Abstract
The main objectives of this study were to obtain clones of Paracoccidioides brasiliensis by two methods (micromanipulation and plating assay) and to determine if the secretion of the 43-kDa glycoprotein (gp43) is dependent on the clonal culture. The results show that the secretion of gp43 is not dependent on clonal cultures. Clones that originally were secretors of this molecule, after subculturing, lost this characteristic; on the other hand, clones that originally did not secrete gp43 began to secrete gp43 after subculturing.
Paracoccidioides brasiliensis is a dimorphic fungus that causes paracoccidioidomycosis. P. brasiliensis secretes and excretes various proteins and glycoproteins, and the 43-kDa molecule (gp43) is the immunodominant antigen and the main antigenic component, useful for diagnosis of paracoccidioidomycosis.
The occurrence of instability in the synthesis of antigenic components by the same P. brasiliensis isolate under controlled incubation conditions was first observed by Franco et al. (5). When a specific isolate is used to produce antigens for immunodiffusion tests or Western blot assays, the gp43 molecule must be present in these preparations. However, this condition is not reproducible, and sometimes the final preparation does not contain gp43. Our hypothesis was that a culture is composed of different clones and for some unknown reason only some of them express gp43. A more reasonable approach to obtain a homogenous antigen preparation is to work with clonal cultures in which all cells contain the same genetic information.
In the present study, we compared two ways to isolate single cells (clones) of P. brasiliensis, i.e., by a micromanipulation technique and by plating of single cells, and determined which medium is better in order to obtain clonal cultures. In addition, these clonal cultures were tested for their potential to secrete gp43 in order to confirm or reject our hypothesis about the relationship between gp43 secretion and clonal cultures. Also, clonal cultures were analyzed by rapidly amplified polymorphic DNA to verify possible polymorphism at the DNA level.
Three isolates of P. brasiliensis were used in this study, Pb-113, Pb-18, and Pb-339 (isolates obtained from human chronic paracoccidioidomycosis in Brazil). Three media were tested in order to obtain clones of P. brasiliensis: YPD agar medium (Difco), Sabouraud agar (Difco), and BHI agar (Difco) incorporated with growth factor obtained according to Castañeda et al. (4), plus bovine fetal serum (Gibco-BRL) and 10 μg of streptomycin and 10, 000 U of penicillin (Sigma). The same lot of medium was used for all experiments.
To obtain clones, P. brasiliensis (Pb-113, Pb-18, and Pb-339) yeast forms were grown on YPD agar slants for 3 days at 36°C. The total growth slant from one tube was transferred to a 500-ml Erlenmeyer flask containing 100 ml of YPD broth and incubated at 36°C in a rotary incubator at 50 rpm (ETICA Equipamentos, São Paulo, Brazil). After 3 days of incubation, 10 ml of this culture was transferred to a 50-ml sterile Falcon tube and treated with 5% sodium heparin for 5 min; 4 ml of this suspension was transferred to a sterile15-ml Falcon tube, sonicated in a cold bath sonicator (Branson Ultrasonics 2200, 6 in. by 11.5 in. by 6 in. inner diameter) for a total of 10 min (1 min of sonication followed by 3-min intervals on ice) and mixed in a Vortex mixer for 1 min. A drop of this solution was then placed on a slide and observed under a light microscope (×400). Viability was always higher than 90% as determined by trypan blue staining. Isolated cells were counted with a hemacytometer, and a solution was prepared and adjusted to 1,000 cells per ml of sterile saline.
In this step, two plating methods were tested in order to obtain isolated cultures from a single cell. (i) We spread 100 μl of this solution containing 100 cells on the surface of a prepared BHI agar plate (onto one half of a petri dish 9 cm in diameter) with a Drigalsky loop. Then, with the aid of a micromanipulator microscope (Leitz), each cell was picked up with a microneedle (by way of a water meniscus) and placed on the other side of the plate (without dragging), and the site was marked with a pen. The plate was incubated at 36°C until visible colonies were obtained. (ii) We pipetted 100 μl of this solution containing 100 cells on a petri dish containing the medium described above and homogenized by hand. The plate was immediately observed under a light micromanipulator microscope (×400) (Leitz MTR-27), and each isolated cell was located and marked with a circle on the petri plate (the side containing the medium), with a fine-tip marker (Sigma). The plate was incubated at 36°C until visible colonies were obtained.
