Abstract
The Multiple Sclerosis Outcome Assessments Consortium (MSOAC) includes representatives from advocacy organizations, Food and Drug Administration (FDA), European Medicines Agency (EMA), National Institute of Neurological Disorders and Stroke (NINDS), academic institutions, and industry partners along with persons living with multiple sclerosis (MS). One of the MSOAC goals is acceptance and qualification by regulators of performance outcomes that are highly reliable and valid, practical, cost-effective, and meaningful in MS. This article addresses the history, application, and psychometric properties of one such MSOAC metric of ambulation or walking namely, the timed 25-foot walk (T25FW). The T25FW has strong reliability over both brief and long periods of time in MS across a large range of disability levels. The outcome of walking speed from the T25FW has obvious real-world relevance and has correlated strongly with other measures of walking and lower extremity function. The T25FW is responsive for capturing intervention effects in pharmacological and rehabilitation trials and has an established value for capturing clinically meaningful change in ambulation. Directions for future research involve validating clinically meaningful improvements on the T25FW as well as determining whether 20% change is clinically meaningful across the disability spectrum. Researchers might further consider synchronizing accelerometers and motion sensors with the T25FW for capturing walking speed in everyday life and the patient’s real environment.
Keywords: Ambulation, walking, gait, multiple sclerosis, validity, reliability
Overview and historical perspective
Walking is defined as advancing or traveling on foot such that there is always one foot on the ground in bipedal locomotion. Walking has historical and clinical underpinnings as well as patient centrality and importance in multiple sclerosis (MS). Walking dysfunction was recognized as a cardinal feature of MS in the earliest historical accounts of the disease1 and currently represents a primary construct for monitoring patients with MS in clinical research and practice.2 Of note, walking is one of the most important and valued functions for patients with MS,3,4 and its dysfunction represents a primary burdensome feature of the disease for quality of life and participation.4,5 Such observations underscore the importance of walking as an outcome in clinical research and practice involving MS patients.
Walking can be readily measured in MS. The Expanded Disability Status Scale (EDSS),6 which is the most common scale to measure disability in MS, classifies walking or ambulatory dysfunction based on EDSS scores of 4.0 or greater (e.g. able to walk 500 vs 300 m without aid or rest differentiates a 4.0 and 4.5, respectively, on the EDSS). To that end, scores above 4.0 on the EDSS are primarily based on gait dysfunction, particularly scores of 6.0–7.5. This makes the EDSS and 500-m walk a poor choice for measuring ambulation in clinical research and practice at earlier stages of MS, and the EDSS has well-recognized limitations related to reliability and sensitivity.2,7
The timed 25-foot walk (T25FW) was first seen in the Ambulatory Index (AI).8 The AI is a rating scale that assesses mobility based on time and degree of assistance required when walking 25 feet as quickly as possible but safely. The AI is scored on a 10-point scale with anchors of asymptomatic and fully active (0) through bedridden (10). The AI captured the effect of intensive cyclophosphamide–adrenocorticotropic hormone (ACTH) treatment in progressive MS,8 and this provided an initial platform for including the T25FW as a measure of ambulation for clinical research and practice of MS. The expression of T25FW performance was based on an ordinal scale, and presented a possibility of reduced precision for capturing small changes in ambulation compared with its expression as a continuous outcome.9
The T25FW was later integrated along with the paced auditory serial addition test (PASAT)10 and 9-hole peg test (9-HPT)11 into the original version of the multiple sclerosis functional composite (MSFC).12 This three-part, standardized assessment was developed for clinical trials in MS by a task force on clinical outcomes assessment by the National Multiple Sclerosis Society (NMSS) and provided a multidimensional outcome that (a) reflects the varied clinical expression of MS, (b) contains dimensions that can change independently over time, and (c) measures cognitive function as well as leg function/ambulation and arm/hand function. The MSFC has been included in a large number of clinical trials but has been criticized based on its (a) expression as a z-score that is not intuitive for interpretation and (b) dependence on a reference population for z-score calculation.2 The MSFC further has not been accepted by regulators as a primary endpoint in clinical trials.
