Abstract
The tumor microenvironment (TME) in the liver plays an important role in primary and metastatic liver tumor formation and tumor growth promotion. Cellular and non-cellular components of the TME significantly influence tumor development, growth, metastatic spread, anti-tumor immunity and response to tumor therapy. The cellular components of the TME in the liver not only consist of infiltrating immune cells, but also of liver-resident cells such as liver sinusoidal endothelial cells (LSEC) and hepatic stellate cells (HSC), which promote tumor growth by negatively regulating tumor-associated immune responses. In this review, we characterize cells of the TME with pro- and anti-tumor function in primary and metastatic liver tumors. Furthermore, we summarize mechanisms that permit growth of hepatic tumors despite the occurrence of spontaneous anti-tumor immune responses and how novel therapeutic approaches targeting the TME could unleash tumor-specific immune responses to improve survival of liver cancer patients.
Keywords: Liver immunology, tumor immunology, hepatocellular carcinoma, liver metastases, immunotherapy, tumor microenvironment
1. Introduction
The TME of solid tumors has gained enormous attention in recent years, as it has become clear that its cellular and non-cellular components significantly influence tumor development and growth, metastatic spread, anti-tumor immunity and response to tumor therapy (Hanahan & Weinberg, 2011; Joyce & Fearon, 2015). The cellular component of the TME can be subdivided into immune cells and non-immune cells. Lymphocytes and myeloid cells form the immune cell compartment and endothelial cells, pericytes and fibroblast form the non-immune cell compartment of the TME. Importantly, the liver contains specialized endothelial cells and specialized fibroblast, namely LSEC and HSC, respectively, that perform immune functions (Jenne & Kubes, 2013; Thomson & Knolle, 2010). All of these aforementioned cell types can influence the tumor and in turn can be influenced by the tumor through a variety of mechanisms. Thus, understanding the tumor-promoting, therapy-inhibiting mechanisms of the TME is instrumental for the design of successful treatment approaches.
In the liver, it is important to distinguish between the TME of primary hepatic tumors, commonly residing in inflamed liver tissue, and the TME of liver metastases of non-hepatic tumors, residing in otherwise healthy liver tissue. Hepatic immune cells play an important role in primary liver cancer formation and growth and also in ‘preparing’ the liver for metastatic seeding of tumors from different primary sites. In patients with liver metastasis but without underlying liver disease, the primary tumor, e.g. colon carcinoma, recruits myeloid cells to the liver to enable metastatic seeding of colon cancer cells (L. Zhao, et al., 2013). These myeloid cells also induce down-regulation of angiopoietin-like 7 protein in cancer cells to promote angiogenesis and metastasis formation (Lim, et al., 2015). Moreover, hepatic resident non-immune cells can also support metastasis development in the liver. Because of the liver’s unique anatomical location and metabolic functions in the human body, the resident immune and non-immune cells of the liver are endowed with distinctive functional properties (Thomson & Knolle, 2010). For example, LSEC sample the blood, which contains nutrients and microbial antigens from the gut, and induce T cell tolerance to antigens to which no preexisting immunity exists (Schurich, et al., 2009). However, the same mechanism also leads to hepatic tolerance towards tumor-associated antigens and enables metastatic spread into the liver (Arteta, et al., 2010; Hochst, et al., 2012), illustrating one non-immune cell-mediated mechanism of tumor immune escape that is unique to the liver.
The TME of hepatocellular carcinomas (HCC), the most frequent primary liver cancer, is even more complex, because HCC usually develop in chronically inflamed livers. Indeed, chronic liver inflammation, induced by non-alcoholic steatohepatitis (NASH) or viral hepatitis, can progress to liver cirrhosis and eventually to carcinomas (El-Serag & Rudolph, 2007). Therefore, both the tumor and the underlying chronic liver inflammation shape the TME of HCC. This is particularly important for the function of myeloid cells. Macrophages, for example, can prevent outgrowth of HCC in otherwise healthy livers by clearing precancerous hepatocytes (Kang, et al., 2011), but they can also initiate HCC development in inflamed livers (Lanaya, et al., 2014). The chronic inflammation that drives liver cancer formation is also responsible for the regular occurrence of tumor neoantigens, which can trigger an anti-tumor T cell immune response (Schumacher & Schreiber, 2015). Consequently, tumor antigen-specific T cells can infiltrate HCC and several HCC-associated epitomes that are recognized by the infiltrating T cells have been identified (Flecken, et al., 2014; Mizukoshi, et al., 2011). Despite the recognition of and infiltration into the tumor by the immune system, HCC remains a cancer with a high mortality and poor 5-year survival (Njei, Rotman, Ditah, & Lim, 2015). This suggests that potent tumor-associated immunosuppressive mechanisms are in place to escape the immune response in patients with HCC. Indeed, myeloid-derived suppressor cells (MDSC) and regulatory T cells (T reg) are commonly found immune suppressive cells in HCC (Fu, et al., 2007; Hoechst, et al., 2008). Together, these findings underscore the significance of immune cells in primary and metastatic tumor development and tumor control in the liver (Figure 1).
Fig. 1.
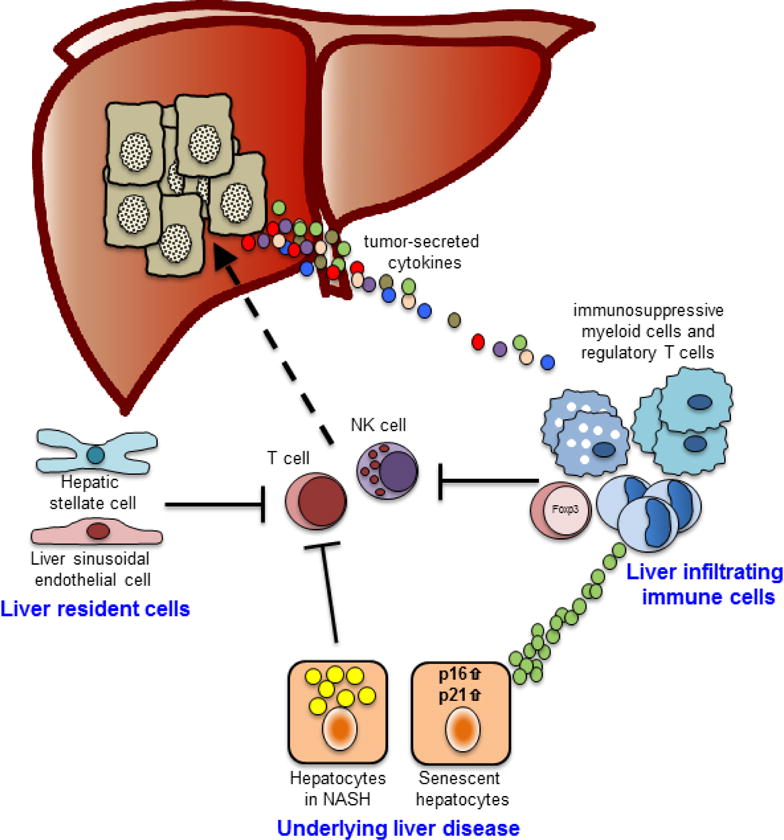
Components of the tumor microenvironment restraining anti-tumor immune cells. Following activation, T cells and NK cells target tumors for tumor cell killing. However, liver resident cells and/or liver infiltrating immune cells such as immunosuppressive myeloid cells or regulatory T cells inhibit their anti-tumor function. Furthermore, the underlying liver disease also negatively regulates T cell and NK cell function either directly by inducing of CD4 T cell death or indirectly by recruiting imunosuppressive myeloid cells.
Thus, the tumor, immune cells and non-immune cells, like LSEC, regulate pro and anti-tumor immune responses in hepatic cancers. In this review, we will highlight the mechanisms that tumors employ to inhibit anti-tumor immune responses. Furthermore, we will demonstrate how hepatic immune cells and non-immune cells contribute to creating an immunosuppressive TME that promotes tumor immune escape and tumor growth. Strategies to therapeutically modulate hepatic immune cell functions will also be discussed (Table 1).
Table 1.
Selected potential treatment approaches for HCC or liver metastases (with the exception of radiofrequency ablation, these therapies have not been approved for the treatment of patients with HCC thus far).
| Targeted immune cell/therapeutic strategy | Therapeutic agent/intervention | Mechanism of action |
|---|---|---|
| Anti-tumor immune cells | ||
|
Cytotoxic T cells enhancing T cell tumoricidal activity |
Nivolumab (anti-PD-1 antibody), Ipilimumab (anti-CTLA-4 antibody) |
blocking T cell inhibitory signals, so called ‘checkpoints’ |
| anti-4-1BB agonistic antibody, anti- OX40 agonistic antibody | activating T cell stimulatory receptors | |
|
Dendritic cells inducing tumor-specific T cell responses |
radiofrequency ablation of HCC |
tumor cell death causes release of tumor-associated antigens ➔ presentation to reactive T cells |
| in vitro-transfection of autologous dendritic cells with heat shock protein 70 mRNA | activation/expansion of T cells targeting heat shock protein 70-expressing HCC | |
|
NK cells enhancing NK cell tumoricidal activity |
Lirilumab (anti-KIR antibody) |
blocking NK cell inhibitory signals |
| Pro-tumor immune cells | ||
|
MDSC reducing MDSC-mediated immunosuppression |
anti-GM-CSF antibody, anti-CXCR2 antibody |
inhibition of MDSC expansion and recruitment |
| Atezolizumab (anti-PD-L1 antibody) | blockade of T cell inhibition through PD-L1 on MDSC | |
| Taldalafil (PDE-5 inhibitor) | blockade of MDSC-mediated T cell inhibition | |
|
Macrophages reducing macrophage-mediated immunosuppression |
CSF-1R inhibitors |
inhibition of macrophage accumulation |
|
Regulatory T cells reducing T reg-mediated immunosuppression |
anti-TGFβ antibody |
inhibition of T reg expansion and blockade of T reg-mediated T cell inhibition |
2. Cells of the hepatic tumor environment and their regulation of tumor growth
2.1 Anti-tumor immune cells in the liver
Three immune cell subsets have been shown to be capable of killing tumor cells and eradicating established tumors, namely cytotoxic T cells (CTL), Natural Killer cells (NK cells) (Figure 2) and macrophages (resident macrophages in the liver are also termed Kupffer cells) (Chao, Weissman, & Majeti, 2012; Pardoll, 2012; Vivier, Ugolini, Blaise, Chabannon, & Brossay, 2012). As discussed below, the first two mentioned subsets are considered to be exclusively anti-tumorigenic in liver cancer. However, because of their plasticity, macrophages can have both pro- and anti-tumor effector functions. After tumor formation, tumors skew macrophages towards an immunosuppressive tumor-promoting phenotype. Therefore, we discuss these cells in the section “Tumor promoting cells in the hepatic tumor environment” below. Similarly, dendritic cells can exert pro- as well as anti-tumor functions. Although dendritic cells do not directly cause tumor cell death, they initiate the anti-tumor T cell response and are therefore discussed in “anti-tumor immune cells in the liver”, in the context of CTL.
Fig. 2.
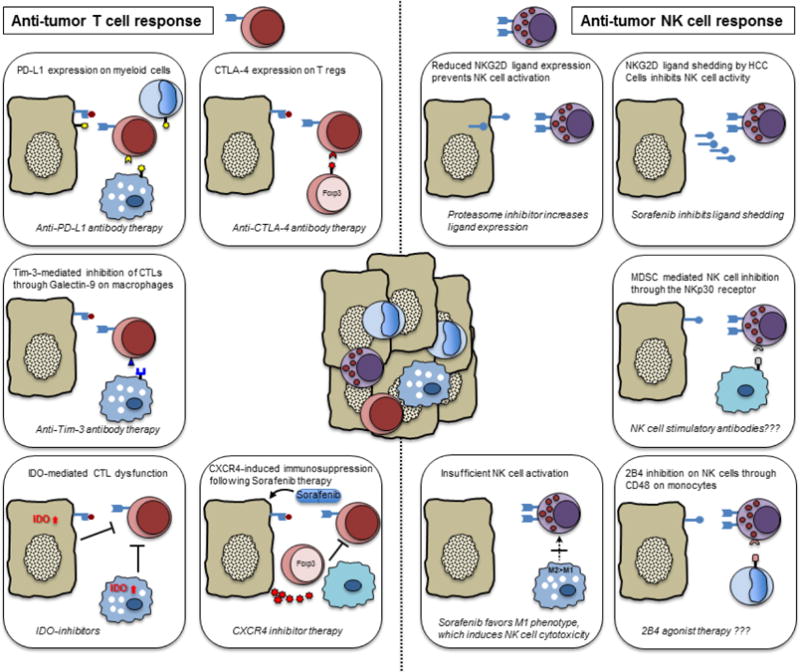
Anti-tumor immune cells and mechanisms restraining their anti-tumor function. Molecular mechanisms inhibiting anti-tumor T cells (left) and anti-tumor NK cells (right) are shown. Therapeutic strategies to reverse this inhibition in order to unleash T cell and NK cell function are provided at the bottom of each box. Question marks indicate proposed, not yet studied approaches.
