Abstract
Background
The p53 protein in mesenchymal stem cells (MSCs) regulates differentiation to osteogenic or adipogenic lineage. Because p53 function is depressed in most malignancies, if MSCs in malignancy also have p53 hypofunction, differentiation therapy to osteogenic or adipogenic lineage may be an effective treatment. We therefore wished to begin to explore this idea by evaluating atypical lipomatous tumor/well-differentiated liposarcoma (ALT/WDL) cells, because murine double minute 2 (MDM2) gene amplification, which leads to p53 hypofunction, is found in almost all ALT/WDLs.
Questions/purposes
We compared osteogenic and adipogenic differentiation potency between MSCs isolated and cultured from normal adipose tissues and ALT/WDLs from the same patients.
Methods
During tumor resections in six patients with ALT/WDL, we analyzed 3 mL of tumor, and for comparison, we harvested a similar amount of normal-appearing subcutaneous adipose tissue from an area remote from the tumor for comparison. Adipogenic differentiation potency was quantitatively assessed using spectrometry after oil red O staining. Osteogenic differentiation potency was semiquantitatively assessed by measuring a specific colored area after alkaline phosphatase (ALP) and alizarin red S staining. ALP is related to preosseous cellular metabolism, and alizarin red is related to calcium deposits in cell culture. There were three observers for each assessment, and each assessment (including induced-differentiation and histologic analysis) was performed in duplicate. We then analyzed the mechanism of the difference of osteogenic differentiation potency using the MDM2-specific inhibitor Nutlin-3 at various concentrations.
Results
In terms of adipogenic differentiation potency, contrary to our expectations, more fatty acid droplets were observed in MSCs derived from normal fat than in MSCs derived from ALT/WDL, although we found no significant difference between MSCs derived from ALT/WDL and MSCs derived from normal fat; the mean differentiation potency values (normal adipose tissue versus ALT/WDL) (± SD) were 0.34 (SD, ± 0.13; 95% CI, 0.24–0.44) versus 0.25 (SD, ± 0.10; 95% CI, 0.18–0.33; p = 0.22). By contrast, we found greater osteogenic differentiation potency in MSCs derived from ALT/WDL than in MSCs derived from normal fat. The mean differentiation potency values (normal adipose tissue versus ALT/WDL) (±SD) based on ALP staining was 1.0 versus 17 (SD, ± 36; 95% CI, −2.8 to 38; p = 0.04). However, we found no differences based on alizarin red S staining; mean differentiation potency value (normal adipose tissue versus ALT/WDL) (± SD) was 1.0 versus 4.2 (SD, ± 4.8; 95% CI, 1.3–7.2; p = 0.58). The gap of osteogenic differentiation potency between MSCs from normal adipose tissue and ALT/WDL was decreased as MDM2-inhibitor Nutlin-3 concentration increased.
Conclusions
MSCs derived from ALT/WDL had higher osteogenic differentiation potency based on ALP staining, which disappeared as Nutlin-3 concentration increased, suggesting that could be caused by amplified MDM2 in ALT/WDL. Future laboratory studies might mechanistically confirm the gene and protein expression, and based on the mechanism of the gap of differentiation potency, if p53 contrast between MSCs in tumor and normal tissue could be stimulated, less-toxic and more-effective differentiation therapy to MSCs in malignancies might be developed.
Keywords: Osteogenic Differentiation, Adipogenic Differentiation, Stromal Vascular Fraction, Differentiation Potency, Adipogenic Lineage
Introduction
Mesenchymal stem cells (MSCs), present in the bone marrow, adipose tissue, and other various tissues, can differentiate into other mesenchymal cells such as adipocyte, osteoblast, and chondrocyte cells. Cancer stem cells derived from mutant MSCs are involved with tumorigenesis, metastasis, and chemoresistance [14]. Considering the failures of conventional therapeutic approaches, novel treatment methods targeting MSCs in malignancies are needed. TP53 mutation, a cancer-regulating gene that codes for a protein that regulates the cell cycle and hence functions as a tumor suppressor, is the most-frequently occurring gene abnormality in human malignant neoplasias [4–6, 11, 15, 18, 19, 22]. The p53 protein gene product in MSCs regulates differentiation into osteogenic or adipogenic lineages by suppressing osterix, runx, and pparγ genes in MSCs [10, 16, 17], and p53 function is depressed in most malignancies [9].
If MSCs in malignancies similarly show a p53 hypofunction, differentiation therapy into osteogenic or adipogenic lineages may be an effective treatment method, suppressing the toxicity on the basis of the gap of differentiation potency between MSCs in malignant and normal tissue. We therefore wished to explore this idea by evaluating atypical lipomatous tumor/well-differentiated liposarcoma (ALT/WDL) cells, because murine double minute 2 (MDM2) gene amplification, which leads to p53 hypofunction, is found in almost all ALT/WDLs [7, 21]. To our knowledge, there are no published studies that have investigated the differentiation potency of MSCs derived from ALT/WDL.
We therefore sought to compare osteogenic and adipogenic differentiation potency between MSCs isolated and cultured from normal adipose tissue and ALT/WDL cells in the same patients.