Single cells (clones) were obtained in both experiments (i and ii), forming colonies only in supplemented BHI agar, as also reported by others (10, 12). The colonies were then transferred to a new petri dish containing supplemented BHI, and after 10 days they measured 1 cm in diameter. Both techniques were efficient in producing clonal cultures, but the number of colonies obtained by micromanipulation (i) was smaller than the number of colonies obtained by smearing (ii). This may have been due to the manipulation of the cell with the microneedle, which might have caused some cell damage, preventing growth. The process used in the second technique was more rapid than that used in the first because it was not necessary to manipulate the cell with the aid of a microneedle (a difficult process) but just to locate the isolated cell and to mark the site.
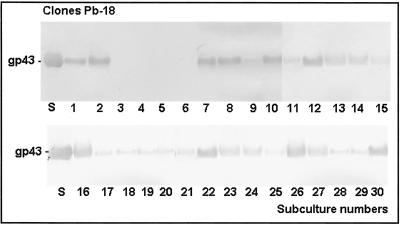
For analysis of gp43 secretion, crude exoantigens from each P. brasiliensis clone were prepared as described in a previous paper (3). Total protein concentration was determined by the method of Bradford (1) and the gp43 concentration was determined by the method of Marques-da-Silva et al. (8). The exoantigens obtained from the cloned cultures were subjected to sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (PAGE) (7) and to Western blotting as previously described (13). Briefly, the membrane was blocked with Tris-buffered saline (20 mM Tris-HCl, 150 mM NaCl, pH 7.2) containing 5% nonfat dry milk for 2 h at room temperature. The membranes were then incubated with an anti-gp43 monoclonal antibody (10 μg/ml) (monoclonal antibody was prepared by Puccia and Travassos [9] and kindly provided for use in this study) for 1 h at room temperature. Next, the membranes were washed three times with phosphate-buffered saline-Tween 20 and incubated with peroxidase-conjugated anti-mouse immunoglobulin G (Sigma) for 1 h at 37°C. The reaction was developed in a solution containing 5 mg of 3,3′diaminobenzidine (DAB; Sigma) and 10 μl of H2O2 in 50 ml of Tris buffer, pH 7.4. The membranes were washed and dried, analyzed, and stored for documentation. The results showed that all exoantigens produced by the 30-Pb113, 30-Pb339 (data not shown), and 26-Pb18 clones presented gp43 recognized by the anti-gp43 monoclonal antibody; however, four exoantigens from Pb-18 clones were not recognized by the anti-gp43 monoclonal antibody (Fig. 1), which agrees with the absence of gp43 in the exoantigens, as previously seen in the SDS-PAGE gel.
FIG. 1.
Representative Western blot reactions obtained with crude exoantigens (10 μg) from 30 clones of P. brasiliensis (Pb-18) probed with anti-gp43 monoclonal antibody (10 μg/ml); Goat anti-mouse immunoglobulin G (γ-chain specific) serum conjugated with peroxidase was used as the second antibody. Lanes 1 to 30, clones; S, standard purified gp43. Most of the exoantigens were recognized by the anti-gp43 monoclonal antibody, but four clones did not secrete gp43 and consequently were not recognized (clones 3, 4, 5, and 6 presented no gp43).
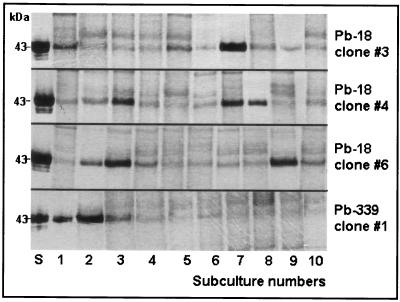
The clones that were negative for gp43 in their exoantigens were subcultured 10 times, and exoantigens were prepared and analyzed by SDS-PAGE in each subculture. Clones 3, 4, and 6 (clone 5 was not viable) from Pb-18 started to secrete gp43 again, but the amount of gp43 varied in each subculture. Pb-339 clone 1 (gp43 positive) was also subcultured 10 times and tested as clones 3, 4, and 6 from Pb-18; it was observed that the fungus was losing its capacity to secrete gp43 after the third subculture (Fig. 2 shows gp43 variability in Pb-339 clone 1, Pb-18 clone 3, Pb-18 clone 4, and Pb-18 clone 6). The intensities of gp43 bands visualized in blots for each clone were proportional to their concentration in the crude exoantigen. For example, clone 1 from Pb-339 had a broad gp43 band and 3.75 μg of gp43 per ml, and its fourth subculture had a small gp43 band and 0.75 μg of gp43 per ml.
