Abstract
Apigenin (4′,5,7,-trihydroxyflavone) is a flavonoid found in certain herbs, fruits, and vegetables. Apigenin can attenuate inflammation, which is associated with many chronic diseases of aging. Senescent cells—stressed cells that accumulate with age in mammals—display a pro-inflammatory senescence-associated secretory phenotype (SASP) that can drive or exacerbate several age-related pathologies, including cancer. Flavonoids, including apigenin, were recently shown to reduce the SASP of a human fibroblast strain induced to senesce by bleomycin. Here, we confirm that apigenin suppresses the SASP in three human fibroblast strains induced to senesce by ionizing radiation, constitutive MAPK (mitogen-activated protein kinase) signaling, oncogenic RAS, or replicative exhaustion. Apigenin suppressed the SASP in part by suppressing IL-1α signaling through IRAK1 and IRAK4, p38-MAPK, and NF-κB. Apigenin was particularly potent at suppressing the expression and secretion of CXCL10 (IP10), a newly identified SASP factor. Further, apigenin-mediated suppression of the SASP substantially reduced the aggressive phenotype of human breast cancer cells, as determined by cell proliferation, extracellular matrix invasion, and epithelial-mesenchymal transition. Our results support the idea that apigenin is a promising natural product for reducing the impact of senescent cells on age-related diseases such as cancer.
Electronic supplementary material
The online version of this article (doi:10.1007/s11357-017-9970-1) contains supplementary material, which is available to authorized users.
Keywords: Flavonoids, Human fibroblasts, Proliferation, Invasion, IL-6, IL-1A, IRAK1/4, NF-κB
Introduction
Aging is the largest risk factor for a host of age-related diseases, both degenerative and hyperplastic, raising the possibility that interventions into basic aging processes hold promise for ameliorating multiple age-related diseases, including cancer (Kennedy et al. 2014). Nine basic aging processes—or hallmarks of aging—were recently defined as targets for interventions to extend the health span (years of healthy aging) and, possibly the life span, of mammalian organisms (López-Otín et al. 2013). Among these hallmarks is cellular senescence.
Cellular senescence is a multifaceted stress response adopted by cells that have the ability to undergo cell division (Campisi 2013). The senescence response entails an essentially irreversible arrest of cell proliferation, coupled to a complex senescence-associated secretory phenotype (SASP) that includes numerous cytokines, chemokines, growth factors, and proteases (Coppe et al. 2008). The growth arrest is a potent tumor suppressive mechanism. However, because senescent cells accumulate with age, their chronic presence—most likely through the SASP—can drive age-related pathologies, including, ironically, cancer (Campisi 2013, Muñoz-Espín and Serrano 2014, Baker et al. 2016). This activity of senescent cells is likely attributable to the pro-inflammatory nature of the SASP (Freund et al. 2010; Lasry and Ben-Neriah 2015).
To identify novel SASP regulators and small molecules capable of suppressing the SASP, we screened a library of FDA-approved drugs for ability to suppress the secretion of IL-6, a robust and common SASP component (Coppe et al. 2008, Coppe et al. 2010) without compromising cell viability (Laberge et al. 2012a). This screen identified glucocorticoids as SASP inhibitors. However, a number of other candidates emerged from the screen that warranted further investigation. Among these candidates was apigenin (4′,5,7,-trihydroxyflavone; MW, 270.24), a naturally occurring flavonoid present in several plants and having little or no toxicity when obtained from the diet (Ross and Kasum 2002, Shukla and Gupta 2010).
Apigenin belongs to the flavone subclass of flavonoids (Kim 2003, Osada et al. 2004) and is abundant in a variety of fruits, vegetables, and herbs. It often exists in food sources as a glycoside, which improves its solubility and bioavailability (Ross and Kasum 2002). Like other flavonoids, apigenin has a variety of biological activities, including the ability to inhibit proliferation and induce apoptosis in several cancer cell lines (Reiners et al. 1999, Gupta et al. 2001, Way et al. 2004, Brusselmans et al. 2005), as well as an ability to inhibit angiogenesis (Kim 2003, Osada et al. 2004). Targets of apigenin that could contribute to these anti-cancer activities include heat shock proteins (Osada et al. 2004), fatty acid synthase (Brusselmans et al. 2005), the aryl hydrocarbon receptor (Reiners et al. 1999), HER2/neu (Way et al. 2004), and matrix metalloproteinases (Kim 2003). Apigenin can also reduce markers of inflammation in response to lipopolysaccharides, TNF-α, and allergens and was shown to suppress activities of the inflammatory mediators cyclooxygenase and NF-κB (Liang et al. 1999, Ruiz and Haller 2006, Li et al. 2010, Kang et al. 2011, Duarte et al. 2013).
Apigenin was recently shown to reduce the secretion of several SASP factors in a human fibroblast strain induced to senesce by the clastogen bleomycin via the IRAK1/IκBα signaling pathway (Lim et al. 2015). To determine how broadly apigenin suppresses the SASP, we analyzed its effects using several human fibroblast strains and senescence inducers, multiplex ELISAs, and several indicators of IL-1/IRAK/NF-κB signaling. Importantly, apigenin suppressed the ability of senescent fibroblasts to stimulate aggressive phenotypes of human breast cancer cells, supporting the idea that apigenin has potential as an anti-cancer agent.
Materials and methods
Cell culture
Except where noted otherwise, primary human primary fibroblasts (IMR-90 from fetal lung; BJ and HCA2 from neonatal foreskin) were seeded at 10,000 cells/cm2 and incubated at 37 °C in a 3% O2/10% CO2 atmosphere and Dulbecco’s modified Eagle medium (DMEM) (VWR, Cat# 45000-312) supplemented with glucose (4.5 g/l), glutamine (4 mM), 10% fetal bovine serum (FBS), and antibiotics (100 IU penicillin, 100 μg/ml streptomycin). Media were replaced every 48 h. HCA2 cells were used because of their higher SASP when compared to other fibroblast cell strains. The other two cell strains were used to confirm and extend our results in a different tissue type and gender. Human breast cancer cells (MDA-MB231, ZR75.1) were cultured under standard conditions in the same medium in 20% oxygen.
Induction of senescence by ionizing radiation and apigenin treatment of cells
Human fibroblasts were seeded and incubated overnight. The following day (day 0), cells were induced to senesce by ionizing irradiation (10 Gy X-ray), and immediately given media containing DMSO or apigenin, and incubated. Non-senescent cells were mock-irradiated (placed inside the irradiator for the same interval used for the irradiated samples, but without exposure to radiation), then given serum-free media containing DMSO or apigenin, and incubated. Twenty-four hours later (day 1), conditioned media were collected from non-senescent cells; senescent cells were left to incubate, with media replaced every 48 h. On day 9, senescent cells were given serum-free media with DMSO or apigenin, and conditioned media were collected 24 h later.
Quantification of IL-6 in conditioned media
IL-6 levels in conditioned media were quantified using the Perkin-Elmer AlphaLISA protocol (Cat# AL322F).
SA-β-gal assay
Senescence-associated beta-galactosidase (SA-β-gal) activity was determined using the BioVision Senescence Detection Kit (Cat# K320-250) protocol. For each experiment, approximately 100–150 cells were counted.
EdU proliferation assay
DNA synthesis was assessed by incorporation of EdU and visualization by fluorescence microscopy using the Invitrogen Click-iT Edu HCS Assay (Cat# C10350/C10351) protocol. For each experiment, approximately 100–150 cells were counted.
Invasion assay
MDA-MB231 cells (80,000 cells/well) were plated atop a layer of Matrigel in the upper chambers of Transwells (BD Biosciences). The lower chambers were filled with conditioned media (lacking apigenin) from non-senescent or senescent HCA2 fibroblasts previously treated with DMSO or apigenin for 10 days. After 18 h, cells that migrated to the underside of the upper chamber filter were stained and counted by light microscopy, as described (Coppé et al. 2010).
