Abstract
Redox imbalance in cells induces lipid peroxidation and generates a class of highly reactive metabolites known as lipid-derived electrophiles (LDEs) that can modify proteins and affects their functions. Identifying targets of LDEs is critical to understand how such modifications are functionally implicated in oxidative-stress associated diseases. Here we report a quantitative chemoproteomic method to globally profile protein targets and sites modified by LDEs. In this strategy, we designed and synthesized an alkyne-functionalized aminooxy probe to react with LDE-modified proteins for imaging and proteomic profiling. Using this probe, we successfully quantified >4000 proteins modified by 4-hydroxy-2-nonenal (HNE) of high confidence in mammalian cell lysate and combined with a tandem-orthogonal proteolysis activity-based protein profiling (TOP-ABPP) strategy, we identified ~400 residue sites targeted by HNE including reactive cysteines in peroxiredoxins, an important family of enzymes with anti-oxidant roles. Our method expands the toolbox to quantitatively profile protein targets of endogenous electrophiles and the enlarged inventory of LDE-modified proteins and sites will contribute to functional elucidation of cellular pathways affected by oxidative stress.
Keywords: Lipid-derived electrophile, 4-hydroxy-2-nonenal, Chemoproteomics, Aminooxy probe, Activity-based protein profiling
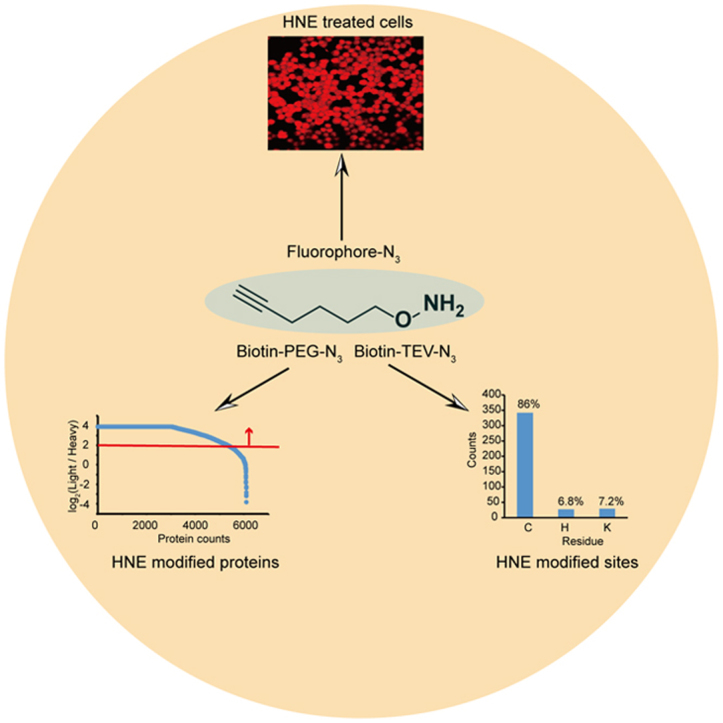
Graphical abstract
Highlights
-
•
An aminooxy-based probe to capture targets of lipid-derived electrophiles (LDEs).
-
•
The probe enables imaging and chemoproteomic profiling of LDE modifications.
-
•
The method quantified >4000 protein targets modified by 4-hydroxy-2-nonenal (HNE).
-
•
The method identified ~400 residue sites modified by HNE.
1. Introduction
Reactive oxygen species (ROS) is closely related to human diseases [1]. ROS leakage can attack lipids in biological membranes such as arachidonic acid (AA) and linoleic acid (LA), and produce a series of reactive electrophiles, including 4-hydroxy-2-nonenal (HNE), 4-oxo-2-nonenal (ONE) and acrolein (ACR) [2]. These so-called lipid derived electrophiles (LDEs) have a common α, β-unsaturated aldehyde or ketone group, and are chemically reactive with nucleophilic amino acids such as cysteine, histidine and lysine. Covalent modification of proteins by LDEs and the resulting functional perturbation have been implicated with onset and development of many pathological conditions, including neurodegeneration, cancers, diabetes, and cardiovascular diseases [3], [4], [5], [6], [7], [8]. It is therefore of high importance to identify protein targets and specific sites of LDE in order to help mechanistically elucidate the functional consequences of such modifications in pathogenesis of oxidative-stress associated diseases.
Several methods have been developed to profile LDE modifications in proteomes [2]. Using a “bioorthogonal analogue” approach, Marnett and co-workers synthesized azido and alkynyl derivatives of HNE and used these bioorthogonal probes to identify protein targets of HNE in RKO cells [9]. The concept was further demonstrated to assess alkylation damage by LDE using the alkyne-functionalized HNE and ONE probes [10]. Combined with a photo-release strategy [11], the bioorthogonal probe has also been used to quantitatively analyze proteins and sites modified by HNE [12]. While these studies have revealed a large number of targets in proteomes that are sensitive to LDE modification, the “unnatural surrogate” approach is not suitable for detecting endogenous modifications and in principle is not applicable for LDEs with small structures, such as ACR, because any chemical modification will perturb its reactivity. Recently, Wang et al. reported a competitive activity-based protein profiling (ABPP) strategy which enables the quantification of >1000 cysteine sites of LDE modifications in proteomes by a cysteine-reactive iodoacetamide(IA)-alkyne probe [13]. Though it can be applied with any cysteine-reactive small molecule including LDEs, this method indirectly identifies sites of LDE modification by competitive displacement and it remains possible that, in certain instances, blockade of IA-probe labeling can be due to modification of a neighboring residue rather than the IA-labeled cysteine itself.