Cytotoxic T cells (CTL)
Unleashing anti-tumor T cell responses in some solid cancers, primarily melanoma, through so-called ‘checkpoint-inhibitors’, like anti-CTLA-4 and anti-PD-1 antibodies, have recently produced astounding results in a fraction of cancer patients, achieving long-term remissions (Robert, et al., 2015; Schadendorf, et al., 2015). These results uncovered the huge potential that T cell responses against tumors have. The reason as to why this treatment is only successful in some cancers has been partially attributed to the frequency of somatic mutations in tumor cells that generate neoantigens, which allow for the induction of an anti-tumor T cell response (Schumacher & Schreiber, 2015). These tumor-associated antigens enable recognition of the tumor whereas ‘Checkpoint-inhibitors’ disinhibit T cells by interfering with the inhibitory signal that would otherwise be relayed through receptors on T cells, which down-modulate the amplitude of T cell activation (Pardoll, 2012).
In HCC patients, tumor-specific T cell responses against numerous HCC-associated antigens have been reported and patients with tumor-specific T cell responses have improved progression-free survival (Flecken, et al., 2014; Mizukoshi, et al., 2011). Furthermore, local ablative therapies like radiofrequency ablation or transarterial chemoembolization, which are primary HCC treatment modalities, enhance the anti-tumor T cell response by triggering tumor cell death (Hiroishi, et al., 2010; Mizukoshi, et al., 2013). In one study, radiofrequency ablation not only enhanced the number of tumor-specific T cells, it also induced responses against additional tumor-associated antigens, which were not detectable before ablation (Mizukoshi, et al., 2013), indicating that T cells can successfully recognize HCC. However, in patients with spontaneous tumor-specific T cell responses, the function of these T cells is impaired, but their activation could be enhanced in vitro by administration of anti-CTLA-4 antibodies (Mizukoshi, et al., 2011). ‘Checkpoint-inhibitors’ are therefore plausible treatment approaches in HCC patients and are currently evaluated in clinical trials using the anti-CTLA-4 antibody (Duffy AG, et al., 2015; Sangro, et al., 2013).
Anti-PD-1 antibody is another ‘checkpoint-inhibitor’ with the potential to improve patient survival. Expression of PD-L1, the ligand for PD-1, was increased on peritumoral monocytes (Kuang, et al., 2009) and intratumoral Kupffer cells (K. Wu, Kryczek, Chen, Zou, & Welling, 2009) of HCC patients. Increased expression of PD-L1 on either monoctyes or Kupffer cells was also associated with poorer survival, indicating that HCC patients could benefit from anti-PD-1 antibody therapy as well. Indeed, a Phase I/II trial studying anti-PD-1 antibody in patients with advanced HCC showed promising results (El-Khoueiry AB, et al., 2015). 18% of patients had a partial response and 5% had a complete remission, which lasted 14 – 17+ months.
A third albeit less studied inhibitory ligand on Kupffer cells within HCC tumors is Galectin-9, which interacts with Tim-3 on T cell (Li, et al., 2012), causing reduced T cell activation and worsening of prognosis. However, antibodies blocking Tim-3 are still in preclinical evaluation and have not yet reached clinical trials. A similar but slightly different approach to enhance anti-tumor T cell responses is to activate surface molecules that directly stimulate T cells instead of preventing T cell inhibition. Examples are 4-1BB and OX40, which are stimulatory receptors on T cells (L. Chen & Flies, 2013). In a murine orthotropic liver tumor model, the triple combination of anti-PD-L1 antagonistic antibody and anti-4-1BB and anti-OX40 agonistic antibodies lead to significant improvement of survival, which was dependent on tumor infiltrating T cells (Morales-Kastresana, et al., 2013).
In summary, patients with HCC develop spontaneous anti-tumor T cell responses, which, however, do not lead to tumor growth control. Enhancing these T cell responses through novel cancer immunotherapy approaches might unleash their full potential, significantly improving patient survival.
Dendritic cells
In order for CTL to carry out their effector function of killing tumor cells, T cells require careful stimulation and subsequent activation to exert their tumor-specific anti-tumor properties. Dendritic cells provide these cues, i.e. antigen-specific T cell receptor engagement, co-stimulatory molecule activation and paracrine cytokine signaling, necessary for proper anti-tumor T cell function. Following phagocytosis of tumor-associated antigens, dendritic cells themselves are activated through pattern recognition receptors, such as toll-like receptors, or cytokine receptor signaling. This enables dendritic cells to induce a tumor-specific T cell response by presenting epitopes of the captured antigens to CTL. Because of their central role in initiating this response, efforts have been made to employ dendritic cells therapeutically to improve spontaneously occurring T cell responses. In patients with HCC, one such effort is to enhance phagocytosis of tumor-associated antigens and dendritic cell activation in situ by causing tumor cell death and release of antigens through local tumor ablation, e.g. radiofrequency ablation (Ali, et al., 2005). Another approach is to expand and activate autologous dendritic cells in vitro and reinfuse these cells into the tumor following transarterial chemoembolization. This treatment yielded prolonged recurrence-free survival in HCC patients, albeit in comparison with historical controls (Nakamoto, et al., 2011). While dendritic cells capture antigens released by tumor cells in situ, it is also possible to manipulate dendritic cells in vitro to direct the ensuing T cell response against a specific antigen. Heat-shock protein 70 was reported to be overexpressed in hepatitis C virus-related HCC. Thus, a phase 1 clinical trial, in which dendritic cells transfected with heat-shock protein 70 messenger RNA were given to patients with hepatitis C virus-related HCC, showed promising clinical responses, including complete remission in two patients (Maeda, et al., 2015). Nevertheless, as of now, large clinical trials of dendritic cell-based therapies in HCC to corroborate these findings are missing.
NK cells
Although novel cancer immunotherapies targeting T cell have been in the limelight recently, preclinical data suggest that NK cells are also capable of controlling HCC growth and therefore constitute another anti-tumor immune cell with promising implications for HCC immunotherapy. In a study using 3 independent HCC patient cohorts, gene expression analysis and immunohistochemistry of tumors revealed a superior survival of patients with high intratumoral NK cell accumulation (Chew, et al., 2012). These results were confirmed in another independent study, which in addition showed that NK cell frequency and function decreased in advanced disease stages (Y. Wu, et al., 2013). In contrast to T cells, NK cells do not recognize tumors through tumor-associated antigen expression on the tumor cell surface. Rather, NK cells possess a fine-tuned equilibrium of activating and inhibiting receptors on their surface. Depending on ligand expression by target cells, signaling through combinations of these NK cell receptors either leads to activation or control of NK cell cytotoxicity (Long, Kim, Liu, Peterson, & Rajagopalan, 2013). Accordingly, in patients with HCC, reduced expression of the ligand for NKG2D, an activating NK cell receptor, is associated with tumor progression and early tumor recurrence (Kamimura, et al., 2012). These data illustrate the significance of NK cells in controlling liver cancer growth.
However, given the high mortality and poor survival of primary liver cancer patients, these tumors ultimately also evade NK cell cytotoxicity. Accordingly, many NK cell-inhibiting mechanisms in HCC patients have been uncovered. In addition to the cited mechanism of NKG2D ligand down-regulation on cancer cells, HCC cell can also shed these ligands, causing elevated NKG2D ligand levels in blood of HCC patients and subsequent down-regulation of NKG2D expression on NK cells (Jinushi, et al., 2005). Not only the cancer cell itself, but also tumor-infiltrating MDSC and monocytes can inhibit NK cell activation by blocking activating-receptors NKp30 and 2B4 on NK cells (Hoechst, et al., 2009; Y. Wu, et al., 2013). Pharmacologically, the NKG2D ligand down-regulation and the ligand shedding can be reversed in vitro by a proteasome inhibitor and sorafenib, respectively (Kamimura, et al., 2012; Kohga, et al., 2010). However, the biological in vivo significance of these findings has not yet been investigated.
Similarly to ‘Checkpoint-inhibitors’ for T cell activation, antibodies that block inhibitory receptors on NK cells have entered clinical trials. One such antibody, lirilumab, targets the killer cell immunoglobulin-like receptor, KIR, and is currently being evaluated in a phase I trial recruiting, among others, HCC patients (Segal NH, et al., 2014). Therefore, NK cells, like T cells, can recognize and infiltrate HCC, affecting tumor growth. However, pharmacological activation is necessary to tap their full potential to overcome the tumor-mediated resistance mechanisms in order to improve survival of HCC patients.
2.2 Tumor promoting cells in the hepatic tumor environment
Immune cells as well as non-immune cells play an important role in tumor-induced immune evasion in patients with liver cancer. As described above, T cells and NK cells have the potential to control growth of HCC or even eradicate established tumors. Despite their potential, survival of HCC patients is poor, indicating that vigorous immunosuppressive mechanisms are in place to protect the tumor from immune attack. Indeed, liver cancers induce a plethora of cellular and non-cellular mechanisms to evade the immune system. Here, we will highlight the tumor-elicited cellular mechanisms that aid growth and progression of liver cancers (Figure 3).
Fig. 3.
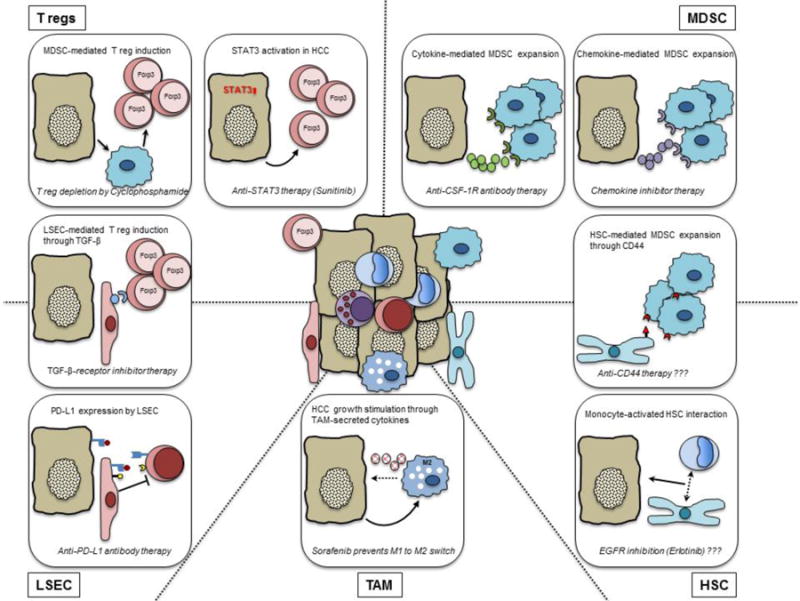
Pro-tumor effector mechanisms of immune and non-immune cells in HCC. Molecular mechanisms of regulatory T cells (upper left), myeloid-derived suppressor cells (upper right), liver sinusoidal endothelial cells (lower left), tumor-associated macrophages (bottom center) and hepatic stellate cells (lower right) that support tumor growth are shown. Therapeutic strategies to counter these mechanisms are provided at the bottom of each box. Question marks indicate proposed, not yet studied approaches.
Myeloid cells/MDSC
MDSC have been shown to accumulate in cancer patients of virtually all tumor types, in which they can suppress innate and adaptive anti-tumor immune responses (Poschke & Kiessling, 2012). Likewise in patients with HCC, MDSC suppress T cell proliferation, inhibit NK cell activity and induce tumor-promoting T reg (Hoechst, et al., 2008; Hoechst, et al., 2009). To suppress T cell proliferation, MDSC express the enzyme arginase to deprive T cells of the amino acid arginine, which is necessary for T cell proliferation. Consequently, low levels of arginine induce cell cycle arrest of T cells (Rodriguez, Quiceno, & Ochoa, 2007). As noted above, NK cell activity is inhibited by engagement or blockade of inhibitory or activating NK cell receptors, respectively (Hoechst, et al., 2009). The poor overall survival of HCC patients despite spontaneous anti-tumor T cell and NK cell responses against HCC suggests that potent immunosuppressive cells, like MDSC, are significant contributors to the tumor immune escape. Accordingly, MDSC accumulate in peripheral blood and within tumors of HCC patients (Hoechst, et al., 2008). Furthermore, with disease progression, the frequency of MDSC in peripheral blood increases and elevated numbers of MDSC correlate with shorter recurrence-free survival of HCC patients after local ablative therapies (Arihara, et al., 2013), confirming a negative association of MDSC accumulation and tumor control; illustrating their significance for tumor immune escape in patients with liver cancer.