H-RASV12 and MKK6EE-induced senescence
HCA2 and BJ fibroblasts were infected with a control lentivirus (L3P) or lentiviruses expressing H-RASV12 or MKK6EE, and selected as described (Coppe et al. 2008, Freund et al. 2011). After selection, infected cells were reseeded and given media containing DMSO or apigenin (10 μM), and the media were refreshed after 48 h. After another 48 h, the cells were given serum-free media containing DMSO or apigenin, conditioned media were collected 24 h later, and IL-6 levels were quantified by AlphaLISA, normalizing to cell number.
NF-κB reporter assay
HCA2 fibroblasts were infected with a lentivirus carrying a NF-κB-firefly luciferase reporter (SA Biosciences, Cat# CLS-013L) according to the supplier’s protocol. After selection, the cells were seeded at 10,000 cells/cm2, induced to senesce by X-irradiation, then given media containing DMSO or apigenin, and cultured for 10 days, with media refreshed every 48 h. On day 8, non-senescent cells were seeded at 5000 cells/cm2. On day 9, all cultures were given serum-free media with DMSO or apigenin. Twenty-four hours later, cells were lysed and luciferase activity measured using the Promega Luciferase Assay System (Cat# E1500).
IL-1 alpha stimulation of cytokine secretion
Fibroblasts were seeded at 10,000 cells/cm2 in two 12-well plates, irradiated to induce senescence, given media containing DMSO or apigenin, and incubated for 10 days. On day 8, two additional plates were seeded with non-senescent cells at 5000 cells/cm2 and incubated overnight. On day 9, non-senescent cells were given media containing DMSO or apigenin and incubated for 48 h. On day 11, all the cultures were washed, given serum-free media containing DMSO or apigenin plus the indicated amounts of recombinant IL-1 alpha (R&D Systems, Cat# 200-LA-010), and incubated overnight. The following day, conditioned media were collected and analyzed for IL-6 using an AlphaLISA, normalizing to cell number.
Western blotting
Cell lysates (25 μg protein) were resolved using Life Technologies NuPage 4–12% Bis-Tris Gels (Cat# NP0336) and transferred onto PVDF membranes. The membranes were blocked using 5% BSA and incubated with primary antibodies in blocking buffer overnight at 4 °C. The membranes were then incubated with the appropriate secondary antibody (BioRad Cat# 172-1011, anti-mouse; or #170-6515, anti-rabbit) followed by detection using a chemiluminescence ECL kit (Amersham LifeSciences Inc.). To control for protein loading, the membrane was stripped and probed with an antibody against actin or tubulin. Primary antibodies were as follows: p38 Thr180/Tyr182 phosphorylation—PhosphoSolutions, Cat# 1901802; total p38—Cell Signaling, Cat# 9212; IRAK4 Thr345/Ser346 phosphorylation—Cell Signaling, Cat# 7652; total IRAK4—Santa Cruz, Cat# SC34770; IRAK1 Thr387 phosphorylation—Cell Signaling, Cat# 4365; actin—Chemicon International, Cat# MAB3128; ZO-1—Santa Cruz, Cat# sc10804; cytokeratin 18—Santa Cruz, Cat# sc28284; vimentin—Millipore, Cat# ab1620; tubulin—Sigma, Cat# T8203.
Immunofluorescence
Cells were cultured in 8-well chamber slides, fixed in 4% formaldehyde (Sigma) for 10 min at 4 °C and permeabilized in PBS + 0.5% Triton for 10 min at 4 °C. Slides were blocked for 30 min in 4% goat serum (Invitrogen). Primary antibody (anti-ZO1 (described above) was diluted in blocking buffer at 1:100 dilution and incubated with cells for 1 h at room temperature. Cells were washed, incubated with secondary antibody (Invitrogen AlexaFluor 488 goat/anti-rabbit, Cat# A-11008) 1:1000 for 30 min at room temperature, washed, and mounted with Slow-Fade Gold (Molecular Probes). Images were acquired using an Olympus BX20 fluorescence microscope with Spotfire software (Diagnostics Instruments) and processed with Photoshop CS (Adobe).
Statistical analysis
Results presented are representative of at least three separate experiments using three biological replicates. We used the Student’s T test to compare the results from treated to untreated samples, normalized to DMSO-treated non-senescent controls where applicable. An asterisk indicates significance of p < 0.05, double asterisks indicate significance of p < 0.01, and triple asterisks indicate significance of p < 0.005.
Results
Suppression of IL-6 expression upon apigenin treatment
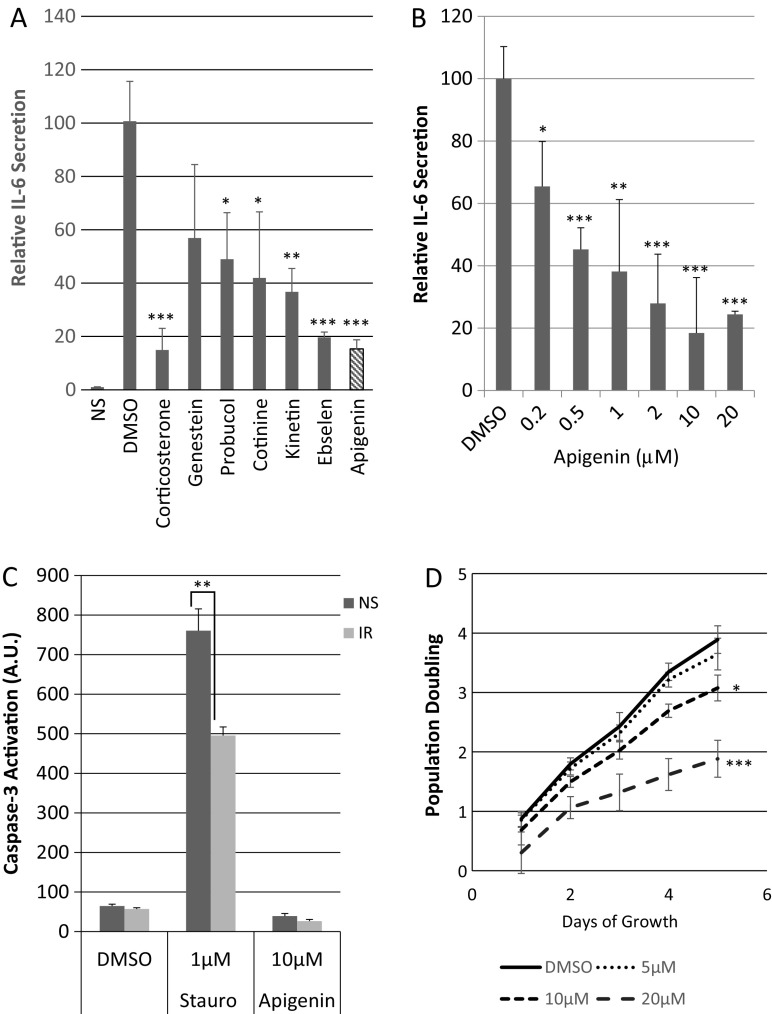
Upon screening a library of FDA-approved compounds for ability to regulate the secretion of IL-6 (Laberge et al. 2012a), a prominent component of the human and mouse SASP (Coppe et al. 2008; Coppe et al. 2010), we identified apigenin as significantly more active than the vehicle (DMSO) control. Apigenin reduced IL-6 secretion by primary human fibroblasts (HCA2, from neonatal foreskin) made senescent by ionizing radiation (IR; 10 Gy X-irradiation) to a greater extent than several other compounds in the library, with activity similar that of the most active compound in the library (corticosterone) (Fig. 1a).
Fig. 1.