Given LDE can react with nucleophilic residues through Michael addition, its modification on proteins often results in a free aldehyde or ketone group, which is also known as “protein carbonylation” [14]. Based on this unique chemical feature, many studies have implemented carbonyl-reactive reagents to capture LDE modifications from diversified biological samples [15], [16], [17], [18], [19], [20]. Among these “aldehyde directed” probes, hydrazide(HZ) or aminooxy(AO) are the most prevalent warheads and in combination with fluorophore or biotin, such probes allows visualization of both exogenous and endogenous LDE modified proteins directly in a complex system as well as enrichment for further analysis by mass spectrometry(MS)-based proteomics.
While the aldehyde directed probes have been widely used in capturing LDE modified proteins, their applications in site-specific analysis of LDE modifications are less often reported. This is probably due to the bulky fluorophore or biotin groups that hinder the release and identification of LDE-adducted peptides during MS analysis. In order to overcome this limitation, we synthesized HZ and AO derivative probes with a bioorthognal alkyne group and evaluated their capability in capturing both HNE-modified proteins and residue sites for MS identification. We discovered that the AO probe is more reactive in labeling LDE modifications and together with a reductive dimethylation-based strategy [21], [22], we were able to successfully quantify >4000 HNE modified proteins in mammalian cell lysates. Using the AO probe in a TOP-ABPP strategy [23], we identified ~400 HNE-adducted sites of high confidence including both catalytic and non-catalytic cysteines of peroxiredoxin 6 that we further confirmed using biochemical means.
2. Materials and methods
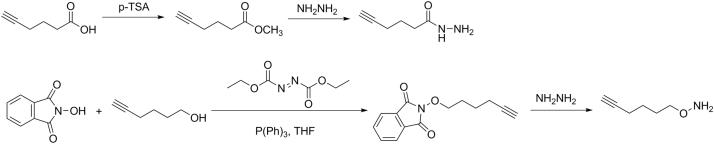
2.1. Synthesis of the HZyne and AOyne probes
The routes for synthesizing HZyne and AOyne probes are designed based on previous reports [24], [25] and shown as below.
.
2.2. Test of HZyne and AOyne reactivity using a GSH model
1 mM Glutathione(GSH) is reacted with 1 mM HNE in Tris-HCl buffer (pH=7.4) for 1 h at room temperature. The reaction is then adjusted to pH 5.0 and added with 5 mM AOyne for another 30 min. The reaction solutions are filtered through a 0.22 µm membrane. The samples are analyzed on an ACQUITY H-Class ultra performance liquid chromatography-electrospray ionization-mass spectrometry (UPLC-ESI-MS) system (Waters Corp.) with a peptide BEH C18 column (Waters 300, 1.7 2.1 100 mm) and a quadrupole rods SQ Detector 2 mass spectrometer (Waters Corp.) The ultra-pure water and acetonitrile are used as the mobile phase in a 7 min gradient and the ion chromatographic traces are extracted at m/z 306 for GSH, 462 for GSH-HNE, 557 for GSH-HNE-OAyne, and 572 for GSH-HNE-HZyne.
2.3. Cell preparation
HEK293T cells are cultured at 37 ℃ under 5% CO2 atmosphere in DMEM culture medium supplemented with 10% FBS and 1% Penicillin-Streptomycin. For in gel fluorescence and mass spectrometry experiments, cells are grown to 100% confluence and washed 3 times with PBS before collection. The cells are centrifuged at 1000 rpm for 3 min at 25 ℃ and the cell pallets are stored at −80 ℃. For the fluorescence imaging experiment, cells are grown to 70% confluence in normal media and transferred to serum-free media. The cells are incubated with HNE at indicated concentrations for 1 h and washed with pre-warmed PBS buffer. The cells are then fixed in PBS containing 4% (v/v) formaldehyde at 37 ℃ for 15 min. After fixation, the cells are rinsed three times with PBS buffer and permeabilized in PBS containing 0.2% (v/v) Triton X-100 at 37 ℃ for 10 min. After permeabilization, the cells are rinsed 3 times with PBS buffer and labeled with AOyne for 30 min before conjugation with the Cy5 dye.
2.4. Capture of LDEs modified proteins using aminooxy-alkyne
Cell pallets are lysed by sonication in ice-cold PBS containing 0.1% (v/v) Triton X-100, centrifuged at 100,000g for 30 min to remove cell debris, and protein concentrations are determined by BCA protein assay (Pierce™ BCA Protein Assay Kit, Thermo Fisher Scientific). Proteomes are normalized to 2 mg/mL, incubated with 100 μM HNE and labeled by 5 mM AOyne at pH 5.0 for 30 min (optimized conditions). Proteomes labeled by AOyne are precipitated with methanol/chloroform(v/v=4:1) and resuspended in 1.2% (w/v) SDS/PBS.
2.5. RD-ABPP
HNE treated proteomes or DMSO treated proteomes are separately labeled by AOyne as mentioned in Section 2.4. Click chemistry is performed in 1.2% SDS/PBS in 1 mL volume. The sample is added with a premix of 1 mM CuSO4, 100 μM TBTA ligand and 100 μM biotin-(PEG)2-N3 tag and then added with 1 mM TCEP to reduce the Cu2+ and trigger this reaction. After incubation at room temperature for 1 h, the proteome are precipitated again with methanol/chloroform and resuspended in 1.2% SDS/PBS. The proteomes are boiled at 90 ℃ for 5 min and after centrifugation at 1400g for 1 min at room temperature, the supernatant is diluted to 0.2% SDS/PBS and subjected to streptavidin enrichment. The enriched proteins are digested by trypsin on-bead in 100 mM TEAB buffer and subjected to reductive dimethylation labeling [22]. Briefly, 4 μL (per 100 μL of sample) of 4% (w/w) light or heavy formaldehyde is added to HNE treated proteomes or DMSO treated proteomes, respectively. At the same time, 4 μL (per 100 μL of sample) of 0.6 M sodium cyanoborohydride is added. After labeling for 1 h at room temperature, the reaction is sequentially quenched by 1% (w/w) ammonia (16 μL per 100 μL of sample) and 5% (w/w) formic acid (8 μL per 100 μL of sample). Finally, heavy and light labeled peptide samples are mixed, concentrated, separated by the Fast-seq protocol [26], and analyzed on a Q Exactive mass spectrometer (Thermo Fisher) as described in Section 2.7.
