Abstract
Left-right (LR) asymmetry is a fundamental feature of internal anatomy, yet the emergence of morphological asymmetry remains one of the least understood phases of organogenesis. Asymmetric rotation of the intestine is directed by forces outside the gut, but the morphogenetic events that generate anatomical asymmetry in other regions of the digestive tract remain unknown. Here, we show in mouse and Xenopus that the mechanisms that drive the curvature of the stomach are intrinsic to the gut tube itself. The left wall of the primitive stomach expands more than the right wall, as the left epithelium becomes more polarized and undergoes radial rearrangement. These asymmetries exist across several species, and are dependent on LR patterning genes, including Foxj1, Nodal and Pitx2. Our findings have implications for how LR patterning manifests distinct types of morphological asymmetries in different contexts.
KEY WORDS: Left-right, Asymmetry, Stomach, Gut, Morphogenesis, Pitx2, Xenopus, Mouse
Highlighted Article: Left-right patterning creates morphogenetic differences between the left and right walls of the vertebrate foregut that generate the asymmetric curvature of the stomach.
INTRODUCTION
The external symmetry of bilateria belies the marked left-right (LR) asymmetry of their internal organs. In vertebrates, the LR axis is established by nodal cilia, which activate expression of the TGFβ molecule Nodal exclusively on the left side of the embryo (Levin et al., 1995; Lowe et al., 1996; Murcia et al., 2000; Okada et al., 1999). Downstream of Nodal, the transcription factor Pitx2c is then expressed along the left side of developing organs as they acquire morphological asymmetry (Campione et al., 1999). These patterning events culminate in anatomical laterality, including left- or right-side organ position, LR asymmetry in the shapes/sizes of paired organs, or intrinsic morphological disparity between the sides of an individual organ (Capdevila et al., 2000).
The J-shaped curvature of the stomach is an archetype of such laterality. The exact configuration of this organ varies among species with different diets, yet its distinctive shape, which is characterized by a longer ‘greater’ curvature on the left, and shorter ‘lesser’ curvature on the right, is highly conserved and integral to normal function (Stevens and Hume, 2004). Early embryologists contended that stomach anatomy results from a developmental rotation in which the ostensibly enlarged dorsal surface turns leftward to become the greater curvature (Liebermann-Meffert, 1969) (Fig. 1A). However, some have argued that rotation is illusory and that curvature emerges via an intrinsic process, e.g. asymmetric growth (Kanagasuntheram, 1957; Liebermann-Meffert, 1969) (Fig. 1B). Remarkably, the cellular and molecular events that drive stomach curvature have never been experimentally addressed.
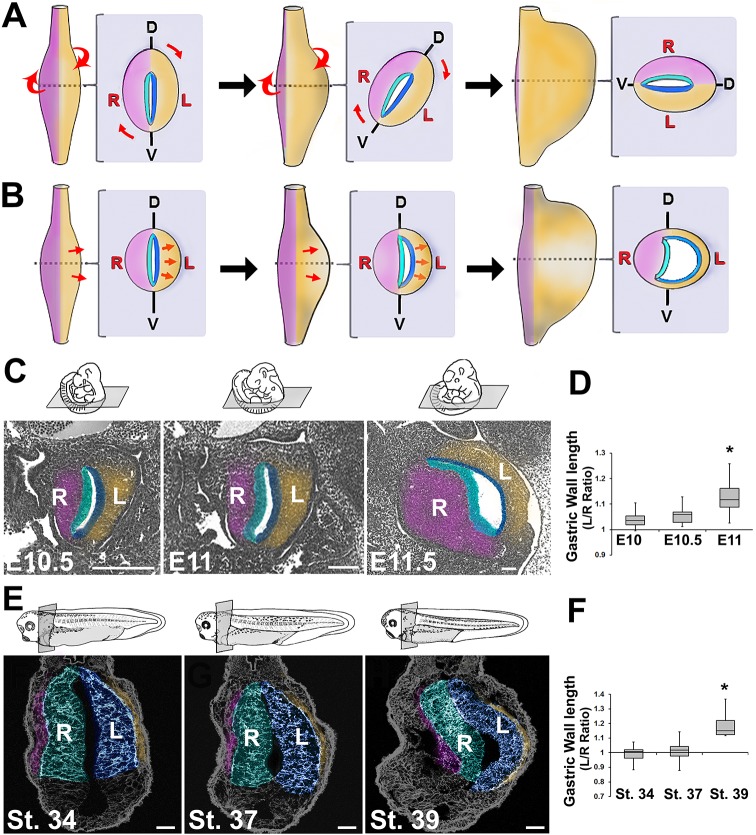
Fig. 1.
Early stomach undergoes leftward expansion. The rotation model (A) posits that the embryonic stomach (shown in ventral views and cross-sections at successive stages) rotates around its longitudinal axis, shifting its dorsal face leftward. An alternative model (B) theorizes that the left wall expands more than the right. Sections of E10.5, E11 and E11.5 mouse embryos (C) or stage 34, 37 and 39 frog embryos (E) reveal the leftward expansion of the early stomach. The left/right ratio of the lengths of the stomach walls becomes significantly greater than 1 in mouse by E11 (D) and in frog by stage 39 (F); *P<0.05. Sections in C and E are false-colored to match diagrams in A and B, highlighting layers of the stomach: right mesoderm, pink; right endoderm, teal; left endoderm, blue; left mesoderm, gold. In all sections, dorsal is upwards and the left side of animal is on right side of image. D, dorsal; V, ventral; L, left; R, right. Scale bars: 500 µm in C (E11.5, 150 µm); 75 µM in E.