Apigenin downregulates IL-6 secretion and moderately reduces fibroblast proliferation but does not induce apoptosis. a The indicated compounds from the Prestwick Library were used at 2 μM to treat primary human HCA2 fibroblasts immediately following X-irradiation at 10 Gy. Cells were treated for 10 days, after which conditioned media (CM) were collected and analyzed using the Perkin-Elmer IL-6 AlphaLISA assay. CM from non-senescent (NS) cells treated with vehicle (DMSO) provided a baseline for IL-6 secretion. Senescent cells were also treated with DMSO (negative control) and corticosterone (100 nM) (positive control). b HCA2 fibroblasts were induced to senesce (10 Gy X-irradiation) and immediately treated with increasing concentrations of apigenin for 10 days. CM were collected and analyzed for IL-6 secretion. IL-6 levels secreted by senescent cells were set at 100%. c HCA2 cells were induced to senesce as above and 7 days later were reseeded in 96-well plates (10,000 cells/well) and allowed to recover for 24 h. In parallel, non-senescent cells were seeded at 2500 cells/well. After 24 h, both senescent and non-senescent cells were treated with DMSO, 1 μM staurosporin or 10 μM apigenin, incubated for 48 h, and then caspase-3 activity was measured by luminescence using the Promega ApoTox Glo kit and arbitrary units. d HCA2 fibroblasts were seeded at 5000 cells/well in five 12-well plates, one for each day of a 5-day treatment with DMSO, 5, 10, or 20 μM apigenin. Three samples corresponding to each concentration were counted from one plate daily to determine cell number
We determined 10 μM to be the lowest dose at which apigenin maximally attenuated IL-6 secretion by senescent HCA2 fibroblasts (Fig. 1b) and showed a similar dose response for BJ fibroblasts (Fig. S1A), also from neonatal foreskin, as described (Lim et al. 2015). We used 10 μM apigenin for subsequent experiments.
Apigenin does not cause apoptosis and moderately reduces fibroblast proliferation
To better understand how apigenin suppressed senescence-associated IL-6 secretion and determine whether it had deleterious effects on non-senescent cells, we asked whether it induced apoptosis or inhibited cell proliferation. Apigenin was reported to induce apoptosis of cancer cells (Jayasooriya et al. 2012). To determine whether this was the case for normal cells, we treated non-senescent and IR-induced senescent HCA2 cells with DMSO or 10 μM apigenin and assessed apoptosis by activated caspase-3 levels (Fig. 1c). Apigenin failed to increase caspase-3 activity in both cell types, whereas 1 μM staurosporine (positive control) increased activity both types of cells. In addition, 10 μM apigenin moderately reduced proliferation (Fig. 1d). We treated proliferating HCA2 cells with 0, 5, 10, and 20 μM apigenin for 5 days. Over this period, untreated cells increased in number approximately 4-fold, whereas 5, 10, and 20 μM apigenin reduced this cell number by approximately 5, 25, and 50%, respectively.
Apigenin does not affect SA-β-gal expression, cell morphology, or growth arrest
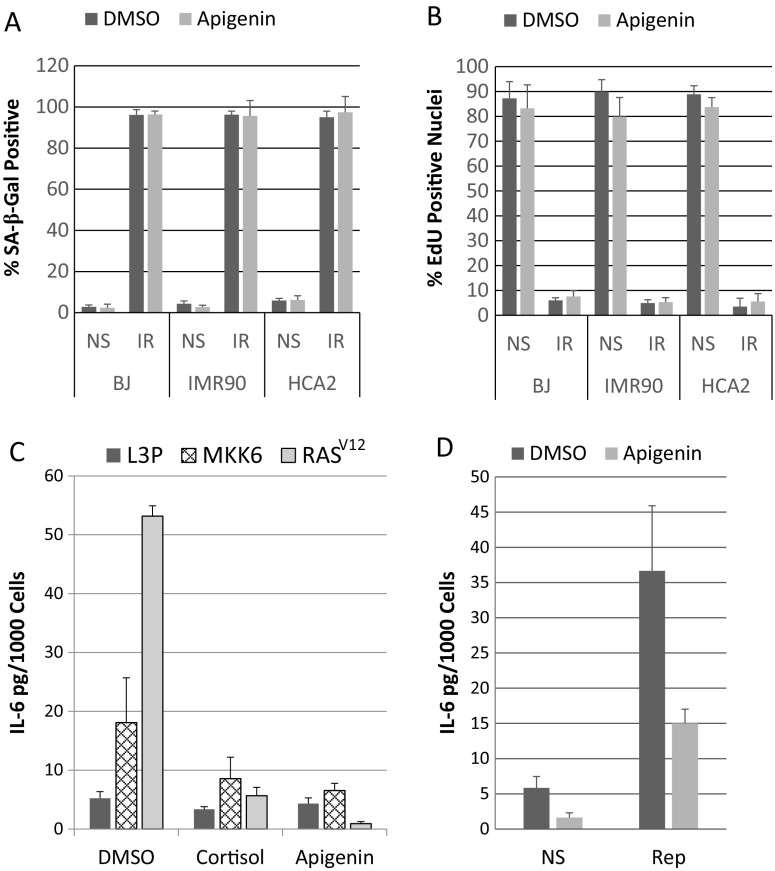
In addition to arresting growth, senescent cells develop an enlarged morphology and express a neutral senescence-associated β-galactosidase (SA-β-gal) (Dimri et al. 1995). Apigenin did not significantly alter SA-β-gal expression by non-senescent or senescent populations of three human fibroblast strains (BJ, HCA2, as well as IMR-90 from female fetal lung) (Fig. 2a). In addition, apigenin had no significant effect on the senescence growth arrest, as measured by incorporation of the fluorescent thymidine analogue EdU into newly synthesized DNA over a 24-h period (Fig. 2b). Finally, the enlarged morphology of senescent cells remained unchanged by apigenin (data not shown).
Fig. 2.
Effects of apigenin treatment on senescence-associated phenotypes. a Human BJ, IMR90, and HCA2 fibroblasts were non-senescent (NS) or induced to senesce as described in the legend to Fig. 1 (IR) and treated with DMSO or apigenin for 10 days. Thirty-six hours before collection of senescent samples, NS cells were plated at 10,000/cm2 and allowed to recover overnight, and media on NS and IR cultures were replaced with serum-free media containing DMSO or apigenin. The following day, cells were stained for SA-β-gal activity and 100–150 cells were counted. b Cells were prepared as in (a) except 10,000/cm2 of senescent or non-senescent cells were seeded into each well of a 8-well slide, and EdU was added to the serum-free media and 100–150 cells were counted. c HCA2 fibroblasts were infected with L3P (insertless vector), MKK6EE, or H-RASV12 containing lentiviruses. Infected cells were selected with 1 μg/ml puromycin for 24 h, allowed to recover for 4 days, and reseeded. Five days later, all media were replaced with serum-free media containing apigenin or DMSO for 24 h. Then, CM was collected and analyzed for IL-6 secretion. d Replicatively senescent (PD 60.2) HCA2 fibroblasts were seeded at 10,000/cm2 onto 12-well plate and allowed to recover for 48 h. Cells were then treated with DMSO or apigenin (media refreshed every 48 h). On day 8, non-senescent cells were seeded onto 6-well plates at 10,000/cm2 and allowed to recover overnight. On day 9, all samples were given serum-free media supplemented with DMSO or apigenin, and collected 24 h later (day 10) and analyzed for IL-6 secretion
Apigenin suppresses oncogene- and replication-induced IL-6 secretion
In addition to the genotoxic stress caused by IR, other stressors induce a senescence response and SASP, including activated oncogenes or signaling kinases and telomere erosion caused by repeated replication (Coppe et al. 2008, Rodier et al. 2009, Freund et al. 2011). To determine whether apigenin was capable of suppressing IL-6 secretion by human fibroblast strains induced to senescence by activated oncogenes or signaling kinases, we infected HCA2 cells with an insertless lentivirus (L3P) or lentiviruses expressing oncogenic RAS (H-RASV12) (Beausejour et al. 2003) or a constitutively active MAP kinase kinase 6 mutant (MKK6EE) (Freund et al. 2011), both of which induce a strong senescence response. After selection, the infected cells were reseeded and given DMSO or 10 μM apigenin. Four days later, we measured secreted IL-6 levels (Fig. 2c). Whether cells senesced in response to oncogenic H-RASV12 or MKK6EE, apigenin (as well as the positive control, cortisol) significantly suppressed IL-6 secretion (Fig. 2c). Similarly, apigenin reduced the level of IL-6 secreted by replicatively senescent HCA2 fibroblasts (60 population doublings) by almost 60% (Fig. 2d).
