Abstract
The prevalence of chronic kidney disease (CKD) is estimated to be 8–16% worldwide, and it is increasing. CKD is a risk factor for heart attack and stroke, and it can progress to kidney failure requiring dialysis or transplantation. Recently, diabetic nephropathy has become the most common cause of CKD. In Japan, the cumulative probability of requiring hemodialysis by the age 80 years is 1/50 in males and 1/100 in females. The number of patients under hemodialysis in Japan exceeded 320,000 in 2014, among which 38,000 were newcomers and 27,000 died.
The annual medical costs of hemodialysis are 1.25 trillion yen in Japan, representing 4% of the total national medical expenditures in 2014. A low-protein diet (less than 0.5 g/kg b.wt.) is a very effective intervention. Low-protein rice (1/10 to 1/25 of the normal protein contents) is helpful to control the consumption of proteins, decreasing at the same time the intake of potassium and phosphate.
Protein restriction is indicated as soon as the eGFR becomes lower than 60 ml/min/1.73 m2 body surface, in order, to slow disease progression. The newly developed low-protein Indica rice is expected to help many CKD patients in China and Southeast Asia.
Keywords: chronic kidney disease, low-protein rice, low protein diet, dietary therapy, prevention, CKD, eGFR
The global burden of chronic kidney disease
Chronic kidney disease (CKD) is an important public-health issue because of its progression to adverse outcomes and high health-care costs.1–3) The prevalence is estimated to be 8–16% worldwide. It is also linked to other major lifestyle-related diseases, such as diabetes, cardiovascular diseases and hypertension.4)
CKD is defined as a reduced glomerular filtration rate (GFR), an increased urinary albumin excretion, or both.1) Complications include: an increase in all-cause mortality and cardiovascular mortality, kidney-disease progression, acute kidney injury, cognitive decline, anemia, mineral and bone disorders, and fractures.5)
Recently, diabetes mellitus has become the most common cause of CKD worldwide.6) The increasing prevalence of diabetes has led to CKD becoming the leading cause of end-stage kidney disease (ESRD) in many countries.4) Screening and adequate interventions can prevent CKD to progress toward ESRD.7,8) Diabetic kidney disease (DKD) occurs in 20% to 40% of patients with diabetes mellitus and it is the leading cause of CKD and end-stage renal failure in the United States9) and in Japan. DKD causes a progressive decline in renal function that is largely irreversible.
Renal replacement therapy, such as peritoneal dialysis, hemodialysis or kidney transplantation are often needed.10,11) The cumulative risk of hemodialysis until age 80 years is 1/50 in males and 1/100 in females.12)
The economic cost of DKD will grow to enormous amounts, and strategies to prevent its onset or progression are urgently awaited. There is emerging evidence that changes in renal function occurring early in the course of diabetes predict future outcomes.13) In Japan, population-based health checks include the screening of eGFR and urinary protein, allowing the detection of declines in renal function at an early stage.15)
When are dialysis and/or transplantation indicated? Typical criteria are: glomerular filtration rate (GFR) <10%, uncontrolled symptoms, uremic syndrome, vomiting, low appetite, edema, cardiac failure, hyperkalemia, and significant acidosis.
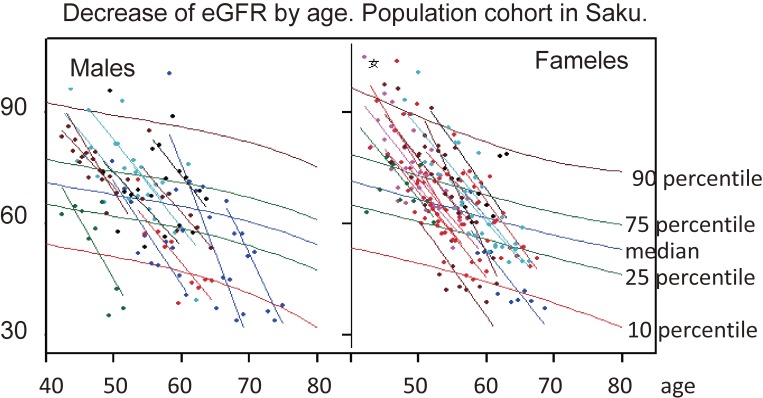
We have been monitoring changes in eGFR in a population-based cohort study in Saku municipality.16) Usually the eGFR decreases at a rate of 1 ml/year, when appropriate amounts (male 60 g/day, female 50 g/day) of proteins are consumed. In some people, however, the decrease in eGFR accelerates up to 5 ml per year (Fig. 1). Their dietary habits feature a preference for broiled or baked beef or other meat dishes.
Figure 1.
Annual change in eGFR in a population-based cohort study. Usually 1 ml/year decrease is observed, when the proper amount of protein is consumed. If the protein intake increases, the decreasing rate accelerates up to 5 ml/year. When we examine the dietary habits of participants, all of them claimed to prefer eating meat.
Social burdens of chronic kidney disease
The number of patients under hemodialysis in Japan exceeded 320,000 in 2014, among which 38,000 were newcomers and 27,000 died.17) Reasons for initiating hemodialysis were: diabetic nephropathy (43.3%), chronic glomerulonephritis (17.8%), nephrosclerosis (14.2%), and others. Chronic glomerulonephritis decreased, while other complications of diabetes increased. Chronic pyelonephritis and acute progressive glomerulonephritis decreased, in parallel with a lower incidence of polycystic kidney and systemic lupus erythematosus (SLE).