FIG. 2.
SDS-PAGE of exoantigens (2 μg) obtained from 10 subcultures of P. brasiliensis (Pb-18) clones 3, 4, and 6 and of exoantigens (2 μg) obtained from 10 subcultures of Pb-339 clone 1. Observe the variability in gp43 expression among the subcultures. In Pb-18 clone 3, exoantigens numbers 1 and 7 secreted great amounts of gp43. In Pb-18 clone 4, exoantigens 3, 7, and 8 secreted large amounts of gp43. In Pb-18 clone 6, exoantigens 2, 3, and 9 secreted large amounts of gp43. In Pb-339 clone 1, gp43 was in abundance in exoantigens 1 and 2 but began to decrease after 3. S, standard purified gp43. Lanes 1 to 10, exoantigens of consecutive subcultures.
For analysis of possible polymorphism at the DNA level, genomic DNA was obtained from each individual yeast culture (11) and rapidly amplified polymorphic DNA analysis was performed (14) with OPG 05 (5′-CTG AGA CGG A-3′), OPG 14 (5′-GGA TGA GAC C-3′), and OPG 16 (5′-AGC GTC CTC C-3′) (Operon-Biotechnology) as primers. Rapidly amplified polymorphic DNA analysis of Pb-113 and Pb-18 clones, including the nonsecretor gp43 clones 3, 4, and 6, was performed to determine genomic DNA variations between them, and no differences were observed concerning the profiles of each isolate (data not shown).
It is our hypothesis that these P. brasiliensis isolates originated from polyclonal cultures present in nature, where humans are infected. The isolates conserved in collections are composed of polyclonal cells, and this implies that when a specific isolate is used in the laboratory, we are working with cells with different genetic information that may differ in their biological, antigenic, and biochemical properties.
A typical problem that occurs in serology laboratories concerns the antigen used for paracoccidioidomycosis diagnosis by immunodiffusion. If gp43 is absent, it is not possible to use the exoantigen preparation for diagnosis or follow-up of paracoccidioidomycosis patients under treatment. In various laboratories, this antigen is prepared with the P. brasiliensis Pb-339 isolate, which is known to produce a good antigenic preparation useful for diagnostic purposes (2, 3). This antigen consists of a concentrated filtrate of a 7-day-old culture that must contain in its composition gp43, the molecule responsible for the specific diagnostic precipitating band by immunodiffusion. However, some antigen lots obtained from Pb-339 are unable to precipitate confirmed paracoccidioidomycosis sera even when prepared in the same laboratory by the same method and by the same people. When SDS-PAGE and a blot are performed to verify the antigenic profiles of such preparations, the gp43 fraction may be absent, justifying the negativity of confirmed paracoccidioidomycosis sera. In this situation, immunodiffusion results may yield a false-negative result. Franco et al. (5) have already demonstrated that the same isolate presented variability in antigenic profile. It was our hypothesis that the Pb-339 isolate was composed of different clones, and during subculture these different clones might or might not express gp43. To obtain an antigen useful for diagnostic purposes, it was necessary to have a culture composed of cells secreting gp43. Thus, in this field of research it is necessary to work with a strain that secretes gp43, and this could be obtained by the use of a clonal culture that secretes this major antigen.
After obtaining 90 clones, we observed that all clones from Pb-113 and Pb-339 and 26 clones from Pb-18 produced gp43 in their exoantigens, but the quantity of gp43 secreted differed among them. Four clones from Pb-18 were gp43 negative, and during the next 10 subcultures, these clones started to secrete gp43 in their exoantigens again, and the amount of gp43 varied according to the subculture. On the other hand, clone 1 from Pb-339, which originally secreted a large amount of gp43, showed a reduced capacity to secrete this glycoprotein in subsequent subcultures.