Patients and Methods
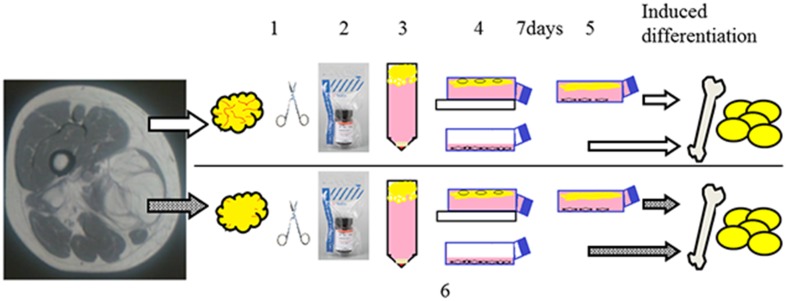
We compared osteogenic and adipogenic differentiation potency between MSCs isolated and cultured from normal adipose tissues and ALT/WDLs from the same patients (Fig. 1). We made the comparisons using quantitative or semiquantitative histologic techniques.
Fig. 1.
Dedifferentiated fat cells were isolated as follows: Fat tissue fragments were minced using scissors (1), and were digested in 0.12% collagenase type I medium (2). The resulting material was centrifuged (3). The floating fraction was placed in an inverted culture flask that was completely filled with culture medium (4). After 7 days, the flask was reversed (5). Adipose-derived stem cells were isolated the same as dedifferentiated fat cells (1, 2, and 3). The stromal vascular fraction was filtered through a 100-µm mesh and centrifuged and then resuspended in a culture medium (6).
During tumor resections in six patients with ALT/WDL, we analyzed 3 mL of tumor, and for comparison, we harvested a similar amount of normal-appearing subcutaneous adipose tissue from an area remote from the tumor. The study group included five men and one woman, with a mean age of 58 ± 7 years and mean BMI of 26 ± 3 kg/m2. The patients did not have diabetes or severe systemic illnesses, and none were taking medications that were known to affect the adipose tissue mass or metabolism. The study protocol was approved by the ethics committees at our respective universities. All patients provided written informed consent.
The MDM2 copy numbers of ALT/WDL tissues of six patients were analyzed with TaqMan® Copy Number Assays (Catalog Number: 4400291, assay number: Hs02873318_cn; Applied Biosystems, Foster City, CA, USA). Genomic DNA (gDNA) from formalin-fixed paraffin-embedded ALT/WDL tissues was isolated using DNeasy kits (Qiagen, Hamburg, Germany). gDNA concentrations were determined by A260 measurements (NanoDrop; Thermo Scientific, Wilmington, DE, USA), and purity was measured by A260/A280 measurement (all gDNA samples were > 1.70). The TaqMan® copy number reference assay (Applied Biosystems 4403326) and the TaqMan® copy number assay (Applied Biosystems 4400291, Assay ID Hs02873318_cn) were used according to the manufacturer’s instructions. Real-time quantitative PCR was performed using the StepOne™ Real Time PCR system (Applied Biosystems). Thermocycling conditions were 95° C for 10 minutes followed by 40 cycles of 95° C for 15 seconds and 60° C for 60 seconds. Quantification was performed using the ΔΔCt method in triplicate.
The CopyCaller software v2.0 (Applied Biosystems) was used to perform the copy number analysis. In ALT/WDL tissues of all six patients, the copy number of the MDM2 gene was greater than 3 (mean = 6). Human MSCs were isolated as previously described [8], with minor modifications. We used adipose-derived stem cells or dedifferentiated fat cells as MSCs. Tissues from three patients were examined using adipose-derived stem cells, and the other three were examined using dedifferentiated fat cells. Fat tissue fragments were minced using scissors and were digested in 0.12% collagenase type I medium with gentle shaking at 37° C for 45 minutes. The resulting material was centrifuged at 1300 rpm for 6 minutes at room temperature, resulting in adipocytes and free oil being separated from the stromal vascular fraction. The floating fraction comprising pure isolated adipocytes was placed in an inverted 25-cm2 culture flask that was completely filled with a culture medium (Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum, 100 units/mL penicillin, and 0.1 mg/mL streptomycin). The primary MSCs grown at the top of the inverted flask were cultured for 7 days after which the flask was reversed. The culture medium was changed every 3 days until the cultures reached confluence (defined as dedifferentiated fat cells passage 0). In contrast, the stromal vascular fraction was filtered through a 100-µm mesh and centrifuged at 1300 rpm for 6 minutes at room temperature and then resuspended in the culture medium (defined as adipose-derived stem cells passage 0). The cells were used for the experiments after passage 3. Both cells were incubated at 37° C in 5% CO2. Because primary cells have a finite lifespan and limited expansion capacity, gene and protein expression analyses were not performed.