RESULTS AND DISCUSSION
Stomach asymmetry is independent of rotation
We compared stomach curvature formation in two animals, the mouse and the frog (Xenopus laevis). In both species, the slit-like lumen of the still-straight primitive stomach [mouse stage E10 and frog stage 34 (Nieuwkoop and Faber, 1994)] is initially aligned with the dorsal-ventral (DV) axis of the embryo (Fig. 1C,E). If the dorsal face of the organ rotates leftward during development, we would expect its DV axis to shift clockwise with respect to the embryonic midline as asymmetric morphology emerges (in posterior view, Fig. 1A). Surprisingly, we find no indication of such a shift in either species. However, in both the mouse (Fig. 1C,D) and frog (Fig. 1E-F), the left wall of the early stomach becomes longer than the right wall and begins to bend outwards, skewing the nascent lumen leftward. These LR differences are also evident in orthogonal planes of section (not shown), indicating that a general expansion of the left wall occurs concomitantly with the emergence of overt laterality. This intrinsic asymmetry is evident prior to mesogastrial remodeling; indeed, in the frog, a mesogastrium is not detectable when stomach asymmetry becomes obvious. Thus, external tissues/forces may not be required for stomach curvature. These observations are congruent with a model of asymmetric stomach morphogenesis driven by preferential expansion of the left wall (i.e. Fig. 1B).
Differential tissue architecture develops in the left and right stomach walls
To define the morphogenetic events that drive asymmetric expansion, we compared contralateral tissue architecture during stomach development (Fig. 2). The walls of the gut tube comprise an inner layer of endoderm, which will become the epithelium, and an outer layer of mesoderm, which will become visceral muscle and connective tissue. Although the relative thickness of each layer varies between mouse and frog embryos, the tissues of the left stomach wall become significantly thinner than the right wall at stages preceding and coinciding with curvature in both species (Fig. 2A,B,E,F,I,J,M,N).
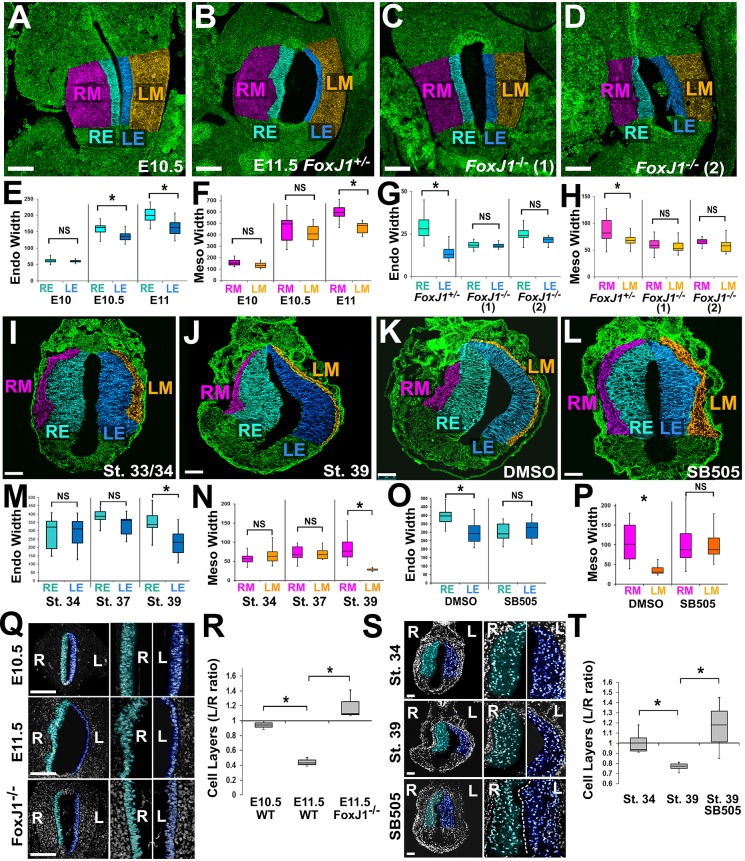
Fig. 2.
Asymmetries in tissue architecture are regulated by left-right patterning. Sections through mouse (A-D) or frog (I-L) stomachs were stained for β-catenin (A-D) or integrin (I-L) (green) and false color-coded as in Fig. 1 (RE, right endoderm; LE, left endoderm; RM, right mesoderm; LM, left mesoderm). The widths of the endoderm (Endo) and mesoderm (Meso) are significantly different by E10.5-11 in mouse (E,F) and stage 39 in frog (M,N). In Foxj1+/− controls (E11.5), the lumen expands leftward and left-right differences in tissue width are evident (B); however, in Foxj1−/− mutants, the normal leftward expansion of the stomach is perturbed (C,D), and left-right differences in tissue width are eliminated (G,H). Likewise, frog embryos exposed to DMSO show normal leftward expansion of the stomach lumen (K); this is eliminated in embryos exposed to a Nodal inhibitor (SB505124; L), as are normal left-right differences in widths of endoderm and mesoderm (O,P). Nuclear staining reveals asymmetry in the number of endoderm cell layers in the right (teal) versus left (blue) stomach walls by E11.5 in mouse (Q,R) and stage 39 in frog (S,T); this asymmetry is perturbed in Foxj1−/− mutants (Q,R) and in frog embryos exposed to SB505124 (S,T). Scale bars: 100 µm in A-D,Q; 75 µm in I-L,S. *P<0.01; NS, not significant.
