Abstract
Several expanded-spectrum β-lactamase blaCTX-M genes are associated with ISEcp1-like elements in Enterobacteriaceae. We found that ISEcp1B was able to mobilize the adjacent blaCTX-M-19 gene by a transpositional mechanism in Escherichia coli by recognizing a variety of DNA sequences as right inverted repeats.
An increasing number of reports of expanded-spectrum β-lactamases of the CTX-M type and plasmid-mediated cephalosporinases of the AmpC type in Enterobacteriaceae raise the question of their acquisition (5, 24, 26). The blaCTX-M genes belonging to three out of the five known clusters (the CTX-M-1, -M-2, and -M-9 clusters) are associated with ISEcp1-like insertion sequences (IS) (2, 5-7, 11, 12, 18, 21, 23). Nevertheless, the distance that separates the β-lactamase gene from ISEcp1 varies within a given cluster of CTX-M-type genes, indicating that different insertion events have occurred (13, 19). Analysis of GenBank databases reveals that some plasmid-encoded cephalosporinase genes, such as the blaCMY-type genes, may also be associated with ISEcp1 (3, 10, 17).
ISEcp1 is an IS weakly related to other IS elements and belonging to the IS1380 family (IS Database HomePage [http://www-is.biotoul.fr/page-is.html]) (8). Nothing is known about the mobilization properties of ISEcp1. Recently, we characterized ISEcp1B associated with a blaCTX-M-19 gene in a Klebsiella pneumoniae ILT-3 clinical isolate from Vietnam (21). ISEcp1B differs from ISEcp1 (GenBank accession no. AJ242809) by three nucleotide substitutions. Their inverted repeat (IR) sequences are identical, and their transposases differ by a single amino acid change (18). Since ISEcp1-like elements are located upstream of several β-lactamase genes, analysis of the variable sequences separating these IS elements from the initiation codons of these genes allowed us to determine its boundaries.
Recently, it has been shown that ISEcp1B brought promoter sequences for high-level expression of the blaCTX-M-14/-18, blaCTX-M-17, and blaCTX-M-19 β-lactamase genes that belong to the same blaCTX-M-9 subgroup (5, 6, 18) and might be also responsible for their mobilization. The aim of this study was to analyze the mobilization process mediated by ISEcp1B.
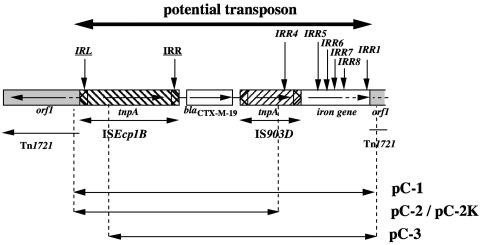
ISEcp1B possesses two imperfect IRs likely made of 14 bp, previously estimated to be 18 bp (18), with 12 of these 14 bp being complementary (Table 1). A detailed analysis of a ca. 9.5-kb DNA fragment from natural plasmid pILT-3 from K. pneumoniae ILT-3 revealed that the gene encoding ORF1 in transposon Tn1721 was interrupted by a 4.8-kb potential transposon bracketed on the left side by the left IR (IRL) of ISEcp1B and on the right side by an imperfect right IR (IRR) named IRR1 made of a 14-bp sequence belonging to a gene encoding a putative iron transport protein (Fig. 1) (18). This potential transposon comprises ISEcp1B, blaCTX-M-19, IS903D, and part of the gene encoding an iron transport protein. We have hypothesized that ISEcp1B could mobilize adjacent sequences by transposition.
TABLE 1.
