ABSTRACT
Purpose of Review: As individuals age, the quality of cognitive function becomes an increasingly important topic. The concept of mild cognitive impairment (MCI) has evolved over the past 2 decades to represent a state of cognitive function between that seen in normal aging and dementia. As such, it is important for health care providers to be aware of the condition and place it in the appropriate clinical context.
Recent Findings: Numerous international population-based studies have been conducted to document the frequency of MCI, estimating its prevalence to be between 15% and 20% in persons 60 years and older, making it a common condition encountered by clinicians. The annual rate in which MCI progresses to dementia varies between 8% and 15% per year, implying that it is an important condition to identify and treat. In those MCI cases destined to develop Alzheimer disease, biomarkers are emerging to help identify etiology and predict progression. However, not all MCI is due to Alzheimer disease, and identifying subtypes is important for possible treatment and counseling. If treatable causes are identified, the person with MCI might improve.
Summary: MCI is an important clinical entity to identify, and while uncertainties persist, clinicians need to be aware of its diagnostic features to enable them to counsel patients. MCI remains an active area of research as numerous randomized controlled trials are being conducted to develop effective treatments.
INTRODUCTION
Identifying pending cognitive impairment at an early stage has become an increasingly important challenge to physicians. Decades ago, it was satisfactory to distinguish dementia from typical cognitive aging, but in recent years, the desire to make a more fine-grained decision on incipient disease has become apparent. In evaluating persons for suspected Alzheimer disease (AD), the clinical spectrum from dementia has extended back to mild cognitive impairment (MCI) and ultimately to preclinical AD, at which point people are cognitively normal but harbor the underlying biological features of AD.1 This puts the clinician in the challenging but opportunistic position of detecting very early clinical features of incipient disease.
Central to this diagnostic scheme is the clinical construct of MCI.2,3 MCI is generally regarded as the borderland between the cognitive changes of aging and very early dementia, but while conceptually reasonable, the construct poses difficulties in clinical practice (Case 2-1A).4
Case 2-1A
A 66-year-old retired high school teacher presented for symptoms of increasing forgetfulness, which he had begun to experience in the past 1 to 2 years. His other cognitive functions, including language, attention, executive function, problem solving, and visuospatial skills, were all intact. He continued to drive without difficulty and handled the financial matters for his family without a problem. His wife had noted a slight increase in forgetfulness, but this had not been of concern to her. The patient noted that he had been taking more time to remember previously well-remembered events such as appointments with his doctor, scheduled meetings with friends, and planned visits with the children. He was not particularly disturbed by these symptoms, and he had not made any major social mistakes. His behavior was otherwise intact, and his mood was stable. He generally slept well and did not admit to any features of dream enactment behavior. He was concerned, however, because his mother had developed dementia later in life and died of what was probably Alzheimer disease at age 81, and the patient wanted to be assessed for the disease.
Comment. This scenario represents a common presentation to clinicians with an aging patient population. This patient is more forgetful than he formerly was and is worried about the possibility of a problem beyond aging. This patient appears to have had progressing memory difficulties in recent months to years. It did not appear that he had other extant medical comorbidities that were influencing his condition. Mental status testing is indicated and may provide further useful information, as is discussed in the continuation of this case later in the article.
HISTORICAL ASPECTS OF THE CONCEPT OF MILD COGNITIVE IMPAIRMENT
Historically, the term MCI has been in the literature for almost 4 decades, with the initial use coming from investigators at New York University who referred to Stage 3 on the Global Deterioration Scale as being MCI.5 In 1999, a group at the Mayo Clinic described subjects in their community aging study who had a memory concern beyond what was expected for age and who demonstrated a slight memory impairment yet did not meet criteria for dementia.6 The research criteria used to characterize these subjects were described, and the clinical outcomes were noted.
MULTIPLE TERMINOLOGIES
Over the years, several sets of terminology for MCI and related conditions have evolved, many referring to similar constructs in the general MCI range. The Mayo Clinic criteria previously noted focused on a memory disturbance and were developed to elucidate the earliest symptomatic stages of AD. However, it soon became apparent that not all intermittent cognitive states represented incipient AD nor did all patients have just a memory impairment. To address this situation, the Key Symposium was held in Stockholm, Sweden, in 2003, and criteria of a more broad scope were published in 2004.2,7 These criteria accomplished two goals: (1) to broaden the classification scheme beyond memory, and (2) to recognize that MCI could result from a variety of etiologies and not just AD. These criteria are outlined in Figure 2-1,8 demonstrating the syndromic phenotypes and how they can be paired with possible etiologies to assist the clinician in diagnosis. The Key Symposium characterization of MCI led to the distinction between the amnestic form of MCI and the nonamnestic form of MCI, since these clinical syndromes appeared to be aligned with etiologies in a differential fashion and may have variable outcomes. Traditionally, amnestic MCI is the typical prodromal stage of dementia due to AD, but other phenotypes can also lead to this type of dementia, such as logopenic aphasia, posterior cortical atrophy (also known as the visual variant), or a frontal lobe–dysexecutive presentation of AD.9 The essential feature of this he essential feature of this portrayal is that not all MCI is early AD.
Figure 2-1.
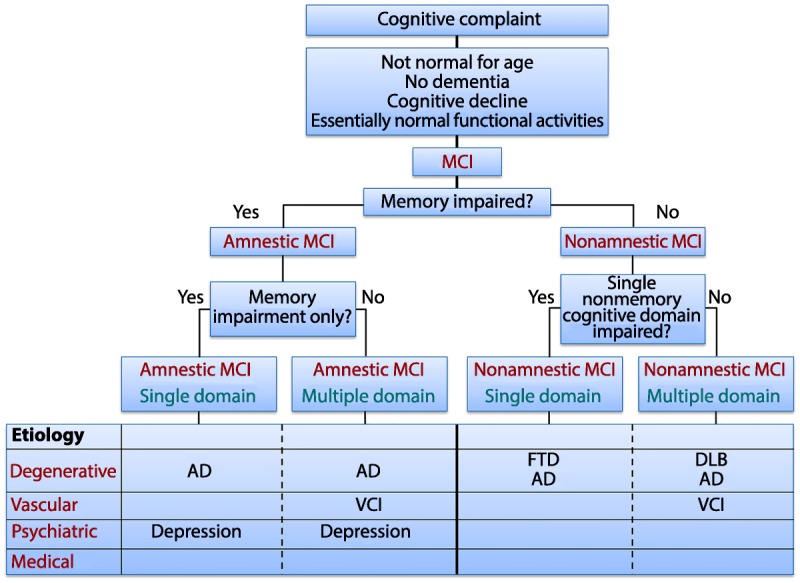
Key Symposium criteria. First Key Symposium criteria demonstrating the syndromic phenotypes and how they can be paired with possible etiologies to assist the clinician in making a diagnosis.
