Abstract
Paragangliomas of the organ of Zuckerkandl (OZ-PGL) are rare tumors that, in >70% of cases, occur in association with succinate dehydrogenase complex iron sulfur subunit B (SDHB) or SDHD gene mutations. The aim of the current study was to determine whether a somatic genetic defect in the hypoxia-inducible factor 2α (HIF2α) gene was present in a case of sporadic OZ-PGL. A 32-year-old African female presented with uncontrolled hypertension during the first trimester of pregnancy. A diagnostic hysteroscopy was performed 3 months after delivery, precipitating a hypertensive crisis. Thereafter, the patient was diagnosed with noradrenaline-secreting OZ-PGL. A complete blood count identified mild normocytic anemia of an inflammatory origin. Surgical removal of the tumor resulted in normalization of plasma and urinary normetanephrine levels. Genetic testing for germline mutations (including large deletions) in the von Hippel-Lindau tumor suppressor, SDHB, SDHC and SDHD genes was normal. However, a heterozygous missense mutation (c.1589Cys>Tyr) was detected in exon 12 of HIF2α, which results in a substitution of alanine 530 with valine (Ala530Val) in the HIF2α protein. A germline mutation was excluded based on the negative results of blood DNA testing. A three-dimensional homology model of Ala530Val was constructed, which showed impaired HIF2α/VHL interaction and decreased HIF2α ubiquitination. 1H-high-resolution magic-angle-spinning nuclear magnetic resonance spectroscopy detected low succinate levels and high α and β glucose levels. To the best of our knowledge, the present case represents the first of its kind to associate a somatic HIF2α gain-of-function mutation with OZ-PGL. It is therefore recommended that patients without germline SDHx mutations should be tested for HIF2α mutations.
Keywords: paraganglioma, nuclear magnetic resonance spectroscopy, endothelial PAS domain-containing protein 1
Introduction
In 1901, Emile Zuckerkandl first described the abdominal para-aortic paraganglia in fetal and newborn humans as a paired retroperitoneal organ located laterally to the abdominal aorta at the level of the inferior mesenteric aorta (1). This paraganglionic complex, known as the organ of Zuckerkandl (OZ), also includes smaller accessory paraganglia located anteriorly to the aorta between the lateral organs or below the aortic bifurcation (2). In 1903, Alfred Kohn established that the OZ commonly originated from chromaffin cells of the adrenal medulla (3), and it has later been established that it constitutes the largest accumulation of extradrenal chromaffin cells in mammals. In humans, the OZ reaches its maximal size at the age of ~3 years and subsequently regresses after reaching its peak by autophagy (4). The OZ is considered to be most important physiologically throughout the early gestational period, during which it secretes catecholamines into the fetal circulation, functioning as a homeostatic regulator of blood pressure (5). The OZ represents a site of origin for paragangliomas (PGLs) that preferentially secrete norepinephrine and induce symptoms of catecholamine excess (6). OZ-PGLs are rare tumors typically located close to the origin of the inferior mesenteric artery or between the proximal common iliac arteries (1). These lesions may occur sporadically or, in ~70% of cases, in association with succinate dehydrogenase complex iron sulfur subunit B (SDHB) or, less commonly, SDHD gene mutations (7). In addition, OZ-PGLs are particularly aggressive with high rates of metastatic spread (8). At least 150 cases of OZ-PGLs have been reported in the literature. They are strongly associated with an aggressive behavior, likely associated with the SDHB mutation status (7). Due to the rarity of this disease, not much is known about its natural history. A single-center retrospective study of 371 patients with either pheochromocytoma or sympathetic paraganglioma revealed only 21 cases of OZ-PGLs, 14 of which (66%) had metastases at diagnosis (9). To the best of our knowledge, the current case demonstrates that somatic HIF2α [also known as endothelial PAS domain-containing protein 1 (EPAS1)] mutations may be associated with OZ-PGL for the first time.