In vertebrates, the transcription factor Foxj1 is a master regulator of motile ciliogenesis required for proper establishment of the LR axis (Brody et al., 2000; Chen et al., 1998; Stubbs et al., 2008; Tamakoshi et al., 2006; Walentek et al., 2012). To determine whether the differential morphogenesis of the stomach walls is dependent on LR patterning, we analyzed Foxj1 mutant mouse embryos. In Foxj1 heterozygotes, which have normal LR patterning (Muthusamy et al., 2014), the preferential lengthening of the left stomach wall and asymmetric lumen expansion proceed normally (Fig. 2B). However, in Foxj1 homozygous-null embryos, in which nodal cilia are defective and LR patterning and organ morphology are consequently randomized, the length of the opposing stomach walls is equivalent and the stomach appears straightened (Fig. 2C,D; Fig. S1). Moreover, there is no LR asymmetry in the thickness of the tissue layers (Fig. 2G,H).
LR asymmetry is propagated by the expression of nodal on the left side of the embryo (Levin et al., 1995; Lowe et al., 1996; Murcia et al., 2000; Okada et al., 1999). Xenopus neurulae exposed to a small molecule inhibitor of the Nodal receptor (e.g. SB505124; Fig. S2A,B) have decreased Nodal signaling, perturbed laterality and aberrant organogenesis (Dush et al., 2011). In such embryos, the lengths of the contralateral stomach walls are equal (Fig. S2C), and the leftward curvature of the stomach is eliminated (Fig. 2K,L; 56%; n=38; Fig. S2D). Furthermore, there is no difference in endoderm/mesoderm thickness (Fig. 2O,P). Thus, the early morphological differences between the sides of the stomach are a manifestation of LR patterning events.
Stomach curvature is accompanied by left-right asymmetric endoderm rearrangement
As radial rearrangement of the endoderm thins and expands other regions of the gut tube (Chalmers and Slack, 2000; Reed et al., 2009), we compared the number of endoderm cell layers within the left and right stomach walls. Prior to curvature, there are no LR differences (L/R ratio ∼1). However, as curvature emerges, fewer layers are apparent in the left wall (L/R ratio<1; average mouse=0.44, P<0.01; frog=0.77, P<0.01; Fig. 2Q-T). As the total number and shape of cells on each side remains equivalent during curvature (Fig. S3A-C), these results indicate that preferential expansion of the left wall is driven by LR asymmetric cell rearrangement.
Appearing concomitantly with LR differences in cell rearrangement are asymmetries in endoderm cell polarity. This is not surprising, as polarity is a prerequisite for cells to undergo radial rearrangement (Marsden and Desimone, 2001) and endoderm cells acquire apicobasal polarity as they rearrange to lengthen the intestine and form the mature digestive epithelium (Grosse et al., 2011; Matsumoto et al., 2002; Reed et al., 2009). In Foxj1 heterozygous mice, the endoderm of the posterior right stomach wall becomes irregularly stratified, whereas the left endoderm is highly polarized (as indicated by apical enrichment of tubulin; Fig. 3A,D,G). This LR asymmetric tissue architecture is variably perturbed in heterotaxic Foxj1−/− embryos, consistent with the randomization of LR patterning cues (Zhang et al., 2004). In one Foxj1−/− embryo, both left and right walls of the straightened stomach are highly polarized (Fig. 3B,E,H), and the number of endoderm cell layers remains equivalent (average L/R ratio=1.19, P<0.01; Fig. 2Q,R); in another case, however, both sides exhibit irregular architecture (Fig. 3C,F,I).
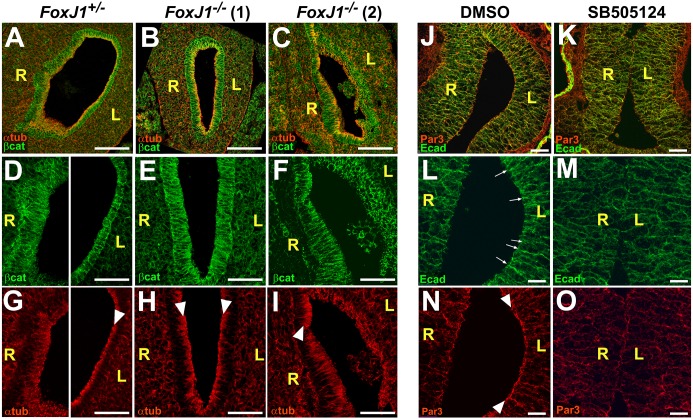
Fig. 3.