AD = Alzheimer disease; DLB = dementia with Lewy bodies; FTD = frontotemporal dementia; MCI = mild cognitive impairment; VCI = vascular cognitive impairment.
Modified with permission from Petersen RC, Continuum (Minneap Minn).8 journals.lww.com/continuum/Fulltext/2004/02000/MILD_COGNITIVE_IMPAIRMENT.3.aspx. © 2004, American Academy of Neurology.
The Key Symposium criteria prevailed in the field and influenced the development of several randomized controlled trials for possible intervention.10–14 In 2011, the National Institute on Aging (NIA) and the Alzheimer’s Association convened workgroups to develop criteria for the entire AD spectrum.1,9,15,16 The criteria for MCI due to AD essentially adopted the Key Symposium criteria while making some of the diagnostic features more explicit. These criteria also added biomarkers for underlying AD pathophysiology in an attempt to refine the underlying etiology and, hence, predict outcome. These criteria did not differentiate between amnestic and nonamnestic MCI.
At approximately the same time, the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) was being developed.17 For the general category of neurocognitive disorders, the criteria now include a predementia phase called mild neurocognitive disorder. Once again, the construct is very similar to the Key Symposium criteria for MCI and suggests that, in addition to the syndromic classification, certain features would allow the subclassification of the clinical presentation into pathologic etiologies. The category of mild neurocognitive disorder due to AD is very similar to the classification of MCI due to AD formulated by the NIA/Alzheimer’s Association workgroups.17
Finally, over the course of several years, the construct of prodromal AD evolved.18–20 This clinical condition grew from the accumulating literature that had developed regarding the observation that amnestic MCI, when paired with certain biomarkers, approximated the condition of AD. In fact, the proponents believed that a certain type of amnestic MCI coupled with biomarkers for the presence of amyloid or amyloid and tau constituted the earliest symptomatic stages of the AD process.20 The temporal evolution of the criteria for MCI and prodromal AD are presented in Figure 2-2.
Figure 2-3.
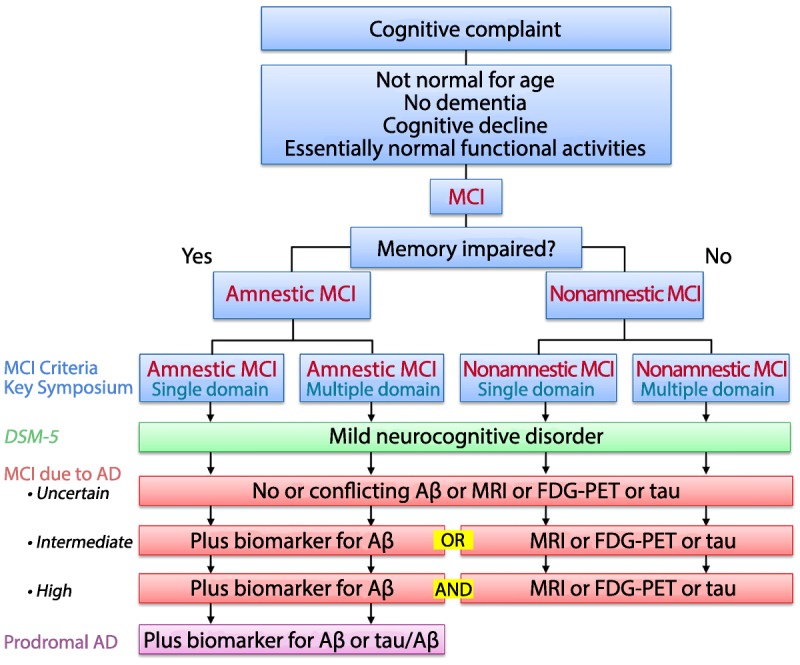
Comparison of common criteria used to characterize mild cognitive impairment (MCI) in various publications. The biomarkers for amyloid-β (Aβ) or tau could be derived from either positron emission tomography (PET) imaging or CSF to accompany the clinical syndromes described above. AD = Alzheimer disease; CSF = cerebrospinal fluid; DSM-5 = Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; FDG-PET = fluorodeoxyglucose positron emission tomography; MRI = magnetic resonance imaging.Reprinted with permission from Petersen RC, et al, J Intern Med.4 onlinelibrary.wiley.com/doi/10.1111/j.1365-2796.2004.01388.x/full#b36. © 2014 The Association for the Publication of the Journal of Internal Medicine.
As is apparent, sufficient overlap exists among these various sets of terminologies, which may reflect the reality that the core features of MCI correspond to the earliest symptomatic stages of a variety of cognitive disorders.4 Figure 2-3 attempts to characterize the common features of these various sets of criteria. Most of the current sets of criteria, regardless of the terminologies, emanate from the Key Symposium criteria for MCI, and most recent approaches embellish these criteria with pathophysiologic biomarkers to lend specificity to the underlying diagnoses. The entire field of aging and dementia is moving in this direction, and these criteria are likely to be important going forward.
Figure 2-2.
Temporal evolution of criteria for mild cognitive impairment (MCI) and prodromal Alzheimer disease (AD). DSM-5 = Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; NIA-AA = National Institute on Aging–Alzheimer’s Association.
PREVALENCE
In the past decade, there have been numerous epidemiologic studies conducted on the prevalence of MCI and the incidence of cognitively normal persons progressing to MCI.21–29 There has been a great deal of variability in the prevalence figures due to methodological variation in the studies and the different implementations of the criteria.4 For example, some of the factors to be considered include the breadth of the MCI—such as all MCI, amnestic MCI, nonamnestic MCI—as well as the methods of data gathering. In addition, factors such as the retrospective application of criteria to previously collected clinical data, versus prospective design using established criteria and applying them as subjects are enrolled in the study, can affect prevalence figures. In general, the prospectively designed studies that establish criteria prior to labeling the participants as having MCI are more reliable and valid. Studies that apply MCI criteria to previously collected data can generate a variety of figures based on the cutoff scores that are used to define MCI. Therefore, since MCI is a clinical diagnosis informed by neuropsychological data, a prospective study is preferred when interpreting epidemiologic data.
