Abstract
Background & objectives:
Since our previous study in 2006, several new modalities for localization of cause of endogenous hyperinsulinemic hypoglycaemia such as multiphasic computed tomography (CT), multiphasic magnetic resonance imaging (MRI), endoscopic ultrasound (EUS), intraoperative ultrasound, and intra-arterial calcium infusion with arterial stimulation venous sampling (ASVS) have become available. Therefore, to evaluate the relative usefulness of various imaging modalities to guide future management in terms of diagnosis and patient care, we analyzed presentation and management of patients of endogenous hyperinsulinemic hypoglycaemia.
Methods:
In this retrospective study, medical records of patients admitted with endogenous hyperinsulinemic hypoglycaemia were retrieved. Data pertaining to clinical features, diagnosis, imaging, surgery and patient outcome were extracted. The localization of insulinoma by preoperative imaging techniques was compared with the findings at surgery to assess the accuracy of localization.
Results:
Fasting hypoglycaemia was present in all, and post-prandial hypoglycaemia (plasma glucose ≤50 mg/dl within four hours of meal) in 25.8 per cent. Mean duration of symptoms before reaching a diagnosis of hyperinsulinemic hypoglycaemia was 3.9 years. Mean duration of provocative fast was 21.8 h (range 6-48 h). Among the currently used imaging modalities, the sensitivity of localizing tumour was 79.3 per cent for multiphasic CT, 85 per cent for multiphasic MRI and 95 per cent for EUS. EUS detected tumour missed by both CT and MRI. All, except one of the operated patients, were cured by surgery.
Interpretation & conclusions:
Our results suggest that patients with insulinoma have a varied presentation. Multiphasic contrast-enhanced MRI/CT scan, EUS and ASVS may be complimentary in pre-operative localization.
Keywords: Hyperinsulinemic hypoglycaemia, insulinoma, intra-arterial calcium infusion test, nesidioblastosis, neurological dysfunction
Endogenous hyperinsulinemic hypoglycaemia presents with a varied spectrum of symptoms. Isolated sporadic insulinomas are generally cured by surgery. Insulinomas are characteristically small, therefore difficult to localize and require the help of multiple imaging modalities. A multidisciplinary team approach is required. Although we have published our experience with a series of 31 cases of insulinoma earlier in 20061, the introduction of newer imaging techniques such as multiphasic contrast-enhanced computed tomography/magnetic resonance imaging (CECT/MRI) scan, endoscopic ultrasound (EUS), arterial calcium stimulation with arterial stimulation venous sampling (ASVS), 68Ga DOTANOC positron emission tomography/CT and intraoperative ultrasound (IOUS) have had a significant impact on the way these patients are being evaluated and managed at our centre. Insulinoma is a rare disease with an incidence of about 1 in 250,0002. Thus, a prospective study is difficult to plan. Therefore, to review our management and guide future management of such patients, a retrospective study was conducted with an objective to assess the clinical features, accuracy of newer diagnostic and localizing techniques and see their impact on the treatment outcomes of patients with hyperinsulinemic hypoglycaemia.
Material & Methods
This was a retrospective analysis of patients with endogenous hyperinsulinemic hypoglycaemia admitted under the care of departments of Endocrinology and Gastrointestinal Surgery, All India Institute of Medical Sciences, New Delhi, India, and managed over the last eight years (January 2006-December 2013). The clinical characteristics, modalities for diagnosis and treatment were compared to that used in the earlier protocol (1992-2005)1.
Inclusion criteria were patients who presented with hypoglycaemia and on investigation were found to have hyperinsulinemic hypoglycaemia3 i.e., serum insulin and C-peptide levels of ≥3 IU/ml and ≥0.6 ng/ml, respectively, during hypoglycaemia (≤50 mg/dl). Exclusion criteria were patients with diabetes who had developed hypoglycaemia secondary to insulin or oral hypoglycaemic agent intake, hypoglycaemia due to insulin autoantibodies, hypopituitarism and hypoadrenalism. Insulin and C-peptide were measured on an autoanalyzer (Roche Elecsys 2010, Germany) using electrochemiluminometric assay using Roche kit after 2006. Before 2006, these were measured by immunoradiometric assay using Immunotech kit.
Ethical clearance for conducting the study was obtained from the institutional ethics committee. Data on clinical features, diagnosis, imaging studies and surgical outcome were extracted and analyzed.
