Abstract
Background
One important cardiovascular morbidity that is associated with obesity hypoventilation syndrome (OHS) is the development of pulmonary hypertension (PH). However, few studies have assessed PH in OHS patients. Therefore, we prospectively assessed the prevalence of PH in a large sample of OHS patients.
Methods
In this prospective observational study, all consecutive OHS patients referred to the sleep disorders clinic during the study period were included. All patients underwent overnight polysomnography (PSG), spirometry, arterial blood samples and thyroid tests. Transthoracic echocardiography was performed for patients who agreed to participate in the study. PH was defined as systolic pulmonary artery pressure (SPAP) >40 mmHg.
Results
Echocardiographic data were available for 77 patients with a mean age of 60.5±11.7 years, a BMI of 43.2±10.4 kg/m2, and an Epworth Sleepiness Scale (ESS) score of 11.4±5.5. SPAP was >40 mmHg in 53 patients (68.8%), with a mean SPAP of 64.1±17.1 mmHg. There were no differences between the OHS patients with PH and those with normal PAP in terms of age, BMI, presenting symptoms, comorbidities, arterial blood gasses (ABG), and spirometric and PSG parameters. Approximately 71.4% of women and 61.9% of men with OHS also had PH. SPAP was >40–55 mmHg in 19 (24.7%) patients (18 women), >55–70 mmHg in 15 (19.5%) patients (6 women) and >70 mmHg in 19 (24.7%) patients (16 women). Severe PH (SPAP >70 mmHg) was diagnosed in 28.6% of the women and 14.3% of the men.
Conclusions
PH is very common among patients with OHS who have been referred to sleep disorders clinics. PH should be considered in the regular clinical assessment of all patients with OHS.
Keywords: Hypercapnia, obstructive sleep apnea (OSA), obesity, hypoventilation, pulmonary hypertension (PH), echocardiography, corpulmonale
Introduction
Obesity hypoventilation syndrome (OHS) was described initially in 1955 and was later associated with the term Pickwickian syndrome to reflect its association with obesity (1,2). OHS is defined as the presence of obesity [body mass index (BMI) ≥30 kg/m2], chronic hypercapnia during wakefulness [arterial partial pressure of carbon dioxide (PaCO2) >45 mmHg], and sleep-disordered breathing in the absence of other causes of chronic hypoventilation (3). OHS patients are more exposed than obese control subjects to a considerable hospitalization rate and excessive comorbidities such as hypertension, heart failure, pulmonary hypertension (PH), asthma and diabetes mellitus, resulting in higher medical resource utilization and shorter life expectancy (3,4). The incidence of OHS is increasing in parallel with the rising global incidence of extreme obesity and is therefore considered a rapidly growing public health problem.
The relationship between obstructive sleep apnea (OSA) and cardiovascular disease (CVD) is well established (5). In contrast, few studies have systematically described the cardiovascular comorbidities of patients with OHS. One important cardiovascular morbidity that has been reported in OHS is the development of right ventricular overload (RVO) and PH. Only a few studies have assessed the prevalence of PH among OHS patients, and these studies have reported that up to 50–88% of patients with OHS present with peripheral edema, PH and right heart failure (6-10). However, most previous studies have been retrospective, included small numbers of patients and lacked strict inclusion criteria to exclude PH due to other causes, and the reported severity of PH in OHS was variable (6). Therefore, we designed this study to assess the prevalence of PH in a large sample of OHS patients.
Methods
Ethics statement
The study protocol was approved by the institutional review board at King Saud University (ID: 09/2361/E), and informed consent was obtained from all participants prior to inclusion in this study.
Study protocol
This prospective observational study is a part of a large project to assess OHS patients and was conducted between January 2002 and December 2012 (4,11). Consecutive positive airway pressure therapy-naive patients (≥18 years old) who were referred to the University Sleep Disorders Centre (USDC) with clinical suspicion of OSA and had polysomnography (PSG) were included. The exclusion criteria consisted of hypnotic medications and disease that may cause hypoventilation and/or hypoxemia such as chronic lung or neurological diseases, congestive heart failure, and thoracic vertebral deformities. Figure 1 shows a flowchart of the patient recruitment process. Comorbidities were determined from the patients’ medical files. Medication lists were obtained from patients and caregivers to confirm which medications were being used at the time of the assessment. As per the USDC protocol, all patients with sleep-disordered breathing underwent arterial blood gas (ABG) analysis, spirometry, and thyroid function tests. For this study, when OHS was diagnosed, the patients were asked to undergo a transthoracic echocardiogram.
Figure 1.