It is very important to delay the conditions that make hemodialysis a necessity, both to decrease medical costs and to improve the quality of life of the patients. The annual medical costs for hemodialysis are 1.25 trillion yen in Japan, representing 4% of the total national medical expenditure (31.5 trillion yen) in 2014.18)
Peritoneal dialysis, hemodialysis and transplantation are the classical treatments for advanced renal failure. In Japan, 96% of dialysed patients receive hemodialysis, and only 3–4% undergo peritoneal dialysis.17) The one-year survival rate after induction of hemodialysis exceeds 90% in Japan, but it is 80% in the USA and 85% in Europe. Technical difference and/or lead time bias could account for such differences. The 10-years CKD survival in Japan is nearly 80%, and the 20-years survival is 60%. The longest recorded surviving period is 46.5 years now.
Supportive care during hemodialysis is important.19) Water-soluble vitamins and some minerals are easily lost by hemodialysis, so the monitoring and the prescription of supplementary or therapeutic doses are often necessary. Dietary control is also important. The recommended protein intake during hemodialysis is 1.2–1.5 g/kg b.wt. usually, but 0.8–1.0 g/kg b.wt. is often enough and it can reduce the frequency of dialysis.
Low-protein diet for the prevention and treatment of CKD
Diet is one of the oldest treatments of CKD, and it has been classically considered to be the mainstay of its management.20) Over the past 50 years, protein-restricted diets have been successfully used to treat chronic renal failure in Japan.21) Even if associated with comprehensive management, prescribing a dietary therapy is often insufficient because of the need for adjustments in eating habits. The empathic educational approach may support a successful personalized dietary intervention.22)
Accumulating evidence from recent randomized controlled trials (RCTs) and cohort studies with long observation periods call for revolutionary changes in dietary therapy. With the increase of lifestyle-related diseases, the improvement of dietary habits and the practice of physical exercise have become more important than drug therapy.23)
Despite the protecting effects of a low-protein diet, patients with severe renal failure ultimately will need dialysis or transplantation. A diet adjusted to hemodialysis also has therapeutic effects. The substitution of animal proteins with vegetable proteins improves the prognosis.24) The replacement of half of the animal proteins (70%) by soy proteins showed an improvement of many biomarkers in follow-up studies of type-2 diabetic patients with nephropathy (Table 1). Blood urea nitrogen (BUN) is a good index of the effect of dietary therapy, and it should remain less than 15 mg/dl.
Table 1.
Longitudinal effect of soy protein substitution to animal protein. This longitudinal randomized clinical trial was conducted among 41 type-2 diabetic patients with nephropathy (18 men and 23 women). Twenty patients in the soy protein group consumed a diet containing 0.8 g protein/kg b.wt. (35% animal proteins, 35% textured soy protein, and 30% vegetable proteins) and 21 patients in the control group consumed a similar diet containing 70% animal proteins and 30% vegetable proteins for 4 years. The addition of soy protein improved their prognosis
| 70% animal + 30% vegetable P | 35% animal + 35% soy P + 30% vegetable P | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 4th yr | 3rd yr | 2nd yr | 1st yr | baseline | 4th yr | 3rd yr | 2nd yr | 1st yr | baseline | |
| Weight (kg) | 73 | 69 | 73 | 71 | 72 | 71 | 73 | 72 | 70 | 71 |
| SBP (mmHg) | 148 | 147 | 150 | 155 | 153 | 147 | 149 | 158 | 148 | 150 |
| DBP (mmHg) | 93 | 94 | 96 | 95 | 91 | 93 | 94 | 90 | 92 | 96 |
| FPG (mg/dl) | 147* | 146 | 145 | 142 | 137 | 121* | 129 | 13 | 130 | 141 |
| TC (mg/dl) | 228* | 227 | 225 | 221 | 218 | 201* | 207 | 209 | 216 | 225 |
| LDL-C (mg/dl) | 158* | 156 | 148 | 153 | 151 | 128 | 132 | 138 | 141 | 149 |
| HDL-C (mg/dl) | 45* | 43 | 40 | 46 | 43 | 53* | 50 | 52 | 47 | 49 |
| UUN (mg/dl) | 19.5* | 18.9 | 18.5 | 17.8 | 18.1 | 12.8* | 14.3 | 15.7 | 16.0 | 17.2 |
| Proteinuria | 725* | 719 | 717 | 684 | 691 | 513* | 543 | 603 | 621 | 667 |
| Serum Cr (mg/dl) | 1.61* | 1.54 | 1.51 | 1.45 | 1.49 | 1.41* | 1.49 | 1.44 | 1.53 | 1.56 |
| Urine Cr (mg/dl) | 5.2* | 5.4 | 4.8 | 4.6 | 4.5 | 3.1* | 3.5 | 3.8 | 4.0 | 4.8 |
| BUN (mg/dl) | 22 | 21 | 20 | 20 | 19 | 18* | 18 | 19 | 19 | 20 |
| CRP (mg/dl) | 3.9 | 3.8 | 3.6 | 3.7 | 3.5 | 2.4 | 2.5 | 3.1 | 3.2 | 3.8 |
| GFR (ml/min) | 81 | 75 | 86 | 80 | 78 | 88 | 82 | 79 | 89 | 84 |
| Plasma phytoE | 1.08 | 1.11 | 1.06 | 1.10 | 1.07 | 1.29* | 1.27 | 1.19 | 1.33 | 1.02 |
*p < 0.05.
