Abstract
Primary ovarian insufficiency (POI) is a genetically heterogeneous disorder that occurs in familial or sporadic fashion. Through whole exome sequencing in a Chinese pedigree with POI, we identified a novel homozygous missense mutation (ENST00000375755: c.1459G > T, p.D487Y) in the MSH5 gene in two sisters with POI. The homologous mutation in mice resulted in atrophic ovaries without oocytes, and in vitro functional study revealed that mutant MSH5 impaired DNA homologous recombination repair. From sanger sequencing of MSH5 in 200 sporadic POI patients, we identified three heterozygous mutations (ENST00000375755: c.1057C > A, p.L353M; c.1459G > T, p.D487Y and c.2107 A > G, p.I703V). Considering the heterozygous p.D487Y carrier in the POI pedigree was fertile, the causality of the three heterozygous mutations in POI need more evidence. Our studies confirmed that perturbation of genes involved in DNA damage repair could lead to non-syndromic POI. The underlying mechanism-inability to repair DNA damage-will receive increasing attention with respect to POI.
Introduction
Primary ovarian insufficiency (POI), also known as premature ovarian failure (POF [MIM 311360]), is a hypergonadotropic disorder characterized by cessation of menses before 40 years of age and elevated level of follicle stimulating hormone (FSH) (1). Approximately, 1% of women in childbearing age are affected (2). POI is heterogeneous in etiology, including chromosomal abnormalities and single gene mutations, as well as autoimmune, metabolic, infectious and iatrogenic factors. While evidence from genetic factors, provided by population and candidate gene studies, is responsible for the pathogenesis of about 25% of cases, most cases remain unexplained (1). Recently, some novel causative genes have been identified by whole exome sequencing (WES) in POI pedigrees, such as HFM1 (MIM 615684), STAG3 (MIM 608489), MCM8 (MIM 608187), MCM9 (MIM 610098) and CSB-PGBD3 (MIM 609413) (3–7). Interestingly, all of these genes are involved in DNA repair or meiosis, which thus proposes a plausible brand-new concept for POI pathogenesis-inability to repair DNA damage.
MSH5 (MutS homologue 5) is a member of the MutS family, which is principally linked to mismatch repair (MMR). Among all the MSH homologs identified in eukaryotes, the MSH4-MSH5 heterodimers play an important role in homologous recombination (HR) repair for DNA double strand breaks (DSBs) (8). Meiotic crossing-over is processed by SPO11-dependent DSB and HR, hence, MSH5 is also involved in stabilizing and protecting the meiotic recombination intermediate (9). Here, we present an autosomal recessive causative mutation in MSH5 responsible for two sisters with POI in a Chinese non-syndromic kindred.
Results
Homozygous missense mutation in MSH5 identified in POI pedigree
Two sisters (III4 and III5) from the non-consanguineous Han Chinese family (Fig. 1A), aged 31 and 29 years, experienced oligomenorrhea since menarche (14 and 13 years old), and amenorrhea occurred approximately 10 years later (Table 1). Both of them have elevated serum FSH, infantile uteri, and atrophic ovaries devoid of follicles. Chromosomal abnormalities, FMR1 premutation, autoimmune disorders, previous ovarian surgery or chemo-/radiotherapy were absent in any of the family members.
Figure 1.
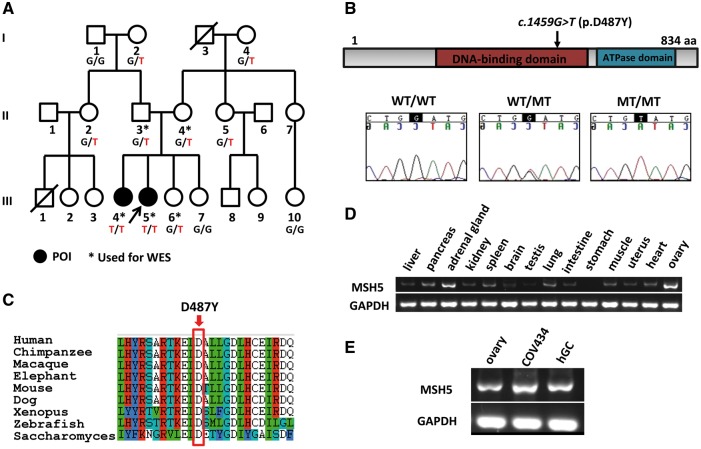
Pedigree of a family with two daughters afflicted by POI and homozygous MSH5 p.D487Y variant. (A) The pedigree of the index family, ascertained through III5. WES was performed on the family members labeled with asterisk, and those labeled with genotypes were available for Sanger sequencing. “T” denotes the mutant MSH5 allele, and “G” wild type. Arrow indicates the proband. (B) The location of p.D487Y variant is in the DNA-binding domain of MSH5, and the residue is conserved from saccharomyces to human (C). (D) shows the mRNA level of MSH5 in fetal tissues, which is significantly higher in ovary than others. (E) The RT-PCR in fetal ovary, human granulosa cells (hGCs, obtained from one patient receiving in vitro fertilization treatment) and COV434 cells, shows that MSH5 is also highly expressed in adult granulosa cells. MT, mutant; and WT, wild type.