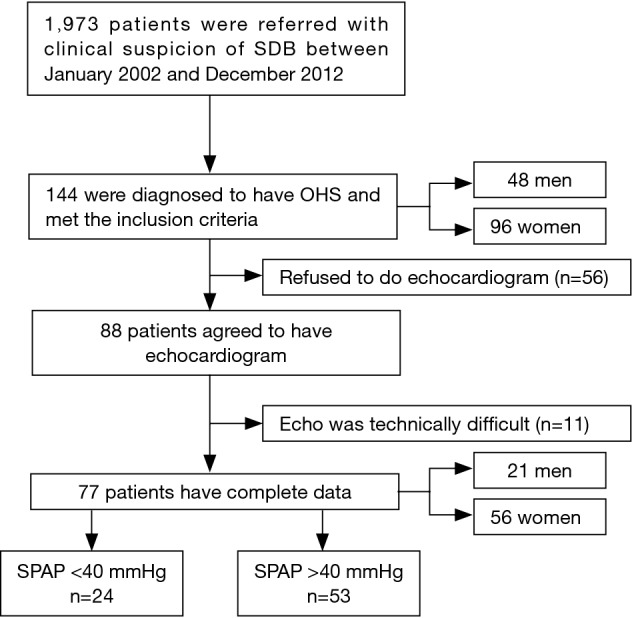
A flowchart of the patient recruitment process.
Details of the thyroid function tests and spirometry measurements have been described previously (4,11). All patients with hypothyroidism were on thyroxine replacement and their thyroxine levels were normal at the time of performing echocardiography.
PSG
All patients underwent a standard type I overnight attended sleep study with neurological, cardiac and respiratory monitoring using Alice® diagnostic equipment (Philips, Respironics Inc., Murrysville, PA, USA) (4,11). Details of the procedure have been previously described (4,11). Scoring was performed manually according to the American Academy of Sleep Medicine (AASM) scoring criteria (12). The desaturation index was defined as the number of desaturation events [≥3% decrease in oxygen (O2) saturation from the pre-event baseline] per hour of sleep.
OHS diagnosis
The AASM criteria were used to diagnose OHS: (I) awake hypercapnia (PaCO2 >45 mmHg), based on arterial PaCO2; (II) obesity (BMI >30 kg/m2); and (III) hypoventilation not primarily due to a chronic lung disease, a chest wall deformity, medication use, a neuromuscular disorder, or a known congenital or idiopathic central alveolar hypoventilation syndrome (13).
Echocardiography
Transthoracic echocardiography and Doppler were performed while the patient was awake in the morning after PSG by an experienced sonographer. As echocardiogram had been performed over several years, different echocardiographic machines had been used for the image acquisition. However, most of the studies were done using a Philips iE33 (Philips Ultrasound Bothell, WA, USA) cardiac ultrasound machine, with an electronic transducer of variable frequency and capacity for two-dimensional, M-mode, continuous and pulsed wave Doppler and color images. The data were stored digitally using commercial software (Xcelera Cardiovascular) and reported by a senior echocardiologist in accordance with published guidelines (14). Standard complete two-dimensional echocardiography was performed in all participants with chamber quantification assessment according to published criteria (15). Systolic pulmonary artery pressure (SPAP) was estimated by imaging the peak velocity of the tricuspid regurgitant jet velocity (TRV) and applying the simplified Bernoulli equation (right ventricular systolic pressure SPAP =4 TRV2+ right atrial pressure) (16). For standardization, a right atrial pressure of 10 mmHg was assumed for all patients unless clear features were present such as an inferior vena cava (IVC) diameter of >21 mm with <50% collapsibility. In such cases, 15 mmHg was used (17). Because the resting physiologic range of SPAP of normal subjects depends on age and BMI and can be as high as 40 mmHg in both normal subjects >50 years and obese people (BMI >30 kg/m2) (18,19), and to avoid false positives, PH was defined as an SPAP >40 mmHg.
The severity of PH was categorized in this study as follows: 41–55, >55–70 and >70 mmHg.
None of the included patients was on home noninvasive ventilation (NIV) treatment prior to the echocardiographic assessment.
Statistical analysis
The data are expressed as the mean and standard deviation (SD) in both the text and tables. Student’s t-tests were used to compare the means for continuous data and the Chi-square test was used for categorical variables. The results were considered statistically significant if P<0.05. The data were analyzed using Statistical Package for Social Sciences (SPSS), IBM version 22 (SPSS Inc., Chicago, IL, USA) software.
Results
During the study period, 144 patients were diagnosed with OHS. Among them, 88 patients agreed to undergo echocardiography. Echocardiography was not possible in 11 of these patients (12.5%) due to technical difficulties (Figure 1). There was no difference between the OHS patients who underwent echocardiography (n=77) and those who did not (n=67) in terms of demographics, respiratory function, and sleep parameters. Table 1 presents the demographics, baseline characteristics and respiratory findings of the 77 patients who underwent a proper SPAP assessment. The participants had a mean age of 60.5±11.7 years, a BMI of 43.2±10.4 kg/m2, and an ESS score of 11.4+5.5, with women representing 72.7% of the population. Hemoglobin level was normal in both groups, which may reflect that OHS patients have a mixed picture of chronic illness anemia and some degree of hypoxemia induced polycythemia and hence hemoglobin appears normal.
