in Vietnam [11, 24]. One article, retrieved from a Vietnamese website assessed the prevalence of HCV in the Vietnamese ethnic minority [38].
Nine clinical trials were found registered with ClinicalTrials.com (see Table 2). Five out of nine were completed but no results have been published to-date, two are completed with results available, one is active but no recruiting, while one is actively recruiting (Fig. 1). Only the two clinical trials that had published results were included in the review, the remaining seven studies have been excluded.
Table 2.
List of the clinical trials conducted in Vietnam to date a
| Year | Title | City | Sponsor | Status |
|---|---|---|---|---|
| 2010–2016 | Hepatic Safety of Raltegravir Versus Efavirenz as HIV Therapy for Patients With HIV and HCV Coinfection | Ho Chi Minh City, Hai Phong | University of Hawaii | Active, not recruiting |
| 2014–2016 | The Study of Safety, Pharmacokinetics, Pharmacodynamics of Peglamda (Peginterferon Lamda 1) on Healthy Volunteers and the Preliminary Evaluation of Peglamda and Hepasig (Ribavirin) Treatment’s Effects on Chronic Hepatitis C Patients | Unknown | Nanogen Pharmaceutical Biotechnology Co., Ltd | Completed |
| 2015–2016 | Feasibility of Interventions on People Who Inject Drugs in Vietnam | |||
| Implementation of a Sexual Health Intervention for Young Men Who Have Sex With Men (MSM) in Two Vietnamese Cities | Ho Chi Minh City, Hanoi | Inserm-ANRS | Completed | |
| 2013–2016 | HCV Treatment in HIV Co-Infected Patients in Asia | Hanoi | amfAR, | Completed |
| 2016 | Long Term Follow-up Study to Assess Durability of Sustained Virologic Response in Alisporivir-treated Hepatitis C Patients | Unknown | Debiopharm International SA | Completed, has results |
| 2011–2016 | Efficacy and Safety of Alisporivir Triple Therapy in Chronic Hepatitis C Genotype 1 Treatment-naïve Participants | Unknown | Debiopharm International SA | Completed, has results |
| 2016–2017 | Efficacy and Safety of Sofosbuvir/Velpatasvir Fixed Dose Combination for 12 Weeks in Participants With Chronic HCV | Unknown | Gilead Sciences | Recruiting |
| 2015 | 3-year Follow-up Study to Assess the Viral Activity in Hepatitis C Patients Who Failed Feeder DEB025/Alisporivir Study | Novartis Pharmaceuticals | Completed | |
| 2014–2016 | Implementation of a Sexual Health Intervention for Young Men Who Have Sex With Men (MSM) in Two Vietnamese Cities | Hanoi | National Development and Research Institutes, Inc. | Completed |
aFor further details please see the following website: ClinicalTrials.gov
The epidemiology of hepatitis C in Vietnam
In Vietnam, the background prevalence of HCV infection in the general population, at least historically, appears to vary depending on the region studied. In 1994 it was estimated that the seroprevalence in individuals without liver disease was 9% (43/491; 95% CI, 6.4%–11.5%) and 4% (18/511; 95% CI, 2.3%–5.6%) in HCMC and Hanoi, respectively [53] (Fig. 2). By 1998 it was estimated that in HCMC, the HCV seroprevalence in patients with underlying liver disease was 23% (69/289; 95% CI, 18.1%–27.8%) and in this population, HCV RNA was detectable in blood of 61% (42/69; 95% CI, 49.4%–72.5%) of the individuals [47]. This same study reported an HCV seroprevalence in healthy individuals in Da Lat (in the Southern Highlands) of 1% (9/890; 95% CI, 0.9%–1.0%); HCV RNA was detected in 44% (4/9) of these individuals [47]. Four years later in 2002, HCV RNA detection in HCMC was estimated to be 2% (2/100; 95% CI, 1.3%–5.3%) in apparently healthy individuals and 33% (45/234; 95% CI, 27.0%–39.0%) in patients with liver disease [15]. In Binh Thuan province (212 km northeast of HCMC) HCV seroprevalence in the general population between 2005 and 2007 was estimated to be 3.3% (17/509; 95% CI, 1.7%–4.8%); the male population exhibited a higher burden of seropositive samples (3.3%) than women (1.8%), and HCV RNA was detected in 52.9% (9/17) of the identified anti-HCV positive individuals (Fig. 2) [30]. Furthermore, one article was retrieved from a Vietnamese journal. The original manuscript was in Vietnamese and only the abstract was translated in English. The manuscript assessed the seroprevalence of HCV in the Vietnamese ethnic minority in the north of Vietnam and the detected seroprevalence in 1305 individuals was 1.22% (95% CI, 0.6%–1.7%) in 2011 [38].
Fig. 2.