To answer the question whether MSCs isolated from ALT/WDL tissue have more adipogenic differentiation potency than MSCs isolated from normal adipose tissue, we compared histologic findings, and the amount of fat droplets was quantitatively analyzed. For adipogenic differentiation, MSCs, 80,000 cells per well seeded on a 24-well plate, were induced to differentiation 1 day after the cells were seeded in an adipogenesis-induction medium comprising a culture medium supplemented with 1 μmol/L dexamethasone, 0.5 mmol/L 3-isobutyl-1-methyl xanthine (both from Millipore, Billerica, MA, USA), 10 µg/mL human insulin, and 100 µmol/L indomethacin (both from Sigma-Aldrich, St Louis, MO, USA). The medium was replaced every 2 to 3 days for 21 days. Adipogenic differentiation capacities were examined using oil red O (Wako Pure Chemical Industries, Ltd, Osaka, Japan) staining at the end of the 21-day induction with the adipogenic differentiation medium. Cells in the 24-well plate were fixed with 10% formalin neutral buffer solution, and their triacylglycerol content was stained with 0.3% (wt/vol) oil red O in 60% (vol/vol) isopropanol. After repeatedly washing with water, the stained oil red O was extracted with 100% (vol/vol) isopropanol. The absorbance of the extracted oil red O was spectrophotometrically determined at 520 nm to measure triglyceride accumulation.
To answer the question whether MSCs isolated from ALT/WDL tissue have more osteogenic differentiation potency than MSCs isolated from normal adipose tissue, we compared histologic findings and the amount of stains related to osteogenic differentiation were semiquantitatively analyzed. Differentiation into osteoblasts was induced using an osteoblast-inducer reagent, according to the manufacturer’s instructions. MSCs isolated from normal adipose tissue and ALT/WDL were seeded in a density of 5000 cells/cm2 on a 12-well plate. The MSCs were induced to osteoblast differentiation after 1 day in an osteogenesis induction medium. The osteogenesis induction medium was replaced every 4 days for 14 days in ALP staining assays and for 28 days in alizarin red S staining assays. ALP and alizarin red S staining were performed according to the manufacturer’s instructions. ALP is related to preosseous cellular metabolism, and alizarin red S is related to calcium deposits in cell culture. Macrophotographs were taken on the TREVIEWER (Trytec, Kyoto, Japan), a high-quality LED light box, after which osteogenic differentiation potency was assessed by measuring a specific colored area using the Image J (National Institutes of Health, Bethesda, MD, USA; http://rsbweb.nih.gov/ij/) image analysis software. The osteoblast-inducer reagent (Cat. No. MK430) was obtained from Takara Bio Inc (Dairen, China). An ALP staining kit was obtained from CosmoBio (Tokyo, Japan), and the alizarin red S staining kit was obtained from ScienCell Research Laboratories (San Diego, CA, USA).
To answer the question whether the gap of osteogenic differentiation capacity occurs as a result of depressed p53 activity because of MDM2 amplification, we used the MDM2 inhibitor Nutlin-3. Both MSCs were induced into osteogenic differentiation with the osteogenesis induction medium supplemented with Nutlin-3 in the following concentrations: 10, 5, 2.5, 1.25, 0.625, and 0 µmol/L. Osteogenic differentiation capacity was assessed as previously mentioned. The MDM2 inhibitor Nutlin-3 was obtained from Santa Cruz Biotech (Santa Cruz, CA, USA).
Statistical Analysis
Data are presented as mean ± SD. Adipogenic differentiation comparisons between the values were performed using the two-tailed Student’s t-test. For effect sizes we calculated Pearson’s correlation coefficient r and used three levels: small (0.1), medium (0.3), or large (0.5). Osteogenic differentiation potency was assessed by ALP and alizarin red S staining and then expressed with a ratio of the value of ALT/WDL to that of normal fat and statistically analyzed using the Mann-Whitney U test. For effect sizes we calculated Cohen’s d and used three levels: small (0.2), medium (0.5), or large (0.8). For all statistical analyses, the level of significance was set at a probability less than 0.05. All experiments were repeated at least twice.
Results
Adipogenic Differentiation Potency
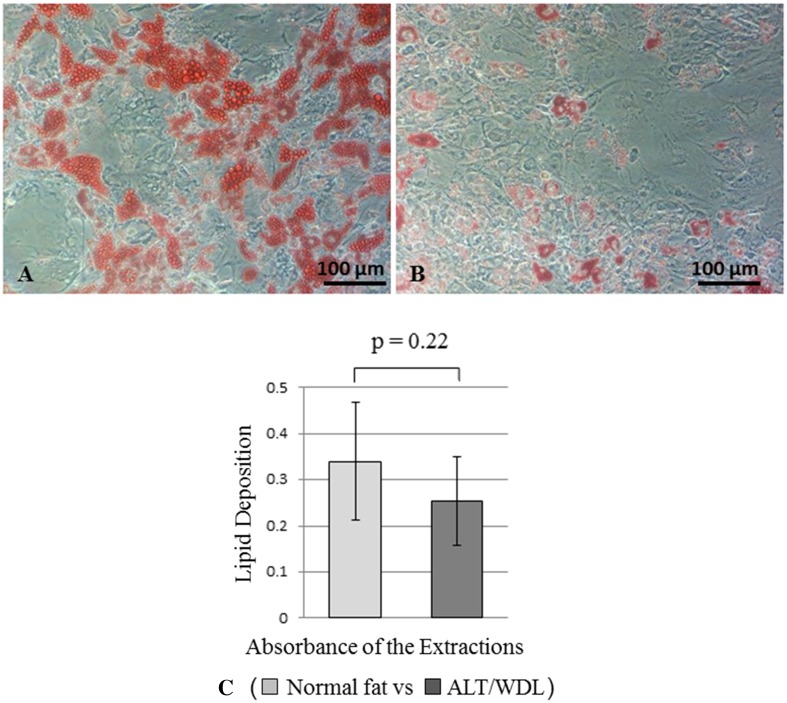
In adipogenic differentiation potency, contrary to our expectations, more fatty acid droplets were observed in MSCs derived from normal fat than in MSCs derived from ALT/WDL, although we found no differences between MSCs derived from ALT/WDL and MSCs derived from normal fat; the mean differentiation potency values (normal adipose tissue versus ALT/WDL) (± SD) were 0.34 (SD, ± 0.13; 95% CI, 0.24-0.44) versus 0.25 (SD, ± 0.10; 95% CI, 0.18–0.33; p = 0.22) (Fig. 2).
