Abstract
Background
Minimally invasive esophagectomy (MIE) is increasingly used for the treatment of esophageal cancer. However, the ideal approach of MIE is not yet standardized. We explore the ideal approach of MIE according to the location of the tumor and compare the clinical outcomes between patients with cancer arising in the upper third of the esophagus and those with tumors involving the middle and lower third of the esophagus.
Methods
We included patients with esophageal carcinoma and had clear indications for MIE. For cancer arising in the upper third of the esophagus, MIE McKeown approach was performed. For tumors involving the middle and lower third of the esophagus, MIE Ivor Lewis approach was adopted.
Results
Of the 251 patients included in this analysis, 200 patients underwent Ivor-Lewis MIE and 51 patients underwent McKeown MIE. The incidence of anastomotic leak, anastomotic stenosis and recurrent laryngeal nerve injury was significantly higher in the McKeown MIE group than that in the Ivor Lewis MIE group. The 30-day postoperative mortality rate was 1.2% (n = 1) in the McKeown MIE group. Lymph nodes harvested were significantly more in the MIE-McKeown group than in Ivor Lewis MIE group (P < 0.05). The median follow-up period was 15 months (1–25 months) and the overall survival rate at 1 year stratified by pathologic stage at esophagectomy was 95.9% (stage 1), 83.8% (stage II), 73.4% (stage III).
Conclusions
MIE for esophageal cancer according to the location and clinical stage of the tumor will decrease all postoperative complications and may yield the greatest benefit from surgery.
Keywords: Esophageal cancer, Esophagectomy laparoscopy, Thoracoscopy
Highlights
-
•
Mid and lower esophageal cancers occurred in most of included patients.
-
•
MIE Ivor Lewis approach is used for the treatment of mid and lower esophageal cancers.
-
•
For cancer arising in the upper third of the esophagus, MIE McKeown approach was performed.
-
•
MIE according to the location of the tumor decrease postoperative complications.
1. Introduction
Cancers arising from the esophagus are relatively common in China. Surgery is the primary therapy for esophageal cancer. However, traditional open esophagectomy carries significantly high risks of operative morbidity and mortality. Advances in surgical treatment have made minimally invasive esophagectomy (MIE) more popular and widely acceptable since the 1990s [1], [2]. Currently, MIE can be performed through the laparoscopic transhiatal, the laparoscopicthoracoscopic Ivor Lewis, or the laparoscopic-thoracoscopic McKeown approach [3].
The prognosis for esophageal cancer is very poor with the median survival time ranging from 1 to 2 years [4]. Traditional open surgical transthoracic and transhiatal esophagectomies are associated with a relatively high morbidity and mortality rate which can impair quality of life [5], [6]. In an effort to improve the outcomes associated with esophagectomy, minimally invasive approach to esophageal resection has been adopted [7], [8], [9], [10], [11]. The use of MIE approaches was associated with a shorter hospital stay, decreased morbidity and improved outcomes compared with open esophagectomy [12], [13], [14], [15], [16], [17], [18]. However, the rapid worldwide use of MIE approaches has not been followed by a rigorous scientific analysis of results. Large scale multicenter surgical clinical trials are difficult to conduct, and few studies have had sufficient follow-up to judge the long-term oncologic results. The numerous technical variables (including the patient's position - prone vs supine or the transoral anvil introduction vs the transthoracic route during an Ivor-Lewis esophagectomy) have jeopardized the results [19]. The ideal approach of MIE is not yet standardized.
The potential benefits of McKeown MIE are a more proximal resection margin and improved lymph node dissection. However, these procedures are associated with a high complication rate and mortality, which is partly due to subtotal resection of esophagus, severe trauma, RLN injury and anastomotic leak, and stricture. And one recent study found that McKeown esophagectomy was an independent predictor of increased perioperative morbidity or mortality, independent of tumor histology. There are some advantages for MIE Ivor-Lewis approach such as the avoidance of subtotal resection of esophagus, the lower anastomotic leakage and stenosis, the better vascular supply of gastric conduit, and preliminarily good clinical outcomes. With the development of operative technic, the MIE Ivor-Lewis has become increasingly prevalent and is safe and feasible for patients with mid and lower esophageal cancer. In the present era of minimally invasive surgery, the choice among these approaches is mainly dominated by institutional preferences and clinical opinions. Until recently, the ideal approach of MIE is not yet standardized. The optimal approach should be tailored for each patient according to the location and clinical stage of the tumor [20]. The purpose of this study was to evaluate the effects of MIE approach according to the location of the tumor and to compare differences in perioperative and long term clinical outcomes between patients with cancer arising in the upper third of the esophagus and those with tumors involving the middle and lower third of the esophagus.
