ABSTRACT
Perfusion-independent regulation of epithelial pattern formation by the vasculature during organ development and regeneration is of considerable interest for application in restoring organ function. During murine submandibular salivary gland development, the vasculature co-develops with the epithelium during branching morphogenesis; however, it is not known whether the vasculature has instructive effects on the epithelium. Using pharmacological inhibitors and siRNA knockdown in embryonic organ explants, we determined that VEGFR2-dependent signaling is required for salivary gland epithelial patterning. To test directly for a requirement for endothelial cells in instructive epithelial patterning, we developed a novel ex vivo cell fractionation/reconstitution assay. Immuno-depletion of CD31+ endothelial cells in this assay confirmed a requirement for endothelial cells in epithelial patterning of the gland. Depletion of endothelial cells or inhibition of VEGFR2 signaling in organ explants caused an aberrant increase in cells expressing the ductal proteins K19 and K7, with a reduction in Kit+ progenitor cells in the endbuds of reconstituted glands. Addition of exogenous endothelial cells to reconstituted glands restored epithelial patterning, as did supplementation with the endothelial cell-regulated mesenchymal factors IGFBP2 and IGFBP3. Our results demonstrate that endothelial cells promote expansion of Kit+ progenitor cells and suppress premature ductal differentiation in early developing embryonic submandibular salivary gland buds.
KEY WORDS: Salivary gland development, Epithelial patterning, Endothelial cells, Ductal differentiation, Progenitor cell, Endothelial cell, Mouse
Summary: In an ex vivo gland reconstitution model, perfusion-independent signaling by CD31+ endothelial cells opposes the emergence of K19+/K7+ cells in the endbuds of early developing submandibular salivary glands.
INTRODUCTION
Organogenesis requires the precise regulation of progenitor cell populations, and how these processes are controlled is currently of great interest. Heterotypic cell interactions are required for organogenesis in many organs, including the salivary gland (Kwon and Larsen, 2015; Nelson and Larsen, 2015). Classical tissue recombination experiments between epithelium and mesenchyme and other studies have demonstrated a requirement for mesenchymal cells in epithelial pattern formation during salivary gland branching morphogenesis. (Hoffman et al., 2002; Tucker, 2007; Wei et al., 2007; Wells et al., 2013; Kera et al., 2014). Additional studies have indicated that Kit+ (Lombaert et al., 2008, 2013; Patel et al., 2014) and cytokeratin 5 (K5; KRT5 – Mouse Genome Informatics)+ (Knox et al., 2013, 2010) epithelial progenitor cells are important for salivary gland development and homeostasis. Kit-expressing progenitor cells were shown to be regulated by fibroblast growth factor produced by the mesenchyme and Kit ligand, which is produced by both the mesenchyme and the epithelium (Lombaert et al., 2013). Developing innervation was shown to regulate the K5+ epithelial progenitor cells (Knox et al., 2010; Nedvetsky et al., 2014; Mattingly et al., 2015), which reciprocally influenced development of the emerging innervation. Involvement of the developing vasculature in salivary gland epithelial development, however, has not been reported.
The circulatory system is connected to every organ and is required for sustaining the viability and function of every adult organ. Studies in several organs, including liver and pancreas, demonstrate a non-nutritional requirement for endothelial cells within the vasculature in early organ patterning prior to the establishment of a functional vascular network (Matsumoto et al., 2001; Lammert et al., 2001; Lazarus et al., 2011; Cleaver and Dor, 2012; Schlieve et al., 2016). Endothelial precursor cells are now acknowledged to produce angiocrine factors, which are paracrine- and juxtacrine-acting factors that control early organ patterning. Multiple angiocrine factors are produced by endothelial cells in every organ, and the profile of angiocrine factors is known to be organ specific (Rafii et al., 2016; Azizoglu and Cleaver, 2016). Angiocrine factors are known to regulate epithelial cell behaviors such as survival and proliferation, polarity, and differentiation (Ingthorsson et al., 2010; Hagiwara et al., 2015; Jaramillo et al., 2015; Kao et al., 2015). Recent evidence demonstrates that vascular endothelial cell-mediated signaling regulates stem/progenitor cell renewal and differentiation during organ development, homeostasis and repair/regeneration (Rafii et al., 2016).
As little is known regarding the relationship between endothelial and epithelial cells during salivary gland development, we hypothesized that endothelial cells have a non-nutritional function in early salivary gland epithelial patterning. To address this hypothesis, we used mouse submandibular salivary gland (SMG) organ explants, which recapitulate the morphogenesis and differentiation of the developing salivary gland in vivo (Yamada and Cukierman, 2007; Larsen et al., 2017). With explant culture, it is possible to manipulate the vasculature, without the complications of doing so in vivo, in a perfusion-independent manner. We previously identified a number of genes that are expressed in endothelial cells during early SMG branching morphogenesis (Larsen et al., 2017), including vascular endothelial growth factor receptor 2 (VEGFR2), which is also known as fetal liver kinase-1 (Flk-1), kinase insert domain receptor (KDR) and cluster of differentiation 309 (CD309). VEGFR2 is a primary mediator of vascular development and angiogenesis that regulates endothelial cell survival, cell proliferation, and migration (Chavakis and Dimmeler, 2002; Schmidt et al., 2007; Marcelo et al., 2013). We found that pharmacological inhibition and siRNA-mediated genetic manipulation of VEGFR2 signaling in SMG explants altered epithelial patterning by reducing the number of immature endbuds.
To test the requirement for endothelial cells specifically in salivary epithelial patterning, we developed a novel SMG cell fractionation/reconstitution assay. Using immuno-depletion of the endothelial cells from the mesenchyme of reconstituted glands, we confirmed a requirement for endothelial cells in epithelial patterning. Interestingly, both assays showed a reduction in the Kit progenitor cell population accompanied by increased K19 and K7 in the endbuds with disruption of endothelial cell signaling, and supplementation with exogenous endothelial cells reversed this trend in the SMG cell fractionation/reconstitution assay. To identify endothelial-produced factors that regulate epithelial progenitor cells, we screened conditioned media produced by endothelial cell-depleted and endothelial cell-supplemented mesenchyme. Identified in the screen were secreted factors, including insulin-like growth factor binding protein 2 and 3 (IGFBP2 and IGFBP3), which contributed to the partial restoration of endothelial cell-depleted and VEGFR2-inhibited epithelial organ patterning. We here provide the first evidence of endothelial cell-mediated regulation of epithelial patterning in the developing salivary gland.
RESULTS
Vascular network development is coordinated with branching of the salivary gland epithelium
To determine how patterning of the vasculature relates to patterning of the developing mouse submandibular salivary gland epithelium, we examined the cluster of differentiation 31 (CD31; PECAM1 – Mouse Genome Informatics)+ vasculature in embryonic day (E) 11.5-E13.5 submandibular salivary glands (SMGs). SMGs were removed from mouse embryos, fixed, and immediately prepared for immunocytochemistry (ICC) (Fig. 1). Between E11.5 and E12.5, a discontinuous vascular network was evident that included CD31+ cell islands and discontinuous vessels within the mesenchyme surrounding the primary epithelial bud (Fig. 1A,B). By E13, fewer endothelial islands were evident with increased CD31+ cells present in elongated vascular networks that were distinct from Tubb3+ innervation. At E13-E13.5, clefts separate the primary epithelial bud into three to five endbuds concomitant with development of a continuous vessel network. CD31+ and VEGFR2+ vasculature was frequently detected inside clefts, which are indentations in the basement membrane that subdivide the buds and subsequently define the boundary between the terminal proacinar structures and the emerging secondary ducts (Fig. 1B,C). The schematic in Fig. 1D summarizes the co-development of the vasculature with the epithelium in the early developing SMG.
Fig. 1.