Modified from Azadbakht and Atabak,24) Diabetes Care. 2008
Low-protein diets are very effective, but many physicians fail to control renal dysfunction because they believe that 0.8 g/kg b.wt. protein intake is already a low-protein diet. How is the recommended dose of protein intake determined in the dietary reference intake (DRI)? In Japan, the recommended range of protein intake has been determined as ±2 SD around the average intake of 0.65 g/kg b.wt. It means that if we take 0.83 g/kg proteins, deficiency would not occur in 97–98% people. The nitrogen equilibrium being 0.3 g/kg, the appropriate range of low-protein diets should vary between 0.3 to 0.5 or 0.6 g/kg b.wt. Actually this low-protein diet induces a good clinical response, as described later.
Many physicians became reluctant to employ low-protein dietary therapy, especially after the Evaluation of Modification of Diet in Renal Disease Study (MDRD), in which very low protein diets resulted in a worse prognosis.25) Yet, a critical reading of the MDRD study is necessary, because the results were included in the Japanese Guideline for CKD.26,27) In the MDRD study, 585 participants in group A (GFR 22–55 ml/min/1.73 m2) were divided between ordinary protein intake (1.3 g/kg/day) and low-protein diet (LPD: 0.6 g/kg/day), while 255 group B participants (GFR = 13–24 ml/min/1.73 m2) were divided between LPD (0.6 g/kg/day) and very low protein diet (VLPD: 0.28 g/kg/day). VLPD participants also received 0.28 g/kg/day of amino acid/keto-acid supplements, and multivitamin tablets. Their energy intake was set at 30 kcal/kg b.wt.
The follow-up period was 2.2 years on average, with an additional 9 months of follow-up revealing a poor prognosis in the VLPD group. Early deaths and deaths after hemodialysis were 33 in the LPD group and 20 in the VLPD group. The numbers of patients remaining free from hemodialysis or kidney transplantation were only 5 and 6, respectively. However, the actual protein intake was above the programed level, while the actual energy intake was only 22–20 kcal/kg, which was about 70 percent of our calculated necessary energy intake (calculated from the participants’ body weight).25) Consequently, many patients could be considered to have died from malnutrition. Arguably, the MDRD study failed to show any effects of LPD and VLPD, because of failing to achieve enough energy intake.
The clinically effective dose for CKD is less than 0.5 g/kg b.wt.22) We observed the case of a man diagnosed with renal failure at age 52 years. When he was advised to undergo hemodialysis, his serum creatinine had reached 3 mg/dl, and he changed hospital to receive a low-protein dietary therapy. As soon as his protein intake was limited to 0.75 g/kg, his BUN and creatinine levels markedly decreased. He was able to stabilize his renal function for more than 10 years with a low-protein diet (0.55 to 0.35 g/kg bodyweight). Since the start of low-protein diet, the body weight remained stable. Serum proteins and albumin concentrations were well kept within normal range. He could live without hemodialysis for more than 10 years, and he saved about 50 million yen (500,000 US $) of medical expenses.
To confirm the effect of a low-protein diet, we performed a cross sectional study comparing 10 individuals in each of the following categories: very low protein diet, hemodialysis patients, family members of the patients, and dietitians.28) CKD patients had diverse causes of renal impairment, but they had received 6.7 years of low protein diet on average. Daily protein intake was 0.39 g/kg, 0.55 g/kg for CKD group, and 1.17 g/kg and 1.25 g/kg for family and dietician group, respectively, but energy source is ideal around 0.4 unit/kg b.wt. Vitamin B1, folic acid, carotene, calcium. and phosphate intakes were less than half of the DRI, but none of the patients had signs of deficiency (Table 2). Serum biochemical data showed no difference compared to controls, except for BUN and creatinine in CKD patients. Bone mineral density, and non-fatty tissue weight, measured by a dual-energy X-ray absorptiometry (DEXA) did not show any significant difference among study groups. The CKD subjects were found not to suffer from sarcopenia, osteoporosis, hyperkalemia, hypo-phosphatemia, hyperuricemia or high levels of uric acid, although a slight anemia was observed.
Table 2.
