Abstract
IN BRIEF Chronic liver disease (CLD) and type 2 diabetes have both been linked to increased morbidity and mortality. In this study, the impact of CLD and diabetes on all-cause mortality was quantified at the population level using U.S. population data. Both type 2 diabetes and CLD were found to be independently associated with increased mortality (age-adjusted hazard ratio [aHR] 1.98 and 1.37 for diabetes and CLD, respectively), and having both diabetes and CLD substantially increased the risk of mortality (aHR 2.41).
Data from the Centers for Disease Control and Prevention’s National Center for Health Statistics suggest that the prevalence of both chronic liver disease (CLD) and diabetes has been rising (1–5). In addition to being common causes of morbidity, both CLD and type 2 diabetes can be important causes of mortality. Indeed, in 2013, diabetes and CLD were the sixth and twelfth leading causes of death, respectively, in the United States (1,2,5).
The most important factor responsible for fueling the epidemics of diabetes and CLD is obesity and its related diseases. Obesity and type 2 diabetes are both associated with nonalcoholic fatty liver disease (NAFLD) and also tend to make the course of other liver diseases more aggressive (3–7). For example, patients with hepatitis C virus (HCV) or other liver diseases and diabetes have higher rates of progression to cirrhosis and hepatocellular carcinoma (8–32).
Because the prevalence rates of both CLD and type 2 diabetes have increased, patients presenting with both diseases are becoming more common. Although data from tertiary care medical centers have suggested that the combination of CLD and diabetes is associated with adverse outcomes (8–32), this issue has not been fully assessed at the population level. In this study, we examined U.S. population data linked to more than 20 years of mortality follow-up with the aim of assessing the impact of type 2 diabetes and CLD on overall mortality.
Methods
Population
The Third National Health and Nutrition Examination Survey (NHANES III) used in this study is a nationwide survey of the noninstitutionalized civilian U.S. population conducted between 1988 and 1994. It includes interviews, standardized physical examinations, and data from blood and urine samples collected from enrolled participants. After demographics-based weighting of the study participants, the sample is representative of the U.S. population. Included participants were ≥18 years of age at the time of examination for NHANES III.
Definitions
For the purpose of this study, type 2 diabetes was defined as a fasting glucose ≥126 mg/dL or self-reported use of oral hypoglycemic agents or insulin.
For assessment of CLD status, we introduced the definitions of excessive alcohol consumption (self-reported use of >20 g/day for men and >10 g/day for women for at least 1 year) and elevated serum aminotransferases (alanine aminotransferase [ALT] >40 U/L or aspartate aminotransferase [AST] >37 U/L in men and ALT or AST >31 U/L in women). CLD was defined as having one of five major etiologies: alcoholic liver disease (ALD, defined as excessive alcohol consumption accompanied by elevated serum aminotransferases), chronic hepatitis B virus (HBV, defined as hepatitis B surface antigen [HBsAg] positivity), HCV (defined as anti-HCV antibody positivity), iron overload (transferrin saturation >50%), or NAFLD (elevated serum aminotransferases in the absence of any other evidence of CLD). Subjects without evidence of liver disease by our inclusion criteria were considered to have no CLD.
All NHANES III participants who were ≥18 years of age had follow-up mortality status calculated using matched data from the U.S. National Death Index (33). A publicly available version of the NHANES III–linked mortality file with individual subjects’ causes of death hidden was used. Mortality follow-up was calculated in months from the day of examination until the date of death or until the end of follow-up, which was 31 December 2011. Subjects with-out available mortality status were excluded.
Statistical Analysis
All participants who were ≥18 years of age and had available CLD, diabetes, and mortality status were included and grouped into four categories: subjects having both CLD and diabetes, CLD without diabetes, diabetes without CLD, and controls with neither CLD nor diabetes. Demographic and clinical parameters were further compared across these four categories using the stratum-specific χ2 test for independence.
Age-adjusted post-survey mortality was calculated in 6-month periods using the population age distribution from U.S. Census 2000 data separately for each of the four DM/CLD categories. Additionally, using the entire follow-up span, a Cox proportional hazards model was used to evaluate independent associations of CLD and diabetes with mortality after adjustment for age, and age-adjusted hazard ratios (aHRs) were calculated.