Sequences identified as IRR boundaries for ISEcp1B-mediated transposition
| Description of sequence | Nucleotide sequence (5′→3′)a | No. of base pairs identical to perfect IRRb | Size of transposed fragment (bp) |
|---|---|---|---|
| IRL of ISEcp1B | GATTCTACGTCAGT | ||
| Deduced perfect IRR of ISEcp1B | ACGTAGAATCTAGG | ||
| IRR of ISEcp1B | ACGTGGAATTTAGG | 12 | |
| IRR1 at the potential transposon end of pILT-3 | GCGTTGATTCCTGG | 9 | 4,798 |
| IRR2 in plasmid pACYC184 | GCATGGATTGTAGG | 9 | 4,726 |
| IRR3 in plasmid pBK-CMV | CCACAGAATCAGGG | 9 | 4,541 |
| IRR4 in the tnpA gene of IS903D | GATGAACTGCGGCG | 3 | 3,273 |
| IRR5 in the iron gene of pILT-3 | GATGTGGAACCTCG | 4 | 3,817 |
| IRR6 in the iron gene of pILT-3 | GCACCGGGATGGCG | 3 | 4,167 |
| IRR7 in the iron gene of pILT-3 | GCGCCGCCGCGGGG | 6 | 4,273 |
| IRR8 in the iron gene of pILT-3 | GGGGTCGAACTGGG | 6 | 4,283 |
Underlined nucleotides are nucleotides identical to those identified at the same positions in the deduced IRR of ISEcp1B.
A perfect IRR contains 14 bp.
FIG. 1.
Schematic map of the potential transposon structure in natural plasmid pILT-3 of K. pneumoniae ILT-3 (13). Open reading frames and genes are shown as boxes with an arrow indicating the orientation of each coding sequence and the gene name shown under the corresponding box. IRL and IRR motifs are indicated by vertical arrows. The IRL and IRR extremities of the ISEcp1B-mobilized fragments are in italics, whereas those of the ISEcp1B element are underlined. The sequences cloned in recombinant plasmids are indicated by double-headed arrows with the corresponding recombinant plasmid names.
Several transposition experiments were performed to determine (i) the mobility of this potential transposon, (ii) the role of ISEcp1B in mobilizing the adjacent β-lactamase gene, and (iii) the role of IS903D. Different recombinant plasmids were constructed by inserting PCR products that encompassed either one or both ISs that bracketed the blaCTX-M-19 gene in plasmid pILT-3 into the EcoRV site of plasmid pACYC184 (Table 2) and then transformed into Escherichia coli reference strain DH10B. Recombinant plasmids were constructed with the pACYC184 backbone to prevent overexpression of the ISEcp1B transposase. Plasmid pC-1 contained the sequences of the potential transposon identified in plasmid pILT-3, whereas the following constructs lacked the right part of it (Fig. 1). Plasmid pC-2 possessed the ISEcp1B sequence but lacked part of IS903D, and plasmid pC-3 possessed the entire IS903D sequence and a truncated ISEcp1B transposase gene (Fig. 1; Table 2). E. coli RR1023 (pOX38) was transformed by electroporation with these recombinant plasmids (Table 2) (9). Plasmid pOX38 is a self-conjugative and IS-free plasmid. Transposition events were searched between inserts of several recombinant plasmids and recipient plasmid pOX38 after 24 h of growth in Trypticase soy broth. Transfer of the recombinant plasmids with the pOX38 backbone into streptomycin-resistant E. coli reference strain DH10B (Table 2) was then performed by conjugation as described previously (20), and transconjugants were selected on agar plates containing 100 μg of amoxicillin per ml (plasmid resistance marker) and 20 μg of streptomycin per ml (chromosomal marker). Several amoxicillin-resistant transconjugants were extracted and sequenced in part.
TABLE 2.