MDSC, or myeloid cells in general, not only promote growth of primary liver cancer by inhibiting anti-tumor immune responses, but they also play an important role in “paving” the way for the development of metastases in the liver. It has been well established, that before metastatic seeding occurs in organs like lungs and livers, tumors “prepare” these organs by expanding metastasis-enabling myeloid cells (Kaplan, et al., 2005). In mice with pancreatic intraepithelial neoplasia, which commonly develop liver metastases upon progression to full-blown pancreatic cancer, accumulation of hepatic immunosuppressive myeloid cells, i.e. MDSC, precedes and promotes metastatic spread into the liver (Connolly, et al., 2010). In summary, tumor-induced expansion of myeloid cells/MDSC in the liver is crucial for escaping anti-tumor immune responses and facilitating metastatic seeding. Therefore, pharmacological inhibition of MDSC/myeloid cell accumulation could improve spontaneous or therapeutically induced anti-tumor immune responses, prevent hepatic metastasis formation and lead to improved patient survival.
The accumulation of immunosuppressive MDSC is mediated by tumor-secreted cytokines and/or chemokines (Gabrilovich & Nagaraj, 2009). G-CSF, GM-CSF, IL-6, CCL2 and VEGF are commonly found in tumor-bearing hosts and also cause MDSC expansion in HCC-bearing mice (Eggert, et al., 2014; Kapanadze, et al., 2013) and humans (Arihara, et al., 2013). These cytokines and chemokines induce proliferation of immature myeloid cells in the bone marrow and subsequent migration to the tumor. In patients with hepatic metastases from colorectal cancers, the expansion of metastasis-enabling myeloid cells in the liver is triggered by tumor-produced chemokines like CCL2 or CCL15 (Itatani, et al., 2013; L. Zhao, et al., 2013), suggesting that interfering with any of these pathways could reduce MDSC accumulation. Indeed, blocking MDSC-inducing cytokine or chemokine receptors has been shown to decrease their numbers in different animal tumor models and enhances efficacy of T cell immunotherapy (Highfill, et al., 2014; Mok, et al., 2014). For example, in a murine model of rhabdomyosarcoma, MDSC express the chemokine receptor CXCR2 and blocking this receptor by anti-CXCR2-monoclonal antibody therapy enhanced anti-PD-1 antibody treatment success (Highfill, et al., 2014). Another factor frequently involved in MDSC expansion is GM-CSF. Accordingly, neutralizing GM-CSF via antibody administration in a murine liver metastases model improved efficacy of chimeric-antigen receptor (CAR) T cell therapy (Burga, et al., 2015).
It is important to note however, that other mechanisms inducing MDSC accumulation and conferring immunosuppressive function, especially within the tumor, have been reported and could potentially limit the efficacy of the aforementioned treatment approaches to decrease MDSC numbers. For example, endoplasmatic reticulum stress (ER-stress) in combination with hypernutrition causes NASH and subsequent HCC development, indicating that ER-stress contributes to primary liver cancer initiation. On the other hand, ER-stress also triggers MDSC expansion and enhances immunosuppressive function (Condamine, et al., 2014; Thevenot, et al., 2014). Additional tumor-associated mechanisms altering MDSC function or triggering accumulation, independent of cytokine receptor signaling, include tumor-associated hypoxia and tumor-derived lactate (Corzo, et al., 2010; Husain, Huang, Seth, & Sukhatme, 2013). Hypoxia within tumors can skew tumor-infiltrating MDSC into more potently immunosuppressing tumor-associated macrophages (TAM). In contrast, tumor-derived lactate enhances MDSC expansion and NK cell dysfunction. In summary, numerous pathways exist how tumors regulate MDSC expansion and function, either directly by secreting cytokines or indirectly by factors that are characteristic for the tumor microenvironment, like hypoxia, high lactate levels or ER-stress. Additional approaches to alleviate MDSC-mediated immunosuppression could therefore further enhance the efficacy of cytokine neutralization or cytokine receptor blockade. Approaches include depletion of MDSC or restraining their immunosuppressive phenotype. Multiple molecules have been studied for their therapeutic effect on MDSC. These have been recently summarized in comprehensive reviews discussing MDSC biology in the liver and approaches to target MDSC in general (Marvel & Gabrilovich, 2015; Medina-Echeverz, Eggert, Han, & Greten, 2015). Approaches to deplete MDSC include low-dose gemcitabine or 5-fluorouracil treatment and approaches to inhibit their immunosuppressive function include molecules that interfere with ROS and NO production by MDSC, such as PDE-5 inhibitors or nitric oxide-releasing aspirin (De Santo, et al., 2005; Serafini, et al., 2006; Suzuki, Kapoor, Jassar, Kaiser, & Albelda, 2005; Vincent, et al., 2010).
Macrophages
In contrast to MDSC, macrophages naturally reside in healthy livers, where they are termed Kupffer cells. Upon liver injury, these liver resident macrophages proliferate and accumulate. Nevertheless, circulating monocytes also migrate to the liver and proliferate in response to inflammatory triggers; thereby contributing to the overall hepatic macrophage pool, i.e. resident macrophages plus monocyte-derived macrophages (Lavin, Mortha, Rahman, & Merad, 2015). It is still unclear, whether macrophage origin (resident vs. monocyte-derived) in the TME shapes their function and if this causes different macrophage responses against tumors. Therefore, we collectively summarize findings herein on macrophages in the hepatic TME independent of cell origin.
Because of their plasticity, i.e. polarization between a pro-inflammatory (M1) and anti-inflammatory (M2) phenotype, macrophages can have both pro- and anti-tumor effects (Mantovani, Sozzani, Locati, Allavena, & Sica, 2002). Precancerous hepatocytes overexpressing oncogenic Nras in an otherwise healthy liver are cleared by macrophages, preventing outgrowth of aggressive HCC (Kang, et al., 2011); exemplifying one mechanism of macrophage-mediated anti-tumor function. Macrophages can also recognize and phagocytose tumors cells upon down-regulation of CD47, a so-called “don’t eat me”-signal, on the tumor cell surface. However, many tumors up-regulate CD47 as an immune escape mechanism, including HCC (Xiao, et al., 2015). Accordingly, a study found that antibody-mediated blockade of CD47 enhances macrophage killing of HCC tumor cells. However, this study did not take into account the inflammatory environment of primary liver cancers, because tumor cells were injected into healthy livers. Macrophage polarization is strongly affected by the tumor-surrounding milieu and liver diseases that precede HCC development, like fibrosis, are associated with the tumor-promoting M2 macrophage phenotype (Sica, Invernizzi, & Mantovani, 2014). Hence, several lines of evidence suggest that macrophages in inflamed livers in mice and humans actually support growth of liver cancers, for example, by secreting the pro-inflammatory cytokine interleukin-6, which induces proliferation of hepatocytes (Lanaya, et al., 2014; Zhu, et al., 2008). If antibody-mediated targeting of CD47 also enhances cancer killing in inflamed livers, remains to be investigated.
Like MDSC or myeloid cells in general, TAM can inhibit both T cell and NK cell responses against HCC through expression of inhibitory receptors on their cell surface (Li, et al., 2012; Y. Wu, et al., 2013; Yan, et al., 2015). As discussed in the section on CTL, these receptors are possible targets for immunotherapeutic agents. Furthermore, macrophage numbers can increase in livers harboring HCC through mechanisms that induce proliferation and recruitment analogous to MDSC, leading to poorer survival in HCC patients (Zhu, et al., 2008). These mechanisms involve cytokines and chemokines like M-CSF and, as in MDSC, can be targeted pharmacologically. Accordingly, interfering with these pathways could dampen tumor-promoting effects of myeloid cells, i.e. TAM and MDSC.
In addition to their immunosuppressive properties, macrophages can promote tumor growth through a direct effect on the tumor cells themselves. It is well established that they induce epithelial-mesenchymal transition of tumor cells and thereby enhance aggressiveness and metastatic spreading of cancers, including HCC (Fan, et al., 2014; Xia, et al., 2014; Yeung, et al., 2015). Thus, macrophages can promote tumor growth through inducing proliferation and/or aggressiveness of cancer cells and through restraining anti-tumor immune responses.
Regulatory T cells (T reg)
In contrast to MDSC, which are believed to accumulate only in pathological settings, T reg possess important physiological functions in healthy humans, because dysfunctional T reg can be a cause of autoimmune diseases, allergy and other immunopathologies (Sakaguchi, Miyara, Costantino, & Hafler, 2010). However, tumors can take advantage of their strong immunosuppressive abilities to evade immune attack. Like with MDSC, T reg accumulate in HCC tumors and accumulation increases with disease progression. T reg also suppress anti-tumor CTL and an increase of the CTL to T reg ratio is an independent prognostic factor for improved survival (Gao, et al., 2007). Moreover, elevated levels in peripheral blood of HCC patients are associated with poorer overall survival (Fu, et al., 2007; X. H. Yang, et al., 2006). Analogous to MDSC, T reg are associated with suppression of T cell responses and aiding liver metastases development in colorectal cancers patients (Pedroza-Gonzalez, et al., 2013). Furthermore, T reg can curb the efficacy of tumor immunotherapy. In an orthotopic murine model of primary liver cancer, non-responders to interleukin-12 therapy had increased intratumoral T reg numbers and interleukin-10 levels (Zabala, et al., 2007). T reg inhibit T cells either in a contact dependent manner, through various membrane-bound molecules like TGF-β (Nakamura, Kitani, & Strober, 2001) or CTLA-4 (Peggs, Quezada, Chambers, Korman, & Allison, 2009; Wing, et al., 2008) or by secreting immunosuppressive cytokines like interleukin-10 and interleukin-35 (Collison, Pillai, Chaturvedi, & Vignali, 2009; Collison, et al., 2007). In summary, despite being of different cellular origin (lymphocytic vs. myeloid), T reg and MDSC share similarities in that they both dampen anti-tumor immune responses and promote growth of primary and metastatic liver cancer, although their effector mechanisms of immunosuppression may differ.
Two mechanisms responsible for T reg accumulation within tumors have been proposed (Bilate & Lafaille, 2012). Either, pre-existing Treg can be recruited by the tumor and proliferate locally (Curiel, et al., 2004; Ghiringhelli, et al., 2005), or tumors induce the conversion of conventional CD4+ T cells to T reg (Liu, et al., 2007; Valzasina, Piconese, Guiducci, & Colombo, 2006). In support of the latter concept, tumor-induced MDSC from HCC patients can also prompt a phenotypic switch from conventional CD4+ T cells to immunosuppressive T reg (Hoechst, et al., 2008). However, it is likely that both mechanisms exist in parallel and contribute to the overall expansion of tumor-associated T reg (Zhou & Levitsky, 2007). In any case, TGF-β has been identified to be a key cytokine for the induction of T reg in cancer bearing hosts (Ghiringhelli, et al., 2005; Liu, et al., 2007; Zhou & Levitsky, 2007). Thus, several mechanisms of intratumoral T reg induction exist, ultimately promoting tumor immune escape and facilitating cancer growth.
Efforts have been made to pharmacologically reduce T reg numbers or modulate their function. Because of its central role in not only inducing T reg but also mediating their immunosuppressive function (see above), targeting TGF-β is a promising approach to ease the detrimental effects of intratumoral T reg accumulation and several molecules and antibodies are in different stages of preclinical and clinical evaluation (Flavell, Sanjabi, Wrzesinski, & Licona-Limon, 2010). Another strategy to minimize T reg-mediated immunosuppression is anti-CTLA-4 antibody therapy. Anti-CTLA-4 antibodies do not only disinhibit CTL, but also restrain the suppressive abilities of CTLA-4 expressing T reg. Blockade of CTLA-4 on both T cell subsets contributes to the anti-tumor activity (Peggs, et al., 2009) and could potentially lead to enhanced anti-tumor T cell responses in patients with HCC (Duffy AG, et al., 2015). Other approaches to target T reg, of which only a few are mentioned here, include low-dose cyclophosphamide administration (Greten, et al., 2010) and toll-like receptor stimulation (Nishikawa, et al., 2006; Y. Yang, Huang, Huang, & Pardoll, 2004).