2.6. TOP-ABPP
HNE treated proteomes are labeled by AOyne as described in Section 2.4. Click chemistry is performed in 1.2% SDS/PBS and 2 mg/mL in 1 mL volume. The sample is added with a premix of 1 mM CuSO4, 100 μM TBTA ligand and 100 μM biotin-TEV-N3 tag and then added with 1 mM TCEP to reduce the Cu2+ and trigger this reaction. After incubation at room temperature for 1 h, the proteome are precipitated again with methanol/chloroform and resuspended in 1.2% SDS/PBS. The proteomes are boiled at 90 ℃ for 5 min and after centrifugation at 1400g for 1 min at room temperature, the supernatant is diluted to 0.2% SDS/PBS and subjected to streptavidin enrichment followed by on-bead trypsin digestion. The peptide supernatant is collected and the beads are further subjected to TEV digestion. Finally, the supernatant is collected, desalted, and analyzed on a Q Exactive mass spectrometer (Thermo Fisher) as described in Section 2.7.
2.7. LC-MS/MS and data analysis
The peptides for protein and sites identification are analyzed on a Q Exactive mass spectrometer (Thermo Fisher). Under the positive-ion mode, full-scan mass spectra are acquired over the m/z range from 350 to 1800 using the Orbitrap mass analyzer with mass resolution setting of 70000. MS/MS fragmentation is performed in a data-dependent mode, of which the 20 most intense ions are selected from each full-scan mass spectrum for high-energy collision induced dissociation (HCD) and MS2 analysis. MS2 spectra are acquired with a resolution setting of 17500 using the Orbitrap analyzer. Some other parameters in the centroid format: isolation window, 2.0 m/z units; default charge, 2+; normalized collision energy, 28%; maximum IT, 50 ms; dynamic exclusion, 20.0 s.
LC-MS/MS data is analyzed by ProLuCID [27] with static modification of cysteine (+57.0215 Da) and variable oxidation of methionine (+15.9949 Da). For RD-ABPP data, the isotopic modifications (28.0313 and 34.0631 Da for light and heavy labeling respectively) are set as variable modifications on the N-terminal of a peptide and lysines. For TOP-ABPP data, 578.39042 Da are set as variable modifications on cysteines, histidines or lysines. The searching results are filtered by DTASelect [28] and peptides are also restricted to fully tryptic with a defined peptide false positive rate of 1%. The ratios of reductive dimethylation are quantified by the CIMAGE software as described before [29].
2.8. Validation of C47 and C91 of PRDX6 as sites of HNE modification
HEK293T cell line with transient overexpression of PRDX6 (WT, C47A, C91A and the double mutant) are grown to 70% confluency in 10 cm dishes. Cells are harvested, lysed in 0.1% Triton X-100, and centrifuged at 100000g for 30 min to remove cell debris. The concentrations of the soluble fraction are determined by BCA protein assay and normalized to 2 mg/mL. Proteomes are incubated with 100 μM HNE at room temperature for 1 h and adjusted to pH 5 before AOyne is added to label for 30 mins. The labeled proteomes are precipitated with methanol/chloroform and resuspended in 1.2% SDS/PBS. Click chemistry with azido-biotin is done as described above, and after a second precipitation, the proteomes are enriched with streptavidin beads for 3 h. The beads are washed with PBS for 3 times, and eluted with 20 μL of 5X reducing gel loading buffer at 95℃for 20 min. Samples are separated on a SDS-PAGE gel and immunoblotted with an anti-His6 tag antibody.
To confirm the sites of modification by LC-MS/MS, lysates of HEK293T cells overexpressing PRDX6 are incubated with HNE at room temperature for 1 h and reduced with 4 mM NaBH4 for1h. FLAG-tagged PRDX6 are immunoprecipitated by anti-FLAG agarose beads for 2 h at room temperature and subjected to in gel digestion by trypsin. The digested peptides are analyzed by LC-MS/MS and a variable modification of 158.13068 Da on cysteines is searched using Mascot to identify the HNE-adducted sites.
3. Results
3.1. Evaluation of probe reactivity using a glutathione (GSH) model
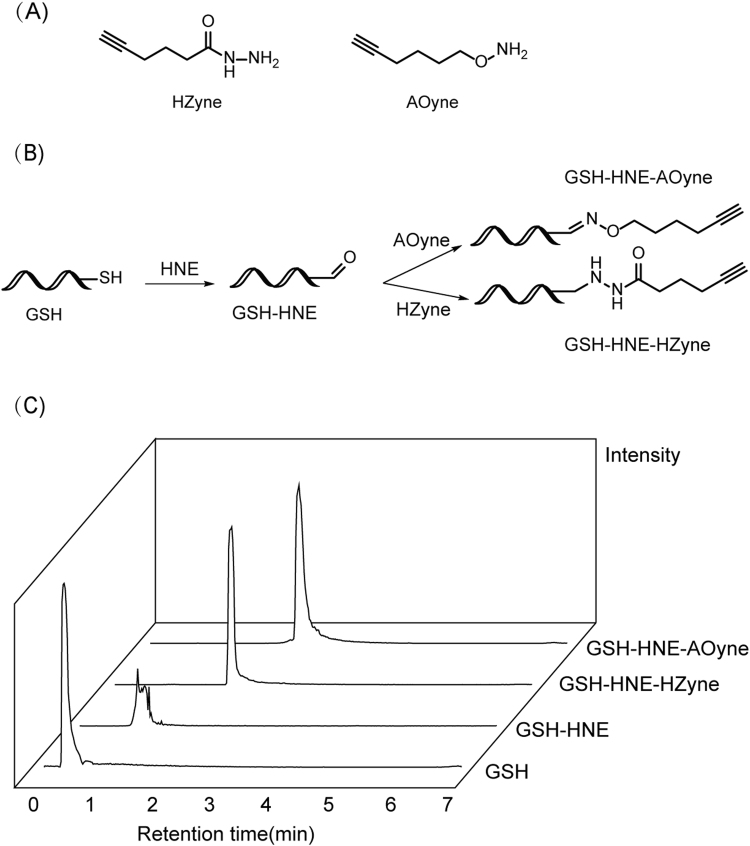
We designed and synthesized the hydrazide-alkyne (HZyne) and aminooxy-alkyne (AOyne) probes (Fig. 1A) based on the routes shown in Section 2.1. Both warheads have been conjugated with biotin to capture LDE-modified proteins before [16], [18], but it remains challenging to identify sites of LDE labeling. The alkyne group in our probes can be flexibly conjugated via copper catalyzed click chemistry [30] with either azido-fluorophore for imaging or with a cleavable azido-biotin tag for enrichment and identification of LDE modified proteins and sites. We firstly use a simple GSH model to validate the chemical reaction between these probes and a representative LDE molecule, HNE (Fig. 1B). LC-MS confirmed that once the HNE-GSH adduct is formed via Michael addition, both HZyne and AOyne probes can efficiently react with the remaining aldehyde group to form the desired final products as expected (Fig. 1C).