Fig. 2A–C.
(A) A photomicrograph shows MSCs derived from normal fat after 21 days of induced adipogenic differentiation. Fat droplets in the cells were stained with 0.3% (wt/vol) oil red O in 60% (vol/vol) isopropanol. (B) A photomicrograph shows MSCs derived from ALT/WDL after 21 days of induced adipogenic differentiation. Fat droplets in the cells were stained with 0.3% (wt/vol) oil red O in 60% (vol/vol) isopropanol. (C) The graph shows the adipogenic differentiation ability of MSCs derived from normal fat and ALT/WDL. The stained droplets were extracted with 100% (vol/vol) isopropanol. The absorbance was spectrophotometrically determined at 520 nm. We found no significant differences between MSCs derived from ALT/WDL and MSCs derived from normal fat.
Osteogenic Differentiation Potency
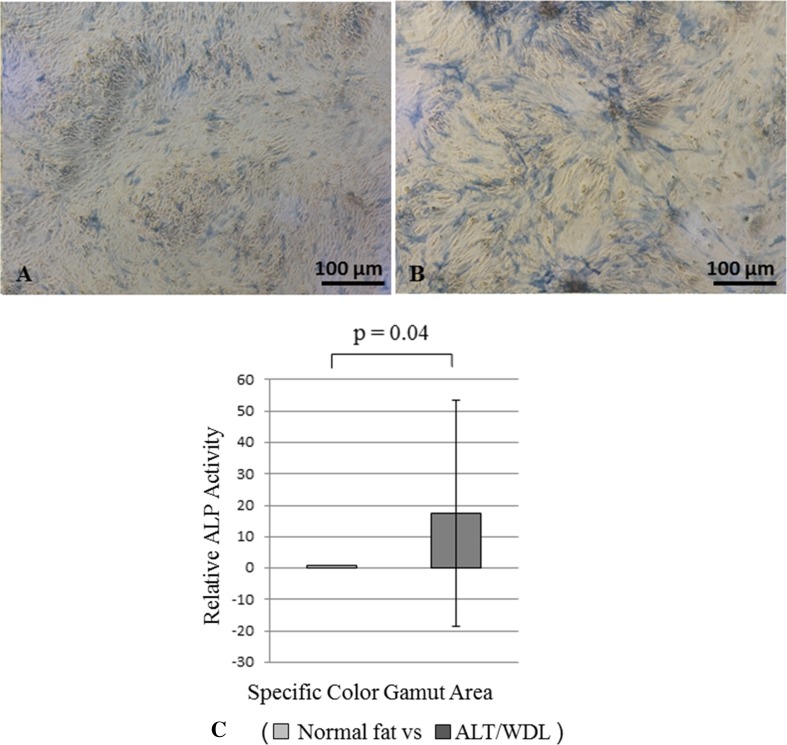
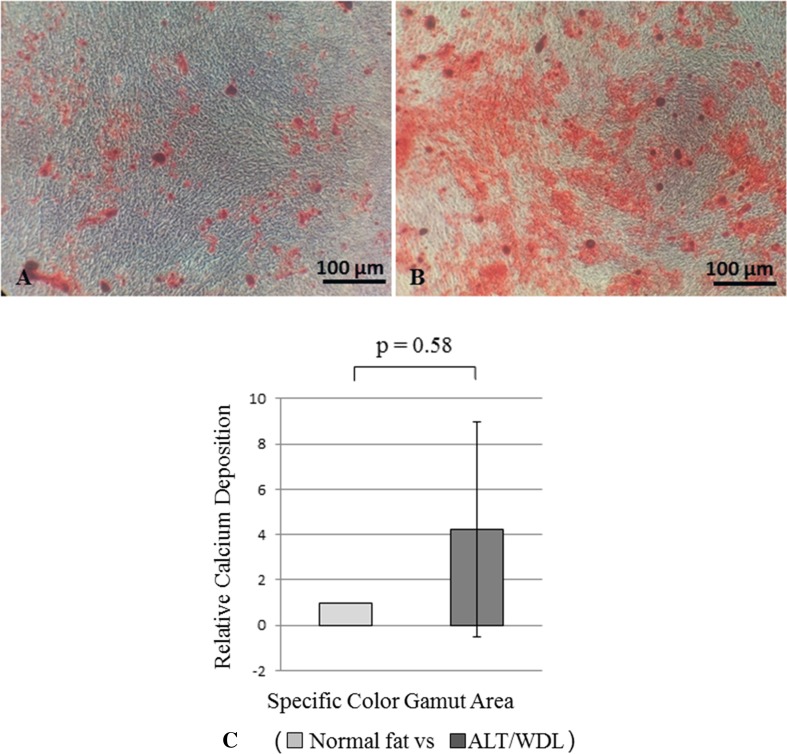
In osteogenic differentiation potency based on ALP staining, we found greater osteogenic differentiation potency in MSCs derived from ALT/WDL than in MSCs derived from normal fat; the mean differentiation potency value (normal adipose tissue versus ALT/WDL) (± SD) based on ALP staining was 1.0 versus 17 (SD, ± 36; 95% CI, −2.8 to 38; p = 0.04) with a medium effect size (d = 0.65) (Fig. 3). However, we found no differences based on alizarin red S staining; the mean differentiation potency value (normal adipose tissue versus ALT/WDL) (±SD) was 1.0 versus 4.2 (SD, ± 4.8; 95% CI, 1.3–7.2; p = 0.58) (Fig. 4).