Case report
In September 2014, a 32-year-old African woman native to Burkina Faso was referred to the hypertension unit of La Timone University Hospital (Marseille, France) for screening for secondary hypertension. Hypertension was initially noted during the first trimester of pregnancy. The patient went into premature labor at 22 weeks and a cesarean delivery was performed 15 days later; the baby did not survive and succumbed a few minutes after birth. Following delivery, the patient experienced persistent and uncontrolled hypertension despite taking nicardipine (60 mg/day) and labetalol (400 mg/day) for 3 months. A diagnostic hysteroscopy was performed 3 months later, precipitating a hypertensive crisis [systolic blood pressure (BP), 300 mmHg; normal, <140 mmHg]. Thereafter, the patient was referred to the hypertension unit of La Timone University Hospital for secondary hypertension screening in September 2014. There was no known family history of tumors, syncope or sudden death. At admission (weight, 51 kg; height, 163 cm; and body mass index, 19.2), the patient presented with headaches, recurring episodes of palpitations and sweating, chest tightness, and polyuria. Treatment with nicardipine and labetalol was replaced with verapamil (240 mg/day). Ambulatory 24-h BP monitoring was performed during treatment with verapamil and demonstrated that the patient maintained a BP of 155/96 mmHg. Prazosin (2.5 mg once per day) was subsequently administered to reduce blood pressure further until surgical intervention.
Additional laboratory tests identified highly elevated 24 h urinary normetanephrine levels [20,140 nmol/24 h; upper reference limit (URL), <1900 nmol/24 h] and normal metanephrine levels (380 nmol/24 h; URL, <1600 nmol/24 h). In addition, serum chromogranin A was observed to be elevated (223 µg/l; URL, <100 µg/l). A complete blood count revealed mild normocytic anemia (hemoglobin count, 108.0 g/l; normal hemoglobin count for female adults, 11.5–15.0 g/dl; mean corpuscular volume, 83.4 fl; normal mean corpuscular volume of adults, 80–100 fl) of an inflammatory origin with normal platelets and leukocytes. Diagnostic computed tomography (CT) revealed a 40-mm hypervascular, heterogeneous, left para-aortic mass located at the level of the inferior mesenteric artery (Fig. 1A). 