Table 1.
Clinical features of familial and sporadic POI patients with mutations in MSH5
| Patient No. |
MSH5 Mutation |
Geno- type | Age (year) | Menstrual History |
FSH (IU/L) | Uterus (cm ×cm) | Left Ovary (cm ×cm) | Right Ovary (cm ×cm) | Age at Diagnosis (year) | Prior Hormonal Treatment | Childbearing | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| sequence variation | Amino acid variation | Age at Menarche (yr) | Age of Amenorrhea (year) | ||||||||||
| Index Family | |||||||||||||
| III4 | c.1459 G>T | p.D487Y | TT | 31 | 14 oligomeno-rrhea | 25 | 44.46 | 3.5 × 2.7 | 1.3×0.4 Follicle: 0 | 1.6×0.4 Follicle: 0 | 28 | Yesa | None |
| III5 | c.1459 G>T | p.D487Y | TT | 29 | 13 oligomeno-rrhea | 23 | 94.2 | 2.5 × 2.1 | Invisible | Invisible | 25 | Yesa | None |
| Sporadic POI | |||||||||||||
| Case 1 | c.1057 C>A | p.L353M | AC | 38 | 16 oligomeno-rrhea | 29 | 69.64 | 3.6 × 2.5 | Invisible | Invisible | 32 | Yes a | None |
| Case 2 | c.1459 G>T | p.D487Y | GT | 32 | 13 oligomeno-rrhea | 19 | 87.61 | 3.8 × 2.7 | 1.4 × 0.9 Follicle: 0.1cm×2 | Invisible | 28 | Yes a | None |
| Case 3 | c.2107 A>G | p.I703V | AG | 37 | 14 | 24 | 127.4 | 2.5 × 1.1 | Invisible | Invisible | 33 | Yes a | G1P0L0A1, amenorrhea occurred after induced abortion |
a1–2 mg daily dose of oral estradiol valerate tablet for 21 days plus oral micronized progesterone (200 mg/days) for 12 days each month.
In this POI pedigree, two affected siblings (III4, III5), both parents (II3, II4), and one unaffected daughter (III6) had been chosen to perform WES (Fig. 1A). In an autosomal-recessive inheritance model, 2 nonsynonymous homozygous variants in MSH5 (MIM 603382, chromosome 6p21.33) and ZNF391 (Zinc Finger Protein 391, chromosome 6p22.1) were revealed. Sanger sequencing confirmed that neither of the two homozygous variants were present in unaffected family members and both of the variants were absent in 400 fertile women. The ZNF391 variant (ENST00000244576: c.187A > G, p.S63G) was predicted to be benign by Polyphen2, and the Serine residue mutated was not conserved among species (Supplementary Material, Fig. S1). Furthermore, ZNF391 has not been related to any human disease and no mutation was found in 200 sporadic patients with POI. Therefore, the MSH5 variant (ENST00000375755: c.1459G > T, p.D487Y) remained as the only potential candidate for this POI family. Sanger sequencing for MSH5 in sporadic cases with POI identified 3 additional heterozygous mutations (ENST000 00375755: c.1057C > A, p.L353M; c.1459G > T, p.D487Y and c.2107 A > G, p.I703V), which had not been reported in either the Exome Variant Server or 1000 Genomes database. Among them, p.L353M and p.D487Y located in the DNA-binding domain and the original residues were highly conserved among species from yeast to human, while p.I703V occurred at the less conserved residue located at the ATPase domain (Supplementary Material, Fig. S2).
