Abstract
AIM
To evaluate the association between egg consumption and risk of non-alcoholic fatty liver disease (NAFLD) development.
METHODS
This case-control study was conducted on individuals who were referred to two hepatology clinics in Tehran, Iran in 2015. The study included 169 patients with NAFLD and 782 controls. Egg consumption was estimated using a validated food frequency questionnaire. The participants were categorized according to the frequency of their egg consumption during the previous year: Less than two eggs per week, two to three eggs per week, and four or more eggs per week.
RESULTS
In the crude model, participants who consumed 2 to 3 eggs per week, were 3.56 times more likely to have NAFLD in comparison to those who consumed less than 2 eggs per week (OR: 3.56; 95%CI: 2.35-5.31). Adjustment for known risk factors of NAFLD strengthened this significant association so that individuals have consumed two to three eggs per week had 3.71 times higher risk of NAFLD than those who have eaten less than two eggs per week (OR: 3.71; 95%CI: 1.91, 7.75).
CONCLUSION
Our data indicate that higher egg consumption in common amount of usage is associated with higher risk of NAFLD.
Keywords: Egg, Diet, Non-alcoholic fatty liver disease, Dietary cholesterol
Core tip: The data indicate that egg consumption in common amount of usage is associated with risk of non-alcoholic fatty liver disease. According to the case-control design of this study, it can not show the causality effect; thus, these findings should be confirmed in future prospective studies with separate parts of eggs to find the etiological relationships.
INTRODUCTION
Non-alcoholic fatty liver disease (NAFLD) includes a spectrum of liver disordersfrom simple steatosis to non-alcoholic steatohepatitis (NASH), fibrosis, cirrhosis, and even hepatocellular carcinoma[1]. NAFLD is the most common cause of chronic liver diseases around the world[2] and may be considered as hepatic manifestation of metabolic syndrome[3]. The increasing prevalence of obesity, together with insulin resistance, hypertension, dyslipidemia, and eventually the metabolic syndrome dispose many people to the risk of NAFLD development in the futureyears[4].
Increasing evidence showed that dietary factors contribute to the pathophysiology and treatment of NAFLD[5-7]. Among the known dietary factors that involved in the development of NAFLD, dietary cholesterol has drown a great deal of attention. Current studies of animal models propose that excess dietary cholesterol is regarded as the key factorrelated to the riskof steatohepatitis and hepatic inflammation[8-10]. Addition of cholesterol to the diet of obese, diabetic mice increasedthe accumulation of hepatic free cholesterol, hepatocyte apoptosis, and liver fibrosis[11]. Moreover, an association between raised cholesterol intake and the risk or severity of NAFLD has been addressed by epidemiological studies[12-14].
Among single foods, eggs are regarded as a main source of dietary cholesterol, with one large egg containing almost 210 mg of cholesterol; on the other hand, eggs are rich in proteins, and other nutrients[15], which can improve human health. There is limited evidence on the relationship between egg consumption and NAFLD and its risk factorswith controversial results[16-18]. Therefore, the present study was designed to examine the association between egg consumption and risk of NAFLD development.
MATERIALS AND METHODS
Participants
The present case-control study was conducted on individuals who were undertaken a liver Ultrasound, and were referred to two Hepatology clinics in Tehran, Iran in 2015. The study included 169 patients with NAFLD and 782 controls. The cases were patients with NAFLD, which was diagnosed by a gastroenterologistaccording to the presence of hepatic steatosis in Ultrasound exam within previous month, and referred to our clinics to be examined by Fibroscan®, and the Fibroscan resultsshowed a Controlled Attenuation Parameter score of more than 263, and fibrosis score of more than 7. These patients were selected with the conveniencesampling procedure. Controls were randomly selected age- and sex-matched subjects from the sameclinic among patients with pancreatobiliay disorders who had been undertaken an Ultrasound showing no hepatic steatosis. The age ranges for matching were 20-40, 40-60 and > 60 years old. Data on each pair of cases and controls were collected at the same time. The participation rate in the study was 94% for cases and 98% for controls. Written informed consent was obtained from all the participants. The study protocol was approved by the local Ethics Review Committee.
Assessment of dietary intake
Dietary intake of patients was assessed using a valid and reliablesemi-quantitative food frequency questionnaire (FFQ), which included 168 items of foods with standard servingsizes, as commonly consumed by Iranians[19]. The consumptionfrequency of each food item was questioned ona daily, weekly or monthly basis and converted to dailyintakes. In the case of egg consumption, the participants were categorized according to the frequency of their egg consumptionduring the previous year: Less than two eggs per week, two to three eggs per week, and four or more eggs per week. Dietary nutrients intakes were calculated using NUTRITIONIST V (First Databank, Hearst Corp,San Bruno, CA, United States). The patients who had completed less than 90% of dietary questionnaires and subjects who reported extremely low or high energy intakes (< 500 or > 5000 kcal/d) were excluded from the study[20].