Table 1. Demographic and sleep characteristics of all participants and comparisons between patients with low and high PAP.
| Variables | All (n=77) | High SPAP (n=53) (SPAP >40 mmHg) | Normal SPAP (n=24) (SPAP ≤40 mmHg) | P value |
|---|---|---|---|---|
| Males (%) | 21 (27.3) | 13 (24.5) | 8 (33.3) | 0.5 |
| Age (years) | 60.5±11.7 | 62.0±11.5 | 56.3±11.4 | 0.057 |
| BMI (kg/m2) | 43.2±10.4 | 44.3±11.6 | 42.8±10.1 | 0.6 |
| Smoking | ||||
| Active (%) | 4 (5.2) | 4 (7.5) | 0 | 1.0 |
| Ex-smoker (%) | 10 (13.0) | 6 (11.3) | 4 (16.6) | 0.5 |
| ESS | 11.4±5.5 | 11.4±5.5 | 9.7±4.3 | 0.2 |
| pH | 7.38±0.08 | 7.4±0.1 | 7.4±0 | 0.5 |
| PaCO2 (mmHg) | 55.7±9.2 | 56.5±12.7 | 41.6±6.7 | 0.6 |
| PaO2 (mmHg) | 61.6±13.1 | 61.0±13.2 | 78.0±12.9 | 0.3 |
| HCO3 (mmol/L) | 33.0±6.3 | 33.0±6.2 | 25.7±3.4 | 0.7 |
| Hemoglobin level (g/dL) | 13.3±2.5 | 14.1±1.8 | 13.7±2.5 | 0.1 |
| Hematocrit | 39.5±7.2 | 41.6±5.3 | 40.6±7.3 | 0.2 |
| FEV1 (% predicted) | 57.8±26.3 | 56.8±26.1 | 85.3±18.0 | 0.2 |
| FVC (% predicted) | 57.3±29.0 | 57.4±24.6 | 86.6±19.3 | 0.2 |
| FEV1/FVC (%) | 82.1±9.9 | 85.5±10.1 | 84.5±11.2 | 0.4 |
| SPAP (mmHg) | 54.1±22.5 | 65.5±16.3 | 27.0±4.3 | <0.001 |
PAP, pulmonary artery pressure; BMI, body mass index; ESS, Epworth Sleepiness Scale; PaCO2, arterial partial pressure of carbon dioxide; PaO2, arterial partial pressure of oxygen; FEV1 forced expiratory volume in 1 second; FVC, forced vital capacity; SPAP, systolic pulmonary artery pressure.
Among the study group, 53 (68.8%) had elevated SPAP (PH), with a mean SPAP of 64.1±17.1 mmHg. There were no differences between the OHS patients with PH and those with normal PAP in terms of BMI, ABG, and PFT parameters (Table 1). However, OHS patients with PH were older and therefore might have a greater history of disease without therapy. Moreover, there was no difference between the OHS patients with PH and those with normal SPAP with regard to presenting symptoms and comorbidities (Table 2). At the time of the assessment, 20 (26%) patients were already on home O2. Table 2 presents a list of medications that may affect the SPAP in the entire group as well as in patients with normal and elevated SPAP. No difference was found between the two groups. Twenty patients were on bronchodilators as they were diagnosed to have bronchial asthma.
Table 2. Symptoms, comorbidities and medications of the study group.
| Variables | Whole group, % (n) | High PAP (SPAP >40 mmHg), % (n) | Normal PAP (SPAP ≤40 mmHg), % (n) | P value |
|---|---|---|---|---|
| Symptom | ||||
| Excessive daytime sleepiness | 57.1 [44] | 56.6 [30] | 58.3 [14] | 0.97 |
| Difficulty falling asleep | 19.5 [15] | 15.1 [8] | 29.2 [7] | 0.29 |
| Snoring | 75.3 [58] | 73.6 [39] | 79.2 [19] | 0.13 |
| Witnessed apnea | 36.4 [28] | 32.1 [17] | 45.8 [11] | 0.94 |
| Acidic mouth | 31.2 [24] | 28.3 [15] | 37.5 [9] | 0.48 |
| Chest pain | 26.0 [20] | 24.5 [13] | 29.2 [7] | 0.86 |
| Choking attacks during sleep | 40.3 [31] | 42.5 [22] | 37.5 [9] | 0.77 |
| Palpitation | 22.1 [17] | 24.5 [13] | 16.7 [4] | 1.00 |
| Morning headache | 36.4 [28] | 32.1 [17] | 45.8 [11] | 0.50 |
| Dry mouth | 55.8 [43] | 52.8 [28] | 62.5 [15] | 0.49 |
| Nocturia | 48.1 [37] | 49.1 [26] | 45.8 [11] | 0.95 |
| Comorbidity | ||||
| Hypertension | 76.6 [59] | 79.2 [42] | 70.8 [17] | 0.95 |
| Ischemic heart disease | 15.6 [12] | 18.9 [10] | 8.3 [2] | 0.20 |
| Diabetes mellitus | 55.8 [43] | 54.7 [29] | 58.3 [14] | 0.90 |