Cross-sectional study comparing CKD patients with very low protein diet and controls. All patients carried out very low protein diet, averagely 6.7 years. Daily protein intake was 0.39, 0.55, 1.17 and 1.25 g/kg b.wt., respectively. The serum biochemistry data showed no difference with controls, except for the BUN and Cr of CKD group
| VLPD | Hemodialysis | Family | Dietitian | ||||||
|---|---|---|---|---|---|---|---|---|---|
| mean | sd | mean | sd | mean | sd | mean | sd | ||
| Nutrient intake | Body weight (kg) | 54.3 | 4.0 | 52.1 | 6.1 | 48.8 | 3.8 | 50.1 | 3.4 |
| Energy (kcal/kg) | 31.7 | 5.0 | 33.5 | 2.8 | 33.1 | 4.5 | 32.8 | 6.9 | |
| Protein (g/kg) | 0.39 | 0.08 | 0.55 | 0.10 | 1.17 | 0.26 | 1.25 | 0.28 | |
| Salt (g/day) | 2.1 | 0.5 | 2.1 | 0.3 | 2.9 | 0.8 | 4.3 | 1.5 | |
| Vit. B1 (ug/day) | 0.21 | 0.09 | 0.19 | 0.05 | 0.45 | 0.14 | 0.55 | 0.21 | |
| Follic acid (ug/day) | 77 | 20 | 94 | 12 | 213 | 73 | 231 | 120 | |
| Carotene (ug/day) | 1072 | 551 | 1451 | 293 | 2923 | 1530 | 2558 | 1477 | |
| Ca (mg/day) | 96 | 50 | 105 | 38 | 302 | 121 | 370 | 199 | |
| P (mg/day) | 227 | 39 | 265 | 14 | 570 | 126 | 595 | 209 | |
| Biochem data | TP (g/dl) | 7.1 | 0.3 | 7.5 | 0.3 | 7.3 | 0.2 | 7.4 | 0.3 |
| Alb (g/dl) | 4.3 | 0.2 | 4.4 | 0.0 | 4.3 | 0.3 | 4.3 | 0.3 | |
| BUN (mg/dl) | 17.7 | 8.7 | 24.7 | 1.1 | 12.3 | 1.7 | 13.7 | 3.6 | |
| Creatinin (mg/dl) | 3.0 | 3.0 | 8.7 | 0.7 | 0.6 | 0.2 | 0.6 | 0.1 | |
A retrospective observational analysis was conducted on 241 CKD patients (151 males and 90 females; average age 51.5 ± 13.8; average 3.0 ± 2.8 years of treatment, ranging from 0.3 to 16.0 years) participating in the Low Protein Diet Practice for Progressive CKD programme.29) With serum creatinine levels less than 5 mg/dL, a decrease in proteinuria was seen within a relatively short period, and estimated at 1.1 g/day protein in the urine according to the protein intake reduction to 0.5 g/kg b.wt. (p < 0.01). These results suggest that LPD protocols should be developed and applied for progressive CKD patients, starting from an early stages of the disease (eGFR <60 ml/1.73 m2).30–32)
A low-protein diet is essential for patients with CKD progressing to renal failure. The appearance of proteinuria or an eGFR less than 60 ml/min/1.73 m2 are recognized threshold indicating when to start a low-protein diet (below 0.5 g/kg b.wt.).
The benefits of a low-protein diet could be summarized as follows: preservation of kidney function, improvement of hyperphosphatemia and hyperkalemia, decrease in urinary protein excretion, improvement of subjective symptoms, reduction of complications, and prolongation of hemodialysis-free interval. Control of patients after the induction of hemodialysis is also better by protein restriction.
Medical rice for kidney disease
In Japan, low-protein rice is available in packaged units containing five different amounts of protein rice. The rice content is 150 g to 180 g, corresponding to 160 kcal (3 serving units of the Diabetic Society). The lowest protein concentration is 0.1 g, which is 1/25 of normal rice. The expiration period is usually 7 months, but some formulas with a 3.5 year ‘best-before’ period can be stored for disaster preparedness.33)
Food claim of Low-protein rice was approved by the Ministry of Health, Labour and Welfare for advanced (level 3 or more) CKD patients,34) but the admitted protein reduction is only 50%. Substituting white rice by low-protein rice is beneficial for maintaining renal function. It is easy to reduce the protein intake among rice-eating Asian people, by using low-protein rice. The recommended protein intake of Japanese is 60 g for males and 50 g for females, and almost 20–30 g protein comes from rice.
Low protein rice is made by the enzymatic digestion of polished raw rice. After wash out aminoacids and short peptide, the rice is dried and boiled under the pressure steam in a plastic tray. The market price is 190 yen/pack, and it is much cheaper compared to the price of hemodialysis.
The nutritional aspects of low-protein rice are shown in Table 3 (Table 3). The energy intake is preserved and the protein concentration is well controlled. Low potassium and phosphate concentrations in low-protein rice are additional benefits for CKD patients.
Table 3.
Nutrients of low protein rice per pack
| Energy | Protein | Potassium | Phosphate | |
|---|---|---|---|---|
| Whole rice | 168 kcal | 2.5 g | 29 mg | 34 mg |
| 1/3 low protein rice | 158 | 0.81 | 1.3 | 15 |
| 1/6 low protein rice | 161 | 0.5 | 0.6 | 15 |
| 1/10 low protein rice | 167 | 0.25 | 2.2 | 13 |
| 1/20 low protein rice | 167 | 0.12 | 0.3 | 14 |
| 1/25 low protein rice | 167 | 0.1 | 0.0 | 13 |
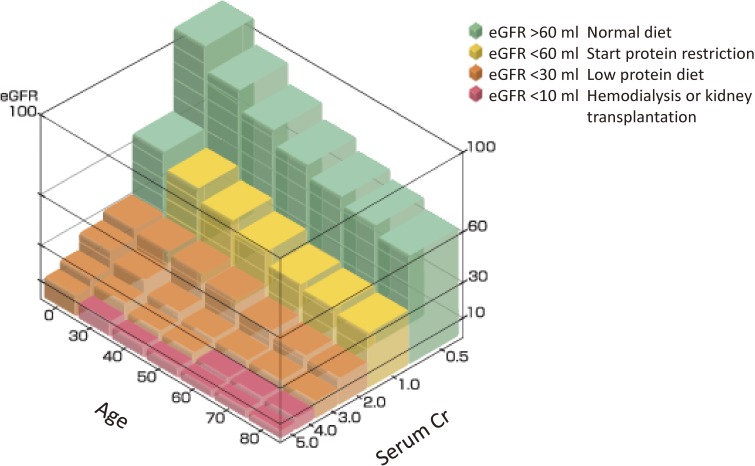
We propose a simple chart to decide on the degree of protein restriction according to the eGFR and age. When the eGFR is above 60 ml/min/1.73 m2 body surface, protein restriction is not necessary, although the recommended dose is 0.8 g/kg b.wt. When the eGFR becomes lower than 60 ml/min, protein restriction should start, aiming at a daily intake of 0.5 g/kg b.wt. When the eGFR becomes less than 30 ml/min, further protein restriction is necessary (0.4 g/kg b.wt.). If the eGFR decreases under 10 ml/min, the patient needs hemodialysis or kidney transplantation.