2. Patients and methods
2.1. Study design
This was a prospective observation trial to evaluate the feasibility and outcomes of tailored therapy for each patient according to the location and the clinical stage of the tumor in an institution that had experience in minimally invasive surgical techniques. Feasibility was defined as the ability to carry out therapy without significant perioperative mortality. Patients included were those scheduled for MIE surgery at the Department of thoracic surgery Chinese PLA General Hospital. from January 2014 onwards. The medical charts were reviewed to obtain clinical data by using a standardized data collection sheet. Variables recorded included sex, age at the time of surgery, comorbid conditions, preoperative symptoms, operative details, and tumor-specific variables. All patients underwent esophagus-gastro-duodenoscopy, a thoracoabdominal CT scan and selective endoscopic ultrasound evaluation every 6 months. All the data was stored in a database that was analyzed for perioperative complications and long term outcomes in patients. The study was approved by the Ethics Committee of Chinese PLA General Hospital, Beijing, China.
2.2. Pretreatment staging
All patients underwent pretreatment staging according to the American Joint Committee on Cancer seventh edition criteria. Diagnostic investigations routinely included a history taking, physical examination, routine laboratory tests, a barium study and an oesophago-gastro-duodenoscopy with biopsies, a neck and thoracoabdominal CT scan, selective endoscopic ultrasound evaluation, and external ultrasonography of the neck, with fine-needle aspiration of lymph nodes when cancer was suspected.
2.3. Eligibility criteria
Patients who were histologically proved suffering from esophageal carcinoma by taking biopsy specimens from the tumor during an endoscopy and the stomach was required to be available for use as a conduit were eligible for inclusion in the study. Patients with tumors of no clinical evidence of metastatic spread (M0) were enrolled. A complete evaluation of cardiac and respiratory functions was made, and all patients were found fit for operation and anesthesia. Informed consent was obtained from all patients. Exclusion criteria included patients with a prior antireflux or gastric operation. Patients who had a prior right thoracotomy or a prior major neck operation were excluded. Patients who were converted to an open procedure were also excluded.
2.4. Operative approach
For tumors involving the middle and lower third of the esophagus, MIE Ivor Lewis approach was adopted (Group A). For cancer arising in the upper third of the esophagus, MIE McKeown approach was performed (Group B). The distance between the incisal edge and the upper edge of the tumor is 5–7 cm. The operative technique has previously been described in detail elsewhere [21], [22], [23], [24]. Two main operative techniques are adopted at our institution, namely a three-incision MIE with cervical anastomosis (MIE McKeown approach) and a two-incision MIE with intrathoracic anastomosis (MIE Ivor Lewis approach). After general anesthesia and endotracheal intubation, the patient was placed in the left lateral decubitus position. The surgeon stands on the right and the assistant on the left. Three ports were placed in the 7th intercostal space at mid-axillary line, the 5th intercostal space at the anterior axillary line and in scapular line at the 8th intercostal space respectively. In short, MIE McKeown approach consists of mobilizing the thoracic esophagus from the hiatus to the thoracic inlet, intrathoracic lymphadenectomy and cervical anastomosis. A staging laparoscopy was performed in the same setting or as a separate procedure to ensure resectability in most patients. Neck wound is closed in layers after placing a corrugated drain.
Laparoscopic and thoracoscopic dissection is also performed and followed by a side-to-side stapled thoracic anastomosis for the MIE Ivor Lewis approach [12].
Lymph nodes along left gastric artery and the lesser curvature of the stomach, mediastinal lymph nodes, abdominal lymph nodes that included paracardial lymph nodes, with bilateral recurrent nerve lymph nodes were routinely dissected. If the primary tumor was located between the upper and mid-thoracic esophagus, supraclavicular lymph nodes were dissected simultaneously.
A feeding jejunostomy tube is placed laparoscopically in most of the patients. One tube-drain is placed close to hiatus, trocars are removed, and ports are closed. Patients are extubated in the operating room. The patients post-operation were given parenteral nutrition through the deep vein or through a nasogastric tube. Patients are discharged on tube feed support with a full liquid diet.
2.5. Adjuvant chemotherapy
For patients with positive lymph node metastasis in a resected specimen, adjuvant chemotherapy using 5-fluororouracil and cisplatin every 3 weeks for two cycles was administered.
2.6. Perioperative and postoperative outcomes
All patients surviving operation were followed until death or time of database closure (January 2016). Perioperative and postoperative data were record, including duration of surgery, blood loss, and postoperative length of stay, surgical outcomes, postoperative complications, operative mortality, recurrence and hospital and late morbidity and mortality. The primary endpoint of this study was all cause mortality. Secondary endpoints were operative and postoperative major morbidity.
2.7. Statistical analysis
Data are reported as the mean ± SD for continuous variables and as frequencies for categorical variables. Continuous variables were compared by Student t-test for normally distributed values. Differences in percentages were evaluated using the Chi-square test. Survival curves during follow-up were plotted by the Kaplane Meier method. Significance was considered to be present for values of p < 0.05. A commercial statistical software package (SPSS for Windows, version 17.0; SPSS, Chicago, IL) was used for data analysis.