Sample weights were used to account for nonresponse and unequal selection probabilities for certain categories of the population. Stratum and sampling units accounted for the survey design effects. P values <0.05 were considered significant. All analyses were run using Standalone SUDAAN 11.0 (Research Triangle Institute, Cary, N.C.).
The study was approved by the Inova institutional review board.
Results
The NHANES III data included 20,050 adult participants. After applying inclusion and exclusion criteria, 16,413 subjects were included in this study (96 degrees of freedom for the stratified sample). Of those, after weighting, 12.2% had CLD, and 5.8% had diabetes. Additionally, 1.0% had both CLD and diabetes, and 83.0% had neither CLD nor diabetes. Of the CLD cohort, 11.3% had ALD, 3.3% had chronic HBV, 17.64% had HCV, 29.26% had iron overload, and 43.59% had presumed NAFLD. The rates of CLD etiologies were similar between CLD patients with and without diabetes except for iron overload (30.91% in those without diabetes vs. 10.63% in those with diabetes) and NAFLD (65.03% in those with diabetes vs. 41.69% in those without diabetes) (both P <0.0001).
Demographics and Age-Adjusted Mortality
The demographic data show that subjects with diabetes were more commonly black and >55 years of age; more likely to be obese, hypertensive, and hyperlipidemic; and had more cardiovascular disease than those without diabetes (Table 1). During follow-up of 240–280 months, 64.0% of the subjects with diabetes and 20.5% of those without diabetes died; the overall age-adjusted mortality rates were 40.1 and 23.4%, respectively (both P <0.0001). In fact, age-adjusted mortality in subjects with diabetes was greater starting 1.5 years after the survey (2.51 vs. 1.12%, respectively, P <0.05) and remained greater throughout follow-up.
TABLE 1.
Clinico-Demographic Data of NHANES III Participants According to Presence of Diabetes and CLD
| Participants Without Diabetes |
Participants With Diabetes |
|||||
|---|---|---|---|---|---|---|
| CLD | No CLD | P | CLD | No CLD | P | |
| n | 1,897 | 13,111 | — | 222 | 1,183 | — |
| Proportion (%) | 11.25 ± 0.48 | 83.00 ± 0.52 | — | 1.00 ± 0.09 | 4.75 ± 0.29 | — |
| Race/ethnicity (%) | ||||||
| Caucasian | 71.64 ± 2.81 | 77.35 ± 2.06 | 0.0002 | 69.60 ± 5.10 | 73.76 ± 3.22 | 0.38 |
| African-American | 10.54 ± 1.19 | 10.43 ± 1.16 | 0.89 | 9.89 ± 2.35 | 14.48 ± 2.00 | 0.06 |
| Hispanic | 7.99 ± 1.40 | 4.69 ± 0.77 | <0.0001 | 8.52 ± 1.75 | 5.44 ± 1.11 | 0.0141 |
| Other | 9.84 ± 1.95 | 7.53 ± 1.10 | 0.09 | 11.98 ± 3.98 | 6.32 ± 1.64 | 0.15 |
| Male (%) | 58.57 ± 1.77 | 46.68 ± 0.57 | <0.0001 | 45.31 ± 5.78 | 49.42 ± 2.54 | 0.53 |
| Age in years (%) | ||||||
| <45 | 69.55 ± 1.84 | 59.38 ± 1.21 | <0.0001 | 26.20 ± 4.75 | 16.31 ± 2.44 | 0.07 |
| 45–54 | 14.66 ± 1.23 | 13.68 ± 0.54 | 0.46 | 21.08 ± 4.31 | 13.69 ± 1.70 | 0.12 |
| 55–64 | 7.41 ± 0.94 | 11.27 ± 0.44 | 0.0002 | 22.24 ± 4.08 | 27.84 ± 2.15 | 0.23 |
| ≥65 | 8.38 ± 0.89 | 15.67 ± 1.03 | <0.0001 | 30.48 ± 5.01 | 42.16 ± 2.18 | 0.0296 |
| Obesity (BMI ≥30 kg/m2) (%) | 23.99 ± 1.49 | 19.74 ± 0.67 | 0.0088 | 52.27 ± 5.73 | 47.00 ± 2.16 | 0.36 |
| Hypercholesterolemia (%) | 64.70 ± 2.08 | 67.86 ± 0.92 | 0.16 | 85.84 ± 3.30 | 86.91 ± 1.94 | 0.75 |
| Hypertension (%) | 21.71 ± 1.67 | 21.20 ± 0.80 | 0.74 | 57.05 ± 5.58 | 56.01 ± 2.08 | 0.87 |
| Current smoking (%) | 37.07 ± 1.78 | 29.50 ± 1.08 | 0.0004 | 22.77 ± 3.91 | 20.67 ± 2.12 | 0.60 |
Proportions are expressed as a percentage ± SD.