There has since been increasing interest in the application of the T25FW alone in MS. For example, the T25FW was the primary endpoint in a Phase II trial of extended release, oral dalfampridine (4-aminopyridine) for improving walking in MS.13 This application of the T25FW as a primary endpoint has been repeated in a subsequent Phase III trial of extended release, oral dalfampridine in MS,14 and the T25FW represents a primary outcome for trials of rehabilitation interventions5 including exercise training.15
Review objective
The T25FW is considered the “best characterized objective measure of walking disability and can be used across a wide range of walking disabilities” in MS,16 based on its ease of administration; application among a wide range of patients (e.g. disability level); and evidence for reliability, validity, responsiveness, and clinical meaningfulness of scores in MS.16 Indeed, there is a robust literature on the psychometric characteristics of the T25FW in MS (e.g. reliability and validity of scores), as well as standardization of its administration and scoring. Such features collectively justify its role in the Multiple Sclerosis Outcome Assessments Consortium (MSOAC), an effort to adopt a clinical outcome assessment tool for clinical trials to better capture MS-related disability.17 The MSOAC was born out of a consensus paper by the International Advisory Committee on Clinical Trials in Multiple Sclerosis. The goals of the consortium are acceptance and qualification by regulators of performance outcomes that reflect core MS impairments that are highly reliable and valid, practical, cost-effective, and meaningful to persons with MS. This review benefitted from a formal MSOAC-sponsored literature search, conducted in Embase, MEDLINE, PsycInfo, and Cumulative Index of Nursing and Allied Health Literature, followed by an enrichment technique (key papers identified by MSOAC members added and informed search criteria) including work identified from prior reviews. Similar to the companion reviews of the symbol digit modalities test, 9-HPT, and low contrast letter acuity, this article begins with psychometric validity and concludes with an appraisal of the clinical meaningfulness of the measure. This paper includes sections on the (a) description, administration, and scoring of the T25FW; (b) reliability and precision of its scores over time; (c) content and construct validity of the T25FW and its scores; and (d) responsiveness of the T25FW and clinical meaningfulness of changes in its scores.
Description, administration, and scoring
Of considerable importance, there is a standardized protocol for administering and scoring the T25FW.18 Standardization is critical for consistent use of the instrument across raters and practice sites, allowing for comparison across studies and pooling of the results. The standardized protocol is quite simple. The subject is instructed to walk as fast and safely as possible (i.e. maximal walking speed) across a clearly marked, linear 25-foot or 7.62-m course (Figure 1). There are no turns in the course, and the T25FW starts with a static start (i.e. standing upright and still). The subject may use an assistive device. The subject is timed walking the 25-foot course twice, and T25FW score is the average in seconds of the two successive trials. Per T25FW trial, the stopwatch is started when the subject begins walking (i.e. foot leaves the ground) and stopped when the subject crosses the line marking 25 feet (i.e. foot breaks the plane of the clearly marked course); the subject is instructed to walk past the finish line before slowing and stopping. The T25FW can be expressed in speed by dividing 25 feet by time in seconds (i.e. feet/second or velocity). The T25FW does not measure gait quality, although velocity has its underpinnings in spatial and temporal gait parameters.5 The T25FW is practical, requires minimal equipment (i.e. stopwatch, clipboard, and recording form), and minimal space (i.e. 25-foot, unobstructed hallway). Collectively, this makes the T25FW a highly attractive, easily administered, and inexpensive outcome for clinical research and practice in MS.
Figure 1.
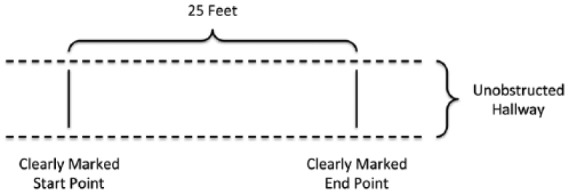
Diagram of the T25FW field. The person starts by standing behind the line on the left of the field. The person walks across the 25-foot field that is clearly marked with start and end points as quickly and safely as possible. The time in seconds is recorded when the person lifts one foot for starting the T25FW and ends upon breaking the plane of the end point with a foot. The test is performed twice, and time is averaged between trials in seconds.