18Fluorine-L-dihydroxyphenylalanine positron emission tomography/CT (Fig. 1B) and iodine-123-metaiodobenzylguanidine scintigraphy (Fig. 1C) confirmed the diagnosis of OZ-PGL without multifocal disease. The tumor also exhibited moderate heterogeneous 18F-fluorodeoxyglucose uptake (Fig. 1B). In October 2014, complete surgical resection was performed. Histopathological analysis of the tumor tissue revealed typical PGL features, including a low Ki-67 index (<1%) (monoclonal mouse antibody; clone, MIB-1; catalogue no., M7240; dilution, 1:100: Dako, Glostrup, Denmark). Genetic testing for germline mutations (including large deletions) in the von Hippel-Lindau tumor suppressor (VHL), succinate dehydrogenase complex iron sulfur subunit B (SDHB), SDHC and SDHD genes was normal. Immunostaining demonstrated that the tumor cells were positive for SDHB. Further genetic testing revealed a heterozygous cysteine to tyrosine substitution at base 1589 (c.1589Cys>Tyr) in the HIF2α coding sequence of the OZ-PGL, resulting in the replacement of alanine with valine at amino acid position 530 (Ala530Val). This leads to HIF2α stabilization as described by a previous in vitro experiment (10). A germline HIF2α mutation was excluded based on the negative results of blood DNA testing.
Figure 1.
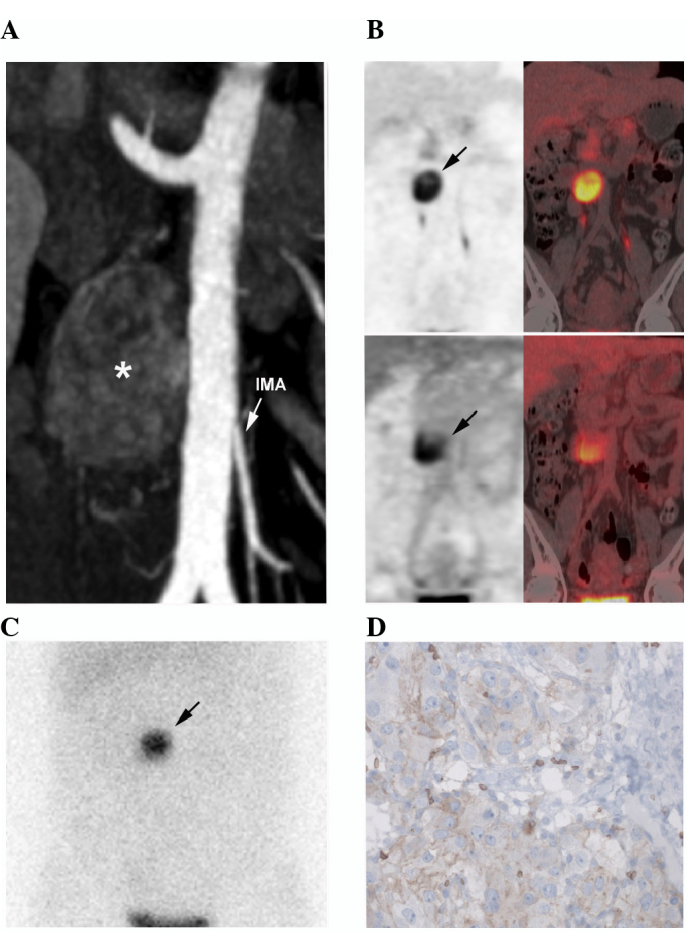
Imaging and pathological features of the OZ-PGL. (A) Contrast-enhanced CT (arterial phase) showing a 40-mm hypervascular and heterogeneous left para-aortic mass located at the level of the IMA (asterisk). (B) 18F-FDOPA (upper image) and 18F-FDG PET/CT (lower image) imaging showing a single tumor. (C) Iodine-123-metaiodobenzylguanidine scintigraphy also positively located the mass (planar anterior view). (D) Immunohistochemical analysis of the tumor demonstrated positive glucose transporter-1 immunostaining (~10%). CT, computed tomography; IMA, inferior mesenteric artery; 18F-FDOPA, 18fluorine-L-dihydroxyphenylalanine; 18F-FDG, 18F-fluorodeoxyglucose; PET, positron emission tomography.
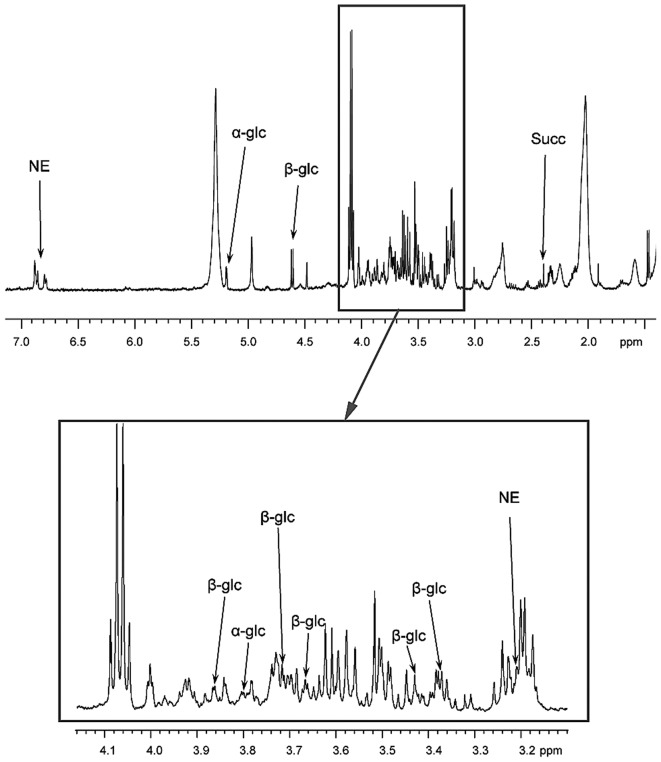
In order to assess the metabolic properties of the tumor, the present study performed 1H-high-resolution magic-angle-spinning (HRMAS) nuclear magnetic resonance spectroscopy-based global metabolomic profiling on tumor samples. A one-dimensional proton spectrum (1.5–7.2 ppm) using a Carr-Purcell-Meiboom-Gill pulse sequence with water presaturation was acquired from each intact tissue sample Low levels of succinate were detected, and according to our previous study (11) this excludes a SDH deficiency. Notably, the tumor also exhibited abnormally high levels of α- and β-glucose isomers as identified by HRMAS (Fig. 2). The patient is currently in remission, and regular clinical follow up occured every 6 months with normal metanephrines. Conventional radiological imaging (MRI) and functional imaging (PET-FDOPA) at 1 year post-intervention were also normal.
Figure 2.