Subjects with both diabetes and CLD were more likely to be female, were predominantly >55 years of age, and more commonly had all the components of metabolic syndrome when compared to CLD subjects without diabetes (all P <0.05) (Table 1). Also, during follow-up, subjects with both diabetes and CLD were substantially more likely to die than CLD patients without diabetes (crude 58.19 vs. 18.32%; age-adjusted 44.97 vs. 27.60%, both P <0.001).
Among subjects with diabetes, those who also had CLD were more frequently Hispanic and slightly younger than those who did not have CLD (Table 1). Also, although mortality in people who had both diabetes and CLD was slightly higher than in those who had diabetes alone, this difference was not significant at any time point (Table 2).
TABLE 2.
Age-Adjusted Mortality in NHANES III Participants
| Follow-Up Year | No CLD, No DM Group | CLD, No DM Group | DM, No CLD Group | DM, CLD Group |
|---|---|---|---|---|
| 1 | 0.66 ± 0.08 | 0.65 ± 0.24 | 1.75 ± 0.71 | 1.18 ± 0.62 |
| 3 | 2.30 ± 0.12 | 3.84 ± 0.70 | 4.82 ± 0.82 | 7.55 ± 2.41 |
| 5 | 4.37 ± 0.21 | 7.13 ± 0.87 | 12.19 ± 2.65 | 12.67 ± 3.45 |
| 8 | 7.88 ± 0.25 | 10.51 ± 1.06 | 18.39 ± 2.73 | 22.09 ± 3.80 |
| 10 | 10.44 ± 0.28 | 13.14 ± 1.07 | 20.93 ± 2.81 | 27.19 ± 4.29 |
| 13 | 14.32 ± 0.39 | 18.35 ± 1.05 | 26.05 ± 2.81 | 31.06 ± 4.07 |
| 15 | 16.74 ± 0.43 | 20.62 ± 1.11 | 29.00 ± 2.84 | 37.70 ± 4.45 |
| 18 | 20.24 ± 0.50 | 24.92 ± 1.33 | 35.44 ± 2.80 | 41.63 ± 4.58 |
| 20 | 21.77 ± 0.53 | 26.85 ± 1.34 | 39.16 ± 2.99 | 43.38 ± 4.56 |
Age-adjusted mortality is expressed as a percentage ± SD.
Among patients without diabetes, the crude mortality rate was slightly lower in those with CLD than in those without CLD (18.32 vs. 20.74%, P = 0.09). However, after adjustment for age, the mortality rate was consistently higher in subjects without diabetes who had CLD than in those without either condition (27.6 vs. 22.9%, P = 0.0011). This difference started at year 2 of follow-up (2.52 vs. 1.37%, P = 0.03) and continued throughout the full follow-up period (Table 2).
Contribution of CLD and Diabetes to Mortality
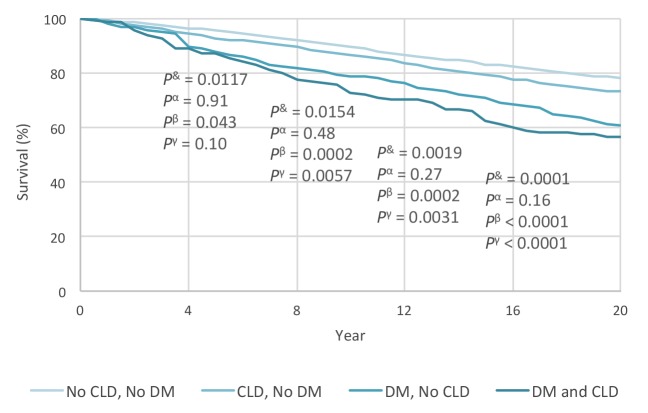
Through ∼20 years of follow-up, 5,088 of 16,413 NHANES III participants in this study died (crude mortality rate 22.95%). We calculated the survival curves in the four DM/CLD groups at 6-month intervals (Figure 1). The data clearly show that patients with both diabetes and CLD have the highest mortality, followed by those with diabetes but without CLD, then by those with CLD but without diabetes, with the lowest mortality in those without either condition.