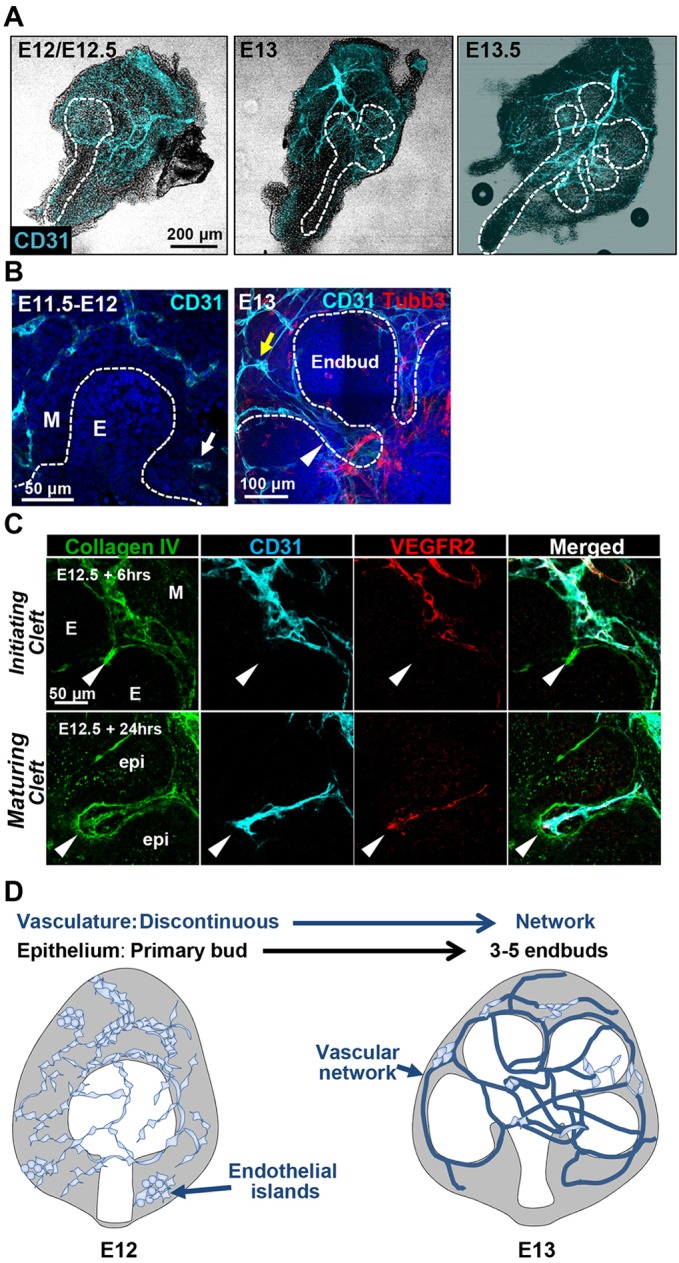
Co-development of epithelium and vasculature in early developing submandibular gland. (A) Mouse embryonic salivary glands were excised and subjected to ICC and confocal imaging to detect CD31+ (cyan) endothelial cells, shown superimposed on a brightfield image. (B) Single confocal images of the glands without brightfield are shown at higher magnification. A CD31+ semi-discontinuous vasculature with some isolated endothelial islands (white arrow) was observed in E11.5-E12.5 mesenchyme (M) that was largely isolated from the emerging epithelial bud (E). In E13-E13.5 glands, a largely continuous vessel network (yellow arrow) was observed that progressively surrounds the epithelium and is distinct from the developing nerves (Tubb3+, red). Note that the CD31+ vessels penetrate into the maturing epithelial clefts at E13 (white arrowhead). (C) CD31+ vessels persist in E12.5 glands cultured ex vivo (shown here after 6 h and after 24 h). CD31+ endothelial cells also express VEGFR2 (red) and collagen IV (green). Because collagen IV is incorporated into the basement membrane of both the endothelial cells and the epithelial cells, it defines the boundary between the epithelium and mesenchyme. CD31+/VEGFR2+/collagen IV+ endothelial cells are found adjacent to the initiating epithelial clefts at E12+6 h growth (arrowheads in upper panels) and ingress into maturing clefts after 24 h of culture (arrowheads in lower panels). (D) Schematic summarizing co-development of the primary epithelial bud with vasculature in the early submandibular gland. Endothelial cells comprising discontinuous vasculature are found in mesenchyme at E12/E12.5. Developing vessels subsequently undergo elongation and maturation concomitant with branching of the primary epithelial bud.
VEGFR2 signaling and vasculature development promote epithelial patterning in SMG organ explants to favor endbud over duct formation
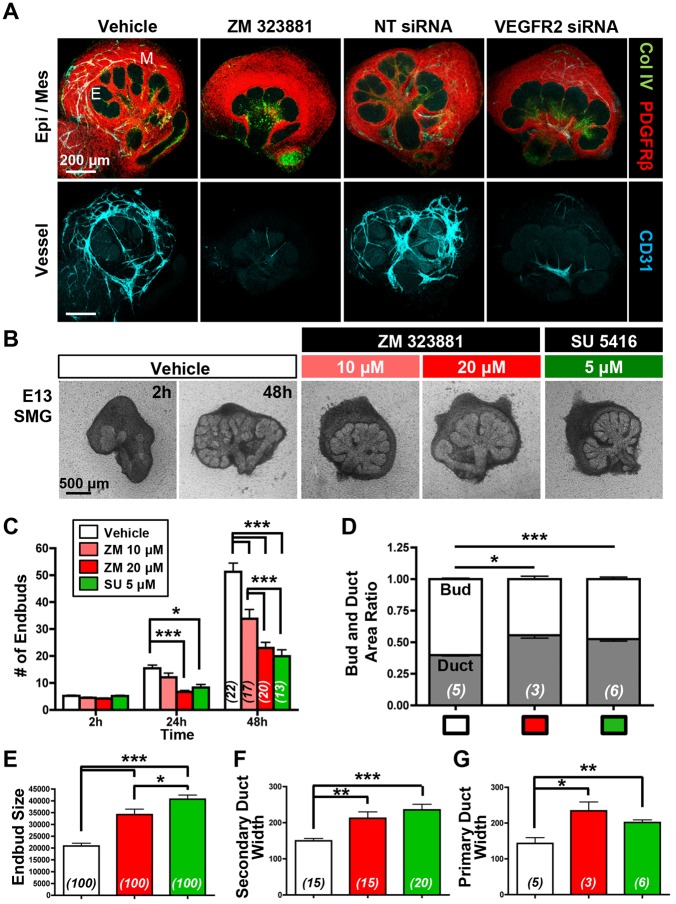
We previously identified endothelial genes enriched in the clefts of developing salivary glands through data mining of a publicly available salivary gland gene expression database (Larsen et al., 2017). Because one of the cleft-enriched endothelial genes, VEGFR2, is expressed in CD31+ endothelial cells (Fig. 1C), and VEGFR2 influences epithelial development in other organs (Matsumoto et al., 2001; Lammert et al., 2001; Lazarus et al., 2011; Magenheim et al., 2011), we manipulated VEGFR2 in salivary gland organ explant cultures. The use of organ explants is ideal for examining the influence of endothelial cell signaling on epithelial pattern formation in a perfusion-independent manner as the explants lack perfusion but recapitulate organ morphogenesis and differentiation in vivo. Both pharmacological inhibition with ZM 323881, a selective inhibitor of VEGFR2 tyrosine kinase activity (Whittles et al., 2002), and VEGFR2 siRNA knockdown reduced vasculature development relative to negative controls (Fig. 2A). ICC for cleaved caspase 3 (CC3), an executioner caspase that is activated by cleavage by both extrinsic and intrinsic apoptosis pathways (Salvesen and Dixit, 1997), demonstrated a correlation of CC3 with CD31+ cells, consistent with a loss of endothelial cells due to apoptosis with VEGFR2 inhibition (Fig. S1). Interestingly, VEGFR2 inhibition with ZM 323881 or with SU 5416, a chemically distinct VEGFR2-specific pharmacological inhibitor (Fong et al., 1999), also significantly reduced epithelial branching morphogenesis and altered epithelial patterning in the explants relative to vehicle control (Fig. 2A,B). Similar responses were obtained with VEGFR2 inhibition in cultures of SMGs from E12-E13.5 embryos (Fig. S1). Quantitative evaluation of epithelial morphology in the explants cultured for 48 h indicated that there was reduced branching morphogenesis, as demonstrated by a reduction in total number of endbuds (Fig. 2C), and an increase in the average endbud size (Fig. 2E) with VEGFR2 inhibition. Interestingly, there was an increase in the ratio of ductal area relative to bud area (Fig. 2D), and the widths of the main and secondary ducts were increased upon VEGFR2 inhibition (Fig. 2F,G). Together, these data indicate that VEGFR2 signaling in developing vasculature influences epithelial patterning by promoting branching morphogenesis to favor endbud formation over duct formation.
Fig. 2.
VEGFR2 signaling and vasculature development promote epithelial patterning in SMG organ culture. (A) Both pharmacological VEGFR2 inhibition (ZM 323881, 20 μM) and VEGFR2 siRNA (400 nM)-mediated knockdown inhibited vascular formation in E12.5 SMG organ explants cultured for 48 h, with reduced CD31+ (cyan) vasculature relative to negative controls, vehicle (DMSO) and non-targeting (NT) siRNA treatment. ICC and confocal microscopy were performed to outline the epithelial basement membrane (Col IV, green), mesenchyme (PDGFRβ, red) and vasculature (Col IV, green; CD31, cyan). Note that the epithelium appears to be less branched upon VEGFR2 inhibition or knockdown. E, epithelial bud; M, mesenchyme. (B) E13 glands were cultured as organ explants in the presence of two distinct VEGFR2 inhibitors (ZM 323881 and SU 5416) for 48 h at the indicated doses, again demonstrating disrupted epithelial organ patterning. (C-G) Brightfield images from multiple glands (n indicated on bars) were used to quantify epithelial structures. (C) Endbud numbers in VEGFR2-inhibitor treated glands were significantly reduced in ex vivo culture for 24 h and 48 h (n=total number of glands from four experiments). (D) The bud/duct ratio of treated glands, compared with vehicle control. Buds were reduced but ducts were enlarged (n=number of glands). In inhibitor-treated glands, individual bud size (E) was enlarged (n=total number of endbuds from five experiments) and the widths of secondary duct and primary duct (F,G) were widened, respectively (n=total number of ducts from five experiments). Data are mean±s.e.m. Two-way ANOVA (C) and Student's t-test (one-tailed) (D-G) were performed for statistical analysis (*P<0.05, **P<0.01, ***P<0.001).