3. Results
3.1. Participants
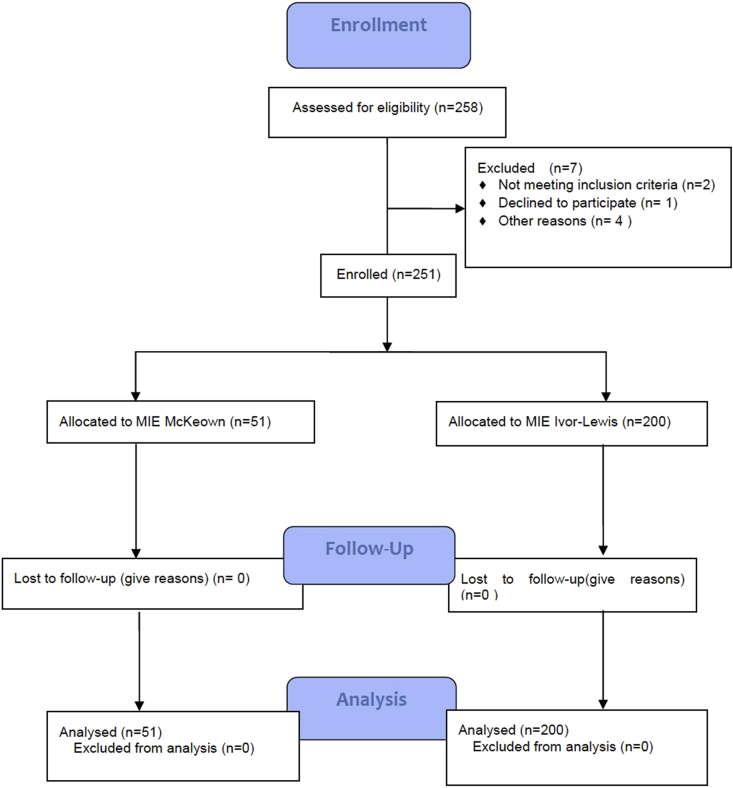
The study was started in January 2014. We enrolled 258 patients. Two patients were converted to open surgery for bleeding. One patient withdrew consent after registration. Of the remaining 255 patients, two patients had a prior right thoracotomy, one patient had a prior gastric operation, and one patient did not undergo resection, leaving 251 patients eligible for the analysis (Fig. 1).
Fig. 1.
Consort diagram.
Clinical characteristics of the patients with tumor arising in the middle and lower third of the esophagus (Group A) and the patients with esophageal cancer arising in the upper third of the esophagus (Group B) are listed in Table 1. The total number of included cases was 251, and the male/female ratio was approximately 4:1. At presentation, 47 patients had comorbid conditions in the form of diabetes and 49 patients suffered from coronary artery disease. Of the 251 patients with esophageal cancer, 155 (61.7%) patients had dysphagia. All patients were histologically proved suffering from esophageal carcinoma and had clear indications and clinical purpose for MIE. Preoperative nutrition of these patients was maintained by high protein calorie liquid or soft diet. The demographic characteristics were similar between two groups.
Table 1.
Characteristics of patients who underwent esophagectomy: Comparing MIE-McKeown and MIE-Ivor Lewis.
| Preoperative characteristics | Group A (n = 200) | Group B (n = 51) | Total, n = 251 | P |
|---|---|---|---|---|
| Age (years) | 59.9 ± 8.4 | 61.4 ± 9.1 | 60.4 ± 8.7 | 0.192 |
| Sex (Male) | 169 (84.5%) | 38 (74.7%) | 207 (82.5%) | 0.102 |
| BMI, kg/m2, median (IQR) | 23.5 (20.1–26.8) | 22.9 (20.8–25.4) | 23.1 (21.0–26.8) | 0.134 |
| Pretreatment weight loss, n (%) | 81 (40.5) | 20 (38.6) | 101 (40.2) | 1.000 |
| Comorbid conditions | ||||
| History of gastroesophageal reflux disease | 152 (76.0) | 40 (78.4) | 192 (76.9) | 0.854 |
| Diabetes mellitus, n (%) | 37 (18.5) | 10 (19.6) | 47 (18.7) | 0.842 |
| Coronary artery disease, n (%) | 40 (20.0) | 9 (17.6) | 49 (19.5) | 0.844 |
| COPD/emphysema, n (%) | 44 (22.0) | 12 (23.5) | 56 (22.3) | 0.851 |
| Chronic renal insufficiency, baseline Cr > 2 mg/dL or HD, n (%) | 4 (2.0) | 1 (2.0) | 5 (2.0) | 1.000 |
| Smoking history | ||||
| Current | 28 (14) | 6 (11.7) | 34 (13.6) | 0.820 |
| Former | 118 (59.0) | 24 (47.1) | 142 (56.6) | 0.202 |
| Never | 54 (27.0) | 21 (41.2) | 75 (29.8) | 0.059 |
BMI indicates body mass index; HD, hemodialysis.
3.2. Operative and postoperative details
For cancer arising in the upper third of the esophagus, MIE McKeown approach was performed. For tumors involving the middle and lower third of the esophagus, MIE Ivor Lewis approach was adopted. Intraoperative and postoperative data of patients are presented in Table 2. Anastomotic leak was detected in eight patients. The leak was minor and managed conservatively. Three patients experienced hoarseness of voice reflecting recurrent laryngeal injury (paresis), which resolved within 5 weeks. Three patients (1.2%) required conversion to open thoracotomy due to adhesions or emergency thoracotomy for bleeding. One patient died of anastomotic leakage postoperatively. Twelve patients experienced long-term complications in the form of stenosis, with dysphagia that required serial dilations.