Fig. 1.
Evaluation of probe reactivity using a glutathione (GSH) model. (A) Structures of the HZyne and AOyne probes. (B) Scheme of the GSH model. After the thiol group of GSH reacts with HNE through Michael addition, the HZyne and AOyne probes are used to capture the remaining aldehyde group on the adduct. (C) The extracted ion chromatographic peaks of GSH, the HNE-GSH adduct, and the final products formed with the HZyne and AOyne probes, respectively.
3.2. Optimization of aminooxy-alkyne for proteomic labeling and imaging
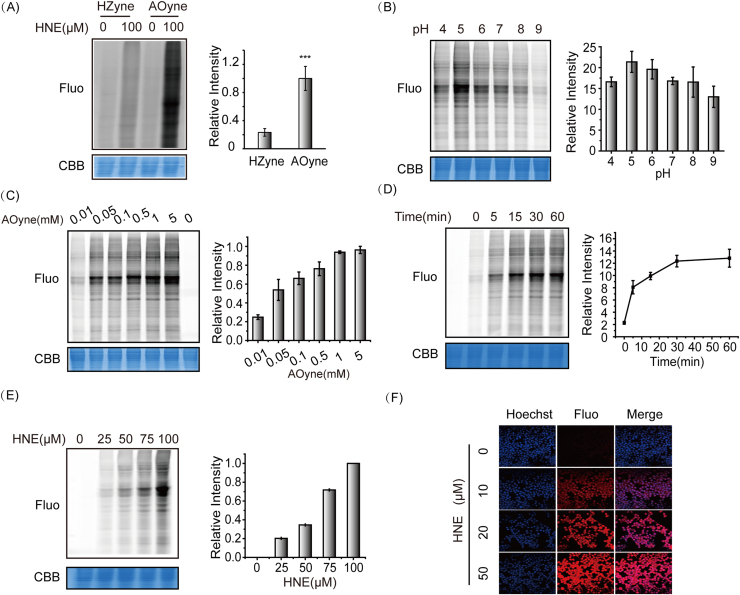
We next examine the reactivity of the HZyne and AOyne probes in detecting LDE modifications at the proteomic level. The HNE-treated lysates of HEK293T were labeled with 5 mM of each probe and conjugated with azido-fluorophore by click chemistry. In-gel fluorescence showed that AOyne exhibits much higher labeling signals in proteomes than HZyne (Fig. 2A). The observed difference in reactivity is consistent with a previous study using biotinylated HZ and AO probes to label human serum albumins [20]. We therefore proceed to further optimize the labeling conditions of AOyne including pH, time and probe concentration. We found the optimal labeling of AOyne at pH 5.0 (Fig. 2B) and 5 mM probe concentration (Fig. 2C). In addition, the labeling is fast, already reaching its maximum after 30 min (Fig. 2D). With these optimized labeling conditions, we showed that AOyne is able to monitor HNE-modifications in a dose-dependent manner in both cell lysates (Fig. 2E) and living cells (Fig. 2F).
Fig. 2.
Optimization of AOyne for proteomic labeling and imaging. (A) AOyne shows much stronger labeling in proteomes than HZyne. (B) Optimization of pH for AOyne labeling. (C) Optimization of probe concentration for AOyne labeling. (D) Optimization of time for AOyne labeling. (E) AOyne is able to label HNE modifications in cell lysates in a dose-dependent manner. (F) AOyne is able to monitor HNE modification in living cells by fluorescence imaging. In A-E, the representative images of in-gel fluorescence (Fluo) and coomassie blue staining (CBB) are shown on the left side and the quantification of fluorescence intensity averaged from multiple replicates are shown on the right.