Psychometric validity
Reliability and precision of scores
Reliability provides an indication of a measure’s consistency and precision over time in the absence of a change and is often based on the intraclass correlation (ICC) coefficient (i.e. test–retest reliability), standard error of measurement (SEM), and coefficient of variation (COV). One study reported strong reliability of T25FW scores across a 1- to 2-hour period in a sample of 151 MS patients (EDSS between 0 and 6.5) based on an ICC of 0.96.19 Another study reported an ICC of 0.99 for T25FW scores over a 1-week period in a combined sample of 10 MS patients (EDSS of 1.0–5.0) and 10 healthy controls.20 T25FW scores have been reliable over a 1-week time period in those with MS who have EDSS scores between 5 and 6.5 based on an ICC of 0.94 and SEM of 4.56 seconds.21
Another study provided a comprehensive examination of the reliability of T25FW scores over a 6-month period without intervention and compared its reliability with other ambulatory measures in 82 persons with MS.20 Such an analysis is important for understanding the stability of the measure and walking itself over time. T25FW scores were reliable over 6 months based on an ICC of 0.99, and this was stronger than the reliability for the 6-minute walk (ICC = 0.96), Multiple Sclerosis Walking Scale-12 (MSWS-12; ICC = 0.93), and accelerometry (steps/day ICC = 0.91, counts/day ICC = 0.88).22 Of note, the SEM was smaller for T25FW scores (SEM = 1.0) than other ambulatory measures (e.g. MSWS-12 = 8), as was the COV for T25FW scores (COV = 6.2%) compared against other measures (e.g. MSWS-12 = 27%). The ICC, SEM, and COV for T25FW scores were comparable between samples differentiated by EDSS levels (i.e. 0–3.5 and 4–6.5).22
Collectively, the data indicate that the T25FW has adequate reliability over both brief and long periods of time in MS across a large range of disability levels, and this is comparable (e.g. 6 MW), and in some cases better (e.g. accelerometry or MSWS-12) than other walking outcomes.
Content validity
This type of validity involves evidence that the T25FW includes content (i.e. its outcome) that has relevance and representativeness for ambulation, particularly during everyday life.23 To this end, the T25FW does have content validity given that most periods of walking in life are very short and can involve tasks that require maximal walking speed. For example, maximal walking speed matters for tasks such as crossing the street safely, before a traffic light changes colors and traffic begins moving again, answering the telephone before the caller disconnects, or getting to the bathroom quickly to avoid urge incontinence. These tasks are performed over a short walking distance. Walking speed might be important in job situations requiring moving quickly between meetings in different locations. Overall, the outcome of walking speed from the T25FW has obvious real-world relevance, enhancing the potential for supportive research pertaining to other forms of validity.23–25
Criterion validity, known-groups evidence
The most common approach for establishing the validity of scores on the T25FW within this category involves comparing MS patients with healthy controls;26–28 one would expect that persons with MS would walk slower than healthy controls without MS. To this end, one study compared T25FW performance between a sample of 141 patients with MS and 104 age- and sex-matched healthy controls and reported a statistically significant difference in the median scores of 4.4 and 3.7 seconds for MS patients and controls, respectively.27 Another study included 31 persons with MS and 31 matched controls and reported a statistically significant difference in the mean T25FW scores of 4.8 and 3.7 seconds for MS and controls, respectively; this reflected an effect size of 0.67 standardized units for a moderately longer or slower T25FW in MS than controls.28 These statistically significant differences were replicated in another study of 275 MS patients and 109 healthy controls, whereby the mean scores were 9.2 and 4.4, respectively.26 This later study likely reported a larger difference in T25FW scores between MS and controls than previous research27 based on the broader range of disability (i.e. EDSS of 0–7.5 vs 0–5.5).