Strains and plasmids used in this study
| Strain or plasmid | Relevant genotype or phenotype | Source or reference |
|---|---|---|
| Strains | ||
| E. coli RR1023 | pOX38 recA56; susceptible strain | 9 |
| E. coli DH10B | araD139 Δ(ara leu)7697 deoR endA1 galK1 galU nupG recA1 rpsL F′ mrcA Δ(mrr-hsdRMS-mrcBC)φ80dlacZΔM15 ΔlacX74; streptomycin resistant | Stratagene |
| K. pneumoniae ILT-3 | Extended-spectrum cephalosporin-resistant isolate | 18 |
| Plasmids | ||
| pOX38 | 55 kb, F derivative, conjugative | 9 |
| pACYC184 | Chloramphenicol resistant | New England Biolabs |
| pILT-3 | Natural ca. 50-kb plasmid of K. pneumoniae ILT-3 producing β-lactamase CTX-M-19 | 18 |
| pC-1 | Recombinant plasmid pACYC184 with a 4,877-bp PCR fragment (primers PreA [5′-GCAGGTCTTTTTCTGCTCC-3′] and PreB [5′-TTTCCGCAGCACCGTTTGC-3′]) encompassing the potential transposon identified in pILT-3 | This study |
| pC-2 | Recombinant plasmid pACYC184 with a 3,635-bp PCR fragment (primers PreA and IS903B [5′-CCGTAGCGGGTTGTGTTTTC-3′]) containing ISEcp1B, blaCTX-M-19 gene, and truncated IS903D | This study |
| pC-2K | Recombinant plasmid pBK-CMV with the same 3,635-bp PCR fragment inserted into pC-2 | This study |
| pC-3 | Recombinant plasmid pACYC184 with a 4,606-bp PCR fragment (primers ISEcp1A [5′-GCAGGTCTTTTTCTGCTCC-3′] and PreB) containing blaCTX-M-19 and IS903D but lacking part of ISEcp1B | This study |
Susceptibility to chloramphenicol was always observed in transconjugant strains, thus ruling out full integration (chloramphenicol resistance) of the recombinant plasmids derived from pACYC184 into plasmid pOX38. Detection of ISEcp1B and IS903D was performed using two sets of primers (primer ISEcp1A [Table 2] in combination with primer ISEcp1B [5′-TTTCCGCAGCACCGTTTGC-3′] and primer IS903A [5′-CACATGAAATCATCTGCGC-3′] in combination with primer IS903B [Table 2], respectively).
Amoxicillin-resistant transconjugants were not obtained when E. coli RR1023 (pC-3) lacking ISEcp1B (Fig. 1) was used as a donor, suggesting that the IS903D element located downstream of the β-lactamase gene did not mobilize this latter gene. On the other hand, amoxicillin-resistant transconjugants were obtained when E. coli RR1023 (pC-1) and E. coli RR1023 (pC-2) were used as donors with a transposition efficiency of ca. 10−7 per donor. These results indicated that ISEcp1B was essential for movement of the blaCTX-M-19 gene.
Using E. coli RR1023 (pC-1) as a donor, all amoxicillin-resistant transconjugants except one contained ISEcp1B and IS903D elements, with the blaCTX-M-19 gene and DNA sequences being located on the right part of IS903D in plasmid pILT-3 (Fig. 1). Surprisingly, none of the 11 transconjugants that were sequenced had the entire potential transposon identified in natural plasmid pILT-3. Nevertheless, the transposition mechanism mediated by ISEcp1B was evident, since 5-bp duplications were observed at the insertion sites in plasmid pOX38 (see below). These data suggested that the IRR sequences recognized by ISEcp1B were located inside the potential transposon. Analysis of the boundaries of the transposed fragments identified sequences, similar to that of the IRR of ISEcp1B and defined as IRR-like sequences, that had been recognized as such by the transposase of ISEcp1B during a mobilization process.
Five different IRRs (IRR4 to IRR8) (Table 1) were identified in five amoxicillin-resistant transconjugants that were analyzed, four of them being located in the gene encoding the iron transport protein and one of them being located in the tnpA gene of IS903D (Table 2; Fig. 1). The number of identical base pairs among the sequence defined as IRR varied from three to six, corresponding to less than 50%.