Numerous international studies have been completed involving several thousand subjects, and these studies tend to estimate the overall prevalence of MCI in the 12% to 18% range in persons over the age of 60 years.21–26 The Mayo Clinic Study of Aging, which is a population-based study in Olmsted County, Minnesota, found the overall prevalence of MCI to be 16% in residents age 70 years and older.27 MCI is clearly an age-related condition, and to the extent that the evaluation suggests a degenerative etiology, AD is most likely.30
A similar situation pertains to the normal cognition to MCI transition, with certain methodological issues lending themselves to some of the variation. Several longitudinal epidemiologic studies have followed cognitively normal subjects sufficiently long enough to characterize the progression rate, which also can be variable.28,31 The Mayo Clinic Study of Aging followed subjects 70 years and older for a median of 5 years and found the progression rate to be in the 5% to 6% per year range.28 The rates are lower in younger subjects and rise considerably with age. All these data speak to the frequency of the condition of MCI and why it is important to recognize in clinical practice.
CLINICAL EVALUATION
If a clinician adopts the flowchart for fulfilling the criteria outlined in Figure 2-1, the MCI diagnosis for a patient can be approached as follows. At the top of the figure, presuming the patient presents with a cognitive concern, the clinician is then faced with the question of how to evaluate this symptom. Obtaining a history from the patient and confirming this history with someone who knows the patient well is critical. It is important that a cognitive concern is elicited either on the part of the patient, the patient’s informant, or the physician. The cognitive concern is important since it reflects a change in the person’s performance. That is, MCI is not meant to reflect lifelong low cognitive function; rather, it is meant to reflect a change for this individual person. As such, in the absence of formal longitudinal cognitive data on an individual, the clinical history is critical. Here, the clinician should focus on the types of cognitive changes the patient has noted. If the primary concern is in the memory domain, the physician should focus on instances of forgetfulness that are relatively new (ie, appearing approximately in the last 6 months to a year). In particular, the physician should be interested in instances of forgetfulness involving recently experienced events, appointments, visits of friends, or conversations. If the patient is starting to repeat himself or herself, then this is an index that some memory deficits are evolving. Again, the clinician should focus on a change in memory or cognitive function for that individual.
Next, the clinician should explore the breadth of the cognitive concern. Is the concern related to memory alone, or does it involve memory and other cognitive domains such as attention and concentration? Patients will often describe cognitive changes in the domain of memory when, in fact, they may mean attention or language problems. The breadth of the cognitive change is important to characterize. At this point, the clinician will need to do a mental status examination and explore the various cognitive domains. If time is a limitation, a mental status assessment involving an instrument such as the Montreal Cognitive Assessment (MoCA) or the Short Test of Mental Status can be useful, but the clinician must be mindful that these screening instruments are insufficient to make the diagnosis; nevertheless, they can be important to isolate domains of impairment and advise the clinician on further assessments.32,33 If the primary question with the patient concerns the differentiation between the patient’s symptoms and the changes in cognitive function seen in normal aging, neuropsychological testing can be particularly helpful. The neuropsychologist can characterize the profile of cognitive function and assess whether the level of function is appropriate for the patient’s age, sex, and education. If this is not available to the clinician, then the clinician must make the best estimate of function possible.
Other elements in the history to be assessed include functional performance, which, in a patient with MCI, should indicate that the patient’s daily function is largely preserved. The patient may be inefficient at doing certain tasks and may take more time but ultimately can do them without any assistance. This type of assessment fulfills the preserved cognitive function aspect of the criteria. This can be a very subjective assessment, and here, a reliable informant can be helpful. However, if the person is still functioning in daily life, driving, paying bills, doing taxes, and to the casual observer appears normal, generally speaking, function is preserved. Finally, given all of these considerations, the patient does not meet criteria for dementia. That is, the person’s cognitive impairments are not of sufficient severity to compromise daily functioning. Hence, the chief criterion for dementia is not met.
The clinician following the flowchart in Figure 2-1 should determine whether the person has experienced a change in cognition, whether there is some objective corroboration of this, whether function is relatively well preserved, and whether the patient does or does not meet criteria for dementia. If MCI is determined to be a reasonable diagnosis, the clinician then needs to determine if memory is a salient part of the cognitive impairment, and if so, the arm in the diagram in Figure 2-1 that outlines amnestic MCI would be appropriate. If, however, the person is experiencing a cognitive decline but memory is relatively well preserved, then the nonamnestic arm of the diagram would apply.
After the clinical syndrome has been determined, as outlined above, then the clinician needs to determine the cause or etiology of that syndrome. Figure 2-1 characterizes possible explanations of the various clinical syndromes and can help determine the further diagnostic workup. If the history of the onset of the disorder is slow and gradual, a degenerative disease is a likely candidate. Alternatively, if the patient has a history of vascular risk factors and has experienced cerebral ischemic events, a vascular contribution needs to be considered. In addition, some aspects of psychiatric conditions such as major depression or generalized anxiety disorder can have cognitive components, and consequently, in the early stages of these disorders, cognition may be impaired. The clinician must always consider other medical conditions such as uncompensated heart failure, poorly controlled diabetes mellitus, or chronic obstructive pulmonary disease as contributors to cognitive impairment. Some of these medical comorbidities may be treatable, and their medications may play a role in the clinical syndrome as well.
If the clinician believes that a degenerative condition is most likely the underlying explanation, then the clinical syndromes can be useful in suggesting an underlying diagnosis. If the patient appears to have a typical amnestic syndrome leading to MCI and is in the appropriate age range, AD is a likely consideration. However, if the patient is experiencing attention, concentration, and visuospatial difficulties, a forme fruste of dementia with Lewy bodies might be considered, and if the person is experiencing behavioral changes, inappropriate behavior, apathy, lack of insight, and attention and concentration are impaired, frontotemporal lobar degeneration may be possible. Of course, the most common degenerative disease of aging, AD, can have atypical presentations involving attention, concentration, and language.