Liver sinusoidal endothelial cells (LSEC)
LSEC are specialized endothelial cells that line the hepatic sinusoids. Fenestration of LSEC allows for substrate transfer from the sinusoidal blood to hepatocytes and vice versa. LSEC have a high endocytosis rate and take up macromolecules from the sinusoidal blood by scavenger receptors (Smedsrod, et al., 2009). Moreover, LSEC have also important immunological functions. LSEC can efficiently cross-present antigens to CD8 T cells. Cross-presentation, the ability of antigen-presenting cells to present extrinsic peptides on their MHC-I molecule for interaction with CD8 T cells, is usually attributed to macrophages and dendritic cells only. Using this pathway, LSEC induce tolerance to CD8 T cells towards antigens form sinusoidal blood (Limmer, et al., 2000). However, virally infected LSEC can sufficiently induce T cell immunity, even in the absence of dendritic cells (Kern, et al., 2010).
In tumor-bearing mice, LSEC can induce CD8 T cell tolerance towards circulating antigen from apoptotic tumors cells (Berg, et al., 2006). Tolerance towards common antigens associated with colorectal cancer, like carcinoembryonic antigen, does also occur in human patients, representing a liver-specific tumor immune escape mechanism (Hochst, et al., 2012). The endocytotic uptake of tumor-associated antigens by LSEC is, at least partially, mediated by mannose receptors on LSEC. Pharmacological blockade of the mannose receptor abrogates tumor-induced and LSEC-mediated immunosuppression (Arteta, et al., 2010) and could serve as a potential target in therapy of hepatic metastases. Furthermore, LSEC can contribute to an immunosuppressive environment in the liver by PD-L1 expression or by inducing T reg through membrane-bound TGF-β (Carambia, et al., 2014). In summary, the ability of LSEC to confer T cell tolerance towards cancer-associated antigens and to create an immunosuppressive environment has been established. However, the significance of these pathways for HCC development and progression in patients and ultimately for patient survival has yet to be confirmed.
Hepatic stellate cells (HSC)
HSC reside in the space of Disse, between LSEC and hepatocytes, and are best known for their central role in liver fibrosis development, which ultimately can lead to HCC (Pellicoro, Ramachandran, Iredale, & Fallowfield, 2014). However, they are also capable of shaping hepatic immune responses (Thomson & Knolle, 2010). In patients with HCC, activated HSC have been reported to induce an immunosuppressive environment and are associated poor clinical outcome (Ji, et al., 2015; W. Zhao, et al., 2014). Upon activation, HSC skew monocytes from an inflammatory to an immunosuppressive phenotype and induce features of aggressive growth in HCC cells. This HSC-monocyte interaction results in early tumor recurrence and poor survival of liver cancer patients (Ji, et al., 2015). Moreover, they can induce the contact-dependent accumulation of MDSC, either through CD44 on HSC or through hydrogen peroxide depletion by catalase (Hochst, et al., 2013; Resheq, et al., 2015). HSC can also directly trigger T cell dysfunction through pathways analogous to other immunosuppressive cells. They are capable of inducing T cell apoptosis through PD-L1 expression (also known as B7-H1) (Yu, et al., 2004) and inhibit T cell proliferation and function through TGF-β (Y. Li, et al., 2015). Therapeutically, these immunosuppressive pathways are targetable using the approaches described above.
Under physiological conditions, HSC are quiescent. Upon inflammation-induced activation, HSC differentiate to myofibroblast-like cells and exert their fibrogenic and immunological functions (Pellicoro, et al., 2014; Thomson & Knolle, 2010). Inhibition of the epidermal growth factor receptor on HSC by the tyrosine kinase inhibitor Erlotinib has been shown to reduce activation of HSC in different mouse models of liver fibrosis (Fuchs, et al., 2014). Furthermore, Erlotinib prevent progression of fibrosis and ultimately protected from HCC development. Unfortunately, however, the effect of Erlotinib-mediated inhibition of HSC activation on immunological functions of HSC and its effect on HCC growth remains to be investigated. In summary, activated HSC promote HCC initiation and growth through immunological and non-immunological mechanisms.
3. Effect of the tumor microenvironment on primary liver cancer growth
In the vast majority of cases, HCC develop in livers with an underlying liver disease such as viral hepatitis or NASH. These underlying liver diseases not only fuel HCC initiation through the production of inflammatory, proliferation-inducing cytokines (He & Karin, 2011; Luedde & Schwabe, 2011; Sun & Karin, 2012), but they are also associated with strong negative regulation of spontaneous anti-tumor immune responses. Hypoxia, for example, is known to be an early phenomenon in liver cirrhosis development (Corpechot, et al., 2002) and skews myeloid cells in the TME towards a potent immunosuppressive phenotype (Corzo, et al., 2010). In HCC, hypoxia induces secretion of CCL20 by tumor cells, which in turn causes inhibition of T cell function through IDO up-regulation in macrophages (Ye, et al., 2016). Lipid accumulation in patients with NASH, preceding HCC development, is another metabolic factor, which negatively influences anti-tumor immune responses. A recent study showed that accumulation of a specific fatty acid, linoleic acid, in hepatocytes causes a selective loss of CD4 T cells through mitochondrial ROS-mediated apoptosis (Ma, et al., 2016). Decreased numbers of CD4 T cells in NASH livers accelerated growth of genetically and carcinogen-induced liver cancers. Thus, metabolic factors like hypoxia or lipid accumulation that occur before HCC initiation and persist during HCC growth restrain anti-tumor immune responses (Figure 1).
In addition to the aforementioned metabolic factors, an oncogene-induced intracellular anti-proliferative response program, i.e. cellular senescence, that has primarily been linked to cancer suppression, can surprisingly also promote growth of neighboring tumor cells in the liver (Eggert, et al., 2016). Senescent hepatocytes accumulate in inflamed livers through activation of oncogenic pathways or DNA damage (Jurk, et al., 2014; Mudbhary, et al., 2014). In livers without proliferating tumor cells, these senescent hepatocytes are cleared by macrophages, which have developed from infiltrating monocyte precursors, and thus prevent HCC initiation (Kang, et al., 2011). However, if full-blown tumor cells arise in livers with senescent hepatocytes, e.g. through bypassing the senescence program or additional senescence-inhibiting oncogenic mutations (Mudbhary, et al., 2014), the nascent tumors secrete factors to impede maturation of senescence-induced monocyte precursors, causing accumulation of immunosuppressive immature myeloid cells that inhibit anti-tumor NK cell responses (Eggert, et al., 2016). The accumulation of these senescence-induced immature myeloid cells is mediated by CCR2. Other studies have recently also implicated HCC-secreted CCL2 in murine and human tumor progression and blocking the CCL2-CCR2 axis led to improved mouse survival by disinhibiting anti-tumor T cells (X. Li, et al., 2015). Thus antagonists of CCR2 constitute an interesting target to inhibit tumor- and TME-induced immunosuppressive myeloid cell accumulation. In summary, in patients with HCC, the tumor by itself and in combination with the underlying liver disease shapes anti-tumor immune responses towards a favorable milieu for tumor growth.
In many cancer settings, studies are now underway investigating the effect of combining immunotherapy with targeted-therapies, e.g. kinase inhibitors, to improve patient survival. Because a plethora of clinical trials using targeted-therapies alone have failed to show an improvement of survival in HCC patients (Llovet, Villanueva, Lachenmayer, & Finn, 2015) and because immune responses are shaped by the tumor and the underlying liver inflammation, it is important to explore the efficacy of combination treatments in preclinical models that closely resemble the situation in humans, i.e. HCC growth in inflamed or cirrhotic livers. New murine HCC models take these two components into account (Reiberger, et al., 2015). After cirrhosis induction through carbon tetrachloride administration, tumor cells are either implanted orthotopically into the liver or initiated in genetically engineered models using viral expression of Cre recombinase. Employing this model, the authors showed that Sorafenib treatment alone led to hypoxia-mediated immunosuppression in liver tumors. However, combining Sorafenib with inhibition of the stromal cell-derived 1 alpha receptor (CXCR4) and ant-PD-1 antibody therapy enhanced anti-tumor immunity and reduced tumor growth (Y. Chen, et al., 2015), demonstrating that unleashing powerful anti-tumor immune responses against HCC is possible in cirrhotic livers. However, in addition to pharmacological T cell activation, it is necessary to block the potent immunosuppressive mechanisms associated with HCC and the underlying liver disease. With the wealth of new targeted-therapy and immunotherapy options, the number of combinations seems unlimited. Preclinical studies, such as the aforementioned, will aid in selecting mechanistically synergistic and promising combinations for further investigation in humans.
In summary, several lines of evidence have validated the importance of the immune system in HCC development and progression, but also its ability to control HCC growth. This suggests that employing the immune system in HCC by enhancing anti-tumor immune cells or interfering with pro-tumor immunosuppression constitutes a promising strategy to improve the survival of patients with HCC. Nevertheless, potent liver tumor-associated immunosuppression within the TME needs to be overcome for successful therapy.
4. Regulation of metastatic seeding into the liver by immune cells
Development of liver metastases from non-hepatic tumors differs from the development of HCC in that they usually form in an otherwise healthy liver without underlying liver inflammation or cirrhosis. Furthermore, metastatic tumors originate from various tissues, like lung, pancreas, colon, stomach or breast and are therefore heterogeneous in their phenotype compared to primary liver cancers (Goodman, 2007). Recently, it has also become clear that the primary tumor expands different myeloid cell populations in the liver through secretion of various factors before metastatic seeding; thereby “priming” the target organ for successful settlement of circulating tumor cells and causing metastatic organotropism (Hoshino, et al., 2015; Kaplan, Psaila, & Lyden, 2006).
Like with primary liver cancers, the immune system has the ability to not only support metastatic tumor growth, but also to suppress metastatic tumor development. The ability of the immune system to recognize and infiltrate primary liver cancers is established, but studies comprehensively investigating the immunogenicity of liver metastases from different primary tumors are missing. However, it has been shown that metastasis-infiltrating lymphocytes from patients with gastrointestinal cancers can recognize cancer cell lines generated from the same tumor, indicating that metastases are possibly also amenable to lymphocyte recognition and killing (Turcotte, et al., 2014). Further evidence for metastasis surveillance in the liver by lymphocytes is provided by studies in mice. In the liver, NK cells suppress metastasis through tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) expression (Takeda, et al., 2001). These NK cell responses towards colon cancer metastases can be triggered through inflammasome-induced IL-18 expression in Kuppfer cells (Dupaul-Chicoine, et al., 2015). These findings establish surveillance of non-hepatic cancer cells by lymphocytes in the liver as a means to prevent metastasis formation. Nevertheless, liver metastasis in advanced stages of gastrointestinal and non-gastrointestinal malignancies is common, illustrating that mechanisms, facilitating the development of these tumors distant from the primary site, must be in place.
In recent years, it has been shown that metastasis formation is merely a process of settlement of cancers cells, detached from the primary tumor, in a distant organ. Rather, the primary tumor “primes” the target organs through tumor produced and secreted factors that induce myeloid cell expansion and migration to the target organ (Hoshino, et al., 2015; Kaplan, et al., 2006). Haematopoietic bone marrow progenitor cells accumulate in organs like lungs and livers distant from the primary tumor before tumor cell arrival to “prepare” these tissues for metastatic seeding by expression of matrix metalloproteinase 9 (MMP 9) (Kaplan, et al., 2005). This process is called “pre-metastatic niche” formation. Pancreatic cancer secreted exosomes induce pre-metastatic niche formation by macrophage migration inhibitory factor (MIF) mediated macrophage recruitment (Costa-Silva, et al., 2015). Accordingly, patients with early stage pancreatic cancer with high MIF expression in exosomes were more likely to develop liver metastases.
In addition to initiating metastatic niche formation, myeloid cells in the liver of pre-invasive abdominal tumor-bearing mice can also enable metastatic tumor growth by inhibiting CTL (Connolly, et al., 2010). Analogous to primary liver tumors, these myeloid cells are recruited to the liver through chemokine expression in cancer cells. For example, colorectal cancer cells recruit CCR1+ myeloid cells through CCL15 or CCR2+ myeloid cells through CCL2 to facilitate liver metastases development (Itatani, et al., 2013; L. Zhao, et al., 2013). Metastasis promoting myeloid cells can also be recruited through tumor-necrosis factor receptor-2 on myeloid cells (Ham, et al., 2015). Therapeutically, interfering with early myeloid cell accumulation by blocking chemokines or cytokines before metastatic seeding occurs poses therefore an interesting strategy to prevent or reduce metastatic burden in patients at risk. Once liver metastases have formed, approaches to break tumor-induced immune tolerance similar to those employed in primary hepatic tumors, like radiofrequency ablation in combination with PD-1 blockade, can be used (Shi, et al., 2016). In summary, myeloid cells play a significant role not only in primary liver cancer development, but also in aiding successful metastatic seeding into the liver. Furthermore, as in HCC, they can confer tumor tolerance through inhibition of anti-tumor lymphocytes and can be targeted therapeutically.