Analysis of E. coli RR1023 (pOX38) transconjugants resistant to amoxicillin, obtained with plasmid pC-2 as a donor, and thus lacking the sequence used as IRR in pILT-3, also showed that transposition had occurred using nearby sequences of pACYC184. The IRR-like sequences bracketing the transposed fragment in these transconjugants remained unchanged. They shared 9 of 14 bp of the perfect IRR sequence (Table 1) and corresponded to a segment belonging to the cloning vector. In order to rule out that the specific sequence of pACYC184 may play a role in this transposition process, the same inserted PCR product of pC-2 was cloned into the SmaI site of pBK-CMV, giving rise to pC-2K (Table 2; Fig. 1). Analysis of several amoxicillin-resistant transconjugants obtained with plasmid pC-2K as a donor also showed that transposition had occurred by involving sequences of pBK-CMV. The IRR-like sequences bracketing the transposed fragment in these transconjugants were always the same and shared 9 of the 14 bp of a perfect IRR (Table 1). These results indicated that sequences of pACYC184 (IRR2) or of pBK-CMV (IRR3) might be used as IRRs by ISEcp1B, but further repeated experiments are needed to assess this hypothesis. The reason why the IRR1 of the potential transposon of pILT-3 was not the boundary of the transposed fragments in these in vitro experiments remains intriguing. One explanation could be that IRR2 and IRR3 share greater nucleotide identity in their 3′ extremities (7 and 8, respectively, of last 10 bp) to a perfect IRR than IRR1 (only 6 of last 10 bp).
No consensus sequence could be determined by comparing these 14-bp-long IRR sequences of recombinant plasmids and the IRR sequences of ISEcp1B in pILT-3 (Table 1). Nevertheless, a guanosine residue located at the 3′ end of these IRRs was always found, likely indicating that this nucleotide was necessary in the transposition process. In addition, the last two nucleotides of these IRR sequences were either GG or CG. These data indicated that ISEcp1B was involved in a transposition process with weakly related IRRs.
Analysis of the insertion sites of ISEcp1B-mediated transposition revealed variable sequences in recombinant plasmids (AAGAA, TATAA, TATTA, GCTGA, ATATC, TAATA, TTTAC, AATGA, TTATA, ATAAG, and TGATT). No consensus sequence was identified among the 5-bp duplicated sites, whereas an A+T-rich content that may target ISEcp1-mediated transposition as observed for IS1 was noticed (16, 27). In silico analysis of six ISEcp1-like sequences associated with blaCTX-M-like or blaCMY-like β-lactamase genes available in the GenBank databases did not identify target site duplications just bracketing the insertion sites of ISEcp1-like sequences. This result likely indicates that after insertion, ISEcp1-like elements may be responsible for mobilization of the adjacent sequences. In addition, this in silico analysis confirmed that the 5-bp sequences adjacent to the IRLs and IRRs of ISEcp1 elements were always A+T rich.
ISEcp1B may use a wide range of DNA sequences as IRRs during a mobilization process that involves its adjacent sequences, therefore explaining its ability to mobilize structurally unrelated β-lactamase genes. Similar observations have been made for IS1247 of Xanthobacter autotrophicus, which also belongs to the IS1380 family (25). A single copy of IS1247 may mobilize sequences located at its right-end extremity in what is considered a one-ended transposition process since only a single IS is involved.
These results may also be compared to those obtained with other IS elements, such as IS91 and IS911, that are responsible for mobilization of adjacent sequences by a one-ended transposition mechanism (4, 22). In addition, transposition of other elements belonging to the Tn3 family, such as Tn21, generates variable endpoints as a consequence of one-ended transposition (1, 15). This peculiar process is the consequence of recognition of several different IR sequences by the Tn21 transposase, generating novel joint molecules (14). However, in our case, some homology between the different IRRs used by ISEcp1B is noticed, whereas one-ended transposition (for example, with Tn21) requires one fixed end and another random one.
In the present study, we have demonstrated the ability for ISEcp1B to mobilize the blaCTX-M β-lactamase gene. In addition, the IS903D element present in the potential transposon in pILT-3 had no role in the mobilization process.
In conclusion, it remains to be determined why the emerging expanded-spectrum β-lactamases of the CTX-M type that are structurally related to several chromosomally located β-lactamase genes of enterobacterial species are associated specifically with ISEcp1-like sequences.
Acknowledgments
We thank Keith Derbyshire for giving us the E. coli RR1023 strain harboring plasmid pOX38 and for precious advice. We also thank Hesna Yigit for contributive discussion.