Assessment of other variables
Physical activity was evaluated using the metabolic equivalent task (MET) questionnaire[21,22]. Other covariate information including age, gender, smoking habits, alcohol consumption,medical history, and current use of medications were assessed using questionnaires. Weight and height of all participants were measured.
Statistical analysis
Baseline characteristics and dietary intakes were compared between cases and controls using t-test for continuous variables and χ2 for categorical variables. Egg consumptionwas dividedinto three ascending categories on an ordinal scale. Mean or prevalence of baseline characteristics wascomputed for each category. Baseline characteristicswere also compared using ANOVA for continuous variables and χ2 categorical variables. The relationship between NAFLDand egg consumptionwas assessed using multipleregression analysis. Estimates were presented inthree models; the first model was adjusted for age (continuous),and total energy intake (kcal/d). In thesecond model, we further controlled for body mass index (BMI), history of diabetes and smoking (non-smoker, current smoker). Finally, we further adjusted for physical activity (MET) and gender. All models were conducted by treating thefirstcategory of egg consumption (< 2/wk) as a reference. All the statistical analyses were done using SPSS for Windows (version 19; SPSS Inc., Chicago, IL).
RESULTS
Baseline characteristics, biochemical parametersand dietary intakes of the cases and controls are shown in Table 1. Mean age of the total study population was 43.54 ± 14.13 years and 41.5% (395) of participants were male. By design, cases and controls had the similar age and sex distribution. Patients with NAFLD had significantly more BMI, lower physically activity, lowerconsumption of alcohol, and were more likely to be smoker, and have diabetes in comparison to controls. Furthermore, the cases had elevatedfasting blood glucose (FBS), low-density lipoprotein cholesterol (LDL), Triglycerides, and reduced high density lipoprotein cholesterol (HDL) levels and increasedintake of protein, cholesterol, fiber and red/processed meats compared with the controls (Table 1).
Table 1.
Baseline characteristics, biochemical parameters and dietary intakes of study participants based on the patients with non-alcoholic fatty liver disease and control group
| Cases (n = 169) | Controls (n = 782) | P valuea | |
| Age (yr), mean ± SD | 42.65 ± 12.21 | 43.71 ± 14.52 | 0.373 |
| Male n (%) | 81 (47.9) | 314 (40.2) | 0.063 |
| BMI (kg/m2), mean ± SD | 33.19 ± 8.71 | 27.74 ± 4.495 | < 0.001 |
| Physical activity (MET), mean ± SD | 31.89 ± 3.15 | 34.33 ± 2.85 | < 0.001 |
| Current smokers, n (%) | 151 (89.9) | 145 (18.5) | < 0.001 |
| Drank alcohol in past year, n (%) | 22 (13.1) | 68 (8.7) | 0.077 |
| Diabetes type 2, n (%) | 26 (15.6) | 53 (6.8) | < 0.001 |
| FBS (mg/dL), mean ± SD | 109.29 ± 39.39 | 90.09 ± 29.24 | < 0.001 |
| Total cholesterol (mg/dL), mean ± SD | 184.79 ± 54.94 | 177.72 ± 38.74 | 0.221 |
| LDL (mg/dL), mean ± SD | 121.17 ± 43.04 | 104.26 ± 31.65 | < 0.001 |
| HDL (mg/dL), mean ± SD | 41.26 ± 16.72 | 47.72 ± 10.51 | 0.001 |
| Triglycerides (mg/dL), mean ± SD | 180.40 ± 123.81 | 131.97 ± 81.59 | < 0.001 |
| Total energy (kcal), mean ± SEM | 2627.67 ± 61.39 | 2746.69 ± 27.23 | 0.068 |
| Carbohydrate (% of total energy), mean ± SEM | 58.12 ± 0.95 | 59.82 ± 0.44 | 0.001 |
| Protein (% of total energy), mean ± SEM | 15.84 ± 0.18 | 14.07 ± 0.08 | < 0.001 |
| Fat (% of total energy), mean ± SEM | 29.23 ± 0.30 | 33.78 ± 0.20 | < 0.001 |
| Dietary cholesterol (mg/d), mean ± SEM | 315.31 ± 11.50 | 263.41 ± 5.35 | < 0.001 |
| Saturated fat (g/d), mean ± SEM | 30.62 ± 5.72 | 62.67 ± 2.67 | < 0.001 |
| Monounsaturated fat (g/d) (mg/d), mean ± SEM | 29.85 ± 0.48 | 32.00 ± 0.23 | < 0.001 |
| Polyunsaturated fat (g/d) (mg/d), mean ± SEM | 18.51 ± 5.74 | 59.58 ± 2.67 | < 0.001 |
| Dietary fiber (g/d), mean ± SEM | 19.21 ± 0.50 | 14.68 ± 0.23 | < 0.001 |
| Red/processed meats (g/d), mean ± SEM | 70.95 ± 2.66 | 36.00 ± 1.24 | < 0.001 |
Independent t-test for quantitative variables and χ2 test for qualitative variables. Dietary intakes (except total energy) were adjusted for age and total energy intake. BMI: Body mass index; MET: Metabolic equivalent task; FBS: Fasting blood sugar; LDL: Low-density lipoprotein cholesterol; HDL: High density lipoprotein cholesterol.