3.3. Identification of HNE-modified proteins by RD-ABPP using aminooxy-alkyne
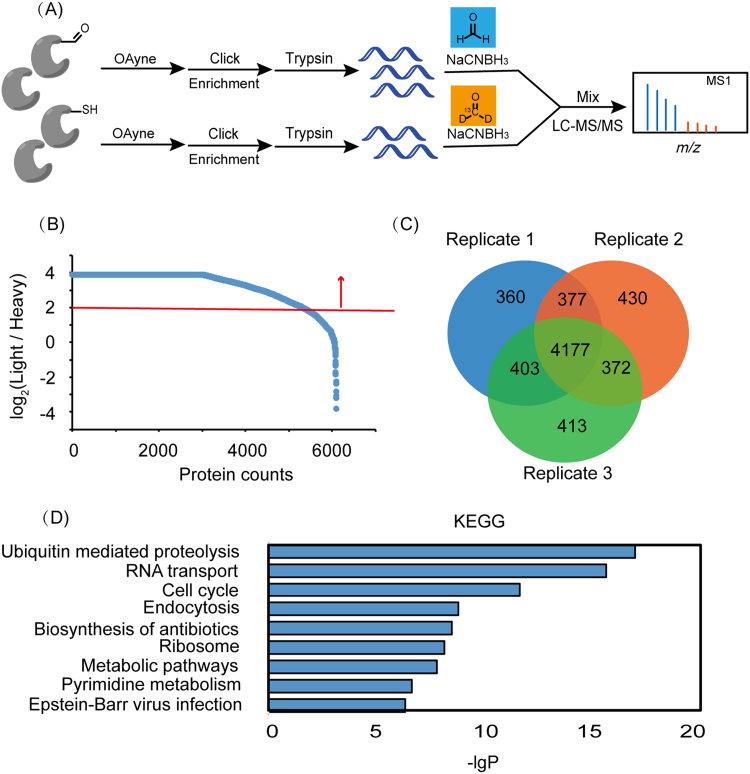
We next used the AOyne probe in a quantitative RD-ABPP platform [31] that combines reductive dimethylation [21], [22] labeling with ABPP [32], [33] to enrich and identify HNE modified proteins (Fig. 3A). Two aliquots of HEK293T proteomes were first treated with either 100 μM of HNE or DMSO, respectively. The “experiment” (HNE-treated) and “control” (DMSO-treated) proteomes are independently labeled by with AOyne, conjugated with azido-biotin, enriched by streptavidin beads and digested with trypsin. After reductive dimethylation labeling, the heavy (“control”) and light (“experiment”) peptides are combined and subjected to LC-MS/MS analysis. We chose a light/heavy ratio of 4.0 as the enrichment cutoff (Fig. 3B), and collectively identified >5000 HNE modified proteins from each of the three replicates of experiments which have 4177 proteins in common (Fig. 3B and C, Supplementary Table 1). We performed the KEGG pathway analysis of these HNE-modified proteins and found that ubiquitin mediated proteolysis, RNA transport, and cell cycle are the three most enriched functional pathways (Fig. 3D).
Fig. 3.
Identification of HNE-modified proteins by reductive dimethylation (RD)-ABPP. (A) Scheme of identifying HNE modified proteins by RD-ABPP using the AOyne probe. (B) Ratio distribution of a representative RD-ABPP profiling experiment in which ~6000 proteins are quantified and those with a light/heavy (HNE/DMSO) ratio >=4.0 are considered as HNE modified proteins. (C) A total number of 4177 HNE modified proteins are commonly identified from three replicates of RD-ABPP profiling experiments. (D) The KEGG pathway analysis of the HNE-modified proteins.
3.4. Identification of HNE-modified sites by TOP-ABPP using aminooxy-alkyne
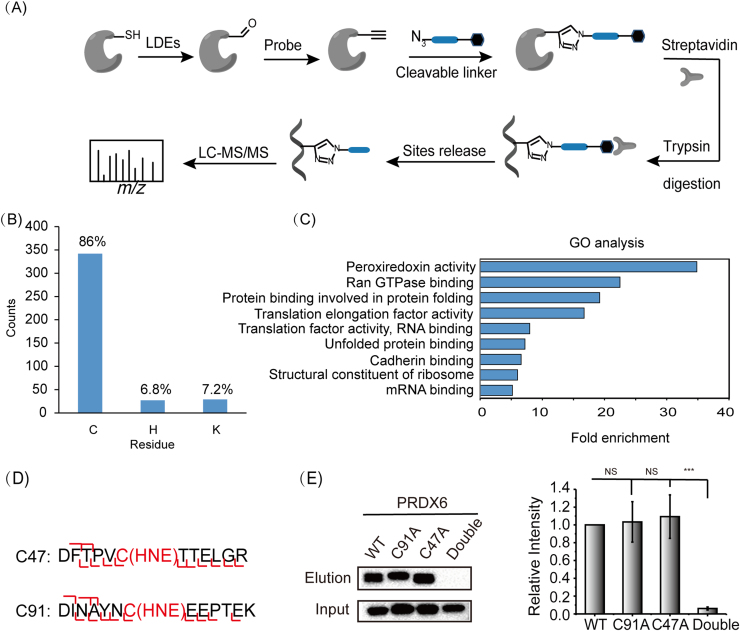
One of the major advantages of the AOyne probe is to, in combination with a cleavable azido-biotin tag, allow identification of sites of LDE modifications in proteomes. To demonstrate this utility, we employed AOyne in a TOP-ABPP strategy [23] in which a special azido-biotin tag with a TEV protease cleavable linker was used for conjugation with the AOyne labeled proteins. After enrichment by streptavidin and on-bead trypsin digestion, the HNE modified peptides are released from the beads by an orthogonal round of TEV digestion and then subjected to LC-MS/MS analysis (Fig. 4A). Using this method, we identified a total number of 398 HNE modified sites including 342 cysteines, 27 histidines and 29 lysines (Fig. 4B, Supplementary Table 2). We performed the gene ontology (GO) analysis of the proteins containing these HNE-modified cysteines and found that the “peroxiredoxin activity” was highly enriched in the category of GO molecular function (Fig. 4C). Peroxiredoxins (PRDXs) are a family of antioxidant enzymes that catalyze the removal of hydroperoxides and maintain redox balance in cells [34]. In human genome, the PRDX family consists of 6 members (PRDX 1–6) and four of them (PRDX 1, 2, 3 and 6) were identified in our profiling data with HNE-modified cysteines. Among these four peroxiredoxins, PRDX6 is unique because it has only one cysteine (Cys47) involved in catalysis (the so-called “1-Cys” peroxiredoxin). This protein was previously found to be adducted with HNE in the livers of rat chronically fed an ethanol-containing diet and its peroxiredoxin activity was inhibited upon HNE adduction in vitro [35]. However, the authors observed the modification only on the non-catalytic cysteine, Cys91, and therefore proposed based on molecular modeling that the adduction on Cys91 caused a change in the protein active site conformation. Our TOP-ABPP profiling identified both the catalytic Cys47 and non-catalytic Cys91 as the sites of HNE modification in PRDX6 and to further confirm these results, we overexpressed PRDX6 in HEK293T cells for biochemical verification. We treated the cell lysates with 100 μM HNE and immunoprecipitated PRDX6 for trypsin digestion. LC-MS/MS analysis of the digested peptides unambiguously mapped the sites of modification as Cys47 and Cys91 (Fig. 4D). We also immunoprecipitated the C47A and C91A mutants of PRDX6 and found that the labeling by AOyne is much reduced only in the double mutant but not in either of the single mutants (Fig. 4E). This data confirmed our profiling results that both Cys47 and Cys91 are modified by HNE and it provided additional evidence for explaining the partial loss of PRDX6 activity upon HNE treatment [35].