This work was financed by a grant from the Ministère de l'Education Nationale et de la Recherche (UPRES grant EA3539), Université Paris XI, Paris, France, and by the European Community (6th PCRD, LSHM-CT-2003-503-335). L.P. is a researcher from the INSERM, Paris, France.
REFERENCES
- 1.Avila, P., J. Grinsted, and F. De La Cruz. 1988. Analysis of the variable endpoints generated by one-ended transposition of Tn21. J. Bacteriol. 170:1350-1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baraniak, A., J. Fiett, W. Hryniewicz, P. Nordmann, and M. Gniadkowski. 2002. Ceftazidime-hydrolysing CTX-M-15 extended-spectrum β-lactamase (ESBL) in Poland. J. Antimicrob. Chemother. 50:393-396. [DOI] [PubMed] [Google Scholar]
- 3.Bauernfeind, A., I. Schneider, R. Jungwirth, H. Sahly, and U. Ullmann. 1999. A novel type of AmpC β-lactamase, ACC-1, produced by a Klebsiella pneumoniae strain causing nosocomial pneumonia. Antimicrob. Agents Chemother. 43:1924-1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bernales, I., M. V. Mendiola, and F. De La Cruz. 1999. Intramolecular transposition of insertion sequence IS91 results in second-site simple insertions. Mol. Microbiol. 33:223-234. [DOI] [PubMed] [Google Scholar]
- 5.Bonnet, R. 2004. Growing group of extended-spectrum β-lactamases: the CTX-M enzymes. Antimicrob. Agents Chemother. 48:1-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cao, V., T. Lambert, and P. Courvalin. 2002. ColE1-like plasmid pIP843 of Klebsiella pneumoniae encoding extended-spectrum β-lactamase CTX-M-17. Antimicrob. Agents Chemother. 46:1212-1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chanawong, A., F. H. M'Zali, J. Heritage, J. H. Xiong, and P. M. Hawkey. 2002. Three cefotaximases, CTX-M-9, CTX-M-13, and CTX-M-14, among Enterobacteriaceae in the People's Republic of China. Antimicrob. Agents Chemother. 46:630-637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chandler, M., and J. Mahillon. 2002. Insertion sequences revisited, p. 305-366. In N. L. Craig, R. Craigie, M. Gellert, and A. M. Lambowitz (ed.), Mobile DNA II, ASM Press, Washington, D.C.
- 9.Derbyshire, K. M., L. Hwang, and N. D. Grindley. 1987. Genetic analysis of the interaction of the insertion sequence IS903 transposase with its terminal inverted repeats. Proc. Natl. Acad. Sci. USA 84:8048-8053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Girlich, D., T. Naas, S. Bellais, L. Poirel, A. Karim, and P. Nordmann. 2000. Biochemical-genetic characterization and regulation of expression of an ACC-1-like chromosome-borne cephalosporinase from Hafnia alvei. Antimicrob. Agents Chemother. 44:1470-1478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karim, A., L. Poirel, S. Nagarajan, and P. Nordmann. 2001. Plasmid-mediated extended-spectrum β-lactamase (CTX-M-3-like) from India and gene association with insertion sequence ISEcp1. FEMS Microbiol. Lett. 201:237-241. [DOI] [PubMed] [Google Scholar]
- 12.Lartigue, M. F., L. Poirel, C. Héritier, V. Tolün, and P. Nordmann. 2003. First description of CTX-M-15-producing Klebsiella pneumoniae in Turkey. J. Antimicrob. Chemother. 52:315-316. [DOI] [PubMed] [Google Scholar]
- 13.Lartigue, M. F., L. Poirel, and P. Nordmann. 2004. Diversity of genetic environment of the blaCTX-M genes. FEMS Microbiol. Lett. 234:201-207. [DOI] [PubMed] [Google Scholar]