Pre-operative localization of tumour was attempted using a combination of available radiological techniques which included multiphase CT, multiphase MRI, EUS and 68Ga DOTANOC positron emission tomography (PET)/CT. Intra-arterial calcium infusion test (ASVS) was used in four patients in whom a discrete lesion could not be identified on CT, MRI and EUS.
Technique of calcium stimulation test: A selective arterial calcium infusion test was performed preoperatively, in which after taking a baseline blood sample from hepatic vein, bolus of 1.2 mEq of calcium solution (3 ml of 8.5% calcium gluconate) was injected sequentially into splenic artery, gastroduodenal artery and superior mesenteric artery. Hepatic venous blood sampling was done before, 30 sec, one, two and three minutes after injection of calcium into each of the arteries.
During surgery, lesions were identified based on a combination of intraoperative bimanual palpation of pancreas and IOUS (since 2000)1. The operative findings were corroborated with the pre-operative imaging before proceeding with enucleation/pancreatic resection. After tumour removal, the specimen was submitted for frozen section analysis to confirm the presence of a neuroendocrine tumour (NET). The localization of insulinoma by pre-operative imaging modalities was compared with the findings at surgery to assess the accuracy of localization.
Results
Table I summarizes the clinical presentation of patients with hyperinsulinemic hypoglycaemia. The mean age of presentation was 37.9 yr with a range of 14-65 yr. The male to female ratio was 35:31. Mean body mass index (BMI) was 25.1 kg/m2 (17.3-39.1 kg/m2). History of weight gain was present in 42 of 66 patients (63.6%) and history of weight loss in none. Post-prandial hypoglycaemia was present in 25.8 per cent patients (17/66, Table I) including one with nesidioblastosis. The mean duration of symptoms before a diagnosis of hyperinsulinemic hypoglycaemia was made was 3.9 years with a range of two months to 15 yr.
Table I.
Clinical features in patients with hyperinsulinemic hypoglycaemia
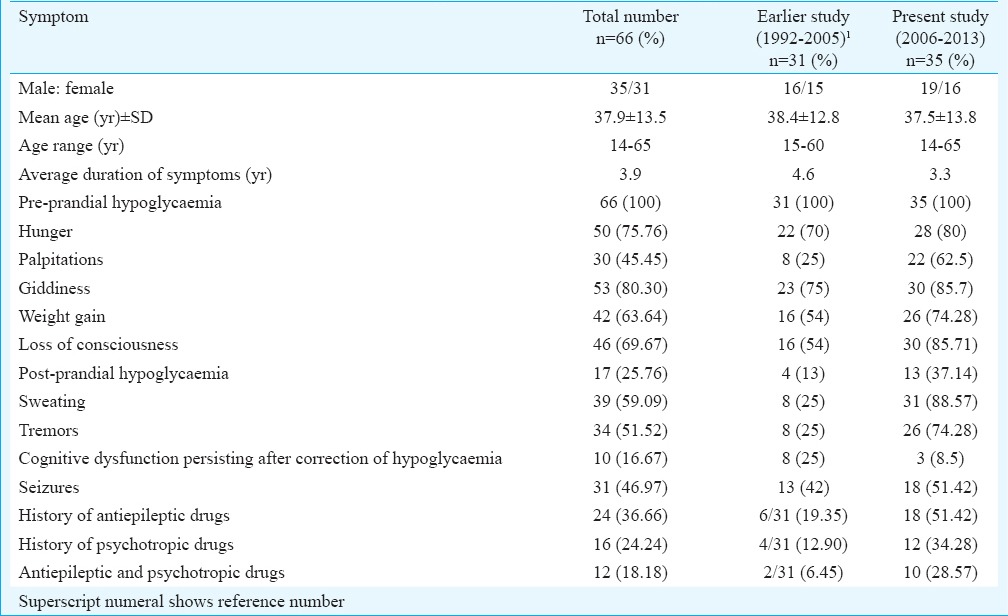
Neurological dysfunction normalized totally after immediate correction of hypoglycaemia in all except three among 35 (8.6%) patients. In three patients, cognitive dysfunction persisted even after immediate correction of hypoglycaemia. Among these three, in two, neurological deficit reverted after surgical removal of tumour. In the third patient who had presented in a comatose state, there was a persistent neurological sequel of hemiparesis and slurred speech post-surgery. She was discharged with advice of speech therapy and physiotherapy.
