Abstract
Here we describe the use of the loop-mediated isothermal amplification (LAMP) method to detect human influenza viruses (H1 to H3). Our results were correlated 100% with results deduced from routine clinical diagnostic tests. In addition, we also developed a LAMP assay specific for human β-actin cDNA as a quality control test.
Influenza A viruses are medically important viral pathogens causing significant mortality, morbidity, and financial burden throughout the world. Of 15 hemagglutinin subtypes and 9 neuraminidase subtypes, only 3 hemagglutinin (H1 to H3) and 2 NA (N1 and N2) subtypes have established stable lineages in the human population since the last century (14). Due to the highly contagious nature of influenza A viruses (4), the disease could be rapidly spread in close communities. Early identification of the virus is therefore critical for the control of the disease. Several molecular techniques have been developed as rapid tests for clinical diagnosis of influenza virus (1, 10).
The recent invention of loop-mediated isothermal amplification (LAMP) provides a new alternative for molecular diagnosis (8). One of the major advantages of this technique is the simplicity of the test. In addition, it has great potential for use as a field test (2). The application of LAMP for clinical diagnosis was previously demonstrated by other studies (2, 6, 9, 15). We recently reported use of this approach for detection of the severe acute respiratory syndrome (SARS) coronavirus (11). In this study, we explore the possibilities of using this method for clinical diagnosis of human influenza A virus.
Nasopharyngeal aspirates collected from patients with respiratory illness (n = 47) or from healthy individuals (n = 5) were included in this study (Table 1). cDNAs were synthesized as previously described (12). Virological diagnosis had been previously confirmed by direct immunofluorescent antibody staining or PCR. Influenza A/WSN/33 virus (H1N1) was used as a reference virus in this study. Clinical isolates of human influenza A virus were obtained from the Department of Microbiology, Queen Mary Hospital, Hong Kong. Avian influenza viruses were isolated in routine surveillance of influenza virus in this geographical region (5).
TABLE 1.
Clinical samples used in the study
| Respiratory pathogena | No. of clinical samples | No. of positive samples in LAMP |
|---|---|---|
| Adenovirus* | 5 | 0 |
| Human metapneumovirus* | 5 | 0 |
| Influenza A virus* | 22 | 22 |
| Influenza B virus* | 2 | 0 |
| Respiratory syncytial virus** | 5 | 0 |
| SARS coronavirus** | 5 | 0 |
| Rhinovirus** | 4 | 0 |
| Healthy control | 5 | 0 |
*, diagnosed by using direct fluorescent-antibody assay; **, diagnosed by using virus-specific PCR assay.
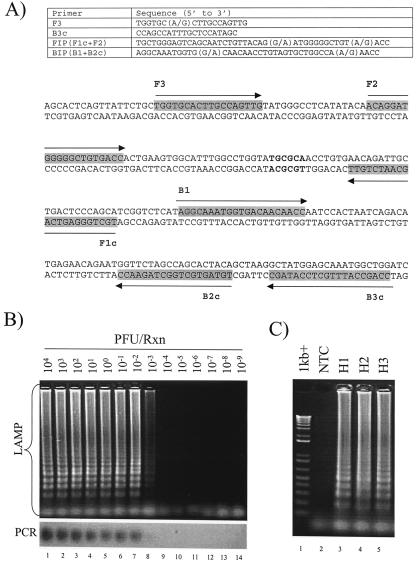
Primers were designed on the basis of sequence information obtained from the Influenza Sequence Database (http://www.flu.lanl.gov). All available human H1, H2, and H3 influenza virus sequences from the database were used in the analysis (data not shown). In conventional PCR, a pair of primers is required for DNA amplification. By contrast, two pairs of primers specific for six independent binding sites are required for LAMP (Fig. 1A and 2B). The great genetic diversity of influenza A viruses prompted us to adopt two strategies for developing a LAMP test for influenza A virus. First, we chose the most conserved viral RNA segment (the M gene) as the target gene in this study (3). Second, we used degenerate primers to target the M gene sequence (Fig. 1A).
FIG. 1.
LAMP assay for human influenza A virus. (A) Primers and representative M gene sequence used in the assay. The first and last nucleotides of the sequence are the 369th and 609th nucleotides, respectively, of the referenced sequence used in this study (GenBank accession number L25818). Locations of primer binding sequences are indicated by arrows. The FspI restriction sites are bolded. (B) Detection limits of LAMP (upper panel) and PCR (lower panel) assays. Serial diluted cDNA derived from influenza A/WSN/33 was tested by these assays. The concentrations of cDNA used in these reactions are indicated (lanes 1 to 14). (C) Detection of different human influenza A viruses. Lane 1, DNA markers; lane 2, no template control; lane 3, H1 subtype lane 4, H2 subtype; lane 5, H3 subtype.
FIG. 2.
