Abstract
Many viruses require the host endoplasmic reticulum protein-folding machinery in order to correctly fold one or more of their glycoproteins. Iminosugars with glucose stereochemistry target the glucosidases which are key for entry into the glycoprotein folding cycle. Viral glycoproteins are thus prevented from interacting with the protein-folding machinery leading to misfolding and an antiviral effect against a wide range of different viral families. As iminosugars target host enzymes, they should be refractory to mutations in the virus. Iminosugars therefore have great potential for development as broad-spectrum antiviral therapeutics. We outline the mechanism giving rise to the antiviral activity of iminosugars, the current progress in the development of iminosugar antivirals and future prospects for this field.
Keywords: calnexin, drug discovery and design, glucosidase, glycobiology, iminosugar
Introduction
Enveloped viruses have a membrane surrounding their capsid, which is composed of lipids derived from host membranes and viral glycoproteins. These viral glycoproteins are typically dependent upon the host endoplasmic reticulum (ER) protein-folding machinery to form the three-dimensional structure necessary for secretion and/or activity [1–3]. Partial inhibition of the ER-folding mechanisms in order to prevent properly folded viral glycoproteins from being incorporated into the virus is therefore a promising target for the development of broad-spectrum antivirals [4,5].
That effective treatments for viral diseases are an urgent requirement has been highlighted by the recent outbreaks of Ebola and Zika. Whilst vaccines are considered the gold standard in this area, the production of effective vaccines can in some cases take many years and factors such as the presence of multiple viral serotypes, as seen for dengue virus (DENV), can pose an added challenge for vaccine development [6]. A complementary approach to vaccination is the development of antiviral drugs. Antiviral drugs fall into two classes: direct-acting drugs, which target one of the essential components of the virus itself, and host-directed drugs, which target a host process required for viral replication. The development of host-directed antivirals is challenging due to the need to ensure selective toxicity towards the virus; however, they avoid the problem of resistance mutations arising in the viral target, which can limit the efficacy of direct-acting antivirals (see, for example, recent reviews [7,8]).
The iminosugar class of compounds includes promising candidates for the development of host-directed antivirals (Figure 1). Iminosugars are sugar mimetics in which the cyclic oxygen is replaced with a nitrogen [9]. Their structural similarity to sugar molecules means that many iminosugars are competitive inhibitors of enzymes that act on sugar substrates. As such, their therapeutic potential has been explored against a range of diseases including Gaucher's disease for which the enzymatic target is glucosylceramide synthase [10,11], and type II diabetes where the targets are intestinal α-glucosidases [12]. Key host enzyme targets for antiviral activity are the ER α-glucosidases I and II (α-glu I and α-glu II), which control entry to the calnexin cycle, which is required for the folding of many glycoproteins [2,13–17]. Iminosugars that inhibit these enzymes have antiviral activity against a range of enveloped viruses including DENV [18,19], influenza virus [20,21], hepatitis C virus (HCV) [22,23] and human immunodeficiency virus [24,25]. Support for the approach of targeting α-glu I in antiviral development is provided by the observations that two patients deficient in this enzyme have no clinical evidence of recurrent viral infections and that cells derived from these patients have a greatly reduced ability to support replication of a variety of different viruses [26]. The α-glu I-deficient patients present with multiple neurologic complications; however, such effects are not expected from antiviral therapy targeting α-glu I as only partial inhibition of the enzyme is necessary for an antiviral effect. In addition, antiviral treatment will not be administered during early development where a lack of α-glu I may be more likely to influence neurologic parameters. The sensitivity of viruses to inhibition of α-glu I and II appears to exceed that of the host proteins, giving a potential therapeutic window in which an antiviral effect occurs without a detrimental effect on the host.
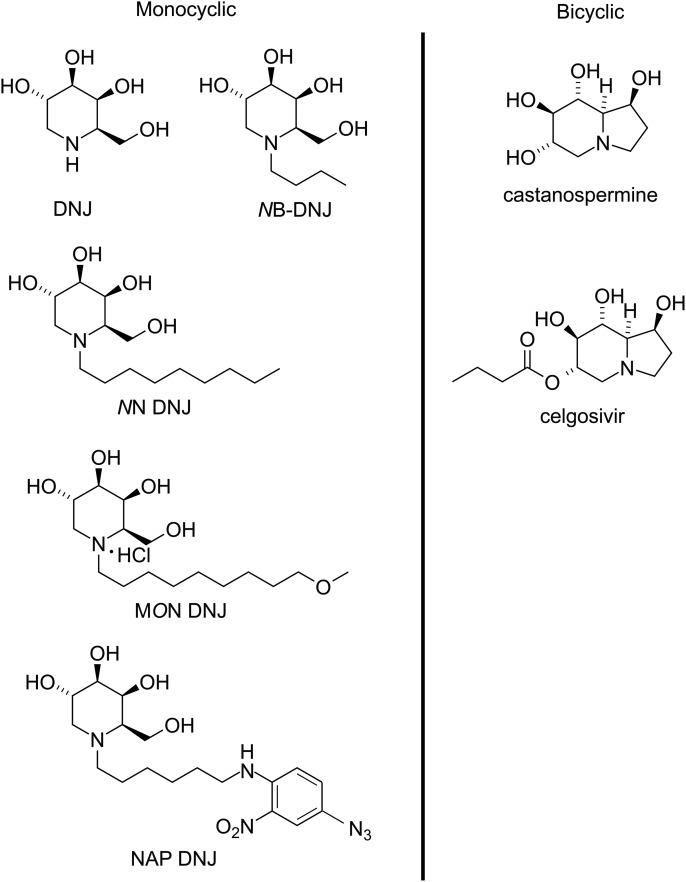
Figure 1. Structures of iminosugars discussed in this mini-review.
The left panel shows the structure of the naturally occurring iminosugar DNJ, along with derivatives modified to increase antiviral efficacy. MON-DNJ, currently in clinical trial against dengue, is also referred to as UV4B. The right panel shows the structure of the naturally occurring iminosugar CAST along with the prodrug celgosivir which is in a phase II clinical trial against dengue.
Mechanism of action
It is widely accepted that a key mechanism by which iminosugars act as antivirals is their ability to disrupt glycoprotein folding, arising from the inhibition of ER α-glu I and II. Early work on haemagglutinin (HA), an influenza envelope glycoprotein, extensively characterised the folding pathway of this protein. Experiments using the bicyclic iminosugar castanospermine (CAST) demonstrated that calnexin was required for efficient folding of HA and that the association with calnexin was dependent on the nature of the glycan [2,15,27,28]. The misfolded states of other viral proteins, such as HIV [29], have also been characterised and direct links observed between iminosugars, misfolding and the antiviral effect. However, in the majority of the cases the evidence for this mechanism of action is less direct; addition of iminosugar leads to impaired virion secretion or secretion of non-infectious virions. Here, we describe the calnexin cycle in more detail and briefly touch on other potential mechanisms of action which may be relevant in some viruses.