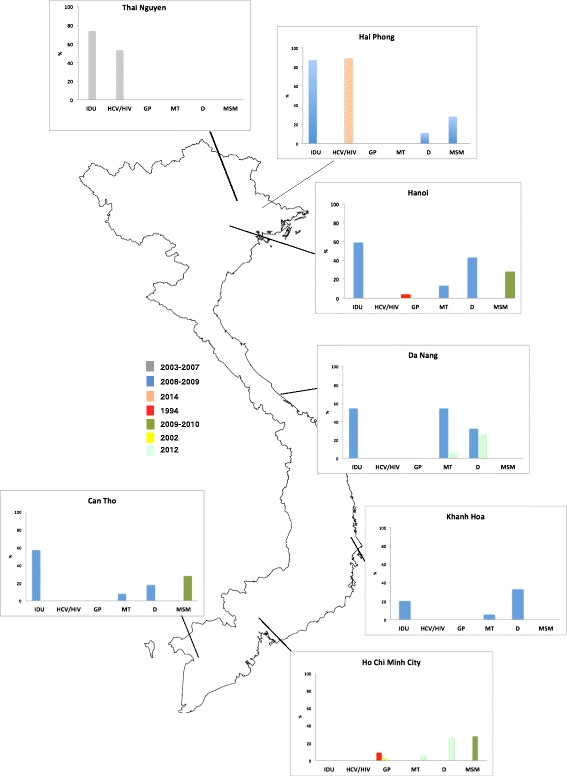
Vietnam Map and summary of the main studies conducted so far
A summary of the main studies performed in Vietnam to-date measuring the prevalence of HCV viral detection (PCR positivity) in selected populations by year of study publication and geographic location. * Data for ‘General adult healthy populations’ is for measured anti-HCV antibody seroprevalence. The map represents the location where the main studies have been conducted and summarized the HCV prevalence or seroprevalence observed during the studies in different groups, such as IDU, MSM, liver disease, hemodialysis and multi-blood transfusion and general population
The majority of the remaining publications evaluated HCV prevalence in specific high-risk groups (Table 1). These data estimated that 89% (1255/1434; 95% CI, 87.3%–90.6%) of IDU in Nguyen province (Northern Vietnam) were HCV RNA positive between 2005 and 2007 and 35% (502/1434; 95% CI, 32.5%-37.4) were HIV/HCV co-infected [29]. Similar figures were reported in Hai Phong province (northern Vietnam) in 2014 (Fig. 2), where the prevalence of HCV in HIV-infected individual was 89.4% (93/104; 95% CI, 82.1%–96.6%). This study also reported that the majority of HCV cases presented with clinical features of advanced liver fibrosis and cirrhosis [26].
Between 2005 and 2012, several studies were conducted around Vietnam (including HCMC, Hanoi, Hai Phong Province, Da Nang, Khanh Hoa Province and Can Tho), which aimed to determine HCV seroprevalence in patients undergoing renal haemodialysis or receiving multiple blood transfusions [23, 37]. These six hospital-based studies found an apparent iatrogenic HCV seroprevalence of 26% (n = 153/575; 95% CI, 22.4%–29.5%) and was similar at all sites assessed.
In 2010, a study estimated the prevalence of HCV infection in MSM in the south of Vietnam (Can Tho and HCMC) and in the north of Vietnam (Hai Phong Province and Hanoi) [25]. The overall seroprevalence of infection in 1588 male participants was 28.4% (95% CI, 26.1%–30.6%) [25]; 29% of the individuals were co-infected with HIV and more northerly regions observed a higher HCV and HCV/HIV co-infection prevalence [25]. A single publication from 1997 assessed the frequency of HCV infection in patients with chronic hepatitis in HCMC, and concluded that HCV was the underlying etiology in 19–27% of chronic hepatitis cases [19]. However, no indication of the sample size used to derive this estimate was provided.
The molecular epidemiology of HCV in Vietnam
A study conducted in four major conurbations in Vietnam – Hanoi, Hai Phong Province, Da Nang, Khanh Hoa Province and Can Tho, identified HCV genotypes 1, 6, 3 and 2 in 59.9% (n = 169), 37.9% (n = 107), 1.8% (n = 5) and 0.4% (n = 1) of samples collected from mixed healthy and high-risk populations, respectively [37]. While genotype 1 was the most prevalent HCV genotype in all four areas, the genotype distribution ranged from 47% in Khanh Hoa Province to 81% in Da Nang [37]. These four genotypes (1–3, 6, and) were identified in dialysis and multi-transfused individuals with nine recognized subtypes (1a, 1b, 2a, 3a, 3b, 6a, 6e, 6 h, 6 l) [39]. A further study similarly identified HCV genotype 1 as the predominant virus circulating in MSM in Hanoi, Hai Phong Province, HCMC and Can Tho [25]. Additionally, plasma samples from 97 HCV infected individuals were molecularly characterized between 2003 and 2010 in HCMC. The RNA sequences obtained from these samples suggested that genotypes 1b and 2a were imported from East Asia, 1a from the United States, 2i, 2j and 2 m imported from France [33, 58]. They also identified an additional 4 subtypes of genotype 6 (6a, 6ax, 6xb and 6xc) with no obvious international link [34].
Clinical trials
Of the nine trials registered on ClinicalTrials.gov, only two had published results available at time of review [61, 62]. Both studies were sponsored by Debiopharm International SA and neither the source country information nor the locations where trial participant recruitment was performed within the country have been specified. Both studies were multicenter international trials to assess the treatment responses (SVR to single agent or combination treatment) to the synthetic cyclosporine-like agent, Alisporivir, a novel host-targeting antiviral (HTA) agent [61, 62].
Discussion
Our literature search identified a paucity of published data regarding the nature or burden of HCV infection in Vietnam. Studies in the publications identified were generally conducted in high-risk populations and focused in the more populous northern provinces (around Hanoi) and southern provinces (around HCMC) of the country. In addition, most studies have measured HCV antibody seroprevalence rather than HCV RNA prevalence, which would more reliably indicate the numbers of individuals with active infection. As elsewhere, the available literature suggested that blood transfusions, intravenous drug use, and unsafe therapeutic injections were a likely common source of HCV infection in the Vietnamese populations studied. Furthermore, the data suggests while the HCV seroprevalence in the general population is approximately 4% (18/511; 95% CI, 2.3%–5.6%) in Hanoi and 9% (43/491; 95% CI, 6.4%–11.5%) in Ho Chi Minh City, which is high in comparison to America or Europe [63, 64], this is likely to be even higher in IDU (55.6%, n = 556/1000; 95% CI, 52.8%–58.3%), MSM (38.8%; n = 1558; 95% CI, 36.3%–41.2%), and hemodialysis patients (26.6%, n = 153/575; 95% CI, 22.4%–29.5%) [23, 31, 53, 57] . There are still substantial gaps in our understanding of the molecular epidemiology of HCV infection in Vietnam, however the limited findings from the few studies that have been conducted suggest that multiple (≥4) HCV genotypes and subtypes are circulating in the population [25, 37].