Asymmetries in stomach epithelial polarity are regulated by left-right signaling. Stomach sections immunostained for α-tubulin (αtub, red) and β-catenin (β-cat, green) in the E11.5 mouse (A-I), or Par3 (red) and E-cadherin (Ecad, green) in the stage 39 frog (J-O), reveal left-right differences in endoderm morphogenesis. In the posterior stomach of Foxj1+/− mice, right endoderm is irregular, whereas left endoderm is more organized (D) and highly polarized, as indicated by tight apical concentration of α-tubulin (αtub, red; arrowhead, G). Both walls of Foxj1−/− mutant embryo [Foxj1−/−(1), B,E,H], comprise polarized cells with tight apical concentration of αtub (arrowheads, H); both sides resemble control left epithelium. In another mutant [Foxj1−/−(2), C,F,I] both sides are irregular and resemble control right epithelium, although sub-regions exhibit greater organization and polarity (arrowhead, I). In control (DMSO) frog embryos (J,L,N), left endoderm is more polarized than right, as indicated by enrichment of E-cadherin (Ecad; green) at apical adherens junctions (arrows, L), and the concentration of the apical polarity marker Par3 at the lumenal surface (red; arrowheads, N). By contrast, SB505124 (K,M,O) results in right isomerism, with left endoderm resembling right endoderm, as indicated by absence of apical enrichment of E-cadherin (M) and Par3 (O). L, left; R, right. Scale bars: 100 µm in A-C; 50 µm in D-K; 25 µm in L-O. (See Fig. S4 for higher magnification images.)
In frogs, the endoderm is substantially thicker than in the mouse; nonetheless, similar LR differences appear as the left endoderm adopts a more polarized morphology (Fig. 3J,L,N; Fig. S4A,B,E,H). Interestingly, in the straightened stomachs of Nodal-deficient (i.e. SB505124-treated) frog embryos, the left endoderm closely resembles the normal right side (Fig. 3K,M,O) and the number of cell layers remains equivalent (average L/R ratio=1.16, P<0.01; Fig. 2S-T). Thus, in both species, the emergence of morphological stomach laterality correlates with LR asymmetries in endoderm polarity and rearrangement.
Pitx2 controls left stomach epithelial morphogenesis
The expression of Pitx2c is limited to the left wall during stomach development (Liu et al., 2001; Logan et al., 1998) and is required for normal organ laterality (Bamforth et al., 2004; Campione et al., 1999; Liu et al., 2001; Logan et al., 1998; Ryan et al., 1998). Pitx2c-null mice may exhibit reversed or midline stomach orientations (Bamforth et al., 2004), although the mechanism by which Pitx2 impacts stomach morphogenesis is unknown. In other regions of the digestive tract, Pitx isoforms are associated with polarized epithelial morphogenesis and tissue expansion (e.g. intestine lengthening, cecal budding) (Al et al., 2012; Campione et al., 1999; Chung et al., 2011). Thus, asymmetric expression of Pitx2c in left stomach may drive the observed asymmetries in endoderm morphogenesis.
Consistent with this hypothesis, CRISPR-Cas9-mediated mutation of Xenopus pitx2c results in straightened stomachs in which the left wall does not lengthen or thin, and exhibits abnormal epithelial architecture (Fig. S5) and failed cell rearrangement [as indicated by an increased number of cell layers in the left epithelium (average L/R ratio=1.31, P<0.05)], confirming that pitx2c is specifically required for key aspects of asymmetric stomach morphogenesis. However, as CRISPR-Cas9 editing introduces random mutations (Fig. S5E), it is impossible to define spatially the exact cells harboring null mutations and the extent to which Pitx2 activity is altered in each F0 animal. Therefore, to specifically determine the role of pitx2c within left stomach, we also capitalized on the amenability of Xenopus for targeting gain- or loss-of-function reagents specifically to the left or right side of developing organs by microinjection, a strategy not easily feasible in mouse (Blum et al., 2009; Duncan and Khokha, 2016).
First, we employed two independent morpholino oligonucleotides (MOs) to deplete Pitx2c translation in a targeted manner (see Fig. S6A,B). When pitx2c MO is microinjected to target the right stomach, stomach curvature is unaffected (8%; n=37); however, if pitx2c MO is targeted to the left side, the organ fails to curve [41% reduced/absent greater curvature (n=37), compared with control MO, 4% (n=41)], recapitulating the CRISPR-Cas9 phenotype (Fig. 4A,B; Fig. S6C). Concomitant with lack of curvature, the Pitx2c-deficient left epithelium now resembles the less organized and unpolarized right epithelium (Fig. 4E,F,I,J,M,N,Q,R; Fig. S4C,F,I). The pitx2c MO-injected left wall does not thin (Fig. S6D,E), nor do the endoderm cells rearrange, as indicated by increased cell layers on the left (Fig. S6F; average L/R ratio control MO=0.83; pitx2c MO=1.34). Interestingly, the endoderm and mesoderm layers of the stomach wall may influence each other during asymmetric morphogenesis, as MO-injected clones limited primarily to the endoderm or mesoderm layer also perturb the architecture of neighboring uninjected tissue (Fig. S7). This reciprocal influence is well established in foregut patterning (Womble et al., 2016).