CD31+ cell-dependent vasculature promotes epithelial patterning by promoting branching morphogenesis in a SMG cell fractionation/reconstitution assay
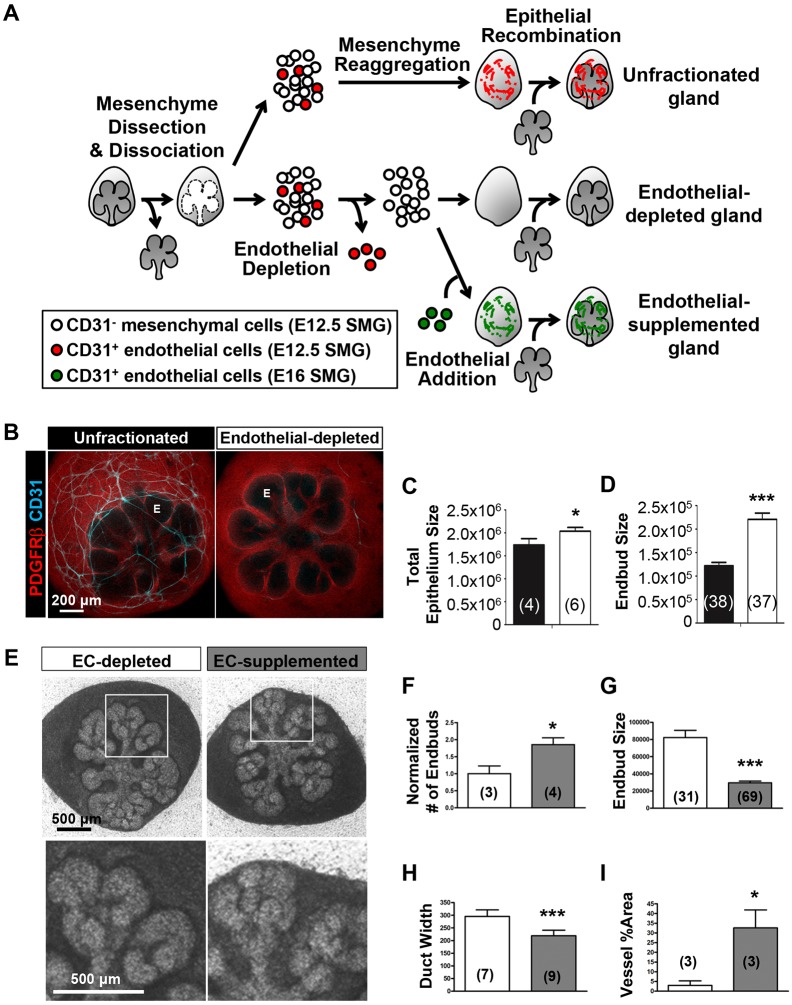
To investigate further whether CD31+ endothelial cells can influence SMG epithelial patterning, we developed a novel ex vivo SMG cell fractionation/reconstitution assay (Fig. 3A). We first microdissected SMG epithelium and mesenchyme and then enzymatically dissociated SMG mesenchyme cells. We then used magnetic-activated cell sorting (MACS)-based immunodepletion to remove CD31+ endothelial cells from the mesenchyme. Unfractionated total mesenchyme cells, mesenchyme immunodepleted of CD31+ endothelial cells, or mesenchyme immunodepleted of CD31+ endothelial cells and supplemented with exogenous CD31+ endothelial cells were then recombined with intact SMG epithelial rudiments. The gland is reconstituted over 48 h of culture, and the vasculature reassembles in conditions where CD31+ endothelial cells are included.
Fig. 3.
CD31 cell-dependent vasculature development promotes epithelial patterning in an SMG cell fractionation/reconstitution assay. (A) SMG cell fractionation/reconstitution assay schematic. Unfractionated SMG mesenchyme amenable to cell immunodepletion was generated by microdissection of the mesenchyme from the epithelium followed by enzymatic dissociation of the mesenchyme to single cells and re-aggregation of the isolated mesenchymal cell population. Re-aggregated mesenchyme was then reconstituted with an intact microdissected E13 epithelial rudiment. For endothelial cell depletion, CD31+ endothelial cells were immunodepleted from fully dissociated mesenchyme cells using MACS with CD31 microbeads prior to re-aggregation of the dissociated mesenchyme and reconstitution with an intact epithelium. For endothelial cell supplementation, endothelial-depleted mesenchymal cells were mixed with MACS-isolated endothelial cells prior to re-aggregation of the mesenchyme and reconstitution with an intact epithelium. The reconstituted glands were cultured ex vivo for 48 h post-reconstitution. (B) Confocal images (maximum projection images) consistently showed a change in the epithelial patterning (no marker, black) with a mesenchymal marker (PDGFRβ in red) defining the mesenchymal shape. CD31+ vasculature (cyan) was present in unfractionated, but not in endothelial-depleted mesenchyme. E, endbud. (C,D) Epithelial area (C) (n=number of reconstituted glands) and endbud size (D) (n=number of endbuds from reconstituted glands shown in C) were enlarged in the absence of CD31+ endothelial cells. Black and white bars indicate unfractionated and endothelial-depleted glands, respectively. (E) Supplementation with E16 SMG-derived endothelial cells was performed with endothelial-depleted mesenchyme and epithelial recombination. Reconstituted glands were cultured for 48 h. Boxed areas are enlarged below. (F-H) Endothelial supplementation promoted epithelial branching (F) (n=number of reconstituted glands), smaller bud size (G) (n=number of endbuds in two reconstituted glands) and thinner ducts (H) (n=number of ducts in two reconstituted glands). (I) Increased vessel area was confirmed by CD31 staining in ICCs (n=number of reconstituted glands). In F-I, white and gray bars indicate endothelial-depleted (EC-depleted in E) and endothelial-supplemented (EC-supplemented in E), respectively. Data are mean±s.e.m. Student's t- test (one-tailed) was performed for statistical analysis (*P<0.05 ***P<0.001).
To assess the role of endothelial cells in epithelial patterning in the SMG cell fractionation/reconstitution assay, we compared the ability of unfractionated mesenchyme with that of CD31+ endothelial cell-depleted gland mesenchyme to support epithelial branching morphogenesis. ICC confirmed that CD31+ vasculature was absent from CD31+ cell-depleted, reconstituted glands (Fig. 3B). Depletion of endothelial cells and loss of vasculature development lead to significant changes in epithelial morphology 48 h post-reconstitution, with larger endbuds and a considerable increase in the separation of the endbuds relative to unfractionated glands (Fig. 3B). The differences in epithelial morphology between the two conditions were quantified. In endothelial cell-depleted glands, epithelial area (Fig. 3C) and endbud size (Fig. 3D) were increased. To determine whether addition of endothelial cells back into the culture could rescue normal epithelial pattern formation in CD31+ cell-depleted glands, endothelial cell supplementation was performed. Indeed, epithelial branching was promoted by endothelial supplementation (Fig. 3E). Quantitative analysis showed that supplementation promoted more endbuds (Fig. 3F), smaller endbuds (Fig. 3G) and thinner ducts (Fig. 3H). ICC to detect CD31 confirmed partial re-vascularization in the reconstituted glands (Fig. 3I). Together, these data indicate that endothelial cells and vascular development promote branching morphogenesis and normal epithelial patterning during early SMG development and corroborate data obtained using VEGFR2 inhibition/knockdown.
VEGFR signaling and vasculature of SMGs regulate epithelial progenitor cell differentiation
Because depletion of CD31+ endothelial cells impacted the size of developing ducts and the number of endbuds, we wondered whether endothelial cells impact the differentiation of salivary gland epithelial cells within the endbuds. We examined the productal marker K19 together with Kit, a tyrosine kinase expressed by proacinar cells in developing salivary glands (Nelson et al., 2013; Lombaert et al., 2013; Matsumoto et al., 2016). In whole organ explant cultures, VEGFR signaling and vasculature development were manipulated using the pharmacological VEGFR2 inhibitors ZM 323881 and SU 5416. In vehicle-treated E12.5 explants, Kit+ cells and K19+ cells were present in endbuds and ducts, respectively (Fig. 4A). In both ZM 323881- and SU 5416-treated glands, the epithelial pattern was perturbed with apparent longer, wider K19+ developing ducts and expansion of the K19+ productal cells farther from the main duct into the developing buds than in vehicle control glands (Fig. 4A). Quantitative analyses confirmed that gland area positive for Kit decreased whereas the gland area positive for K19 increased in treated glands relative to vehicle-treated controls (Fig. 4B). Analysis of gland areas positive for Kit and K19 in the SMG cell fractionation/reconstitution assay corroborated the inhibitor data in whole gland explant cultures (Fig. 4C). Similarly, increased cells expressing the ductal marker K7 were detected in endbuds both after VEGFR2 inhibition in whole gland explant cultures and after CD31+ endothelial depletion in the SMG cell fractionation/reconstitution assay, which was rescued by CD31+ cell supplementation in the latter assay (Fig. S2). Taken together, these data indicate that VEGFR2-dependent/CD31+ vascular endothelial cells promote the proacinar progenitor cell phenotype in a coordinated manner with branching morphogenesis, and their absence leads to aberrant ductalization of the endbuds.
Fig. 4.