Basic characteristics and dietary intakes of the studied participants by categories of egg consumption are presented in Table 2. Compared to egg consumption of lower than two per week, higher egg consumption was associated with a lower average age, male sex, current smoking, higher energy intake, lower percent of total energy from carbohydrate and fat. Additionally, the subjects with higher egg consumption tended toconsume more protein, cholesterol, monounsaturated fat and red/processed meats, but less saturated and polyunsaturated fatty acids (Table 2).
Table 2.
Basic characteristics and dietary intakes of study participants by frequency of egg consumption n (%)
|
Egg consumption categories |
P valuea | |||
| < 2/wk (n = 589) | 2-3/wk (n = 142) | ≥ 4 /wk (n = 220) | ||
| Age (yr) | 45.65 ± 12.26 | 39.73 ± 13.18 | 40.35 ± 13.30 | < 0.001 |
| Male gender | 218 (37.0) | 56 (39.4) | 121 (55) | < 0.001 |
| BMI (kg/m2), mean ± SD | 28.58 ± 5.44 | 29.60 ± 7.34 | 28.51 ± 5.87 | 0.150 |
| Physical activity (MET), mean ± SD | 33.99 ± 3.05 | 33.42 ± 3.21 | 33.94 ± 2.95 | 0.136 |
| Current smokers | 155 (26.3) | 59 (41.8) | 82 (37.3) | < 0.001 |
| Total energy (kcal), mean ± SEM | 2580.59 ± 30.68 | 2744.94 ± 57.45 | 3101.07 ± 51.20 | < 0.001 |
| Carbohydrate (% of total energy), mean ± SEM | 60.44 ± 0.67 | 59.48 ± 0.63 | 58.14 ± 0.85 | 0.001 |
| Protein (% of total energy), mean ± SEM | 14.09 ± 0.10 | 14.71 ± 0.20 | 14.95 ± 0.17 | 0.001 |
| Fat (% of total energy), mean ± SEM | 33.06 ± 0.24 | 32.56 ± 0.49 | 32.97 ± 0.40 | < 0.001 |
| Dietary cholesterol (mg/d) | 226.40 ± 5.75 | 291.95 ± 11.60 | 383.90 ± 9.53 | < 0.001 |
| Saturated fat (g/d) | 56.70 ± 3.16 | 64.70 ± 6.38 | 52.57 ± 5.24 | < 0.001 |
| Monounsaturated fat (g/d) (mg/d), mean ± SEM | 31.20 ± 0.26 | 31.32 ± 0.53 | 32.91 ± 0.44 | < 0.001 |
| Polyunsaturated fat (g/d) (mg/d), mean ± SEM | 53.10 ± 3.20 | 57.26 ± 6.45 | 46.71 ± 5.30 | < 0.001 |
| Dietary fiber (g/d) | 15.65 ± 0.28 | 16.25 ± 0.57 | 14.60 ± 0.47 | < 0.001 |
| Red/processed meats (g/d) | 37.76 ± 1.53 | 47.79 ± 3.10 | 50.51 ± 2.54 | < 0.001 |
Dietary intakes (except total energy) were adjusted for age and total energy intake. BMI: Body mass index; MET: Metabolic equivalent task.
In secondary analysis, there wasa similar egg-NAFLD associationsin women (P-trend 0.001) and men (P-trend 0.048) (Table 3).
Table 3.
Odds ratio for non-alcoholic fatty liver diseaseaccording to egg consumption stratified by gender
|
Multivariate adjusted modela |
||
| Egg consumption | Female | Male |
| < 2/wk | 1.00 | 1.00 |
| 2-3/wk | 5.55 (2.30-13.37) | 1.90 (0.50-7.16) |
| ≥ 4/wk | 1.67 (0.68-4.10) | 0.25 (0.06-1.01) |
| P for trend | 0.001 | 0.048 |
Adjusted for age, energy intake, body mass index, history of diabetes, smoking, and physical activity.