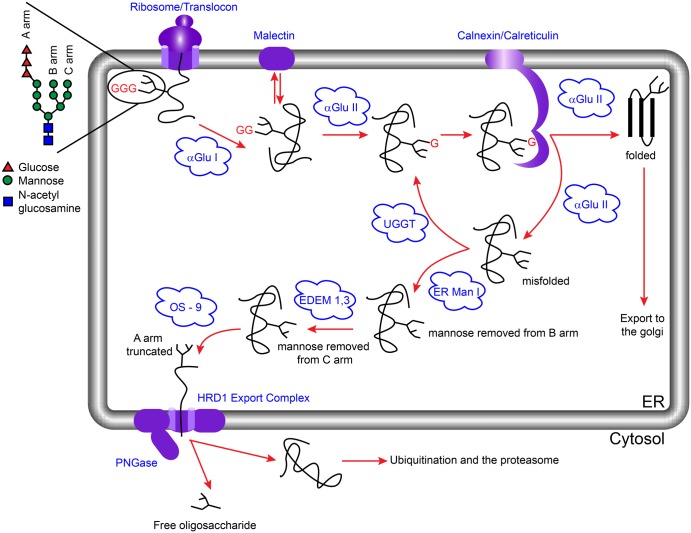
N-linked glycoproteins are co-translationally modified at the luminal face of the ER through en bloc addition of one or more precursor glycans in the form of Glc3Man9GlcNAc2 by oligosaccharyltransferase [30]. The glycans are recognised and processed by a variety of ER- and Golgi-resident factors that assist protein folding and assembly, mediate flow of secretory cargo and trigger ER-associated degradation (ERAD) [31,32]. Sequential cleavage of the two terminal glucose residues is important for interaction of the nascent polypeptide chain with calnexin, which forms a core part of the ER quality control (ERQC) mechanism [2,15,33,34]. ER α-glu I and α-glu II are the gatekeepers for the calnexin cycle, with binding to ERQC components dependent on the glycoform that the nascent polypeptide retains. ER α-glu I cleaves the terminal glucose residue of the N-linked glycan to give a Glc2Man9GlcNAc2 species. This diglucosylated glycan can be specifically bound by malectin, a membrane-bound ER-resident lectin [35]. Expression of malectin is induced by the unfolded protein response [36], and the protein is proposed to preferentially associate with non-native conformers of folding glycoproteins [37]. The glycan-bound form of malectin potentially associates with the translocon-associated oligosaccharyl transferase acting as an early pathway misfolding sensor [38]. Cleavage of the second glucose residue by α-glu II results in Glc1Man9GlcNAc2, which competes for binding with calnexin/calreticulin and α-glu II [33]. Binding by calnexin retains the protein in the ER where it can interact with chaperones such as binding immunoglobulin protein (BiP) and protein disulfide-isomerase (PDI) [34]. Binding to α-glu II results in cleavage of the third glucose residue after which there are several possible outcomes. If the protein is correctly folded, it can move to the Golgi apparatus for further processing of the glycans. If the protein is misfolded, this may be recognised by UDP-glucose:glycoprotein glucosyl transferase (UGGT), which reglucosylates the glycan such that the protein is once again a substrate for calnexin [39,40]; alternatively, the protein may encounter an α-mannosidase which removes a specific terminal mannose residue targeting the protein for degradation (Figure 2) [41,42].
Figure 2. The calnexin cycle and ERAD.
The precursor glycan Glc3Man9GlcNAc2 (represented here for simplicity with the glucose residues as red triangles and the remaining portion of the glycan shown as black lines) is added to a peptide co-translationally. Cleavage of the terminal glucose residue by α-glu I leads to a form that can either bind to malectin or be further trimmed by α-glu II to become a substrate for calnexin/calreticulin. On release from calnexin/calreticulin, α-glu II can remove the remaining glucose residue. At this point properly folded proteins are exported to the Golgi for further processing, whilst misfolded proteins are either reglucosylated by UGGT for a ‘second chance’ at folding or directed to the ERAD pathway by ER mannosidase I (ER Man I), which removes a mannose residue from the B-arm of the glycan [42,79]. ER degradation-enhancing α-mannosidase-like proteins 1–3 (EDEM1–3) then act on the C-arm of the glycan followed by OS-9/XTP3-B-mediated delivery of the substrate to the Hrd1 ubiquitination complex through the interaction with a membrane-spanning adaptor protein, SEL1L [80–87]. PNGase separates the glycan from the protein and both segments are degraded [44,88].
The presence of large quantities of misfolded proteins will trigger ERAD [32]. This pathway targets misfolded proteins for translocation from the ER into the cytosol, ubiquitination and subsequent hydrolysis by the proteasome. The ERAD targeting presumably occurs through a variety of mechanisms, depending on the nature of the substrate as well as the localisation of the misfolded region within the protein. Glycoproteins degraded through ERAD have their glycan portion released prior to the proteasomal destruction in the cytosol by a peptide:N-glycanase (PNGase) [43–45]. As a result, free oligosaccharides (FOS) are produced. These FOS are an excellent biomarker for ERAD and provide both qualitative and quantitative insight into glycoprotein degradation in the ER on a cellular level.
The FOS produced by PNGase initially carry two GlcNAc residues at the reducing terminus. The cytosolic pathway for FOS catabolism in normal conditions involves rapid processing by endo-β-N-acetylglucosaminidase (ENGase), which removes the terminal GlcNAc residue, resulting in a mono-GlcNAc form of FOS [46,47]. This is trimmed by a cytosolic neutral α-mannosidase (NAM, MAN2C1) and subsequently transported into the lysosome for further hydrolysis to monosaccharides [48–50]. Inhibition of α-glu I and II leads to the presence of glucosylated FOS [51,52]. These are processed in the cytosol to the final form of Glc3Man5GlcNAc1 (and slowly to Glc3Man4GlcNAc1; Figure 3). However, glucosylated FOS are unable to access the lysosome, resulting in their cytosolic accumulation [50]. The use of the ER glucosidase iminosugar inhibitor N-butyl-deoxynojirimycin (NB-DNJ) has demonstrated that the generation of glucosylated FOS is both dose- and treatment time-dependent. Analysing the level of glucosylated FOS in the cytosol is a valuable test for novel iminosugars in a cellular context [53]. This is particularly pertinent, as although there are in vitro assays for α-glucosidase inhibition, these do not address the question of cellular uptake. Entry of iminosugars into the ER needs to be achieved and demonstrated for these compounds to be developed for clinical trials.
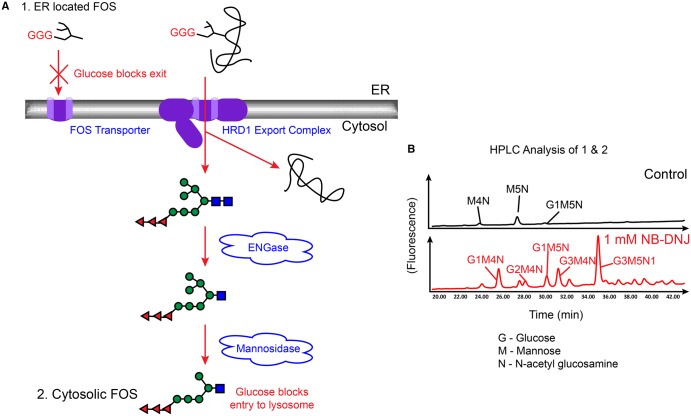
Figure 3. FOS analysis of cells grown in the presence of iminosugars.
(A) FOS are produced by the activity of two PNGase enzymes: one located in the ER, and the other in the cytosol. In the absence of iminosugar inhibitors FOS produced in the ER will be exported via a FOS transporter to the cytosol for degradation. The presence of terminal glucose residues on the A-arm of the glycan prevents export from the ER, leading to an increase in glucosylated FOS in the ER in the presence of iminosugars. Misfolded glycoproteins targeted for degradation through ERAD are trimmed by the enzymes ENGase and a cytosolic mannosidase. In the absence of iminosugars the resulting glycans enter the lysosome for further degradation; however, the presence of glucose residues prevents this leading to a build-up of glucosylated FOS in the cytosol. (B) Following isolation, purification and fluorescent labelling, total cellular FOS containing both the ER pool (1 in A) and cytosolic FOS pool (2 in A) can be analysed by NP-HPLC. Shown here are representative chromatograms of total cellular FOS from control HL60s cells (upper panel) and HSL60s cells treated with 1 mM NB-DNJ for 24 h (lower panel). The glucose capped glycans visible in the lower panel are indicative of α-glucosidase inhibition.