Fig. 3A–C.
The photomicrographs show MSCs derived from (A) normal fat after 14 days of induced osteogenic differentiation, and from (B) ALT/WDL after 14 days of induced osteogenic differentiation. ALP staining was performed in both cases. Greater ALP staining was observed in MSCs derived from ALT/WDL. (C) A graph shows the osteogenic differentiation ability of MSCs derived from normal fat and ALT/WDL using ALP staining. Osteogenic differentiation potency was assessed by measuring a specific colored area using Image J image analysis software. MSCs derived from ALT/WDL had higher differentiation potency than MSCs derived from normal adipose tissue. ALP = alkaline phosphatase.
Fig. 4A–C.
The photomicrographs show MSCs derived from (A) normal fat and from (B) ALT/WDL after 28 days of induced osteogenic differentiation. Alizarin red S staining was performed. Greater calcium deposition was observed in MSCs derived from ALT/WDL. (C) The graph shows the osteogenic differentiation ability of MSCs derived from normal fat and ALT/WDL using alizarin red S staining. Osteogenic differentiation potency was assessed by measuring a specific colored area using the Image J image analysis software. We found no significant differences between MSCs derived from ALT/WDL and MSCs derived from normal fat.
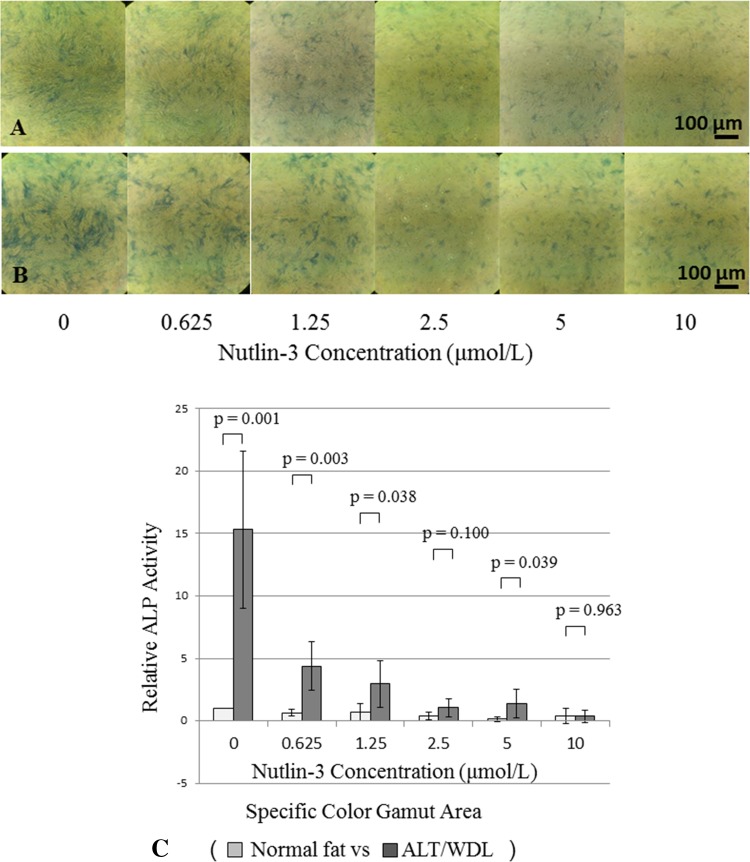
We then analyzed the mechanism of the difference of osteogenic differentiation potency using the MDM2-specific inhibitor Nutlin-3 at various concentrations. The gap of osteogenic differentiation potency between MSCs from normal adipose tissue and ALT/WDL was decreased as the Nutlin-3 concentration increased; the mean differentiation potency values (normal adipose tissue versus ALT/WDL) (±SD) based on ALP staining in each Nutlin-3 concentration (0, 0.625, 1.25, 2.5, 5, and 10 µmol/L) were 1.0 versus 15 (SD, ± 6.3; 95% CI, 9.8–20; p = 0.001) with a large effect size (d = 3.7), 0.66 (SD, ± 0.28; 95% CI, 0.42–0.91) versus 4.4 (SD, ± 2.0; 95% CI, 2.7–6.1; p = 0.003) with a large effect size (d = 2.7), 0.71 (SD, ± 0.71; 95% CI, 0.09–1.3) versus 3.0 (SD, ± 1.9; 95% CI, 1.3–4.6; p = 0.038) with a large effect size (d = 1.6), 0.40 (SD, ± 0.28; 95% CI, 0.16–0.65) versus 1.1 (SD, ± 0.73; 95% CI, 0.41–1.7; p = 0.10), 0.14 (SD, ± 0.18; 95% CI, −0.01 to 0.30) versus 1.4 (SD, ± 1.1; 95% CI, 0.41–2.4; p = 0.039) with a large effect size (d = 1.6), and 0.39 (SD, ± 0.61; 95% CI, −0.14 to 0.93) versus 0.38 (SD, ± 0.47; 95% CI, −0.04 to 0.79; p = 0.96) (Fig. 5).