Multivariate adjusted odds ratios for NAFLD based on egg consumption categories are indicated in Figure 1. In the crude model, participants that consumed 2 to 3 eggs per week, were 3.56 times more likely to have NAFLD in comparison to those who consumed less than 2 eggs per week (OR: 3.56; 95%CI: 2.35-5.31). After controlling for ageand total energy intake, consuming 2 to 3 eggs per week was positively associated with the risk of NAFLD (OR: 3.83; 95%CI: 2.49-5.89). These associations remained significant even after additionally controlling for BMI, history of diabetes and smoking (OR: 3.57; 95%CI: 1.89-6.75). Further adjustment for physical activity, and gender strengthened this significant association so that individuals who have consumed two to three eggs per week had 3.71 times higher risk of NAFLD than those who have eaten less than two eggs per week (OR: 3.71; 95%CI: 1.91-7.75). Egg consumption more than four per week was not significantly associated with the NAFLD risk.
Figure 1.
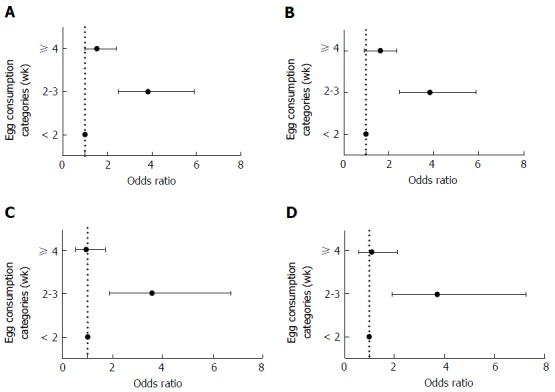
Multivariate-adjusted odds ratio for non-alcoholic fatty liver diseaseaccording to egg consumption. A: Crude model; B: Model 2, multivariate adjusted for age and energy intake; C: Model 3, further controlled for, body mass index, history of diabetes and smoking; D: Model 4, additionally adjusted for physical activity, and gender. Data are presented as the odds ratio (95%CI).
DISCUSSION
The results of the present study showed that the egg consumption increases the risk of NAFLD in common range of its consumption (two tothree eggs per week).This relationship was also significant after adjustment forage, gender, BMI, history of diabetes, smoking, and physical activity.
The role of diet and dietary supplements on the pathogenesis of NAFLD have been shown previously[23-36]; however, to our knowledge, no study has yet evaluated the association of egg consumption and NAFLD risk. It is well established that eggscontain a wide variety of essential nutrients and bioactive compounds that can affecthuman health. Their high quality protein, fats and micronutrients and low price make theman important part of many people’s diet[37]; despite the nutritional benefits of egg consumption, there are concerns abouttheir high content of cholesterol and saturatedfat and theirinfluences on metabolic disorders[38]. Thus, one possible explanation for theinverse association between egg consumption and risk of NAFLD development may be due to the highcholesterol content of egg. Previous studieshave shown that a higher consumption of cholesterolis associated with NAFLD and its exacerbation[12,13,39,40]. In addition, the presence of high amount of cholesterol in diet is necessary for development of NAFLD[41]. Baumgartner et al[39] have shown that daily egg consumption increases serum cholesterol and LDL-C concentrations inwomen; however, there was no effects on markers for inflammation, endothelial activity, and liver function. Interestingly, the consumption of egg white hydrolyzed with pepsin considerably improved hepatic steatosis[42]. Thus, it seems that the association between egg consumption and NAFLD is mainly due to high cholesterol content of it, and might not be seen when people consume only the white part of it. Therefore, more studies are recommended to evaluate the effects of consumption of different parts of egg on NAFLD risk[13].
An unexpected finding of the present study was that more than 4 eggs consumptionper week was not significantly associated with risk of NAFLD. This may be explained by the fact that nutritional factors are correlated with each other, and determining of the effect of particular nutrients or particular foods on a risk factor is difficult. The effects of egg cholesterol on serum cholesterol concentrations depends on the content of individuals’ diet specially the fiber content of it[43,44]. It is possible that those who ate more than 4 eggs per week, consumed it in mixed dishes containing vegetables, which reduces the absorption of cholesterol. Thus, we suggest that future studies assess the type of dishes with egg to find the possible interactions of different constituent of them.