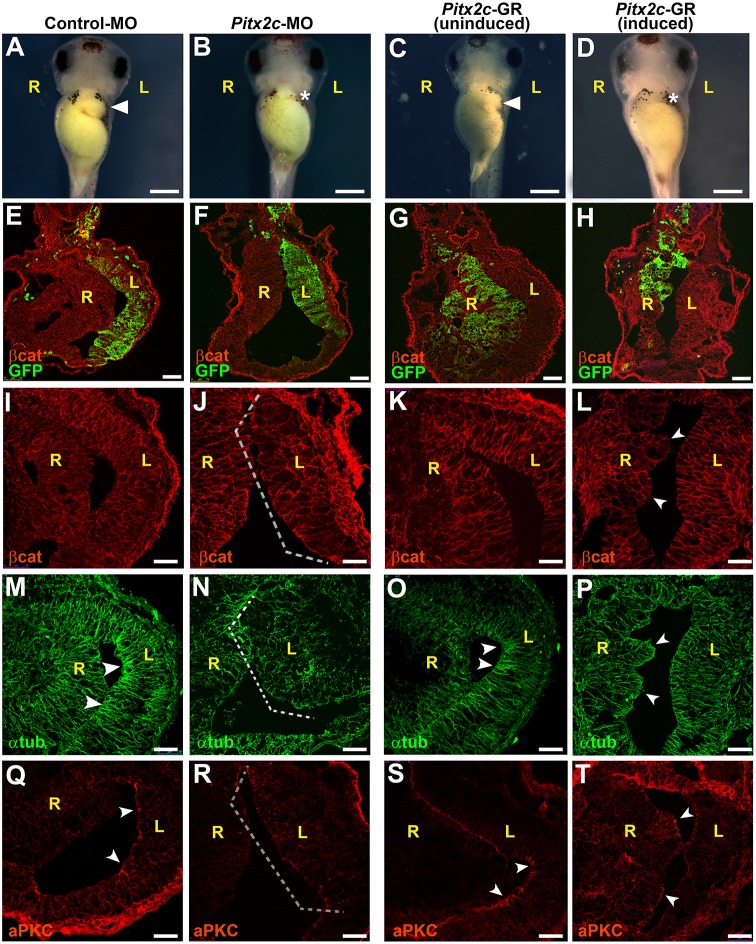
Fig. 4.
Pitx2c controls epithelial morphogenesis in left stomach wall. Frog embryos were injected with control morpholino (control-MO; A,E,I,M,Q) or Pitx2c-MO (B,F,J,N,R) targeted to the left side of the stomach, or injected with Pitx2c-GR mRNA (C,G,K,O,S and D,H,L,P,T) targeted to the right side. (See Fig. S6A,B for morpholino validation.) In injected embryos (A-D), the greater curvature of the stomach at stage 42 is indicated by an arrowhead (A,C); absence of curvature is specified by an asterisk (B,D). Sections through stage 39 stomachs (E-T) were stained for β-catenin (βcat; red; E-L), α-tubulin (αtub; green; M-P) or atypical PKC (aPKC; red; Q-T). GFP mRNA was co-injected as a lineage tracer to confirm proper targeting (green; E-H). MO depletion on the left (F) or ectopic activity of Pitx2 on the right (H) results in a loss of asymmetry within the stomach compared with controls (E,G, respectively). In addition, compared with control-MO injected embryos, in which αtub and aPKC are concentrated at the apical surface of the left stomach wall (arrowheads in M,Q, respectively), MO depletion of Pitx2c disrupts epithelial architecture (brackets in J,N,R). Meanwhile, dexamethasone induction of Pitx2c activity in the right wall polarizes stomach endoderm, as indicated by ectopic regions of polarized epithelial architecture (arrowheads in L,P,T), correlating with ectopic αtub (P) and aPKC (T), which are not observed in right wall of uninduced controls (K,O,S). Scale bars: 500 µm in A-D; 75 µm in E-H; 50 µm in I-T. L, left; R, right.
To determine whether pitx2c alone is sufficient to impart apicobasal polarity to the endoderm, we targeted mRNA encoding a glucocorticoid-inducible version of Pitx2c, pitx2c-GR (Chung et al., 2011), to the right stomach. Induction of ectopic Pitx2c activity by dexamethasone eliminates stomach curvature (60%, n=82; Fig. 4C,D), while uninduced control stomachs remain unaffected (10%; n=90). Strikingly, inducing Pitx2c-GR results in ectopic polarization of the right endoderm, with the affected tissue expanding into the stomach lumen (Fig. 4G,H,K,L) and expressing higher levels of polarity markers compared with adjacent uninduced tissue (Fig. 4O,P,S,T; Fig. S4D,G,J). Thus, pitx2c is necessary and sufficient to promote epithelial polarization in stomach endoderm.
How anatomical asymmetries are generated is relevant to the origin of novelty during evolution and integral to the etiology of birth defects but, for most organs, this crucial phase of organogenesis is poorly understood. Recent work suggests asymmetrical intestinal rotation is generated by LR differences in the dorsal mesentery that connects the midgut to the body wall – not in the gut tube itself, which remains symmetrical during the rotation process (Davis et al., 2008; Kurpios et al., 2008; Welsh et al., 2013). By contrast, our results indicate that stomach curvature is driven by asymmetric morphogenesis within the organ itself.