| Renal impairment | 10.4 [8] | 7.5 [4] | 16.6 [4] | 1.00 |
| Compensated heart failure | 16.9 [13] | 20.8 [11] | 8.3 [2] | 0.20 |
| Bronchial asthma | 28.6 [22] | 24.5 [13] | 37.5 [9] | 0.65 |
| Hypothyroidism | 23.4 [18] | 18.9 [10] | 33.3 [8] | 0.26 |
| Medication | ||||
| ACEIs and ARBs | 24.7 [19] | 22.4 [12] | 29.2 [7] | 0.60 |
| β-Blocker | 13.0 [10] | 11.3 [6] | 16.7 [4] | 0.50 |
| Calcium-channel antagonists | 22.1 [17] | 22.6 [12] | 20.8 [5] | 0.90 |
| Diuretics | 33.8 [26] | 28.3 [15] | 45.8 [11] | 0.20 |
| Bronchodilator agents | 26.0 [20] | 24.5 [13] | 29.2 [7] | – |
| Anti-platelet agents | 40.3 [31] | 50.9 [27] | 16.7 [4] | 0.01 |
| Warfarin | 5.2 [4] | 5.7 [3] | 4.2 [1] | 0.90 |
| Sildenafil | 1.3 [1] | 1.9 [1] | 0 | 0.90 |
| Home O2 | 26 [20] | 26.4 [14] | 25.0 [6] | 0.80 |
PAP, pulmonary artery pressure; ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin II receptor antagonists.
Table 3 presents the PSG parameters of all OHS patients, OHS patients with PH and those with normal PAP. There were no significant differences between the OHS patients with PH and the OHS patients with normal SPAP.
Table 3. Polysomnographic findings of all OHS patients, patients with high SPAP and patients with normal SPAP.
| Variables | All (n=88) | High PAP (SPAP >40 mmHg) (n=53) | Normal PA (SPAP ≤40 mmHg) (n=24) | P value |
|---|---|---|---|---|
| Total sleep time (min) | 195.1±89.0 | 186.3±88.4 | 145.9±54.9 | 0.04 |
| Sleep efficiency (%) | 63.4±22.1 | 73.3±18.7 | 64.8±21.2 | 0.50 |
| Stage N1 (%) | 14.0±14.0 | 12.0±11.8 | 13.7±13.7 | 0.60 |
| Stage N2 (%) | 72.5±16.9 | 68.8±14.7 | 73.4±17.7 | 0.50 |
| Stage N3 (%) | 3.4±8.4 | 4.5±8.4 | 3.2±7.9 | 0.07 |
| Stage REM (%) | 9.2±11.4 | 12.7±9.9 | 8.3±11.4 | 0.20 |
| AHI (events/hr) | 65.6±45.4 | 70.2±44.3 | 55.9±46.5 | 0.15 |
| AHI during NREM (events/hr) | 64.3±47.3 | 69.3±46.0 | 53.4±49.1 | 0.15 |
| AHI during REM (events/hr) | 54.8±41.7 | 66.8±40.7 | 47.0±31.7 | 0.12 |
| Desaturation index (events/hr) | 48.5±41.7 | 50.4±42.2 | 43.0±40.5 | 0.50 |
| Duration of nSpO2 <90% (min) | 83.1±35.1 | 92.2±35.3 | 64.1±34.7 | 0.30 |
| t<90 (%TST) | 47.2±24.4 | 48.9±25.1 | 45.3±23.7 | 0.60 |
| Lowest recorded nSpO2 (%) | 64.7±17.3 | 65.1±17.4 | 63.3±17.4 | 0.70 |
| Mean nSpO2 (%) | 84.3±7.9 | 83.7±8.4 | 86.1±5.7 | 0.20 |
| Arousal index (arousals/hr) | 64.9±42.3 | 66.8±43.0 | 59.6±41.1 | 0.50 |
OHS, obesity hypoventilation syndrome; SPAP, systolic pulmonary artery pressure; N1, stage 1 sleep; N2, stage 2 sleep, N3, stage 3 sleep; REM, rapid eye movement sleep; AHI, apnea hypopnea index; nSpO2, nocturnal oxygen saturation; t<90 (%TST), percentage of sleep time with SpO2 <90%.
Echocardiography
Ten patients (13%) exhibited systolic left ventricular (LV) ejection function (EF) <55% (range, 15–50%). LV diastolic dysfunction was reported in nine patients (12%) (with normal LVEF): grade I in 6 patients, grade II in 2 patients and grade III in 1 patient. Figure 2 presents the distribution of the severity of PH in the OHS patients based on SPAP. Approximately 71.4%of women and 61.9% of men with OHS also had PH. SPAP was >40–55 mmHg in19 (24.7%) patients (18 women), >55–70 mmHg in 15 (19.5%) patients (6 women) and >70 mmHg in 19 (24.7%) patients (16 women). In women with OHS (n=56), the SPAP was 55.0±23.0 mmHg, and in men (n=20), the SPAP was 51.5±21.6 mmHg (P=0.26). Among women, 28.6% had normal SPAP versus 38.1% of the men. Severe PH (SPAP >70 mmHg) was diagnosed in 28.6% of the women and 14.3% of the men.