Fig. 4.
Identification of the sites of HNE modification by TOP-ABPP. (A) Scheme of identifying the sites of HNE modification by TOP-ABPP using the AOyne probe. (B) Number of cysteine, histidine and lysine sites identified with HNE modification by TOP-ABPP. (C) The gene ontology (GO) analysis of the molecular functions of proteins that contain these HNE-modified cysteines. (D) C47 and C91 of PRDX6 are modified by HNE as confirmed by LC-MS/MS. (E) The double mutant of C47A and C91A showed much reduced probe labeling compared to the wild-type PRDX6.
4. Discussion
In this study we reported a novel chemoproteomic method to profile LDE-modified proteins and residue sites in native proteomes. Using an aminooxy-alkyne probe that can specifically capture the aldehyde group on the Michael adducts, we successfully identified >4000 HNE-modified proteins and ~400 residue sites in proteomes of HEK293T cells. We demonstrated the alkyne-functionalized probe can be conjugated with an azido reporter group of choice, maintaining its full flexibility and compatibility for versatile applications including cell imaging and ABPP-based quantitative chemical proteomics [33].
Both hydrazide and aminooxy warheads have been installed onto the so-called “aldehyde-reactive probe” (ARP) [36], [37] to capture LDE-modified proteins, and literature survey indicated that hydrazide is so far a more popular choice. Our comparison showed that AOyne has much better labeling efficiency in HNE-treated proteomes than HZyne and it is possible that the extra step of reduction to stabilize the hydrazone adduct needs to be further optimized if a similar labeling efficiency is achieved for the HZyne probe. Along this direction, exploration of new chemistry to develop novel ARP warheads with improved labeling efficiency and adduct stabilization is well warranted.
Our list of HNE-modified residues includes a large portion of cysteines, which is consistent with another chemoproteomic profiling study in which a surrogate HNE-akyne probe was used [12]. The results are also supported by previous theoretical studies, which concluded that HNE reacts nucleophilic residues in the following order: Cys >> His > Lys [38]. It is worth noting that, while our manuscript is in preparation, Lu and coworkers published their work of identifying HNE-modified sites in MCF7 cell lysates using an aminooxy probe with fluorous derivatization and found majority of them are histidines [39]. The striking difference on the amino acid preferences could be attributed to different cell lines used (HEK293T vs. MCF7), different labeling conditions (25 ℃, 1 h vs. 37 ℃, 4 h) or different enrichment strategies (biotin-avidin vs. fluorous tag-fluorous solid phase extraction). Nevertheless, both studies confirmed that the aminooxy group is an effective and reliable warhead to capture LDE modifications in native proteomes and chemical proteomics have been proven as powerful strategies to globally identify and quantify events of LDE modifications in cells, which will contribute to our understanding of oxidative stress-mediated damage and signaling.
Author contributions
Y.C. and C.W. designed the experiments. Y.C., Y.Cong., B.Q., T.L., X.C., Z.Y., and X.H. conducted experiments. Y.C. and C.W. wrote the manuscript.
Conflict of interest
The authors declare no conflicts of interest.
Acknowledgements
We thank the Computing Platform of the Center for Life Science for supporting the proteomic data analysis. This work was supported by National Science Foundation of China (21472008 and 81490741), Ministry of Science and Technology of China (2016YFA0501500) and a "1000 Talents Plan" Young Investigator Award (C.W).
Footnotes
Supplementary data associated with this article can be found in the online version at doi:10.1016/j.redox.2017.04.001.
Appendix A. Supplementary material
Supplementary material: The Supplementary information contains Supplementary Table 1 and 2 that contain the lists of HNE-modified proteins identified by RD-ABPP and HNE-modified residues identified by TOP-ABPP, respectively.
.