Table 2.
Technical and perioperative aspects of patients: Comparing MIE-Ivor Lewis and MIE-McKeown.
| Group A (n = 200) | Group B (n = 51) | P | |
|---|---|---|---|
| Duration of surgery (min) | 322.8 ± 50.5 | 296.1 ± 35.9 | 0.013 |
| Blood loss (ml) | 181.5 ± 80.0 | 172.6 ± 90.1 | 0.594 |
| Conversion to open, n (%) | 2 (1.0) | 1 (2.0) | 0.496 |
| Reoperations, n (%) | 2 (1.0) | 2 (3.9) | 0.184 |
| Anastomotic leakage, n (%) | 1 (0.5) | 4 (7.8) | 0.007 |
| Anastomotic stenosis, n (%) | 2 (1.0) | 6 (11.8) | 0.001 |
| RLN injury, n (%) | 0 (0) | 3 (5.9) | 0.008 |
| Pulmonary complications | 10 (5.0) | 8 (15.7) | 0.103 |
| Chylothorax, n (%) | 0 (0) | 1 (1.9) | 0.203 |
| Cardiac arrhythmia, n (%) | 6 (3.0) | 2 (3.9) | 0.666 |
| ICU stay, n (%) | 3 (1.5) | 2 (3.9) | 0.268 |
| Hospital length of stay (d) | 16.7 ± 5.5 | 18.6 ± 6.5 | 0.621 |
| Mortality at 30 days, n (%) | 0 (0) | 1 (2.0) | 0.203 |
RLN, recurrent laryngeal nerve.
The mean operative time of McKeown MIE group is significant shorter than the Ivor Lewis MIE group (296.1 ± 35.9s vs. 322.8 ± 50.5s, p = 0.013, respectively). However, the occurrence of anastomotic leak, anastomotic stenosis and RLN injury in the McKeown MIE group was significant higher than those in the Ivor Lewis MIE group. There was no statistical significance with the blood loss, adequacy of cancer resection, reoperations, disparity of chylothorax, cardiac arrhythmia, ICU length of stay and hospital length of stay between two groups.
There were no intraoperative mortalities. The 30-day postoperative mortality rate was 1.2% (n = 1) in the McKeown MIE group. This patient died of multiple organ dysfunction syndromes resulting from anastomotic leakage which was confirmed by gastrograf in swallow.
3.3. Pathologic findings
For 215 patients (85.7%), the malignancy was squamous cell cancer. Twenty-eight patients (11.2%) had adenocarcinoma. Six patients had neuroendocrine carcinoma. The remaining 2 patients had Melanoma (Table 3). Lymph nodes harvested were significantly more in the MIE-McKeown group than that of in Ivor Lewis MIE group (P < 0.05). There was no significant difference in the R0 resection rate between groups A and B (95.0%vs. 96.1%). As shown in Table 3, 129 patients were pathologically positive for LN metastasis, which was evaluated as an indication for adjuvant chemotherapy. Of these 129 patients, 98 (76%) received adjuvant chemotherapy. The remaining 31 (24%) patients did not receive chemotherapy because of age (n = 12, 9.3%), other cancer diagnosis (n = 8, 6.2%), postoperative complications (n = 6, 4.7%), and patient's instances (n = 5, 3.9%).
Table 3.
Pathologic findings after operation: Comparing MIE-Ivor Lewis and MIE-McKeown.
| Tumor specific variables | Group A (n = 200) | Group B (n = 51) | P |
|---|---|---|---|
| AJCC stage, n (%) | |||
| I | 20 (10.0) | 7 (13.7) | 0.451 |
| IIa | 53 (26.5) | 11 (21.6) | 0.590 |
| IIb | 48 (24.0) | 11 (21.6) | 0.854 |
| III | 99 (49.5) | 22 (43.1) | 0.437 |
| Squamous tumor type, n (%) | 170 (85.0) | 45 (88.2) | 0.659 |
| Adenocarcinoma tumor type, n (%) | 24 (12.0) | 4 (7.9) | 0.618 |
| Neuroendocrine carcinoma, n (%) | 4 (2.0) | 2 (3.9) | 0.605 |
| Melanoma, n (%) | 2 (1.0) | 0 (0) | 1.000 |
| Adequacy of cancer resection | |||
| Negative margins, n (%) | 190 (95.0) | 49 (96.1) | 0.496 |
| Lymph nodes harvested(n) | 19.5 ± 9.6 | 27.5 ± 10.1 | 0.031 |
| Lymph nodes metastasis rate, n (%) | 105 (52.5) | 24 (47.1) | 0.532 |
| Adjuvant chemotherapy n (%) | 80 (40.0) | 18 (35.3) | 0.562 |
AJCC, American Joint Committee on Cancer.