Using FOS to probe iminosugar mechanism of action
The calnexin cycle provides a simplistic picture of the current targets involved in inhibiting glycoprotein folding — namely the enzymes α-glu I and α-glu II. The sequential removal of glucose residues, the flux through the ER and the multiple equilibria (between enzymes, inhibitors and competing substrates) involved in the processing of the glycoprotein make it difficult to predict the extent to which each of the two enzymes will be inhibited for any given concentration of iminosugar. We might have expected, for example, that inhibition of α-glu I, which iminosugars such as NB-DNJ inhibit at least 10 times more strongly than α-glu II (Table 1), would dominate. However, FOS analysis shows that, at physiologically significant iminosugar concentrations, where an antiviral effect is observed, the result of all the competing reactions and processes is that inhibition of the α-glu II enzyme at the mono-glucosylated stage is key, and that higher concentrations of iminosugars are needed to inhibit predominately α-glu I [53].
The FOS assay carried out on cells incubated with NB-DNJ revealed an initial build-up of mono-glucosylated FOS at short treatment periods and low concentrations of iminosugar, before triglucosylated FOS are generated [53]. This reveals the potent cellular inhibition of α-glu II, compared with α-glu I, and contrasts markedly with the inhibition of these enzymes using in vitro assays, where, on the basis of IC50 values, NB-DNJ is 100 times more efficient at inhibiting α-glu I than II. This result is mirrored by more potent α-glu I inhibitors such as NAP-DNJ, which inhibits at low nanomolar concentrations (Table 1) [54]. The very small amount of diglucosylated FOS produced by iminosugar inhibition in the cell demonstrates that iminosugars are relatively poor at preventing the removal of the first α1,2-linked glucose. This reflects the kinetics of α-glu I and II action in the ER, which results in the efficient and rapid hydrolysis of tri- and di-glucosylated glycans to mono-glucosylated glycans, allowing interaction with calnexin/calreticulin chaperones.
The longer half-life of mono-glucosylated glycans, compared with that of tri- and di-glucosylated species, is due to the slower hydrolysis rate of the proximal glucose residue by α-glu II [55]. This contributes to a more favourable environment for iminosugar inhibition of this step. Removal of the first glucose residue by α-glu I and the second glucose residue by α-glu II in cultured cells probably occurs close to their limiting rates (Vmax), where addition of a competitive inhibitor has a limited effect on the observed rate. Removal of the third glucose residue by α-glu II is much slower, suggesting that this rate is not close to its Vmax, and under such conditions a competitive inhibitor has a greater effect on the rate. The calnexin molecule acts as an anchor, while chaperones, such as BiP and PDI, help with the folding process. Since α-glucosidase II and calnexin both compete for the same substrate in the ER, Glc1Man9GlcNac2-protein, if α-glu II is unable to hydrolyse the substrate bound to calnexin, the presence of calnexin will significantly reduce the free substrate concentration, hence reducing the rate. Alternatively, if α-glu II was able to hydrolyse the substrate bound to calnexin (although such a scenario is difficult to envisage), then the presence of calnexin is likely to change the Km. An increase in Km would also result in a reduced rate. Both possibilities have the same functional outcome: the rate of removal of the proximal glucose residue is reduced in cells and hence is more sensitive to the presence of a competitive inhibitor, resulting in a greater accumulation of mono-glucosylated glycans. The retention of the mono-glucosylated form by inhibition of α-glu II by an iminosugar will result in the glycoprotein failing to pass the quality control and it being targeted for degradation in the ER. It is important to note that not all glycoproteins require the calnexin cycle to fold correctly. The Glc1Man9GlcNAc2 glycan exists in a dynamic equilibrium between two structures, a major and a minor conformer for the Glcα1–3Manα linkage [56]. Modelling of the binding of Glc1Man9GlcNAc2 to calnexin suggests that it is the minor conformer that is recognised by calnexin. This may be one of the mechanisms for controlling the rate of recruitment of proteins into the calnexin/calreticulin chaperone system and enabling proteins that do not use this particular quality control mechanism for folding to bypass the system.
Inhibition of glycolipid processing
Although it is clear that inhibition of ER α-glu I and/or II has an antiviral effect against many viruses, this may not be the whole story. Monocyclic compounds such as 1-deoxynojirimycin (DNJ) and its derivatives are also inhibitors of glycolipid processing enzymes [52]. Glycolipids play an important role in the life-cycle of many viruses; neutral sphingolipids are thought to be important for the binding of DENV to mammalian and mosquito cell surfaces [57,58], whilst glycosphingolipids are a major component of the lipid rafts necessary for the replication of HCV [59,60]. It is therefore possible that the antiviral effects of iminosugars against these viruses arise from inhibition of glycolipid processing in addition to inhibiting glycoprotein folding. The extent to which inhibition of glycolipid processing contributes to antiviral effects will almost certainly be virus-dependent. Recently, Sayce et al. demonstrated that, in the case of DENV, the antiviral effects arise via inhibition of glycoprotein folding and that iminosugar effects on glycolipid processing do not contribute to the antiviral effect [61,62]. For HCV and other viruses the picture is less clear and investigations in this area are ongoing.
Cellular targets visualised by structural biology
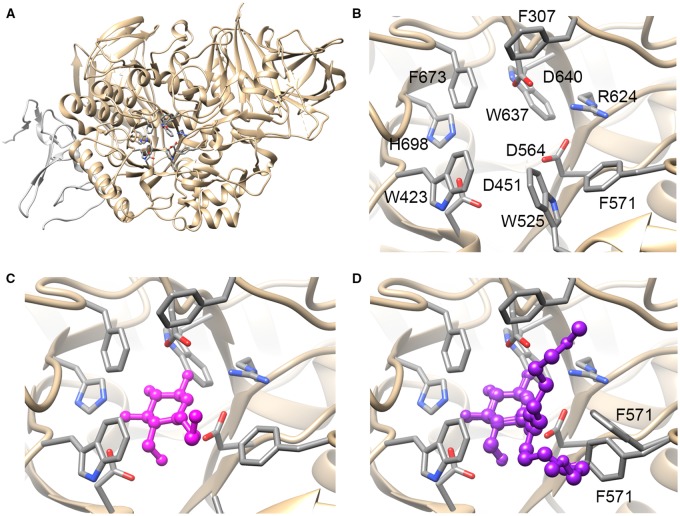
It is clear that both α-glucosidases are important antiviral targets. Recent structural biology approaches have attempted to examine their structures to both understand the mechanism of inhibition and provide insights into the design and evaluation of more selective and potent inhibitors [63–65]. Inhibition is based on a DNJ structure, which has glucose stereochemistry, and hence, with the abundance of glucose usage in the cell, the chances of multiple binding partners for the drugs are high. Indeed, DNJ compounds with alkyl chains have shown efficacy against enzymes including ER α-glucosidases, gut α-glucosidases, ceramide glucosyl transferase Β and glucocerebrosidases (GBA 1, 2 and 3). The 2.04 Å crystal structure of ER α-glu I determined in 2013 provides clear indication of the catalytic residues, but unfortunately the crystal packing prohibits experimental active-site investigations due to occlusion of the active site by a His6 purification tag from a crystal contact [63]. More recently, Caputo et al. [64] have solved the structure of mouse α-glu II (Mmα-GluII) in the presence of a series of substrate analogues, inhibitors and products (Figure 4). When compared with the structure of an intestinal α-glucosidase, the structure has revealed some crucial differences including an ‘exclusion loop’ unique to α-glu II that hangs over the active site. Generating compounds that bind to the exclusion loop region may therefore lead to increased specificity for ER α-glu II over the intestinal α-glucosidases. In addition, crystal structures with inhibitors gave insight into potential methods for designing inhibitors with increased affinity. Alkylated DNJ derivatives, for example, occupy the site of the terminal glucose residue as expected, but the extent to which they occupy the binding sites for the adjacent sugar moieties depends upon the length of the alkyl chain. NB-DNJ, with its four carbon alkyl chain, occupies the +1 sugar-binding site, adjacent to the terminal glucose site, whilst MON-DNJ with its much longer chain extends towards the +2 sugar-binding site. The co-crystal structure with MON-DNJ suggests that the aromatic residues forming a ring between the +1 and +2 site would be good targets for inhibitors with increased potency (Figure 4C,D).