The eGFR is easily estimated from a chart checking age and serum creatinine. For example, if the patient is 40 years-old and the serum creatinine is 2.0 mg/dL, the color of the crossing area is orange, so the corresponding protein restriction should be 0.4 g/kg b.wt. (Fig. 2).
Figure 2.
Simple step model of eGFR by age and serum creatinine. When the eGFR is above 60 ml/min/1.73 m2 body surface, protein restriction is not necessary, although the recommended dose is 0.8 g/kg b.wt. When the eGFR is lower than 60 ml/min, protein restriction should start, aiming at a daily intake of 0.5 g/kg b.wt. When the eGFR becomes less than 30 ml/min/, further protein restriction is necessary (0.4 g/kg). If the eGFR decreases under 10 ml/min/, the patient needs hemodialysis or kidney transplantation. The eGFR is easily estimated from a chart computing age and serum creatinine. For example, if the patient is 40-years old and the serum creatinine is 2.0 g/dL, the color of the crossing area is orange, so the corresponding protein restriction should be 0.4 g/b.wt.
Obesity and hypertension should be controlled for preventing CKD progression.35,36) We propose a new energy unit (E-unit) of 80 kcal, such that healthy adults’ caloric needs (in energy units) can be calculated with a simple equation.37) The necessary amount of energy units for daily intake is given by the product of the body weight (kg)*0.4. This remains constant throughout most of the adult life for both sexes, for the elderly and even for pregnant women. This formula, in conjunction with the comprehensive food icon (FI), which is a colored guide for balanced nutrition used in Shokuiku (food and nutrition education) in Japan, will allow individuals to adjust their energy intake to achieve an adequate energy balance. The comprehensive food icon includes information about (i) energy units, (ii) the balance of major nutrients, and (iii) antioxidant units (AOU) as a surrogate marker of vegetables and fruits. The widespread adoption and utilization of this food icon by food producers, suppliers, and consumers will help people who want to control their body weight. Life behavior modification is important in case of CKD prevention.38)
The food industry can play a significant role in promoting healthy diets by reducing the fat, sugar and salt contents of processed foods.37) They can also provide useful and comprehensive information to consumers, using the proposed FI. Food labels with detailed contents of nutrients are often too complicated and difficult to understand by ordinary consumers. Consumers primarily want to eat cheap and tasty foods, and are generally not concerned with the exact nutrient contents of foods.
Export of low protein rice to Southeast Asia
Low protein rice for Shanghai study.
We performed a RCT in Huadong Hospital (Shanghai) with Professor Sun,39) to examine the effect of 12-weeks dietary therapy for CKD patients. PLC rice (FORICA FOODS Co., LTD) provided 3 packs/day containing 1.35 g proteins and 900 kcal energy. Compared with baseline levels, the total dietary energy intake increased from 1606 kcal/d (27.9 kcal/kg b.wt.) to 1748 kcal (30.8 kcal/kg b.wt.). The total serum protein concentration increased from 74 g/L to 77 g/L (p > 0.05). Meanwhile, body weight, BMI, and hemoglobin did not significantly change. After 12 weeks, urinary protein levels decreased from 0.40 g/d to 0.10 g/d (p < 0.05). Urine albumin also decreased from 130.8 mg/24 h to 60.8 mg/d (p < 0.05). Twenty-four urinary protein excretion, 24 h urinary albumin excretion, and urinary albumin/creatinine ratio decreased by 63.7%, 55.0% and 52.0% respectively.39)
Low-protein rice was well accepted by Chinese CKD patients as a staple food. It is an important tool for CKD dietary therapy as it increases the intake of energy and micronutrients, and it improves the nutritional status. Unfortunately, this RCT was interrupted by the Fukushima Atomic Plant Explosion, because rice exports to China have been prohibited since then.
Low-protein rice plant in Bangkok.
The increasing trend in CKD is the same in Thailand. According to the National Health Security Office, the number of CKD patients above stage 3 is estimated to be 10 million (17.6%).40) Expenditures reached 160 million Bahts in 2008, but they steadily increased to 3,858 million Bahts in 2012.
So, we have planned to build a small plant in Thailand, because the number of CKD patients is increasing in Thailand in parallel to obesity, hypertension and diabetes. JICA supported our plan to build a small plant in the Kassart University with the collaboration of Professor Patcharee Tungtrakul. Researchers from FORICA Food Co., Ltd. succeeded in making a 1/10 low-protein rice from Indica rice. As soon as September 2016, we are ready to start a RCT in the Air Base Hospital in Bangkok.
A long-term and large sample size study is planned in Thailand, to confirm the protective effects of low-protein rice on CKD progression. Medical expenditures for CKD patients has reached record amounts of 200,000 Baht per capita/year. In terms of yearly number of hospitalizations in Thailand, diabetes steadily increased from 33/100,000 in 1985 to 91/100,000 in 1994, and 737/100,000 in 2009. The prevalence of hypertension was 982/100,000 in 2009. The number of overweight persons (BMI > 25) is estimated to be 8.8 million. Obesity (BMI > 30) affects 1.7 million people, hypertension 4.3 million, and diabetes 1.8 million. This forebodes the progression to CKD to become a serious problem.