References
- 1.Valko M., Leibfritz D., Moncol J., Cronin M.T., Mazur M., Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int J. Biochem Cell Biol. 2007;39:44–84. doi: 10.1016/j.biocel.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 2.Chen Y., Qin W., Wang C. Chemoproteomic profiling of protein modifications by lipid-derived electrophiles. Curr. Opin. Chem. Biol. 2016;30:37–45. doi: 10.1016/j.cbpa.2015.10.029. [DOI] [PubMed] [Google Scholar]
- 3.Reed T.T. Lipid peroxidation and neurodegenerative disease. Free Radic. Biol. Med. 2011;51:1302–1319. doi: 10.1016/j.freeradbiomed.2011.06.027. [DOI] [PubMed] [Google Scholar]
- 4.Di Domenico F., Pupo G., Tramutola A., Giorgi A., Schinina M.E., Coccia R., Head E., Butterfield D.A., Perluigi M. Redox proteomics analysis of HNE-modified proteins in Down syndrome brain: clues for understanding the development of Alzheimer disease. Free Radic. Biol. Med. 2014;71:270–280. doi: 10.1016/j.freeradbiomed.2014.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Butterfield D.A., Gu L., Di Domenico F., Robinson R.A. Mass spectrometry and redox proteomics: applications in disease. Mass Spectrom. Rev. 2014;33:277–301. doi: 10.1002/mas.21374. [DOI] [PubMed] [Google Scholar]
- 6.Sauriasari R., Andrajati R., Dharmeizar Azizahwati, Saputri D.A., Muris R.U., Manfaatun A., Amanda O.F., Setiawan H., Sakano N., Wang D.H., Ogino K. Marker of lipid peroxidation related to diabetic nephropathy in Indonesian type 2 diabetes mellitus patients. Diabetes Res Clin. Pract. 2015;108:193–200. doi: 10.1016/j.diabres.2014.12.016. [DOI] [PubMed] [Google Scholar]
- 7.Uchida K. Role of reactive aldehyde in cardiovascular diseases. Free Radic. Biol. Med. 2000;28:1685–1696. doi: 10.1016/s0891-5849(00)00226-4. [DOI] [PubMed] [Google Scholar]
- 8.Zhang P.Y., Xu X., Li X.C. Cardiovascular diseases: oxidative damage and antioxidant protection. Eur. Rev. Med Pharmacol. Sci. 2014;18:3091–3096. [PubMed] [Google Scholar]
- 9.Vila A., Tallman K.A., Jacobs A.T., Liebler D.C., Porter N.A., Marnett L.J. Identification of protein targets of 4-hydroxynonenal using click chemistry for ex vivo biotinylation of azido and alkynyl derivatives. Chem. Res Toxicol. 2008;21:432–444. doi: 10.1021/tx700347w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Codreanu S.G., Ullery J.C., Zhu J., Tallman K.A., Beavers W.N., Porter N.A., Marnett L.J., Zhang B., Liebler D.C. Alkylation damage by lipid electrophiles targets functional protein systems. Mol. Cell Proteom. 2014;13:849–859. doi: 10.1074/mcp.M113.032953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kim H.Y., Tallman K.A., Liebler D.C., Porter N.A. An azido-biotin reagent for use in the isolation of protein adducts of lipid-derived electrophiles by streptavidin catch and photorelease. Mol. Cell Proteom. 2009;8:2080–2089. doi: 10.1074/mcp.M900121-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang J., Tallman K.A., Porter N.A., Liebler D.C. Quantitative chemoproteomics for site-specific analysis of protein alkylation by 4-hydroxy-2-nonenal in cells. Anal. Chem. 2015;87:2535–2541. doi: 10.1021/ac504685y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang C., Weerapana E., Blewett M.M., Cravatt B.F. A chemoproteomic platform to quantitatively map targets of lipid-derived electrophiles. Nat. Meth. 2014;11:79–85. doi: 10.1038/nmeth.2759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dalle-Donne I., Giustarini D., Colombo R., Rossi R., Milzani A. Protein carbonylation in human diseases. Trends Mol. Med. 2003;9:169–176. doi: 10.1016/s1471-4914(03)00031-5. [DOI] [PubMed] [Google Scholar]
- 15.Tamarit J., de Hoogh A., Obis E., Alsina D., Cabiscol E., Ros J. Analysis of oxidative stress-induced protein carbonylation using fluorescent hydrazides. J. Proteom. 2012;75:3778–3788. doi: 10.1016/j.jprot.2012.04.046. [DOI] [PubMed] [Google Scholar]
- 16.Codreanu S.G., Zhang B., Sobecki S.M., Billheimer D.D., Liebler D.C. Global analysis of protein damage by the lipid electrophile 4-hydroxy-2-nonenal. Mol. Cell Proteom. 2009;8:670–680. doi: 10.1074/mcp.M800070-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Roe M.R., Xie H., Bandhakavi S., Griffin T.J. Proteomic mapping of 4-hydroxynonenal protein modification sites by solid-phase hydrazide chemistry and mass spectrometry. Anal. Chem. 2007;79:3747–3756. doi: 10.1021/ac0617971. [DOI] [PubMed] [Google Scholar]
- 18.Chavez J., Wu J., Han B., Chung W.G., Maier C.S. New role for an old probe: affinity labeling of oxylipid protein conjugates by N′-aminooxymethylcarbonylhydrazino d-biotin. Anal. Chem. 2006;78:6847–6854. doi: 10.1021/ac0607257. [DOI] [PubMed] [Google Scholar]
- 19.Chavez J.D., Wu J., Bisson W., Maier C.S. Site-specific proteomic analysis of lipoxidation adducts in cardiac mitochondria reveals chemical diversity of 2-alkenal adduction. J. Proteom. 2011;74:2417–2429. doi: 10.1016/j.jprot.2011.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Coffey C.M., Gronert S. A cleavable biotin tagging reagent that enables the enrichment and identification of carbonylation sites in proteins. Anal. Bioanal. Chem. 2016;408:865–874. doi: 10.1007/s00216-015-9176-2. [DOI] [PubMed] [Google Scholar]