Moreover, whereas Pitx2c elicits tissue condensation in left dorsal mesentery (Davis et al., 2008), Pitx2c elicits tissue expansion in the left stomach, suggesting that the final manifestation of Pitx2c activity in different organs is not universal, but is influenced by geometric context (e.g. internal or external to the gut tube) and tissue type. Our study suggests that the final topology of the digestive tract is the end-product of at least two Pitx2-mediated symmetry-breaking processes: mesentery distortion, which positions the gut tube asymmetrically in the body cavity; and asymmetric gut morphogenesis, which sculpts disparate morphologies within the contralateral walls of the gut itself.
MATERIALS AND METHODS
Animals and genotyping
Animals used in accordance with NCSU IACUC regulations. Xenopus in vitro fertilization, embryo culture and staging were as described previously (Nieuwkoop and Faber, 1994; Sive et al., 1998). Heterozygous Foxj1+/creERT2 mice (Muthusamy et al., 2014) were used in timed pregnancy matings to obtain Foxj1+/creERT2 (i.e. Foxj1+/−) and Foxj1creERT2/creERT2 (i.e. Foxj1−/−) embryos. Noon on day of vaginal plug discovery was defined as E0.5. Embryos were harvested in ice-cold 0.1 M phosphate-buffered saline (PBS) and fixed overnight in 4% paraformaldehyde in PBS. PCR was used to identify homozygous creERT2 insertion into Foxj1 locus (Muthusamy et al., 2014); see supplementary Materials and Methods for genotyping primers.
Xenopus loss- and gain-of-function experiments
Pharmacological (e.g. SB505124) dosing of Xenopus neurulae (stage 19/20) was conducted as described previously (Dush et al., 2011). SB505124 was diluted to 5 µM in culture medium (0.1× MMR); an equal volume of DMSO (solvent) was used as a control.
Microinjection was performed at 1-cell (CRISPR) or 8-cell stages (morpholinos and mRNA), using established methods (Sive et al., 1998). Co-injected GFP mRNA validated tissue targeting. Pitx2-GR mRNA-injected embryos were exposed to dexamethasone (10 µM) or to ethanol (control) from stage 19/20. Synthetic mRNA was transcribed in vitro (mMessage mMachine kit; ThermoFisher). Cas9 mRNA and gRNAs were synthesized as described previously (Zhang et al., 2014). See supplementary Materials and Methods for morpholino/gRNA sequences and mutation analyses. Embryos were cultured through stages 34, 37, 39 or 42 and collected for phenotyping/immunofluorescence.
Immunofluorescence
Tissue processing, sectioning and immunostaining were as described previously (Dush and Nascone-Yoder, 2013). See supplementary Materials and Methods for antibodies. Mouse sections in Fig. 1 were downloaded from the EMAP eMouse Atlas Project (http://www.emouseatlas.org) (Richardson et al., 2014).
Methodology and statistics
All experiments were performed at least in triplicate, with similar results. Morphometric measurements performed with Image J, Photoshop or eMouse measuring tools, EMAP eMouse Atlas Project (http://www.emouseatlas.org). Statistical significance was determined by ANOVA with post-hoc Tukey HSD; see supplementary Materials and Methods for details.
Acknowledgements
We thank N. Muthasamy for assistance with mouse genotyping.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
A.D., N.M.A., C.J. and K.B. performed experiments; N.N.-Y. conceived of the project and wrote manuscript; H.T.G. provided mouse strains.
Funding
This work was supported by the National Institutes of Health (R01DK085300 and R21OD017963 to N.N-Y., R01NS062182 to H.T.G, T32ES007046 to A.D.). Deposited in PMC for release after 12 months.