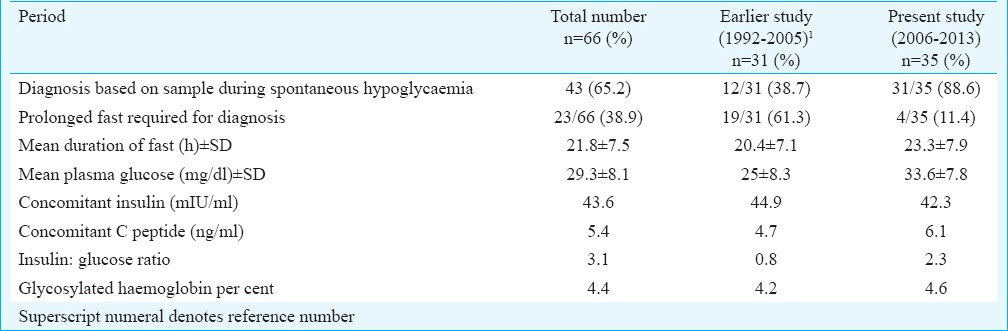
Biochemical parameters: All patients presented with fasting hypoglycaemia with Whipple's triad. Spontaneous hypoglycaemia occurred in most admitted patients when they were being monitored. Prolonged fast was required in 38.9 per cent patients (23 of 66) and the mean duration of fast was 21.8 h (range 6-48 h) (Table II). The mean plasma glucose was 29.3 mg/dl (range 8-45 mg/dl). Concomitant mean plasma insulin during episodes of hypoglycaemia was 43.6 mIU/ml (range 5.6-172.8 mIU/ml). Simultaneous mean C-peptide was 5.4 ng/ml (range 0.55-24.20 ng/ml). The mean plasma insulin-glucose ratio was 3.05 (range 0.2-21.5). Mean glycosylated haemoglobin was 4.4 per cent (range 4.1-5.6%).
Table II.
Biochemical parameters of patients
After confirmation of hyperinsulinemic hypoglycaemia, localization of insulinoma was done by various imaging modalities. Their accuracy in localizing tumour was compared to the actual tumour site at surgery (Table III). During 2006-2013, multiphasic CT was done in 29 patients, of whom insulinoma was localized in 23. Among these, in one patient, though lesion was at the junction of head and neck on CT, at surgery, the actual location of tumour was at the body. In the other patient, there was a focal contour abnormality at the actual site. The sensitivity of multiphase CT in localizing tumour was 79 per cent. Pancreas was falsely normal in five, and generalized enlargement was seen in one which turned out to be nesidioblastosis. During the same period, multiphasic MRI was done in 20 patients, of whom insulinoma localization was done in 17. This included a focal contour abnormality in one patient which turned out to be an insulinoma and generalized pancreas enlargement in another which turned out to be nesidioblastosis. Multiphasic MRI was falsely normal in three patients. EUS revealed lesion in 21 of the 24 patients of hyperinsulinemic hypoglycaemia in whom it was performed. Among the cases missed, one was a case of nesidioblastosis, second ectopic insulin secretion from the kidney4 and the third with actual location of insulinoma in the tail of pancreas. Thus, it was positive in 21 of the 22 patients, in whom insulinoma was present in pancreas. This gives EUS a sensitivity of 95 per cent.
Table III.