3.4. Clinical outcomes
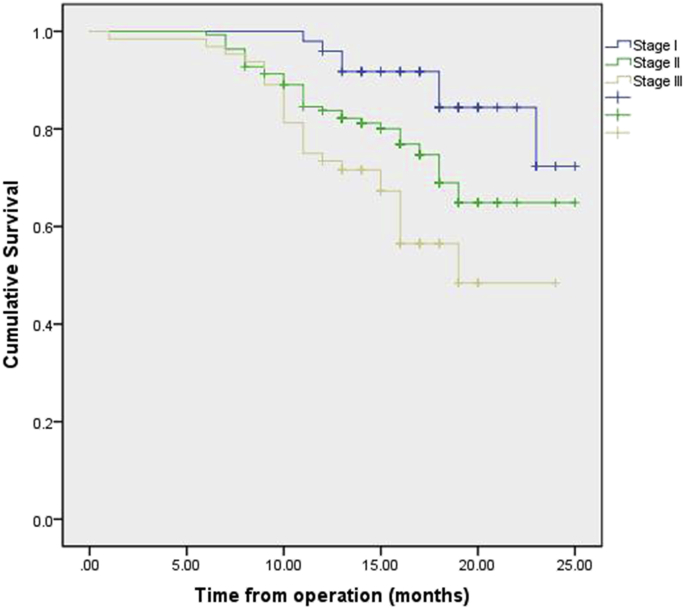
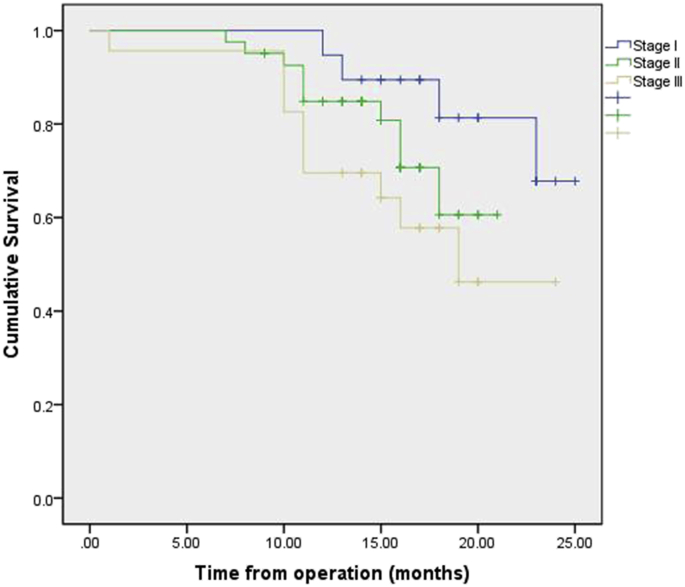
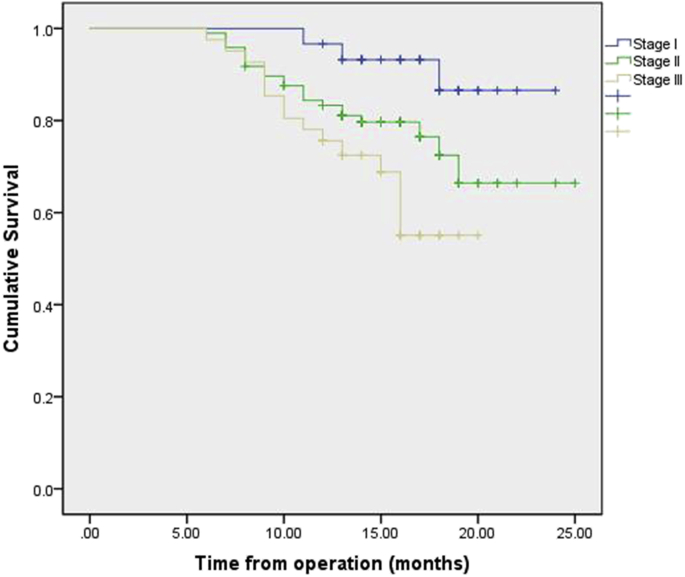
Follow-up was 100% complete in 251 patients. No patient was lost to follow-up. The median follow-up period was 15 months (range, 1–25 months). The overall survival rate at 1 year stratified by pathologic stage at esophagectomy was 95.9% (stage I), 83.8% (stage II), 73.4% (stage III). Survival by pathological stage is demonstrated in Fig. 2. Survival by pathological stage and operation procedures are demonstrated in Fig. 3, Fig. 4. Recurrence was mainly systemic in the form of liver and lung metastasis. At this writing, no patient has had any intrathoracic recurrence.
Fig. 2.
Kaplan–Meier plot of the estimated overall survival of patients stratified by stage.
Fig. 3.
Kaplan–Meier plot of the estimated overall survival of patients who accepted MIE-Ivor Lewis operation, stratified by stage.
Fig. 4.
Kaplan–Meier plot of the estimated overall survival of patients who accepted MIE-McKeown operation, stratified by stage.
4. Discussion
This experience has demonstrated that MIE can be performed safely without intraoperative mortality, with ICU stay rate of 2.0% (n = 5) and a median hospital stay of 17 days. MIE procedure was better than most published series of open esophagectomy [5]. In our comparison of short-term and long-term outcomes between the MIE-chest and MIE-neck groups, the mean operative time of McKeown MIE group is significant shorter than the Ivor Lewis MIE group (p = 0.013). However, the occurrence of anastomotic leak, anastomotic stenosis and RLN injury in the McKeown MIE group was significant higher than those in the Ivor Lewis MIE group. These findings are consistent with the first prospective multicenter study of MIE [7].