Kinetics of apigenin suppression of IL-6 secretion
To determine the kinetics with which apigenin reduces IL-6 secretion by senescent HCA2 fibroblasts, we conducted three experiments.
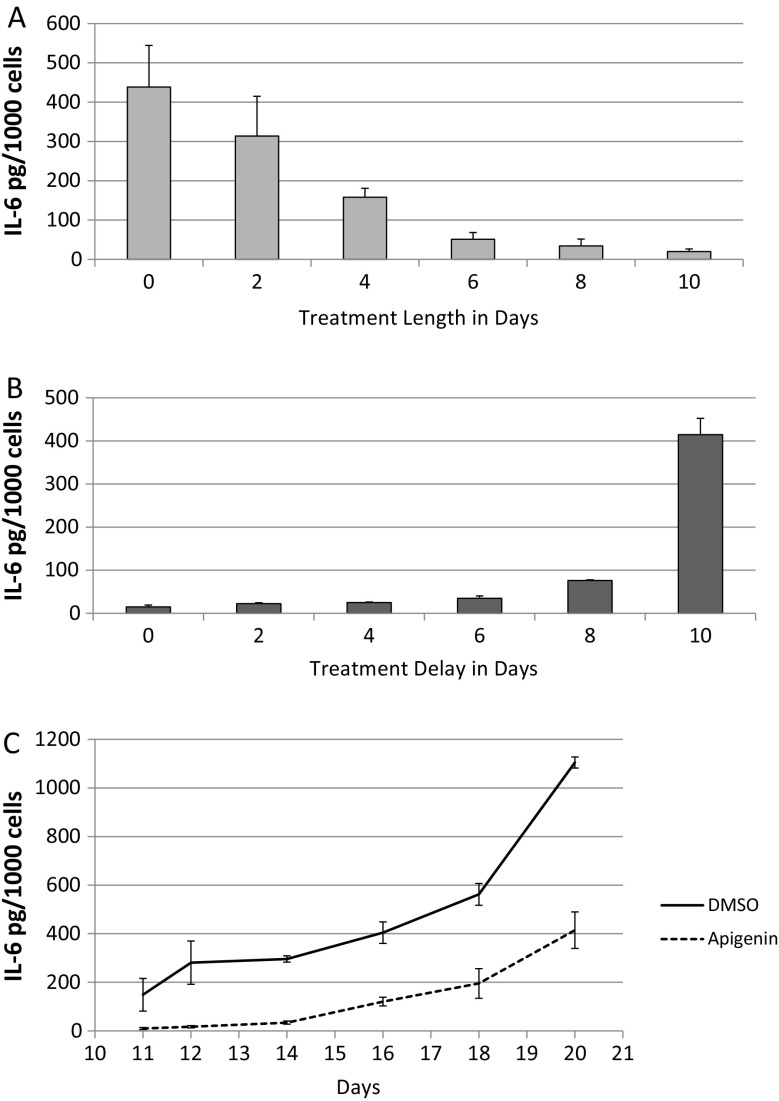
First, we treated cells with DMSO or apigenin immediately after IR (day 0) (Fig. 3a). On each following day, for 10 consecutive days, we replaced the apigenin-containing media with DMSO-containing media, thus releasing the cells from the treatment. On day 10, we collected conditioned media and quantified IL-6. As expected, senescent cells that were never exposed to apigenin secreted the highest levels of IL-6. IL-6 secretion progressively decreased with increasing time of apigenin exposure, reaching maximal reduction after 6 days of treatment.
Fig. 3.
Effect of timing of apigenin treatment on IL-6 secretion. a, b HCA2 fibroblasts were seeded at 10,000/cm2 into two 24-well plates and induced to senesce by IR. 10 days later, we varied the length of continuous treatment with apigenin starting immediately after IR in one plate (a), and in the other plate, we varied the day of initiation of treatment with apigenin after IR (b). Duplicate samples were treated with media containing apigenin or DMSO and refreshed every 48 h. On day 9, media were replaced with serum-free media containing apigenin or DMSO, and 24 h later, cells were counted and conditioned media analyzed for IL-6 secretion. c HCA2 fibroblasts were seeded at 10,000/cm2 into a 24-well plate and induced to senesce by IR. Immediately following IR, media were refreshed with DMSO or apigenin and incubated for 10 days (media refreshed every 48 h with DMSO or apigenin). On day 10, cells were washed and incubated with serum-containing media except for the first sample (day 11) that was replaced with serum-free media. Samples for subsequent time points were similarly washed and media replaced with serum-free media 24 h before collection. After the final time point on day 20, CM for all time points were analyzed for IL-6
In the second 10-day experiment (Fig. 3b), we delayed the initiation of apigenin treatment after IR for varying intervals and then quantified IL-6 secretion. As expected, continuous treatment (no treatment delay) reduced IL-6 secretion maximally. No treatment (day 10) yielded the highest IL-6 secretion level. Interestingly, treatment at day 8 for 48 h was sufficient to produce a robust decrease in IL-6 secretion (>4-fold), indicating that apigenin can suppress a fully formed SASP, which takes about 7 days to develop (Coppe et al. 2008).
Finally, we examined the recovery of IL-6 secretion after apigenin treatment. We treated senescent cells with apigenin or DMSO continuously for 10 days, then washed the cells, and added a standard culture medium (Fig. 3c). Over the next 10 days, we collected conditioned media and then quantified IL-6 levels. The ability of apigenin to suppress IL-6 secretion lasted 4–5 days after removal, reaching levels achieved by DMSO-treated cells in 5–6 days. Interestingly, IL-6 secretion by DMSO-treated cells also rose during this period, possibly due to nutrient replenishment and mTOR signaling, which drives the SASP (Laberge et al. 2015).
Secretion profile of apigenin-treated senescent cells
To obtain a broader view of how apigenin suppressed the SASP, we used a bead-based ELISA assay (Luminex) to quantify the levels of 51 cytokines, some, but not all, of which were shown to be SASP factors by semi-quantitative antibody arrays. We used IMR90 and BJ cells to compare their responses and determine whether effects were cell of origin or sex specific (Fig. S1B; Fig. S2).