Based on the history and the examination, the clinician may be able to make a likely supposition regarding the nature of the condition. At this point, further testing such as an MRI scan, fluorodeoxyglucose positron emission tomography (FDG-PET) or positron emission tomography (PET) for amyloid imaging along with a CSF analysis could be considered. These further assessments may assist in depicting the underlying etiology of the clinical syndromes. While no pharmacologic therapies are currently approved by the US Food and Drug Administration (FDA) for MCI due to AD, lifestyle modifications and cognitive and behavioral therapies can be useful. Also, counseling patients on expectations can be quite important. The contribution of medical comorbidities, as outlined previously, including sleep disorders such as sleep apnea, also need to be considered since some of these have treatable components (Case 2-1B).
Case 2–1B
A mental status examination was performed, and while the patient did quite well, there was a suggestion of memory impairment with impaired delayed recall of the words. Neuropsychological testing was pursued, which showed a profile that looked normal for his age, sex, and education in virtually all cognitive domains except for memory. Here, his delayed recall of lists, paragraphs, and nonverbal materials was mildly impaired.
Further interview of the patient and a family member revealed that his function was largely preserved. In particular, he functioned in the community quite well without difficulty, and while slightly more inefficient at some tasks, he still completed everything quite well.
Comment. Based on examination findings, neuropsychological testing, and discussions with the patient and his family member, he does not appear to have dementia. A reasonable diagnosis at this point would be amnestic MCI, but the etiology, given the patient’s young age and negative family history, is uncertain. At this point, a discussion with the patient might include possible etiologies and education about lifestyle alterations, planning for the future, and consideration of enrollment in randomized controlled trials.
PREDICTORS OF PROGRESSION
There has been a great deal of data generated in recent years concerning the progression of persons diagnosed with MCI. In particular, there are clinical variables such as severity of cognitive impairment that predict progression, and many imaging and fluid biomarker studies.30,34 The Alzheimer’s Disease Neuroimaging Initiative (ADNI) has been very active for the past 10 years evaluating individuals with amnestic MCI who have been followed for several years. These data as well as others indicate that, in general, medial temporal lobe atrophy on MRI tends to predict progression as does a hypometabolic pattern consistent with AD on FDG-PET.34–37 Figure 2-4 shows individuals who are cognitively normal along with those who have MCI and those who have dementia on MRI scans, FDG-PET, amyloid PET, and the newest imaging modality, tau PET. Several studies have indicated that individuals with MCI who have a positive amyloid PET scan are more likely to progress rapidly and, again, ADNI data confirm this.38,39 For many years, it has been known that carriers of the apolipoprotein E4 (APOE4) genotype are more likely to progress rapidly, and this has been borne out in numerous studies; however, in clinical practice APOE testing does not add significantly to the diagnostic evaluation.34,40
Figure 2-4.
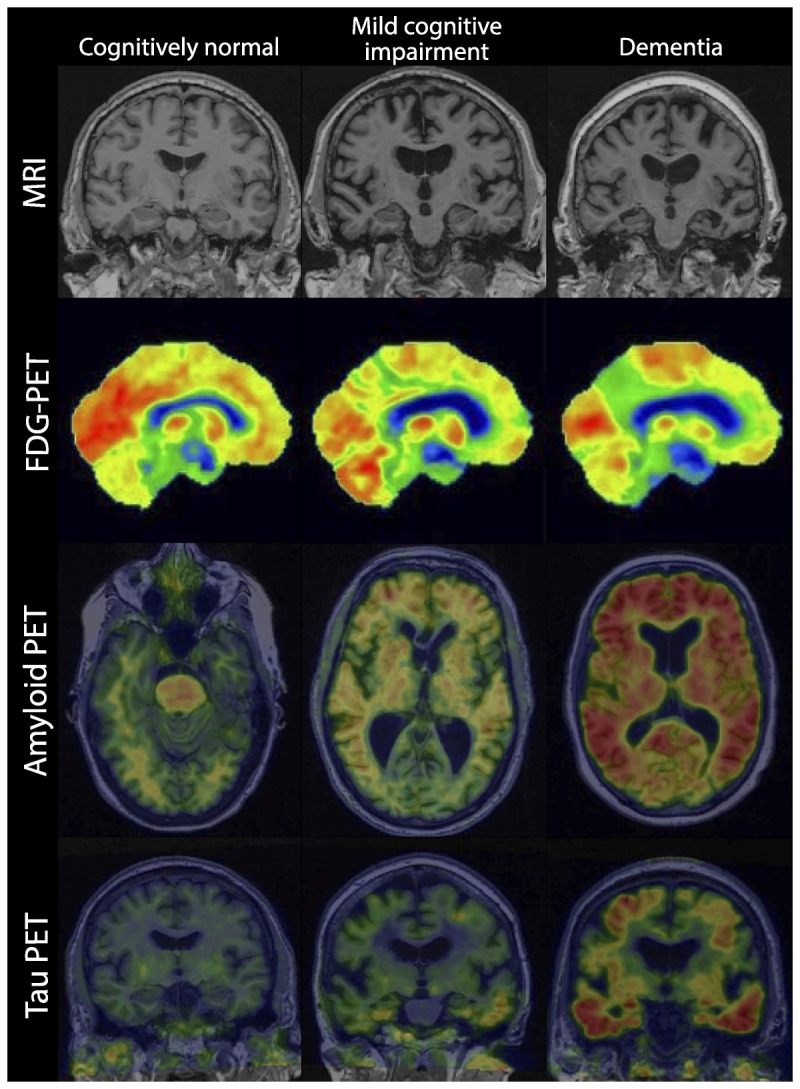
Progression of imaging features from cognitively normal to mild cognitive impairment to dementia.
FDG-PET = fluorodeoxyglucose positron emission tomography; MRI = magnetic resonance imaging; PET = positron emission tomography.
The newest PET tracer that is emerging allows investigators to evaluate the role of tau in clinical progression, and these data are evolving.41 It is quite likely that a tau PET scan that shows the spread of tau outside of the medial temporal lobe into lateral temporal lobe structures portends a poorer prognosis and, more likely, a rapid progression from MCI to AD dementia, but these data need to be amplified (Figure 2-4).