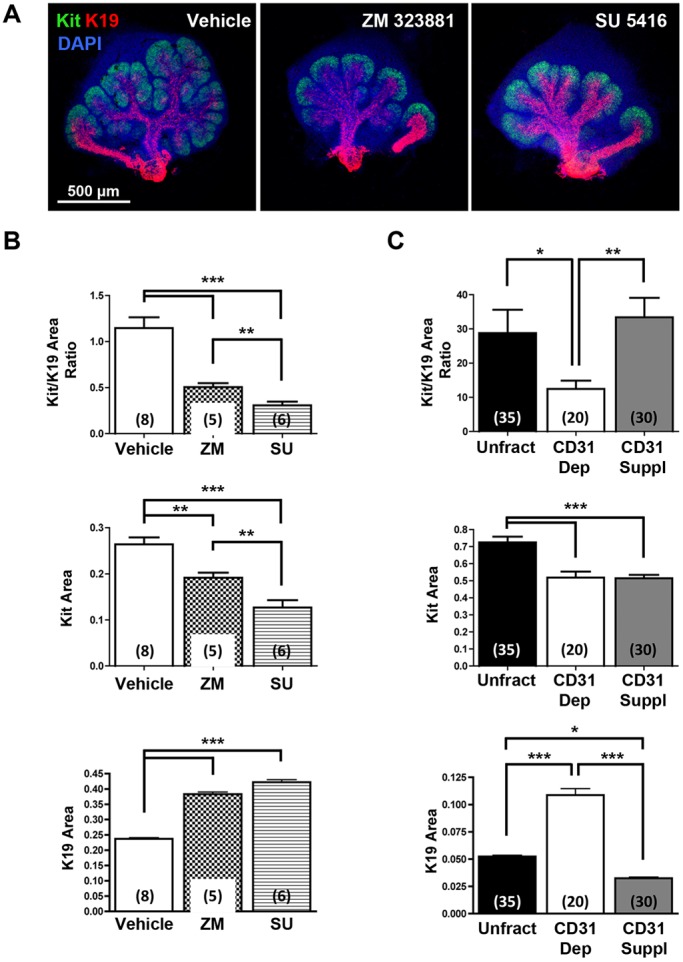
VEGFR signaling and vasculature development regulate SMG epithelial progenitor cell differentiation. (A) ICC and confocal microscopy show that pharmacological inhibition of VEGFR2 with ZM 323881 (20 µM) or SU 5416 (5 µM) expands the productal K19+ (red) cell population relative to the Kit+ (green) cell population in E12.5 SMGs organ explants grown for 48 h versus control glands. Nuclei were stained with DAPI (blue). (B) Maximum projection intensity images of the glands shown in A were used to quantify areas positive for Kit or K19 within the submandibular epithelium only. Oral epithelium at the base of the main duct was avoided to measure K19 expression area. The Kit+/K19+ ratio was decreased with both inhibitors, and Kit expression area was significantly decreased whereas K19 expression area was significantly increased. (C) In the SMG cell fractionation/reconstitution assay, similar CD31-dependent changes in SMG progenitor cells were also observed, with a marked increase in the K19+ productal cell population. Reconstituted glands were grown for 48 h. Total unfractionated dissociated mesenchyme (Unfract), endothelial depleted (CD31 Dep), endothelial supplementation after endothelial depletion (CD31 Suppl). ICC and confocal microscopy (single section of the middle of endbud) was performed to quantify Kit+ and K19+ expression areas (n=number of endbuds). Number of experiments: 7 unfractionated, 4 CD31-depleted and 6 CD31-supplemented. Endothelial cell immunodepletion significantly decreased the Kit+/K19+ ratio, which was partially rescued with endothelial supplementation and re-vascularization. Data are mean±s.e.m. Student's t-test was performed for statistical analysis (*P<0.05, **P<0.01, ***P<0.001).
CD31+ endothelial cells influence soluble factors that regulate SMG epithelial patterning and differentiation
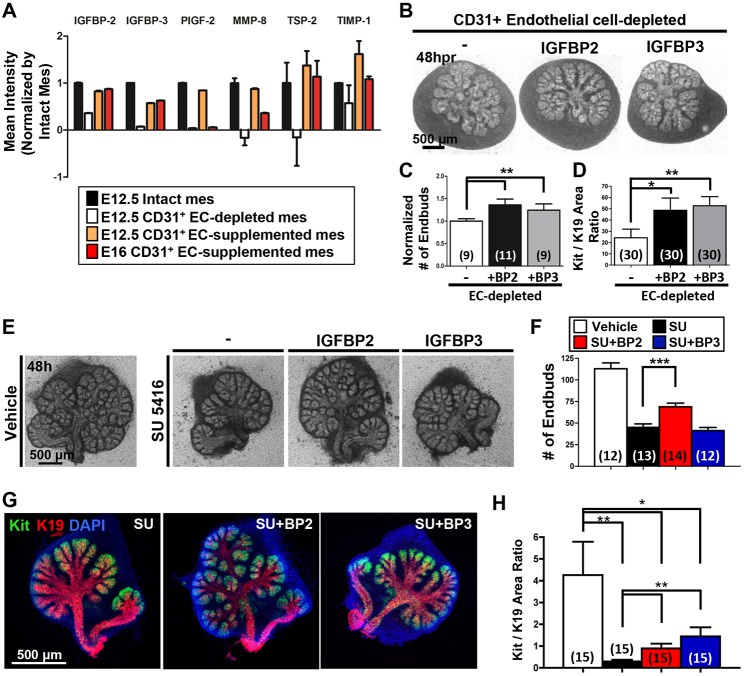
Angiocrine factors are juxtacrine- and paracrine-acting factors produced by endothelial cells that influence parenchymal epithelial cell differentiation, homeostasis and regeneration (Rafii et al., 2016). To screen for endothelial cell-dependent soluble factors that might influence the developing SMG epithelium, conditioned media from cultured endothelial cell-depleted mesenchyme and endothelial supplemented mesenchyme were applied to an antibody-based proteome array containing 53 soluble factors. Intact mesenchyme pieces were used as a positive control. Multiple factors were more abundant in the conditioned media collected from the CD31+ cell-supplemented mesenchyme cell cultures relative to the CD31+ cell-depleted mesenchyme, including IGFBP2, IGFBP3, PIGF-2 (PGF), MMP8, TSP-2 (THBS2 – Mouse Genome Informatics) and TIMP1 (Fig. 5A).
Fig. 5.
Vascular endothelial cells influence soluble factors that regulate SMG epithelial patterning and differentiation. (A) Conditioned media collected from intact (black bar) or recombined mesenchyme, including endothelial depletion (white), E12.5 (orange) and E16 (red) CD31+ endothelial supplementation was analyzed with an angiogenesis proteome array. Select soluble factors abundance of which in the conditioned medias changed in response to CD31+ cell condition are shown, normalized to intact mesenchyme. Full normalized data for the array are available in Table S2. (B) Brightfield images of SMG cell glands reconstituted after CD31+ endothelial cell depletion cultured for 48 h without or with addition of recombinant IGFBP2 or IGFBP3 (1 μg/ml). (C,D) Quantitative analyses demonstrate that both IGFBP2 and IGFBP3 supplementation increased epithelial branching (C) (n=number of reconstituted glands) and the Kit+/K19+ area ratio (D) (n=total number of endbuds from six reconstituted glands). (E) Brightfield images of E13 SMG explants cultured ex vivo for 48 h in the absence or presence of 5 µM SU 5416 (SU), either with or without addition of recombinant IGFBP2 (BP2; 2 μg/ml) or IGFBP3 (BP3; 5 μg/ml). (F) Quantitative analysis of VEGFR2-inhibited glands shows disrupted epithelial patterning expressed as endbud numbers (three experiments). (G) ICC and confocal images representative of the partial rescue of Kit (green) relative to K19 (red) expression areas in the IGFBP2- and IGFBP3-supplemented, SU 5416-treated glands. (H) The Kit+/K19+ ratio was partially rescued with exogenous IGFBPs (n=total number of endbuds from three experiments). Data are mean±s.e.m. Student's t-test was performed for statistical analysis (*P<0.05, **P<0.01, ***P<0.001).
As IGFBP2 and IGFBP3 gave robust signals on the proteome array and have previously been identified as angiocrine factors (Remédio et al., 2012; Minuzzo et al., 2015; Rafii et al., 2016), we tested whether these factors can rescue epithelial patterning upon vasculature disruption using recombinant IGFBP2 or IGFBP3 added to SMG culture assays. In the SMG cell fractionation/reconstitution assay, addition of recombinant IGFBP2 or IGFBP3 promoted both epithelial patterning (Fig. 5B,C) and differentiation (Fig. 5D) relative to CD31+ cell depleted samples. We also tested the effects of recombinant IGFBP2 or IGFBP3 in intact gland explant assays, in which IGFBP2, but not IGFBP3, showed a partial rescue of disrupted epithelial branching in the SU 5416-inhibited glands (Fig. 5E,F). Similar to the SMG cell fractionation/reconstitution assay, ICC of Kit+ and K19+ cells in the endbuds of SU 5416-treated glands revealed that the reduction of the Kit+/K19+ ratio with VEGFR2 inhibition was partially rescued by exogenous IGFBP2 or IGFBP3 (Fig. 5G,H). We performed additional arrays to interrogate the cell type that makes IGFBP2 and IGFBP3. We found that IGFBP2 and IGFBP3 are made primarily by the mesenchyme relative to the epithelium. Interestingly, we noted that both the endothelial cells and the non-endothelial mesenchyme make these proteins, with more robust production by the non-endothelial mesenchyme (Fig. S3). Together, these data indicate that vascular endothelial cells promote epithelial patterning and differentiation at least in part through effects on soluble factors, such as IGFBPs.
DISCUSSION
Signaling from mesenchymal cell subpopulations is required for elaboration of epithelial tissue structure during development. Using an SMG cell fractionation/reconstitution assay that allowed us to deplete CD31+ endothelial cells from the mesenchyme of reconstituted embryonic submandibular salivary glands, we demonstrate for the first time that CD31+ endothelial cells influence epithelial patterning during salivary gland development in a perfusion-independent manner. The endothelial cells function through VEGFR2-dependent signaling to moderate ductalization of the buds and support the Kit+ bipotent progenitor cell population. We observed that inhibition of this vasculature-dependent support of progenitor cell expansion caused altered epithelial patterning through reduction of the Kit+ cell population that was accompanied by increased localization of K19+ and K7+ ductal cells in the endbuds of the gland. This would be predicted to prevent normal elaboration of the secretory compartment of the gland with accumulation of elongated and dilated ducts. In fact, similar pathologies are associated with diseases that result in xerostomia, such as Sjögren's syndrome and irradiation damage as a result of treatment of head and neck cancers, suggesting that a continued interaction of the vasculature and the epithelium may be a component of homeostasis or a response to injury in adult glands. Consistent with our findings in development, several studies have suggested that the vasculature is damaged in salivary gland pathologies and that gland regeneration is enhanced by restoration of the vasculature (Cotrim et al., 2007; Zhang et al., 2014; An et al., 2015).