Figure 4. The structure of α-glu II; active site and in complex with inhibitors.
(A) Ribbon diagram illustrating the structure of α-glu II with the key catalytic residues shown as sticks. The catalytic subunit is shown in gold, whilst the fragment of the accessory subunit remaining after protease digestion is shown in silver. (B) The catalytic binding site of α-glu II. Key residues for catalytic activity and interaction with the substrate glycan are shown as sticks. In the catalytic cycle D564 acts as a nucleophile and D640 as the catalytic acid/base. As is typical for sugar-binding sites, there are many aromatic residues and polar groups offering a wide range of potential interactions for inhibitors. (C) The catalytic binding site of α-glu II with NB-DNJ shown in magenta as a ball-and-stick representation. In this structure, the glucose-like portion occupies a similar position to the natural substrate and the alkyl chain occupies the +1 site, which would be occupied by the sugar moiety adjacent to that cleaved in the reaction in the natural substrate. The side chain of W525 becomes disordered and is not shown in the structure. (D) The catalytic binding site of α-glu II with the inhibitor MON-DNJ shown in purple as a ball-and-stick representation. In this structure, the alkyl chain of the iminosugar fully occupies the +1 site and extends beyond; the alkyl chain adopts two main conformations, one of which is located towards the +2 sugar-binding site. Addition of the inhibitor leads to disorder in the catalytic site including displacement of the 523–528 loop (containing W525) and multiple conformations for F571. In all figures, H atoms are removed for clarity and all structures are shown in the same orientation. Protein databank file 5foe.pdb was shown in A and B; 5ieg.pdb and 5ief.pdb were used for C and D, respectively.
The consequences of misfolding
Iminosugars show antiviral activity against viruses from a wide range of different families, thus it appears that glycoproteins from many viruses are dependent upon the calnexin cycle for proper folding. There is, however, indirect evidence that the extent of misfolding caused by iminosugar inhibition of α-glucosidases varies between viruses. For some viruses, such as HIV, the level of secreted virus decreases by only a small amount in the presence of iminosugars, but the secreted viral particles have greatly reduced infectivity [29]. This implies that the degree of misfolding is small enough to escape detection by the cellular unfolded protein response but large enough to impair function. In contrast, for other viruses (including DENV, Hepatitis B virus and bovine viral diarrhea virus) the presence of iminosugars leads to greatly reduced secretion of virions [1,19,66]. This suggests that the misfolding of the glycoproteins in this case is more severe. In the case of Hepatitis B, at high doses of iminosugars (where α-glu I is inhibited and viral glycoproteins with the unmodified Glc3Man9GlcNAc2 glycoform will predominate) a considerable fraction of the viral glycoproteins are retained within the cell. These glycoproteins appear as aggregates and have an intracellular half-life in excess of 24 h. It was hypothesised that one particular viral glycoprotein (M) when in the triglucosylated state may act as a kind of ‘poison pill' in the ER preventing the secretion of the virus. In this case a viral protein acts as a ‘dominant negative' poison of viral secretion and may itself be considered the antiviral drug. The long intracellular half-life of the ‘poison pill’ may be due to aberrant trafficking between the ER and Golgi that has recently been observed for glycoproteins retaining terminal glucose residues on their glycans. This additional mechanism could lead to a reservoir of unfolded protein interfering with the ERQC. These are areas that need to be investigated further to understand the structural requirements for viral assembly, secretion and fusion to host cells in an iminosugar protein folding compromised context.
Currently, we cannot yet predict which glycoproteins will be secreted and which degraded in the presence of an iminosugar, and it remains a challenge to identify whether inhibition of ER α-glu I versus α-glu II will affect the extent of misfolding observed.
Clinical prospects and challenges
Naturally occurring iminosugars including DNJ and CAST have been used as the starting point for medicinal chemistry strategies aimed at antiviral drug discovery. Compounds modified from these starting materials have been, or are currently being, evaluated in clinical trials against HIV, HCV and DENV [67–71].
The n-butylated form of DNJ (NB-DNJ) and celgosivir, a prodrug of CAST, were both evaluated in clinical trials against HIV; however, neither proceeded further than phase II [67,68]. In the case of NB-DNJ, although some effects on viraemia were observed, it proved impossible to maintain therapeutic concentrations of the drug in serum. Observations that the concentrations of NB-DNJ necessary to inhibit the ER α-glucosidases in whole-cell assays greatly exceed that necessary to achieve the same inhibition of isolated enzymes suggested that difficulties in penetration of the drug into the ER were responsible for these poor results. These trials did, however, demonstrate that both DNJ and celgosivir are well tolerated by patients. The principal side effect of DNJ-derived iminosugars is osmotic diarrhoea, which arises due to the inhibition of the gut α-glucosidases [68,72]. This side effect is reversible and can be controlled by diet; however, improvements to limit the unwanted effect would be desirable, particularly in the development of iminosugar antivirals to treat chronic viral infections.
The current most promising targets for iminosugar antivirals appear to be DENV, for which clinical trials are in process [70,71], and influenza, where promising results have been observed in preclinical studies [21,73]. DENV is a mosquito-borne disease estimated to infect up to 390 million people per annum [74]. Many of these infections have no clinical manifestation, whilst others require hospitalisation, and a small percentage develop into dengue haemorrhagic fever, which can be fatal [75]. Both celgosivir and the DNJ-derivative UV-4 (MON-DNJ) are currently in clinical trials against DENV. UV-4 is in a phase I clinical trial, with a study evaluating the pharmacokinetics of the hydrochloride salt of UV-4 (UV-4B) administered as a multiple ascending dose [71]. Celgosivir has recently been approved for a phase II clinical trial [70]. In a proof of concept phase 1b study in which 50 patients were recruited, administration of celgosivir did not appear to reduce the viral load or fever burden in patients with uncomplicated dengue fever [76]. However, further investigations using a mouse model suggest that the dosing regime may be critical for efficacy. Mouse studies carried out prior to the phase 1b study suggested that a twice-daily treatment regime would be sufficient to reduce viraemia [77]; however, work carried out in light of the results from the phase 1b clinical trial suggests that this treatment regime is inadequate when the treatment is begun at the peak of viraemia [78]. More recent work suggests that a four-time daily regime is likely to be more effective against established infections and this will be investigated in the upcoming trial. A range of iminosugars, including NB-DNJ, NN-DNJ and UV-4B, have been tested for efficacy against the influenza virus [20,21,73]. Intriguingly, the antiviral efficacy observed varies between different influenza strains and subtypes in addition to varying with the specific iminosugar inhibitor tested. In a recent study Warfield et al. demonstrated that, at high concentrations, UV4-B shows activity against both influenza A and influenza B strains in cell culture, and that it gave a significant survival benefit against influenza A H1N1 and H3N2, and influenza B/Sichuan/379/99 in lethal mouse models. Future studies on the efficacy against influenza in humans are planned [21].