Numerous studies have shown that the various CSF markers consistent with AD predict progression to AD dementia.42,43 The 2006 study by Hansson and colleagues44 was most informative with regard to these data and corroborates the suspicion that those individuals, particularly with amnestic MCI, who harbor low CSF levels of Aβ42 and elevated total tau and phosphorylated tau are at an increased risk for progressing more rapidly than those subjects with the same clinical phenotype but normal CSF biomarkers.
In general, these predictors all refer to individuals who are on the AD spectrum. Biomarkers for other degenerative disorders are less certain at this point and need to be fully evaluated. As biomarkers for other disorders evolve, it is likely that they will be matched with a particular phenotype of MCI and increase the ability to predict the clinical outcome.
CRITERIA FOR MILD COGNITIVE IMPAIRMENT DUE TO ALZHEIMER DISEASE
The NIA and the Alzheimer’s Association convened groups of experts to revise the criteria for AD across the spectrum.8 The groups divided the AD spectrum into three overlapping areas: preclinical AD in which individuals are clinically normal but possess biomarker evidence for the AD process, MCI due to AD whereby individuals meet the clinical criteria for MCI and have varying levels of biomarker specificity for AD, and AD dementia in which individuals meet clinical criteria for dementia and similarly have varying degrees of biomarker support.1 These criteria have proved useful in characterizing the entire spectrum of AD and expanding it beyond the dementia phase. Prior sets of criteria had focused only on the dementia phase, but it has become apparent in recent years that the AD process likely begins years and perhaps decades before clinical symptoms appear.45 As such, the use of biomarkers has afforded an important tool for specificity for the clinician. It must be emphasized, however, that these criteria involving biomarkers are still in the research phase, and specific recommendations regarding their use in clinical practice remain to be determined. However, based on the literature cited above, it is apparent that the use of biomarkers is working its way into clinical practice.
In addition, the American Psychiatric Association has recently published the DSM-5, and this set of criteria gives consideration to the diagnosis of mild neurocognitive disorder. In general, the term neurocognitive disorder is used for the syndromes of cognitive impairment irrespective of specific etiology.17 The spectrum was divided into mild neurocognitive disorder, which is very similar to MCI, and major neurocognitive disorder, which is very similar to dementia. The criteria for mild neurocognitive disorder essentially incorporate the MCI criteria from the Key Symposium in 2004 and suggest that, as data accumulate, specificity of etiology will be added through the use of biomarkers. After the syndrome of either mild or major neurocognitive disorder is made, DSM-5 then gives recommendations as to how the clinician can determine the underlying etiology of the syndrome.17 As such, conditions such as AD, frontotemporal lobar degeneration, dementia with Lewy bodies, vascular cognitive impairment, human immunodeficiency (HIV)-related disorders, alcohol and substance abuse, Parkinson disease, and a variety of other possible etiologies are explicated. As noted above, the process of combining the clinical syndrome with patient history, clinical examination findings, and, while still evolving, biomarker information is used to specify the clinical syndrome. As is seen in DSM-5, there are often degrees of certainty added to the clinical diagnoses based on the preponderance of evidence for a specific underlying disorder.17
In 2007 and updated in 2010 and 2013, investigators have proposed the term prodromal AD as an alternative for characterizing individuals along the AD spectrum.18–20 The most recent version of these criteria embodies the notion of amnestic MCI and embellishes it with evidence for amyloid deposition via PET scanning or amyloid and tau information using CSF.20 These investigators contend that the combination of amnestic MCI with specific biomarkers is highly suggestive of the AD process and should be labeled as AD. These criteria have been useful for randomized control trials for evaluation of pharmacologic therapeutics intended to target underlying AD pathophysiology.46,47
TREATMENTS
Currently, there are no accepted pharmacologic treatments for MCI approved by the FDA, the European Medicines Agency, or the Pharmaceuticals and Medical Devices Agency in Japan. Numerous randomized control trials have been conducted in the MCI spectrum, but none has been successful at demonstrating effectiveness at delaying the progression from MCI to AD dementia.10–14 One of the first trials, conducted by the Alzheimer’s Disease Cooperative Study, evaluated donepezil and high-dose vitamin E in amnestic MCI.13 This study indicated that donepezil may be effective at slowing the rate of progression in all subjects with amnestic MCI for the first year of the trial and perhaps up to 2 years in subjects with amnestic MCI who were positive for the APOE4 isoform. However, since the study was designed to continue for 36 months, no treatments demonstrated effectiveness at that time and, as such, the trial was negative. Other studies involving cholinesterase inhibitors that have been used for the treatment of AD dementia were also unsuccessful.10,11,13
Lifestyle modifications and other nonpharmacologic therapies have also been investigated, and there is a suggestion that some of these modifications or therapies, such as aerobic exercise, may be effective at reducing the rate of progression from MCI to dementia.48 However, a state-of-the-science report from the National Institutes of Health (NIH) in 2010 failed to document any successful interventions for progression to dementia.49 However, subsequent data suggest that there may be some efficacy to be gleaned from lifestyle modifications, and these need to be explored further.
CLINICAL ACCEPTANCE
The construct of MCI has been in the medical literature for many years and, over time, has been accepted in clinical practice to certain degrees. The American Academy of Neurology (AAN) completed an evidence-based medicine review of the literature and concluded that the construct of MCI is useful for clinicians to identify since the condition does lead to a higher risk of progression to dementia.50 Numerous epidemiologic studies have been done around the world that have suggested the utility of the identification of MCI as a clinical entity.
Roberts and colleagues51 evaluated members of the Behavioral Neurology section and the Geriatric Neurology section of the AAN and documented that MCI is used frequently in clinical practice and that practitioners find the construct useful. There were concerns about its specificity and the lack of treatments, but, nevertheless, it was believed to be useful.
Recently, a similar exercise was done in Europe assessing the utility of the construct of MCI in clinical practice and the outcome was quite similar to that found in polling members of the AAN. As such, it appears that the construct of MCI is accepted in clinical practice and serves a function for clinicians in communicating clinical diagnoses to patients. It has also been a useful construct for research as literally thousands of studies have been generated in the past decade assessing various aspects of the condition.