Ascertaining the burden of hepatitis C in Vietnam
Ascertaining the true likely burden of infection in Vietnam would facilitate advocacy for the implementation of disease specific strategies and guidelines, and provide useful baseline data for further epidemiological measurements. Our search identified that HCV seroprevalence in the population may be high (1–4%) compared to other countries in the region [3, 27, 53]. For example, several large-scale seroprevalence studies have reported HCV estimates of asymptomatic Chinese ranging from 0.39 to 3.2% depending of the region [65]. Other Asian countries such as Japan, India or Indonesia reported an HCV seroprevalence of between 2.3, 0.9 and 2.1% respectively [4].
Observations supporting the speculation that the true seroprevalence might be underestimated include data from neighboring countries such as China, where the reported rate of HCV infections increased more than 10-fold (from 21,000 reported cases/year to 210,000 reported cases/year) between 2003 and 2011 [66]. Reasons for this increase may include an increase in life expectancy and/or the increased detection/occurrence of new infections due to better surveillance and improved diagnostics. The ageing of the population creates two potentially major pressures on health care finances: increased utilization of health services and decreased revenues (as a declining share of the population is economically active).
Further insights into the current HCV situation in Vietnam may be provided by two non-resident populations; data on HCV infection in USA veterans after the Vietnam/American war suggest that returning veterans had a higher risk (10–17%) of being HCV infected than the general North American population (1.3%) [63, 64, 67]. This conflict was associated with the highest rate of HCV acquisition in comparison to any that the US has been engaged in. More recently, it was observed that Vietnamese migrants to other countries have a higher prevalence (5.8%) of HCV infection when compared to the autochthonous population (1.3%) [60, 68].
The transmission of HCV infection in clinical settings is still particularly concerning in Vietnamese healthcare facilities; the high HCV prevalence (26%) in haemodialysis patients [23, 31, 57] has not been observed in other Asian-Pacific countries (0.7–18.1%) [69]. This literature review also found a high seroprevalence (27%) in other high-risk populations including IDU, MSM, and CSW, similar data has been reported from studies conducted between the Myanmar and Chinese border region [70].
We therefore speculate that the seroprevalence in the general population might be higher than that currently estimated as the limited data currently available (only 42 publications published in over two decades) is from studies performed in only a few locations across Vietnam and which mostly had relatively small sample sizes upon which to base these calculations. Of note, the two most recent and larger studies performed to estimate seroprevalence in the general population have found an HCV prevalence of 3.3 and 4.7%, respectively [27, 30].
Identifying the genetic characteristics of HCV to improve public health
In addition to assessing the burden of HCV disease, further data is required regarding HCV genotype, viral mutations and viral loads in HCV infected patients in order to assess how to implement the recent HCV treatment guidance, and to estimate the cost-benefit of an integrated HCV control program.
While currently available data are limited, they imply a broad genetic diversity within circulating HCV strains in all parts of the country [33, 34, 37, 39, 58]. More detailed phylogenetic approaches have proposed that the HCV population has been influenced by the international turmoil of the past, with importation of genotypes that are more typically associated with North America, Europe and other parts of Asia [34]. This genetic variation can also be observed in high-risk groups, meaning that the formation of a local treatment policy may be more complex than in other locations. For example, the distribution of HCV genotype in haemodialysis patients is variable according to the geographical region, this may be due to a lack of infection control and or point source outbreaks [37]. This genotype distribution is in contrast to other countries in Asia, such as China, where hemodialysis patients are predominantly affected by genotype 2 HCV [71, 72]. An improved understanding of the genotype distribution and frequency of viral treatment mutations among Vietnamese patients will facilitate the development of appropriate local guidelines.
Current treatment options, molecular challenges and social taboos
Early diagnosis and access to treatment are essential components for an effective HCV control program in Vietnam. As in most LMICs, current treatment for HCV infection in Vietnam is limited to interferon-based (IFN-gamma and pegylated-IFN) therapy [10]. More recently, some patients have been treated with newer DAA agents at the Hospital for Tropical Diseases (HTD) in HCMC, where more than 18,000 patients with viral hepatitis are assessed each year. Further clinical treatment studies have received funding and are planned to commence in the near future [73].
The more widespread introduction of the DAAs is hindered by a lack of data regarding HCV genotype distribution and viral treatment mutations [74]. This lack of data is especially problematic for the early detection of treatment resistance mutations, which occurred previously with hepatitis B treatment and HCV treatment in Japan and North America [75–77]. Furthermore, the efficacy of DAAs against some of the less common genotypes, such as genotype 6 which was frequently identified in previously studies, is not well established [78].
Other barriers to increasing the utilization of HCV treatment in resource-limited settings such as Vietnam include high costs, a perceived complexity in treatment regimes and bureaucratic support for importation, licensing and distribution of medicines for which specific clinical trial data in the local populations is lacking. Vietnam is a rapidly developing country, whose GDP per-capita has increased from $1095 to $1684 from 2004 until 2014 [79]. However, the per capita income remains low (~ $1024) [80] with limited annual spending on healthcare (according to WHO, approximately $264 in 2006) [80]. A large proportion of people in Vietnam do not invest in health insurance and even those who do may not have full coverage to expensive treatment courses: pegylated-interferon courses are reimbursed at 30% ($10,000–18,000 for a 48 week treatmentcourse), and this partial reimbursement is based on doctor opinion only, with no allowance given for those in high-risk populations such as haemodialysis patients [81, 82].