Figure 2.
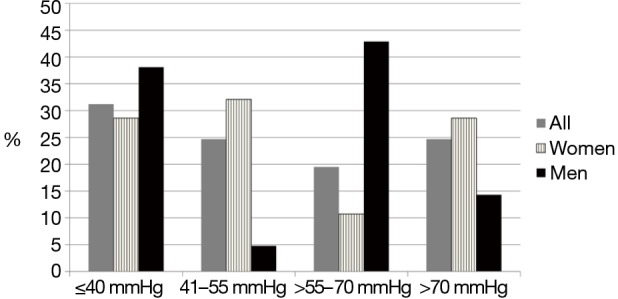
The distribution of the severity of pulmonary hypertension (PH) in obesity hypoventilation patients based on systolic pulmonary artery pressure (SPAP).
Discussion
Although PH is one of the characteristic comorbidities of OHS, only a few studies have addressed this topic. Our study is the largest study to assess the prevalence of PH among OHS patients. This study revealed that PH is prevalent among patients with OHS presenting to a sleep medicine clinic: 71.4% of women and 61.9% of men with OHS also had PH. In an earlier small study, Kessler et al. used right heart catheterization (RHC) in 27 OHS patients and reported PH, as defined as a mean pulmonary artery pressure (mPAP) >20 mmHg, in 59% of patients (9). A recent retrospective study that used echocardiography to assess SPAP reported a prevalence of PH of 52% in 25 patients with OHS (10). In another study that used RHC to assess PH in 26 OHS patients undergoing bariatric surgery and defined PH as an mPAP >20 mmHg, PH was diagnosed in 88% of the patients (20). OHS patients are more prone to develop PH and corpulmonale compared with patients with isolated OSA (21). In a small study of 20 patients with OHS (obtained from a population registry), Berg et al. reported that OHS patients were significantly more likely to suffer corpulmonale compared with OSA patients (odds ratio of 9; 95% CI: 1.4–57.1) (22). Table 4 summarizes the studies that have assessed PH in patients with OHS, sample characteristics, PH diagnostic criteria and PH prevalence. The differences in the prevalence of PH among the different studies could be related to the different study designs, definitions used to diagnose OHS and PH and the wide range of OHS severity in the studied patients; in addition, there were differences in the BMI and age of the participants.
Table 4. A summary of previous studies that assessed pulmonary hypertension (PH) in patients with obesity hypoventilation syndrome.
| Study | Study design | Sample characteristics | PAP assessment method | Definition of PH | Prevalence of PH |
|---|---|---|---|---|---|
| Kessler et al. (9) | Prospectively collected clinical data | n=27; age =61 yr; BMI =40 kg/m2 | Right-heart catheterization | Mean PAP >20 mmHg | 59% |
| Kauppert et al. (23) | Prospective cross-sectional study of patients who were on NIV therapy | n=21; BMI =45 kg/m2; prior to enrollment, patients were treated with NIV for a mean of 1.9 yr. PCO2 and PO2 data prior to NIV were not reported | Right-heart catheterization | Mean PAP >20 mmHg | 81% had mild to moderate PH |
| Alawami et al. (10) | A retrospective analysis of dataset | n=25 | Transthoracic echocardiography | Systolic PAP >30 mmHg | 88% |
| Sugerman et al. (24) | Prospective study | n=26; age =44 yr; BMI not reported; did not use the standard diagnostic criteria for OHS; pre-operative assessment for Bariatric surgery | Right-heart catheterization | Mean PAP ≥20 mmHg | 88% |
| Held et al. (8) | Retrospective study | n=12 (3 of them had co-existing COPD); age =62 yr; BMI =36 kg/m2 | Right-heart catheterization and transthoracic echocardiography | Mean PAP at rest ≥25 mmHg or mean PAP at rest <25 mmHg and mean PAP at exercise ≥50 mmHg or, an echocardiographic systolic PAP ≥50 mmHg at rest | Not reported (mean PAP =49±13 mmHg) |
| Marik & Desai (25) | Retrospective study | n=61 (77% women and 92% black); age =59 yr; BMI =48.9 kg/m2 | Transthoracic echocardiography; the peak tricuspid regurgitant velocity was used; to estimate the systolic PAP | Systolic PAP >35 mmHg; systolic PAP >45 mmHg (moderate to severe PH) | 77%; 41% (moderate to severe PH) |
| Current study | Prospective observational cross-sectional study | n=77; age =60.5 yr; no prior treatment with NIV | Transthoracic echocardiography | Systolic PAP >40 mmHg | 68.8% |
The underlying mechanism of PH in patients with OHS is thought to be chronic diurnal and nocturnal hypoxia, hypercapnia, and acidosis (21,26). Other factors that contribute to PH in patients with OHS include restrictive lung disease related to severe obesity and the large intrathoracic pressure shifts in the respiratory cycle due to increased upper airway resistance (21). Upper airway obstruction results in profound negative intrathoracic pressures during inspiration, which increases venous return and right ventricle filling, causing a leftward shift of the interventricular septum. Hence, LV filling will be reduced, and LV stroke volume will decrease.