FIGURE 1.
Age-adjusted mortality in the U.S. population with and without CLD and/or type 2 diabetes. Zero time point represents screening for NHANES. P values are reported at 4, 8, 12, and 16 years of follow-up, respectively (P& between CLD and no CLD in subjects without diabetes; Pα between CLD and no CLD in those with diabetes; Pβ between diabetes and no diabetes in subjects without CLD; Pγ between diabetes and no diabetes in those with CLD).
In multivariate survival analysis of the entire cohort, after adjusting for age, both diabetes and CLD were independently associated with increased mortality, although the presence of diabetes was associated with an ∼50% greater hazard ratio (Table 3). Additionally, although having both CLD and diabetes was not significantly associated with an increased risk of mortality (aHR 1.12, 95% CI 0.87–1.44, P = 0.39), having CLD without diabetes was associated with increased mortality (aHR 1.44, 95% CI 1.21–1.70). On the other hand, having diabetes was associated with increased mortality in both patients with CLD (aHR 1.77, 95% CI 1.29–2.42) and in those without CLD (aHR 2.03, 95% CI 1.80–2.29) (both P <0.001). Finally, in comparison to subjects who did not have diabetes or CLD, the overall mortality risk in those with both conditions was more than two times higher (aHR 2.41, 95% CI 1.83–3.17, P <0.0001).
TABLE 3.
Association of Diabetes and CLD With Mortality in the U.S. Population
| aHR (95% CI) | P | |
|---|---|---|
| Age, per year | 1.09 (1.09–1.10) | <0.0001 |
| Type 2 diabetes | 1.98 (1.75–2.24) | <0.0001 |
| CLD | 1.37 (1.19–1.57) | <0.0001 |
Discussion
This large, population-based study clearly showed the negative effect of diabetes on the mortality of people with CLD. In this study, we included CLD in general, regardless of its etiology, to provide a broader view of the scope of this issue. Smaller studies have shown that the presence of diabetes in people with NAFLD, HCV, or HBV leads to a faster progression of their liver disease to more advanced liver disease such as cirrhosis or hepatocellular carcinoma and eventually death (8–32,34). The current study, using a large population database with a substantial number of clinical variables and long-term mortality data, broadens these findings by showing that the presence of diabetes with any type of CLD significantly raises the risk of death.
We believe this study has a number of important implications. First, CLD and diabetes, separately, are already among the most common and rising causes of mortality in the United States, and each is associated with significant mortality, morbidity, and resource utilization (1–6). In our clinics, we increasingly see not only patients with NAFLD and diabetes, but also those with other types of liver disease (i.e., ALD, HBV, and HCV) and diabetes. Thus, referring to these data, we suggest that both CLD and diabetes should be treated aggressively.
Also, increasing vaccination rates in people with diabetes for hepatitis B (and for hepatitis A in the endemic areas) has the potential to save lives at minimal cost. However, although major health organizations (e.g., the American Diabetes Association and the American Association for the Study of Liver Diseases) recommend routine vaccination, a minority of people with diabetes have been vaccinated (35–37).
Furthermore, both diabetes and CLD negatively affect work productivity and patient-reported outcomes, including health-related quality of life, and place a substantial economic burden on society, especially in patients with advanced liver disease requiring liver transplantation (38–42). A sound and practical strategy to deal with both chronic diseases would have important health policy implications.
In summary, our data show that the presence of diabetes in patients with CLD has dire consequences through increased mortality. Future clinical and policy implications of this issue need to be assessed.
Funding
This work was funded by the Beatty Liver Obesity Research Fund, Inova Health System, Falls Church, Va.
Duality of Interest
No potential conflicts of interest relevant to this article were reported.