Conclusion
CKD becomes more common with increasing age. After the age of 40, kidney filtration begins to fall by approximately 1% per year. On top of the natural aging of the kidneys, many conditions which damage the kidneys are more common in older people including diabetes, high blood pressure and heart diseases.
This is important because CKD increases the risk of heart attack and stroke, and in some cases can progress to kidney failure requiring dialysis or transplantation. The appearance of micro-albuminemia and/or low eGFR is an opportunity to start low-protein diet to prevent complications and improve the quality of life.41–43)
The recent popularity of low-carbon diets entails the risk of high-protein and high-lipid regimens to result in renal dysfunction and/or hyperlipidemia with atherosclereosis.44,45) In addition, meat consumption is a risk factor of diabetes.46)
More studies should be conducted on dietary therapies, including low-carbohyrate ketogenic diet.47)
Acknowledgement
The author thanks Dr. Shigeru Beppu (FORICA Food Co., Ltd.), Dr. Sun Jing Jang and Dr. Patchalee for their constructive discussion of clinical trial in Shanghai and Bangkok. Dr. Philippe Calain contributed to the editing of English language.
COI: The author does not have any conflict of interest with any company.
Abbreviations
- eGFR
estimated glomerular filtration rate.
Profile
Shaw Watanabe was born in Pyongyang in 1941 and graduated from Keio University School of Medicine in 1965. After completing his internship, he entered Keio Graduate School and majored in human pathology. As a distinguished hematopathologist at the National Cancer Institute (U.S.A.) and the Pathology Division, National Cancer Center (Japan), he defined criteria for T-cell lymphoma/leukemia and histiocytic tumors. He was then appointed Director of the Epidemiology Division, National Cancer Center (1985–1996), where he established a population based cohort study (JPHC). As a consultant, he also contributed to the success of the WHO Framework Convention on Tobacco Control. After being diagnosed with diabetes at the age of 50, he moved to Tokyo University of Agriculture (1995–2005) to promote diabetic control through appropriate diet and physical activity. As a professor of public nutrition, he became one of the pioneers of the use of functional foods. He was promoted Director General, National Institute of Health and Nutrition (2005–2009), and he was commissioned by the Cabinet Office to develop a national policy on Shoku-iku (Eating education). After retirement he became the President of the Life Science Promoting Association (2009–present) and the President of the Japanese Society of Integrative Medicine (2011–present). He chaired the Asia Pacific Conference on Clinical Nutrition 2014 in Tokyo and the East Asia Conference on Standardization of Rice Function in Kyoto. He is currently the chief editor of ‘Clinical & Functional Nutriology’ ‘Life Science’, and the editor-in-chief of the ‘Diabetes Research Open Journal’. He is an emeritus member of the Japan Pathological Society the Japan Epidemiology Society, the Society of Functional Food Factors, and other academic societies. He served also as member of scientific boards appointed by: the Ministry of Health, Labour and Welfare; the Ministry of Agriculture, Forest and Fishery; and the Cabinet Office and Environmental Agency.
For his accomplishments, he received the WHO Tobacco or Health Medal (1991), the Japan Medical Association Award, the Japan Epidemiological Association Award, and the Award of Contribution to Society from the Keio University Medical School.
References
- 1).Jha V., Garcia-Garcia G., Iseki K., Li Z., Naicker S., Plattner B., Saran R., Wang A.Y., Yang C.W. (2013) Chronic kidney disease: global dimension and perspectives. Lancet. 382, 260–272, doi:10.1016/S0140-6736(13)60687-X. [DOI] [PubMed] [Google Scholar]
- 2).http://www.worldkidneyday.org/faqs/chronic-kidney-disease/.