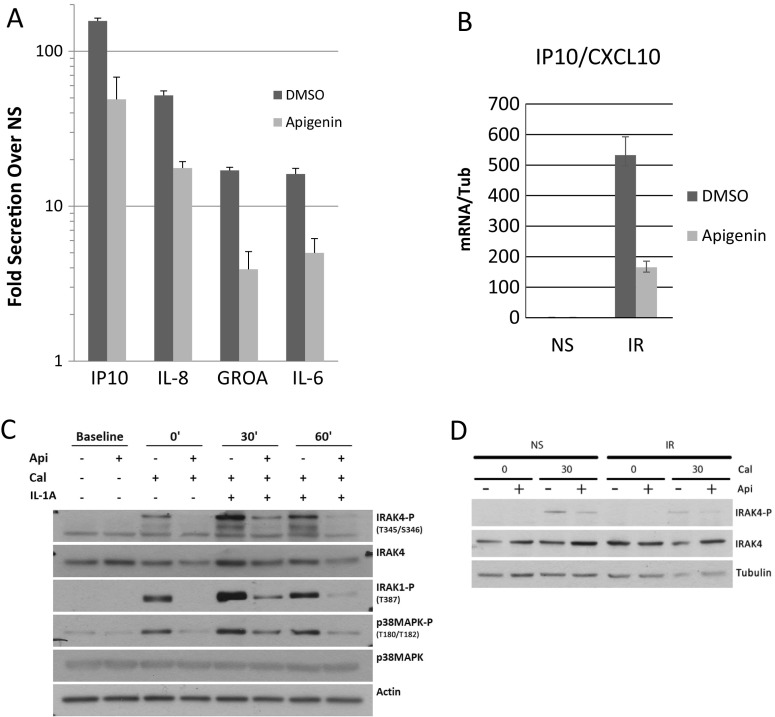
Ten days after IR, both fibroblast strains developed a typical SASP compared to sham-irradiated controls, showing increased expression and secretion of several pro-inflammatory cytokines, including IL-8, IL-6, and GROA (Fig. 4a; Fig. S2A-B, S3) as described (Coppe et al. 2008, Lim et al. 2015). This increase was reduced by apigenin in both strains to similar extents.
Fig. 4.
Effects of apigenin on the SASP and its regulation. a We compared the levels of selected cytokines (from the Luminex screening) secreted by treated (apigenin) and untreated (DMSO), NS, or IR senescent IMR90 fibroblasts. Levels of IP10, IL-8, GROA, and IL-6 in the CM from senescent cells are shown relative to NS. b Using qPCR, we determined the mRNA level of IP10 in DMSO- and apigenin-treated non-senescent (NS) and senescent (IR) BJ fibroblasts. c The kinetics of phosphorylation of IRAK4, IRAK1, and p38MAPK in non-senescent BJ fibroblasts stimulated with IL-1A and treated or not with apigenin, was examined using stabilization of phosphorylation by calyculin A (Cal). d IRAK4 phosphorylation in response to apigenin treatment was compared between senescent and non-senescent BJ fibroblasts
As expected (Coppe et al. 2008), certain markers of acute inflammation, such as TGFβ, were relatively low in the secretory profile of senescent IMR90 and BJ cells, whereas many other markers of inflammation, such as IL-6 and IL-8, were markedly upregulated (Fig. S2A, B). In addition to known SASP factors, we identified a novel SASP factor, the chemokine interferon-γ-inducible protein 10 (IP10/CXCL10). This chemokine was secreted at high levels by senescent fibroblasts, equaling the level of IL-6. Similar to other prominent SASP factors, IP10/CXCL10 secretion was robustly reduced by apigenin (Fig. 4b).
Apigenin reduces NF-κB activity stimulated by IL-1A
The NF-κB transcription factor is a major positive regulator of inflammatory responses (Baker et al. 2011), including much of the SASP (Freund et al. 2011). Further, apigenin reduced NF-κB activity and improved survival after an acute, otherwise lethal, inflammatory stimulus (Nicholas et al. 2007). To determine whether apigenin reduced the SASP by suppressing NF-κB activity, we used a lentiviral-delivered reporter consisting of NF-κB transcriptional response elements driving firefly luciferase. We expressed this reporter in HCA2 fibroblasts, induced the cells to senesce by IR, and then measured IL-6 secretion and luciferase activity. As expected, senescent cells showed a robust increase in IL-6 secretion, which was abolished by apigenin (Fig. S4A). In parallel, senescent cells showed a 4- to 5-fold increase in NF-κB activity, which was reduced to <2-fold by apigenin (Fig. S4B).
The NF-κB activity in senescent cells is driven in large measure by an IL-1A-NF-κB-positive feedback loop: the cells express cell surface-bound IL-1A, which acts in a juxtacrine manner to induce several genes, including IL-1A itself (Orjalo et al. 2009). To determine whether apigenin’s ability to reduce the SASP depended on this IL-1A feeback loop, we treated non-senescent and senescent HCA2 cells with DMSO or apigenin, added increasing concentrations of recombinant IL-1A, and then analyzed conditioned media for IL-6. Apigenin reduced IL-1A-stimulated IL-6 secretion in both non-senescent and senescent cells at all IL-1A concentrations tested (Fig. S4C), consistent with its ability to suppress NF-κB activity. Thus, the anti-inflammatory effects of apigenin are likely mediated in part by its ability to suppress NF-κB activity initiated by IL-1A.
Apigenin reduces IRAK1/4 and p38MAPK phosphorylation
IL-1A signaling stimulates the SASP owing to phosphorylation the signal transduction proteins IRAK1 and IRAK4, then ultimately of p38MAPK on Thr180 (Freund et al. 2011). Apigenin was shown to inhibit p38MAPK phosphorylation in prostate cancer cells, albeit at a higher concentration (40 μM) (Shukla and Gupta 2007). To determine whether apigenin alters IRAK1/IRAK4/p38MAPK phosphorylation in normal cells at a concentration that inhibited the SASP (10 μM), we stimulated non-senescent BJ fibroblasts with IL-1A in the presence of the phosphatase inhibitor calyculin. Using western blotting, we then analyzed for the phosphorylated forms of IRAK1 (Thr387), IRAK4 (Thr345/Ser346), and p38MAPK (Thr180/Tyr182) (Fig. 4c). Phosphorylation of the three proteins was substantially reduced by apigenin.
Noting the reduction of IRAK4 phosphorylation, proximal to the start of the IL-1A cascade, we sought to examine the effects in senescent cells without exogenous stimulation by IL-1A. We therefore made primary BJ fibroblasts senescent by irradiation and showed they also exhibited a decrease in IL-1A-mediated IRAK4 phosphorylation with apigenin treatment (Fig. 4d). This decrease suggests that NF-κB activity in senescent cells could at least in part be reduced by apigenin through inhibition of IRAK4.
Apigenin suppresses ability of the SASP to stimulate breast cancer cell aggressiveness
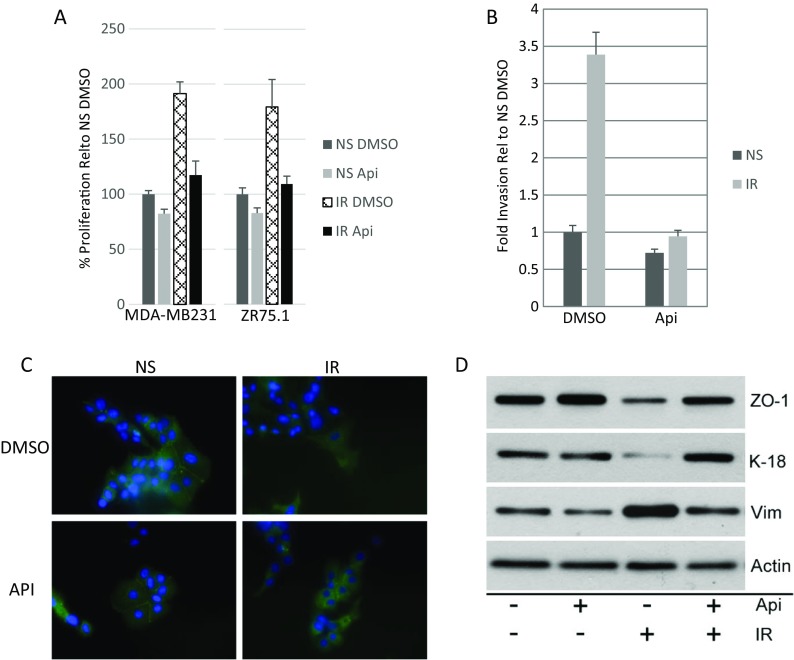
The SASP can disrupt epithelial organization and promotes premalignant epithelial cell proliferation and invasion in culture and in vivo (Krtolica et al. 2001, Coppe et al. 2008, Coppe et al. 2010, Laberge et al. 2015). We therefore determined the ability of apigenin to counterbalance the effects of the SASP on breast cancer cell aggressiveness. We treated senescent (IR-induced) HCA2 cells with vehicle or apigenin for 10 days, washed the cells, and then collected conditioned media (CM) over the next 24 h. We added the CM to relatively aggressive MDA-MB231 and non-aggressive ZR75.1 human breast cancer cells and monitored their ability to proliferate (Fig. 5a). As expected, the SASP-containing CM from senescent cells stimulated the proliferation of both breast cancer cell lines to a significantly greater extent than CM from non-senescent cells. Importantly, apigenin significantly reduced the ability of the SASP to stimulate cancer cell proliferation.