Expression of MSH5 in the primate ovary
Through RT-PCR in various tissues of human fetuses, which were induced abortion at 21 weeks, we found MSH5 was highly expressed in fetal ovary and adrenal gland (Fig. 1D). We also found MSH5 was highly expressed in adult human granulosa cells (hGCs), including the hGCs obtained from one patient receiving in vitro fertilization treatment and COV434 (human ovarian granulosa tumor cell line) cells (Fig. 1E).
Homozygous mutant mouse model carrying Msh5 p.D486Y point mutation displayed POI phenotype
The homologous residue for human MSH5 c.G1459 (ENST00 000375755: p.D487) in mouse is Msh5 c.G1456 (EN SM UST00000007250: p.D486), which is highly conserved (Fig. 1C). To examine the functional effect of p.D487Y identified in human POI, the mouse model carrying Msh5 p.D486Y point mutation was generated using a CRISPR/Cas9 system (Supplementary Material, Fig. S3A and B). The homozygous mutant mice Msh5D486Y/D486Y were obtained by intercrossing of Msh5+/D486Y mice (Supplementary Material, Fig. S3C). The homozygous mutant mice were viable at birth and no obvious developmental defects were observed in adults (Fig. 2A). However, all the Msh5D486Y/D486Yfemales were infertile. The size of the ovary was dramatically reduced compared to that of control females (Fig. 2B). The results of histological studies showed that the follicles at different developmental stages (black arrows) were observed in control ovaries (Fig. 2C), whereas no developing follicle was noted in Msh5D486Y/D486Y ovaries (Fig. 2D) at 2 month of age. Numerous Ddx4-positive germ cells (black arrows) were observed in control ovaries (Fig. 2E), but no germ cells were noted in Msh5D486Y/D486Y ovaries (Fig. 2F).
Figure 2.
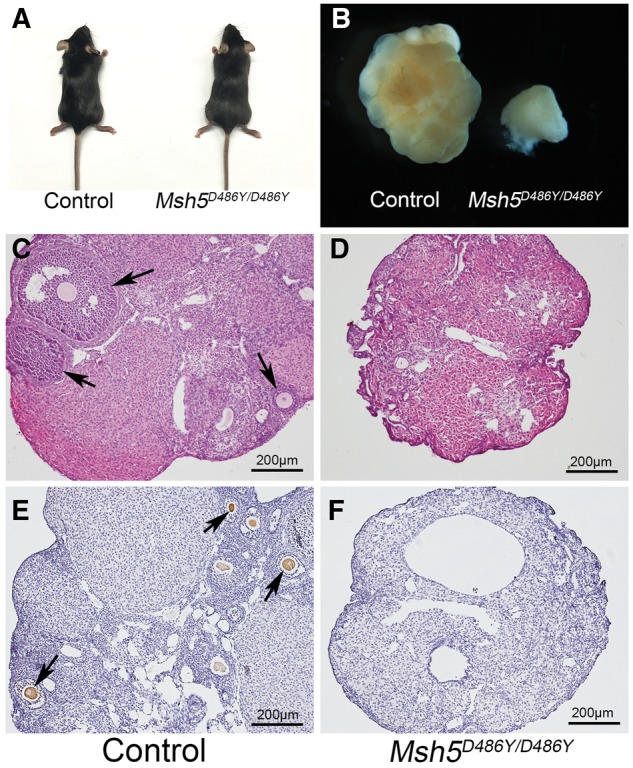
Germ cell loss in ovaries from Msh5D486Y/D486Y mice. (A) The homozygous mutant mice were viable at birth and no obvious developmental defects were observed in adults. (B) The size of ovaries from homozygous mutant females was dramatically reduced compared to the control ovaries. The follicles at different developmental stages (arrows) were observed in control ovaries (C), whereas no developing follicle was noted in Msh5D486Y/D486Y ovaries (D). Numerous Ddx4-positive germ cells (arrows) were observed in control ovaries (E), but no germ cells were noted in Msh5D486Y/D486Y ovaries (F). Scale bars: 200 μm.