There was no difference between OHS with PH and those without in most measured parameters. However, OHS patients with PH were older, which may indicate a greater history of disease without therapy, and hence a higher risk of developing PH. Moreover, some patients may genetically have a better adaptation to chronic hypoxia (27). Recent data suggest that there are several candidate genes that may underlie the adaptation or maladaptation to chronic hypoxia (27).
The established mainstay treatment of OHS is NIV, which is effective at improving blood gas and respiratory parameters, sleep quality, daytime vigilance and quality of life (3,28). Despite limited data, current evidence suggests that NIV is effective at treating PH secondary to OHS. Therefore, proper assessment of PAP is needed in all patients with OHS. In an earlier study, Castro-Añón et al. used echocardiography to assess PAP and right ventricular dilatation among 30 OHS patients and reported RVO in 43.3% of the patients (20). After 6 months of NIV, the authors reported a reduction in PAP from 58 to 44 mmHg (P=0.014); however, there were no significant changes in the RVO percentage (20). Two recent studies assessed the effect of NIV on PAP in patients with OHS. In 21 OHS patients who were treated with NIV for a mean of 1.9 years, Kauppert et al. used RHC to assess PH (defined as Mpap >25 mmHg) and then assessed the effect of NIV on mPAP (NIV ≥3 months) (23). PH was diagnosed in 43% of patients; however, 3 of the 9 patients with PH had a pulmonary capillary wedge pressure (PCWP) >15 mmHg (23). mPAP was inversely related to the long-term use of NIV. Nevertheless, PH persisted in some patients despite regular use of NIV, which suggests that some degree of vascular remodeling or endothelial dysfunction may persist even after amelioration of nocturnal gas exchange (23). In another study, Held et al. reported 18 patients with severe PH due to alveolar hypoventilation, who markedly improved after 3 months of NIV (8). At baseline, mPAP and pulmonary vascular resistance (PVR) were 50 mmHg and 6–7 Wood units, respectively, which decreased to 30 mmHg and 3–4 Wood units with NIV (8). The above findings indicate the need for early recognition and treatment of patients with OHS using NIV to prevent the progression of PH and the occurrence of permanent vascular remodeling changes. Nevertheless, compliance with NIV in patients with OHS is a major problem (29,30). Recent data suggest that bariatric surgery may improve PH in the short-term (31).
The strengths of this study include the size of the cohort and that every single subject who was referred underwent PSG, provided an arterial blood sample, and underwent thyroid and pulmonary function tests (PFT). However, there are some limitations that must be acknowledged. One limitation of this study is the use of echocardiography to assess PH because assessment of SPAP by echocardiography may not be accurate (32). Nevertheless, echocardiography tends to underestimate SPAP rather than overestimate it (33-35). Moreover, Doppler techniques have been used to assess pulmonary vascular hemodynamics with accuracy and good reproducibility in obese patients with OSA. In one study, Sajkov et al. showed a correlation of r=0.96 (P<0.001) between catheter-measured and Doppler-estimated PAP (36). In another study of patients with OSA and a BMI of 34±6 kg/m2, Alchanatis et al. showed that Doppler echocardiography-derived PAP is valid and accurate with a good correlation with catheter-measured PAP (r=0.957, P<0.001) (37). Unfortunately, using cardiac catheterization in patients with OHS is too invasive to complete safely on a presentation on NIV naïve OHS patients. In one study that assessed PAP using RHC, OHS patients were treated with NIV for a mean of 1.9 years prior to performing RHC (23). Another potential limitation is the fact that most of the studied samples are women. However, recent data suggest that OHS is more prevalent among women (11,25,29).
In conclusion, our data suggest that PH is common among patients with OHS who are referred to sleep disorders clinics. Although the role of PH in the long-term prognosis of OHS patients is unknown, PH should be considered in the regular clinical assessment of all patients with OHS.
Acknowledgements
Funding: The work was funded by the Strategic Technologies Program of the National Plan for Sciences and Technology and Innovation in the Kingdom of Saudi Arabia (08-MED511-02).
Ethical Statement: The study protocol was approved by the institutional review board at King Saud University (ID: 09/2361/E), and informed consent was obtained from all participants prior to inclusion in this study.
Disclaimer: The study sponsor played no role in the study design; the collection, analysis or interpretation of the data; the writing of the manuscript; or the decision to submit the manuscript.