Fig. 5.
Apigenin suppresses the ability of the SASP to induce cancer cell aggressiveness. a MDA-MB231 (left panel) and ZR75.1 (right panel) breast cancer cells were cultured in presence of conditioned media (CM) from DMSO- or apigenin (Api)-treated fibroblasts and monitored for proliferation over 3 days. b CM were prepared from non-senescent (NS) cells or senescent (IR) cells, treated or not with apigenin (Api). CM were assayed for ability to stimulate MDA-MB231 human breast cancer cells to invade a basement membrane, as described in the “Materials and methods” section. Invasion stimulated by NS CM was given a value of one, and other conditions were normalized to this value. Error bars indicate the standard deviation around the mean. c ZR75.1 cells were incubated with the indicated CM for 3 days and immunostained for the tight junction protein ZO-1. d Using western blotting, we analyzed the expression of ZO-1, the epithelial marker cytokeratin 18 (K-18), and the mesenchymal marker vimentin (Vim) in ZR75-1 cells. The effect of CM from apigenin-treated NS and IR fibroblasts on the expression of these three markers is presented on lanes 2 and 4, whereas lanes 1 and 3 show the expression in cells cultured in CM from DMSO-treated NS and IR fibroblasts, respectively
Apigenin also suppressed the ability of the SASP to stimulate MDA-MB231 cells to invade a basement membrane in Boyden chambers (Fig. 5b). SASP-containing CM from senescent fibroblasts stimulated 3.5-fold more invasion than CM from non-senescent fibroblasts, and apigenin reduced this stimulation to non-senescent levels. Consistent with this finding, apigenin suppressed the ability of the SASP to induce an epithelial-mesenchymal transition (EMT) and confer on epithelial cells their invasive and metastatic properties, which is an important step during cancer progression (Laberge et al. 2012b). By immunofluorescence (Fig. 5c) and/or western blotting (Fig. 5d), control, non-aggressive, ZR75.1 cells expressed the tight junction protein ZO-1 and epithelial cytoskeletal protein keratin (K)-18, as well as detectable levels of the mesenchymal cytoskeletal protein vimentin. As expected (Coppe et al. 2008, Coppe et al. 2010), SASP-containing CM reduced ZO-1 and K-18 expression and increased vimentin expression, consistent with inducing an EMT. However, CM from apigenin-treated senescent fibroblasts reestablished the higher expression of ZO-1 and K-18 and lower expression of vimentin (Fig. 5c, d). Thus, apigenin can indirectly reduce the aggressive phenotype of breast cancer cells stimulated by the SASP.
Discussion
Discovering new molecules that can prevent or attenuate the deleterious effects of the SASP, and also have low or non-existent negative side effects, holds promise for the development of safe therapeutic interventions into the many age-related diseases in which the SASP has been implicated. We followed up on earlier results from a high content screen to identify compounds that either eliminate senescent cells or suppress the SASP (Laberge et al. 2012a). Among the compounds that robustly reduced the secretion of IL-6, a sentinel SASP factor, was apigenin, one of the few natural products included in the library (the Prestwick Library of FDA-approved compounds). Here, we examined the effects of apigenin on several aspects of the senescent phenotype of normal human fibroblasts.
Apigenin reduced IL-6 secretion by two independent strains of senescent human fibroblasts (HCA2 and BJ) in a dose-dependent manner. At 10 μM, apigenin significantly and near-maximally reduced IL-6 secretion by these normal cells. Because apigenin was shown to induce apoptosis and inhibit cell proliferation in cancer cell lines (Reiners et al. 1999, Gupta et al. 2001, Way et al. 2004, Brusselmans et al. 2005, Shukla and Gupta 2007, Jayasooriya et al. 2012), we tested it for these activities in normal human fibroblasts. At 10 μM, apigenin did not induce apoptosis in non-senescent or senescent normal cells and, while robustly reducing IL-6 secretion, only decreased proliferation of non-senescent cells by ∼25% while the flavonoid was present. We saw significant decreases in IL-6 secretion with concentrations as low as 1 μM. These data suggest that a plasma concentrations of ∼1.5 μM apigenin, which is achievable through dietary sources (Gradolatto et al. 2005), can be an effective suppressor of the SASP.
One proposed mechanism for the ability of apigenin to suppress inflammation is inhibition of NF-κB activity. At 100 μM, apigenin suppressed TNF-α-activated NF-κB activity in transformed mouse intestinal cells at the level of phosphorylation and degradation of the inhibitory network (Ruiz and Haller 2006). By contrast, at 20 μM, apigenin reduced TNF-α-stimulated NF-κB-reporter activity without affecting the phosphorylation or degradation of NF-κB pathway components, DNA binding activity or translocation of NF-κB to the nucleus in NIH-3T3 immortalized fibroblasts (Funakoshi-Tago et al. 2011a, 2011b). Thus, apigenin can act on NF-κB activity by different mechanisms in different cell types. We found that apigenin inhibited NF-κB activity at 10 μM in normal human fibroblasts primarily by inhibiting signaling through IL-1A, an important upstream regulator of the inflammatory arm of the SASP (Orjalo et al. 2009). Thus, apigenin did not alter the phosphorylation state of MSK1, which phosphorylates the activation domain of NF-κB (Reber et al. 2009).
However, apigenin was shown to inhibit the phosphorylation state of p38MAPK in cancer cell lines (Noh et al. 2010), and we showed p38MAPK is important for the SASP of normal human cells (Freund et al. 2011). Indeed, here we show that apigenin inhibited p38MAPK phosphorylation on thr380, a critical residue for its activation, in two normal human fibroblast strains. Accordingly, apigenin suppressed the expression and secretion of several SASP factors. These factors included IL-6 and IL-8, which are important pro-inflammatory cytokines, and GROA, a potent growth factor; all three were shown to promote aggressive phenotypes in human breast cancer cells (Coppe et al. 2008; Coppe et al. 2010).
Of interest, we also identified IP10 as a SASP factor and target of suppression by apigenin. As its name suggests, IP10 is induced by interferon-gamma, along with CXCL9 and CXCL11, which together promote a Th1 response to viral infection, in essence a response to cellular damage. Interestingly, serum levels of IP10 increase during normal aging (Antonelli et al. 2005) and may promote abnormal immune responses in the elderly. The receptor for IP10, CXCR3, activates NF-κB which has a binding site in the promoter of IP10. Thus, IP10 can stimulate its own transcription, creating a feedback loop that has the potential to reinforce the SASP in a fashion to that of IL-1A (Orjalo et al. 2009).
In flavones such as apigenin, the ABC three-ring backbone is similar in structure to ATP (Funakoshi-Tago et al. 2011a, 2011b). Because flavones are known to inhibit ATP hydrolysis, apigenin could compete with ATP for binding to p38MAPK, and other kinases as well, and thus inhibit multiple kinases simultaneously. The pleotropic nature of this inhibition could make determining the precise mechanism(s) of action of apigenin challenging. However, as a potential intervention into cancer and possibly aging, having multiple targets involved in inflammation and growth could result in a synergy that would make it a promising compound for regulating several pathways involved in healthy longevity.