Conclusions
Our understanding of the mechanism of action of iminosugar antivirals has developed enormously since DNJ was first reported to have an antiviral effect against HIV. Medicinal chemistry approaches have improved the efficacy and pharmacokinetics of natural iminosugars, and current compounds represent good prospects for iminosugar antivirals reaching the clinic. However, medicinal chemists are already leading the way towards the ‘next generation’ of iminosugars. The compounds currently under investigation are well tolerated by patients and offer hope against current and future viral challenges; however, it is always desirable to limit off-target effects and increase ER uptake. The structures of mammalian ER α-glucosidases, together with those of off-target α-glucosidases, have the potential to facilitate rational design of new inhibitors with increased specificity for the ER-resident enzyme(s).
Table 1. Iminosugar IC50 values against purified rat α-glucosidase I and II; experiments were performed using a Glc1-3Man7GlcNAc2 substrate.
| Iminosugar | α-glu I | α-glu II | |
|---|---|---|---|
| Glc2 | Glc1 | ||
| NB-DNJ | 0.68 ± 0.05 | 10.80 ± 05 | 53.00 ± 6.6 |
| NN-DNJ | 0.54 ± 0.08 | 2.10 ± 0.42 | 14.90 ± 3.6 |
| MON-DNJ | 0.015 ± 0.04 | 0.90 ± 0.19 | 1.60 ± 0.2 |
| NAP-DNJ | 0.017 ± 0.01 | 0.30 ± 0.1 | 0.83 ± 0.13 |
Abbreviations
- BiP
binding immunoglobulin protein
- CAST
castanospermine
- DENV
dengue virus
- ENGase
endo-β-N-acetylglucosaminidase
- ER
endoplasmic reticulum
- ERAD
ER-associated degradation
- ERQC
ER quality control
- FOS
free oligosaccharides
- HA
haemagglutinin
- HCV
hepatitis C virus
- MON-DNJ
N-(9′-methoxynonyl)-1-deoxynojirimycin
- NAP-DNJ
N-(6′-4″-azido-2″-nitrophenylamino) hexyl-1-deoxynojirimycin
- NB-DNJ
N-butyl-deoxynojirimycin
- NN-DNJ
N-nonyl-deoxynojirimycin
- PDI
protein disulfide-isomerase
- PNGase
peptide:N-glycanase
- UGGT
UDP-glucose:glycoprotein glucosyl transferase
Competing Interests
The Authors declare that there are no competing interests associated with the manuscript.
References
- 1.Block T.M., Lu X., Platt F.M., Foster G.R., Gerlich W.H., Blumberg B.S. et al. (1994) Secretion of human hepatitis B virus is inhibited by the imino sugar N-butyldeoxynojirimycin. Proc. Natl Acad. Sci. U.S.A. 91, 2235–2239 doi: 10.1073/pnas.91.6.2235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hammond C., Braakman I. and Helenius A. (1994) Role of N-linked oligosaccharide recognition, glucose trimming, and calnexin in glycoprotein folding and quality control. Proc. Natl Acad. Sci. U.S.A. 91, 913–917 doi: 10.1073/pnas.91.3.913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mehta A., Lu X., Block T.M., Blumberg B.S. and Dwek R.A. (1997) Hepatitis B virus (HBV) envelope glycoproteins vary drastically in their sensitivity to glycan processing: evidence that alteration of a single N-linked glycosylation site can regulate HBV secretion. Proc. Natl Acad. Sci. U.S.A. 94, 1822–1827 doi: 10.1073/pnas.94.5.1822 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dwek R.A., Butters T.D., Platt F.M. and Zitzmann N. (2002) Targeting glycosylation as a therapeutic approach. Nat. Rev. Drug Discovery 1, 65–75 doi: 10.1038/nrd708 [DOI] [PubMed] [Google Scholar]
- 5.Chang J., Guo J.-T., Du Y. and Block T. (2013) Imino sugar glucosidase inhibitors as broadly active anti-filovirus agents. Emerg. Microbes Infect. 2, e77 doi: 10.1038/emi.2013.77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Scott L.J. (2016) Tetravalent dengue vaccine: a review in the prevention of dengue disease. Drugs 76, 1301–1312 doi: 10.1007/s40265-016-0626-8 [DOI] [PubMed] [Google Scholar]
- 7.Ahmed A. and Felmlee D.J. (2015) Mechanisms of hepatitis C viral resistance to direct acting antivirals. Viruses 7, 6716–6729 doi: 10.3390/v7122968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Iyidogan P. and Anderson K.S. (2014) Current perspectives on HIV-1 antiretroviral drug resistance. Viruses 6, 4095–4139 doi: 10.3390/v6104095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Paulsen H. (1966) Carbohydrates containing nitrogen or sulfur in the ‘hemiacetal’ ring. Angew. Chem. Int. Ed. Engl. 5, 495–510 doi: 10.1002/anie.196604951 [DOI] [Google Scholar]
- 10.Platt F.M., Neises G.R., Dwek R.A. and Butters T.D. (1994) N-butyldeoxynojirimycin is a novel inhibitor of glycolipid biosynthesis. J. Biol. Chem. 269, 8362–8365 PMID: [PubMed] [Google Scholar]
- 11.Cox T., Lachmann R., Hollak C., Aerts J., van Weely S., Hrebícek M. et al. (2000) Novel oral treatment of Gaucher's disease with N-butyldeoxynojirimycin (OGT 918) to decrease substrate biosynthesis. Lancet 355, 1481–1485 doi: 10.1016/S0140-6736(00)02161-9 [DOI] [PubMed] [Google Scholar]
- 12.Joubert P.H., Venter C.P., Joubert H.F. and Hillebrand I. (1985) The effect of a 1-deoxynojirimycin derivative on post-prandial blood glucose and insulin levels in healthy black and white volunteers. Eur. J. Clin. Pharmacol. 28, 705–708 doi: 10.1007/BF00607920 [DOI] [PubMed] [Google Scholar]
- 13.Shailubhai K., Pukazhenthi B.S., Saxena E.S., Varma G.M. and Vijay I.K. (1991) Glucosidase I, a transmembrane endoplasmic reticular glycoprotein with a luminal catalytic domain. J. Biol. Chem. 266, 16587–16593 PMID: [PubMed] [Google Scholar]
- 14.Karlsson G.B., Butters T.D., Dwek R.A. and Platt F.M. (1993) Effects of the imino sugar N-butyldeoxynojirimycin on the N-glycosylation of recombinant gp120. J. Biol. Chem. 268, 570–576 PMID: [PubMed] [Google Scholar]