LAMP assay for β-actin. (A) Primers and β-actin sequence used in the assay. The first and last nucleotides of the sequence are the 300th and 599th nucleotides, respectively, of the referenced sequence in this study (GenBank accession number NM_001101). Corresponding recognition sequences of the primers are indicated by arrows. The BseYI and MboI restriction sites are boldfaced and italicized, respectively. Apart from the primer sequences, the reaction condition was identical to the one for the detection of influenza viruses. (B) Detection of β-actin sequences in NPA samples. Lane 1, DNA markers; lane 2, influenza A virus; lane 3, human metapneumovirus; lane 4, respiratory syncytial virus; lane 5, adenovirus; lane 6, SARS coronavirus; lane 7, rhinovirus; lane 8, influenza B virus.
In the initial phase of the study, the detection limit of the LAMP assay was first determined by using cDNA converted from titrated influenza A/WSN/33 virus. cDNA at a concentration equivalent to 5 × 103 PFU per μl was serially diluted, and these diluted cDNA samples were subjected to the LAMP reaction. Two microliters of cDNA was amplified in a 12.5-μl reaction mixture containing 0.4 mM deoxynucleoside triphosphates, 1.6 μM (each) forward inner primer and backward inner primer, 0.2 μM (each) F3 and B3c, 8 U of Bst DNA polymerase (New England Biolabs), and 1× Bst polymerase reaction buffer. Reactions were incubated at 60°C for 2 h, followed by heat inactivation at 80°C for 5 min. As shown in Fig. 1B, a positive reaction would be shown as a ladder-like pattern in a gel electrophoresis analysis. The detection limit of the assay was found to be 10−3 PFU per reaction (Fig. 1B, upper panel). The identities of these amplified products were confirmed by FspI restriction digestion analysis (Fig. 1A and data not shown). To compare the sensitivity of the assay to that of conventional PCR, the same batch of cDNA was amplified by a pair of M gene-specific primers for 35 cycles (forward primer, 5′-GACCAATCCTGTCACCTC-3′; reverse primer, 5′-GATCTCCGTTCCCATTAAGAG-3′). The detection limit of the PCR assay was 10−2 PFU per reaction (Fig. 1B, lower panel). These observations agreed with previous findings that the sensitivities of LAMP assays are comparable to those of conventional PCR methods (2, 6, 9, 11, 15).
To evaluate the specificity of the assay, both human (two H1N1, one H2N2, and three H3N2) and avian (two H5N1, two H6N1, and one H9N2) isolates were examined by the test. Positive signals were observed with all of human isolates (Fig. 1C, lanes 3 to 5; also data not shown). By contrast, none of the avian viruses was positive in the study (data not shown). In addition, we also evaluated the test by using clinical samples. NPA from healthy individuals and retrospective nasopharyngeal aspirate (NPA) specimens with known etiological agents were tested by the assay (Table 1). Of these clinical specimens, all virologically confirmed influenza samples were positive in the assay (Table 1). No positive signal was observed for healthy individuals or for non-influenza A virus samples (Table 1). These results highlight the potential use of degenerate primers for LAMP. In addition, our data also demonstrated the potential application of LAMP for clinical diagnosis of human influenza viruses. Nonetheless, the reliability of this assay should be further evaluated by large-scale investigations and by different types of clinical specimens.
In reverse transcription-PCR assays, to monitor the quality of extracted RNA and the performance of various reverse transcription-PCR procedures, housekeeping genes or exogenous RNA molecules are frequently used as positive controls (7). In this study, we also applied this concept to the LAMP detection system. A LAMP assay specific for human β-actin mRNA was developed for this purpose (Fig. 2A). The detection limit of the assay was 10 copies per reaction (data not shown). The authenticity of these amplified products was further confirmed by BseYI and MboI restriction digestion analyses (Fig. 2A; also data not shown). In the absent of cDNA synthesis, RNA samples were negative in this test (data not shown), indicating that this β-actin assay is highly specific for cDNA of β-actin mRNA. As shown in Fig. 2B, all clinical specimens were positive in the β-actin assay (lanes 2 to 8; also data not shown), demonstrating the potential use of this assay as a quality control test.
Although the present study only concentrated on the diagnosis of human influenza viruses, we are currently modifying our assays to detect highly pathogenic avian H5 influenza viruses. In an event like the recent H5N1 outbreak (5, 13), a field test for avian H5 influenza virus would be extremely valuable for prompt identification of affected poultry or farms. Recently, by detecting the amount of magnesium pyrophosphate precipitates generated in LAMP reactions, Parida et al. (9) have demonstrated that the positivity of reactions can be determined by the naked eye. This new approach further eliminates the need of gel electrophoresis and increases the feasibility of using LAMP for field applications.