Electronic supplementary material
Apigenin down-regulates the SASP in BJ fibroblasts. (A) Primary human BJ fibroblasts were induced to senesce (10 Gy X-irradiation) and immediately treated with increasing concentrations of apigenin for 10 days. CM were collected and analyzed for IL-6 secretion. (B) We investigated the levels of IL-8, GROA, IL-6 and IP10 secreted by treated (apigenin) and untreated (DMSO), NS or senescent BJ fibroblasts. Samples were in quadruplicate and levels of individual cytokines in senescent samples were normalized to the levels in DMSO-treated NS cells. (PDF 23 kb)
Apigenin reduces cytokine secretion of senescent IMR90 and BJ fibroblasts. (A) Primary IMR90 (PD 37) and (B) primary BJ (PD 34) human fibroblasts were induced to senesce by IR, and treated with apigenin or DMSO. CM were collected and the levels of 51 cytokines were analyzed by Luminex. Samples were in quadruplicate and levels of individual cytokines in senescent samples were normalized to the levels in DMSO-treated NS cells. (PDF 26 kb)
Effect of apigenin on the expression of major SASP factors in BJ fibroblasts. Using qPCR we determined the mRNA levels of major SASP factors in DMSO- and apigenin-treated non-senescent and senescent BJ fibroblasts (IL-6, IL-8, and GROA). IL-1A and IL-1B were also investigated at the mRNA level. (PDF 15 kb)
Apigenin reduces the senescence-induced inflammatory pathway activation. (A) IL-6 secretion of cells infected with a lentivirus NF-κB-luciferase reporter construct, induced to senesce by IR in the presence of apigenin, is shown. (B) NF-κB reporter activation in the senescent cells in (A) was measured by luminescence using the Promega Luciferase Assay System. (C) Induction of IL-6 secretion in NS and senescent HCA2 cells upon administration of three concentrations of recombinant IL-1A was measured by AlphaLISA. (PDF 18 kb)
Acknowledgments
We thank Dr. Remi-Martin Laberge for comments and discussions. This work was supported by the SENS Research Foundation to KP and by NIH grants AG009909 and AG017242 to JC.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflicts of interest.
References
- Antonelli A, Rotondi M, Fallahi P, Romagnani P, Ferrari S, Ferrannini E, Serio M. Age-dependent changes in CXC chemokine ligand 10 serum levels in euthyroid subjects. J Interf Cytokine Res. 2005;25:547–552. doi: 10.1089/jir.2005.25.547. [DOI] [PubMed] [Google Scholar]
- Baker DJ, Childs BG, Durik M, Wijers ME, Sieben CJ, Zhong J, Saltness R, Jeganathan KB, Casaclang Versoza GC, Pezeshki A, Khazaie K, Miller JD, van Deursen JM. Naturally occurring p16Ink4a-positive cells shorten healthy lifespan. Nature. 2016;530:184–189. doi: 10.1038/nature16932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker RG, Hayden MS, Ghosh S. NF-κB, inflammation, and metabolic disease. Cell Metab. 2011;13:11–22. doi: 10.1016/j.cmet.2010.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beausejour CM, Krtolica A, Galimi F, Narita M, Lowe SW, Yaswen P, Campisi J. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 2003;22:4212–4222. doi: 10.1093/emboj/cdg417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brusselmans K, Vrolix R, Verhoeven G, Swinnen JV. Induction of cancer cell apoptosis by flavonoids is associated with their ability to inhibit fatty acid synthase activity. J Biol Chem. 2005;280:5636–5645. doi: 10.1074/jbc.M408177200. [DOI] [PubMed] [Google Scholar]
- Campisi J. Aging, cellular senescence, and cancer. Annu Rev Physiol. 2013;75:685–705. doi: 10.1146/annurev-physiol-030212-183653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppé J-P, Desprez P-Y, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010;5:99–118. doi: 10.1146/annurev-pathol-121808-102144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppe JP, Patil CK, Rodier F, Krtolica A, Beausejour C, Parrinello S, Hodgson G, Chin K, Desprez PY, Campisi J. A human-like senescence-associated secretory phenotype is conserved in mouse cells dependent on physiological oxygen. PLoS One. 2010;5 doi: 10.1371/journal.pone.0009188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppe JP, Patil CK, Rodier F, Sun Y, Munoz D, Goldstein J, Nelson PS, Desprez PY, Campisi J. Senescence-associated secretory phenotypes reveal cell non-automous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853–2868. doi: 10.1371/journal.pbio.0060301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, Medrano EE, Linskens M, Rubelj I, Pereira-Smith OM, Peacocke M, Campisi J. A novel biomarker identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci U S A. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duarte S, Arango D, Parihar A, Hamel P, Yasmeen R, Doseff AI. Apigenin protects endothelial cells from lipopolysaccharide (LPS)-induced inflammation by decreasing caspase-3 activation and modulating mitochondrial function. Internatl J Molec Sci. 2013;14:17664–17679. doi: 10.3390/ijms140917664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freund A, Orjalo A, Desprez PY, Campisi J. Inflammatory networks during cellular senescence: causes and consequences. Trends Molec Med. 2010;16:238–248. doi: 10.1016/j.molmed.2010.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freund A, Patil PK, Campisi J. p38MAPK is a novel DNA damage response-independent regulator of the senescence-associated secretory phenotype. EMBO J. 2011;30:1536–1548. doi: 10.1038/emboj.2011.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Funakoshi-Tago M, Nakamura K, Tago K, Mashino T, Kasahara T. Anti-inflammatory activity of structurally related flavonoids, apigenin, luteolin and fisetin. Int Immunopharmacol. 2011;11:1050–1059. doi: 10.1016/j.intimp.2011.03.012. [DOI] [PubMed] [Google Scholar]
- Funakoshi-Tago M, Nakamura K, Tago K, Mashino T, Kasahara T. Anti-inflammatory activity of structurally related flavonoids, apigenin, luteolin and fisetin. Int Immunopharmacol. 2011;11:1150–1159. doi: 10.1016/j.intimp.2011.03.012. [DOI] [PubMed] [Google Scholar]
- Gradolatto A, Basly JP, Berges R, Teyssier C, Chagnon MC, Siess MH, Canivenc-Lavier MC. Pharmacokinetics and metabolism of apigenin in female and male rats after a single oral administration. Drug Metab Dispos. 2005;33(1):49–54. doi: 10.1124/dmd.104.000893. [DOI] [PubMed] [Google Scholar]
- Gupta S, Afaq F, Mukhtar H. Selective growth-inhibitory, cell-cycle deregulatory and apoptotic response of apigenin in normal versus human prostate carcinoma cells. Biochem Biophys Res Comm. 2001;287:914–920. doi: 10.1006/bbrc.2001.5672. [DOI] [PubMed] [Google Scholar]
- Jayasooriya RG, Kang SH, Kang CL, Hyun Y, Moon DO, Hyun JW, Chang WY, Kim GY. Apigenin decreases cell viability and telomerase activity in human leukemia cell lines. Food Chem Tox. 2012;50:2605–2611. doi: 10.1016/j.fct.2012.05.024. [DOI] [PubMed] [Google Scholar]
- Kang OH, Lee JH, Kwon D. Apigenin inhibits release of inflammatory mediators by blocking the NF-κB activation pathways in HMC-1 cells. Immunopharmacol Immunotoxicol. 2011;33:473–479. doi: 10.3109/08923973.2010.538851. [DOI] [PubMed] [Google Scholar]