While the treatment of HCV infection requires prescription from a medical practitioner, a further concern is the non-completion of treatment courses due to high costs and changes in family/patient financial circumstances. Some patients may also travel to other countries in the region where medications, which may be substandard, might be more affordable. Incomplete or inadequate treatment courses are likely to lead to increased rates of viral resistance, which has been previously widely observed in hepatitis B treatment programs in Vietnam [83, 84]. Further, PEG + INF + RBV remains (as 2016) the first line of therapy for chronic patients in many part of Asia, Vietnam included [85].
Study limitations
The majority of the studies included here had small sample sizes and concentrated on groups of individuals at known high-risk of acquiring HCV infection thus risking an overestimation bias. Given the heterogeneity of the study populations sampled, in addition to the broad time period and diverse geographic locations described, more accurate meta-analysis is not currently feasible. Thus, a considerable knowledge gap remains regarding the likely true burden of chronic HCV infection in the general, healthy adult Vietnamese population.
Conclusions
Relatively little is known regarding the burden of HCV infection in the general population in Vietnam. Available data suggest that the seroprevalence could be higher than previously assumed, especially in known high-risk groups such as patients undergoing haemodialysis. Limited data predict a broad distribution of HCV genotypes and, to-date, very few HCV treatment trials have been performed in Vietnam. While HCV control programs in Vietnam are in their infancy, there is an urgent need for more detailed disease burden and viral genotyping data, which, in addition to planned treatment studies, could be used to inform the development of local HCV treatment strategies and thus eventual attainment of the 2030 elimination target.
Acknowledgments
Funding
No funding was used for this manuscript.
Availability of data and materials
All data generated or analysed during this study are included in this published article [and its supplementary information files].
Authors’ contributions
AB performed the literature search and wrote the draft manuscript. TD made substantial contributions to conception and design; JD, SB and GET contributed with study revision and gave final approval. NG helped to retrieve data from HTD hospital. All authors contributed to this paper with revision editing, and final approval.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
“Not applicable”.
Ethics approval and consent to participate
This manuscript reports previously published data about HCV in Vietnam, no clinical studies were performed, therefore there was no need for ethics approval.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- HCV
Hepatitis C virus
- DAA
Direct acting anti-viral
- HTD
Hospital for Tropical Diseases
- HCMC
Ho Chi Minh City
- IFN
Interferon-based
- LMIC
Low to middle income countries
- WHO
World Health organization
- IDU
Injecting drug users
- MSM
Men that have sex with men
- CSW
Commercial sex workers
- SVR
Sustained virological response
Contributor Information
Alessandra Berto, Phone: +84 89241761, Email: aberto@oucru.org.
Jeremy Day, Email: jday@oucru.org.
Nguyen Van Vinh Chau, Email: chaunvv@oucru.org.
Guy E. Thwaites, Email: gthwaites@oucru.org
Ngoc Nghiem My, Email: ngocnm@oucru.org.
Stephen Baker, Email: sbaker@oucru.org.
Thomas C. Darton, Email: tdarton@oucru.org
References
- 1.Gower E, Estes C, Blach S, Razavi-Shearer K, Razavi H. Global epidemiology and genotype distribution of the hepatitis C virus infection. J Hepatol. 2014;61(1 Suppl):S45–S57. doi: 10.1016/j.jhep.2014.07.027. [DOI] [PubMed] [Google Scholar]
- 2.Simmonds P. Genetic diversity and evolution of hepatitis C virus--15 years on. J Gen Virol. 2004;85(Pt 11):3173–3188. doi: 10.1099/vir.0.80401-0. [DOI] [PubMed] [Google Scholar]
- 3.Blach S, Zeuzem S, Manns M, Altraif I, Duberg A-S, Muljono DH, et al. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol Hepatol. 2016;2(3):161–176. doi: 10.1016/S2468-1253(16)30181-9. [DOI] [PubMed] [Google Scholar]
- 4.Shepard CW, Finelli L, Alter MJ. Global epidemiology of hepatitis C virus infection. Lancet Infect Dis. 2005;5(9):558–567. doi: 10.1016/S1473-3099(05)70216-4. [DOI] [PubMed] [Google Scholar]
- 5.Poordad F, Dieterich D. Treating hepatitis C: current standard of care and emerging direct-acting antiviral agents. J Viral Hepat. 2012;19(7):449–464. doi: 10.1111/j.1365-2893.2012.01617.x. [DOI] [PubMed] [Google Scholar]
- 6.Jacobson IM, McHutchison JG, Dusheiko G, Di Bisceglie AM, Reddy KR, Bzowej NH, et al. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med. 2011;364(25):2405–2416. doi: 10.1056/NEJMoa1012912. [DOI] [PubMed] [Google Scholar]
- 7.Poordad F, McCone JJ, Bacon BR, Bruno S, Manns MP, Sulkowski MS, et al. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med. 2011;364(13):1195–1206. doi: 10.1056/NEJMoa1010494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hezode C, Bronowicki JP. Ideal oral combinations to eradicate HCV: the role of ribavirin. J Hepatol. 2016;64(1):215–225. doi: 10.1016/j.jhep.2015.09.009. [DOI] [PubMed] [Google Scholar]
- 9.WHO. Combating hepatitis B and C to reach elimination by 2030. World Health Organisation; 2016.