- 15.Chen W., Helenius J., Braakman I. and Helenius A. (1995) Cotranslational folding and calnexin binding during glycoprotein synthesis. Proc. Natl Acad. Sci. U.S.A. 92, 6229–6233 doi: 10.1073/pnas.92.14.6229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Caramelo J.J. and Parodi A.J. (2008) Getting in and out from calnexin/calreticulin cycles. J. Biol. Chem. 283, 10221–10225 doi: 10.1074/jbc.R700048200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Eisenberg R.J., Atanasiu D., Cairns T.M., Gallagher J.R., Krummenacher C. and Cohen G.H. (2012) Herpes virus fusion and entry: a story with many characters. Viruses 4, 800–832 doi: 10.3390/v4050800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wu S.-F., Lee C.-J., Liao C.-L., Dwek R.A., Zitzmann N. and Lin Y.-L. (2002) Antiviral effects of an iminosugar derivative on flavivirus infections. J. Virol. 76, 3596–3604 doi: 10.1128/JVI.76.8.3596-3604.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Courageot M.-P., Frenkiel M.-P., Dos Santos C.D., Deubel V. and Desprès P. (2000) Alpha-Glucosidase inhibitors reduce dengue virus production by affecting the initial steps of virion morphogenesis in the endoplasmic reticulum. J. Virol. 74, 564–572 doi: 10.1128/JVI.74.1.564-572.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hussain S., Miller J.L., Harvey D.J., Gu Y., Rosenthal P.B., Zitzmann N. et al. (2015) Strain-specific antiviral activity of iminosugars against human influenza A viruses. J. Antimicrob. Chemother. 70, 136–152 doi: 10.1093/jac/dku349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Warfield K.L., Barnard D.L., Enterlein S.G., Smee D.F., Khaliq M., Sampath A. et al. (2016) The iminosugar UV-4 is a broad inhibitor of influenza A and B viruses ex vivo and in mice. Viruses 8, 71 doi: 10.3390/v8030071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chapel C., Garcia C., Bartosch B., Roingeard P., Zitzmann N., Cosset F.-L. et al. (2007) Reduction of the infectivity of hepatitis C virus pseudoparticles by incorporation of misfolded glycoproteins induced by glucosidase inhibitors. J. Gen. Virol. 88, 1133–1143 doi: 10.1099/vir.0.82465-0 [DOI] [PubMed] [Google Scholar]
- 23.Steinmann E., Whitfield T., Kallis S., Dwek R.A., Zitzmann N., Pietschmann T. et al. (2007) Antiviral effects of amantadine and iminosugar derivatives against hepatitis C virus. Hepatology 46, 330–338 doi: 10.1002/hep.21686 [DOI] [PubMed] [Google Scholar]
- 24.Gruters R.A., Neefjes J.J., Tersmette M., de Goede R.E.Y., Tulp A., Huisman H.G. et al. (1987) Interference with HIV-induced syncytium formation and viral infectivity by inhibitors of trimming glucosidase. Nature 330, 74–77 doi: 10.1038/330074a0 [DOI] [PubMed] [Google Scholar]
- 25.Fleet G.W.J., Karpas A., Dwek R.A., Fellows L.E., Tyms A.S., Petursson S. et al. (1988) Inhibition of HIV replication by amino-sugar derivatives. FEBS Lett. 237, 128–132 doi: 10.1016/0014-5793(88)80185-6 [DOI] [PubMed] [Google Scholar]
- 26.Sadat M.A., Moir S., Chun T.-W., Lusso P., Kaplan G., Wolfe L. et al. (2014) Glycosylation, hypogammaglobulinemia, and resistance to viral infections. N. Engl. J. Med. 370, 1615–1625 doi: 10.1056/NEJMoa1302846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tatu U., Hammond C. and Helenius A. (1995) Folding and oligomerization of influenza hemagglutinin in the ER and the intermediate compartment. EMBO J. 14, 1340–1348 PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hebert D.N., Foelimer B. and Helenius A. (1996) Calnexin and calreticulin promote folding, delay oligomerization and suppress degradation of influenza hemagglutinin in microsomes. EMBO J. 15, 2961–2968 PMID: [PMC free article] [PubMed] [Google Scholar]
- 29.Fischer P.B., Karlsson G.B., Butters T.D., Dwek R.A. and Platt F.M. (1996) N-butyldeoxynojirimycin-mediated inhibition of human immunodeficiency virus entry correlates with changes in antibody recognition of the V1/V2 region of gp120. J. Virol. 70, 7143–7152 PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kelleher D.J. and Gilmore R. (2006) An evolving view of the eukaryotic oligosaccharyltransferase. Glycobiology 16, 47R–62R doi: 10.1093/glycob/cwj066 [DOI] [PubMed] [Google Scholar]
- 31.Xu C. and Ng D.T.W. (2015) Glycosylation-directed quality control of protein folding. Nat. Rev. Mol. Cell Biol. 16, 742–752 doi: 10.1038/nrm4073 [DOI] [PubMed] [Google Scholar]
- 32.Ruggiano A., Foresti O. and Carvalho P. (2014) ER-associated degradation: protein quality control and beyond. J. Cell Biol. 204, 869–879 doi: 10.1083/jcb.201312042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hebert D.N., Foellmer B. and Helenius A. (1995) Glucose trimming and reglucosylation determine glycoprotein association with calnexin in the endoplasmic reticulum. Cell 81, 425–433 doi: 10.1016/0092-8674(95)90395-X [DOI] [PubMed] [Google Scholar]
- 34.Hammond C. and Helenius A. (1994) Quality control in the secretory pathway: retention of a misfolded viral membrane glycoprotein involves cycling between the ER, intermediate compartment, and Golgi apparatus. J. Cell Biol. 126, 41–52 doi: 10.1083/jcb.126.1.41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schallus T., Jaeckh C., Fehér K., Palma A.S., Liu Y., Simpson J.C. et al. (2008) Malectin: a novel carbohydrate-binding protein of the endoplasmic reticulum and a candidate player in the early steps of protein N-glycosylation. Mol. Biol. Cell 19, 3404–3414 doi: 10.1091/mbc.E08-04-0354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Galli C., Bernasconi R., Soldà T., Calanca V. and Molinari M. (2011) Malectin participates in a backup glycoprotein quality control pathway in the mammalian ER. PLoS ONE 6, e16304 doi: 10.1371/journal.pone.0016304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen Y., Hu D., Yabe R., Tateno H., Qin S.-Y., Matsumoto N. et al. (2011) Role of malectin in Glc2Man9GlcNAc2-dependent quality control of α1-antitrypsin. Mol. Biol. Cell 22, 3559–3570 doi: 10.1091/mbc.E11-03-0201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Qin S.-Y., Hu D., Matsumoto K., Takeda K., Matsumoto N., Yamaguchi Y. et al. (2012) Malectin forms a complex with ribophorin I for enhanced association with misfolded glycoproteins. J. Biol. Chem. 287, 38080–38089 doi: 10.1074/jbc.M112.394288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Trombetta S.E. and Parodi A.J. (1992) Purification to apparent homogeneity and partial characterization of rat liver UDP-glucose:glycoprotein glucosyltransferase. J. Biol. Chem. 267, 9236–9240 PMID: [PubMed] [Google Scholar]
- 40.Taylor S.C., Ferguson A.D., Bergeron J.J.M. and Thomas D.Y. (2004) The ER protein folding sensor UDP-glucose glycoprotein–glucosyltransferase modifies substrates distant to local changes in glycoprotein conformation. Nat. Struct. Mol. Biol. 11, 128–134 doi: 10.1038/nsmb715 [DOI] [PubMed] [Google Scholar]
- 41.Weng S. and Spiro R.G. (1993) Demonstration that a kifunensine-resistant α-mannosidase with a unique processing action on N-linked oligosaccharides occurs in rat liver endoplasmic reticulum and various cultured cells. J. Biol. Chem. 268, 25656–25663 PMID: [PubMed] [Google Scholar]
- 42.Tokunaga F., Brostrom C., Koide T. and Arvan P. (2000) Endoplasmic reticulum (ER)-associated degradation of misfolded N-linked glycoproteins is suppressed upon inhibition of ER mannosidase I. J. Biol. Chem. 275, 40757–40764 doi: 10.1074/jbc.M001073200 [DOI] [PubMed] [Google Scholar]