Sensitivity of various pre-operative imaging techniques
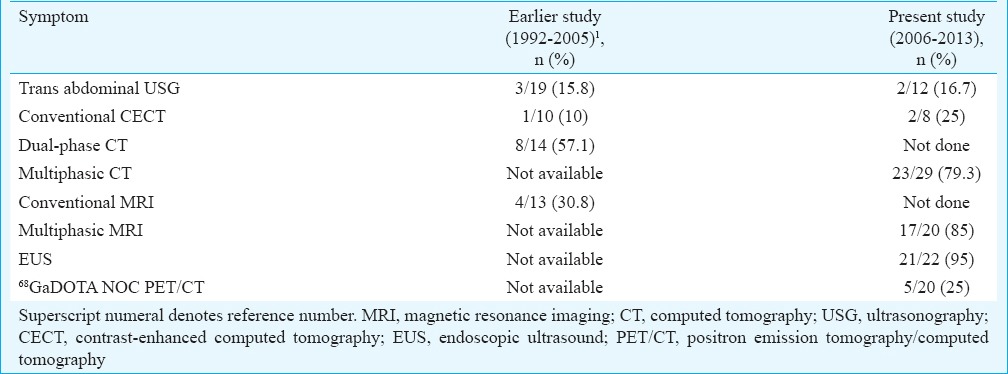
68Ga DOTANOC PET/CT rightly localized tumour in 5 of 20 patients. It was falsely normal in 13 and localized to wrong place in two. It was particularly helpful in picking up ectopic NET from the kidney4. In one patient, multiphase CT picked up a lesion in tail of pancreas which was missed on multiphasic MRI. In three patients, multiphasic MRI picked up tumour in the body of pancreas which was missed by multiphasic CT. EUS localized tumour in three patients in whom CT/MRI could not locaalize these tumours. In another patient, EUS helped to define exact location in head of pancreas while on CT and MRI, lesion was located in pancreaticoduodenal groove. 68Ga DOTANOC PET/CT did not localize any additional tumour and failed to localize tumours which were picked up by multiphasic CT and multiphasic MRI. In two cases, the uptake on 68Ga DOTANOC PET/CT was different from the actual location of tumour at surgery which was rightly picked up by multiphasic CT and/or multiphasic MRI.
In selective arterial calcium stimulation test, hepatic venous sampling following selective arterial infusion of calcium gluconate test was done in four patients in whom tumour could not be localized by other methods. Two patients were not operated. In one, CT, MRI and DOTONOC PET were normal, and EUS showed lesion in the head of pancreas. This patient was discharged on request and was lost to follow up. In the other patient, multiphasic CT showed doubtful lesion in tail while other localizing modalities were normal. In the absence of convincing localization, he was advised frequent complex carbohydrate meals and plasma glucose monitoring at the time of symptoms. The medical management of hypoglycaemia in patients who were not operated included diazoxide, octreotide and verapamil in addition to frequent complex carbohydrate meals.
Of the 35 patients from 2006 to 2013, none was found with malignant insulinoma. In the previous study1 from 1992 to 2005, one of 31 had insulinoma with metastasis to the liver. Actual location of tumour was in pancreatic head (12), tail (6), between junction of body and tail (3), body (4), neck (1), junction between head and neck (2) and uncinate process (2). One patient had ectopic insulin-secreting tumour arising from a NET in kidney4, one had nesidioblastosis and another had multiple endocrine neoplasia type-1 (MEN-1) with parathyroid adenoma and prolactinoma. In the patient with MEN-1, insulinoma was multifocal, present in the head, body, tail and uncinate process. In all others, lesion was single.
Enucleation was done in 22, distal pancreatectomy in eight, median pancreatectomy in one and Whipple's procedure in one. Patient with ectopic insulin secretion from NET kidney underwent nephrectomy as the tumour was large and vascular with propensity to bleed. During 2006 to 2013 on surgery, the actual size of tumour (considering length & breath) was ≤1 cm in nine, between 1 and 2 cm in 13, between 2 and 3 cm in seven and ≥3 cm in ne patient had ectopic insulinoma in kidney, one had nesidioblastosis and two were not operated.
Post-operative blood glucose monitoring: Post-operative rebound hyperglycaemia occurred in 10 out of 33 (30.3%) patients operated. These patients required insulin for glycaemic control, which was tapered down gradually such that at discharge (mean duration 10 days post-operative stay); only one of the 33 operated (3%) patients required insulin. Patients who had no recurrence of hypoglycaemia during post-operative period were considered cured. All, except two patients who were operated, were cured in single surgery. Among the patients who were not cured in single surgery, one was a case of nesidioblastosis and was managed postoperatively with long-acting octreotide. The other had a 5 mm single lesion in the head of pancreas and continued to have post-operative hypoglycaemia. Five months later, he presented in coma with hypoglycaemia. CT revealed a residual lesion at the head of pancreas. He underwent second surgery after which his hypoglycaemia disappeared.
Histology of all patients except two was consistent with NET of pancreas. Cells were immunoreactive for chromogranin A and synaptophysin. Immunohistochemistry confirmed insulinoma tissue to be immunopositive for insulin. Among the remaining two patients, one was nesidioblastosis and the other was insulin-secreting NET arising from kidney.