Results of HRMAS NMR spectroscopy (500 MHz) performed on tumor samples from the organ of Zuckerkandl paraganglioma. Partial metabolite assignment is indicated. The metabolic content may be directly compared as the spectrum intensity was normalized with respect to the weight of each examined sample. For display purposes, the amplitude of the lactate peak at 4.09 ppm has been cut out. The top image shows a representative spectrum with highly elevated levels of α-glc and β-glc. Spectrum regions ranging from 3.10–4.15 ppm are magnified in the lower box. Amongst catecholamines, only an NE signal was detected in all the examined tissue samples. Finally, the level of Succ was low, which confirmed the absence of a succinate dehydrogenase complex deficiency. HRMAS, 1H-high-resolution magic-angle-spinning; NMR, nuclear magnetic resonance; α-glc, α-glucose; β-glc, β-glucose; NE, norepinephrine; Succ, succinate.
Written informed consent was obtained from the patient for publication of the present case report and any accompanying images.
Discussion
To the best of our knowledge, the present case demonstrates, for the first time, that patients with somatic HIF2α mutations may present with OZ-PGL.
Germline mutations in the HIF2α/EPAS1 gene have been previously associated with congenital polycythemia (12). A syndromic association has been reported between somatic gain-of-function mutations in HIF2α and congenital polycythemia, multiple PGL, duodenal somatostatinoma and ocular vascular abnormalities (for example, Pacak-Zhuang syndrome) (10,13–16). Mutations in HIF2α have also been observed in apparently sporadic pheochromocytomas (PHEOs)/PGLs without polycythemia (17–19). In one study, mutations (exon 12) were identified in 2 cases of solitary PHEO and 1 para-adrenal PGL (18). In an additional study, 6/42 cases of apparently sporadic PHEOs were identified to have HIF2α mutations (3 in exon 9 and 3 in exon 12) (17). HIF2α protein stability is dependent on the hydroxylation of two specific proline residues (Pro405 and Pro531) located in the O2-dependent degradation domain (10). Until present, all mutations described were known to be located in hot spots adjacent to hydroxylation sites (16). These specific mutations disturb HIF2α prolyl hydroxylation and subsequent recognition by the VHL protein, resulting in the failure of HIF2α degradation via ubiquitination (16). As mutant HIF2α protein has a longer half-life compared with the wild-type protein, it has a targeted effect downstream of HIF2α (10).
The mutation identified in the present study had previously been reported in a case of apparently sporadic PHEO/PGL (18). The mutation involved Ala530, which is located in close proximity to the second hydroxylation site (Pro531) and at the interface with VHL and Egl-9 family hypoxia-inducible factor 1 (EGLN1) client-proteins. Homology modeling was performed to outline the biological properties of the Val530 mutant (Fig. 3). These three-dimensional models were generated with IBM SPSS Modeler v14 (IBM SPSS, Armonk, NY, USA) using the crystal structures of HIF1α in interaction with EGLN1 or VHL as templates. HIF1α and HIF2α exhibit a sequence identity of 65% in the region modeled, which guarantees (>50% identity) that the models are of a high quality. The model anticipates that valine, a larger residue than alanine, increases steric hindrance at Pro531, leading to: i) A reduction in its accessibility to EGLN1 by inhibition of Pro531 hydroxylation; and ii) impairment of HIF2α/VHL interaction with decreased HIF2α ubiquitination. The present study also identified a novel metabolomic pattern with low succinate and high glucose levels associated with HIF2α mutation. Abnormally high levels of glucose may be explained by increased glucose uptake induced by HIF2α stabilization (20).
Figure 3.