Footnotes
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- 1.Bickelmann AG, Burwell CS, Robin ED, et al. Extreme obesity associated with alveolar hypoventilation; a Pickwickian syndrome. Am J Med 1956;21:811-8. 10.1016/0002-9343(56)90094-8 [DOI] [PubMed] [Google Scholar]
- 2.Auchincloss JH, Jr, Cook E, Renzetti AD. Clinical and physiological aspects of a case of obesity, polycythemia and alveolar hypoventilation. J Clin Invest 1955;34:1537-45. 10.1172/JCI103206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Al Dabal L, Bahammam AS. Obesity hypoventilation syndrome. Ann Thorac Med 2009;4:41-9. 10.4103/1817-1737.49411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.BaHammam AS Prevalence, clinical characteristics, and predictors of obesity hypoventilation syndrome in a large sample of Saudi patients with obstructive sleep apnea. Saudi Med J 2015;36:181-9. 10.15537/smj.2015.2.9991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thomas JJ, Ren J. Obstructive sleep apnoea and cardiovascular complications: perception versus knowledge. Clin Exp Pharmacol Physiol 2012;39:995-1003. 10.1111/1440-1681.12024 [DOI] [PubMed] [Google Scholar]
- 6.Naeije R. Pulmonary hypertension in hypoventilation syndromes. Eur Respir J 2014;43:12-5. 10.1183/09031936.00185213 [DOI] [PubMed] [Google Scholar]
- 7.Mokhlesi B, Tulaimat A. Recent advances in obesity hypoventilation syndrome. Chest 2007;132:1322-36. 10.1378/chest.07-0027 [DOI] [PubMed] [Google Scholar]
- 8.Held M, Walthelm J, Baron S, et al. Functional impact of pulmonary hypertension due to hypoventilation and changes under noninvasive ventilation. Eur Respir J 2014;43:156-65. 10.1183/09031936.00147712 [DOI] [PubMed] [Google Scholar]
- 9.Kessler R, Chaouat A, Schinkewitch P, et al. The obesity-hypoventilation syndrome revisited: a prospective study of 34 consecutive cases. Chest 2001;120:369-76. 10.1378/chest.120.2.369 [DOI] [PubMed] [Google Scholar]
- 10.Alawami M, Mustafa A, Whyte K, et al. Echocardiographic and electrocardiographic findings in patients with obesity hypoventilation syndrome. Intern Med J 2015;45:68-73. 10.1111/imj.12620 [DOI] [PubMed] [Google Scholar]
- 11.BaHammam AS , Pandi-Perumal SR, Piper A, et al. Gender differences in patients with obesity hypoventilation syndrome. J Sleep Res 2016;25:445-53. 10.1111/jsr.12400 [DOI] [PubMed] [Google Scholar]
- 12.Berry RB, Brooks R, Gamaldo CE, et al. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications, Version 2.1. Darien, Illinois: American Academy of Sleep Medicine, 2014. [Google Scholar]
- 13.American Academy of Sleep Medicine (AASM). International classification of sleep disorders (ICSD), 3rd ed. Darien, IL: AASM, 2014. [Google Scholar]
- 14.Thomas JD, Adams DB, Devries S, et al. Guidelines and recommendations for digital echocardiography. J Am Soc Echocardiogr 2005;18:287-97. 10.1016/j.echo.2005.01.010 [DOI] [PubMed] [Google Scholar]
- 15.Lang RM, Bierig M, Devereux RB, et al. Recommendations for chamber quantification: a report from the American Society of Echocardiography's Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr 2005;18:1440-63. 10.1016/j.echo.2005.10.005 [DOI] [PubMed] [Google Scholar]
- 16.Hatle L, Angelsen BA, Tromsdal A. Non-invasive estimation of pulmonary artery systolic pressure with Doppler ultrasound. Br Heart J 1981;45:157-65. 10.1136/hrt.45.2.157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rudski LG, Lai WW, Afilalo J, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr 2010;23:685-713; quiz 86-8. 10.1016/j.echo.2010.05.010 [DOI] [PubMed] [Google Scholar]
- 18.Bossone E, D'Andrea A, D'Alto M, et al. Echocardiography in pulmonary arterial hypertension: from diagnosis to prognosis. J Am Soc Echocardiogr 2013;26:1-14. 10.1016/j.echo.2012.10.009 [DOI] [PubMed] [Google Scholar]
- 19.McQuillan BM, Picard MH, Leavitt M, et al. Clinical correlates and reference intervals for pulmonary artery systolic pressure among echocardiographically normal subjects. Circulation 2001;104:2797-802. 10.1161/hc4801.100076 [DOI] [PubMed] [Google Scholar]
- 20.Castro-Añón O, Golpe R, Pérez-de-Llano LA, et al. Haemodynamic effects of non-invasive ventilation in patients with obesity-hypoventilation syndrome. Respirology 2012;17:1269-74. 10.1111/j.1440-1843.2012.02252.x [DOI] [PubMed] [Google Scholar]