5. Conclusions
The TME in the liver plays an important role in primary and metastatic liver tumor formation and tumor growth promotion. Like in malignancies of other tissues, immune cells can enhance malignant transformation, facilitate metastatic seeding or induce or restrain anti-tumor immune responses. In addition, non-immune cells unique to the liver, namely LSEC and HSC, can alter tumor-associated immune responses. In both, primary or metastatic tumors, cytotoxic lymphocytes are capable of killing cancer cells and spontaneous anti-tumor immune responses are readily detectable. However, several clinical trials aiming at enhancing anti-tumor immune responses via different means, e.g. vaccines, cytokines or adoptive cell therapy, in liver cancer patients have been conducted, but clinical responses overall were disappointing after single agent immunotherapeutic approaches (Makarova-Rusher, Medina-Echeverz, Duffy, & Greten, 2015), most likely, because tumors evaded immune responses through many potent tumor- and TME-induced immunosuppressive mechanisms. Thus, new approaches like ‘checkpoint-blockade’, especially in combination with targeted molecular therapies or local ablative therapies, seem promising in ‘unleashing’ the host anti-tumor immune response. It is noteworthy that recent research has shown that targeted molecular therapies, originally designed to hinder tumor cell proliferation, can also positively influence anti-tumor immune cell responses by alleviating myeloid cell-mediated resistance to ‘checkpoint-blockade’ (De Henau, et al., 2016). Treatment with a selective inhibitor of the gamma isoform of phosphoinositide 3-kinase can reshape the tumor immune microenvironment by blocking this enzyme in myeloid cells, ultimately promoting anti-tumor T cell responses. The ideal combination of immunotherapy and targeted molecular therapy would therefore, based on the aforementioned observations, achieve the following: i) enhance anti-tumor immune responses, ii) alleviate tumor-associated immunosuppression and iii) hinder tumor cell proliferation. In summary, harnessing immune responses by enhancing anti-tumor immune cells or interfering with pro-tumor immunosuppression has the potential to significantly improve patient survival.
Abbreviations
- CTL
cytotoxic T cells
- HCC
hepatocellular carcinomas
- HSC
hepatic stellate cells
- LSEC
liver sinusoidal endothelial cells
- MDSC
myeloid-derived suppressor cells
- NASH
non-alcoholic steatohepatitis
- NK cells
Natural Killer cells
- TAM
tumor-associated macrophages
- TME
tumor microenvironment
- T reg
regulatory T cells
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of Interest
The authors declare that there are no conflicts of interest.
References
- Ali MY, Grimm CF, Ritter M, Mohr L, Allgaier HP, Weth R, Bocher WO, Endrulat K, Blum HE, Geissler M. Activation of dendritic cells by local ablation of hepatocellular carcinoma. J Hepatol. 2005;43:817–822. doi: 10.1016/j.jhep.2005.04.016. [DOI] [PubMed] [Google Scholar]
- Arihara F, Mizukoshi E, Kitahara M, Takata Y, Arai K, Yamashita T, Nakamoto Y, Kaneko S. Increase in CD14+HLA−DR −/low myeloid-derived suppressor cells in hepatocellular carcinoma patients and its impact on prognosis. Cancer Immunol Immunother. 2013;62:1421–1430. doi: 10.1007/s00262-013-1447-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arteta B, Lasuen N, Lopategi A, Sveinbjornsson B, Smedsrod B, Vidal-Vanaclocha F. Colon carcinoma cell interaction with liver sinusoidal endothelium inhibits organ-specific antitumor immunity through interleukin-1-induced mannose receptor in mice. Hepatology. 2010;51:2172–2182. doi: 10.1002/hep.23590. [DOI] [PubMed] [Google Scholar]
- Berg M, Wingender G, Djandji D, Hegenbarth S, Momburg F, Hammerling G, Limmer A, Knolle P. Cross-presentation of antigens from apoptotic tumor cells by liver sinusoidal endothelial cells leads to tumor-specific CD8+ T cell tolerance. Eur J Immunol. 2006;36:2960–2970. doi: 10.1002/eji.200636033. [DOI] [PubMed] [Google Scholar]
- Bilate AM, Lafaille JJ. Induced CD4+Foxp3+ regulatory T cells in immune tolerance. Annu Rev Immunol. 2012;30:733–758. doi: 10.1146/annurev-immunol-020711-075043. [DOI] [PubMed] [Google Scholar]
- Burga RA, Thorn M, Point GR, Guha P, Nguyen CT, Licata LA, DeMatteo RP, Ayala A, Joseph Espat N, Junghans RP, Katz SC. Liver myeloid-derived suppressor cells expand in response to liver metastases in mice and inhibit the anti-tumor efficacy of anti-CEA CAR-T. Cancer Immunol Immunother. 2015;64:817–829. doi: 10.1007/s00262-015-1692-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carambia A, Freund B, Schwinge D, Heine M, Laschtowitz A, Huber S, Wraith DC, Korn T, Schramm C, Lohse AW, Heeren J, Herkel J. TGF-beta-dependent induction of CD4(+)CD25(+)Foxp3(+) Tregs by liver sinusoidal endothelial cells. J Hepatol. 2014;61:594–599. doi: 10.1016/j.jhep.2014.04.027. [DOI] [PubMed] [Google Scholar]
- Chao MP, Weissman IL, Majeti R. The CD47-SIRPalpha pathway in cancer immune evasion and potential therapeutic implications. Curr Opin Immunol. 2012;24:225–232. doi: 10.1016/j.coi.2012.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat Rev Immunol. 2013;13:227–242. doi: 10.1038/nri3405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Ramjiawan RR, Reiberger T, Ng MR, Hato T, Huang Y, Ochiai H, Kitahara S, Unan EC, Reddy TP, Fan C, Huang P, Bardeesy N, Zhu AX, Jain RK, Duda DG. CXCR4 inhibition in tumor microenvironment facilitates anti-programmed death receptor-1 immunotherapy in sorafenib-treated hepatocellular carcinoma in mice. Hepatology. 2015;61:1591–1602. doi: 10.1002/hep.27665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chew V, Chen J, Lee D, Loh E, Lee J, Lim KH, Weber A, Slankamenac K, Poon RT, Yang H, Ooi LL, Toh HC, Heikenwalder M, Ng IO, Nardin A, Abastado JP. Chemokine-driven lymphocyte infiltration: an early intratumoural event determining long-term survival in resectable hepatocellular carcinoma. Gut. 2012;61:427–438. doi: 10.1136/gutjnl-2011-300509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collison LW, Pillai MR, Chaturvedi V, Vignali DA. Regulatory T cell suppression is potentiated by target T cells in a cell contact, IL-35- and IL-10-dependent manner. J Immunol. 2009;182:6121–6128. doi: 10.4049/jimmunol.0803646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collison LW, Workman CJ, Kuo TT, Boyd K, Wang Y, Vignali KM, Cross R, Sehy D, Blumberg RS, Vignali DA. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature. 2007;450:566–569. doi: 10.1038/nature06306. [DOI] [PubMed] [Google Scholar]
- Condamine T, Kumar V, Ramachandran IR, Youn JI, Celis E, Finnberg N, El-Deiry WS, Winograd R, Vonderheide RH, English NR, Knight SC, Yagita H, McCaffrey JC, Antonia S, Hockstein N, Witt R, Masters G, Bauer T, Gabrilovich DI. ER stress regulates myeloid-derived suppressor cell fate through TRAIL-R-mediated apoptosis. J Clin Invest. 2014;124:2626–2639. doi: 10.1172/JCI74056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connolly MK, Mallen-St Clair J, Bedrosian AS, Malhotra A, Vera V, Ibrahim J, Henning J, Pachter HL, Bar-Sagi D, Frey AB, Miller G. Distinct populations of metastases-enabling myeloid cells expand in the liver of mice harboring invasive and preinvasive intra-abdominal tumor. J Leukoc Biol. 2010;87:713–725. doi: 10.1189/jlb.0909607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corpechot C, Barbu V, Wendum D, Kinnman N, Rey C, Poupon R, Housset C, Rosmorduc O. Hypoxia-induced VEGF and collagen I expressions are associated with angiogenesis and fibrogenesis in experimental cirrhosis. Hepatology. 2002;35:1010–1021. doi: 10.1053/jhep.2002.32524. [DOI] [PubMed] [Google Scholar]
- Corzo CA, Condamine T, Lu L, Cotter MJ, Youn JI, Cheng P, Cho HI, Celis E, Quiceno DG, Padhya T, McCaffrey TV, McCaffrey JC, Gabrilovich DI. HIF-1alpha regulates function and differentiation of myeloid-derived suppressor cells in the tumor microenvironment. J Exp Med. 2010;207:2439–2453. doi: 10.1084/jem.20100587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H, Xiang J, Zhang T, Theilen TM, Garcia-Santos G, Williams C, Ararso Y, Huang Y, Rodrigues G, Shen TL, Labori KJ, Lothe IM, Kure EH, Hernandez J, Doussot A, Ebbesen SH, Grandgenett PM, Hollingsworth MA, Jain M, Mallya K, Batra SK, Jarnagin WR, Schwartz RE, Matei I, Peinado H, Stanger BZ, Bromberg J, Lyden D. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat Cell Biol. 2015;17:816–826. doi: 10.1038/ncb3169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P, Evdemon-Hogan M, Conejo-Garcia JR, Zhang L, Burow M, Zhu Y, Wei S, Kryczek I, Daniel B, Gordon A, Myers L, Lackner A, Disis ML, Knutson KL, Chen L, Zou W. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004;10:942–949. doi: 10.1038/nm1093. [DOI] [PubMed] [Google Scholar]
- De Henau O, Rausch M, Winkler D, Campesato LF, Liu C, Cymerman DH, Budhu S, Ghosh A, Pink M, Tchaicha J, Douglas M, Tibbitts T, Sharma S, Proctor J, Kosmider N, White K, Stern H, Soglia J, Adams J, Palombella VJ, McGovern K, Kutok JL, Wolchok JD, Merghoub T. Overcoming resistance to checkpoint blockade therapy by targeting PI3Kgamma in myeloid cells. Nature. 2016;539:443–447. doi: 10.1038/nature20554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Santo C, Serafini P, Marigo I, Dolcetti L, Bolla M, Del Soldato P, Melani C, Guiducci C, Colombo MP, Iezzi M, Musiani P, Zanovello P, Bronte V. Nitroaspirin corrects immune dysfunction in tumor-bearing hosts and promotes tumor eradication by cancer vaccination. Proc Natl Acad Sci U S A. 2005;102:4185–4190. doi: 10.1073/pnas.0409783102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duffy AG, Makarova-Rusher OV, Kerkar SP, Kleiner DE, Fioravanti S, Walker M, Carey S, Figg WD, Steinberg SM, Anderson V, Abi-Jaoudeh N, Levi E, Wood BJ, G TF. A pilot study of tremelimumab – a monoclonal antibody against CTLA-4 – in combination with either trans catheter arterial chemoembolization (TACE) or radiofrequency ablation (RFA) in patients with hepatocellular carcinoma (HCC) J Clin Oncol. 2015;32 [Google Scholar]
- Dupaul-Chicoine J, Arabzadeh A, Dagenais M, Douglas T, Champagne C, Morizot A, Rodrigue-Gervais IG, Breton V, Colpitts SL, Beauchemin N, Saleh M. The Nlrp3 Inflammasome Suppresses Colorectal Cancer Metastatic Growth in the Liver by Promoting Natural Killer Cell Tumoricidal Activity. Immunity. 2015;43:751–763. doi: 10.1016/j.immuni.2015.08.013. [DOI] [PubMed] [Google Scholar]