Adverse effect of MSH5 p.D487Y on DNA repair for DSBs
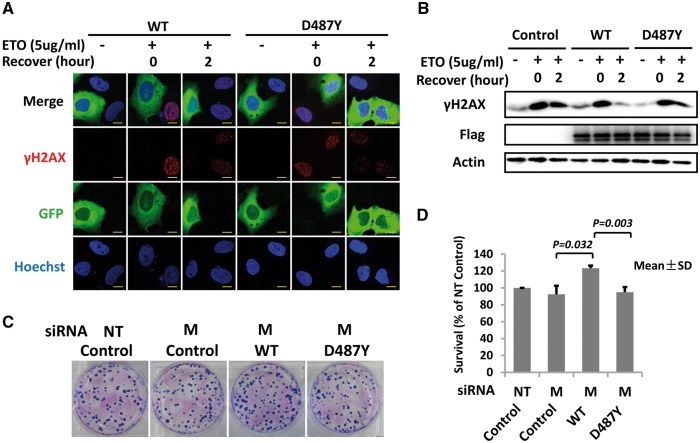
DSBs were induced by ETO treatment, and γH2AX foci were observed at the DSBs sites, which gradually disappeared along with the repair processing (Fig. 3A). The result showed that γH2AX level was much higher in the U2OS cells overexpressing mutant MSH5 than wild type (Fig. 3B). Furthermore, HeLa cells overexpressing mutant MSH5 showed a lower clonogenic survival rate than wild type, indicating the adverse effect of p.D487Y on DNA repair capacity or cellular resistance for DSBs. (Fig. 3C and D).
Figure 3.
MSH5 p.D487Y impaired DNA repair. (A) Immunofluorescence showed the γH2AX foci formation in U2OS cells overexpressing wild type (WT) or mutant (D487Y) MSH5-GFP protein when suffering from ETO treatment. Scale bars: 5μm. (B) The γH2AX concentration among U2OS cells overexpressing blank vector (Control), wild type (WT) or mutant (D487Y) MSH5-Flag protein was detected by western blot. The expression of wild type and mutant MSH5 was detected by Flag antibody and β-actin was used as the loading control. (C) and (D) showed the clonogenic survival rate of WT- and D487Y-overexpressing cells in response to ETO treatment. The experiments on U2OS and HeLa cells both had 3 replicates independently. Data in the figure are shown as mean ± SD. M, siRNA targeting at MSH5; NT, non-targeting siRNA; and WT, wild type.
Discussion
In a Han Chinese family having two siblings with non-syndromic POI, we identified an autosomal recessive mutation in MSH5 (p.D487Y) by WES, which impaired DNA repair for DSBs. Furthermore, the mice carrying homologous mutation present with POI phenotype, indicating that p.D487Y in MSH5 might be the causative mutation of POI pathogenesis.
Oocyte genome is particularly vulnerable to ubiquitous external damage, such as DSBs and single-strand breaks (SSBs). Ability to repair DNA damage is essential to maintain the supply of oocytes necessary for reproduction. DSBs are likely the most detrimental damage, normally repaired by HR and non-homologous end joining (NHEJ) (10,11). The MSH4-MSH5 heterodimer is one of the crucial factors in HR by recognizing Holliday junctions and forming a hydrolysis-independent sliding clamp that embracing adjacent homologous duplex arms (8).
In the Msh5-/- female mice, meiosis is arrested due to the disruption of chromosome pairing at prophase I, subsequently, resulting in progressive loss of oocytes and infertility (12). Mandon-Pépin et al. identified a novel heterozygous mutation p.P29S (2/41, 4.9%) in the MSH5 gene in 41 women with POI (13); however, functional validation studies were lacking. In the current study, the homozygous mutation p.D487Y identified is located at a position that is highly conserved; surprisingly the variation of its homologous residue in yeast (Msh5 p.D527A) has been reported to result in poor spore viability (14,15). In our study, the Msh5D486Y/D486Y mice were infertile with atrophic ovaries, confirmed the adverse effect of human MSH5 p.D487Y on oogenesis. Moreover, human cells overexpressing MSH5 p.D487Y showed diminished survival rate and remaining more DSBs unrepaired compared with cells overexpressing wild type MSH5, indicating the impaired function in the process of HR repair. As shown by RT-PCR, besides fetal ovary, MSH5 was also expressed in granulosa cells. These results suggested that perturbations in MSH5 might not only disturb meiosis in oocytes, but also affect HR repair in granulosa cells during mitosis, followed by accelerated follicle loss through atresia, finally causing POI.