Supplementary information
Supplementary information available online at http://dev.biologists.org/lookup/doi/10.1242/dev.143701.supplemental
References
- Al A. D., Sala F. G., Baptista S., Galzote R., Danopoulos S., Tiozzo C., Gage P., Grikscheit T., Warburton D., Frey M. R. et al. (2012). FGF9-Pitx2-FGF10 signaling controls cecal formation in mice. Dev. Biol. 369, 340-348. 10.1016/j.ydbio.2012.07.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bamforth S. D., Bragança J., Farthing C. R., Schneider J. E., Broadbent C., Michell A. C., Clarke K., Neubauer S., Norris D., Brown N. A. et al. (2004). Cited2 controls left-right patterning and heart development through a Nodal-Pitx2c pathway. Nat. Genet. 36, 1189-1196. 10.1038/ng1446 [DOI] [PubMed] [Google Scholar]
- Blum M., Beyer T., Weber T., Vick P., Andre P., Bitzer E. and Schweickert A. (2009). Xenopus, an ideal model system to study vertebrate left-right asymmetry. Dev. Dyn. 238, 1215-1225. 10.1002/dvdy.21855 [DOI] [PubMed] [Google Scholar]
- Brody S. L., Yan X. H., Wuerffel M. K., Song S.-K. and Shapiro S. D. (2000). Ciliogenesis and left-right axis defects in forkhead factor HFH-4-null mice. Am. J. Respir. Cell Mol. Biol. 23, 45-51. 10.1165/ajrcmb.23.1.4070 [DOI] [PubMed] [Google Scholar]
- Campione M., Steinbeisser H., Schweickert A., Deissler K., van Bebber F., Lowe L. A., Nowotschin S., Viebahn C., Haffter P., Kuehn M. R. et al. (1999). The homeobox gene Pitx2: mediator of asymmetric left-right signaling in vertebrate heart and gut looping. Development 126, 1225-1234. [DOI] [PubMed] [Google Scholar]
- Capdevila J., Vogan K. J., Tabin C. J. and Izpisúa Belmonte J. C. (2000). Mechanisms of left-right determination in vertebrates. Cell 101, 9-21. 10.1016/S0092-8674(00)80619-4 [DOI] [PubMed] [Google Scholar]
- Chalmers A. D. and Slack J. M. (2000). The Xenopus tadpole gut: fate maps and morphogenetic movements. Development 127, 381-392. [DOI] [PubMed] [Google Scholar]
- Chen J., Knowles H. J., Hebert J. L. and Hackett B. P. (1998). Mutation of the mouse hepatocyte nuclear factor/forkhead homologue 4 gene results in an absence of cilia and random left-right asymmetry. J. Clin. Invest. 102, 1077-1082. 10.1172/JCI4786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung M.-I., Nascone-Yoder N. M., Grover S. A., Drysdale T. and Wallingford J. B. (2011). Direct activation of Shroom3 transcription by Pitx proteins drives epithelial morphogenesis in the developing gut. Development 137, 1339-1349. 10.1242/dev.044610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis N. M., Kurpios N. A., Sun X., Gros J., Martin J. F. and Tabin C. J. (2008). The chirality of gut rotation derives from left-right asymmetric changes in the architecture of the dorsal mesentery. Dev. Cell 15, 134-145. 10.1016/j.devcel.2008.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan A. R. and Khokha M. K. (2016). Xenopus as a model organism for birth defects-Congenital heart disease and heterotaxy. Semin. Cell Dev. Biol. 51, 73-79. 10.1016/j.semcdb.2016.02.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dush M. K., McIver A. L., Parr M. A., Young D. D., Fisher J., Newman D. R., Sannes P. L., Hauck M., Deiters A. and Nascone-Yoder N. (2011). Heterotaxin: a novel TGF-β signaling inhibitor identified in a multi-phenotype profiling screen in Xenopus embryos. Chem. Biol. 18, 252-263. 10.1016/j.chembiol.2010.12.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dush M. K. and Nascone-Yoder N. M. (2013). Jun N-terminal kinase maintains tissue integrity during cell rearrangement in the gut. Development 140, 1457-1466. 10.1242/dev.086850 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grosse A. S., Pressprich M. F., Curley L. B., Hamilton K. L., Margolis B., Hildebrand J. D. and Gumucio D. L. (2011). Cell dynamics in fetal intestinal epithelium: implications for intestinal growth and morphogenesis. Development 138, 4423-4432. 10.1242/dev.065789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanagasuntheram R. (1957). Development of the human lesser sac. J. Anat. 91, 188-206. [PMC free article] [PubMed] [Google Scholar]
- Kurpios N. A., Ibanes M., Davis N. M., Lui W., Katz T., Martin J. F., Belmonte J. C. I. and Tabin C. J. (2008). The direction of gut looping is established by changes in the extracellular matrix and in cell:cell adhesion. Proc. Natl. Acad. Sci. USA 105, 8499-8506. 10.1073/pnas.0803578105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M., Johnson R. L., Stern C. D., Kuehn M. and Tabin C. (1995). A molecular pathway determining left-right asymmetry in chick embryogenesis. Cell 82, 803-814. 10.1016/0092-8674(95)90477-8 [DOI] [PubMed] [Google Scholar]
- Liebermann-Meffert D. (1969). [Development of form and position of the human stomach and its mesenteries]. Acta Anat. (Basel.) 72, 376-410. 10.1159/000143257 [DOI] [PubMed] [Google Scholar]
- Liu C., Liu W., Lu M. F., Brown N. A. and Martin J. F. (2001). Regulation of left-right asymmetry by thresholds of Pitx2c activity. Development 128, 2039-2048. [DOI] [PubMed] [Google Scholar]