It has been reported that dietary intake of patients with NAFLD was richer in saturated fat, cholesterol and was poorer in polyunsaturated fat[12]. Subramanian et al[40] have concluded that dietary cholesterol confers in progression of NAFLD to NASH. Furthermore, Zelber-Sagi et al[18] found that NAFLD patients have a higher intake of meat, which is another source of dietary cholesterol; however, some other studies only found a significant association between NAFLD and high dietary intake of carbohydrate and simple sugars[45,46], and some studies did find an association only between NAFLD and low intake of n-3 fatty acids and some antioxidants[16]. These dietary habits may accelerate the development of NAFLD by directly affecting steatosis of liver and oxidative injury[12].
This study was the first study that examined the relationship between egg consumption and risk of NAFLD in newly diagnosed patients who have not probably changed their diet due to the disease diagnosis; other strengths of this study includes its relatively large sample size, the high participation rate of participants, and socioeconomic differences of participants, which affects their dietary intakes.
Although we used a validated FFQ for measurement of dietary intakes, measurement error, and recall bias are unavoidable errors. Moreover, there might be some unknown risk factors that affect our results. Therefore, we recommend this analysis to be done in other populations.
In conclusion, our data indicate that egg consumption in common amount of usage is associated with riskof NAFLD. According to the case-control design of this study, it can not show the causality effect; thus, these findings should be confirmed in future prospective studies with separate parts of eggs to find the etiological associations.
ACKNOWLEDGMENTS
This work was supported by agrant from “National Nutrition and Food Technology Research Institute” of Shahid Beheshti University of Medical Sciences, and Digestive Disease Research Institute, Tehran University of Medical Sciences, Tehran, Iran.
COMMENTS
Background
Among the known dietary factors that affect the pathogenesis of non-alcoholic fatty liver disease (NAFLD), dietary cholesterol has drown a great deal of attention. Current studies propose that excess dietary cholesterol is regarded as the key factor related to the risk of steatohepatitis and hepatic inflammation. Among individual foods, eggs are regarded as a main source of dietary cholesterol; on the other hand, eggs are rich in proteins, and other nutrients. Limited research has assessed therelationship between egg consumption and risk of (NAFLD) development.
Research frontiers
Understanding of the associationbetween egg consumption andrisk of NAFLD developmentcan contribute to clarifyhow intake of special food groups correlate with the disease and could lead to more particularguidelinesfor NAFLD prevention.
Innovations and breakthroughs
This study showed that egg consumption in common amount of usage is associated with risk of NAFLD. It seems that this association is mainly due to high cholesterol content of it, and might not be seen when people consume only the white part of it.
Applications
According to the results of this study, the authors recommend low intake of eggs specially the yolk part of it for prevention of NAFLD; however, further studies are recommended to reach to a consus in this regard.
Peer-review
This is an interesting paper evaluating the association between egg consumption and NAFLD.
Footnotes
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Iran
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
Institutional review board statement: The study was approved by the ethics committee of National Nutrition and Food Technology Research Institute, Tehran, Iran.
Informed consent statement: All patients signed the informed consent form.
Conflict-of-interest statement: None of the authors had any personal or financial conflicts of interest to report.
Data sharing statement: Technical appendix, statistical code, and dataset available from the corresponding author at a_hekmat2000@yahoo.com.
Peer-review started: August 29, 2016
First decision: September 27, 2016
Article in press: March 17, 2017
P- Reviewer: Bellanti F, Tziomalos K S- Editor: Kong JX L- Editor: A E- Editor: Li D