- 21.Hsu J.L., Huang S.Y., Chow N.H., Chen S.H. Stable-isotope dimethyl labeling for quantitative proteomics. Anal. Chem. 2003;75:6843–6852. doi: 10.1021/ac0348625. [DOI] [PubMed] [Google Scholar]
- 22.Boersema P.J., Raijmakers R., Lemeer S., Mohammed S., Heck A.J. Multiplex peptide stable isotope dimethyl labeling for quantitative proteomics. Nat. Protoc. 2009;4:484–494. doi: 10.1038/nprot.2009.21. [DOI] [PubMed] [Google Scholar]
- 23.Weerapana E., Speers A.E., Cravatt B.F. Tandem orthogonal proteolysis-activity-based protein profiling (TOP-ABPP)--a general method for mapping sites of probe modification in proteomes. Nat. Protoc. 2007;2:1414–1425. doi: 10.1038/nprot.2007.194. [DOI] [PubMed] [Google Scholar]
- 24.Xu R., Sim M.K., Go M.L. Synthesis and pharmacological characterization of O-alkynyloximes of tropinone and N-methylpiperidinone as muscarinic agonists. J. Med Chem. 1998;41:3220–3231. doi: 10.1021/jm9708588. [DOI] [PubMed] [Google Scholar]
- 25.Malik G., Guinchard X., Crich D. Asymmetric synthesis of polyhydroxylated N-alkoxypiperidines by ring-closing double reductive amination: facile preparation of isofagomine and analogues. Org. Lett. 2012;14:596–599. doi: 10.1021/ol203213f. [DOI] [PubMed] [Google Scholar]
- 26.Ding C., Jiang J., Wei J., Liu W., Zhang W., Liu M., Fu T., Lu T., Song L., Ying W., Chang C., Zhang Y., Ma J., Wei L., Malovannaya A., Jia L., Zhen B., Wang Y., He F., Qian X., Qin J. A fast workflow for identification and quantification of proteomes. Mol. Cell Proteom. 2013;12:2370–2380. doi: 10.1074/mcp.O112.025023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Xu T., Park S.K., Venable J.D., Wohlschlegel J.A., Diedrich J.K., Cociorva D., Lu B., Liao L., Hewel J., Han X., Wong C.C., Fonslow B., Delahunty C., Gao Y., Shah H., Yates J.R., 3rd ProLuCID: an improved SEQUEST-like algorithm with enhanced sensitivity and specificity. J. Proteom. 2015;129:16–24. doi: 10.1016/j.jprot.2015.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tabb D.L., McDonald W.H., Yates J.R., 3rd DTASelect and Contrast: tools for assembling and comparing protein identifications from shotgun proteomics. J. Proteome Res. 2002;1:21–26. doi: 10.1021/pr015504q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Weerapana E., Wang C., Simon G.M., Richter F., Khare S., Dillon M.B., Bachovchin D.A., Mowen K., Baker D., Cravatt B.F. Quantitative reactivity profiling predicts functional cysteines in proteomes. Nature. 2010;468:790–795. doi: 10.1038/nature09472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rostovtsev V.V., Green L.G., Fokin V.V., Sharpless K.B. A stepwise huisgen cycloaddition process: copper(i)-catalyzed regioselective "ligation" of azides and terminal alkynes. Angew. Chem. Int Ed. Engl. 2002;41:2596–2599. doi: 10.1002/1521-3773(20020715)41:14<2596::AID-ANIE2596>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 31.Inloes J.M., Hsu K.L., Dix M.M., Viader A., Masuda K., Takei T., Wood M.R., Cravatt B.F. The hereditary spastic paraplegia-related enzyme DDHD2 is a principal brain triglyceride lipase. Proc. Natl. Acad. Sci. USA. 2014;111:14924–14929. doi: 10.1073/pnas.1413706111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Niphakis M.J., Cravatt B.F. Enzyme inhibitor discovery by activity-based protein profiling. Annu Rev. Biochem. 2014;83:341–377. doi: 10.1146/annurev-biochem-060713-035708. [DOI] [PubMed] [Google Scholar]
- 33.Wang C., Chen N. Activity-based protein profiling. Acta Chim. Sin. 2015;73:657–668. [Google Scholar]
- 34.Rhee S.G., Chae H.Z., Kim K. Peroxiredoxins: a historical overview and speculative preview of novel mechanisms and emerging concepts in cell signaling. Free Radic. Biol. Med. 2005;38:1543–1552. doi: 10.1016/j.freeradbiomed.2005.02.026. [DOI] [PubMed] [Google Scholar]
- 35.Roede J.R., Carbone D.L., Doorn J.A., Kirichenko O.V., Reigan P., Petersen D.R. In vitro and in silico characterization of peroxiredoxin 6 modified by 4-hydroxynonenal and 4-oxononenal. Chem. Res Toxicol. 2008;21:2289–2299. doi: 10.1021/tx800244u. [DOI] [PubMed] [Google Scholar]
- 36.Kubo K., Ide H., Wallace S.S., Kow Y.W. A novel, sensitive, and specific assay for abasic sites, the most commonly produced DNA lesion. Biochemistry. 1992;31:3703–3708. doi: 10.1021/bi00129a020. [DOI] [PubMed] [Google Scholar]
- 37.Atamna H., Cheung I., Ames B.N. A method for detecting abasic sites in living cells: age-dependent changes in base excision repair. Proc. Natl. Acad. Sci. USA. 2000;97:686–691. doi: 10.1073/pnas.97.2.686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Doorn J.A., Petersen D.R. Covalent modification of amino acid nucleophiles by the lipid peroxidation products 4-hydroxy-2-nonenal and 4-oxo-2-nonenal. Chem. Res Toxicol. 2002;15:1445–1450. doi: 10.1021/tx025590o. [DOI] [PubMed] [Google Scholar]
- 39.Yuan W., Zhang Y., Xiong Y., Tao T., Wang Y., Yao J., Zhang L., Yan G., Bao H., Lu H. Highly selective and large scale mass spectrometric analysis of 4-hydroxynonenal modification via fluorous derivatization and fluorous solid-phase extraction. Anal. Chem. 2017 doi: 10.1021/acs.analchem.6b04850. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material: The Supplementary information contains Supplementary Table 1 and 2 that contain the lists of HNE-modified proteins identified by RD-ABPP and HNE-modified residues identified by TOP-ABPP, respectively.