Endothelial cells instruct organ development in other organs, such as liver (Matsumoto et al., 2001), pancreas (Lammert et al., 2001; Cleaver and Dor, 2012) and lung (Lazarus et al., 2011), through perfusion-independent mechanisms prior to the formation of functional vasculature. In the liver, VEGFR2-dependent signaling is required for early hepatocyte morphogenesis and differentiation (Matsumoto et al., 2001) and cell fate specification of the central zone of the liver (Rocha et al., 2015). Similarly, early dorsal pancreas budding and insulin production by β cells requires endothelial cell signaling (Lammert et al., 2001; Yoshitomi and Zaret, 2004). However, endothelial cell signaling can also restrict cell fate. VEGFR2-dependent endothelial cell signaling restricts branching morphogenesis and restricts acinar differentiation later in development of the pancreas (Magenheim et al., 2011) and differentiation in liver (Ramasamy et al., 2015; Rocha et al., 2015). Determining which cells are directly impacted by endothelial-dependent signaling in developing salivary glands and whether cell fate choices are directly regulated by VEGFR2 signaling will require investigation by cell lineage tracking.
Although specific angiocrine factors produced by endothelial cells can regulate cell specification, such as R-spondin 3 in specification in the liver (Rocha et al., 2015), endothelial cell signaling can also activate signaling by other mesenchyme cells to regulate progenitor cells indirectly in the epithelium. For example, endothelial-produced factors are known to recruit and support stromal pericytes, which are important for organogenesis. Additionally, endothelial progenitor cell-produced platelet-derived growth factor (PDGF)-BB was identified in recruitment and sustaining the function of the PDGFRβ+ mesenchymal stem cells (MSCs) after therapeutic engraftment during MSC cell therapy (Lin et al., 2014). Although IGFBP2 is known to be one of many angiocrine factors produced by endothelial cells in an organ-specific manner (Mouhieddine et al., 1996; Besnard et al., 2001; Bridgewater and Matsell, 2003; Huynh et al., 2011; Perri et al., 2014), in the salivary gland, IGFBP2 and IGFBP 3 appear to be produced primarily by CD31− mesenchyme cells.
Our data are consistent with a model in which endothelial cells stimulate IGFBP2 and IGFBP3 production by neighboring mesenchymal cells, and IGFBP2 and IGFBP3 produced by mesenchymal cells modulate epithelial signaling in developing salivary glands to favor Kit+ cells over K19+ cells in the developing endbuds. IGFBP2 in circulation can stimulate or inhibit the growth-promoting effects of the IGFs (Binkert et al., 1992; Bourner et al., 1992). However, IGFBP2 has an RGD site that can activate integrin signaling independently of IGF receptors (Feng et al., 2015), and modulates the expansion and survival of hematopoietic stem cells (Huynh et al., 2011). Whether IGFBP2 and IGFBP3 function in an IGF-dependent or -independent manner in an in vivo context during salivary gland development and how they regulate epithelial progenitor cells remains to be elucidated. Although our data do not delineate a specific mechanism for endothelial cell signaling in controlling SMG cell fate, they are consistent with data from other embryonic organs indicating that the developing vasculature supports expansion of the primitive progenitor populations, which in turn recruits developing vasculature to the emerging epithelium.
A similar premature ductal differentiation was recently reported during early development of the salivary gland that is Wnt dependent (Matsumoto et al., 2016). Similar to our data demonstrating premature ductal formation and expansion of the ductal area of the gland with compromised VEGFR2/CD31+ endothelial cell function, Matsumoto et al. observed that excess Wnt signaling can induce premature ductal formation and expansion of the ductal compartment of the gland. In early SMG development, mesenchymal Wnt signaling antagonized FGF-mediated proacinar cell differentiation to expand the Kit+ distal progenitor cell population. As mesenchymal Wnt signaling declines at E15 concomitant with the onset of cytodifferentiation, they show that the expanded bipotent Kit+ progenitor population then creates the secretory proacinar and emerging secondary duct structures required for development of the mature branched organ. Wnt signaling can also suppress Kit expression at the RNA level through Myb, demonstrating that Wnt activity must be tightly balanced to allow proper progenitor expansion followed by spatially restricted differentiation.
In our studies, the VEGFR2/CD31+ endothelial cell population might promote epithelial patterning and expansion of the Kit+ endbud epithelial progenitor cell population in part by modulating Wnt signaling from the mesenchyme. Loss of VEGFR2/CD31+ endothelial cell function would then be expected to increase canonical Wnt signaling to the epithelium, prevent proacinar differentiation, downregulate Kit expression and accelerate ductal formation. Wnt signaling is regulated by balancing activation of Wnt receptors by ligands at many levels, and several reports indicate that the Wnt family has complex roles in SMG development (Haara et al., 2011; Musselmann et al., 2011; Patel et al., 2011; Knosp et al., 2015; Maimets et al., 2016; Matsumoto et al., 2016). Thus, VEGFR2/CD31+ endothelial cells could act in many ways to modulate mesenchymal Wnt signaling to the epithelium, and might confer this regulation in a spatially restricted manner during branching morphogenesis. Thus, the mesenchyme, including the PDGFRβ+ fibroblasts, vasculature and innervation (Knox et al., 2010), develops in a coordinated manner with the epithelium, and this co-patterning is likely to confer essential spatial restriction to the reciprocal epithelial-mesenchymal signaling pathways that control organ development.
Diverse, reciprocal signaling pathways between the epithelium and mesenchyme are known to orchestrate SMG development, and the mechanisms by which these signals interact and are decoded by the progenitor cell populations to coordinate morphogenesis and differentiation are of great interest. Reciprocal interactions between the developing vasculature and epithelium are likely to be multifactorial, similar to the contribution of innervation to gland development (Knox et al., 2010; Nedvetsky et al., 2014; Knosp et al., 2015). The fact that our assays with IGFBP2 and IGFBP3 show modest effects on SMG morphogenesis and differentiation is consistent with the concept that there are likely to be numerous endothelial-produced factors that impact parenchymal development. As addition of excess recombinant proteins cannot recapitulate spatial restriction of signaling factors, additional studies will be needed to confirm a role for these factors in SMG in vivo development and elucidate the mechanism by which they control epithelial patterning of differentiation.
Importantly, our SMG cell fractionation/gland reconstitution assay provides a platform for more extensive screens for putative angiocrine factors (soluble or cell-cell mediated) as well as for mechanistic studies that cannot be performed with classical organ recombination experiments using intact mesenchyme. Additionally, the SMG cell fractionation/reconstitution assay enables screening for synthetic or interacting pathways, for example with CD31+ cell depletion/cell supplementation in the presence of inhibitors or genetic knockdown of other pathways known to control gland development. Recent work in lung branching morphogenesis and differentiation revealed that mesenchymal glucocorticoid receptor and STAT3 act in parallel pathways to specify lung alveolar differentiation, such that neither is absolutely required but either pathway can promote epithelial differentiation (Laresgoiti et al., 2016). Our gland reconstitution assay will facilitate future investigation of these types of parallel, or similarly synergistic, activities in epithelial cell fate choices. The assay is also amenable for testing the interaction between mesenchymal cell subsets, such as vascular cells and nerves, which are known to have reciprocal patterning effects in other systems.
Elucidation of the molecular mechanisms through which the vasculature directly instructs epithelial patterning and differentiation, as well pathways through which it affects other mesenchymal cell types to indirectly affect the parenchymal epithelium, will ultimately enable therapeutic manipulations. Similarities and differences in the angiocrine control of organ development and regeneration in diverse organs can be exploited to enhance both general and organ-specific regenerative medicine approaches. Importantly, the angiocrine mechanisms that control organ homeostasis and response to injury are currently being characterized and exploited for therapeutic interventions to improve organ regeneration (Rafii et al., 2016). Ongoing characterization of angiocrine control of salivary glands might similarly improve therapeutic options for restoration of salivary gland function in disease pathologies.
MATERIALS AND METHODS
Ex vivo SMG organ explant culture with pharmacological and siRNA manipulation
Salivary gland dissection and ex vivo culture were performed according to previously established methods (Daley et al., 2009) and protocols approved by the University at Albany Institutional Animal Care and Use Committee. Embryos were harvested from timed pregnant CD-1 female mice (Charles River) at the embryonic stages indicated for each figure (with day of plug discovery designated as E0). SMGs were removed from the embryos under a dissecting microscope. The SMGs were placed on a polycarbonate filter with 0.1 µm pores (Nuclepore, GE Healthcare Life Sciences) in a glass-bottomed, single-microwell dish (MatTek) and cultured ex vivo in complete DMEM/F12 medium [1:1 DMEM/Ham's F12 lacking Phenol Red (Invitrogen) supplemented with 50 μg/ml transferrin, 150 μg/ml L-ascorbic acid, 100 U/ml penicillin and 100 μg/ml streptomycin].
Pharmacological and siRNA treatments were performed in whole explants. For pharmacological VEGFR2 inhibition, ZM 323881 (10 μM and 20 μM in DMSO; Tocris) (Whittles et al., 2002) and SU 5416 (5 μM in DMSO; Tocris) (Fong et al., 1999) were supplemented during the explant culture. Vehicle-treated controls contained the same volume of DMSO. siRNA genetic manipulation was performed as previously described (Daley et al., 2009). VEGFR2 siRNAs (Silencer Select s68715/s68716, Thermo Fisher Scientific) and negative control non-targeting siRNA (Silencer Select Negative Control #2 siRNA) were transfected using RNAiFECT (Qiagen), both at a final concentration of 400 nM.