- 3).Zhang Q.L., Rothenbacher D. (2008) Prevalence of chronic kidney disease in population-based studies: systematic review. BMC Public Health 8, 117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4).Levey A.S., Atkins R., Coresh J., Cohen E.P., Collins A.J., Eckardt K.U., Nahas M.E., Jaber B.L., Jadoul M., Levin A., Powe N.R., Rossert J., Wheeler D.C., Lameire N., Eknoyan G. (2007) Chronic kidney disease as a global public health problem: approaches and initiatives — a position statement from Kidney Disease Improving Global Outcomes. Kidney Int. 72, 247–259. [DOI] [PubMed] [Google Scholar]
- 5).El Nahas M. (2005) The global challenge of chronic kidney disease. Kidney Int. 68, 2918–2929. [DOI] [PubMed] [Google Scholar]
- 6).Zimmer P., Alberti K.G., Shaw J. (2001) Global and societal implications of the diabetes epidemic. Nature 414, 782–787. [DOI] [PubMed] [Google Scholar]
- 7).Atkins R.C., Zimmet P. (2010) World Kidney Day 2010: diabetic kidney disease-act now or pay later. Am. J. Kidney Dis. 55, 205–208. [DOI] [PubMed] [Google Scholar]
- 8).Bilous R. (2008) Microvascular disease: what does the UKPDS tell us about diabetic nephropathy? Diabet Med. 25 (Suppl. 2), 25–29. [DOI] [PubMed] [Google Scholar]
- 9).Couser W.G., Remuzzi G., Mendis S., Tonelli M. (2011) The contribution of chronic kidney disease to the global burden of major noncommunicable diseases. Kidney Int. 80, 1258–1270. [DOI] [PubMed] [Google Scholar]
- 10).Reutens A.T. (2013) Epidemiology of diabetic kidney disease. Med. Clin. North Am. 97, 1–18, doi:10.1016/j.mcna.2012.10.001. [DOI] [PubMed] [Google Scholar]
- 11).National Kidney Foundation (2002) K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am. J. Kidney Dis. 39 (Suppl. 1), S1–S266. [PubMed] [Google Scholar]
- 12).Watanabe S., Morita A., Aiba N., Miyachi M., Sasaki S., Morioka M., Noda M., Takebayashi T., Kimira M., SCOP (2007) Study design of the SAKU Control Obesity Program (SCOP). Anti-aging Med. 7, 71–73. [Google Scholar]
- 13).Huang E.S., Basu A., O’Grady M., Capretta J.C. (2009) Projecting the future diabetes populaton size and related costs for the U.S. Diabetes Care 32, 2225–2229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14).Shibuya K., Hashimoto H., Ikegami N., Nishi A., Tanimoto T., Miyata H., Takemi K., Reich M.R. (2016) Future of Japan’s system of good health at low cost with equity: beyond universal coverage. Lancet 378, 1265–1273. [DOI] [PubMed] [Google Scholar]
- 15).Ninomiya T., Kiyohara Y., Tokuda Y., Doi Y., Arima H., Harada A., Ohashi Y., Ueshima H., Japan Arteriosclerosis Longitudinal Study Group (2008) Impact of kideny disease and blood pressure on the development of cardiovascular disease: an overview from the Japan Arteriosclerosis Longtudinal Study. Circulation 118, 2694–2701. [DOI] [PubMed] [Google Scholar]
- 16).Morimoto A., Tatsumi Y., Deura K., Mizuno S., Ohno Y., Miyamatsu N., Watanabe S. (2014) Impact of impaired insulin secretion and insulin resistance on the incidence of type 2 diabetes mellitus in a Japanese population: the Saku study. Diabetologia, doi:10.1007/s00125-013-2932-y. [DOI] [PubMed] [Google Scholar]
- 17).Imai E., Horio M., Iseki K., Yamagata K., Watanabe T., Hara S., Ura N., Kiyohara Y., Hirakata H., Moriyama T., Ando Y., Nitta K., Inaguma D., Narita I., Iso H., Wakai K., Yasuda Y., Tsukamoto Y., Ito S., Makino H., Hishida A., Matsuo S. (2007) Prevalence of chronic kidney disease (CKD) in the Japanese general population predicted by the MDRD equation modified by a Japanese coefficient. Clin. Exp. Nephrol. 11, 156–163. [DOI] [PubMed] [Google Scholar]
- 18).http://www.mhlw.go.jp/toukei/saikin/hw/k-iryohi/14/dl/kekka.pdf.
- 19).Kanazawa Y., Nakao T. (2009) Nutritional and dietary management of diabetic nephropaty: Focus on low protein diets for stages 4 and 5 CKD. Clin. Funct. Nutr. 1, 31–34. [Google Scholar]
- 20).Piccoli G.B., Capizzi I., Vigotti F.N., Leone F, D’Alessandro C., Giuffrida D., Nazha M., Roggero S., Colombi N., Mauro G., Castelluccia N., Cupisti A., Avagnina P. (2016) Low protein diets in patients with chronic kidney disease: a bridge between mainstream and complementary-alternative medicines? BMC Nephrol. 17, 76 doi:10.1186/s12882-016-0275-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21).Watanabe S. (2014) Effects of low carbohydrate diet for obesity control: meta-analysis. Clin. Funct. Nutr. 6, 300–303. [Google Scholar]
- 22).Saito J. (2009) Effects of low protein diet for chronic renal failure. Clin. Funct. Nutr. 1, 251–256. [Google Scholar]
- 23).Morita A., Watanabe S. (2011) Dietary therapy for the elderly. Clin. Funct. Nutr. 3, 186–190. [Google Scholar]
- 24).Azadbakht L., Atabak S., Esmaillzadeh A. (2008) Soy protein intake, cardiorenal indices, and C-reactive protein in type 2 diabetes with nephropathy: a longitudinal randomized clinical trial. Diabetes Care 31, 648–654. doi:10.2337/dc07-2065. [DOI] [PubMed] [Google Scholar]
- 25).Watanabe S. (2009) Evaluation of Modification of Diet in Renal Disease (MDRD) Study. Clin. Funct. Nutr. 1, 238–241. [Google Scholar]
- 26).Japan Nephrology Society (2014) Dietary recommendations for chronic kidney disease. Jpn. J. Renal. Soc. (Nichi-jin-kai-shi) 56, 553–599. [Google Scholar]
- 27).Yamagata K., Ideura T., Watanabe S. (2009) Low protein diet therapy for renal failure. Clin. Funct. Nutriol. 1, 230–236. [Google Scholar]
- 28).Watanabe S., Noboru M., Yasunaga M., Ideura T. (2010) A cross-sectional study on the effects of long term very low protein diets in patients with chronic kidney disease. Serum and urine DEXA and amino acid profiles. Anti-aging Med. 7, 7–13. [Google Scholar]
- 29).Mizuno S. (2009) A secondary analysis of Ideura Data of low protein diet practice for progressive chronic kidney disease patients. Clin. Funct. Nutr. 1, 242–245. [Google Scholar]
- 30).James M.T., Hemmelgam B.R., Tonelli M. (2010) Early recognition and prevention of chronic kidney disease. Lancet. 375, 1296–1309. [DOI] [PubMed] [Google Scholar]
- 31).Hemmelgarn B.R., Manns B.J., Lloyd A., James M.T., Klarenbach S., Quinn R.R., Wiebe N., Tonelli M., Alberta Kidney Disease Network (2010) Relaton between kidney function, proteinuria, and adverse outcomes. JAMA 303, 423–429. [DOI] [PubMed] [Google Scholar]
- 32).Rifkin E.D., Katz R., Chonchol M., Fried L.F., Cao J., de Boer I.H., Siscovick D.S., Shlipak M.G., Sarnak M.J. (2010) Alubuminuria, impaird kidney function and cardiovascular outcomes or mortality in the elderly. Nephrol. Dial Transplant. 25, 1560–1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33).Watanabe S., Hirakawa A., Nishijima C., Ohtsubo K., Nakamura K., Beppu S., Tungtrakul P., Sun J.-Q., Tee E.S., Tsuno T., Ohigashi H. (2016) Food as medicine: The new concept of “medical rice”. Adv. Food Technol. Nutr. Sci. Open J. 2, 38–50. doi:10.17140/AFTNSOJ-2-129. [Google Scholar]
- 34).Ministry of Health, Labour and Welfare (2007) Standard of foods for diseased. www.mhlw.go.jp/shingi/2007/11/dl/s1121-13m.pdf.