- 10.World Health Organization . Guidelines for the screening, care and treatment of persons with hepatitis C infection. 2016. pp. 1–135. [PubMed] [Google Scholar]
- 11.Hser Y-I, Liang D, Lan Y-C, Vicknasingam BK, Chakrabarti A. Drug abuse, HIV, and HCV in Asian countries. J NeuroImmune Pharmacol. 2016;11:383. doi: 10.1007/s11481-016-9665-x. [DOI] [PubMed] [Google Scholar]
- 12.Ministry of Health . Plan of preventing viral Hepatits Deseases in Vietnam 2015–2019. 2015. [Google Scholar]
- 13.Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nakano T, Lu L, Liu P, Pybus OG. Viral gene sequences reveal the variable history of hepatitis C virus infection among countries. J Infect Dis. 2004;190(6):1098–1108. doi: 10.1086/422606. [DOI] [PubMed] [Google Scholar]
- 15.Tran HT-T, Ushijima H, Quang VX, Phuong N, Li TC, Hayashi S, et al. Prevalence of hepatitis virus types B through E and genotypic distribution of HBV and HCV in ho chi Minh City. Vietnam Hepatol Res. 2003;26(4):275–280. doi: 10.1016/S1386-6346(03)00166-9. [DOI] [PubMed] [Google Scholar]
- 16.Tran HT-T, Ushijima H, Ngoc TT, Ha LD, Hayashi S, Sata T, et al. Recombination of genotypes B and C in hepatitis B virus isolated from a Vietnamese patient with fulminant hepatitis. Jpn J Infect Dis. 2003;56(1):35–37. [PubMed] [Google Scholar]
- 17.Nerurkar VR, Woodward CL, Nguyen HT, DeWolfe MF, Tashima LT, Zalles-Ganley A, et al. Lack of association between acquisition of TT virus and risk behavior for HIV and HCV infection in Vietnam. Int J Infect Dis. 1999;3(4):181–185. doi: 10.1016/S1201-9712(99)90021-8. [DOI] [PubMed] [Google Scholar]
- 18.Follezou JY, Lan NY, Lien TX, Lafon ME, Tram LT, Hung PV, et al. Clinical and biological characteristics of human immunodeficiency virus-infected and uninfected intravascular drug users in ho chi Minh City, Vietnam. AmJTrop Med Hyg. 1999;61(3):420–424. doi: 10.4269/ajtmh.1999.61.420. [DOI] [PubMed] [Google Scholar]
- 19.Ha MV. Some peculiarities of hepatobiliary diseases in Vietnam. J Gastroenterol Hepatol. 1997;12(5):S15–S18. doi: 10.1111/j.1440-1746.1997.tb00438.x. [DOI] [PubMed] [Google Scholar]
- 20.Durier N, Nguyen C, White LJ. Treatment of hepatitis C as prevention: a modeling case study in Vietnam. PLoS One. 2012;7(4):e34548. doi: 10.1371/journal.pone.0034548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sereno L, Mesquita F, Kato M, Jacka D, Van Nguyen TT, Nguyen TN. Epidemiology, responses, and way forward: the silent epidemic of viral hepatitis and HIV coinfection in Vietnam. J Int Assoc Physicians AIDS Care (Chic) 2012;11(5):311–320. doi: 10.1177/1545109712453939. [DOI] [PubMed] [Google Scholar]
- 22.Duong CM, Olszyna DP, Nguyen PD, ML ML. Challenges of hemodialysis in Vietnam: experience from the first standardized district dialysis unit in ho chi Minh City. BMC Nephrol. 2015;16:122. doi: 10.1186/s12882-015-0117-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Duong CM, McLaws M-L. An investigation of an outbreak of hepatitis C virus infections in a low-resourced hemodialysis unit in Vietnam. Am J Infect Control. 2016;44(5):560–566. doi: 10.1016/j.ajic.2016.01.014. [DOI] [PubMed] [Google Scholar]
- 24.Martinello M, Amin J, Matthews GV, Dore GJ. Prevalence and disease burden of HCV Coinfection in HIV cohorts in the Asia Pacific region: a systematic review and meta-analysis. AIDS Rev. 2016;18(2):68–80. [PubMed] [Google Scholar]
- 25.Nadol P, O’Connor S, Duong H, Mixson-Hayden T, Tram TH, Xia G-L, et al. High hepatitis C virus (HCV) prevalence among men who have sex with men (MSM) in Vietnam and associated risk factors: 2010 Vietnam Integrated Behavioural and Biologic Cross-Sectional Survey. Sex Transm Infect. 2016. [DOI] [PMC free article] [PubMed]
- 26.Nguyen Truong T, Laureillard D, Lacombe K, Duong Thi H, Pham Thi Hanh P, Truong Thi Xuan L, et al. High proportion of HIV-HCV Coinfected patients with advanced liver fibrosis requiring hepatitis C treatment in Haiphong, northern Vietnam (ANRS 12262) PLoS One. 2016;11(5):e0153744. doi: 10.1371/journal.pone.0153744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Quesada P, Whitby D, Benavente Y, Miley W, Labo N, Chichareon S, et al. Hepatitis C virus seroprevalence in the general female population from 8 countries. J Clin Virol. 2015;68:89–93. doi: 10.1016/j.jcv.2015.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wasitthankasem R, Vongpunsawad S, Siripon N, Suya C, Chulothok P, Chaiear K, et al. Genotypic distribution of hepatitis C virus in Thailand and Southeast Asia. PLoS One. 2015;10(5):e0126764. doi: 10.1371/journal.pone.0126764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang L, Celentano DD, Le Minh N, Latkin CA, Mehta SH, Frangakis C, et al. Prevalence and correlates of HCV monoinfection and HIV and HCV coinfection among persons who inject drugs in Vietnam. Eur J Gastroenterol Hepatol. 2015;27(5):550–556. doi: 10.1097/MEG.0000000000000321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Do SH, Yamada H, Fujimoto M, Ohisa M, Matsuo J, Akita T, et al. High prevalences of hepatitis B and C virus infections among adults living in Binh Thuan province, Vietnam. Hepatol Res. 2015;45(3):259–268. doi: 10.1111/hepr.12350. [DOI] [PubMed] [Google Scholar]