In the present study, we choose the approach of MIE according to the location of the tumor. Ivor-Lewis MIE approach is used for the treatment of mid and lower esophageal cancers, and with fewer complications such as RLN injury, anastomosis leak and anastomosis stenosis. These findings are consistent with other reported studies [24], [12], [25], [26], [27], [28]. The lower rate of anastomosis leak in the MIE Ivor-Lewis group mainly due to the reduced tension at the anastomosis and the ability to remove the potentially ischemic gastric tip. However, Ivor-Lewis MIE is technically demanding and requires extensive experience.
For cancer arising in the upper third of the esophagus, MIE McKeown approach was performed in our study. The main cause is the intrathoracic anastomosis and the embedding of the staple line in the MIE Ivor-Lewis group are more complex and waste more time. Significantly, more lymph nodes were resected in patients who underwent McKeown MIE in the present study (P < 0.05). The extent of lymph node dissection required for patients with esophageal cancer also remains controversial [29], [30]. However, adequate lymph node sampling is required for accurate staging [30]. One of the potential advantages of the McKeown MIE approach is better exposure and improved cervical lymphadenectomy. Esophagectomy is a complex and technically challenging operation. Specialty training of the surgeon and surgeon volume has an impact on clinical outcomes. In the current study, the operative time was significantly different between two groups (322.8 ± 50.5vs. 296.1 ± 35.9) because the intrathoracic anastomosis and the embedding of the staple line in the MIE Ivor-Lewis group were more complex and wasted more time. Intrathoracic anastomosis is technically demanding and learning curve is relatively long. McKeown approach avoids the most difficult problem of intrathoracic anastomosis. Cervical anastomosis is relatively simple and easy to operate. Thus, the MIE McKeown approach is more convenient and easier to grasp for the beginners.
In the reports of experienced centers, open esophagectomy approaches are frequently associated with significant morbidity and a mortality rate in the range of 6–23% [31], [32]. In one study of 1777 patients undergoing open esophagectomy, the most frequent reported complications were pneumonia (21%), respiratory failure (16%) and prolonged ventilator support (22%) [33]. Given concerns over this high morbidity and mortality, minimally invasive esophagectomy has been performed with increasing frequency. Our mortality of 0.4% and the low incidence of pneumonia (7.17%) and RLN injury (1.2%) suggest an advantage for the MIE.
At a median follow-up of 15 months, no significant difference in survival was noted between the two groups of patients. The overall survival rate at 1 year stratified by pathologic stage at esophagectomy was 95.9% (stage I), 83.8% (stage II), 73.4% (stage III). Furthermore, the oncological outcomes in the current study were good with an estimated 1-year overall survival of 81.7%. The estimated overall survival is acceptable when compared with published series of open esophagectomy. These results suggest that MIE can provide equivalent oncological outcomes as compared with open transthoracic esophagectomy.
4.1. Strength and limitations
The study is prospective trial designed to investigate the effects of MIE approach according to the location of tumor and compare the clinical outcomes between the Ivor Lewis and McKeown MIE procedures. However, there are several limitations in this study. Firstly, the main problem is the relative low proportion of upper third of the esophagus, thus may limited the statistical power. Secondly, the optimal approach to esophagectomy is controversial. The selection of MIE approach is lack of standard and is mainly based on the location of the tumor. Thirdly, the median follow-up period was short and lack of exploration of the long-term effects, especially on quality of life. Longer follow-up period is required to fully evaluate the oncologic results of MIE. it is a single-institution study. Largescale, prospective multicenter trials are required to fully judge the long-term results of MIE.
Ethical approval
The study was approved by the Ethics Committee of Chinese PLA General Hospital, Beijing, China.
Sources of funding
This work was supported by a general financial grant from the China Postdoctoral Science Foundation (2012T50865, to L. Gao.).
Author contribution
Chen L, Gao LG, Wang R, Liu X and Gao DW contributed to the study design, data collections.
Chen L, Zhang T, Liu X contributed to the data analysis, writing.
Conflicts of interest
The authors declare no conflict of interest.
Guarantor
Gao DW.
Research registration unique identifying number (UIN)
Researchregistry1949.
Acknowledgment
This work was supported by a general financial grant from the China Postdoctoral Science Foundation (2012T50865, to L. Gao.).
Contributor Information
Dewei Gao, Email: gaodeweigdw@163.com.
Linggen Gao, Email: gaolinggen@163.com.