Another approach for establishing the construct validity of scores on the T25FW within this category involves comparing performance across disability status among MS patients;27,29,30 one would expect that persons with MS who have severe neurological disability based on EDSS scores would walk slower on the T25FW than those with mild or moderate disability. Accordingly, one study compared T25FW performance across mild (EDSS of 0–2.0), moderate (EDSS of 2.5–3.5), and severe (EDSS of 4–5.5) levels of disability in a sample of 141 patients with MS and reported a statistically significant difference in the median scores of 3.9, 4.5, and 5.8 seconds across those three levels of disability, respectively.29 Another study included 96 persons with MS who were stratified across mild (EDSS of 0–3.0), moderate (EDSS of 4.0–5.5), and severe (EDSS of 6.0–6.5) levels of disability and reported a statistically significant difference in the mean scores of 4.8, 6.3, and 9.0 seconds across those three levels of disability.30 The effect sizes between mild and moderate and moderate and severe groups were 1.04 and 1.02 standardized units.
One final approach is to examine T25FW performance as a function of employment status in MS;26,31 one would expect that persons with MS who have MS-related alterations in work/employment status would have MS-related gait problems and walk slower on the T25FW. Indeed, one study compared T25FW performance based on employment status (working outside home vs disabled by self-report) in a sample of 169 patients with MS and reported a statistically significant difference in the median scores of 4.6 and 6.9 seconds for the employment groups.31 Another study compared T25FW performance across groups of MS work disabled, MS work challenged, and MS work stable in a sample of 275 patients with MS and reported a statistically significant difference in the mean scores of 7.4, 5.6, and 4.7 seconds across those three levels of employment status.26 Collectively, these data comparing T25FW performance across three separate group comparisons (i.e. controls, disability, and employment) indicate that T25FW scores can be interpreted as a valid measure of walking and its dysfunction in MS, and its relevance for employment status might even reflect ecological validity.
Correlational evidence of construct validity
A common approach for establishing construct validity involves examining the hypothesized pattern of correlations between T25FW scores and other measures of walking and lower extremity functioning (i.e. convergent validity) and measures of hand or upper extremity functioning (i.e. divergent validity). One would hypothesize stronger correlations with constructs related to lower extremity functioning and walking but weaker correlations with constructs associated with upper extremity functioning, vision, or cognition. To this end, researchers have reported that T25FW scores strongly correlated with 100-m timed walk (r = 0.92) and 6-minute walk (r = −0.83) performance as well as Timed Up and Go (r = 0.85), six spot step test (SSST; r = 0.92), and MSWS-12 (r = 0.78) scores in MS.19,27,32,33 There is evidence of strong correlations between T25FW performance and overall gait (r = −0.82)34 and objectively measured lower extremity functioning (r = 0.77)35 and moderate associations with oxygen cost of walking (r = 0.60) and steps/day from accelerometry (r = −0.64) in MS.36,37 By comparison, T25FW scores have weaker associations with measures of upper extremity function (e.g. 9-HPT); cognition (e.g. symbol digit modalities test and PASAT; rs = −0.48 and −0.36, respectively);38,39 body weight (d = .29, r = 0.14);40 and depression (R2 = 0.029, r⩽ 0.17), fall frequency (R2 = 0.053, r⩽ 0.23), and quality of life (R2 = 0.026, r⩽ 0.16).41 Collectively, these data indicate that T25FW scores can be interpreted as a valid measure of walking in MS.
Responsiveness and clinical meaningfulness of the T25FW
Responsiveness refers to the ability of the T25FW to capture intervention effects (i.e. sensitivity to change) regardless of clinical relevance. By comparison, clinical meaningfulness is the idea that a given amount of change or difference in T25FW scores is reflective of a relevant difference in real-life activities for a person living with MS. These are both pertinent aspects of T25FW validity, as these reflect its ability to capture changes over time that might be regarded as important in the context of an intervention or treatment.