Acknowledgments
We acknowledge research funding from Public Health Research Grant A195357 from the National Institute of Allergy and Infectious Diseases and The Research Grant Council of Hong Kong (HKU 7356/03 M to L.L.M.P. and HKU 7542/03 M to J.S.M.P.).
REFERENCES
- 1.Ellis, J. S., and M. C. Zambon. 2002. Molecular diagnosis of influenza. Rev. Med. Virol. 12:375-389. [DOI] [PubMed] [Google Scholar]
- 2.Enosawa, M., S. Kageyama, K. Sawai, K. Watanabe, T. Notomi, S. Onoe, Y. Mori, and Y. Yokomizo. 2003. Use of loop-mediated isothermal amplification of the IS900 sequence for rapid detection of cultured Mycobacterium avium subsp. paratuberculosis. J. Clin. Microbiol. 41:4359-4365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fouchier, R. A. M., T. M. Bestebroer, S. Herfst, L. Van Der Kemp, G. F. Rimmelzwaan, and A. D. M. E. Osterhaus. 2000. Detection of influenza A viruses from different species by PCR amplification of conserved sequences in the matrix gene. J. Clin. Microbiol. 38:4096-4101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fraser, C., S. Riley, R. M. Anderson, and N. M. Ferguson. 2004. Factors that make an infectious disease outbreak controllable. Proc. Natl. Acad. Sci. USA 101:6146-6151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Guan, Y., L. L. Poon, C. Y. Cheung, T. M. Ellis, W. Lim, A. S. Lipatov, K. H. Chan, K. M. Sturm-Ramirez, C. L. Cheung, Y. H. Leung, K. Y. Yuen, R. G. Webster, and J. S. Peiris. 2004. H5N1 influenza: a protean pandemic threat. Proc. Natl. Acad. Sci. USA 101:8156-8161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kuboki, N., N. Inoue, T. Sakurai, F. Di Cello, D. J. Grab, H. Suzuki, C. Sugimoto, and I. Igarashi. 2003. Loop-mediated isothermal amplification for detection of African trypanosomes. J. Clin. Microbiol. 41:5517-5524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nolte, F. S. 2004. Novel internal controls for real-time PCR assays. Clin. Chem. 50:801-802. [DOI] [PubMed] [Google Scholar]
- 8.Notomi, T., H. Okayama, H. Masubuchi, T. Yonekawa, K. Watanabe, N. Amino, and T. Hase. 2000. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 28:E63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Parida, M., G. Posadas, S. Inoue, F. Hasebe, and K. Morita. 2004. Real-time reverse transcription loop-mediated isothermal amplification for rapid detection of West Nile virus. J. Clin. Microbiol. 42:257-263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Playford, E. G., and D. E. Dwyer. 2002. Laboratory diagnosis of influenza virus infection. Pathology 34:115-125. [DOI] [PubMed] [Google Scholar]
- 11.Poon, L. L., C. S. Leung, M. Tashiro, K. H. Chan, B. W. Wong, K. Y. Yuen, Y. Guan, and J. S. Peiris. 2004. Rapid detection of the severe acute respiratory syndrome (SARS) coronavirus by a loop-mediated isothermal amplification assay. Clin. Chem. 50:1050-1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Poon, L. L., O. K. Wong, K. H. Chan, W. Luk, K. Y. Yuen, J. S. Peiris, and Y. Guan. 2003. Rapid diagnosis of a coronavirus associated with severe acute respiratory syndrome (SARS). Clin. Chem. 49:953-955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tran, T. H., T. L. Nguyen, T. D. Nguyen, T. S. Luong, P. M. Pham, V. C. Nguyen, T. S. Pham, C. D. Vo, T. Q. Le, T. T. Ngo, B. K. Dao, P. P. Le, T. T. Nguyen, T. L. Hoang, V. T. Cao, T. G. Le, D. T. Nguyen, H. N. Le, K. T. Nguyen, H. S. Le, V. T. Le, D. Christiane, T. T. Tran, M. de Jong, C. Schultsz, P. Cheng, W. Lim, P. Horby, J. Farrar, and the World Health Organization International Avian Influenza Investigative Team. 2004. Avian influenza A (H5N1) in 10 patients in Vietnam. N. Engl. J. Med. 350:1179-1188. [DOI] [PubMed] [Google Scholar]
- 14.Webby, R. J., and R. G. Webster. 2003. Are we ready for pandemic influenza? Science 302:1519-1522. [DOI] [PubMed] [Google Scholar]
- 15.Yoshikawa, T., M. Ihira, S. Akimoto, C. Usui, F. Miyake, S. Suga, Y. Enomoto, R. Suzuki, Y. Nishiyama, and Y. Asano. 2004. Detection of human herpesvirus 7 DNA by loop-mediated isothermal amplification. J. Clin. Microbiol. 42:1348-1352. [DOI] [PMC free article] [PubMed] [Google Scholar]