References
- 1.McAnena O.J., Rogers J., Williams N.S. Right thoracoscopically assisted oesophagectomy for cancer. Br. J. Surg. 1994;81:236–238. doi: 10.1002/bjs.1800810225. [DOI] [PubMed] [Google Scholar]
- 2.Law S., Fok M., Chu K.M., Wong J. Thoracoscopic esophagectomy for esophageal cancer. Surgery. 1997;122:8–14. doi: 10.1016/s0039-6060(97)90257-9. [DOI] [PubMed] [Google Scholar]
- 3.Yerokun B.A., Sun Z., Jeffrey Yang C.F., Gulack B.C., Speicher P.J., Adam M.A. Minimally invasive versus open esophagectomy for esophageal cancer: a population-based analysis. Ann. Thorac. Surg. 2016;102:416–423. doi: 10.1016/j.athoracsur.2016.02.078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ayiomamitis A. Epidemiology of cancer of the esophagus in Canada: 1931-1984. Gastroenterology. 1988;94:374–380. doi: 10.1016/0016-5085(88)90424-6. [DOI] [PubMed] [Google Scholar]
- 5.Bailey S.H., Bull D.A., Harpole D.H., Rentz J.J., Neumayer L.A., Pappas T.N. Outcomes after esophagectomy: a ten-year prospective cohort. Ann. Thorac. Surg. 2003;75:217–222. doi: 10.1016/s0003-4975(02)04368-0. discussion 22. [DOI] [PubMed] [Google Scholar]
- 6.Birkmeyer J.D., Siewers A.E., Finlayson E.V., Stukel T.A., Lucas F.L., Batista I. Hospital volume and surgical mortality in the United States. N. Engl. J. Med. 2002;346:1128–1137. doi: 10.1056/NEJMsa012337. [DOI] [PubMed] [Google Scholar]
- 7.Luketich J.D., Pennathur A., Franchetti Y., Catalano P.J., Swanson S., Sugarbaker D.J. Minimally invasive esophagectomy: results of a prospective phase II multicenter trial-the eastern cooperative oncology group (E2202) study. Ann. Surg. 2015;261:702–707. doi: 10.1097/SLA.0000000000000993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Luketich J.D., Alvelo-Rivera M., Buenaventura P.O., Christie N.A., McCaughan J.S., Litle V.R. Minimally invasive esophagectomy: outcomes in 222 patients. Ann. Surg. 2003;238:486–494. doi: 10.1097/01.sla.0000089858.40725.68. discussion 94–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Irino T., Tsai J.A., Ericson J., Nilsson M., Lundell L., Rouvelas I. Thoracoscopic side-to-side esophagogastrostomy by use of linear stapler-a simplified technique facilitating a minimally invasive Ivor-Lewis operation. Langenbecks Arch. Surg. 2016;401:315–322. doi: 10.1007/s00423-016-1396-1. [DOI] [PubMed] [Google Scholar]
- 10.Sihag S., Kosinski A.S., Gaissert H.A., Wright C.D., Schipper P.H. Minimally invasive versus open esophagectomy for esophageal cancer: a comparison of early surgical outcomes from the society of thoracic surgeons national database. Ann. Thorac. Surg. 2016;101:1281–1289. doi: 10.1016/j.athoracsur.2015.09.095. [DOI] [PubMed] [Google Scholar]
- 11.Xiao P., Zhuang X., Shen Y., Li Q., Dai W., Yang X.J. Reverse-puncture anastomotic technique for minimally invasive Ivor-Lewis esophagectomy. Ann. Thorac. Surg. 2015;100:2372–2375. doi: 10.1016/j.athoracsur.2015.04.140. [DOI] [PubMed] [Google Scholar]
- 12.Luketich J.D., Pennathur A., Awais O., Levy R.M., Keeley S., Shende M. Outcomes after minimally invasive esophagectomy: review of over 1000 patients. Ann. Surg. 2012;256:95–103. doi: 10.1097/SLA.0b013e3182590603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xie M.R., Liu C.Q., Guo M.F., Mei X.Y., Sun X.H., Xu M.Q. Short-term outcomes of minimally invasive Ivor-Lewis esophagectomy for esophageal cancer. Ann. Thorac. Surg. 2014;97:1721–1727. doi: 10.1016/j.athoracsur.2014.01.054. [DOI] [PubMed] [Google Scholar]
- 14.Kim K., Park J.S., Seo H. Early outcomes of video-assisted thoracic surgery (VATS) Ivor Lewis operation for esophageal squamous cell carcinoma: the extracorporeal anastomosis technique. Surg. Laparosc. Endosc. Percutan Tech. 2013;23:303–308. doi: 10.1097/SLE.0b013e31828b8841. [DOI] [PubMed] [Google Scholar]
- 15.Mu J., Yuan Z., Zhang B., Li N., Lyu F., Mao Y. Comparative study of minimally invasive versus open esophagectomy for esophageal cancer in a single cancer center. Chin. Med. J. Engl. 2014;127:747–752. [PubMed] [Google Scholar]
- 16.Palanivelu C., Prakash A., Senthilkumar R., Senthilnathan P., Parthasarathi R., Rajan P.S. Minimally invasive esophagectomy: thoracoscopic mobilization of the esophagus and mediastinal lymphadenectomy in prone position–experience of 130 patients. J. Am. Coll. Surg. 2006;203:7–16. doi: 10.1016/j.jamcollsurg.2006.03.016. [DOI] [PubMed] [Google Scholar]