Regarding responsiveness, one study examined the influence of intravenous methylprednisolone (IVMP) therapy on recovery of walking outcomes in patients with MS experiencing a period of relapse.42 The study included 49 patients with relapsing–remitting MS who received IVMP (1000 mg/day) over 3 days for relapse with associated walking difficulties. The researchers administered the T25FW along with the 2-minute walk (2-MW) test, EDSS, SSST, and MSWS-12 before and 1 month after IVMP. The T25FW detected a significant improvement in walking 1 month after IVMP (Time 1 = 6.8 seconds; Time 2 = 5.9 seconds), as did the other ambulatory measures (i.e. the T25FW and other measures captured intervention effects on walking). The effect size for the T25FW was 0.27 standardized units, whereas the standardized response mean was 0.55 units. The T25FW had 68.3% efficiency for capturing improvement compared with the EDSS, whereas the 2-MW had 95.1% efficiency, as an example. This means that the T25FW was less efficient for capturing intervention effects than the 2-MW (i.e. other measures were more responsive). Importantly, the T25FW captured statistically significant improvements in walking following a relapse, and this provides evidence for its responsiveness but not necessary clinically meaningful change. There is some additional evidence of the responsiveness of the T25FW for capturing rehabilitation effects, including exercise training, in MS.15,43
There is broad acceptance that a 20% change in T25FW performance represents a meaningful change in walking performance in MS. This was originally derived from an assessment of the reliability of quantitative functional tests including the T25FW on five consecutive days in 63 patients with MS44 and then later supported by validation of 20% change in T25FW scores based on correlation with change in other, external outcomes.45 This metric of meaningful change has been included as a benchmark and confirmed in multiple examinations of dalfampridine in MS.46–48
One recent paper compared the association between walking speed measured by the T25FW and the Physical Health Component Summary (PCS) score of the 36-item, Short Form Health Survey (SF-36) to better understand the clinical meaning of T25FW scores in MS.49 The analysis included retrospective data from three clinical trials of disease modifying therapies that included T25FW and SF-36 data. Of note, the percent change in T25FW over a 2-year period was significantly correlated with change in SF-36 PCS scores in the placebo-treated patients (r = 0.35). Among the placebo-treated patients, 27.5% of participants had a clinically meaningful change in SF-36 PCS scores that exceeded five points (the well-accepted cut point for meaningful change in the PCS score), and this group had an average 21.8% decline in T25FW performance. Additional analyses demonstrated that a change exceeding 20% on the T25FW generally corresponded with a meaningful change in SF-36 PCS scores. Collectively, these data paint a picture that the T25FW is capable of detecting changes in walking and worsening of at least 20% and that this degree of change can be viewed as clinically meaningful.
Conclusion and future directions
The T25FW test has been described as the “best characterized objective measure of walking disability and can be used across a wide range of walking disabilities” in MS.16 This observation is confirmed based on a robust literature review of the T25FW in persons with MS and its application and psychometric properties in the MS population. This review provided information on the (a) history, description, administration, and scoring of the T25FW; (b) its reliability and precision over short and long periods of time; (c) its content and construct validity; and (d) the responsiveness and clinical meaningfulness of changes in its scores. The T25FW represents an ideal primary endpoint for clinical research and practice targeting ambulation in MS, and by virtue of its simplicity, it might be the best test of ambulation for large, multicenter clinical trials.
As with all measures, validation is an ongoing and evolving process that requires the strongest evidence through continued research;23 this is important for application of the T25FW in clinical practice and research. Some important directions for future research involve validating clinically meaningful improvements on the T25FW as well as determining if 20% change is clinically meaningful across the disability spectrum. Researchers might consider synchronizing accelerometers and motion sensors with the T25FW for capturing walking speed in everyday life and the patient’s real environment. There is need for research determining the weighting placed on ambulation compared with other measurable performance domains for judging the effects of MS and clinical/rehabilitation therapies. There is additional research needed on developing benchmarks for interpreting or describing the real-world meaning of T25FW scores that could provide both clinical and research relevance for describing a patient or a study sample.25,30 There is limited understanding of time of day effects on T25FW performance, although there is a report of no difference in 10-m walk test performance between morning and noon/afternoon administrations.50 Finally, we note that the T25FW is not universally applicable across the MS spectrum, as it might have ceiling effects for those with an EDSS above 6.5 (i.e. not able to walk 25 feet). Nevertheless, researchers should continue efforts in validating scores from the T25FW as a measure of ambulation suitable for clinical research and practice involving MS.
Acknowledgments
The authors gratefully acknowledge the expert services of Wendy Kaye (McKing Consulting) and the organizational support from Gary Lundstrom and Alicia West (Critical Path Institute). The Multiple Sclerosis Outcome Assessments Consortium (MSOAC) operates through the Critical Path Institute.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Multiple Sclerosis Society (grant number RG 4869-A-1 to the Critical Path Institute). The authors received no financial compensation for this review.