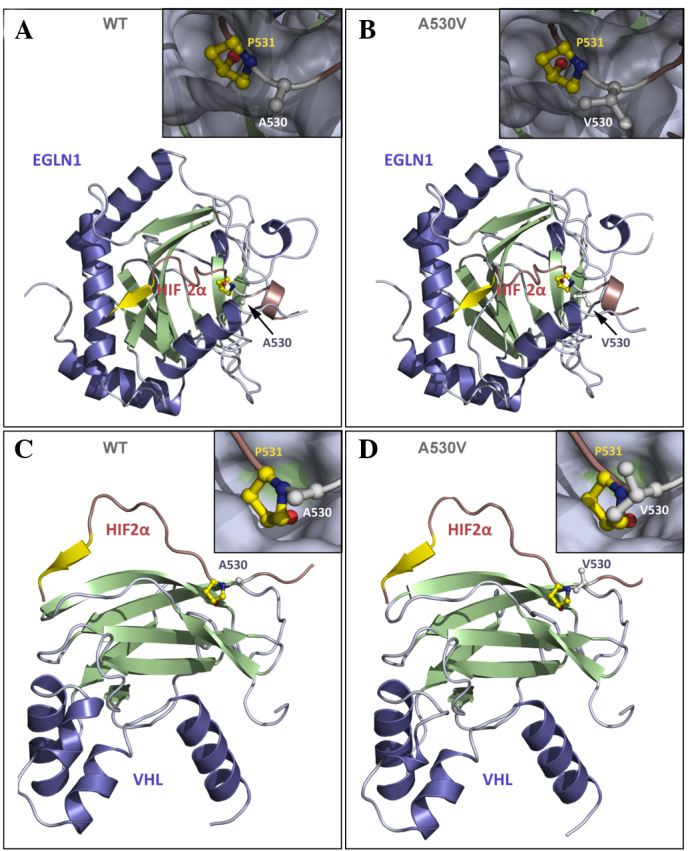
Representation of human HIF2α in the presence of its binding partners EGLN1 and VHL. (A) WT HIF2α (A560) interacting with EGLN1; (B) mutant Val560 interacting with EGLN1; (C) WT HIF2α interacting with VHL; and (D) mutant Val560 interacting with VHL. HIF2α (16 residues) is represented in red and yellow, and the interactive partners (EGLN1 or VHL) are represented in blue and green. (B and D) The inserts present a closer view of Pro531 and Ala530 (or Val530) from HIF2α in the ball-and-stick representation to show the atomic details, while HIF-2α partners, (A and B) EGLN1 or (C and D) VHL are shown as a grey surface showing that residues 530 and 531 bind to small pockets at the surface of the protein partner. Residue Ala530 is located in close proximity to residue Pro531, which is hydroxylated by EGLN1 and at the interface with the binding partners EGLN1 and VHL. Hydroxylation of Pro531 is required for interaction with VHL. It is anticipated that valine, which is a larger residue than alanine, increases steric hindrance at Pro531, resulting in a reduction in its accessibility to EGLN1 by inhibition of Pro531 hydroxylation; therefore, interaction with VHL and subsequent ubiquitination is prevented. Panel D is presented as a model, but VHL interaction should not occur in the Val530 mutant. HIF2α, hypoxia inducible factor 2α; EGLN1, Egl-9 family hypoxia-inducible factor 1; VHL, von Hippel-Lindau tumor suppressor; WT, wild-type.
In conclusion, to the best of our knowledge, the current study identified, for the first time, an association between somatic HIF2α mutations and OZ-PGL. It is therefore recommended that patients with OZ-PGL in the absence of germline SDHx mutations should undergo testing for HIF2α mutations.
References
- 1.Zuckerkandl E. About sympathetic paraganglions in the retroperitoneal space of man. Verh Anat Ges. 1901;15:95–107. (In German) [Google Scholar]
- 2.Unsicker K, Huber K, Schütz G, Kalcheim C. The chromaffin cell and its development. Neurochem Res. 2005;30:921–925. doi: 10.1007/s11064-005-6966-5. [DOI] [PubMed] [Google Scholar]
- 3.Kohn A. The paraganglia. Arch Mikrosk Anat. 1903;52:262–265. (In German) [Google Scholar]
- 4.Schober A, Parlato R, Huber K, Kinscherf R, Hartleben B, Huber TB, Schütz G, Unsicker K. Cell loss and autophagy in the extra-adrenal chromaffin organ of Zuckerkandl are regulated by glucocorticoid signalling. J Neuroendocrinol. 2013;25:34–47. doi: 10.1111/j.1365-2826.2012.02367.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.West GB, Shepherd DM, Hunter RB, McGregor AR. The function of the organs of Zuckerkandl. Clin Sci. 1953;12:317–325. [PubMed] [Google Scholar]
- 6.Martucci VL, Pacak K. Pheochromocytoma and paraganglioma: Diagnosis, genetics, management, and treatment. Curr Probl Cancer. 2014;38:7–41. doi: 10.1016/j.currproblcancer.2014.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lodish MB, Adams KT, Huynh TT, Prodanov T, Ling A, Chen C, Shusterman S, Jimenez C, Merino M, Hughes M, et al. Succinate dehydrogenase gene mutations are strongly associated with paraganglioma of the organ of Zuckerkandl. Endocr Relat Cancer. 2010;17:581–588. doi: 10.1677/ERC-10-0004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Subramanian A, Maker VK. Organs of Zuckerkandl: Their surgical significance and a review of a century of literature. Am J Surg. 2006;192:224–234. doi: 10.1016/j.amjsurg.2006.02.018. [DOI] [PubMed] [Google Scholar]