To determine the contribution of MSH5 gene to sporadic POI, we screened MSH5 by Sanger sequencing in 200 sporadic cases with POI and identified 3 heterozygous mutations (ENS T00000375755: c.1057C > A, p.L353M; c.1459G > T, p.D487Y and c.2107 A > G, p.I703V). The carriers of p.L353M and p.D487Y suffered from oligomenorrhea since menarche occurred, whereas the carrier of p.I703V experienced amenorrhea at age of 24 just after spontaneous pregnancy. While mutation p.I703V located at the ATPase domain, mutations p.L353M and p.D487Y located at the DNA binding domain of MSH5 and were highly conserved among species (Supplementary Material, Fig. S2). However, they might not be responsible for POI pathogenesis due to heterozygosity. In support of this assumption, the mother of the proband (II4) in the POI pedigree, carried heterozygous p.D487Y as well. She gave birth to four children and had no sign of POI or early menopause. Our data suggest that the biallelic causal variants in MSH5 are uncommon in idiopathic POI.
Together with the findings in HFM1, STAG3, MCM8, MCM9 and CSB-PGBD3, our studies in MSH5 further confirmed that perturbation of genes involved in DNA damage repair could lead to non-syndromic POI. The underlying mechanism-inability to repair DNA damage-is plausible and will receive increasing attention with respect to POI.
Materials and Methods
Exome sequencing and data analysis
Genomic DNA was extracted from peripheral blood leukocytes using DNeasy Blood & Tissue Kit (Qiagen). Exome sequences were captured with SureSelect Target Enrichment System for Illumina Paired-End Sequencing Library (Agilent Technologies), and DNA sequencing was performed on the Illumina platform (Illumina HiSeq). Reads were mapped to the hg19 reference genome, and variants were called and annotated using Genome Analysis Toolkit (GATK), ANNOVAR, and custom pipelines. Candidate variants were confirmed in other family members and 400 control women. Sanger sequencing of candidate genes in 200 independent sporadic patients with POI were performed to detect other variations in those genes.
Detection of MSH5 expression in primate ovary
The mRNA level of MSH5 was tested by RT-PCR in ovary, uterus, heart, brain, lung, stomach, liver, kidney, spleen, testis, pancreas, adrenal gland, intestine and muscle tissues of human fetuses (induced abortion at 21 weeks), as well as in hGCs from one patient receiving in vitro fertilization treatment and COV434 cells.
Knock-in mouse model
The mouse model carrying Msh5 p.D486Y point mutation was generated using CRISPR/Cas9 system, the homozygous mutant mice Msh5D486Y/D486Y were obtained by intercrossing of Msh5+/D486Y mice.
DNA damage and recovery assay
The human osteosarcoma U2OS cells were transiently transfected with wild type or mutant MSH5-GFP or MSH5-Flag plasmids, and were incubated in media containing Etoposide (ETO) (5μg/ml) for 1 h at 37°C to induce DSBs, followed with normal media replacement and recovery for 2 h at 37°C. Phosphorylation of the Ser-139 residue of histone variant H2AX, forming γH2AX, is an early cellular response to DSBs, which here was used as a sensitive marker for DSBs. Immunofluorescence and western blot were performed to detect DNA damage and recovery degree by staining γH2AX. The clonogenic survival rate after ETO treatment was calculated in HeLa cells, in which endogenous MSH5 was silenced with siRNA and then wild type or mutant MSH5 was overexpressed.
Statistics
Comparisons of clonogenic survival rate between cell lines were calculated using independent sample t-test. All the P values were two-sided, and P <0.05 was considered statistically significant. SPSS 19.0 computer software (IBM, Armonk, NY, USA) was used for data analysis.
Study approval
The recruitment of POI pedigree, sporadic POI patients and control women from Shandong Provincial Hospital Affiliated to Shandong University was approved by the Institutional Review Board of Center for Reproductive Medicine Shandong University. Written informed consent was obtained from all participating subjects. All animal studies were carried out in accordance with the protocols approved by the Institutional Animal Care and Use Committee at the Institute of Zoology, Chinese Academy of Sciences (CAS).
Supplementary Material
Supplementary Material is available at HMG online.
Supplementary Material
Acknowledgements
We thank the family and all the sporadic patients with POI, as well as control women for their participation in this study. We also thank CAS Key Technology Talent Program for the technical support.
Conflict of Interest statement. None declared.