- 35).Zoungas S., de Galan B.E., Ninomiya T., Grobbee D., Hamet P., Heller S., MacMahon S., Marre M., Neal B., Patel A., Woodward M., Chalmers J., ADVANCE Collaborative Group (2009) Combined effects of routine blood pressure lowering and intensive glucose control on macrovascular and microvascular outcomes in patients with type 2 diabetes: new results from the ADVANCE trial. Diabetes Care. 32, 2068–2074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36).Grimm R.H., Jr., Svendsen K.H., Kasiske B. (1997) Proteinuria is a risk factor for mortality over 10 years of follow-up. MRFIT Research Group. Multiple Risk Factor Intervention Trial. Kidney Int. Suppl. 63, 510–514. [PubMed] [Google Scholar]
- 37).Hirakawa A., Melby M.K., Watanabe S. (2016) Comprehensive food labeling for obesity control. Adv. Obesity Weight Manage. Cont. 4, 00088. [Google Scholar]
- 38).Yamagata K., Yamagata K., Makino H., Iseki K., Ito S., Kimura K., Kusano E., Shibata T., Tomita K., Narita I., Nishino T., Fujigaki Y., Mitarai T., Watanabe T., Wada T., Nakamura T., Matsuo S., Study Group for Frontier of Renal Outcome Modifications in Japan (FROM-J) (2016) Effect of behavior modification on outcome in early- to moderate-stage chronic kidney disease: A cluster-randomized trial. PLoS One 11, e0151422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39).Sun J.-Q., Wang Y. (2014) Effects of low-protein rice on nutrition status and renal function in patients with chronic kidney disease: A pilot study. East-Asia Conference on Standardization of Rice Function, Kyoto, pp. 49–50. [Google Scholar]
- 40).http://www.who.int/bulletin/volumes/88/2/10-010210/en/.
- 41).Romundstad S., Holmen J., Kvenild K., Hallan H., Ellekjaer H. (2003) Microalbuminuria and all-cause mortallity in 2,089 apparently healthy individuals: a 44-year follow-up study. The Nord-Trondelag Health Study (HUNT), Norway. Am. J. Kidney Dis. 42, 466–473. [DOI] [PubMed] [Google Scholar]
- 42).Gansevoort R.T., de Jong P.E. (2009) The case for using albuminuria instaging chronic didney disease. J. Am. Soc. Nephrol. 20, 465–468. [DOI] [PubMed] [Google Scholar]
- 43).Verhave J.C., Gansevoort R.T., Hillege H.L., Bakker S.J., De Zeeuw D., de Jong P.E., PREVEND Study Group (2004) An elevated urinary albumin excretion predicts de novo development of renal function impairment in the general population. Kidney Int. 66 (Suppl. 92), S18–S21. [DOI] [PubMed] [Google Scholar]
- 44).van der Velde M., Halbesma N., de Charro F.T., Bakker S.J., de Zeeuw D., de Jong P.E., Gansevoort R.T. (2009) Screening for albuminuria identifies individuals at increased renal risk. J. Am. Soc. Nephrol. 20, 852–862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45).Watanabe S. (2015) Current controversies around carbohydrate restriction. Diabetes Res. Open J. 1, e7–e10. [Google Scholar]
- 46).Kurotani K., Nanri A., Goto A., Mizoue T., Noda M., Oba S., Kato M., Matsushita Y., Inoue M., Tsugane S., Japan Public Health Center-based Prospective Study Group (2013) Red meat consumption is associated with the risk of type 2 diabetes in men but not in women: a Japan Public Health Center-based Prospective Study. Br. J. Nutr. 110, 1910–1918. doi:10.1017/S0007114513001128. [DOI] [PubMed] [Google Scholar]
- 47).Watanabe S., Hirakawa A., Aoe S., Fukuda K., Muneta T. (2016) Basic keton engine and booster glucose engine. Diabetes Res. Open J. 2, 14–23. doi:10.17140/DROJ-2-125. [Google Scholar]