Fig. 5A–C.
The photomicrographs show MSCs derived from (A) normal fat and (B) ALT/WDL after 14 days of induced osteogenic differentiation with an osteogenesis induction medium supplemented with MDM2 inhibitor Nutlin-3 in various concentrations. ALP staining was performed. The gap of ALP staining between MSCs from normal adipose tissue and ALT/WDL was decreased as the Nutlin-3 concentration increased. (C) The osteogenic differentiation ability of MSCs derived from normal fat and ALT/WDL using ALP staining is presented in this graph. Osteogenic differentiation potency was assessed by measuring a specific colored area using the Image J image analysis software. The gap of differentiation potency between MSCs from normal adipose tissue and ALT/WDL was decreased as the Nutlin-3 concentration increased. ALP = alkaline phosphatase.
Discussion
The p53 protein in MSCs regulates differentiation into osteogenic or adipogenic lineage. Because p53 function is depressed in most malignancies, if MSCs in malignancies also have p53 hypofunction, differentiation therapy into osteogenic or adipogenic lineage may be an effective treatment. To the best of our knowledge, there are no published studies that have investigated the differentiation potency of MSCs derived from ALT/WDL. Therefore, we compared osteogenic and adipogenic differentiation potency between MSCs isolated and cultured from normal adipose tissue and ALT/WDL from the same patients. We selected ALT/WDL for testing the assumption that MSCs derived from the tumors, which have depressed p53 function, will be introduced into adipogenic or osteogenic differentiation more than MSCs derived from normal tissue because ALT/WDL has a relevant counterpart, which is normal adipose tissue. ALT/WDL is used in research because it has a low prevalence of p53 mutation and high prevalence of MDM2 amplification [13]. Even if ALT/WDLs have TP53 missense mutations in this study, the resulting p53 hypofunction should lead MSCs to the same tendency of differentiation as MDM2-amplified tumors.
Some limitations exist in this study. First, there is considerable discussion regarding whether adipose-derived MSCs are a pure or mixed population. Using two kinds of isolation, we assessed all cells that adhere to a plastic flask and that have proliferative and differentiation capacities. Each comparison was performed with the cells isolated using the same method. Second, we did not perform gene and protein expression experiments because of the difficulty in primary culturing of MSCs derived from ALT/WDL, which has relatively low prevalence. Puzio-Kuter et al. [12] reported that using a liposarcoma mouse model with the conditional inactivation of PTEN and p53, MDM2 RNA levels by real-time PCR (TaqMan®) in normal versus tumor tissue were 1.5- to twofold higher (data not shown), and the protein expression of MDM2 by immunohistochemistry was at very high levels in well-differentiated liposarcoma sections in areas with clear lipoblasts. Although that was a special setting, they provided useful information in terms of amplified MDM2 expression in liposarcoma. Third, the sample harvesting sites were different between normal adipose tissues and ALT/WDLs. We harvested the comparison samples from the same human to equalize the background in each comparison. However, most ALT/WDLs were deep (below the fascia), and normal adipose tissues were harvested from subcutaneous tissues in this study. We do not believe that this is a disqualifying limitation because the amplified MDM2 copy numbers of ALT/WDL tissues of six patients were confirmed, indicating, we believe, that a fair comparison was made between normal and tumor tissues.