- 21.Friedman SE, Andrus BW. Obesity and pulmonary hypertension: a review of pathophysiologic mechanisms. J Obes 2012;2012:505274. [DOI] [PMC free article] [PubMed]
- 22.Berg G, Delaive K, Manfreda J, et al. The use of health-care resources in obesity-hypoventilation syndrome. Chest 2001;120:377-83. 10.1378/chest.120.2.377 [DOI] [PubMed] [Google Scholar]
- 23.Kauppert CA, Dvorak I, Kollert F, et al. Pulmonary hypertension in obesity-hypoventilation syndrome. Respir Med 2013;107:2061-70. 10.1016/j.rmed.2013.09.017 [DOI] [PubMed] [Google Scholar]
- 24.Sugerman HJ, Baron PL, Fairman RP, et al. Hemodynamic dysfunction in obesity hypoventilation syndrome and the effects of treatment with surgically induced weight loss. Ann Surg 1988;207:604-13. 10.1097/00000658-198805000-00015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marik PE, Desai H. Characteristics of Patients With the "Malignant Obesity Hypoventilation Syndrome" Admitted to an ICU. J Intensive Care Med 2013;28:124-30. 10.1177/0885066612444261 [DOI] [PubMed] [Google Scholar]
- 26."2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS)." Nazzareno Galie, Marc Humbert, Jean-Luc Vachiery, Simon Gibbs, Irene Lang, Adam Torbicki, Gerald Simonneau, Andrew Peacock, Anton Vonk Noordegraaf, Maurice Beghetti, Ardeschir Ghofrani, Miguel Angel Gomez Sanchez, Georg Hansmann, Walter Klepetko, Patrizio Lancellotti, Marco Matucci, Theresa McDonagh, Luc A. Pierard, Pedro T. Trindade, Maurizio Zompatori and Marius Hoeper. Eur Respir J 2015; 46: 903-975. Eur Respir J 2015;46:1855-6. [DOI] [PubMed] [Google Scholar]
- 27.Ronen R, Zhou D, Bafna V, et al. The genetic basis of chronic mountain sickness. Physiology (Bethesda) 2014;29:403-12. 10.1152/physiol.00008.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bahammam AS, Al-Jawder SE. Managing acute respiratory decompensation in the morbidly obese. Respirology 2012;17:759-71. 10.1111/j.1440-1843.2011.02099.x [DOI] [PubMed] [Google Scholar]
- 29.Masa JF, Corral J, Caballero C, et al. Non-invasive ventilation in obesity hypoventilation syndrome without severe obstructive sleep apnoea. Thorax 2016;71:899-906. 10.1136/thoraxjnl-2016-208501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Masa JF, Corral J, Alonso ML, et al. Efficacy of Different Treatment Alternatives for Obesity Hypoventilation Syndrome. Pickwick Study. Am J Respir Crit Care Med 2015;192:86-95. 10.1164/rccm.201410-1900OC [DOI] [PubMed] [Google Scholar]
- 31.Sheu EG, Channick R, Gee DW. Improvement in severe pulmonary hypertension in obese patients after laparoscopic gastric bypass or sleeve gastrectomy. Surg Endosc 2016;30:633-7. 10.1007/s00464-015-4251-5 [DOI] [PubMed] [Google Scholar]
- 32.Balachandran JS, Masa JF, Mokhlesi B. Obesity Hypoventilation Syndrome Epidemiology and Diagnosis. Sleep Med Clin 2014;9:341-7. 10.1016/j.jsmc.2014.05.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Claessen G, La Gerche A, Voigt JU, et al. Accuracy of Echocardiography to Evaluate Pulmonary Vascular and RV Function During Exercise. JACC Cardiovascular imaging. 2016;9:532-43. 10.1016/j.jcmg.2015.06.018 [DOI] [PubMed] [Google Scholar]
- 34.Fisher MR, Forfia PR, Chamera E, et al. Accuracy of Doppler echocardiography in the hemodynamic assessment of pulmonary hypertension. Am J Respir Crit Care Med 2009;179:615-21. 10.1164/rccm.200811-1691OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Howard LS, Grapsa J, Dawson D, et al. Echocardiographic assessment of pulmonary hypertension: standard operating procedure. Eur Respir Rev 2012;21:239-48. 10.1183/09059180.00003912 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sajkov D, Cowie RJ, Bradley JA, et al. Validation of new pulsed Doppler echocardiographic techniques for assessment of pulmonary hemodynamics. Chest 1993;103:1348-53. 10.1378/chest.103.5.1348 [DOI] [PubMed] [Google Scholar]
- 37.Alchanatis M, Tourkohoriti G, Kakouros S, et al. Daytime pulmonary hypertension in patients with obstructive sleep apnea: the effect of continuous positive airway pressure on pulmonary hemodynamics. Respiration 2001;68:566-72. 10.1159/000050574 [DOI] [PubMed] [Google Scholar]