- 43.Weng S. and Spiro R.G. (1997) Demonstration of a peptide:N-glycosidase in the endoplasmic reticulum of rat liver. Biochem. J. 322, 655–661 doi: 10.1042/bj3220655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Suzuki T., Park H. and Lennarz W.J. (2016) Cytoplasmic peptide:N-glycanase (PNGase) in eukaryotic cells: occurrence, primary structure, and potential functions. FASEB J. 16, 635–641 doi: 10.1096/fj.01-0889rev [DOI] [PubMed] [Google Scholar]
- 45.Suzuki T., Kitajima K., Emori Y., Inoue Y. and Inoue S. (1997) Site-specific de-N-glycosylation of diglycosylated ovalbumin in hen oviduct by endogenous peptide:N-glycanase as a quality control system for newly synthesized proteins. Proc. Natl Acad. Sci. U.S.A. 94, 6244–6249 doi: 10.1073/pnas.94.12.6244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pierce R.J., Spik G. and Montreuil J. (1979) Cytosolic location of an endo-N-acetyl-β-d-glucosaminidase activity in rat liver and kidney. Biochem. J. 180, 673–676 doi: 10.1042/bj1800673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kato T., Fujita K., Takeuchi M., Kobayashi K., Natsuka S., Ikura K. et al. (2002) Identification of an endo-β-N-acetylglucosaminidase gene in Caenorhabditis elegans and its expression in Escherichia coli. Glycobiology 12, 581–587 PMID: [DOI] [PubMed] [Google Scholar]
- 48.Shoup V.A. and Touster O. (1976) Purification and characterization of the α-d-mannosidase of rat liver cytosol. J. Biol. Chem. 251, 3845–3852 PMID: [PubMed] [Google Scholar]
- 49.Suzuki T., Hara I., Nakano M., Shigeta M., Nakagawa T., Kondo A. et al. (2006) Man2c1, an α-mannosidase, is involved in the trimming of free oligosaccharides in the cytosol. Biochem. J. 400, 33–41 doi: 10.1042/BJ20060945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Saint-Pol A., Codogno P. and Moore S.E.H. (1999) Cytosol-to-lysosome transport of free polymannose-type oligosaccharides: kinetic and specificity studies using rat liver lysosomes. J. Biol. Chem. 274, 13547–13555 doi: 10.1074/jbc.274.19.13547 [DOI] [PubMed] [Google Scholar]
- 51.Durrant C. and Moore S.E.H. (2002) Perturbation of free oligosaccharide trafficking in endoplasmic reticulum glucosidase I-deficient and castanospermine-treated cells. Biochem. J. 365, 239–247 doi: 10.1042/bj20011786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mellor H.R., Neville D.C.A., Harvey D.J., Platt F.M., Dwek R.A. and Butters T.D. (2004) Cellular effects of deoxynojirimycin analogues: inhibition of N-linked oligosaccharide processing and generation of free glucosylated oligosaccharides. Biochem. J. 381, 867–875 doi: 10.1042/BJ20031824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Alonzi D.S., Neville D.C.A., Lachmann R.H., Dwek R.A. and Butters T.D. (2008) Glucosylated free oligosaccharides are biomarkers of endoplasmic-reticulum α-glucosidase inhibition. Biochem. J. 409, 571–580 doi: 10.1042/BJ20070748 [DOI] [PubMed] [Google Scholar]
- 54.Rawlings A.J., Lomas H., Pilling A.W., Lee M.J.-R., Alonzi D.S., Rountree J.S.S. et al. (2009) Synthesis and biological characterisation of novel N-alkyl-deoxynojirimycin α-glucosidase inhibitors. ChemBioChem 10, 1101–1105 doi: 10.1002/cbic.200900025 [DOI] [PubMed] [Google Scholar]
- 55.Grinna L.S. and Robbins P.W. (1980) Substrate specificities of rat liver microsomal glucosidases which process glycoproteins. J. Biol. Chem. 255, 2255–2258 PMID: [PubMed] [Google Scholar]
- 56.Mackeen M.M., Almond A., Deschamps M., Cumpstey I., Fairbanks A.J., Tsang C. et al. (2009) The conformational properties of the Glc3Man unit suggest conformational biasing within the chaperone-assisted glycoprotein folding pathway. J. Mol. Biol. 387, 335–347 doi: 10.1016/j.jmb.2009.01.043 [DOI] [PubMed] [Google Scholar]
- 57.Wichit S., Jittmittraphap A., Hidari K.I.P.J., Thaisomboonsuk B., Petmitr S., Ubol S. et al. (2011) Dengue virus type 2 recognizes the carbohydrate moiety of neutral glycosphingolipids in mammalian and mosquito cells. Microbiol. Immunol. 55, 135–140 doi: 10.1111/j.1348-0421.2010.00293.x [DOI] [PubMed] [Google Scholar]
- 58.Perera R., Riley C., Isaac G., Hopf-Jannasch A.S., Moore R.J., Weitz K.W. et al. (2012) Dengue virus infection perturbs lipid homeostasis in infected mosquito cells. PLoS Pathog. 8, e1002584 doi: 10.1371/journal.ppat.1002584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kapadia S.B., Barth H., Baumert T., McKeating J.A. and Chisari F.V. (2007) Initiation of hepatitis C virus infection is dependent on cholesterol and cooperativity between CD81 and scavenger receptor B type I. J. Virol. 81, 374–383 doi: 10.1128/JVI.01134-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Voisset C., Lavie M., Helle F., De Beeck A.O., Bilheu A., Bertrand-Michel J. et al. (2008) Ceramide enrichment of the plasma membrane induces CD81 internalization and inhibits hepatitis C virus entry. Cell. Microbiol. 10, 606–617 doi: 10.1111/j.1462-5822.2007.01070.x [DOI] [PubMed] [Google Scholar]
- 61.Sayce A.C., Alonzi D.S., Killingbeck S.S., Tyrrell B.E., Hill M.L., Caputo A.T. et al. (2016) Iminosugars inhibit dengue virus production via inhibition of ER alpha-glucosidases — not glycolipid processing enzymes. PLoS Negl. Trop. Dis. 10, e0004524 doi: 10.1371/journal.pntd.0004524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Warfield K.L., Plummer E.M., Sayce A.C., Alonzi D.S., Tang W., Tyrrell B.E. et al. (2016) Inhibition of endoplasmic reticulum glucosidases is required for in vitro and in vivo dengue antiviral activity by the iminosugar UV-4. Antiviral Res. 129, 93–98 doi: 10.1016/j.antiviral.2016.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Barker M.K. and Rose D.R. (2013) Specificity of processing α-glucosidase I is guided by the substrate conformation: crystallographic and in silico studies. J. Biol. Chem. 288, 13563–13574 doi: 10.1074/jbc.M113.460436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Caputo A.T., Alonzi D.S., Marti L., Reca I.-B., Kiappes J.L., Struwe W.B. et al. (2016) Structures of mammalian ER α-glucosidase II capture the binding modes of broad-spectrum iminosugar antivirals. Proc. Natl Acad. Sci. U.S.A. 113, E4630––E4638. doi: 10.1073/pnas.1604463113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Satoh T., Toshimori T., Yan G., Yamaguchi T. and Kato K. (2016) Structural basis for two-step glucose trimming by glucosidase II involved in ER glycoprotein quality control. Sci. Rep. 6, 20575 doi: 10.1038/srep20575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zitzmann N., Mehta A.S., Carrouee S., Butters T.D., Platt F.M., McCauley J. et al. (1999) Imino sugars inhibit the formation and secretion of bovine viral diarrhea virus, a pestivirus model of hepatitis C virus: implications for the development of broad spectrum anti-hepatitis virus agents. Proc. Natl Acad. Sci. U.S.A. 96, 11878–11882 doi: 10.1073/pnas.96.21.11878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Hoechst Marion Roussel (1996) A randomized, double-blind active-controlled, dose-ranging study of the safety and efficacy of chronically administered MDL 28,574A in the treatment of HIV-infected patients. NLM identifier: NCT00002151