Contributor Information
Robert W Motl, Department of Physical Therapy, University of Alabama at Birmingham, Birmingham, AL, USA.
Jeffrey A Cohen, Mellen Center, Neurological Institute, Cleveland Clinic, Cleveland, OH, USA.
Ralph Benedict, Department of Neurology, University at Buffalo, Buffalo, NY, USA.
Glenn Phillips, Biogen, Cambridge, MA, USA.
Nicholas LaRocca, National Multiple Sclerosis Society, New York, NY, USA.
Lynn D Hudson, Critical Path Institute, Tucson, AZ, USA.
Richard Rudick, Biogen, Cambridge, MA, USA.
Multiple Sclerosis Outcome Assessments Consortium, Multiple Sclerosis Outcome Assessments Consortium (MSOAC), Critical Path Institute, Tucson, AZ, USA.
References
- 1. Murray TJ. Multiple sclerosis: The history of a disease. New York: Demos, 2005. [Google Scholar]
- 2. Goldman MD, Motl RW, Rudick RA. Possible clinical outcome measures for clinical trials in patients with multiple sclerosis. Ther Adv Neurol Disord 2010; 3: 229–239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Hessen C, Bohm J, Reich C, et al. Patient perception of bodily functions in multiple sclerosis: Gait and visual function are the most valuable. Mult Scler 2008; 14: 988–991. [DOI] [PubMed] [Google Scholar]
- 4. LaRocca NG. Impact of walking impairment in multiple sclerosis: Perspectives of patients and care partners. Patient 2011; 4: 189–201. [DOI] [PubMed] [Google Scholar]
- 5. Motl RW. Ambulation and multiple sclerosis. Phys Med Rehabil Clin N Am 2013; 24: 325–336. [DOI] [PubMed] [Google Scholar]
- 6. Kurtke JF. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983; 33: 1444–1452. [DOI] [PubMed] [Google Scholar]
- 7. Hobart J, Freeman J, Thompson A. Kurtzke scales revisited: The application of psychometric methods to clinical intuition. Brain 2000; 123: 1027–1040. [DOI] [PubMed] [Google Scholar]
- 8. Hauser SL, Dawson DM, Lehrich JR, et al. Intensive immunosuppression in progressive multiple sclerosis: A randomized, three-arm study of high-dose intravenous cyclophosphamide, plasma exchange, and ACTH. N Engl J Med 1983; 308: 173–180. [DOI] [PubMed] [Google Scholar]
- 9. Schwid SR, Goodman AD, Mattson DH, et al. The measurement of ambulatory impairment in multiple sclerosis. Neurology 1997; 49: 1419–1424. [DOI] [PubMed] [Google Scholar]
- 10. Cutter GR, Baier ML, Rudick RA, et al. Development of a multiple sclerosis functional composite as a clinical outcome measure. Brain 1999; 122: 871–882. [DOI] [PubMed] [Google Scholar]
- 11. Gronwall DMA. Paced auditory serial-addition task: A measure of recovery from concussion. Percept Mot Skills 1977; 44: 367–373. [DOI] [PubMed] [Google Scholar]
- 12. Mathiowetz V, Weber K, Kashman N, et al. Adult norms for Nine Hole Peg Test of finger dexterity. Occup Ther J Res 1985; 5: 24–38. [Google Scholar]
- 13. Goodman AD, Brown TR, Cohen JA, et al. Dose comparison trial of sustained-release fampridine in multiple sclerosis. Neurology 2008; 71: 1134–1141. [DOI] [PubMed] [Google Scholar]
- 14. Goodman AD, Brown TR, Edwards KR, et al. A phase 3 trial of extended release oral dalfampridine in multiple sclerosis. Ann Neurol 2010; 68: 494–502. [DOI] [PubMed] [Google Scholar]
- 15. Pearson M, Bieberg G, Smart N. Exercise as a therapy for improvement of walking ability in adults with multiple sclerosis: A meta-analysis. Arch Phys Med Rehabil 2015; 96: 1339–1348. [DOI] [PubMed] [Google Scholar]
- 16. Kieseier BC, Pozzilli C. Assessing walking disability in multiple sclerosis. Mult Scler 2012; 18: 914–924. [DOI] [PubMed] [Google Scholar]