- 31.Duong CM, Olszyna DP, ML ML. Hepatitis B and C virus infections among patients with end stage renal disease in a low-resourced hemodialysis center in Vietnam: a cross-sectional study. BMC Public Health. 2015;15:192. doi: 10.1186/s12889-015-1532-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nadol P, O’connor S, Duong H, Le L-VN, Thang PH, Tram TH, et al. Findings from integrated behavioral and biologic survey among males who inject drugs (MWID) - Vietnam, 2009-2010: evidence of the need for an integrated response to HIV, hepatitis B virus, and hepatitis C virus. PLoS One. 2015;10(2):e0118304. doi: 10.1371/journal.pone.0118304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li C, Yuan M, Lu L, Lu T, Xia W, Pham VH, et al. The genetic diversity and evolutionary history of hepatitis C virus in Vietnam. Virology. 2014;468–470:197–206. doi: 10.1016/j.virol.2014.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Li C, Pham VH, Abe K, Lu L. Nine additional complete genome sequences of HCV genotype 6 from Vietnam including new subtypes 6xb and 6xc. Virology. 2014;468–470:172–177. doi: 10.1016/j.virol.2014.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Viet L, Lan NTN, Ty PX, Bjorkvoll B, Hoel H, Gutteberg T, et al. Prevalence of hepatitis B & hepatitis C virus infections in potential blood donors in rural Vietnam. Indian J Med Res. 2012;136(1):74–81. [PMC free article] [PubMed] [Google Scholar]
- 36.Gish RG, Bui TD, Nguyen CTK, Nguyen DT, Tran HV, Tran DMT, et al. Liver disease in Viet Nam: screening, surveillance, management and education: a 5-year plan and call to action. J Gastroenterol Hepatol. 2012;27(2):238–247. doi: 10.1111/j.1440-1746.2011.06974.x. [DOI] [PubMed] [Google Scholar]
- 37.Dunford L, Carr MJ, Dean J, Waters A, Nguyen LT, Ta Thi TH, et al. Hepatitis C virus in vietnam: high prevalence of infection in dialysis and multi-transfused patients involving diverse and novel virus variants. PLoS One. 2012;7(8):1–10. doi: 10.1371/journal.pone.0041266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nguyễn ThịLan Anh, Dương ThịThu Thủy, Nguyễn Thùy Linh Bùi ThịLan Anh, Nguyễn Tuấn Nghĩa NTH. Tình trạng nhiễm vi rút viêm gan B và Cởcộng đồng dân tộc thiểu sốxã MỏVàng, huyện Văn Yên, tỉnh Yên Bái. Vietnamese J Prev Med. 2013;49(147):1.
- 39.Pham VH, Nguyen HDP, Ho PT, Banh DV, Pham HLT, Pham PH, et al. Very high prevalence of hepatitis C virus genotype 6 variants in southern Vietnam: large-scale survey based on sequence determination. Jpn J Infect Dis. 2011;64(6):537–539. [PMC free article] [PubMed] [Google Scholar]
- 40.Bjoerkvoll B, Viet L, Ol HS, Lan NTN, Sothy S, Hoel H, et al. Screening test accuracy among potential blood donors of HBsAg, anti-HBc and anti-HCV to detect hepatitis B and C virus infection in rural Cambodia and Vietnam. Southeast Asian J Trop Med Public Health. 2010;41(5):1127–1135. [PubMed] [Google Scholar]
- 41.Tanimoto T, Nguyen HC, Ishizaki A, Chung PTT, Hoang TTH, Nguyen VT, et al. Multiple routes of hepatitis C virus transmission among injection drug users in Hai Phong, northern Vietnam. J Med Virol. 2010;82(8):1355–1363. doi: 10.1002/jmv.21787. [DOI] [PubMed] [Google Scholar]
- 42.Clatts MC, Colon-Lopez V, Giang LM, Goldsamt LA. Prevalence and incidence of HCV infection among Vietnam heroin users with recent onset of injection. J Urban Health. 2010;87(2):278–291. doi: 10.1007/s11524-009-9417-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pham DA, Leuangwutiwong P, Jittmittraphap A, Luplertlop N, Bach HK, Akkarathamrongsin S, et al. High prevalence of hepatitis C virus genotype 6 in Vietnam. Asian Pac J Allergy Immunol. 2009;27(2–3):153–160. [PubMed] [Google Scholar]
- 44.Quan VM, Go VF, Nam L Van, Bergenstrom A, Thuoc NP, Zenilman J, et al. Risks for HIV, HBV, and HCV infections among male injection drug users in northern Vietnam: a case-control study. AIDS Care; 2009;21(1):7–16. [DOI] [PMC free article] [PubMed]