- 17.Lazzarino A.I., Nagpal K., Bottle A., Faiz O., Moorthy K., Aylin P. Open versus minimally invasive esophagectomy: trends of utilization and associated outcomes in England. Ann. Surg. 2010;252:292–298. doi: 10.1097/SLA.0b013e3181dd4e8c. [DOI] [PubMed] [Google Scholar]
- 18.Verhage R.J., Hazebroek E.J., Boone J., Van Hillegersberg R. Minimally invasive surgery compared to open procedures in esophagectomy for cancer: a systematic review of the literature. Minerva Chir. 2009;64:135–146. [PubMed] [Google Scholar]
- 19.Bencini L., Moraldi L., Bartolini I., Coratti A. Esophageal surgery in minimally invasive era. World J. Gastrointest. Surg. 2016;8:52–64. doi: 10.4240/wjgs.v8.i1.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zaninotto G., Portale G., Costantini M., Rizzetto C., Salvador R., Rampado S. Minimally invasive enucleation of esophageal leiomyoma. Surg. Endosc. 2006;20:1904–1908. doi: 10.1007/s00464-005-0838-6. [DOI] [PubMed] [Google Scholar]
- 21.Mei X., Xu M., Guo M., Xie M., Liu C., Wang Z. Minimally invasive Ivor-Lewis oesophagectomy is a feasible and safe approach for patients with oesophageal cancer. ANZ J. Surg. 2015 doi: 10.1111/ans.13161. [DOI] [PubMed] [Google Scholar]
- 22.Wee J.O. Minimally invasive oesophagectomy: the Ivor Lewis approach. Multimed. Man. Cardiothorac. Surg. 2015;2015 doi: 10.1093/mmcts/mmv034. pii: mmv034. [DOI] [PubMed] [Google Scholar]
- 23.Chen B., Zhang B., Zhu C., Ye Z., Wang C., Ma D. Modified McKeown minimally invasive esophagectomy for esophageal cancer: a 5-year retrospective study of 142 patients in a single institution. PLoS One. 2013;8:e82428. doi: 10.1371/journal.pone.0082428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhai C., Liu Y., Li W., Xu T., Yang G., Lu H. A comparison of short-term outcomes between Ivor-Lewis and McKeown minimally invasive esophagectomy. J. Thorac. Dis. 2015;7:2352–2358. doi: 10.3978/j.issn.2072-1439.2015.12.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ai B., Zhang Z., Liao Y. Laparoscopic and thoracoscopic esophagectomy with intrathoracic anastomosis for middle or lower esophageal carcinoma. J. Thorac. Dis. 2014;6:1354–1357. doi: 10.3978/j.issn.2072-1439.2014.07.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li H., Hu B., You B., Miao J.B., Fu Y.L., Chen Q.R. Combined laparoscopic and thoracoscopic Ivor Lewis esophagectomy for esophageal cancer: initial experience from China. Chin. Med. J. Engl. 2012;125:1376–1380. [PubMed] [Google Scholar]
- 27.Zhang R., Kang N., Xia W., Che Y., Wan J., Yu Z. Thoracoscopic purse string technique for minimally invasive Ivor Lewis esophagectomy. J. Thorac. Dis. 2014;6:148–151. doi: 10.3978/j.issn.2072-1439.2013.12.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shen G., Pan S.B., Wu M., Zhang S., Xu X.F., Chen G. Use of efficient purse-string stapling technique for esophagogastric anastomosis in minimally invasive Ivor Lewis esophagectomy. J. Thorac. Dis. 2013;5:898–901. doi: 10.3978/j.issn.2072-1439.2013.12.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lerut T., Nafteux P., Moons J., Coosemans W., Decker G., De Leyn P. Three-field lymphadenectomy for carcinoma of the esophagus and gastroesophageal junction in 174 R0 resections: impact on staging, disease-free survival, and outcome: a plea for adaptation of TNM classification in upper-half esophageal carcinoma. Ann. Surg. 2004;240:962–972. doi: 10.1097/01.sla.0000145925.70409.d7. discussion 72–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rizk N., Venkatraman E., Park B., Flores R., Bains M.S., Rusch V. The prognostic importance of the number of involved lymph nodes in esophageal cancer: implications for revisions of the American Joint Committee on Cancer staging system. J. Thorac. Cardiovasc Surg. 2006;132:1374–1381. doi: 10.1016/j.jtcvs.2006.07.039. [DOI] [PubMed] [Google Scholar]
- 31.Kelsen D.P., Ginsberg R., Pajak T.F., Sheahan D.G., Gunderson L., Mortimer J. Chemotherapy followed by surgery compared with surgery alone for localized esophageal cancer. N. Engl. J. Med. 1998;339:1979–1984. doi: 10.1056/NEJM199812313392704. [DOI] [PubMed] [Google Scholar]
- 32.Birkmeyer J.D., Siewers A.E., Finlayson E.V.A., Stukel T.A., Lucas F.L., Batista Hospital volume and surgical mortality in the United States. N. Engl. J. Med. 2002;346:1128–1137. doi: 10.1056/NEJMsa012337. [DOI] [PubMed] [Google Scholar]
- 33.Bailey S.H., Bull D.A., Harpole D.H., Rentz J.J., Neumayer L.A., Pappas T.N. Outcomes after esophagectomy: a ten-year prospective cohort. Ann. Thorac. Surg. 2003;75:217–222. doi: 10.1016/s0003-4975(02)04368-0. [DOI] [PubMed] [Google Scholar]