This is not to say, however, that there is not controversy surrounding the construct. Criticism has been raised regarding the boundaries of the condition with respect to differentiating MCI from changes of cognitive aging and also from dementia. Since cognitive changes with aging are believed to be common, it can be challenging to determine whether a particular person is experiencing those changes or the forme fruste of MCI. A recent publication from the Institute of Medicine has characterized the expectations of cognitive aging, and a distinction is made between these changes and those that represent incipient MCI.52 However, there is a great deal of variability in the literature regarding MCI, and many of these features have been reviewed recently.4 There have been controversies regarding the implementation of MCI criteria involving neuropsychological tests, cutoff scores, types of measures, and cross-sectional versus longitudinal assessment. There has been variability in methodology of studies including the subjects sampled for the studies, how they were evaluated (eg, prospectively or retrospectively), and the availability of normative data against which to make comparisons for cognitive performance. All of these issues, and more, are very salient and require the clinician to use judgment in characterizing individuals with MCI. Nevertheless, a potential asset of a condition such as MCI will be validated when effective treatments are developed, particularly for MCI due to AD.
No doubt, there will be additional research generated regarding the construct of MCI, and these data should help clarify many of the issues surrounding uncertainty. The clinicians must recognize that these arbitrary distinctions in clinical state, particularly when dealing with underlying degenerative conditions and their continua, are artificial. Nevertheless, in communicating with patients and with other clinicians, these constructs can be useful.
CONCLUSION
MCI has become an important topic in clinical practice and research. With the emphasis on earlier identification of incipient forms of cognitive deficits, MCI put boundaries on this intermediate state. Numerous randomized controlled trials are underway for the MCI subtype due to AD, and clinicians need to be aware of these research opportunities for their patients. As treatable etiologies of MCI are identified (such as those due to psychiatric conditions, medications, or medical comorbidities), the cognitive deficits may be reversible. Biomarker development should provide the clinician with new tools to identify and hopefully treat MCI.
KEY POINTS
The Key Symposium criteria for mild cognitive impairment accomplished two goals: (1) to broaden the classification scheme beyond memory, and (2) to recognize that mild cognitive impairment could result from a variety of etiologies and not just Alzheimer disease.
Traditionally, amnestic mild cognitive impairment is the typical prodromal stage of dementia due to Alzheimer disease, but other phenotypes can also lead to this type of dementia, such as logopenic aphasia, posterior cortical atrophy (also known as the visual variant), or a frontal lobe–dysexecutive presentation of Alzheimer disease.
Not all mild cognitive impairment is early Alzheimer disease.
The criteria for mild cognitive impairment due to Alzheimer disease developed by the National Institute on Aging and the Alzheimer’s Association essentially adopted the Key Symposium criteria while making some of the diagnostic features more explicit. These criteria also added biomarkers for underlying Alzheimer disease pathophysiology in an attempt to refine the underlying etiology and, hence, predict outcome.
In the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, for the general category of neurocognitive disorders, the criteria now include a predementia phase called mild neurocognitive disorder.
The construct of prodromal Alzheimer disease evolved from the accumulating literature that had developed regarding the observation that amnestic mild cognitive impairment, when paired with certain biomarkers, approximated the condition of Alzheimer disease. In fact, the proponents believed that a certain type of amnestic mild cognitive impairment coupled with biomarkers for the presence of amyloid or amyloid and tau constituted the earliest symptomatic stages of the Alzheimer disease process.
The multiple sets of criteria referring to mild cognitive impairment actually contain many of the same elements and are quite similar to the original Key Symposium criteria.
Numerous international studies have been completed involving several thousand subjects, and these studies tend to estimate the overall prevalence of mild cognitive impairment in the 12% to 18% range in persons over the age of 60 years.
The Mayo Clinic Study of Aging followed subjects 70 years and older for a median of 5 years and found the progression rate of mild cognitive impairment to be in the 5% to 6% per year range.
Mild cognitive impairment is not meant to reflect lifelong low cognitive function; rather, it is meant to reflect a change for this individual person.
In mild cognitive impairment, the patient’s daily function is largely preserved.
Several studies have indicated that individuals with mild cognitive impairment who have a positive amyloid positron emission tomography scan are more likely to progress rapidly, which is confirmed by data from the Alzheimer’s Disease Neuroimaging Initiative.
The National Institute on Aging and the Alzheimer’s Association divided the Alzheimer disease spectrum into three overlapping areas: preclinical Alzheimer disease in which individuals are clinically normal but possess biomarker evidence for the Alzheimer disease process, mild cognitive impairment due to Alzheimer disease whereby individuals meet the clinical criteria for mild cognitive impairment and have varying levels of biomarker specificity for Alzheimer disease, and AD dementia in which individuals meet clinical criteria for dementia and similarly have varying degrees of biomarker support.
The spectrum of neurocognitive disorder, as defined by the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, is divided into mild neurocognitive disorder, which is very similar to mild cognitive impairment, and major neurocognitive disorder, which is very similar to dementia.
Currently, there are no accepted pharmacologic treatments for mild cognitive impairment approved by the US Food and Drug Administration, the European Medicines Agency, or the Pharmaceuticals and Medical Devices Agency in Japan.
Lifestyle modifications and other nonpharmacologic therapies have also been investigated, and there is a suggestion that some of these modifications or therapies, such as aerobic exercise, may be effective at reducing the rate of progression from mild cognitive impairment to dementia.
Criticism has been raised regarding the boundaries of the condition of mild cognitive impairment with respect to differentiating it from changes of cognitive aging and also differentiating it from dementia.
ACKNOWLEDGMENTS
The author receives grant support from the National Institute on Aging (P50 AG016574, U01 AG006786, U01 AG024904, R01 AG011378, and R01 AG041581).