For mesenchyme-free, epithelial rudiment cultures, epithelial rudiments were physically separated from mesenchyme following a dispase digestion, as previously described (Sequeira et al., 2013), and were cultured ex vivo for 48 h in growth factor-reduced Matrigel (Corning, 356231). FGF7 (200 ng/ml; Peprotech, 100-19) and epidermal growth factor (20 ng/ml; Peprotech, AF-100-15) were additionally supplemented into the complete DMEM/F12 medium.
Primary mesenchyme cell preparation
To prepare unfractionated E12/E13 mesenchyme, dispase digestion of E12/E13 whole SMGs was performed followed by manual separation of the mesenchyme from the epithelium, as previously described (Sequeira et al., 2013). To prepare E12/E13 primary mesenchyme cells, mesenchyme pieces were placed into a MatTek dish, mixed with 200 μl 0.3× collagenase/hyaluronidase (7912, STEMCELL Technologies, Vancouver, Canada), and incubated for 5 min at 37°C. The digested mesenchyme was mixed with 20 μl fetal bovine serum (FBS; Life Technologies), placed in a microcentrifuge tube, and homogenized by trituration 30-40 times to yield single cells.
To prepare primary E16 mesenchyme cells, whole excised E16 SMGs were microdissected with forceps in a 35 mm dish and serially treated with 1× collagenase/hyaluronidase (STEMCELL Technologies) for 10 min and 0.8 U/ml dispase (Life Technologies) for 10-15 min, followed by incubation for 15 min at 37°C. The cell preparation was mixed with 200 μl FBS, placed into a 15 ml conical tube, and homogenized by trituration at least 40 times. To enrich for mesenchymal cells, the epithelial fragments were separated by gravity sedimentation for 5 min. The mesenchyme-enriched gravity supernatant was filtered through a 70 μm pore size cell strainer (Falcon) to enrich for single cells, and washed twice with PBS with 10% FBS/2 mM ethylenediaminetetraacetic acid (EDTA; Sigma-Aldrich) buffer. The enriched primary mesenchymal cells were centrifuged at 450 g for 5 min and re-suspended in 1× PBS buffer with 10% FBS and 2 mM EDTA.
Depletion of CD31+ cells from mesenchyme and collection of CD31+ cells by magnetic-activated cell sorting (MACS)
For CD31+ endothelial cell depletion from E12.5 primary mesenchyme, MACS with mouse CD31 microbeads (130-097-418, Miltenyi Biotech, Bergisch Gladbach, Germany) was performed according to the manufacturer's protocols. Briefly, enriched primary mesenchyme cells were suspended in 99 μl 1× PBS buffer with 10% FBS and mixed with 1 μl mouse CD31 microbeads, followed by incubation at 4°C for 15 min. After washing with 500 μl PBS buffer with 10% FBS, the microbead-incubated cells were centrifuged at 450 g for 5 min and resuspended with 200 μl PBS with 10% FBS for immunomagnetic separation. To collect CD31− mesenchyme, cells were applied to a magnetic column and the flow-through was collected, followed by three washes with 1× PBS with 10% FBS, which were included with the flow-through. The remaining CD31+ cells were collected by centrifugation at 450 g for 5 min and then resuspended in DMEM:F12 medium.
For isolation of CD31+ endothelial cells from E16 SMG, primary E16 mesenchyme cells were prepared as described above, and negative selection was performed to remove contaminating EpCAM+ epithelial cells. Primary mesenchyme cells were resuspended in 90 μl PBS with 10% FBS/2 mM EDTA buffer (wash buffer), mixed with 10 μl mouse EpCAM microbeads (130-105-958, Miltenyi Biotech), followed by incubation at 4°C for 15 min. After washing with 1 ml wash buffer, the microbead-incubated cells were centrifuged at 450 g for 5 min and resuspended in 500 μl wash buffer for magnetic removal of the EpCAM+ cells. The resulting mesenchymal cells were collected and centrifuged at 450 g for 5 min, resuspended in 90 μl wash buffer, mixed with 10 μl mouse CD31 microbeads, and incubated at 4°C for 15 min. Positive selection was performed to collect the CD31+ endothelial cells.
SMG cell fractionation/reconstitution assay
Dissociated whole unfractionated mesenchyme cells, CD31− mesenchyme cells, or CD31− mesenchyme cells supplemented with purified CD31+ endothelial cells were cultured overnight. For overnight cultures, the mesenchyme cell fractions were collected and centrifuged at 450 g for 5 min. Cells were then suspended in 50 μl fresh DMEM:F12 medium containing only antibiotics (100 U/ml penicillin and 100 μg/ml streptomycin) and placed into a V-bottom 96-well plate to encourage mesenchymal tissue reaggregation overnight at 37°C. The contracted mesenchymal tissue was placed on a Nuclepore filter in a MatTek dish with a fresh intact E13 SMG epithelium placed on top of the aggregated mesenchyme to reconstitute the gland. The reconstituted glands were grown with complete DMEM/F12 organ explant medium for 48 h. Each reconstituted gland was considered to be a single experiment.
Immunocytochemistry and confocal microscopy
Immunocytochemistry (ICC) was performed as previously described (Daley et al., 2009; Peters et al., 2015). Briefly, SMG organ explants or SMG cell fractionation/reconstitutions glands were fixed with 4% paraformaldehyde (w/v) (Electron Microscopy Sciences) containing 5% sucrose (w/v) for 20 min at room temperature (RT) and permeablized with 0.1% Triton X-100 (Sigma-Aldrich) or fixed/permeablized in 100% cold MeOH at −20°C. Blocking was performed with 20% donkey serum in 1× PBS with 0.5% Tween-20 for 1 h at RT. Primary and secondary antibodies were diluted in 1× PBS with 0.5% Tween-20 and incubated overnight at 4°C or for 1.5 h at room temperature, and then washed three times in PBS with 0.5% Tween-20. Antibodies and the dilutions used in this study are shown in Table S1. DAPI (Life Technologies) was used for nuclear staining. The tissues were mounted with Fluoro-Gel (Electron Microscopy Sciences) and imaged with a Zeiss 510 or 710 confocal microscope (Carl Zeiss).
Angiogenesis proteomic array
For analysis of soluble factors, microdissected mesenchyme pieces, endothelial-depleted mesenchyme or endothelial-supplemented mesenchyme were cultured for 48 h to collect conditioned media. Conditioned media was applied to a mouse angiogenesis proteome array (Proteome Profiler Mouse Angiogenesis Antibody Array, #ARY015, R&D systems) and samples were processed according to the manufacturer's protocol. For analysis of factor production, cell lysates were prepared as previously described (Larsen et al., 2003). Briefly, gland tissue or cells were collected in a microfuge tube and solubilized with completed RIPA buffer (Thermo Fisher Scientific) containing complete mini, EDTA-free protease inhibitor cocktail (Roche), followed by sonication and centrifugation at 4°C to collect the soluble fraction. The resulting cell lysates of fresh uncultured microdissected E12.5 epithelium, mesenchyme, E12/E13 CD31− depleted mesenchyme, or immunopurified CD31+ endothelial cells were applied to the angiogenesis proteome array. Image processing to measure spot intensities from films was performed using ImageJ software (National Institute of Health). Duplicate spots within each membrane were used to calculate average intensities. The normalized spot intensity was calculated as (individual spot intensity−negative spot intensity)/positive spot intensity. Additional normalization procedures are explained in each figure legend when relevant.
Image data analysis for epithelial patterning and differentiation
To evaluate epithelial structure, measurements of epithelium size, endbud/duct area, endbud size, or duct width/length were collected from brightfield images of SMG organ explants or SMG cell fractionation/reconstitution glands. Area and size were measured as the pixel area value of manually designated areas, and width and length were collected from pixel length value using ImageJ. Percentages of Kit+ and K19+areas were measured from ICC confocal images. First, total epithelium or individual endbuds were manually designated using the polygon selection tool, as indicated in figure legends. Individual Kit+ and K19+ areas were calculated and normalized to total designated area; each marker-positive pixel area value (measured using a threshold option) was divided by the total pixel area of the designated area. To analyze apoptotic endothelial cells (double-positive CD31+ and cleaved caspase 3+ cells), CD31+ areas were manually designated and the cleaved caspase 3+ area was measured using a threshold tool. Vessel area (the area of the segmented CD31+ vessel area/total area) was measured using AngioTool (Zudaire et al., 2011) (https://ccrod.cancer.gov/confluence/display/ROB2/Home). Vessel intensity was evaluated by CD31 staining of confocal images using ImageJ. Graphs were prepared using Prism 5 (GraphPad). Statistics were calculated as described in each figure using Prism 5 and Microsoft Excel.
Acknowledgements
The authors thank Drs Guohao Dai, Paolo Forni and Prashanth Rangan for helpful comments.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
H.R.K., M.L., and D.A.N. conceived the experiments. H.R.K., K.A.D., and J.M.M. performed the experiments. H.R.K., M.L., and D.A.N. interpreted the data. H.R.K., M.L., and D.A.N. wrote and revised the manuscript.
Funding
This work was supported by the National Institutes of Health (RO1 DE022467 to M.L., C06 RR015464) and the University at Albany (SUNY). Deposited in PMC for release after 12 months.