- Eggert T, Medina-Echeverz J, Kapanadze T, Kruhlak MJ, Korangy F, Greten TF. Tumor induced hepatic myeloid derived suppressor cells can cause moderate liver damage. PLoS One. 2014;9:e112717. doi: 10.1371/journal.pone.0112717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eggert T, Wolter K, Ji J, Ma C, Yevsa T, Klotz S, Medina-Echeverz J, Longerich T, Forgues M, Reisinger F, Heikenwalder M, Wang XW, Zender L, Greten TF. Distinct functions of senescence-associated immune responses in liver tumor surveillance and tumor progression. Cancer Cell. 2016 doi: 10.1016/j.ccell.2016.09.003. accepted for publication on September 12th, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Khoueiry AB, Melero I, Crocenzi TS, Welling TH, Yau TC, Yeo W, Chopra A, Grosso J, Lang L, Anderson J, Dela Cruz CM, B S Phase I/II safety and antitumor activity of nivolumab in patients with advanced hepatocellular carcinoma (HCC): CA209-040. J Clin Oncol. 2015;33 2015 (suppl; abstr LBA101) [Google Scholar]
- El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557–2576. doi: 10.1053/j.gastro.2007.04.061. [DOI] [PubMed] [Google Scholar]
- Fan QM, Jing YY, Yu GF, Kou XR, Ye F, Gao L, Li R, Zhao QD, Yang Y, Lu ZH, Wei LX. Tumor-associated macrophages promote cancer stem cell-like properties via transforming growth factor-beta1-induced epithelial-mesenchymal transition in hepatocellular carcinoma. Cancer Lett. 2014;352:160–168. doi: 10.1016/j.canlet.2014.05.008. [DOI] [PubMed] [Google Scholar]
- Flavell RA, Sanjabi S, Wrzesinski SH, Licona-Limon P. The polarization of immune cells in the tumour environment by TGFbeta. Nat Rev Immunol. 2010;10:554–567. doi: 10.1038/nri2808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flecken T, Schmidt N, Hild S, Gostick E, Drognitz O, Zeiser R, Schemmer P, Bruns H, Eiermann T, Price DA, Blum HE, Neumann-Haefelin C, Thimme R. Immunodominance and functional alterations of tumor-associated antigen-specific CD8+ T-cell responses in hepatocellular carcinoma. Hepatology. 2014;59:1415–1426. doi: 10.1002/hep.26731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B, Zhang Z, Yang H, Zhang H, Zhou C, Yao J, Jin L, Wang H, Yang Y, Fu YX, Wang FS. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients. Gastroenterology. 2007;132:2328–2339. doi: 10.1053/j.gastro.2007.03.102. [DOI] [PubMed] [Google Scholar]
- Fuchs BC, Hoshida Y, Fujii T, Wei L, Yamada S, Lauwers GY, McGinn CM, DePeralta DK, Chen X, Kuroda T, Lanuti M, Schmitt AD, Gupta S, Crenshaw A, Onofrio R, Taylor B, Winckler W, Bardeesy N, Caravan P, Golub TR, Tanabe KK. Epidermal growth factor receptor inhibition attenuates liver fibrosis and development of hepatocellular carcinoma. Hepatology. 2014;59:1577–1590. doi: 10.1002/hep.26898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009;9:162–174. doi: 10.1038/nri2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Q, Qiu SJ, Fan J, Zhou J, Wang XY, Xiao YS, Xu Y, Li YW, Tang ZY. Intratumoral balance of regulatory and cytotoxic T cells is associated with prognosis of hepatocellular carcinoma after resection. J Clin Oncol. 2007;25:2586–2593. doi: 10.1200/JCO.2006.09.4565. [DOI] [PubMed] [Google Scholar]
- Ghiringhelli F, Puig PE, Roux S, Parcellier A, Schmitt E, Solary E, Kroemer G, Martin F, Chauffert B, Zitvogel L. Tumor cells convert immature myeloid dendritic cells into TGF-beta-secreting cells inducing CD4+CD25+ regulatory T cell proliferation. J Exp Med. 2005;202:919–929. doi: 10.1084/jem.20050463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman ZD. Neoplasms of the liver. Mod Pathol. 2007;20(Suppl 1):S49–60. doi: 10.1038/modpathol.3800682. [DOI] [PubMed] [Google Scholar]
- Greten TF, Ormandy LA, Fikuart A, Hochst B, Henschen S, Horning M, Manns MP, Korangy F. Low-dose cyclophosphamide treatment impairs regulatory T cells and unmasks AFP-specific CD4+ T-cell responses in patients with advanced HCC. J Immunother. 2010;33:211–218. doi: 10.1097/CJI.0b013e3181bb499f. [DOI] [PubMed] [Google Scholar]
- Ham B, Wang N, D’Costa Z, Fernandez MC, Bourdeau F, Auguste P, Illemann M, Eefsen RL, Hoyer-Hansen G, Vainer B, Evrard M, Gao ZH, Brodt P. TNF Receptor-2 Facilitates an Immunosuppressive Microenvironment in the Liver to Promote the Colonization and Growth of Hepatic Metastases. Cancer Res. 2015;75:5235–5247. doi: 10.1158/0008-5472.CAN-14-3173. [DOI] [PubMed] [Google Scholar]
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- He G, Karin M. NF-kappaB and STAT3 - key players in liver inflammation and cancer. Cell Res. 2011;21:159–168. doi: 10.1038/cr.2010.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Highfill SL, Cui Y, Giles AJ, Smith JP, Zhang H, Morse E, Kaplan RN, Mackall CL. Disruption of CXCR2-mediated MDSC tumor trafficking enhances anti-PD1 efficacy. Sci Transl Med. 2014;6:237ra267. doi: 10.1126/scitranslmed.3007974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiroishi K, Eguchi J, Baba T, Shimazaki T, Ishii S, Hiraide A, Sakaki M, Doi H, Uozumi S, Omori R, Matsumura T, Yanagawa T, Ito T, Imawari M. Strong CD8(+) T-cell responses against tumor-associated antigens prolong the recurrence-free interval after tumor treatment in patients with hepatocellular carcinoma. J Gastroenterol. 2010;45:451–458. doi: 10.1007/s00535-009-0155-2. [DOI] [PubMed] [Google Scholar]
- Hochst B, Schildberg FA, Bottcher J, Metzger C, Huss S, Turler A, Overhaus M, Knoblich A, Schneider B, Pantelis D, Kurts C, Kalff JC, Knolle P, Diehl L. Liver sinusoidal endothelial cells contribute to CD8 T cell tolerance toward circulating carcinoembryonic antigen in mice. Hepatology. 2012;56:1924–1933. doi: 10.1002/hep.25844. [DOI] [PubMed] [Google Scholar]
- Hochst B, Schildberg FA, Sauerborn P, Gabel YA, Gevensleben H, Goltz D, Heukamp LC, Turler A, Ballmaier M, Gieseke F, Muller I, Kalff J, Kurts C, Knolle PA, Diehl L. Activated human hepatic stellate cells induce myeloid derived suppressor cells from peripheral blood monocytes in a CD44-dependent fashion. J Hepatol. 2013;59:528–535. doi: 10.1016/j.jhep.2013.04.033. [DOI] [PubMed] [Google Scholar]
- Hoechst B, Ormandy LA, Ballmaier M, Lehner F, Kruger C, Manns MP, Greten TF, Korangy F. A new population of myeloid-derived suppressor cells in hepatocellular carcinoma patients induces CD4(+)CD25(+)Foxp3(+) T cells. Gastroenterology. 2008;135:234–243. doi: 10.1053/j.gastro.2008.03.020. [DOI] [PubMed] [Google Scholar]
- Hoechst B, Voigtlaender T, Ormandy L, Gamrekelashvili J, Zhao F, Wedemeyer H, Lehner F, Manns MP, Greten TF, Korangy F. Myeloid derived suppressor cells inhibit natural killer cells in patients with hepatocellular carcinoma via the NKp30 receptor. Hepatology. 2009;50:799–807. doi: 10.1002/hep.23054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, Singh S, Williams C, Soplop N, Uryu K, Pharmer L, King T, Bojmar L, Davies AE, Ararso Y, Zhang T, Zhang H, Hernandez J, Weiss JM, Dumont-Cole VD, Kramer K, Wexler LH, Narendran A, Schwartz GK, Healey JH, Sandstrom P, Labori KJ, Kure EH, Grandgenett PM, Hollingsworth MA, de Sousa M, Kaur S, Jain M, Mallya K, Batra SK, Jarnagin WR, Brady MS, Fodstad O, Muller V, Pantel K, Minn AJ, Bissell MJ, Garcia BA, Kang Y, Rajasekhar VK, Ghajar CM, Matei I, Peinado H, Bromberg J, Lyden D. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329–335. doi: 10.1038/nature15756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Husain Z, Huang Y, Seth P, Sukhatme VP. Tumor-derived lactate modifies antitumor immune response: effect on myeloid-derived suppressor cells and NK cells. J Immunol. 2013;191:1486–1495. doi: 10.4049/jimmunol.1202702. [DOI] [PubMed] [Google Scholar]
- Itatani Y, Kawada K, Fujishita T, Kakizaki F, Hirai H, Matsumoto T, Iwamoto M, Inamoto S, Hatano E, Hasegawa S, Maekawa T, Uemoto S, Sakai Y, Taketo MM. Loss of SMAD4 from colorectal cancer cells promotes CCL15 expression to recruit CCR1+ myeloid cells and facilitate liver metastasis. Gastroenterology. 2013;145:1064–1075 e1011. doi: 10.1053/j.gastro.2013.07.033. [DOI] [PubMed] [Google Scholar]
- Jenne CN, Kubes P. Immune surveillance by the liver. Nat Immunol. 2013;14:996–1006. doi: 10.1038/ni.2691. [DOI] [PubMed] [Google Scholar]
- Ji J, Eggert T, Budhu A, Forgues M, Takai A, Dang H, Ye Q, Lee JS, Kim JH, Greten TF, Wang XW. Hepatic stellate cell and monocyte interaction contributes to poor prognosis in hepatocellular carcinoma. Hepatology. 2015;62:481–495. doi: 10.1002/hep.27822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jinushi M, Takehara T, Tatsumi T, Hiramatsu N, Sakamori R, Yamaguchi S, Hayashi N. Impairment of natural killer cell and dendritic cell functions by the soluble form of MHC class I-related chain A in advanced human hepatocellular carcinomas. J Hepatol. 2005;43:1013–1020. doi: 10.1016/j.jhep.2005.05.026. [DOI] [PubMed] [Google Scholar]
- Joyce JA, Fearon DT. T cell exclusion, immune privilege, and the tumor microenvironment. Science. 2015;348:74–80. doi: 10.1126/science.aaa6204. [DOI] [PubMed] [Google Scholar]
- Jurk D, Wilson C, Passos JF, Oakley F, Correia-Melo C, Greaves L, Saretzki G, Fox C, Lawless C, Anderson R, Hewitt G, Pender SL, Fullard N, Nelson G, Mann J, van de Sluis B, Mann DA, von Zglinicki T. Chronic inflammation induces telomere dysfunction and accelerates ageing in mice. Nat Commun. 2014;2:4172. doi: 10.1038/ncomms5172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamimura H, Yamagiwa S, Tsuchiya A, Takamura M, Matsuda Y, Ohkoshi S, Inoue M, Wakai T, Shirai Y, Nomoto M, Aoyagi Y. Reduced NKG2D ligand expression in hepatocellular carcinoma correlates with early recurrence. J Hepatol. 2012;56:381–388. doi: 10.1016/j.jhep.2011.06.017. [DOI] [PubMed] [Google Scholar]
- Kang TW, Yevsa T, Woller N, Hoenicke L, Wuestefeld T, Dauch D, Hohmeyer A, Gereke M, Rudalska R, Potapova A, Iken M, Vucur M, Weiss S, Heikenwalder M, Khan S, Gil J, Bruder D, Manns M, Schirmacher P, Tacke F, Ott M, Luedde T, Longerich T, Kubicka S, Zender L. Senescence surveillance of pre-malignant hepatocytes limits liver cancer development. Nature. 2011;479:547–551. doi: 10.1038/nature10599. [DOI] [PubMed] [Google Scholar]
- Kapanadze T, Gamrekelashvili J, Ma C, Chan C, Zhao F, Hewitt S, Zender L, Kapoor V, Felsher DW, Manns MP, Korangy F, Greten TF. Regulation of accumulation and function of myeloid derived suppressor cells in different murine models of hepatocellular carcinoma. J Hepatol. 2013;59:1007–1013. doi: 10.1016/j.jhep.2013.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan RN, Psaila B, Lyden D. Bone marrow cells in the ‘pre-metastatic niche’: within bone and beyond. Cancer Metastasis Rev. 2006;25:521–529. doi: 10.1007/s10555-006-9036-9. [DOI] [PubMed] [Google Scholar]