REFERENCES
- 1. Jack CR, Jr, Albert MS, Knopman DS, et al. Introduction to the recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s & dementia: the journal of the Alzheimer’s Association 2011; 7(3): 257– 262. doi:10.1016/j.jalz.2011.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med 2004; 256(3): 183– 194. doi:10.1111/j.1365-2796.2004.01388.x. [DOI] [PubMed] [Google Scholar]
- 3. Petersen RC. Clinical practice. Mild cognitive impairment. N Engl J Med 2011; 364(23): 2227– 2234. doi:10.1056/NEJMcp0910237. [DOI] [PubMed] [Google Scholar]
- 4. Petersen RC, Caracciolo B, Brayne C, et al. Mild cognitive impairment: a concept in evolution. J Intern Med 2014; 275(3): 214– 228. doi:10.1111/joim.12190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Reisberg B, Ferris S, de Leon MJ. Stage-specific behavioral, cognitive, and in vivo changes in community residing subjects with age-associated memory impairment and primary degenerative dementia of the Alzheimer type. Drug Dev Res 1988; 15(2–3): 101– 114. doi:10.1002/ddr.430150203. [Google Scholar]
- 6. Petersen RC, Smith GE, Waring SC, et al. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol 1999; 56(3): 303– 308. doi:10.1001/archneur.56.3.303. [DOI] [PubMed] [Google Scholar]
- 7. Winblad B, Palmer K, Kivipelto M, et al. Mild cognitive impairment—beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med 2004; 256(3): 240– 246. doi:10.1111/j.1365-2796.2004.01380.x. [DOI] [PubMed] [Google Scholar]
- 8. Petersen RC.Mild Cognitive Impairment. Continuum (Minneap Minn) 2004; 10(1 Dementia): 9– 28. doi:10.1212/01.CON.0000293545.39683.cc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Albert MS, DeKosky ST, Dickson D, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011; 7(3): 270– 279. doi:10.1016/j.jalz.2011.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Doody RS, Ferris SH, Salloway S, et al. Donepezil treatment of patients with MCI: a 48-week randomized, placebo-controlled trial. Neurology 2009; 72(18): 1555– 1561. doi:10.1212/01.wnl.0000344650.95823.03. [DOI] [PubMed] [Google Scholar]
- 11. Feldman HH, Ferris S, Winblad B, et al. Effect of rivastigmine on delay to diagnosis of Alzheimer’s disease from mild cognitive impairment: the InDDEx study. Lancet Neurol 2007; 6(6): 501– 512. doi:10.1016/S1474-4422(07)70109-6. [DOI] [PubMed] [Google Scholar]
- 12. Thal LJ, Ferris SH, Kirby L, et al. A randomized, double-blind, study of rofecoxib in patients with mild cognitive impairment. Neuropsychopharmacology 2005; 30(6): 1204– 1215. doi:10.1038/sj.npp.1300690. [DOI] [PubMed] [Google Scholar]
- 13. Winblad B, Gauthier S, Scinto L, et al. Safety and efficacy of galantamine in subjects with mild cognitive impairment. Neurology 2008; 70(22): 2024– 2035. doi:10.1212/01.wnl.0000303815.69777.26. [DOI] [PubMed] [Google Scholar]
- 14. Petersen RC, Thomas RG, Grundman M, et al. Vitamin E and donepezil for the treatment of mild cognitive impairment. N Engl J Med 2005; 352(23): 2379– 2388. doi:10.1056/NEJMoa050151. [DOI] [PubMed] [Google Scholar]
- 15. McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011; 7(3): 263– 269. doi:10.1016/j.jalz.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sperling RA, Aisen PS, Beckett LA, et al. Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Assocation workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011; 7(3): 280– 292. doi:10.1016/j.jalz.2011.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.American Psychiatric Association. Diagnostic and statistical manual of mental disorders, fifth edition Washington, DC: American Psychiatric Publishing, 2013. [Google Scholar]
- 18. Dubois B, Feldman HH, Jacova C, et al. Research criteria for the diagnosis of Alzheimer’s disease: revising the NINCDS-ADRDA criteria. Lancet Neurol 2007; 6(8): 734– 746. doi:10.1016/S1474-4422(07)70178-3. [DOI] [PubMed] [Google Scholar]
- 19. Dubois B, Feldman HH, Jacova C, et al. Revising the definition of Alzheimer’s disease: a new lexicon. Lancet Neurol 2010; 9(11): 1118– 1127. doi:10.1016/S1474-4422(10)70223-4. [DOI] [PubMed] [Google Scholar]
- 20. Dubois B, Feldman HH, Jacova C, et al. Advancing research diagnostic criteria for Alzheimer’s disease: the IWG-2 criteria. Lancet Neurol 2014; 13(6): 614– 629. doi:10.1016/S1474-4422(14)70090-0. [DOI] [PubMed] [Google Scholar]
- 21. Busse A, Hensel A, Gühne U, et al. Mild cognitive impairment: long-term course of four clinical subtypes. Neurology 2006; 67(12): 2176– 2185. doi:10.1212/01.wnl.0000249117.23318.e1. [DOI] [PubMed] [Google Scholar]
- 22. Di Carlo A, Lamassa M, Baldereschi M, et al. CIND and MCI in the Italian elderly: frequency, vascular risk factors, progression to dementia. Neurology 2007; 68(22): 1909– 1916. doi:10.1212/01.wnl.0000263132.99055.0d. [DOI] [PubMed] [Google Scholar]
- 23. Ganguli M, Chang CC, Snitz BE, et al. Prevalence of mild cognitive impairment by multiple classifications: The Monongahela-Youghiogheny Healthy Aging Team (MYHAT) project. Am J Geriatr Psychiatry 2010; 18(8): 674– 683. doi:10.1097/JGP.ob013e3181cdee4f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Larrieu S, Letenneur L, Orgogozo JM, et al. Incidence and outcome of mild cognitive impairment in a population-based prospective cohort. Neurology 2002; 59(10): 1594– 1599. doi:10.1212/01.WNL.0000034176.07159.F8. [DOI] [PubMed] [Google Scholar]
- 25. Lopez OL, Jagust WJ, DeKosky ST, et al. Prevalence and classification of mild cognitive impairment in the Cardiovascular Health Study Cognition Study: part 1. Arch Neurol 2003; 60(10): 1385– 1389. doi:10.1001/archneur.60.10.1385. [DOI] [PubMed] [Google Scholar]