Supplementary information
Supplementary information available online at http://dev.biologists.org/lookup/doi/10.1242/dev.142497.supplemental
References
- An H. Y., Shin H. S., Choi J. S., Kim H. J., Lim J. Y. and Kim Y. M. (2015). Adipose mesenchymal stem cell secretome modulated in hypoxia for remodeling of radiation-induced salivary gland damage. PLoS ONE 10, e0141862 10.1371/journal.pone.0141862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azizoglu D. B. and Cleaver O. (2016). Blood vessel crosstalk during organogenesis-focus on pancreas and endothelial cells. Wiley Interdiscip. Rev. Dev. Biol. 5, 598-617. 10.1002/wdev.240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Besnard V., Corroyer S., Trugnan G., Chadelat K., Nabeyrat E., Cazals V. and Clement A. (2001). Distinct patterns of insulin-like growth factor binding protein (IGFBP)-2 and IGFBP-3 expression in oxidant exposed lung epithelial cells. Biochim. Biophys. Acta 1538, 47-58. 10.1016/S0167-4889(00)00136-1 [DOI] [PubMed] [Google Scholar]
- Binkert C., Margot J. B., Landwehr J., Heinrich G. and Schwander J. (1992). Structure of the human insulin-like growth factor binding protein-2 gene. Mol. Endocrinol. 6, 826-836. [DOI] [PubMed] [Google Scholar]
- Bourner M. J., Busby W. H., Siegel N. R., Krivi G. G., McCusker R. H. and Clemmons D. R. (1992). Cloning and sequence determination of bovine insulin-like growth factor binding protein-2 (IGFBP-2) - comparison of its structural and functional properties with IGFBP-1. J. Cell. Biochem. 48, 215-226. 10.1002/jcb.240480212 [DOI] [PubMed] [Google Scholar]
- Bridgewater D. J. and Matsell D. G. (2003). Insulin-like growth factor binding protein-2 modulates podocyte mitogenesis. Pediatr. Nephrol. 18, 1109-1115. 10.1007/s00467-003-1242-x [DOI] [PubMed] [Google Scholar]
- Chavakis E. and Dimmeler S. (2002). Regulation of endothelial cell survival and apoptosis during angiogenesis. Arterioscler. Thromb. Vasc. Biol. 22, 887-893. 10.1161/01.ATV.0000017728.55907.A9 [DOI] [PubMed] [Google Scholar]
- Cleaver O. and Dor Y. (2012). Vascular instruction of pancreas development. Development 139, 2833-2843. 10.1242/dev.065953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cotrim A. P., Sowers A., Mitchell J. B. and Baum B. J. (2007). Prevention of irradiation-induced salivary hypofunction by microvessel protection in mouse salivary glands. Mol. Ther. 15, 2101-2106. 10.1038/sj.mt.6300296 [DOI] [PubMed] [Google Scholar]
- Daley W. P., Gulfo K. M., Sequeira S. J. and Larsen M. (2009). Identification of a mechanochemical checkpoint and negative feedback loop regulating branching morphogenesis. Dev. Biol. 336, 169-182. 10.1016/j.ydbio.2009.09.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng N., Zhang Z., Wang Z., Zheng H., Qu F., He X. and Wang C. (2015). Insulin-like growth factor binding protein-2 promotes adhesion of endothelial progenitor cells to endothelial cells via integrin alpha 5 beta 1. J. Mol. Neurosci. 57, 426-434. 10.1007/s12031-015-0589-3 [DOI] [PubMed] [Google Scholar]
- Fong T. A., Shawver L. K., Sun L., Tang C., App H., Powell T. J., Kim Y. H., Schreck R., Wang X., Risau W. et al. (1999). SU5416 is a potent and selective inhibitor of the vascular endothelial growth factor receptor (Flk-1/KDR) that inhibits tyrosine kinase catalysis, tumor vascularization, and growth of multiple tumor types. Cancer Res. 59, 99-106. [PubMed] [Google Scholar]
- Haara O., Fujimori S., Schmidt-Ullrich R., Hartmann C., Thesleff I. and Mikkola M. L. (2011). Ectodysplasin and Wnt pathways are required for salivary gland branching morphogenesis. Development 138, 2681-2691. 10.1242/dev.057711 [DOI] [PubMed] [Google Scholar]
- Hagiwara M., Peng F. and Ho C.-M. (2015). In vitro reconstruction of branched tubular structures from lung epithelial cells in high cell concentration gradient environment. Sci. Rep. 5, 8054 10.1038/srep08054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman M. P., Kidder B. L., Steinberg Z. L., Lakhani S., HO S., Kleinman H. K. and Larsen M. (2002). Gene expression profiles of mouse submandibular gland development: FGFR1 regulates branching morphogenesis in vitro through BMP- and FGF-dependent mechanisms. Development 129, 5767-5778. 10.1242/dev.00172 [DOI] [PubMed] [Google Scholar]
- Huynh H., Zheng J., Umikawa M., Zhang C., Silvany R., Iizuka S., Holzenberger M., Zhang W. and Zhang C. C. (2011). IGF binding protein 2 supports the survival and cycling of hematopoietic stem cells. Blood 118, 3236-3243. 10.1182/blood-2011-01-331876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingthorsson S., Sigurdsson V., Fridriksdottir A. J. R., Jonasson J. G., Kjartansson J., Magnusson M. K. and Gudjonsson T. (2010). Endothelial cells stimulate growth of normal and cancerous breast epithelial cells in 3D culture. BMC Res. Notes 3, 184 10.1186/1756-0500-3-184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaramillo M., Mathew S., Mamiya H., Goh S. K. and Banerjee I. (2015). Endothelial cells mediate islet-specific maturation of human embryonic stem cell-derived pancreatic progenitor cells. Tissue Eng. Part A 21, 14-25. 10.1089/ten.tea.2014.0013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kao D.-I., Lacko L. A., Ding B.-S., Huang C., Phung K., Gu G., Rafii S., Stuhlmann H. and Chen S. (2015). Endothelial cells control pancreatic cell fate at defined stages through EGFL7 signaling. Stem Cell Rep. 4, 181-189. 10.1016/j.stemcr.2014.12.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kera H., Yuki S. and Nogawa H. (2014). FGF7 signals are relayed to autocrine EGF family growth factors to induce branching morphogenesis of mouse salivary epithelium. Dev. Dyn. 243, 552-559. 10.1002/dvdy.24097 [DOI] [PubMed] [Google Scholar]
- Knosp W. M., Knox S. M., Lombaert I. M. A., Haddox C. L., Patel V. N. and Hoffman M. P. (2015). Submandibular parasympathetic gangliogenesis requires sprouty-dependent Wnt signals from epithelial progenitors. Dev. Cell 32, 667-677. 10.1016/j.devcel.2015.01.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knox S. M., Lombaert I. M. A., Reed X., Vitale-Cross L., Gutkind J. S. and Hoffman M. P. (2010). Parasympathetic innervation maintains epithelial progenitor cells during salivary organogenesis. Science 329, 1645-1647. 10.1126/science.1192046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knox S. M., Lombaert I. M. A., Haddox C. L., Abrams S. R., Cotrim A., Wilson A. J. and Hoffman M. P. (2013). Parasympathetic stimulation improves epithelial organ regeneration. Nat. Commun. 4, 1494 10.1038/ncomms2493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon H. R. and Larsen M. (2015). The contribution of specific cell subpopulations to submandibular salivary gland branching morphogenesis. Curr. Opin. Genet. Dev. 32, 47-54. 10.1016/j.gde.2015.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lammert E., Cleaver O. and Melton D. (2001). Induction of pancreatic differentiation by signals from blood vessels. Science 294, 564-567. 10.1126/science.1064344 [DOI] [PubMed] [Google Scholar]
- Laresgoiti U., Nikolić M. Z., Rao C., Brady J. L., Richardson R. V., Batchen E. J., Chapman K. E. and Rawlins E. L. (2016). Lung epithelial tip progenitors integrate glucocorticoid- and STAT3-mediated signals to control progeny fate. Development 143, 3686-3699. 10.1242/dev.134023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larsen M., Hoffman M. P., Sakai T., Neibaur J. C., Mitchell J. M. and Yamada K. M. (2003). Role of PI 3-kinase and PIP3 in submandibular gland branching morphogenesis. Dev. Biol. 255, 178-191. 10.1016/S0012-1606(02)00047-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larsen M., Bogdanov P., Sood R., Kwon H. R., Nelson D. A., Duffy C., Peters S. B. and Chittur S. V. (2017). Systems biology: salivary gland development, disease, and regenerative medicine (in Press). In Salivary Gland Development and Regenerative Medicine: Advances in Research and Clinical Approaches to Functional Restoration (ed. Cha S.). Switzerland: Springer; (in press). 10.1007/978-3-319-43513-8 [DOI] [Google Scholar]