In adipogenic differentiation, contrary to our expectations, we found no differences between MSCs derived from ALT/WDL and MSCs derived from normal fat. Gopinathan et al. [2] reported MDM2 regulates peroxisome proliferator-activated receptor-α (PPARα)-mediated pathways of lipid metabolism. Moreover, Goto et al. [3] reported that activation of PPARα stimulates differentiation and fatty acid oxidation in adipocytes. Because our study focused on the relationship between MDM2 and p53, adipogenic induced-differentiation was not suitable. Considering differentiation therapy, in at least tumors that have depressed p53 function because of MDM2 amplification, adipogenic-induced differentiation may be ineffective. Demetri et al. [1] and Tontonoz et al. [20] reported induction of human liposarcoma terminal differentiation by the PPAR- γ ligand troglitazone as a potential therapeutic method. According to our observations, those adipogenic-differentiation-inducing methods may be inefficient in tumors with MDM2 amplification, although terminal differentiation of tumor cells is promising in terms of suppressing the tumor growth and metastasis.
In osteogenic differentiation, as expected, MSCs derived from ALT/WDL had higher differentiation potency than MSCs derived from normal adipose tissue. To the best of our knowledge, no published studies have compared the differentiation potency between those MSCs. Because the difference of osteogenic differentiation potency disappeared as Nutlin-3 concentration increased, the difference can be attributed to MDM2 amplification, which leads to depressed p53 function in MSCs derived from ALT/WDL, although we have not confirmed their gene expressions. The osteogenic differentiation capacity was not proportional to the MDM2 amplification copy number. We believe the reason for this is that the copy number was assayed with formalin-fixed paraffin-embedded ALT/WDL tissues including normal various cells and the copy number is the mean of the tissue. Therefore, the copy number is not an accurate number of MDM2 amplified cells.
Previous studies revealed that ALT/WDL tumors have increased MDM2 expression [7, 21]. In the current study, we found that MSCs derived from ALT/WDL tissues have more osteogenic differentiation potency than MSCs derived from the normal adipose tissue of the same human. The gap of osteogenic differentiation potency between MSCs derived from ALT/WDL and normal adipose tissues was depressed by the MDM2 inhibitor Nutlin-3. Future laboratory studies might mechanistically confirm the gene and protein expressions, and based on the mechanism of the gap of differentiation potency, if p53 contrast between MSCs in tumor and normal tissue could be enhanced by some stimulation (such as treatments that encourage p53 expression), less-toxic and more-effective differentiation therapy to MSCs in malignancies might be developed. If future studies confirm osteogenic differentiation potency is higher in MSCs derived from ALT/WDL, and can confirm the mechanism behind this observation, then translational work might subsequently investigate whether tumors with amplified MDM2 could be treated more effectively by osteogenic differentiation-induced therapy with adjuvant chemotherapy or radiotherapy.
Acknowledgments
We thank Mineyoshi Aoyama MD, PhD (Department of Molecular Neurobiology, Nagoya City University Graduate School of Medical Science), Yuko Nagaya MD, PhD, and Seiji Otsuka MD, PhD (both from the Department of Orthopaedic Surgery, Nagoya City University Graduate School of Medical Science) for support with the experiments. We also thank Enago (www.enago.jp) for English language review.
Footnotes
This work was supported by JSPS KAKENHI Grant Number JP26462271 (KH).
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research ® editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research ® neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
Each author certifies that his or her institution has approved the human protocol for this investigation, that all investigations were conducted in conformity with ethical principles of research, and that informed consent was obtained.
This work was performed at Kanazawa University Graduate School of Medical Science, Ishikawa-ken, Japan; and Nagoya City University Graduate School of Medical Science, Nagoya-shi, Japan.
References