- Kaplan RN, Riba RD, Zacharoulis S, Bramley AH, Vincent L, Costa C, MacDonald DD, Jin DK, Shido K, Kerns SA, Zhu Z, Hicklin D, Wu Y, Port JL, Altorki N, Port ER, Ruggero D, Shmelkov SV, Jensen KK, Rafii S, Lyden D. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature. 2005;438:820–827. doi: 10.1038/nature04186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kern M, Popov A, Scholz K, Schumak B, Djandji D, Limmer A, Eggle D, Sacher T, Zawatzky R, Holtappels R, Reddehase MJ, Hartmann G, Debey-Pascher S, Diehl L, Kalinke U, Koszinowski U, Schultze J, Knolle PA. Virally infected mouse liver endothelial cells trigger CD8+ T-cell immunity. Gastroenterology. 2010;138:336–346. doi: 10.1053/j.gastro.2009.08.057. [DOI] [PubMed] [Google Scholar]
- Kohga K, Takehara T, Tatsumi T, Ishida H, Miyagi T, Hosui A, Hayashi N. Sorafenib inhibits the shedding of major histocompatibility complex class I-related chain A on hepatocellular carcinoma cells by down-regulating a disintegrin and metalloproteinase 9. Hepatology. 2010;51:1264–1273. doi: 10.1002/hep.23456. [DOI] [PubMed] [Google Scholar]
- Kuang DM, Zhao Q, Peng C, Xu J, Zhang JP, Wu C, Zheng L. Activated monocytes in peritumoral stroma of hepatocellular carcinoma foster immune privilege and disease progression through PD-L1. J Exp Med. 2009;206:1327–1337. doi: 10.1084/jem.20082173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanaya H, Natarajan A, Komposch K, Li L, Amberg N, Chen L, Wculek SK, Hammer M, Zenz R, Peck-Radosavljevic M, Sieghart W, Trauner M, Wang H, Sibilia M. EGFR has a tumour-promoting role in liver macrophages during hepatocellular carcinoma formation. Nat Cell Biol. 2014;16:972–981. 971–977. doi: 10.1038/ncb3031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavin Y, Mortha A, Rahman A, Merad M. Regulation of macrophage development and function in peripheral tissues. Nat Rev Immunol. 2015;15:731–744. doi: 10.1038/nri3920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Wu K, Tao K, Chen L, Zheng Q, Lu X, Liu J, Shi L, Liu C, Wang G, Zou W. Tim-3/galectin-9 signaling pathway mediates T-cell dysfunction and predicts poor prognosis in patients with hepatitis B virus-associated hepatocellular carcinoma. Hepatology. 2012;56:1342–1351. doi: 10.1002/hep.25777. [DOI] [PubMed] [Google Scholar]
- Li X, Yao W, Yuan Y, Chen P, Li B, Li J, Chu R, Song H, Xie D, Jiang X, Wang H. Targeting of tumour-infiltrating macrophages via CCL2/CCR2 signalling as a therapeutic strategy against hepatocellular carcinoma. Gut. 2015 doi: 10.1136/gutjnl-2015-310514. [DOI] [PubMed] [Google Scholar]
- Li Y, Kim BG, Qian S, Letterio JJ, Fung JJ, Lu L, Lin F. Hepatic Stellate Cells Inhibit T Cells through Active TGF-beta1 from a Cell Surface-Bound Latent TGF-beta1/GARP Complex. J Immunol. 2015;195:2648–2656. doi: 10.4049/jimmunol.1500139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim SY, Gordon-Weeks A, Allen D, Kersemans V, Beech J, Smart S, Muschel RJ. Cd11b myeloid cells support hepatic metastasis through down-regulation of angiopoietin-like 7 in cancer cells. Hepatology. 2015 doi: 10.1002/hep.27838. [DOI] [PubMed] [Google Scholar]
- Limmer A, Ohl J, Kurts C, Ljunggren HG, Reiss Y, Groettrup M, Momburg F, Arnold B, Knolle PA. Efficient presentation of exogenous antigen by liver endothelial cells to CD8+ T cells results in antigen-specific T-cell tolerance. Nat Med. 2000;6:1348–1354. doi: 10.1038/82161. [DOI] [PubMed] [Google Scholar]
- Liu VC, Wong LY, Jang T, Shah AH, Park I, Yang X, Zhang Q, Lonning S, Teicher BA, Lee C. Tumor evasion of the immune system by converting CD4+CD25− T cells into CD4+CD25+ T regulatory cells: role of tumor-derived TGF-beta. J Immunol. 2007;178:2883–2892. doi: 10.4049/jimmunol.178.5.2883. [DOI] [PubMed] [Google Scholar]
- Llovet JM, Villanueva A, Lachenmayer A, Finn RS. Advances in targeted therapies for hepatocellular carcinoma in the genomic era. Nat Rev Clin Oncol. 2015;12:408–424. doi: 10.1038/nrclinonc.2015.103. [DOI] [PubMed] [Google Scholar]
- Long EO, Kim HS, Liu D, Peterson ME, Rajagopalan S. Controlling natural killer cell responses: integration of signals for activation and inhibition. Annu Rev Immunol. 2013;31:227–258. doi: 10.1146/annurev-immunol-020711-075005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luedde T, Schwabe RF. NF-kappaB in the liver–linking injury, fibrosis and hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2011;8:108–118. doi: 10.1038/nrgastro.2010.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma C, Kesarwala AH, Eggert T, Medina-Echeverz J, Kleiner DE, Jin P, Stroncek DF, Terabe M, Kapoor V, ElGindi M, Han M, Thornton AM, Zhang H, Egger M, Luo J, Felsher DW, McVicar DW, Weber A, Heikenwalder M, Greten TF. NAFLD causes selective CD4(+) T lymphocyte loss and promotes hepatocarcinogenesis. Nature. 2016;531:253–257. doi: 10.1038/nature16969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeda Y, Yoshimura K, Matsui H, Shindo Y, Tamesa T, Tokumitsu Y, Hashimoto N, Tokuhisa Y, Sakamoto K, Sakai K, Suehiro Y, Hinoda Y, Tamada K, Yoshino S, Hazama S, Oka M. Dendritic cells transfected with heat-shock protein 70 messenger RNA for patients with hepatitis C virus-related hepatocellular carcinoma: a phase 1 dose escalation clinical trial. Cancer Immunol Immunother. 2015;64:1047–1056. doi: 10.1007/s00262-015-1709-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makarova-Rusher OV, Medina-Echeverz J, Duffy AG, Greten TF. The yin and yang of evasion and immune activation in HCC. J Hepatol. 2015;62:1420–1429. doi: 10.1016/j.jhep.2015.02.038. [DOI] [PubMed] [Google Scholar]
- Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23:549–555. doi: 10.1016/s1471-4906(02)02302-5. [DOI] [PubMed] [Google Scholar]
- Marvel D, Gabrilovich DI. Myeloid-derived suppressor cells in the tumor microenvironment: expect the unexpected. J Clin Invest. 2015:1–9. doi: 10.1172/JCI80005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medina-Echeverz J, Eggert T, Han M, Greten TF. Hepatic myeloid-derived suppressor cells in cancer. Cancer Immunol Immunother. 2015;64:931–940. doi: 10.1007/s00262-015-1736-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizukoshi E, Nakamoto Y, Arai K, Yamashita T, Sakai A, Sakai Y, Kagaya T, Yamashita T, Honda M, Kaneko S. Comparative analysis of various tumor-associated antigen-specific t-cell responses in patients with hepatocellular carcinoma. Hepatology. 2011;53:1206–1216. doi: 10.1002/hep.24149. [DOI] [PubMed] [Google Scholar]
- Mizukoshi E, Yamashita T, Arai K, Sunagozaka H, Ueda T, Arihara F, Kagaya T, Yamashita T, Fushimi K, Kaneko S. Enhancement of tumor-associated antigen-specific T cell responses by radiofrequency ablation of hepatocellular carcinoma. Hepatology. 2013;57:1448–1457. doi: 10.1002/hep.26153. [DOI] [PubMed] [Google Scholar]
- Mok S, Koya RC, Tsui C, Xu J, Robert L, Wu L, Graeber TG, West BL, Bollag G, Ribas A. Inhibition of CSF-1 receptor improves the antitumor efficacy of adoptive cell transfer immunotherapy. Cancer Res. 2014;74:153–161. doi: 10.1158/0008-5472.CAN-13-1816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morales-Kastresana A, Sanmamed MF, Rodriguez I, Palazon A, Martinez-Forero I, Labiano S, Hervas-Stubbs S, Sangro B, Ochoa C, Rouzaut A, Azpilikueta A, Bolanos E, Jure-Kunkel M, Gutgemann I, Melero I. Combined immunostimulatory monoclonal antibodies extend survival in an aggressive transgenic hepatocellular carcinoma mouse model. Clin Cancer Res. 2013;19:6151–6162. doi: 10.1158/1078-0432.CCR-13-1189. [DOI] [PubMed] [Google Scholar]
- Mudbhary R, Hoshida Y, Chernyavskaya Y, Jacob V, Villanueva A, Fiel MI, Chen X, Kojima K, Thung S, Bronson RT, Lachenmayer A, Revill K, Alsinet C, Sachidanandam R, Desai A, SenBanerjee S, Ukomadu C, Llovet JM, Sadler KC. UHRF1 overexpression drives DNA hypomethylation and hepatocellular carcinoma. Cancer Cell. 2014;25:196–209. doi: 10.1016/j.ccr.2014.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamoto Y, Mizukoshi E, Kitahara M, Arihara F, Sakai Y, Kakinoki K, Fujita Y, Marukawa Y, Arai K, Yamashita T, Mukaida N, Matsushima K, Matsui O, Kaneko S. Prolonged recurrence-free survival following OK432-stimulated dendritic cell transfer into hepatocellular carcinoma during transarterial embolization. Clin Exp Immunol. 2011;163:165–177. doi: 10.1111/j.1365-2249.2010.04246.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura K, Kitani A, Strober W. Cell contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T cells is mediated by cell surface-bound transforming growth factor beta. J Exp Med. 2001;194:629–644. doi: 10.1084/jem.194.5.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishikawa H, Sato E, Briones G, Chen LM, Matsuo M, Nagata Y, Ritter G, Jager E, Nomura H, Kondo S, Tawara I, Kato T, Shiku H, Old LJ, Galan JE, Gnjatic S. In vivo antigen delivery by a Salmonella typhimurium type III secretion system for therapeutic cancer vaccines. J Clin Invest. 2006;116:1946–1954. doi: 10.1172/JCI28045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Njei B, Rotman Y, Ditah I, Lim JK. Emerging trends in hepatocellular carcinoma incidence and mortality. Hepatology. 2015;61:191–199. doi: 10.1002/hep.27388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedroza-Gonzalez A, Verhoef C, Ijzermans JN, Peppelenbosch MP, Kwekkeboom J, Verheij J, Janssen HL, Sprengers D. Activated tumor-infiltrating CD4+ regulatory T cells restrain antitumor immunity in patients with primary or metastatic liver cancer. Hepatology. 2013;57:183–194. doi: 10.1002/hep.26013. [DOI] [PubMed] [Google Scholar]
- Peggs KS, Quezada SA, Chambers CA, Korman AJ, Allison JP. Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti-CTLA-4 antibodies. J Exp Med. 2009;206:1717–1725. doi: 10.1084/jem.20082492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellicoro A, Ramachandran P, Iredale JP, Fallowfield JA. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat Rev Immunol. 2014;14:181–194. doi: 10.1038/nri3623. [DOI] [PubMed] [Google Scholar]
- Poschke I, Kiessling R. On the armament and appearances of human myeloid-derived suppressor cells. Clin Immunol. 2012;144:250–268. doi: 10.1016/j.clim.2012.06.003. [DOI] [PubMed] [Google Scholar]
- Reiberger T, Chen Y, Ramjiawan RR, Hato T, Fan C, Samuel R, Roberge S, Huang P, Lauwers GY, Zhu AX, Bardeesy N, Jain RK, Duda DG. An orthotopic mouse model of hepatocellular carcinoma with underlying liver cirrhosis. Nat Protoc. 2015;10:1264–1274. doi: 10.1038/nprot.2015.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Resheq YJ, Li KK, Ward ST, Wilhelm A, Garg A, Curbishley SM, Blahova M, Zimmermann HW, Jitschin R, Mougiakakos D, Mackensen A, Weston CJ, Adams DH. Contact-dependent depletion of hydrogen peroxide by catalase is a novel mechanism of myeloid-derived suppressor cell induction operating in human hepatic stellate cells. J Immunol. 2015;194:2578–2586. doi: 10.4049/jimmunol.1401046. [DOI] [PubMed] [Google Scholar]