- 17. Cohen JA, Reingold SC, Polman CH, et al. Disability outcome measures in multiple sclerosis clinical trials: Current status and future prospects. Lancet Neurol 2012; 11: 467–476. [DOI] [PubMed] [Google Scholar]
- 18. Fischer JS, Rudick RA, Cutter GR, et al. The Multiple Sclerosis Functional Composite Measure (MSFC): An integrated approach to MS clinical outcome assessment. Mult Scler 1999; 5: 244–250. [DOI] [PubMed] [Google Scholar]
- 19. Nieuwenhuis MM, Van Tongeren H, Sorensen PS, et al. The six sport step test: A new measure for walking ability in multiple sclerosis. Mult Scler 2006; 12: 495–500. [DOI] [PubMed] [Google Scholar]
- 20. Rosti-Otajarvi E, Hamaiainen P, Koivisto K, et al. The reliability of the MSFC and its components. Acta Neurol Scand 2008; 117: 421–427. [DOI] [PubMed] [Google Scholar]
- 21. Learmonth YC, Paul L, Miller L, et al. Reliability and clinical significance of mobility and balance assessments in multiple sclerosis. Int J Rehabil Res 2012; 35: 69–74. [DOI] [PubMed] [Google Scholar]
- 22. Learmonth YC, Dlugonski D, Pilutti LA, et al. The reliability, precision, and clinically meaningful change of walking assessments in multiple sclerosis. Mult Scler 2013; 19: 1784–1791. [DOI] [PubMed] [Google Scholar]
- 23. Messick S. Validation of psychological assessment: Validation of inferences from persons’ responses and performances as scientific inquiry into score meaning. Am Psychol 1995; 50: 741–749. [Google Scholar]
- 24. Pearson OR, Busse ME, van Deursen, et al. Quantification of walking mobility in neurological disorders. QJM 2004; 97: 463–475. [DOI] [PubMed] [Google Scholar]
- 25. Landy FJ. Stamp collecting versus science: Validation as hypothesis testing. Am Psychol 1986; 41: 1183–1192. [Google Scholar]
- 26. Benedict RH, Drake AS, Irwin LN, et al. Benchmarks of meaningful impairment on the MSFC and BICAMS. Mult Scler 2016; 22: 1874–1882. [DOI] [PubMed] [Google Scholar]
- 27. Phan-Ba R, Pace A, Calay P, et al. Comparison of the timed 25-foot walk and the 100-meter walk as performance measures in multiple sclerosis. Neurorehabil Neural Repair 2011; 25: 672–679. [DOI] [PubMed] [Google Scholar]
- 28. Sandroff BM, Klaren RE, Motl RW. Relationships among physical inactivity, deconditioning, and walking impairment in persons with multiple sclerosis. J Neurol Phys Ther 2015; 39: 103–110. [DOI] [PubMed] [Google Scholar]
- 29. Learmonth YC, Motl RW, Sandroff BM, et al. Validation of patient determined disease steps (PDDS) scale score in persons with multiple sclerosis. BMC Neurol 2013; 13: 37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Sandroff BM, Motl RW, Sosnoff JJ, et al. Further validation of the Six-Spot Step Test as a measure of ambulation in multiple sclerosis. Gait Posture 2015; 41: 222–227. [DOI] [PubMed] [Google Scholar]
- 31. Goldman MD, Motl RW, Scagnelli J, et al. Clinically meaningful performance benchmarks in MS: Timed 25-foot walk and the real world. Neurology 2013; 81: 1856–1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Goldman MD, Marrie RA, Cohen JA. Evaluation of the six-minute walk in multiple sclerosis subjects and healthy controls. Mult Scler 2008; 14: 383–390. [DOI] [PubMed] [Google Scholar]
- 33. Cavanaugh JT, Gappmaier VO, Dibble LE, et al. Ambulatory activity monitoring in individuals with multiple sclerosis. J Neurol Phys Ther 2011; 35: 26–33. [DOI] [PubMed] [Google Scholar]
- 34. Sosnoff JJ, Weikert M, Dlugonski D, et al. Quantifying gait impairment in multiple sclerosis using GAITRite technology. Gait Posture 2011; 34: 145–147. [DOI] [PubMed] [Google Scholar]