- 1.Demetri GD, Fletcher CD, Mueller E, Sarraf P, Naujoks R, Campbell N, Spiegelman BM, Singer S. Induction of solid tumor differentiation by the peroxisome proliferator-activated receptor-gamma ligand troglitazone in patients with liposarcoma. Proc Natl Acad Sci U S A. 1999;96:3951–3956. doi: 10.1073/pnas.96.7.3951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gopinathan L, Hannon DB, Peters JM, Heuvel JPV. Regulation of peroxisome proliferator-activated receptor-α by MDM2. Toxicol Sci. 2009;108:48–58. doi: 10.1093/toxsci/kfn260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goto T, Lee JY, Teraminami A, Kim YI, Hirai S, Uemura T, Inoue H, Takahashi N, Kawada T. Activation of peroxisome proliferator-activated receptor-alpha stimulates both differentiation and fatty acid oxidation in adipocytes. J Lipid Res. 2011;52:873–884. doi: 10.1194/jlr.M011320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hainaut P, Soussi T, Shomer B, Hollstein M, Greenblatt M, Hovig E, Harris CC, Montesano R. Database of p53 gene somatic mutations in human tumors and cell lines: updated compilation and future prospects. Nucleic Acids Res. 1997;25:151–157. doi: 10.1093/nar/25.1.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Harris CC, Hollstein M. Clinical implications of the p53 tumor-suppressor gene. N Engl J Med. 1993;329:1318–1327. doi: 10.1056/NEJM199310283291807. [DOI] [PubMed] [Google Scholar]
- 6.Hollstein M, Sidransky D, Vogelstein B, Harris CC. p53 mutations in human cancers. Science. 1991;253:49–53. doi: 10.1126/science.1905840. [DOI] [PubMed] [Google Scholar]
- 7.Kimura H, Dobashi Y, Nojima T, Nakamura H, Yamamoto N, Tsuchiya H, Ikeda H, Sawada-Kitamura S, Oyama T, Ooi A. Utility of fluorescence in situ hybridization to detect MDM2 amplification in liposarcomas and their morphological mimics. Int J Clin Exp Pathol. 2013;6:1306–1316. [PMC free article] [PubMed] [Google Scholar]
- 8.Matsumoto T, Kano K, Kondo D, Fukuda N, Iribe Y, Tanaka N, Matsubara Y, Sakuma T, Satomi A, Otaki M, Ryu J, Mugishima H. Mature adipocyte-derived dedifferentiated fat cells exhibit multilineage potential. J Cell Physiol. 2008;215:210–222. doi: 10.1002/jcp.21304. [DOI] [PubMed] [Google Scholar]
- 9.Meek DW. Regulation of the p53 response and its relationship to cancer. Biochem J. 2015;469:325–346. doi: 10.1042/BJ20150517. [DOI] [PubMed] [Google Scholar]
- 10.Molchadsky A, Shats I, Goldfinger N, Pevsner-Fischer M, Olson M, Rinon A, Tzahor E, Lozano G, Zipori D, Sarig R, Rotter V. p53 plays a role in mesenchymal differentiation programs, in a cell fate dependent manner. PLoS One. 2008;3:e3707. doi: 10.1371/journal.pone.0003707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nigro JM, Baker SJ, Preisinger AC, Jessup JM, Hostetter R, Cleary K, Bigner SH, Davidson N, Baylin S, Devilee P, et al. Mutations in the p53 gene occur in diverse human tumour types. Nature. 1989;342:705–708. doi: 10.1038/342705a0. [DOI] [PubMed] [Google Scholar]
- 12.Puzio-Kuter AM, Laddha SV, Castillo-Martin M, Sun Y, Cordon-Cardo C, Chan CS, Levine AJ. Involvement of tumor suppressors PTEN and p53 in the formation of multiple subtypes of liposarcoma. Cell Death Differ. 2015;22:1785–1791. doi: 10.1038/cdd.2015.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ray-Coquard I, Blay JY, Italiano A, Le Cesne A, Penel N, Zhi J, Heil F, Rueger R, Graves B, Ding M, Geho D, Middleton SA, Vassilev LT, Nichols GL, Bui BN. Effect of the MDM2 antagonist RG7112 on the P53 pathway in patients with MDM2-amplified, well-differentiated or dedifferentiated liposarcoma: an exploratory proof-of-mechanism study. Lancet Oncol. 2012;13:1133–1140. doi: 10.1016/S1470-2045(12)70474-6. [DOI] [PubMed] [Google Scholar]
- 14.Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- 15.Roessner A, Schneider-Stock R, Radig K, Neumann W, Mittler U. Alterations of p53 gene in soft tissue and bone tumors. Genet Diagn Pathol. 1997;143:1–13. [PubMed] [Google Scholar]
- 16.Solozobova V, Blattner C. p53 in stem cells. World J Biol Chem. 2011;2:202–214. doi: 10.4331/wjbc.v2.i9.202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tataria M, Quarto N, Longaker MT, Sylvester KG. Absence of the p53 tumor suppressor gene promotes osteogenesis in mesenchymal stem cells. J Pediatr Surg. 2006;41:624–632. doi: 10.1016/j.jpedsurg.2005.12.001. [DOI] [PubMed] [Google Scholar]
- 18.Taubert H, Meye A, Wurl P. Soft tissue sarcomas and p53 mutations. Mol Med. 1998;4:365–372. [PMC free article] [PubMed] [Google Scholar]
- 19.Taubert H, Schmidt H, Wurl P, Hinze R, Meye A, Bache M, Berger D, Holzhausen H, Dralle H, Rath F. Morphological and molecular characterization of an undifferentiated soft tissue sarcoma cell line and derivative clones. Int J Oncol. 1997;11:521–526. doi: 10.3892/ijo.11.3.521. [DOI] [PubMed] [Google Scholar]
- 20.Tontonoz P, Singer S, Forman BM, Sarraf P, Fletcher JA, Fletcher CD, Brun RP, Mueller E, Altiok S, Oppenheim H, Evans RM, Spiegelman BM. Terminal differentiation of human liposarcoma cells induced by ligands for peroxisome proliferator-activated receptor gamma and the retinoid X receptor. Proc Natl Acad Sci U S A. 1997;94:237–241. doi: 10.1073/pnas.94.1.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Weaver J, Kelly DE, Goldblum JR, Turner S, Kulkarni S, Tubbs RR, Rubin BP, Skacel M. Fluorescence in situ hybridization for MDM2 gene amplification as a diagnostic tool in lipomatous neoplasms. Mod Pathol. 2008;21:943–949. doi: 10.1038/modpathol.2008.84. [DOI] [PubMed] [Google Scholar]
- 22.Yoo J, Lee HK, Kang CS, Park WS, Lee JY, Shim SI. p53 gene mutations and p53 protein expression in human soft tissue sarcomas. Arch Pathol Lab Med. 1997;121:395–399. [PubMed] [Google Scholar]