- Robert C, Schachter J, Long GV, Arance A, Grob JJ, Mortier L, Daud A, Carlino MS, McNeil C, Lotem M, Larkin J, Lorigan P, Neyns B, Blank CU, Hamid O, Mateus C, Shapira-Frommer R, Kosh M, Zhou H, Ibrahim N, Ebbinghaus S, Ribas A, investigators, K Pembrolizumab versus Ipilimumab in Advanced Melanoma. N Engl J Med. 2015;372:2521–2532. doi: 10.1056/NEJMoa1503093. [DOI] [PubMed] [Google Scholar]
- Rodriguez PC, Quiceno DG, Ochoa AC. L-arginine availability regulates T-lymphocyte cell-cycle progression. Blood. 2007;109:1568–1573. doi: 10.1182/blood-2006-06-031856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakaguchi S, Miyara M, Costantino CM, Hafler DA. FOXP3+ regulatory T cells in the human immune system. Nat Rev Immunol. 2010;10:490–500. doi: 10.1038/nri2785. [DOI] [PubMed] [Google Scholar]
- Sangro B, Gomez-Martin C, de la Mata M, Inarrairaegui M, Garralda E, Barrera P, Riezu-Boj JI, Larrea E, Alfaro C, Sarobe P, Lasarte JJ, Perez-Gracia JL, Melero I, Prieto J. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J Hepatol. 2013;59:81–88. doi: 10.1016/j.jhep.2013.02.022. [DOI] [PubMed] [Google Scholar]
- Schadendorf D, Hodi FS, Robert C, Weber JS, Margolin K, Hamid O, Patt D, Chen TT, Berman DM, Wolchok JD. Pooled Analysis of Long-Term Survival Data From Phase II and Phase III Trials of Ipilimumab in Unresectable or Metastatic Melanoma. J Clin Oncol. 2015;33:1889–1894. doi: 10.1200/JCO.2014.56.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schumacher TN, Schreiber RD. Neoantigens in cancer immunotherapy. Science. 2015;348:69–74. doi: 10.1126/science.aaa4971. [DOI] [PubMed] [Google Scholar]
- Schurich A, Bottcher JP, Burgdorf S, Penzler P, Hegenbarth S, Kern M, Dolf A, Endl E, Schultze J, Wiertz E, Stabenow D, Kurts C, Knolle P. Distinct kinetics and dynamics of cross-presentation in liver sinusoidal endothelial cells compared to dendritic cells. Hepatology. 2009;50:909–919. doi: 10.1002/hep.23075. [DOI] [PubMed] [Google Scholar]
- Segal NH, Hodi FS, Sanborn RE, Gajewski T, Wolchok JD, Urba WJ, Fox BA, Topalian SL, Pardoll DM, Kollia G, McDonald D, Kim SY, S WH. A phase I dose escalation and cohort expansion study of lirilumab (anti-KIR; BMS-986015) in combination with nivolumab (anti-PD-1; BMS-936558, ONO-4538) in advanced solid tumors. J Clin Oncol. 2014;32 5s, 2014 (suppl; abstr TPS3115) [Google Scholar]
- Serafini P, Meckel K, Kelso M, Noonan K, Califano J, Koch W, Dolcetti L, Bronte V, Borrello I. Phosphodiesterase-5 inhibition augments endogenous antitumor immunity by reducing myeloid-derived suppressor cell function. J Exp Med. 2006;203:2691–2702. doi: 10.1084/jem.20061104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi L, Chen L, Wu C, Zhu Y, Xu B, Zheng X, Sun M, Wen W, Dai X, Yang M, Lv Q, Lu B, Jiang J. PD-1 Blockade Boosts Radiofrequency Ablation-Elicited Adaptive Immune Responses against Tumor. Clin Cancer Res. 2016;22:1173–1184. doi: 10.1158/1078-0432.CCR-15-1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sica A, Invernizzi P, Mantovani A. Macrophage plasticity and polarization in liver homeostasis and pathology. Hepatology. 2014;59:2034–2042. doi: 10.1002/hep.26754. [DOI] [PubMed] [Google Scholar]
- Smedsrod B, Le Couteur D, Ikejima K, Jaeschke H, Kawada N, Naito M, Knolle P, Nagy L, Senoo H, Vidal-Vanaclocha F, Yamaguchi N. Hepatic sinusoidal cells in health and disease: update from the 14th International Symposium. Liver Int. 2009;29:490–501. doi: 10.1111/j.1478-3231.2009.01979.x. [DOI] [PubMed] [Google Scholar]
- Sun B, Karin M. Obesity, inflammation, and liver cancer. J Hepatol. 2012;56:704–713. doi: 10.1016/j.jhep.2011.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki E, Kapoor V, Jassar AS, Kaiser LR, Albelda SM. Gemcitabine selectively eliminates splenic Gr-1+/CD11b+ myeloid suppressor cells in tumor-bearing animals and enhances antitumor immune activity. Clin Cancer Res. 2005;11:6713–6721. doi: 10.1158/1078-0432.CCR-05-0883. [DOI] [PubMed] [Google Scholar]
- Takeda K, Hayakawa Y, Smyth MJ, Kayagaki N, Yamaguchi N, Kakuta S, Iwakura Y, Yagita H, Okumura K. Involvement of tumor necrosis factor-related apoptosis-inducing ligand in surveillance of tumor metastasis by liver natural killer cells. Nat Med. 2001;7:94–100. doi: 10.1038/83416. [DOI] [PubMed] [Google Scholar]
- Thevenot PT, Sierra RA, Raber PL, Al-Khami AA, Trillo-Tinoco J, Zarreii P, Ochoa AC, Cui Y, Del Valle L, Rodriguez PC. The stress-response sensor chop regulates the function and accumulation of myeloid-derived suppressor cells in tumors. Immunity. 2014;41:389–401. doi: 10.1016/j.immuni.2014.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomson AW, Knolle PA. Antigen-presenting cell function in the tolerogenic liver environment. Nat Rev Immunol. 2010;10:753–766. doi: 10.1038/nri2858. [DOI] [PubMed] [Google Scholar]
- Turcotte S, Gros A, Tran E, Lee CC, Wunderlich JR, Robbins PF, Rosenberg SA. Tumor-reactive CD8+ T cells in metastatic gastrointestinal cancer refractory to chemotherapy. Clin Cancer Res. 2014;20:331–343. doi: 10.1158/1078-0432.CCR-13-1736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valzasina B, Piconese S, Guiducci C, Colombo MP. Tumor-induced expansion of regulatory T cells by conversion of CD4+CD25− lymphocytes is thymus and proliferation independent. Cancer Res. 2006;66:4488–4495. doi: 10.1158/0008-5472.CAN-05-4217. [DOI] [PubMed] [Google Scholar]
- Vincent J, Mignot G, Chalmin F, Ladoire S, Bruchard M, Chevriaux A, Martin F, Apetoh L, Rebe C, Ghiringhelli F. 5-Fluorouracil selectively kills tumor-associated myeloid-derived suppressor cells resulting in enhanced T cell-dependent antitumor immunity. Cancer Res. 2010;70:3052–3061. doi: 10.1158/0008-5472.CAN-09-3690. [DOI] [PubMed] [Google Scholar]
- Vivier E, Ugolini S, Blaise D, Chabannon C, Brossay L. Targeting natural killer cells and natural killer T cells in cancer. Nat Rev Immunol. 2012;12:239–252. doi: 10.1038/nri3174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, Nomura T, Sakaguchi S. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008;322:271–275. doi: 10.1126/science.1160062. [DOI] [PubMed] [Google Scholar]
- Wu K, Kryczek I, Chen L, Zou W, Welling TH. Kupffer cell suppression of CD8+ T cells in human hepatocellular carcinoma is mediated by B7-H1/programmed death-1 interactions. Cancer Res. 2009;69:8067–8075. doi: 10.1158/0008-5472.CAN-09-0901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y, Kuang DM, Pan WD, Wan YL, Lao XM, Wang D, Li XF, Zheng L. Monocyte/macrophage-elicited natural killer cell dysfunction in hepatocellular carcinoma is mediated by CD48/2B4 interactions. Hepatology. 2013;57:1107–1116. doi: 10.1002/hep.26192. [DOI] [PubMed] [Google Scholar]
- Xia L, Huang W, Tian D, Zhang L, Qi X, Chen Z, Shang X, Nie Y, Wu K. Forkhead box Q1 promotes hepatocellular carcinoma metastasis by transactivating ZEB2 and VersicanV1 expression. Hepatology. 2014;59:958–973. doi: 10.1002/hep.26735. [DOI] [PubMed] [Google Scholar]
- Xiao Z, Chung H, Banan B, Manning PT, Ott KC, Lin S, Capoccia BJ, Subramanian V, Hiebsch RR, Upadhya GA, Mohanakumar T, Frazier WA, Lin Y, Chapman WC. Antibody mediated therapy targeting CD47 inhibits tumor progression of hepatocellular carcinoma. Cancer Lett. 2015;360:302–309. doi: 10.1016/j.canlet.2015.02.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan W, Liu X, Ma H, Zhang H, Song X, Gao L, Liang X, Ma C. Tim-3 fosters HCC development by enhancing TGF-beta-mediated alternative activation of macrophages. Gut. 2015 doi: 10.1136/gutjnl-2014-307671. [DOI] [PubMed] [Google Scholar]
- Yang XH, Yamagiwa S, Ichida T, Matsuda Y, Sugahara S, Watanabe H, Sato Y, Abo T, Horwitz DA, Aoyagi Y. Increase of CD4+ CD25+ regulatory T-cells in the liver of patients with hepatocellular carcinoma. J Hepatol. 2006;45:254–262. doi: 10.1016/j.jhep.2006.01.036. [DOI] [PubMed] [Google Scholar]
- Yang Y, Huang CT, Huang X, Pardoll DM. Persistent Toll-like receptor signals are required for reversal of regulatory T cell-mediated CD8 tolerance. Nat Immunol. 2004;5:508–515. doi: 10.1038/ni1059. [DOI] [PubMed] [Google Scholar]
- Ye LY, Chen W, Bai XL, Xu XY, Zhang Q, Xia XF, Sun X, Li GG, Hu QD, Fu QH, Liang TB. Hypoxia-Induced Epithelial-to-Mesenchymal Transition in Hepatocellular Carcinoma Induces an Immunosuppressive Tumor Microenvironment to Promote Metastasis. Cancer Res. 2016;76:818–830. doi: 10.1158/0008-5472.CAN-15-0977. [DOI] [PubMed] [Google Scholar]
- Yeung OW, Lo CM, Ling CC, Qi X, Geng W, Li CX, Ng KT, Forbes SJ, Guan XY, Poon RT, Fan ST, Man K. Alternatively activated (M2) macrophages promote tumour growth and invasiveness in hepatocellular carcinoma. J Hepatol. 2015;62:607–616. doi: 10.1016/j.jhep.2014.10.029. [DOI] [PubMed] [Google Scholar]
- Yu MC, Chen CH, Liang X, Wang L, Gandhi CR, Fung JJ, Lu L, Qian S. Inhibition of T-cell responses by hepatic stellate cells via B7-H1-mediated T-cell apoptosis in mice. Hepatology. 2004;40:1312–1321. doi: 10.1002/hep.20488. [DOI] [PubMed] [Google Scholar]
- Zabala M, Lasarte JJ, Perret C, Sola J, Berraondo P, Alfaro M, Larrea E, Prieto J, Kramer MG. Induction of immunosuppressive molecules and regulatory T cells counteracts the antitumor effect of interleukin-12-based gene therapy in a transgenic mouse model of liver cancer. J Hepatol. 2007;47:807–815. doi: 10.1016/j.jhep.2007.07.025. [DOI] [PubMed] [Google Scholar]
- Zhao L, Lim SY, Gordon-Weeks AN, Tapmeier TT, Im JH, Cao Y, Beech J, Allen D, Smart S, Muschel RJ. Recruitment of a myeloid cell subset (CD11b/Gr1 mid) via CCL2/CCR2 promotes the development of colorectal cancer liver metastasis. Hepatology. 2013;57:829–839. doi: 10.1002/hep.26094. [DOI] [PubMed] [Google Scholar]
- Zhao W, Zhang L, Xu Y, Zhang Z, Ren G, Tang K, Kuang P, Zhao B, Yin Z, Wang X. Hepatic stellate cells promote tumor progression by enhancement of immunosuppressive cells in an orthotopic liver tumor mouse model. Lab Invest. 2014;94:182–191. doi: 10.1038/labinvest.2013.139. [DOI] [PubMed] [Google Scholar]
- Zhou G, Levitsky HI. Natural regulatory T cells and de novo-induced regulatory T cells contribute independently to tumor-specific tolerance. J Immunol. 2007;178:2155–2162. doi: 10.4049/jimmunol.178.4.2155. [DOI] [PubMed] [Google Scholar]
- Zhu XD, Zhang JB, Zhuang PY, Zhu HG, Zhang W, Xiong YQ, Wu WZ, Wang L, Tang ZY, Sun HC. High expression of macrophage colony-stimulating factor in peritumoral liver tissue is associated with poor survival after curative resection of hepatocellular carcinoma. J Clin Oncol. 2008;26:2707–2716. doi: 10.1200/JCO.2007.15.6521. [DOI] [PubMed] [Google Scholar]