- 35. Motl RW, Learmonth YC, Wojcicki TR, et al. Preliminary validation of the short physical performance battery in older adults with multiple sclerosis. BMC Geriatr 2015; 15: 157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Motl RW, Dlugonski D, Suh Y, et al. Accelerometry and its association with objective measures of walking limitations in ambulatory adults with multiple sclerosis. Arch Phys Med Rehabil 2010; 91: 1942–1947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Motl RW, Pilutti LA, Sandroff BM, et al. Accelerometry as a measure of walking behavior in multiple sclerosis. Acta Neurol Scand 2013; 127: 384–390. [DOI] [PubMed] [Google Scholar]
- 38. Motl RW, Cadavid D, Sandroff BM, et al. Cognitive processing speed has minimal influence on the construct validity of Multiple Sclerosis Walking Scale-12 scores. J Neurol Sci 2013; 335: 169–173. [DOI] [PubMed] [Google Scholar]
- 39. Benedict RH, Holtzer R, Motl RW, et al. Upper and lower extremity function and cognitive impairment in multiple sclerosis. J Int Neuropsychol Soc 2011; 17: 643–653. [DOI] [PubMed] [Google Scholar]
- 40. Pilutti LA, Dlugonski D, Pula JH, et al. Weight status in persons with multiple sclerosis: Implications for mobility outcomes. J Obes 2012; 2012: 868256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Bethoux FA, Palfy DM, Plow MA. Correlations of the timed 25-foot walk in a multiple sclerosis outpatient rehabilitation clinic. Int J Rehabil Res 2016; 39: 134–139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Filipovic Grcic P, Matijaca M, Lusic I, et al. Responsiveness of walking-based outcome measures after multiple sclerosis relapses following steroid pulses. Med Sci Monit 2011; 17: CR704–CR710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Baert I, Freeman J, Smedal T, et al. Responsiveness and clinically meaningful improvement, according to disability level, of five walking measures after rehabilitation in multiple sclerosis: A European multicenter study. Neurorehabil Neural Reapir 2014; 28: 621–631. [DOI] [PubMed] [Google Scholar]
- 44. Schwid SR, Goodman AD, McDermott MP, et al. Quantitative functional measures in MS: What is a reliable change? Neurology 2002; 58: 1294–1296. [DOI] [PubMed] [Google Scholar]
- 45. Kragt JJ, Van der Linden FA, Nielsen JM, et al. Clinical impact of 20% worsening on Timed 25-Foot Walk and 9-Hole Peg Test in multiple sclerosis. Mult Scler 2006; 6: 286–290. [DOI] [PubMed] [Google Scholar]
- 46. Coleman CI, Sobieraj DM, Marinucci LN. Minimally important clinical difference of the Timed 25-Foot Walk Test: Results from a randomized controlled trial in patients with multiple sclerosis. Curr Med Res Opin 2012; 28: 49–56. [DOI] [PubMed] [Google Scholar]
- 47. Hobart J, Blight AR, Goodman A, et al. Time 25-Foot Walk: Direct evidence that improving 20% or greater is clinically meaningful in MS. Neurology 2013; 80: 1509–1517. [DOI] [PubMed] [Google Scholar]
- 48. Jensen H, Ravnborg M, Mamoei S, et al. Changes in cognition, arm function and lower body function after slow-release Fampridine treatment. Mult Scler 2014; 20: 1872–1880. [DOI] [PubMed] [Google Scholar]
- 49. Cohen JA, Krishnan AV, Goodman AD, et al. The clinical meaning of walking speed as measured by the time 25-foot walk in patients with multiple sclerosis. JAMA Neurol 2014; 71: 1386–1393. [DOI] [PubMed] [Google Scholar]
- 50. Feys P, Gijbels D, Romberg A, et al. Effect of time of day on walking capacity and self-reported fatigue in persons with multiple sclerosis: A multi-center trial. Mult Scler 2012; 18: 351–357. [DOI] [PubMed] [Google Scholar]