Discussion
The most common cause of endogenous hyperinsulinemic hypoglycaemia is insulinomas. For the diagnosis and management of hyperinsulinemic hypoglycaemia, a multidisciplinary team consisting of endocrinologist, gastrointestinal surgeon, radiologist and endosonologist is required2,5. In some studies, the average age of onset has been in the fifth decade with a female preponderance3. We found the mean age at presentation in the fourth decade and did not see a female preponderance.
As symptoms of hypoglycaemia are non-specific, insulinoma can mimic several other conditions. Hypoglycaemia can present as seizures, psychiatric illness, confusion6 and vague symptoms5. In our study, most common symptoms were giddiness, drowsiness and hunger. On correction of hypoglycaemia, there was immediate normalization of neurological dysfunction in most of the patients. It has been reported earlier that cognitive and neurological dysfunctions and even brain MRI changes induced by hypoglycaemia revert after surgical removal of insulinoma over a period of time7.
Since insulinomas are small in size and located intraparenchymally, localization can be challenging8. Pre-operative localization increases the chances of surgical cure, decreases operating time and prevents repeat surgeries9,10,11. Multiphasic pancreas protocol CT has replaced dual phase CT which was done before 2006. Conventional CT is no longer done for suspected insulinoma.
In this study, localization predicted by each pre-operative test was compared to localization at surgery for accuracy. Among non-invasive modalities, multiphasic MRI could localize insulinoma in 85 per cent whereas multiphasic CT in 79 per cent. Three patients in whom insulinoma could not be localized by CT were localized correctly by MRI. In one patient where MRI failed to localize tumour, CT localized tumour. Thus, MRI and CT are complimentary, and there is a need to repeat one if the other is negative. Multiphasic MRI yielded a better result than multiphasic CT.
From 1992 to 20051, conventional MRI accurately picked up insulinoma in only 31 per cent while after 2006, multiphasic MRI localized accurately in 85 per cent. This could be due to increase in field strength of scanner and faster multiphasic sequences. In four cases in whom other methods could not localize definite lesion, selective arterial calcium infusion was performed. It helped to localize tumour in three. The last turned out to be nesidioblastosis. A previous report10 also showed that nesidioblastosis should be suspected in patients with endogenous hyperinsulinemic hypoglycaemia without any detectable pancreatic tumour on pre-operative imaging.
In a case series in 25 patients11, calcium stimulation has been reported to have the highest accuracy (88%) for localizing insulinomas compared to MRI (43%), CT (17%) and ultrasonography (9%). In another series from NIH, in which 45 sporadic insulinomas in whom localization by other methods were negative, calcium stimulation localized correctly in 84 per cent9. The drawback of this study was that EUS was not used9. Moreover, selective arterial calcium infusion is an invasive test which requires expertise.
In our study, EUS had a sensitivity of 95 per cent in localizing insulinoma. Other studies11,12 have shown EUS to be positive in 70-95 per cent cases if an experienced endoscopist is available. Thus it is the imaging modality of choice if the other non-invasive modalities are negative. EUS also helps to know if tumour enucleation is possible as it can estimate the distance between tumour and pancreatic duct which can also be done on MRI12,13.
In our study, 68Ga DOTANOC PET/CT was able to localize insulinoma in only 25 per cent cases. It was particularly helpful in establishing the functionality of NET from kidney which turned out to be ectopic insulinoma on histopathology. Promising results were obtained using PET/CT methods including 68Ga DOTATOC14,15,16 though large-scale studies are lacking. Limited utility of 68Ga DOTANOC PET in insulinoma localization was seen in another study with only 35.1 per cent of patients getting correct localization with this modality17. DOTANOC binds predominantly to somatostatin receptor subtype 2, while it binds to somatostatin receptor 3 and 5 to a lesser extent. Insulinomas express mostly receptor subtype 2 and 4. If the receptor subtype predominantly expressed is type 4, DOTANOC shall be false negative18.
At our centre, IOUS is in use since 2000. IOUS along with bimanual palpation is useful to localize tumour. Therefore invasive tests such as intra-arterial digital subtraction angiography, CT angiography and selective pancreatic angiography (earlier done at our centre) have been abandoned. Previous studies19,20 showed that intraoperative inspection and palpation localized the lesions correctly in 91 per cent and IOUS in 93 per cent of cases.