References
- 1.Angulo P. Nonalcoholic fatty liver disease. N Engl J Med. 2002;346:1221–1231. doi: 10.1056/NEJMra011775. [DOI] [PubMed] [Google Scholar]
- 2.Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34:274–285. doi: 10.1111/j.1365-2036.2011.04724.x. [DOI] [PubMed] [Google Scholar]
- 3.Marchesini G, Brizi M, Bianchi G, Tomassetti S, Bugianesi E, Lenzi M, McCullough AJ, Natale S, Forlani G, Melchionda N. Nonalcoholic fatty liver disease: a feature of the metabolic syndrome. Diabetes. 2001;50:1844–1850. doi: 10.2337/diabetes.50.8.1844. [DOI] [PubMed] [Google Scholar]
- 4.Marchesini G, Bugianesi E, Forlani G, Cerrelli F, Lenzi M, Manini R, Natale S, Vanni E, Villanova N, Melchionda N, et al. Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology. 2003;37:917–923. doi: 10.1053/jhep.2003.50161. [DOI] [PubMed] [Google Scholar]
- 5.Papandreou D, Andreou E. Role of diet on non-alcoholic fatty liver disease: An updated narrative review. World J Hepatol. 2015;7:575–582. doi: 10.4254/wjh.v7.i3.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nseir W, Nassar F, Assy N. Soft drinks consumption and nonalcoholic fatty liver disease. World J Gastroenterol. 2010;16:2579–2588. doi: 10.3748/wjg.v16.i21.2579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zelber-Sagi S, Godos J, Salomone F. Lifestyle changes for the treatment of nonalcoholic fatty liver disease: a review of observational studies and intervention trials. Therap Adv Gastroenterol. 2016;9:392–407. doi: 10.1177/1756283X16638830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wouters K, van Gorp PJ, Bieghs V, Gijbels MJ, Duimel H, Lütjohann D, Kerksiek A, van Kruchten R, Maeda N, Staels B, et al. Dietary cholesterol, rather than liver steatosis, leads to hepatic inflammation in hyperlipidemic mouse models of nonalcoholic steatohepatitis. Hepatology. 2008;48:474–486. doi: 10.1002/hep.22363. [DOI] [PubMed] [Google Scholar]
- 9.Kleemann R, Verschuren L, van Erk MJ, Nikolsky Y, Cnubben NH, Verheij ER, Smilde AK, Hendriks HF, Zadelaar S, Smith GJ, et al. Atherosclerosis and liver inflammation induced by increased dietary cholesterol intake: a combined transcriptomics and metabolomics analysis. Genome Biol. 2007;8:R200. doi: 10.1186/gb-2007-8-9-r200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Savard C, Tartaglione EV, Kuver R, Haigh WG, Farrell GC, Subramanian S, Chait A, Yeh MM, Quinn LS, Ioannou GN. Synergistic interaction of dietary cholesterol and dietary fat in inducing experimental steatohepatitis. Hepatology. 2013;57:81–92. doi: 10.1002/hep.25789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Van Rooyen DM, Larter CZ, Haigh WG, Yeh MM, Ioannou G, Kuver R, Lee SP, Teoh NC, Farrell GC. Hepatic free cholesterol accumulates in obese, diabetic mice and causes nonalcoholic steatohepatitis. Gastroenterology. 2011;141:1393–1403, 1403.e1-5. doi: 10.1053/j.gastro.2011.06.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Musso G, Gambino R, De Michieli F, Cassader M, Rizzetto M, Durazzo M, Fagà E, Silli B, Pagano G. Dietary habits and their relations to insulin resistance and postprandial lipemia in nonalcoholic steatohepatitis. Hepatology. 2003;37:909–916. doi: 10.1053/jhep.2003.50132. [DOI] [PubMed] [Google Scholar]
- 13.Yasutake K, Nakamuta M, Shima Y, Ohyama A, Masuda K, Haruta N, Fujino T, Aoyagi Y, Fukuizumi K, Yoshimoto T, et al. Nutritional investigation of non-obese patients with non-alcoholic fatty liver disease: the significance of dietary cholesterol. Scand J Gastroenterol. 2009;44:471–477. doi: 10.1080/00365520802588133. [DOI] [PubMed] [Google Scholar]
- 14.Enjoji M, Nakamuta M. Is the control of dietary cholesterol intake sufficiently effective to ameliorate nonalcoholic fatty liver disease? World J Gastroenterol. 2010;16:800–803. doi: 10.3748/wjg.v16.i7.800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Song WO, Kerver JM. Nutritional contribution of eggs to American diets. J Am Coll Nutr. 2000;19:556S–562S. doi: 10.1080/07315724.2000.10718980. [DOI] [PubMed] [Google Scholar]
- 16.Han JM, Jo AN, Lee SM, Bae HS, Jun DW, Cho YK, Suk KT, Yoon JH, Ahn SB, Cho YJ, et al. Associations between intakes of individual nutrients or whole food groups and non-alcoholic fatty liver disease among Korean adults. J Gastroenterol Hepatol. 2014;29:1265–1272. doi: 10.1111/jgh.12520. [DOI] [PubMed] [Google Scholar]