- Lazarus A., Del-Moral P. M., Ilovich O., Mishani E., Warburton D. and Keshet E. (2011). A perfusion-independent role of blood vessels in determining branching stereotypy of lung airways. Development 138, 2359-2368. 10.1242/dev.060723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin R.-Z., Moreno-Luna R., Li D., Jaminet S.-C., Greene A. K. and Melero-Martin J. M. (2014). Human endothelial colony-forming cells serve as trophic mediators for mesenchymal stem cell engraftment via paracrine signaling. Proc. Natl. Acad. Sci. USA 111, 10137-10142. 10.1073/pnas.1405388111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lombaert I. M. A., Brunsting J. F., Wierenga P. K., Faber H., Stokman M. A., Kok T., Visser W. H., Kampinga H. H., de Haan G. and Coppes R. P. (2008). Rescue of salivary gland function after stem cell transplantation in irradiated glands. PLoS ONE 3, e2063 10.1371/journal.pone.0002063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lombaert I. M. A., Abrams S. R., Li L., Eswarakumar V. P., Sethi A. J., Witt R. L. and Hoffman M. P. (2013). Combined KIT and FGFR2b signaling regulates epithelial progenitor expansion during organogenesis. Stem Cell Rep. 1, 604-619. 10.1016/j.stemcr.2013.10.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magenheim J., Ilovich O., Lazarus A., Klochendler A., Ziv O., Werman R., Hija A., Cleaver O., Mishani E., Keshet E. et al. (2011). Blood vessels restrain pancreas branching, differentiation and growth. Development 138, 4743-4752. 10.1242/dev.066548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maimets M., Rocchi C., Bron R., Pringle S., Kuipers J., Giepmans B. N. G., Vries R. G. J., Clevers H., de Haan G., van Os R. et al. (2016). Long-term in vitro expansion of salivary gland stem cells driven by Wnt signals. Stem Cell Rep. 6, 150-162. 10.1016/j.stemcr.2015.11.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcelo K. L., Goldie L. C. and Hirschi K. K. (2013). Regulation of endothelial cell differentiation and specification. Circ. Res. 112, 1272-1287. 10.1161/CIRCRESAHA.113.300506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto K., Yoshitomi H., Rossant J. and Zaret K. S. (2001). Liver organogenesis promoted by endothelial cells prior to vascular function. Science 294, 559-563. 10.1126/science.1063889 [DOI] [PubMed] [Google Scholar]
- Matsumoto S., Kurimoto T., Taketo M. M., Fujii S. and Kikuchi A. (2016). The WNT/MYB pathway suppresses KIT expression to control the timing of salivary proacinar differentiation and duct formation. Development 143, 2311-2324. 10.1242/dev.134486 [DOI] [PubMed] [Google Scholar]
- Mattingly A., Finley J. K. and Knox S. M. (2015). Salivary gland development and disease. Wiley Interdiscip. Rev. Dev. Biol. 4, 573-590. 10.1002/wdev.194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minuzzo S., Agnusdei V., Pusceddu I., Pinazza M., Moserle L., Masiero M., Rossi E., Crescenzi M., Hoey T., Ponzoni M. et al. (2015). DLL4 regulates NOTCH signaling and growth of T acute lymphoblastic leukemia cells in NOD/SCID mice. Carcinogenesis 36, 115-121. 10.1093/carcin/bgu223 [DOI] [PubMed] [Google Scholar]
- Mouhieddine O. B., Cazals V., Kuto E., Le Bouc Y. and Clement A. (1996). Glucocorticoid-induced growth arrest of lung alveolar epithelial cells is associated with increased production of insulin-like growth factor binding protein-2. Endocrinology 137, 287-295. 10.1210/endo.137.1.8536625 [DOI] [PubMed] [Google Scholar]
- Musselmann K., Green J. A., Sone K., Hsu J. C., Bothwell I. R., Johnson S. A., Harunaga J. S., Wei Z. and Yamada K. M. (2011). Salivary gland gene expression atlas identifies a new regulator of branching morphogenesis. J. Dent. Res. 90, 1078-1084. 10.1177/0022034511413131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nedvetsky P. I., Emmerson E., Finley J. K., Ettinger A., Cruz-Pacheco N., Prochazka J., Haddox C. L., Northrup E., Hodges C., Mostov K. E. et al. (2014). Parasympathetic innervation regulates tubulogenesis in the developing salivary gland. Dev. Cell 30, 449-462. 10.1016/j.devcel.2014.06.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson D. A. and Larsen M. (2015). Heterotypic control of basement membrane dynamics during branching morphogenesis. Dev. Biol. 401, 103-109. 10.1016/j.ydbio.2014.12.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson D. A., Manhardt C., Kamath V., Sui Y., Santamaria-Pang A., Can A., Bello M., Corwin A., Dinn S. R., Lazare M. et al. (2013). Quantitative single cell analysis of cell population dynamics during submandibular salivary gland development and differentiation. Biol. Open 2, 439-447. 10.1242/bio.20134309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel N., Sharpe P. T. and Miletich I. (2011). Coordination of epithelial branching and salivary gland lumen formation by Wnt and FGF signals. Dev. Biol. 358, 156-167. 10.1016/j.ydbio.2011.07.023 [DOI] [PubMed] [Google Scholar]
- Patel V. N., Lombaert I. M. A., Cowherd S. N., Shworak N. W., Xu Y., Liu J. and Hoffman M. P. (2014). Hs3st3-modified heparan sulfate controls KIT+ progenitor expansion by regulating 3-O-sulfotransferases. Dev. Cell 29, 662-673. 10.1016/j.devcel.2014.04.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perri A. F., Dallard B. E., Baravalle C., Licoff N., Formía N., Ortega H. H., Becú-Villalobos D., Mejia M. E. and Lacau-Mengido I. M. (2014). Cellular proliferation rate and insulin-like growth factor binding protein (IGFBP)-2 and IGFBP-3 and estradiol receptor alpha expression in the mammary gland of dairy heifers naturally infected with gastrointestinal nematodes during development. J. Dairy Sci. 97, 4985-4996. 10.3168/jds.2014-8070 [DOI] [PubMed] [Google Scholar]
- Peters S. B., Nelson D. A., Kwon H. R., Koslow M., DeSantis K. A. and Larsen M. (2015). TGFbeta signaling promotes matrix assembly during mechanosensitive embryonic salivary gland restoration. Matrix. Biol. 43, 109-124. 10.1016/j.matbio.2015.01.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rafii S., Butler J. M. and Ding B.-S. (2016). Angiocrine functions of organ-specific endothelial cells. Nature 529, 316-325. 10.1038/nature17040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramasamy S. K., Kusumbe A. P. and Adams R. H. (2015). Regulation of tissue morphogenesis by endothelial cell-derived signals. Trends Cell Biol. 25, 148-157. 10.1016/j.tcb.2014.11.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remédio L., Carvalho T., Caiado F., Bastos-Carvalho A., Martins D., Duarte A., Yagita H. and Dias S. (2012). Context- and cell-dependent effects of Delta-like 4 targeting in the bone marrow microenvironment. PLoS ONE 7, e52450 10.1371/journal.pone.0052450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rocha A. S., Vidal V., Mertz M., Kendall T. J., Charlet A., Okamoto H. and Schedl A. (2015). The angiocrine factor Rspondin3 is a key determinant of liver zonation. Cell Rep. 13, 1757-1764. 10.1016/j.celrep.2015.10.049 [DOI] [PubMed] [Google Scholar]
- Salvesen G. S. and Dixit V. M. (1997). Caspases: Intracellular signaling by proteolysis. Cell 91, 443-446. 10.1016/S0092-8674(00)80430-4 [DOI] [PubMed] [Google Scholar]
- Schlieve C. R., Mojica S. G., Holoyda K. A., Hou X., Fowler K. L. and Grikscheit T. C. (2016). Vascular Endothelial Growth Factor (VEGF) bioavailability regulates angiogenesis and intestinal stem and progenitor cell proliferation during postnatal small intestinal development. PLoS ONE 11, e0151396 10.1371/journal.pone.0151396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt A., Brixius K. and Bloch W. (2007). Endothelial precursor cell migration during vasculogenesis. Circ. Res. 101, 125-136. 10.1161/CIRCRESAHA.107.148932 [DOI] [PubMed] [Google Scholar]
- Sequeira S. J., Gervais E. M., Ray S. & Larsen M. (2013). Genetic modification and recombination of salivary gland organ cultures. J. Vis. Exp. 71, e50060 10.3791/50060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tucker A. S. (2007). Salivary gland development. Semin. Cell Dev. Biol. 18, 237-244. 10.1016/j.semcdb.2007.01.006 [DOI] [PubMed] [Google Scholar]
- Wei C., Larsen M., Hoffman M. P. and Yamada K. M. (2007). Self-organization and branching morphogenesis of primary salivary epithelial cells. Tissue Eng. 13, 721-735. 10.1089/ten.2006.0123 [DOI] [PubMed] [Google Scholar]
- Wells K. L., Gaete M., Matalova E., Deutsch D., Rice D. and Tucker A. S. (2013). Dynamic relationship of the epithelium and mesenchyme during salivary gland initiation: the role of Fgf10. Biol. Open 2, 981-989. 10.1242/bio.20135306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whittles C. E., Pocock T. M., Wedge S. R., Kendrew J., Hennequin L. F., Harper S. J. and Bates D. O. (2002). ZM323881, a novel inhibitor of vascular endothelial growth factor-receptor-2 tyrosine kinase activity. Microcirculation 9, 513-522. 10.1038/sj.mn.7800164 [DOI] [PubMed] [Google Scholar]
- Yamada K. M. and Cukierman E. (2007). Modeling tissue morphogenesis and cancer in 3D. Cell 130, 601-610. 10.1016/j.cell.2007.08.006 [DOI] [PubMed] [Google Scholar]
- Yoshitomi H. and Zaret K. S. (2004). Endothelial cell interactions initiate dorsal pancreas development by selectively inducing the transcription factor Ptf1a. Development 131, 807-817. 10.1242/dev.00960 [DOI] [PubMed] [Google Scholar]
- Zhang J. Y., Cui L., Xu M. H. and Zheng Y. L. (2014). Restoring the secretory function of irradiation-damaged salivary gland by administrating deferoxamine in mice. PLoS ONE 9, e113721 10.1371/journal.pone.0113721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zudaire E., Gambardella L. S., Kurcz C. and Vermeren S. (2011). A computational tool for quantitative analysis of vascular networks. PLoS ONE 6, e27385 10.1371/journal.pone.0027385 [DOI] [PMC free article] [PubMed] [Google Scholar]