- Logan M., Pagán-Westphal S. M., Smith D. M., Paganessi L. and Tabin C. J. (1998). The transcription factor Pitx2 mediates situs-specific morphogenesis in response to left-right asymmetric signals. Cell 94, 307-317. 10.1016/S0092-8674(00)81474-9 [DOI] [PubMed] [Google Scholar]
- Lowe L. A., Supp D. M., Sampath K., Yokoyama T., Wright C. V. E., Potter S. S., Overbeek P. and Kuehn M. R. (1996). Conserved left-right asymmetry of nodal expression and alterations in murine situs inversus. Nature 381, 158-161. 10.1038/381158a0 [DOI] [PubMed] [Google Scholar]
- Marsden M. and Desimone D. W. (2001). Regulation of cell polarity, radial intercalation and epiboly in Xenopus: novel roles for integrin and fibronectin. Development 128, 3635-3647. [DOI] [PubMed] [Google Scholar]
- Matsumoto A., Hashimoto K., Yoshioka T. and Otani H. (2002). Occlusion and subsequent re-canalization in early duodenal development of human embryos: integrated organogenesis and histogenesis through a possible epithelial-mesenchymal interaction. Anat. Embryol. (Berl) 205, 53-65. 10.1007/s00429-001-0226-5 [DOI] [PubMed] [Google Scholar]
- Murcia N. S., Richards W. G., Yoder B. K., Mucenski M. L., Dunlap J. R. and Woychik R. P. (2000). The Oak Ridge Polycystic Kidney (orpk) disease gene is required for left-right axis determination. Development 127, 2347-2355. [DOI] [PubMed] [Google Scholar]
- Muthusamy N., Vijayakumar A., Cheng G. Jr and Ghashghaei H. T. (2014). A knock-in Foxj1(CreERT2::GFP) mouse for recombination in epithelial cells with motile cilia. Genesis 52, 350-358. 10.1002/dvg.22753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieuwkoop P. D. and Faber J. (1994). Normal Table of Xenopus laevis (Daudin). New York: Garland Publishing Inc. [Google Scholar]
- Okada Y., Nonaka S., Tanaka Y., Saijoh Y., Hamada H. and Hirokawa N. (1999). Abnormal nodal flow precedes situs inversus in iv and inv mice. Mol. Cell 4, 459-468. 10.1016/S1097-2765(00)80197-5 [DOI] [PubMed] [Google Scholar]
- Reed R. A., Womble M. A., Dush M. K., Tull R. R., Bloom S. K., Morckel A. K., Devlin E. W. and Nascone-Yoder N. M. (2009). The morphogenesis of the primitive gut tube is generated by Rho/ROCK/Myosin II-mediated endoderm rearrangements. Dev. Dyn. 238, 3111-3125. 10.1002/dvdy.22157 [DOI] [PubMed] [Google Scholar]
- Richardson L., Venkataraman S., Stevenson P., Yang Y., Moss J., Graham L., Burton N., Hill B., Rao J., Baldock R. A. et al. (2014). EMAGE mouse embryo spatial gene expression database: 2014 update. Nucleic Acids Res. 42, D835-D844. 10.1093/nar/gkt1155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan A. K., Blumberg B., Rodriguez-Esteban C., Yonei-Tamura S., Tamura K., Tsukui T., de la Peña J., Sabbagh W., Greenwald J., Choe S. et al. (1998). Pitx2 determines left-right asymmetry of internal organs in vertebrates. Nature 394, 545-551. 10.1038/29004 [DOI] [PubMed] [Google Scholar]
- Sive H. L., Grainger R. M. and Harland R. M. (1998). Early Development of Xenopus laevis. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press. [Google Scholar]
- Stevens C. E. and Hume I. D. (2004). Comparative Physiology of the Vertebrate Digestive System. Cambridge: Cambridge University Press. [Google Scholar]
- Stubbs J. L., Oishi I., Izpisúa Belmonte J. C. and Kintner C. (2008). The forkhead protein Foxj1 specifies node-like cilia in Xenopus and zebrafish embryos. Nat. Genet. 40, 1454-1460. 10.1038/ng.267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamakoshi T., Itakura T., Chandra A., Uezato T., Yang Z., Xue X.-D., Wang B., Hackett B. P., Yokoyama T. and Miura N. (2006). Roles of the Foxj1 and Inv genes in the left-right determination of internal organs in mice. Biochem. Biophys. Res. Commun. 339, 932-938. 10.1016/j.bbrc.2005.11.097 [DOI] [PubMed] [Google Scholar]
- Walentek P., Beyer T., Thumberger T., Schweickert A. and Blum M. (2012). ATP4a is required for Wnt-dependent Foxj1 expression and leftward flow in Xenopus left-right development. Cell Rep. 1, 516-527. 10.1016/j.celrep.2012.03.005 [DOI] [PubMed] [Google Scholar]
- Welsh I. C., Thomsen M., Gludish D. W., Alfonso-Parra C., Bai Y., Martin J. F. and Kurpios N. A. (2013). Integration of left-right Pitx2 transcription and wnt signaling drives asymmetric gut morphogenesis via daam2. Dev. Cell 26, 629-644. 10.1016/j.devcel.2013.07.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Womble M., Pickett M. and Nascone-Yoder N. (2016). Frogs as integrative models for understanding digestive organ development and evolution. Semin. Cell Dev. Biol. 51, 92-105. 10.1016/j.semcdb.2016.02.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang M., Bolfing M. F., Knowles H. J., Karnes H. and Hackett B. P. (2004). Foxj1 regulates asymmetric gene expression during left-right axis patterning in mice. Biochem. Biophys. Res. Commun. 324, 1413-1420. 10.1016/j.bbrc.2004.09.207 [DOI] [PubMed] [Google Scholar]
- Zhang F., Wen Y. and Guo X. (2014). CRISPR/Cas9 for genome editing: progress, implications and challenges. Hum. Mol. Genet. 23, R40-R46. 10.1093/hmg/ddu125 [DOI] [PubMed] [Google Scholar]