- 17.Shi L, Liu ZW, Li Y, Gong C, Zhang H, Song LJ, Huang CY, Li M. The prevalence of nonalcoholic fatty liver disease and its association with lifestyle/dietary habits among university faculty and staff in Chengdu. Biomed Environ Sci. 2012;25:383–391. doi: 10.3967/0895-3988.2012.04.002. [DOI] [PubMed] [Google Scholar]
- 18.Zelber-Sagi S, Nitzan-Kaluski D, Goldsmith R, Webb M, Blendis L, Halpern Z, Oren R. Long term nutritional intake and the risk for non-alcoholic fatty liver disease (NAFLD): a population based study. J Hepatol. 2007;47:711–717. doi: 10.1016/j.jhep.2007.06.020. [DOI] [PubMed] [Google Scholar]
- 19.Esfahani FH, Asghari G, Mirmiran P, Azizi F. Reproducibility and relative validity of food group intake in a food frequency questionnaire developed for the Tehran Lipid and Glucose Study. J Epidemiol. 2010;20:150–158. doi: 10.2188/jea.JE20090083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jaceldo-Siegl K, Knutsen SF, Sabaté J, Beeson WL, Chan J, Herring RP, Butler TL, Haddad E, Bennett H, Montgomery S, et al. Validation of nutrient intake using an FFQ and repeated 24 h recalls in black and white subjects of the Adventist Health Study-2 (AHS-2) Public Health Nutr. 2010;13:812–819. doi: 10.1017/S1368980009992072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Aadahl M, Jørgensen T. Validation of a new self-report instrument for measuring physical activity. Med Sci Sports Exerc. 2003;35:1196–1202. doi: 10.1249/01.MSS.0000074446.02192.14. [DOI] [PubMed] [Google Scholar]
- 22.Kelishadi R, Rabiei K, Khosravi A, Famouri F, Sadeghi M, Rouhafza H, Shirani S. 2001. Assessment of physical activity of adolescents in Isfahan. [Google Scholar]
- 23.Askari F, Rashidkhani B, Hekmatdoost A. Cinnamon may have therapeutic benefits on lipid profile, liver enzymes, insulin resistance, and high-sensitivity C-reactive protein in nonalcoholic fatty liver disease patients. Nutr Res. 2014;34:143–148. doi: 10.1016/j.nutres.2013.11.005. [DOI] [PubMed] [Google Scholar]
- 24.Emamat H, Foroughi F, Eini-Zinab H, Taghizadeh M, Rismanchi M, Hekmatdoost A. The effects of onion consumption on treatment of metabolic, histologic, and inflammatory features of nonalcoholic fatty liver disease. J Diabetes Metab Disord. 2015;15:25. doi: 10.1186/s40200-016-0248-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Eslamparast T, Eghtesad S, Hekmatdoost A, Poustchi H. Probiotics and Nonalcoholic Fatty liver Disease. Middle East J Dig Dis. 2013;5:129–136. [PMC free article] [PubMed] [Google Scholar]
- 26.Eslamparast T, Eghtesad S, Poustchi H, Hekmatdoost A. Recent advances in dietary supplementation, in treating non-alcoholic fatty liver disease. World J Hepatol. 2015;7:204–212. doi: 10.4254/wjh.v7.i2.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Eslamparast T, Poustchi H, Zamani F, Sharafkhah M, Malekzadeh R, Hekmatdoost A. Synbiotic supplementation in nonalcoholic fatty liver disease: a randomized, double-blind, placebo-controlled pilot study. Am J Clin Nutr. 2014;99:535–542. doi: 10.3945/ajcn.113.068890. [DOI] [PubMed] [Google Scholar]
- 28.Faghihzadeh F, Adibi P, Hekmatdoost A. The effects of resveratrol supplementation on cardiovascular risk factors in patients with non-alcoholic fatty liver disease: a randomised, double-blind, placebo-controlled study. Br J Nutr. 2015;114:796–803. doi: 10.1017/S0007114515002433. [DOI] [PubMed] [Google Scholar]
- 29.Faghihzadeh F, Adibi P, Rafiei R, Hekmatdoost A. Resveratrol supplementation improves inflammatory biomarkers in patients with nonalcoholic fatty liver disease. Nutr Res. 2014;34:837–843. doi: 10.1016/j.nutres.2014.09.005. [DOI] [PubMed] [Google Scholar]
- 30.Ghaemi A, Taleban FA, Hekmatdoost A, Rafiei A, Hosseini V, Amiri Z, Homayounfar R, Fakheri H. How Much Weight Loss is Effective on Nonalcoholic Fatty Liver Disease? Hepat Mon. 2013;13:e15227. doi: 10.5812/hepatmon.15227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hekmatdoost A, Shamsipour A, Meibodi M, Gheibizadeh N, Eslamparast T, Poustchi H. Adherence to the Dietary Approaches to Stop Hypertension (DASH) and risk of Nonalcoholic Fatty Liver Disease. Int J Food Sci Nutr. 2016;67:1024–1029. doi: 10.1080/09637486.2016.1210101. [DOI] [PubMed] [Google Scholar]