- 45.Nguyen VTT, McLaws M-L, Dore GJ. Prevalence and risk factors for hepatitis C infection in rural north Vietnam. Hepatol Int. 2007;1(3):387–393. doi: 10.1007/s12072-007-9008-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Buchy P, Monchy D, An TTN, Srey CT, Tri DV, Son S, et al. Prevalence of hepatitis a, B, C and E virus markers among patients with elevated levels of Alanine aminotransferase and Aspartate aminotransferase in Phnom Penh (Cambodia) and Nha Trang (Central Vietnam) Bull Soc Pathol Exot. 2004;97(3):165–171. [PubMed] [Google Scholar]
- 47.Kakumu S, Sato K, Morishita T, Trinh KA, Nguyen HB, Banh VD, et al. Prevalence of hepatitis B, hepatitis C, and GB virus C/hepatitis G virus infections in liver disease patients and inhabitants in ho chi Minh, Vietnam. J Med Virol. 1998;54(4):243–248. doi: 10.1002/(SICI)1096-9071(199804)54:4<243::AID-JMV2>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 48.Simmonds P, Mellor J, Sakuldamrongpanich T, Nuchaprayoon C, Tanprasert S, Holmes EC, et al. Evolutionary analysis of variants of hepatitis C virus found in South-East Asia: comparison with classifications based upon sequence similarity. J Gen Virol. 1996;77(Pt 12):3013–3024. doi: 10.1099/0022-1317-77-12-3013. [DOI] [PubMed] [Google Scholar]
- 49.Corwin AL, Dai TC, Duc DD, Suu PI, Van NT, Ha LD, et al. Acute viral hepatitis in Hanoi, Viet Nam. Trans R Soc Trop Med Hyg. 1996;90(6):647–648. doi: 10.1016/S0035-9203(96)90418-6. [DOI] [PubMed] [Google Scholar]
- 50.Mellor J, Walsh EA, Prescott LE, Jarvis LM, Davidson F, Yap PL, et al. Survey of type 6 group variants of hepatitis C virus in Southeast Asia by using a core-based genotyping assay. J Clin Microbiol. 1996;34(2):417–423. doi: 10.1128/jcm.34.2.417-423.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tokita H, Okamoto H, Tsuda F, Song P, Nakata S, Chosa T, et al. Hepatitis C virus variants from Vietnam are classifiable into the seventh, eighth, and ninth major genetic groups. Proc Natl Acad Sci U S A. 1994;91(23):11022–11026. doi: 10.1073/pnas.91.23.11022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Song P, Duc DD, Hien B, Nakata S, Chosa T, Watanabe J, et al. Markers of hepatitis C and B virus infections among blood donors in ho chi Minh City and Hanoi, Vietnam. Clin Diagn Lab Immunol. 1994;1(4):413–418. doi: 10.1128/cdli.1.4.413-418.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nakata S, Song P, Duc DD, Nguyen XQ, Murata K, Tsuda F, et al. Hepatitis C and B virus infections in populations at low or high risk in ho chi Minh and Hanoi, Vietnam. J Gastroenterol Hepatol. 1994;9(4):416–419. doi: 10.1111/j.1440-1746.1994.tb01265.x. [DOI] [PubMed] [Google Scholar]
- 54.Cordier S, Le TB, Verger P, Bard D, Le CD, Larouze B, et al. Viral infections and chemical exposures as risk factors for hepatocellular carcinoma in Vietnam. Int J Cancer. 1993;55(2):196–201. doi: 10.1002/ijc.2910550205. [DOI] [PubMed] [Google Scholar]
- 55.Toan NL, Song LH, Kremsner PG, Duy DN, Binh VQ, Koeberlein B, et al. Impact of the hepatitis B virus genotype and genotype mixtures on the course of liver disease in Vietnam. Hepatology. 2006;43(6):1375–1384. doi: 10.1002/hep.21188. [DOI] [PubMed] [Google Scholar]
- 56.Phung TBT, Alestig E, Nguyen TL, Hannoun C, Lindh M. Genotype X/C recombinant (putative genotype I) of hepatitis B virus is rare in Hanoi, Vietnam-genotypes B4 and C1 predominate. J Med Virol. 2010;82(8):1327–1333. doi: 10.1002/jmv.21775. [DOI] [PubMed] [Google Scholar]
- 57.Dunford L, Carr MJ, Dean J, Waters A, Nguyen LT, Ta Thi TH, et al. Hepatitis C virus in Vietnam: high prevalence of infection in dialysis and multi-transfused patients involving diverse and novel virus variants. PLoS One. 2012;7(8):e41266. doi: 10.1371/journal.pone.0041266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fu Y, Qin W, Cao H, Xu R, Tan Y, Lu T, et al. HCV 6a prevalence in Guangdong province had the origin from Vietnam and recent dissemination to other regions of China: phylogeographic analyses. PLoS One. 2012;7(1):e28006. doi: 10.1371/journal.pone.0028006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sy BT, Nguyen HM, Toan NL, Song LH, Tong HV, Wolboldt C, et al. Identification of a natural intergenotypic recombinant hepatitis delta virus genotype 1 and 2 in Vietnamese HBsAg-positive patients. J Viral Hepat. 2015;22(1):55–63. doi: 10.1111/jvh.12228. [DOI] [PubMed] [Google Scholar]
- 60.Caruana S, Bowden S, Kelly H. Transmission of HCV in the Vietnamese community. Aust N Z J Public Health. 2001;25:276. doi: 10.1111/j.1467-842X.2001.tb00578.x. [DOI] [PubMed] [Google Scholar]
- 61.ClinicalTrails.gov. No Title. Efficacy and Safety of Alisporivir Triple Therapy in Chronic Hepatitis C Genotype 1 Treatment-naïve Participants. 2016.
- 62.ClinicalTrials.gov. No Title. Long Term Follow-up Study to Assess Durability of Sustained Virologic Response in Alisporivir-treated Hepatitis C Patients. 2016.