- 9.Ayala-Ramirez M, Feng L, Johnson MM, Ejaz S, Habra MA, Rich T, Busaidy N, Cote GJ, Perrier N, Phan A, et al. Clinical risk factors for malignancy and overall survival in patients with pheochromocytomas and sympathetic paragangliomas: Primary tumor size and primary tumor location as prognostic indicators. J Clin Endocrinol Metab. 2011;96:717–725. doi: 10.1210/jc.2010-1946. [DOI] [PubMed] [Google Scholar]
- 10.Zhuang Z, Yang C, Lorenzo F, Merino M, Fojo T, Kebebew E, Popovic V, Stratakis CA, Prchal JT, Pacak K. Somatic HIF2A gain-of-function mutations in paraganglioma with polycythemia. N Engl J Med. 2012;367:922–930. doi: 10.1056/NEJMoa1205119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Imperiale A, Moussallieh FM, Sebag F, Brunaud L, Barlier A, Elbayed K, Bachellier P, Goichot B, Pacak K, Namer IJ, Taïeb D. A new specific succinate-glutamate metabolomic hallmark in SDHx-related paragangliomas. PLoS One. 2013;8:e80539. doi: 10.1371/journal.pone.0080539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Percy MJ, Furlow PW, Lucas GS, Li X, Lappin TR, McMullin MF, Lee FS. A gain-of-function mutation in the HIF2A gene in familial erythrocytosis. N Engl J Med. 2008;358:162–168. doi: 10.1056/NEJMoa073123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Taïeb D, Yang C, Delenne B, Zhuang Z, Barlier A, Sebag F, Pacak K. First report of bilateral pheochromocytoma in the clinical spectrum of HIF2A-related polycythemia-paraganglioma syndrome. J Clin Endocrinol Metab. 2013;98:E908–E913. doi: 10.1210/jc.2013-1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yang C, Sun MG, Matro J, Huynh TT, Rahimpour S, Prchal JT, Lechan R, Lonser R, Pacak K, Zhuang Z. Novel HIF2A mutations disrupt oxygen sensing, leading to polycythemia, paragangliomas, and somatostatinomas. Blood. 2013;121:2563–2566. doi: 10.1182/blood-2012-10-460972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pacak K, Chew EY, Pappo AS, Yang C, Lorenzo FR, Wilson MW, Aronow MB, Young JA, Popovic V, Zhuang Z. Ocular manifestations of hypoxia-inducible factor-2α paraganglioma-somatostatinoma-polycythemia syndrome. Ophthalmology. 2014;121:2291–2293. doi: 10.1016/j.ophtha.2014.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pacak K, Jochmanova I, Prodanov T, Yang C, Merino MJ, Fojo T, Prchal JT, Tischler AS, Lechan RM, Zhuang Z. New syndrome of paraganglioma and somatostatinoma associated with polycythemia. J Clin Oncol. 2013;31:1690–1698. doi: 10.1200/JCO.2012.47.1912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Welander J, Andreasson A, Brauckhoff M, Bäckdahl M, Larsson C, Gimm O, Söderkvist P. Frequent EPAS1/HIF2α exons 9 and 12 mutations in non-familial pheochromocytoma. Endocr Relat Cancer. 2014;21:495–504. doi: 10.1530/ERC-13-0384. [DOI] [PubMed] [Google Scholar]
- 18.Comino-Méndez I, de Cubas AA, Bernal C, Álvarez-Escolá C, Sánchez-Malo C, Ramírez-Tortosa CL, Pedrinaci S, Rapizzi E, Ercolino T, Bernini G, et al. Tumoral EPAS1 (HIF2A) mutations explain sporadic pheochromocytoma and paraganglioma in the absence of erythrocytosis. Hum Mol Genet. 2013;22:2169–2176. doi: 10.1093/hmg/ddt069. [DOI] [PubMed] [Google Scholar]
- 19.Favier J, Buffet A, Gimenez-Roqueplo AP. HIF2A mutations in paraganglioma with polycythemia. N Engl J Med. 2012;367:2161–2162. doi: 10.1056/NEJMc1211953. [DOI] [PubMed] [Google Scholar]
- 20.Keith B, Johnson RS, Simon MC. HIF1α and HIF2α: Sibling rivalry in hypoxic tumour growth and progression. Nat Rev Cancer. 2011;12:9–22. doi: 10.1038/nrc3183. [DOI] [PMC free article] [PubMed] [Google Scholar]