- 68.Fischl M.A., Resnick L., Coombs R., Kremer A.B., Pottage J.C. Jr, Fass R.J. et al. (1994) The safety and efficacy of combination N-butyl-deoxynojirimycin (SC-48334) and zidovudine in patients with HIV-1 infection and 200-500 CD4 cells/mm3. J. Acquir. Immune Defic. Syndr. 7, 139–147 PMID: [PubMed] [Google Scholar]
- 69.BioWest Therapeutics Inc (2006) An extension study to evaluate the safety and efficacy of Celgosivir and Peginterferon Alfa-2b, with or without Ribavirin, in patients with chronic Hepatitis C genotype 1 infection. NLM identifier: NCT00292084
- 70.Singapore General Hospital (2016) Celgosivir or modipafant as treatment for adult participants with uncomplicated dengue fever in Singapore. NLM identifier: NCT02569827
- 71.Emergent BioSolutions (2016) Safety and pharmacokinetics of UV-4B solution administered orally as multiple ascending doses to healthy subjects. NLM identifier: NCT02696291
- 72.Tierney M., Pottage J., Kessler H., Fischl M.A., Richman D., Merigan T. et al. (1995) The tolerability and pharmacokinetics of N-butyl-deoxynojirimycin in patients with advanced HIV disease (ACTG 100). J. Acquired Immune Defic. Syndr. Hum. Retrovirol. 10, 549–553 doi: 10.1097/00042560-199510050-00008 [DOI] [PubMed] [Google Scholar]
- 73.Stavale E.J., Vu H., Sampath A., Ramstedt U. and Warfield K.L. (2015) In vivo therapeutic protection against influenza a (H1N1) oseltamivir-sensitive and resistant viruses by the iminosugar UV-4. PLoS ONE 10, e0121662 doi: 10.1371/journal.pone.0121662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Bhatt S., Gething P.W., Brady O.J., Messina J.P., Farlow A.W., Moyes C.L. et al. (2013) The global distribution and burden of dengue. Nature 496, 504–507 doi: 10.1038/nature12060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Halstead S.B. (2007) Dengue. Lancet 370, 1644–1652 doi: 10.1016/S0140-6736(07)61687-0 [DOI] [PubMed] [Google Scholar]
- 76.Low J.G., Sung C., Wijaya L., Wei Y., Rathore A.P.S., Watanabe S. et al. (2014) Efficacy and safety of celgosivir in patients with dengue fever (CELADEN): a phase 1b, randomised, double-blind, placebo-controlled, proof-of-concept trial. Lancet Infect. Dis. 14, 706–715 doi: 10.1016/S1473-3099(14)70730-3 [DOI] [PubMed] [Google Scholar]
- 77.Watanabe S., Rathore A.P.S., Sung C., Lu F., Khoo Y.M., Connolly J. et al. (2012) Dose- and schedule-dependent protective efficacy of celgosivir in a lethal mouse model for dengue virus infection informs dosing regimen for a proof of concept clinical trial. Antiviral Res. 96, 32–35 doi: 10.1016/j.antiviral.2012.07.008 [DOI] [PubMed] [Google Scholar]
- 78.Watanabe S., Chan K.W.-K., Dow G., Ooi E.E., Low J.G. and Vasudevan S.G. (2016) Optimizing celgosivir therapy in mouse models of dengue virus infection of serotypes 1 and 2: the search for a window for potential therapeutic efficacy. Antiviral Res. 127, 10–19 doi: 10.1016/j.antiviral.2015.12.008 [DOI] [PubMed] [Google Scholar]
- 79.Sousa M. and Parodi A.J. (1995) The molecular basis for the recognition of misfolded glycoproteins by the UDP-Glc:glycoprotein glucosyltransferase. EMBO J. 14, 4196–4203 PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Oda Y., Hosokawa N., Wada I. and Nagata K. (2003) EDEM as an acceptor of terminally misfolded glycoproteins released from calnexin. Science 299, 1394–1397 doi: 10.1126/science.1079181 [DOI] [PubMed] [Google Scholar]
- 81.Hosokawa N., Wada I., Natsuka Y. and Nagata K. (2006) EDEM accelerates ERAD by preventing aberrant dimer formation of misfolded α1-antitrypsin. Genes Cells 11, 465–476 doi: 10.1111/j.1365-2443.2006.00957.x [DOI] [PubMed] [Google Scholar]
- 82.Hirao K., Natsuka Y., Tamura T., Wada I., Morito D., Natuska S. et al. (2006) EDEM3, a soluble EDEM homolog, enhances glycoprotein endoplasmic reticulum-associated degradation and mannose trimming. J. Biol. Chem. 281, 9650–9658 doi: 10.1074/jbc.M512191200 [DOI] [PubMed] [Google Scholar]
- 83.Olivari S., Cali T., Salo K.E.H., Paganetti P., Ruddock L.W. and Molinari M. (2006) EDEM1 regulates ER-associated degradation by accelerating de-mannosylation of folding-defective polypeptides and by inhibiting their covalent aggregation. Biochem. Biophys. Res. Commun. 349, 1278–1284 doi: 10.1016/j.bbrc.2006.08.186 [DOI] [PubMed] [Google Scholar]
- 84.Kim W., Spear E.D. and Ng D.T.W. (2005) Yos9p detects and targets misfolded glycoproteins for ER-associated degradation. Mol. Cell 19, 753–764 doi: 10.1016/j.molcel.2005.08.010 [DOI] [PubMed] [Google Scholar]
- 85.Buschhorn B.A., Kostova Z., Medicherla B. and Wolf D.H. (2004) A genome-wide screen identifies Yos9p as essential for ER-associated degradation of glycoproteins. FEBS Lett. 577, 422–426 doi: 10.1016/j.febslet.2004.10.039 [DOI] [PubMed] [Google Scholar]
- 86.Bhamidipati A., Denic V., Quan E.M. and Weissman J.S. (2005) Exploration of the topological requirements of ERAD identifies Yos9p as a lectin sensor of misfolded glycoproteins in the ER lumen. Mol. Cell 19, 741–751 doi: 10.1016/j.molcel.2005.07.027 [DOI] [PubMed] [Google Scholar]
- 87.Christianson J.C., Shaler T.A., Tyler R.E. and Kopito R.R. (2008) OS-9 and GRP94 deliver mutant α1-antitrypsin to the Hrd1–SEL1L ubiquitin ligase complex for ERAD. Nat. Cell Biol. 10, 272–282 doi: 10.1038/ncb1689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Suzuki T., Huang C. and Fujihira H. (2016) The cytoplasmic peptide:N-glycanase (NGLY1) — structure, expression and cellular functions. Gene 577, 1–7 doi: 10.1016/j.gene.2015.11.021 [DOI] [PMC free article] [PubMed] [Google Scholar]