- 32.Mofidi F, Yari Z, Poustchi H, Merat S, Nourinayyer B, Malekzadeh R, Hekmatdoost A. Effects of Synbiotics Supplementation in Lean Patients with Nonalcoholic Fatty Liver Disease: Study Protocol of a Pilot Randomized Double-blind Clinical Trial. Arch Iran Med. 2016;19:282–284. [PubMed] [Google Scholar]
- 33.Rahimlou M, Ahmadnia H, Hekmatdoost A. Dietary supplements and pediatric non-alcoholic fatty liver disease: Present and the future. World J Hepatol. 2015;7:2597–2602. doi: 10.4254/wjh.v7.i25.2597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rahimlou M, Yari Z, Hekmatdoost A, Alavian SM, Keshavarz SA. Ginger Supplementation in Nonalcoholic Fatty Liver Disease: A Randomized, Double-Blind, Placebo-Controlled Pilot Study. Hepat Mon. 2016;16:e34897. doi: 10.5812/hepatmon.34897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shavakhi A, Minakari M, Firouzian H, Assali R, Hekmatdoost A, Ferns G. Effect of a Probiotic and Metformin on Liver Aminotransferases in Non-alcoholic Steatohepatitis: A Double Blind Randomized Clinical Trial. Int J Prev Med. 2013;4:531–537. [PMC free article] [PubMed] [Google Scholar]
- 36.Yari Z, Rahimlou M, Eslamparast T, Ebrahimi-Daryani N, Poustchi H, Hekmatdoost A. Flaxseed supplementation in non-alcoholic fatty liver disease: a pilot randomized, open labeled, controlled study. Int J Food Sci Nutr. 2016;67:461–469. doi: 10.3109/09637486.2016.1161011. [DOI] [PubMed] [Google Scholar]
- 37.Miranda JM, Anton X, Redondo-Valbuena C, Roca-Saavedra P, Rodriguez JA, Lamas A, Franco CM, Cepeda A. Egg and egg-derived foods: effects on human health and use as functional foods. Nutrients. 2015;7:706–729. doi: 10.3390/nu7010706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Weggemans RM, Zock PL, Katan MB. Dietary cholesterol from eggs increases the ratio of total cholesterol to high-density lipoprotein cholesterol in humans: a meta-analysis. Am J Clin Nutr. 2001;73:885–891. doi: 10.1093/ajcn/73.5.885. [DOI] [PubMed] [Google Scholar]
- 39.Baumgartner S, Kelly ER, van der Made S, Berendschot TT, Husche C, Lütjohann D, Plat J. The influence of consuming an egg or an egg-yolk buttermilk drink for 12 wk on serum lipids, inflammation, and liver function markers in human volunteers. Nutrition. 2013;29:1237–1244. doi: 10.1016/j.nut.2013.03.020. [DOI] [PubMed] [Google Scholar]
- 40.Subramanian S, Goodspeed L, Wang S, Kim J, Zeng L, Ioannou GN, Haigh WG, Yeh MM, Kowdley KV, O’Brien KD, et al. Dietary cholesterol exacerbates hepatic steatosis and inflammation in obese LDL receptor-deficient mice. J Lipid Res. 2011;52:1626–1635. doi: 10.1194/jlr.M016246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Emamat H, Noori M, Foroughi F, Rismanchi M, Eini-Zinab H, Hekmatdoost A. An Accessible and Pragmatic Experimental Model of Nonalcoholic Fatty Liver Disease. Middle East J Dig Dis. 2016;8:109–115. doi: 10.15171/mejdd.2016.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Garcés-Rimón M, González C, Uranga JA, López-Miranda V, López-Fandiño R, Miguel M. Pepsin Egg White Hydrolysate Ameliorates Obesity-Related Oxidative Stress, Inflammation and Steatosis in Zucker Fatty Rats. PLoS One. 2016;11:e0151193. doi: 10.1371/journal.pone.0151193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cohn JS, Kamili A, Wat E, Chung RW, Tandy S. Reduction in intestinal cholesterol absorption by various food components: mechanisms and implications. Atheroscler Suppl. 2010;11:45–48. doi: 10.1016/j.atherosclerosissup.2010.04.004. [DOI] [PubMed] [Google Scholar]
- 44.Gunness P, Gidley MJ. Mechanisms underlying the cholesterol-lowering properties of soluble dietary fibre polysaccharides. Food Funct. 2010;1:149–155. doi: 10.1039/c0fo00080a. [DOI] [PubMed] [Google Scholar]
- 45.Solga S, Alkhuraishe AR, Clark JM, Torbenson M, Greenwald A, Diehl AM, Magnuson T. Dietary composition and nonalcoholic fatty liver disease. Dig Dis Sci. 2004;49:1578–1583. doi: 10.1023/b:ddas.0000043367.69470.b7. [DOI] [PubMed] [Google Scholar]
- 46.Toshimitsu K, Matsuura B, Ohkubo I, Niiya T, Furukawa S, Hiasa Y, Kawamura M, Ebihara K, Onji M. Dietary habits and nutrient intake in non-alcoholic steatohepatitis. Nutrition. 2007;23:46–52. doi: 10.1016/j.nut.2006.09.004. [DOI] [PubMed] [Google Scholar]