Blind distal pancreatectomy which was advocated often in the previous era, is no longer advocated in the current surgical practice. The success of surgery depends on the successful pre-operative and perioperative localization techniques20,21. In the patient with MEN-1 who had multifocal lesions at the head, body, uncinate process and tail, enucleation of the lesions was done with postoperatively, patient being free of hypoglycaemia. In a study of MEN-1 with insulinoma, enucleation with limited pancreas resection provided long-term cure in MEN-122. Subtotal distal pancreatectomy should be reserved for patients with multiple tumours given the high risk of endocrine and exocrine insufficiency.
Intraoperatively, maintenance of euglycaemia is important as severe hypoglycaemia during tumour handling can occur23. Glucose infusion with frequent plasma glucose monitoring is helpful. Post tumour removal there can be hyperglycaemia which usually recovers within a few days. In the present study (2006-2013), 45.5 per cent patients developed post-operative hyperglycaemia. They were managed with insulin. All, but one, were euglycaemic without insulin at discharge which was on an average around the 10th post-operative day.
This study had limitations. Being a retrospective study, all data were not available, especially from the previously published study from 1992 to 20051 for comparison. The modalities for localization of insulinoma have changed, for example, earlier selective pancreatic angiography, intra-arterial digital subtraction angiography and CT angiography were used earlier but since 2000 we have started using intra-abdominal ultrasound1. The CT and MRI used now were multiphasic compared to dual phase used earlier.
In conclusion, insulinomas have a varied presentation and the lag time before correct diagnosis continues to be long (about four years). Improved imaging techniques such as multiphasic CECT scan and MRI had a sensitivity of 79 and 85 per cent, respectively, in localizing an insulinoma. There were cases in whom CT picked up lesions missed by MRI and vice versa. EUS picked up additional cases missed by both CT and MRI and was true positive in 95 per cent of the cases. A 68Ga DOTANOC PET/CT was of no additional help in localization of pancreatic insulinoma. Surgery (enucleation/resection) achieved a high success rate (97%) in ameliorating symptoms of hypoglycaemia.
Footnotes
Conflicts of Interest: None.
References
- 1.Jyotsna VP, Rangel N, Pal S, Seith A, Sahni P, Ammini AC. Insulinoma: diagnosis and surgical treatment. Retrospective analysis of 31 cases. Indian J Gastroenterol. 2006;25:244–7. [PubMed] [Google Scholar]
- 2.Jensen RT, Cadiot G, Brandi ML, de Herder WW, Kaltsas G, Komminoth P, et al. ENETS Consensus Guidelines for the management of patients with digestive neuroendocrine neoplasms: functional pancreatic endocrine tumor syndromes. Neuroendocrinology. 2012;95:98–119. doi: 10.1159/000335591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Iglesias P, Díez JJ. Management of endocrine disease: a clinical update on tumor-induced hypoglycemia. Eur J Endocrinol. 2014;170:R147–57. doi: 10.1530/EJE-13-1012. [DOI] [PubMed] [Google Scholar]
- 4.Ramkumar S, Dhingra A, Jyotsna VP, Ganie MA, Das CJ, Seth A, et al. Ectopic insulin secreting neuroendocrine tumor of kidney with recurrent hypoglycemia: a diagnostic dilemma. BMC Endocr Disord. 2014;14:36. doi: 10.1186/1472-6823-14-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Okabayashi T, Shima Y, Sumiyoshi T, Kozuki A, Ito S, Ogawa Y, et al. Diagnosis and management of insulinoma. World J Gastroenterol. 2013;19:829–37. doi: 10.3748/wjg.v19.i6.829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jagadheesan V, Suresh SS. Episodic confusional state: due to insulinoma. Indian J Psychiatry. 2008;50:197–9. doi: 10.4103/0019-5545.43636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pakhetra R, Priya G, Jyotsna VP, Seith A, Ammini AC. Insulinoma: reversal of brain magnetic resonance imaging changes following resection. Neurol India. 2008;56:192–4. doi: 10.4103/0028-3886.42001. [DOI] [PubMed] [Google Scholar]