Funding
The National Basic Research Program of China (973 Program) (2012CB944700), and the National Natural Science Foundation of China (81522018, 81430029, 81270662, 81471509, 81525011, 31601198). Funding to pay the Open Access publication charges for this article was provided by the National Natural Science Foundation of China (81522018, 81430029, 81270662, 81471509, 81525011, 31601198).
References
- 1. Nelson L.M. (2009) Clinical practice. Primary ovarian insufficiency. N. Engl. J. Med., 360, 606–614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Haller-Kikkatalo K., Uibo R., Kurg A., Salumets A. (2015) The prevalence and phenotypic characteristics of spontaneous premature ovarian failure: a general population registry-based study. Hum. Reprod., 30, 1229–1238. [DOI] [PubMed] [Google Scholar]
- 3. Wang J., Zhang W., Jiang H., Wu B.L. (2014) Mutations in HFM1 in recessive primary ovarian insufficiency. N. Engl. J. Med., 370, 972–974. [DOI] [PubMed] [Google Scholar]
- 4. Caburet S., Arboleda V.A., Llano E., Overbeek P.A., Barbero J.L., Oka K., Harrison W., Vaiman D., Ben-Neriah Z., Garcia-Tunon I., et al. (2014) Mutant cohesin in premature ovarian failure. N. Engl. J. Med., 370, 943–949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. AlAsiri S., Basit S., Wood-Trageser M.A., Yatsenko S.A., Jeffries E.P., Surti U., Ketterer D.M., Afzal S., Ramzan K., Faiyaz-Ul H.M., et al. (2015) Exome sequencing reveals MCM8 mutation underlies ovarian failure and chromosomal instability. J. Clin. Invest., 125, 258–262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wood-Trageser M.A., Gurbuz F., Yatsenko S.A., Jeffries E.P., Kotan L.D., Surti U., Ketterer D.M., Matic J., Chipkin J., Jiang H., et al. (2014) MCM9 mutations are associated with ovarian failure, short stature, and chromosomal instability. Am. J. Hum. Genet., 95, 754–762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Qin Y., Guo T., Li G., Tang T.S., Zhao S., Jiao X., Gong J., Gao F., Guo C., Simpson J.L., et al. (2015) CSB-PGBD3 Mutations Cause Premature Ovarian Failure. PLoS Genet., 11, e1005419.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Snowden T., Acharya S., Butz C., Berardini M., Fishel R. (2004) hMSH4-hMSH5 recognizes Holliday Junctions and forms a meiosis-specific sliding clamp that embraces homologous chromosomes. Mol. Cell, 15, 437–451. [DOI] [PubMed] [Google Scholar]
- 9. Baudat F., de Massy B. (2007) Regulating double-stranded DNA break repair towards crossover or non-crossover during mammalian meiosis. Chromosome Res., 15, 565–577. [DOI] [PubMed] [Google Scholar]
- 10. Menezo Y., Dale B., Cohen M. (2010) DNA damage and repair in human oocytes and embryos: a review. Zygote, 18, 357–365. [DOI] [PubMed] [Google Scholar]
- 11. Rothkamm K., Kruger I., Thompson L.H., Lobrich M. (2003) Pathways of DNA double-strand break repair during the mammalian cell cycle. Mol. Cell. Biol., 23, 5706–5715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Edelmann W., Cohen P.E., Kneitz B., Winand N., Lia M., Heyer J., Kolodner R., Pollard J.W., Kucherlapati R. (1999) Mammalian MutS homologue 5 is required for chromosome pairing in meiosis. Nat. Genet., 21, 123–127. [DOI] [PubMed] [Google Scholar]
- 13. Mandon-Pépin B., Touraine P., Kuttenn F., Derbois C., Rouxel A., Matsuda F., Nicolas A., Cotinot C., Fellous M. (2008) Genetic investigation of four meiotic genes in women with premature ovarian failure. Eur. J. Endocrinol., 158, 107–115. [DOI] [PubMed] [Google Scholar]
- 14. Nishant K.T., Chen C., Shinohara M., Shinohara A., Alani E. (2010) Genetic analysis of baker's yeast Msh4-Msh5 reveals a threshold crossover level for meiotic viability. PLoS Genet., 6, e1001083.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Rakshambikai R., Srinivasan N., Nishant K.T. (2013) Structural insights into Saccharomyces cerevisiae Msh4-Msh5 complex function using homology modeling. PLoS One, 8, e78753.. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.