- 26. Manly JJ, Tang MX, Schupf N, et al. Frequency and course of mild cognitive impairment in a multiethnic community. Ann Neurol 2008; 63(4): 494– 506. doi:10.1002/ana.21326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Petersen RC, Roberts RO, Knopman DS, et al. Prevalence of mild cognitive impairment is higher in men. The Mayo Clinic Study of Aging. Neurology 2010; 75(10): 889– 897. doi:10.1212/WNL.0b013e3181f11d85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Roberts RO, Geda YE, Knopman DS, et al. The incidence of MCI differs by subtype and is higher in men: the Mayo Clinic Study of Aging. Neurology 2012; 78(5): 342– 351. doi:10.1212/WNL.0b013e3182452862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Unverzagt FW, Gao S, Baiyewu O, et al. Prevalence of cognitive impairment: data from the Indianapolis Study of Health and Aging. Neurology 2001; 57(9): 1655– 1662. doi:10.1212/WNL.57.9.1655. [DOI] [PubMed] [Google Scholar]
- 30. Okello A, Koivunen J, Edison P, et al. Conversion of amyloid positive and negative MCI to AD over 3 years: An 11C-PIB PET Study. Neurology 2009; 73(10): 754– 760. doi:10.1212/WNL.ob013e3181b23564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Lopez OL, Becker JT, Chang YF, et al. Incidence of mild cognitive impairment in the Pittsburgh Cardiovascular Health Study-Cognition Study. Neurology 2012; 79(15): 1599– 1606. doi:10.1212/WNL.0b013e31826e25f0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 2005; 53(4): 695– 699. doi:10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- 33. Kokmen E, Naessens JM, Offord KP. A short test of mental status: description and preliminary results. Mayo Clin Proc 1987; 62(4): 281– 288. doi:10.1016/S0025-6196(12)61905-3. [DOI] [PubMed] [Google Scholar]
- 34. Weiner MW, Veitch DP, Aisen PS, et al. ; Alzheimer’s Disease Neuroimaging Initiative. Impact of the Alzheimer’s Disease Neuroimaging Initiative, 2004 to 2014. Alzheimers Dement 2015; 11(7): 865– 884. doi:10.1016/j.jalz.2015.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Dickerson BC, Salat DH, Greve DN, et al. Increased hippocampal activation in mild cognitive impairment compared to normal aging and AD. Neurology 2005; 65(3): 404– 411. doi:10.1212/01.wnl.0000171450.97464.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Landau SM, Harvey D, Madison CM, et al. Comparing predictors of conversion and decline in mild cognitive impairment. Neurology 2010; 75(3): 230– 238. doi:10.1212/WNL.0b013e3181e8e8b8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Landau SM, Mintun MA, Joshi AD, et al. Amyloid deposition, hypometabolism, and longitudinal cognitive decline. Ann Neurol 2012; 72(4): 578– 586. doi:10.1002/ana.23650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Prestia A, Caroli A, van der Flier WM, et al. Prediction of dementia in MCI patients based on core diagnostic markers for Alzheimer disease. Neurology 2013; 80(11): 1048– 1056. doi:10.1212/WNL.0b013e3182872830. [DOI] [PubMed] [Google Scholar]
- 39. Petersen RC, Aisen P, Boeve BF, et al. Mild cognitive impairment due to Alzheimer disease in the community. Ann Neurol 2013; 74(2): 199– 208. doi:10.1002/ana.23931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Petersen RC, Smith GE, Ivnik RJ, et al. Apolipoprotein E status as a predictor of the development of Alzheimer’s disease in memory-impaired individuals. JAMA 1995; 273(16): 1274– 1278. doi:10.1001/jama.1995.03520400044042. [PubMed] [Google Scholar]
- 41. Johnson KA, Schultz A, Betensky RA, et al. Tau positron emission tomographic imaging in aging and early Alzheimer disease. Ann Neurol 2016; 79(1): 110– 119. doi:10.1002/ana.24546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Mattsson N, Insel PS, Donohue M, et al. ; Alzheimer’s Disease Neuroimaging Initiative. Predicting reduction of cerebrospinal fluid β-amyloid 42 in cognitively healthy controls. JAMA Neurol 2015; 72(5): 554– 560. doi:10.1001/jamaneurol.2014.4530. [DOI] [PubMed] [Google Scholar]
- 43. Mattsson N, Zetterberg H, Hansson O, et al. CSF biomarkers and incipient Alzheimer disease in patients with mild cognitive impairment. JAMA 2009; 302(4): 385– 393. doi:10.1001/jama.2009.1064. [DOI] [PubMed] [Google Scholar]
- 44. Hansson O, Zetterberg H, Buchhave P, et al. Association between CSF biomarkers and incipient Alzheimer’s disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurol 2006; 5(3): 228– 234. doi:10.1016/S1474-4422(06)70355-6. [DOI] [PubMed] [Google Scholar]
- 45. Rowe CC, Ng S, Ackermann U, et al. Imaging beta-amyloid burden in aging and dementia. Neurology 2007; 68(20): 1718– 1725. doi:10.1212/01.wnl.0000261919.22630.ea. [DOI] [PubMed] [Google Scholar]
- 46. Perry D, Sperling R, Katz R, et al. Building a roadmap for developing combination therapies for Alzheimer’s disease. Expert Rev Neurother 2015; 15(3): 327– 333. doi:10.1586/14737175.2015.996551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Romero K, Ito K, Rogers JA, et al. ; Alzheimer’s Disease Neuroimaging Initiative; Coalition Against Major Diseases. The future is now: model-based clinical trial design for Alzheimer’s disease. Clin Pharmacol Ther 2015; 97(3): 210– 214. doi:10.1002/cpt.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Lautenschlager NT, Cox KL, Flicker L, et al. Effect of physical activity on cognitive function in older adults at risk for Alzheimer disease: a randomized trial. JAMA 2008; 300(9): 1027– 1037. doi:10.1001/jama.300.9.1027. [DOI] [PubMed] [Google Scholar]
- 49.National Institute of Health Office of the Director. NIH state-of-the-science conference statement on preventing Alzhimer’s disease and cognitive decline. Volume 27, number 4 Updated April 26–28, 2010. Accessed February 9, 2016. consensus.nih.gov/2010/docs/alz/alz_stmt.pdf [Google Scholar]
- 50. Petersen RC, Stevens JC, Ganguli M, et al. Practice parameter: early detection of dementia: mild cognitive impairment (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2001; 56(9): 1133– 1142. doi:10.1212/WNL.56.9.1133. [DOI] [PubMed] [Google Scholar]
- 51. Roberts JS, Karlawish JH, Uhlmann WR, et al. Mild cognitive impairment in clinical care: a survey of American Academy of Neurology members. Neurology 2010; 75(5): 425– 431. doi:10.1212/WNL.0b013e3181eb5872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Institute of Medicine. Cognitive aging: progress in understanding and opportunities for action. Washington, DC: The National Academies Press, 2015. [PubMed] [Google Scholar]