- 8.Mathur A, Gorden P, Libutti SK. Insulinoma. Surg Clin North Am. 2009;89:1105–21. doi: 10.1016/j.suc.2009.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Guettier JM, Kam A, Chang R, Skarulis MC, Cochran C, Alexander HR, et al. Localization of insulinomas to regions of the pancreas by intraarterial calcium stimulation: the NIH experience. J Clin Endocrinol Metab. 2009;94:1074–80. doi: 10.1210/jc.2008-1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gupta RA, Patel RP, Nagral S. Adult onset nesidioblastosis treated by subtotal pancreatectomy. JOP. 2013;14:286–8. doi: 10.6092/1590-8577/1352. [DOI] [PubMed] [Google Scholar]
- 11.Doppman JL, Chang R, Fraker DL, Norton JA, Alexander HR, Miller DL, et al. Localization of insulinomas to regions of the pancreas by intra-arterial stimulation with calcium. Ann Intern Med. 1995;123:269–73. doi: 10.7326/0003-4819-123-4-199508150-00004. [DOI] [PubMed] [Google Scholar]
- 12.Patel KK, Kim MK. Neuroendocrine tumors of the pancreas: endoscopic diagnosis. Curr Opin Gastroenterol. 2008;24:638–42. doi: 10.1097/MOG.0b013e32830bf7fb. [DOI] [PubMed] [Google Scholar]
- 13.Joseph AJ, Kapoor N, Simon EG, Chacko A, Thomas EM, Eapen A, et al. Endoscopic ultrasonography – A sensitive tool in the preoperative localization of insulinoma. Endocr Pract. 2013;19:602–8. doi: 10.4158/EP12122.OR. [DOI] [PubMed] [Google Scholar]
- 14.Dudczak R, Traub-Weidinger T. PET and PET/CT in endocrine tumours. Eur J Radiol. 2010;73:481–93. doi: 10.1016/j.ejrad.2009.12.024. [DOI] [PubMed] [Google Scholar]
- 15.Kauhanen S, Seppänen M, Minn H, Nuutila P. Clinical PET imaging of insulinoma and beta-cell hyperplasia. Curr Pharm Des. 2010;16:1550–60. doi: 10.2174/138161210791164090. [DOI] [PubMed] [Google Scholar]
- 16.Treglia G, Inzani F, Campanini N, Rindi G, Agnes S, Giordano A, et al. A case of insulinoma detected by (68)Ga-DOTANOC PET/CT and missed by (18)F-dihydroxyphenylalanine PET/CT. Clin Nucl Med. 2013;38:e267–70. doi: 10.1097/RLU.0b013e31825b222f. [DOI] [PubMed] [Google Scholar]
- 17.Sharma P, Arora S, Karunanithi S, Khadgawat R, Durgapal P, Sharma R, et al. Somatostatin receptor based PET/CT imaging with 68Ga-DOTA-Nal3-octreotide for localization of clinically and biochemically suspected insulinoma. Q J Nucl Med Mol Imaging. 2016;60:69–76. [PubMed] [Google Scholar]
- 18.Sharma P, Singh H, Bal C, Kumar R. PET/CT imaging of neuroendocrine tumors with 68Gallium-labeled somatostatin analogues: An overview and single institutional experience from India. Indian J Nucl Med. 2014;29:2–12. doi: 10.4103/0972-3919.125760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gahlot A, Naik DB, Siddiqui SS, Jain P, Sharma B, Chaudhary S, et al. Intraoperative sonographic localization of insulinoma: case reports and review of literature. WJOES. 2011;3:75–8. [Google Scholar]
- 20.Ravi K, Britton BJ. Surgical approach to insulinomas: are pre-operative localisation tests necessary? Ann R Coll Surg Engl. 2007;89:212–7. doi: 10.1308/003588407X179008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Galiber AK, Reading CC, Charboneau JW, Sheedy PF, 2nd, James EM, Gorman B, et al. Localization of pancreatic insulinoma: comparison of pre- and intraoperative US with CT and angiography. Radiology. 1988;166:405–8. doi: 10.1148/radiology.166.2.2827232. [DOI] [PubMed] [Google Scholar]
- 22.Bartsch DK, Albers M, Knoop R, Kann PH, Fendrich V, Waldmann J. Enucleation and limited pancreatic resection provide long-term cure for insulinoma in multiple endocrine neoplasia type 1. Neuroendocrinology. 2013;98:290–8. doi: 10.1159/000357779. [DOI] [PubMed] [Google Scholar]
- 23.Goswami J, Somkuwar P, Naik Y. Insulinoma and anaesthetic implications. Indian J Anaesth. 2012;56:117–22. doi: 10.4103/0019-5049.96301. [DOI] [PMC free article] [PubMed] [Google Scholar]