On the practical side, this implies that when exoantigens are prepared and gp43 is absent or present in a small quantity, the strain is not useful to prepare antigens to be used in serological tests. Our results indicate that the problem of gp43 secretion is not related to polyclonal or monoclonal cultures since both kinds of cultures show the same behavior regarding gp43 secretion. In some occasions, we have observed in our laboratory that when exoantigens obtained from polyclonal cultures are analyzed by SDS-PAGE and immunoblot, the 43-kDa fraction may be absent, justifying the negativity of confirmed paracoccidioidomycosis sera by serological tests, making the exoantigen not useful for diagnosis purposes.
Thus, a hypothesis to explain this fact is that some problem may occur at the molecular level in the mechanism that regulates the expression of the gp43 molecule. This hypothesis is currently under study, and we hope to be able to answer this question in the future.
Acknowledgments
We thank CAPES/ Federal University of São Paulo (UNIFESP) for a fellowship to R.B. and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for financial support.
REFERENCES
- 1.Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248-254. [DOI] [PubMed] [Google Scholar]
- 2.Camargo, Z. P., C. Untekircher, S. P. Campoy, and L. R. Travassos. 1988. Production of Paracoccidioides brasiliensis exoantigens for immunodiffusion tests. J. Clin. Microbiol. 26:2147-2215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Camargo, Z. P., R. Berzaghi, C. C. Amaral, and S. H. Silva. 2003. Simplified method for producing Paracoccidioides brasiliensis exoantigens for use in immunodiffusion tests. Med. Mycol. 41:539-542. [DOI] [PubMed] [Google Scholar]
- 4.Castañeda, E., E. Brummer, Perlman. M., A J. G. McEwen, and D. A. Stevens. 1988. A culture medium for Paracoccidioides brasiliensis with high plating efficiency, and the effect of siderophores. J. Med. Vet. Mycol. 26:351-358. [PubMed] [Google Scholar]
- 5.Franco, M., E. Bagagli, M. Cunha, L. G. Chamma, and D. Fecchio. 1996. Paracoccidioides brasiliensis antigen batches from the same isolate show immunological and biochemical differences. Mycopathologia 135:13-19. [DOI] [PubMed] [Google Scholar]
- 6.Gomes, G. M., P. S. Cisalpino, C. P. Taborda, and Z. P. Camargo. 2000. PCR for diagnosis of paracoccidioidomycosis. J. Clin. Microbiol. 38:3478-3480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Laemmli, U. K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680-685. [DOI] [PubMed] [Google Scholar]
- 8.Marques-da-Silva, S. H., A. L. Colombo, L, Blotta, J. D. Lopes, Queiroz- F. Tellez, and Z. P. Camargo. 2003. Detection of circulating gp43 antigen in serum, cerebrospinal fluid and bronchoalveolar lavage fluid of patients with paracoccidioidomycosis. J. Clin. Microbiol. 41:3675-3680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Puccia, R., and L. R. Travassos. 1991. 43-Kilodalton glycoprotein from Paracoccidioides brasiliensis: immunochemical reactions with sera from patients with paracoccidioidomycosis, histoplasmosis or Jorge Lobo's disease. J. Clin. Microbiol. 29:1610-1615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Restrepo, E., and B. E. Jimenez. 1980. Growth of Paracoccidioides brasiliensis yeast phase in a chemically defined culture medium. J. Clin. Microbiol. 12:279-280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sambrook, J., E. F. Fritsch, and T. Maniatis. 1989. Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
- 12.Singer-Vermes, L. M., M. C. Ciavaglia, S. S. Kashino, E. Burger, and V. L. Calich. 1992. The source of the growth-promoting factor(s) affects the plating efficiency of Paracoccidioides brasiliensis. J. Med. Vet. Mycol. 30:261-264. [DOI] [PubMed] [Google Scholar]
- 13.Towbin, H., T. Staehelin, and J. Gordon. 1979. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. USA 76:4350-4354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Williams, J. G. K., A. R. Kubelik, K. J. Livak, J. A. Rafalski, and S. I. Tingey. 1990. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res. 18:6531-6535. [DOI] [PMC free article] [PubMed] [Google Scholar]