- 63.Zuniga IA, Chen JJ, Lane DS, Allmer J, Jimenez-Lucho VE. Analysis of a hepatitis C screening programme for US veterans. Epidemiol Infect. 2006;134(2):249–257. doi: 10.1017/S095026880500498X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Roselle GA, Danko LH, Kralovic SM, Simbartl LA, Kizer KW. National Hepatitis C Surveillance day in the veterans health Administration of the Department of veterans affairs. Mil Med. 2002;167(9):756–759. [PubMed] [Google Scholar]
- 65.Qin Q, Smith MK, Wang L, Su Y, Wang L, Guo W, et al. Hepatitis C virus infection in China: an emerging public health issue. J Viral Hepat. 2015;22(3):238–244. doi: 10.1111/jvh.12295. [DOI] [PubMed] [Google Scholar]
- 66.Duan Z, Jia J-D, Hou J, Lou L, Tobias H, Xu XY, et al. Current challenges and the management of chronic hepatitis C in mainland China. J Clin Gastroenterol. 2014;48(8):679–686. doi: 10.1097/MCG.0000000000000109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Brau N, Bini EJ, Shahidi A, Aytaman A, Xiao P, Stancic S, et al. Prevalence of hepatitis C and coinfection with HIV among United States veterans in the new York City metropolitan area. Am J Gastroenterol. 2002;97(8):2071–2078. doi: 10.1111/j.1572-0241.2002.05924.x. [DOI] [PubMed] [Google Scholar]
- 68.Richter C, Ter Beest G, Gisolf EH, VAN Bentum P, Waegemaekers C, Swanink C, et al. Screening for chronic hepatitis B and C in migrants from Afghanistan, Iran, Iraq, the former soviet republics, and Vietnam in the Arnhem region, The Netherlands. Epidemiol Infect. 2014;142(10):2140–2146. doi: 10.1017/S0950268813003415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Johnson DW, Dent H, Yao Q, Tranaeus A, Huang C-C, Han D-S, et al. Frequencies of hepatitis B and C infections among haemodialysis and peritoneal dialysis patients in Asia-Pacific countries: analysis of registry data. Nephrol Dial Transplant. 2009;24(5):1598–1603. doi: 10.1093/ndt/gfn684. [DOI] [PubMed] [Google Scholar]
- 70.Li L, Assanangkornchai S, Duo L, McNeil E, Li J. Risk behaviors, prevalence of HIV and hepatitis C virus infection and population size of current injection drug users in a China-Myanmar border city: results from a respondent-driven sampling survey in 2012. PLoS One. 2014;9(9):e106899. doi: 10.1371/journal.pone.0106899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Khan S, Attaullah S, Ali I, Ayaz S, Naseemullah, Khan SN, et al. Rising burden of hepatitis C virus in hemodialysis patients. Virol J. 2011;8:438. doi: 10.1186/1743-422X-8-438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Pybus OG, Barnes E, Taggart R, Lemey P, Markov PV, Rasachak B, et al. Genetic history of hepatitis C virus in East Asia. J Virol. 2009;83(2):1071–1082. doi: 10.1128/JVI.01501-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.DNDI. An alternative reseach and development strategy to deliver afforadble treatments for hepatitis C patients. The Drugs for Neglected Diseases initiative’ s 2016.
- 74.Pawlotsky J, REVIEWS IN. BASIC AND CLINICAL GASTROENTEROLOGY AND HEPATOLOGY in Interferon-Free Regimens. Gastroenterology. 2016;151(1):70–86. doi: 10.1053/j.gastro.2016.04.003. [DOI] [PubMed] [Google Scholar]
- 75.Gu L, Han Y, Li Y, Zhu T, Song X, Huang Y, et al. Emergence of Lamivudine-Resistant HBV during antiretroviral therapy including Lamivudine for patients Coinfected with HIV and HBV in China. PLoS One. 2015;10(8):e0134539. doi: 10.1371/journal.pone.0134539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cento V, Chevaliez S, Perno CF. Resistance to direct-acting antiviral agents: clinical utility and significance. Curr Opin HIV AIDS. 2015;10(5):381–389. doi: 10.1097/COH.0000000000000177. [DOI] [PubMed] [Google Scholar]
- 77.Chayama K, Hayes CN. HCV drug resistance challenges in Japan: the role of pre-existing variants and emerging Resistant strains in direct acting antiviral therapy. Viruses. 2015;7(10):5328–5342. doi: 10.3390/v7102876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Antaki N, Craxi A, Kamal S, Moucari R, Van der Merwe S, Haffar S, et al. The neglected hepatitis C virus genotypes 4, 5 and 6: an international consensus report. Liver Int. 2010;30(3):342–355. doi: 10.1111/j.1478-3231.2009.02188.x. [DOI] [PubMed] [Google Scholar]
- 79.WBG. The Word bank Country Srurvey FY14. Word Bank Group; 2014.
- 80.Res JMB, SP S, Shyamala R, JR R, RRM V, Dwivedi M, et al. Global policy report on the prevention and control of viral hepatitis IN WHO MEMBER STATES. Indian J Gastroenterol. 2012;2(2):146–149. [Google Scholar]
- 81.Mohamed R, Desmond P, Suh D-J, Amarapurkar D, Gane E, Guangbi Y, et al. Practical difficulties in the management of hepatitis B in the Asia-Pacific region. J Gastroenterol Hepatol. 2004;19(9):958–969. doi: 10.1111/j.1440-1746.2004.03420.x. [DOI] [PubMed] [Google Scholar]
- 82.Hipgrave DB, Van NT, Vu MH, Hoang TL, Do TD, Tran NT, et al. Hepatitis B infection in rural Vietnam and the implications for a national program of infant immunization. AmJTrop Med Hyg. 2003;69(3):288–294. [PubMed] [Google Scholar]
- 83.Ding CM, Sung JJY, Chan HLY, Luan J. Viral mutant discovery in hepatitis B virus quasi-species in patients undergoing long-term lamivudine treatment. Hong Kong Med J. 2015;21(Suppl 4):31–34. [PubMed] [Google Scholar]
- 84.Yin F, Wu Z, Fang W, Wu C, Rayner S, Han M, et al. Resistant mutations and quasispecies complexity of hepatitis B virus during telbivudine treatment. J Gen Virol. 2015;96(11):3302–3312. doi: 10.1099/jgv.0.000285. [DOI] [PubMed] [Google Scholar]
- 85.Younossi ZM, Tanaka A, Eguchi Y, Lim YS, Yu ML, Kawada N, et al. The impact of hepatitis C virus outside the liver: Evidence from Asia. Liver Int. 2016;37(2):159-172. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analysed during this study are included in this published article [and its supplementary information files].