- 14.Liebert, C. A., R. M. Hall, and A. O. Summers. 1999. Transposon Tn21, flagship of the floating genome. Microbiol. Mol. Biol. Rev. 63:507-522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Martin, C., J. Grinsted, and F. de la Cruz. 1989. Effects of variation of inverted-repeat sequences on reactions mediated by the transposase of Tn21. J. Bacteriol. 171:3996-4001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Meyer, J., S. Iida, and W. Arber. 1980. Does the insertion element IS1 transpose preferentially into A+T rich DNA segments? Mol. Gen. Genet. 178:471-473. [DOI] [PubMed] [Google Scholar]
- 17.Nadjar, D., M. Rouveau, C. Verdet, L. Donay, J. Herrmann, P. H. Lagrange, A. Philippon, and G. Arlet. 2000. Outbreak of Klebsiella pneumoniae producing transferable AmpC-type β-lactamase (ACC-1) originating from Hafnia alvei. FEMS Microbiol. Lett. 187:35-40. [DOI] [PubMed] [Google Scholar]
- 18.Poirel, L., J. W. Decousser, and P. Nordmann. 2003. Insertion sequence ISEcp1B is involved in the expression and mobilization of a blaCTX-M β-lactamase gene. Antimicrob. Agents Chemother. 47:2938-2945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Poirel, L., M. Gniadkowski, and P. Nordmann. 2002. Biochemical analysis of the ceftazidime-hydrolysing extended-spectrum β-lactamase CTX-M-15 and of its structurally related β-lactamase CTX-M-3. J. Antimicrob. Chemother. 50:1031-1034. [DOI] [PubMed] [Google Scholar]
- 20.Poirel, L., M. Guibert, S. Bellais, T. Naas, and P. Nordmann. 1999. Integron- and carbenicillinase-mediated reduced susceptibility to amoxicillin-clavulanic acid in isolates of multidrug-resistant Salmonella enterica serotype typhimurium DT104 from French patients. Antimicrob. Agents Chemother. 43:1098-1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Poirel, L., T. Naas, I. Le Thomas, A. Karim, E. Bingen, and P. Nordmann. 2001. CTX-M-type extended-spectrum β-lactamase that hydrolyzes ceftazidime through a single amino acid substitution in the omega loop. Antimicrob. Agents Chemother. 45:3355-3361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Prère, M. F., M. Chandler, and O. Fayet. 1990. Transposition in Shigella dysenteriae: isolation and analysis of IS911, a new member of the IS3 group of insertion sequences. J. Bacteriol. 172:4090-4099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Saladin, M., V. T. Cao, T. Lambert, J. L. Donay, J. L. Herrmann, Z. Ould-Hocine, C. Verdet, F. Delisle, A. Philippon, and G. Arlet. 2002. Diversity of CTX-M β-lactamases and their promoter regions from Enterobacteriaceae isolated in three Parisian hospitals. FEMS Microbiol. Lett. 209:161-168. [DOI] [PubMed] [Google Scholar]
- 24.Tzouvelekis, L. S., E. Tzelepi, P. T. Tassios, and N. J. Legakis. 2000. CTX-M-type β-lactamases: an emerging group of extended-spectrum enzymes. Int. J. Antimicrob. Agents 14:137-140. [DOI] [PubMed] [Google Scholar]
- 25.Van der Ploeg, J., M. Willemsem, G. Van Hall, and D. B. Janssen. 1995. Adaptation of Xanthobacter autotrophicus GJ10 to bromoacetate due to activation and mobilization of the haloacetate dehalogenase gene by insertion element IS1247. J. Bacteriol. 177:1348-1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wu, S. W., K. Dornbusch, G. Kronvall, and M. Norgren. 1999. Characterization and nucleotide sequence of a Klebsiella oxytoca cryptic plasmid encoding a CMY-type β-lactamase: confirmation that the plasmid-mediated cephamycinase originated from the Citrobacter freundii AmpC β-lactamase. Antimicrob. Agents Chemother. 43:1350-1357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zerbib, D., P. Gamas, M. Chandler, P. Prentki, S. Bass, and D. Galas. 1985. Specificity of insertion of IS1. J. Mol. Biol. 185:517-524. [DOI] [PubMed] [Google Scholar]