References
- 1.Centers for Disease Control and Prevention Chronic diseases: the leading causes of death and disability in the United States [Internet]. Available from http://www.cdc.gov/chronicdisease/overview. Accessed 20 June 2016
- 2.Schiller JS, Lucas JW, Ward BW, Peregoy JA. Summary health statistics for US adults: National Health Interview Survey, 2010. Vital Health Stat 10 2012;252:1–207 [PubMed] [Google Scholar]
- 3.Centers for Disease Control and Prevention/National Center for Health Statistics Health Interview Survey, 1997–2014: sample adult core component. Early release of selected estimates based on data from the National Health Interview Survey, 2014: diagnosed diabetes [Internet]. Available from http://www.cdc.gov/nchs/data/nhis/earlyrelease/earlyrelease201506_14.pdf. Accessed 20 June 2016
- 4.Centers for Disease Control and Prevention/National Center for Health Statistics Health Interview Survey, 1997–2014: sample adult core component. Early release of selected estimates based on data from the National Health Interview Survey, 2014: diagnosed obesity [Internet]. Available from http://www.cdc.gov/nchs/data/nhis/earlyrelease/earlyrelease201506_06.pdf. Accessed 20 June 2016
- 5.Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. JAMA 2012;307:491–497 [DOI] [PubMed] [Google Scholar]
- 6.Heron M. Deaths: leading causes for 2010. Natl Vital Stat Rep 2013;62:1–96 [PubMed] [Google Scholar]
- 7.El-Serag HB, Tran T, Everhart JE. Diabetes increases the risk of chronic liver disease and hepatocellular carcinoma. Gastroenterology 2004;126:460–468 [DOI] [PubMed] [Google Scholar]
- 8.Reilly ML, Schillie SF, Smith E, et al. . Increased risk of acute hepatitis B among adults with diagnosed diabetes mellitus. J Diabetes Sci Technol 2012;6:858–866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther 2011;34:274–285 [DOI] [PubMed] [Google Scholar]
- 10.Harrison SA, Togerson S, Hayashi PH. The natural history of nonalcoholic fatty liver disease: a clinical histopathological study. Am J Gastroenterol 2008;98:2042–2047 [DOI] [PubMed] [Google Scholar]
- 11.Argo CK, Caldwell SH. Epidemiology and natural history of non-alcoholic steatohepatitis. Clin Liver Dis 2009;13:511–531 [DOI] [PubMed] [Google Scholar]
- 12.Wree A, Broderick L, Canbay A, Hoffman HM, Feldstein AE. From NAFLD to NASH to cirrhosis: new insights into disease mechanisms. Nat Rev Gastroenterol Hepatol 2013;10:627–636 [DOI] [PubMed] [Google Scholar]
- 13.Targher G, Byrne CD. Clinical review: nonalcoholic fatty liver disease: a novel cardiometabolic risk factor for type 2 diabetes and its complications. J Clin Endocrinol Metab 2013;98:483–495 [DOI] [PubMed] [Google Scholar]
- 14.Williams KH, Shackel NA, Gorrell MD, McLennan SV, Twigg SM. Diabetes and nonalcoholic fatty liver disease: a pathogenic duo. Endocr Rev 2013;34:84–129 [DOI] [PubMed] [Google Scholar]
- 15.Ascha MS, Hanouneh IA, Lopez R, Tamimi TA, Feldstein AF, Zein NN. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology 2010;51:1972–1978 [DOI] [PubMed] [Google Scholar]
- 16.Centers for Disease Control and Prevention Hepatocellular carcinoma—United States, 2001–2006. MMWR Morb Mortal Wkly Rep 2010;59:517–520 [PubMed] [Google Scholar]
- 17.Wong RJ, Aguilar M, Cheung R, Perumpail RB, Harrison SA, Younossi ZM. Nonalcoholic steatohepatitis is the second leading etiology of liver disease among adults awaiting liver transplantation in the United States. Gastroenterology 2015;148:547–555 [DOI] [PubMed] [Google Scholar]
- 18.Thuluvath PJ, Guidinger MK, Fung JJ, Johnson LB, Rayhill SC, Pelletier SJ. Liver transplantation in the United States, 1999–2008. Am J Transplant 2010;10:1003–1019 [DOI] [PubMed] [Google Scholar]
- 19.Saab S, Manne V, Nieto J, Schwimmer JB, Chalasani NP. Nonalcoholic fatty liver disease in Latinos. Clin Gastroenterol Hepatol 2016;14:5–12 [DOI] [PubMed] [Google Scholar]
- 20.Younossi ZM, Stepanova M, Saab S, et al. . The impact of type 2 diabetes and obesity on the long-term outcomes of more than 85,000 liver transplant recipients in the US. Aliment Pharmacol Ther 2014;40:686–694 [DOI] [PubMed] [Google Scholar]