- Kennedy BK, Berger SL, Brunet A, Campisi J, Cuervo AM, Epel ES, Franceschi C, Lithgow GJ, Morimoto RI, Pessin JE, Rando TA, Richardson A, Schadt EE, Wyss-Coray T, Sierra F. Geroscience: linking aging to chronic disease. Cell. 2014;159:709–713. doi: 10.1016/j.cell.2014.10.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim MH. Flavonoids inhibit VEGF/bFGF-induced angiogenesis in vitro by inhibiting the matrix-degrading proteases. J Cell Biochem. 2003;89:529–538. doi: 10.1002/jcb.10543. [DOI] [PubMed] [Google Scholar]
- Krtolica A, Parrinello S, Lockett S, Desprez P, Campisi J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc Natl Acad Sci U S A. 2001;98:12072–12077. doi: 10.1073/pnas.211053698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laberge RM, Awad P, Campisi J, Desprez PY. Epithelial-mesenchymal transition induced by senescent fibroblasts. Cancer Microenviron. 2012;5:39–44. doi: 10.1007/s12307-011-0069-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laberge RM, Sun Y, Orjalo AV, Patil CK, Freund A, Zhou L, Curran SC, Davalos AR, Wilson-Edell KA, Liu S, Limbad C, Demaria M, Li P, Hubbard GB, Ikeno Y, Javors M, Desprez PY, Benz CC, Kapahi P, Nelson PS, Campisi J. MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nature Cell Biol. 2015;17:1049–1061. doi: 10.1038/ncb3195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laberge RM, Zhou L, Sarantos MR, Rodier F, Freund A, de Keizer PL, Liu S, Demaria M, Cong YS, Kapahi P, Desprez PY, Hughes RE, Campisi J. Glucocorticoids suppress selected components of the senescence-associated secretory phenotype. Aging Cell. 2012;11:569–578. doi: 10.1111/j.1474-9726.2012.00818.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lasry A, Ben-Neriah Y. Senescence-associated inflammatory responses: aging and cancer perspectives. Trends Immunol. 2015;36:217–228. doi: 10.1016/j.it.2015.02.009. [DOI] [PubMed] [Google Scholar]
- Li RR, Pang LL, Du Q, Shi Y, Dai WJ, Yin KS. Apigenin inhibits allergen-induced airway inflammation and switches immune response in a murine model of asthma. Immunopharmacol Immunotoxicol. 2010;32:364–370. doi: 10.3109/08923970903420566. [DOI] [PubMed] [Google Scholar]
- Liang YC, Huang YT, Tsai SH, Lin-Shiau SY, Chen CF, Lin JK. Suppression of inducible cyclooxygenase and inducible nitric oxide synthase by apigenin and related flavonoids in mouse macrophages. Carcinogenesis. 1999;20:1945–1952. doi: 10.1093/carcin/20.10.1945. [DOI] [PubMed] [Google Scholar]
- Lim H, Park H, Kim HP. Effects of flavonoids on senescence-associated secretory phenotype formation from bleomycin-induced senescence in BJ fibroblasts. Biochem Pharmacol. 2015;96:337–348. doi: 10.1016/j.bcp.2015.06.013. [DOI] [PubMed] [Google Scholar]
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153:1194–1217. doi: 10.1016/j.cell.2013.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muñoz-Espín D, Serrano M. Cellular senescence: from physiology to pathology. Nature Rev Molec Cell Biol. 2014;15:482–496. doi: 10.1038/nrm3823. [DOI] [PubMed] [Google Scholar]
- Nicholas C, Batra S, Vargo MA, Voss OH, Gavrilin MA, Wewers MD, Guttridge DC, Grotewold E, Doseff AI. Apigenin blocks lipopolysaccharide-induced lethality in vivo and proinflammatory cytokines expression by inactivating NF-kappaB through the suppression of p65 phosphorylation. J Immunol. 2007;179:7121–7127. doi: 10.4049/jimmunol.179.10.7121. [DOI] [PubMed] [Google Scholar]
- Noh HJ, Sung E, Kim GJY, Lee TJ, Song IH. Suppression of phorbol-12-myristate-13-acetate-induced tumor cell invasion by apigenin via the inhibition of p38 mitogen-activated protein kinase-dependent matrix metalloproteinase-9 expression. Oncol Rep. 2010;24:277–283. doi: 10.3892/or_00000857. [DOI] [PubMed] [Google Scholar]
- Orjalo A, Bhaumik D, Gengler B, Scott GK, Campisi J. Cell surface IL-1α is an upstream regulator of the senescence-associated IL6/IL-8 cytokine network. Proc Natl Acad Sci U S A. 2009;106:17031–17036. doi: 10.1073/pnas.0905299106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osada M, Imaoka S, Funae Y. Apigenin suppresses the expression of VEGF, an important factor for angiogenesis, in endothelial cells via degradation of HIF-1α protein. FEBS Lett. 2004;575:59–63. doi: 10.1016/j.febslet.2004.08.036. [DOI] [PubMed] [Google Scholar]
- Reber L, Vermeulen L, Haegeman G, Frossard N. Ser276 phosphorylation of NF-kB p65 by MSK1 controls SCF expression in inflammation. PLoS One. 2009;4(2) doi: 10.1371/journal.pone.0004393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiners JJ, Clift R, Mathieu P. Suppression of cell cycle progression by flavonoids: dependence on the aryl hydrocarbon receptor. Carcinogenesis. 1999;20:1561–1566. doi: 10.1093/carcin/20.8.1561. [DOI] [PubMed] [Google Scholar]
- Rodier F, Coppé JP, Patil CK, Hoeijmakers WA, Muñoz DP, Raza SR, Freund A, Campeau E, Davalos AR, Campisi J. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nature Cell Biol. 2009;11:973–979. doi: 10.1038/ncb1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross JA, Kasum C. Dietary flavonoids: bioavailability, metabolic effects, and safety. Annu Rev Nutr. 2002;22:19–34. doi: 10.1146/annurev.nutr.22.111401.144957. [DOI] [PubMed] [Google Scholar]
- Ruiz PA, Haller D. Functional diversity of flavonoids in the inhibition of the proinflammatory NF-kappaB, IRF, and Akt signaling pathways in murine intestinal epithelial cells. J Nutr. 2006;136:664–671. doi: 10.1093/jn/136.3.664. [DOI] [PubMed] [Google Scholar]
- Shukla S, Gupta S. Apigenin-induced cell cycle arrest is mediated by modulation of MAPK, PI3K-Akt, and loss of cyclin D1 associated retinoblastoma dephosphorylation in human prostate cancer cells. Cell Cycle. 2007;6:1102–1114. doi: 10.4161/cc.6.9.4146. [DOI] [PubMed] [Google Scholar]
- Shukla S, Gupta S. Apigenin: a promising molecule for cancer prevention. Pharm Res. 2010;27:962–978. doi: 10.1007/s11095-010-0089-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Way TD, Kao MC, Lin JK. Apigenin induces apoptosis through proteasomal degradation of HER2/neu in HER2/neu-overexpressing breast cancer cells via the phosphatidylinositol 3-kinase/Akt-dependent pathway. J Biol Chem. 2004;279:4479–4489. doi: 10.1074/jbc.M305529200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Apigenin down-regulates the SASP in BJ fibroblasts. (A) Primary human BJ fibroblasts were induced to senesce (10 Gy X-irradiation) and immediately treated with increasing concentrations of apigenin for 10 days. CM were collected and analyzed for IL-6 secretion. (B) We investigated the levels of IL-8, GROA, IL-6 and IP10 secreted by treated (apigenin) and untreated (DMSO), NS or senescent BJ fibroblasts. Samples were in quadruplicate and levels of individual cytokines in senescent samples were normalized to the levels in DMSO-treated NS cells. (PDF 23 kb)
Apigenin reduces cytokine secretion of senescent IMR90 and BJ fibroblasts. (A) Primary IMR90 (PD 37) and (B) primary BJ (PD 34) human fibroblasts were induced to senesce by IR, and treated with apigenin or DMSO. CM were collected and the levels of 51 cytokines were analyzed by Luminex. Samples were in quadruplicate and levels of individual cytokines in senescent samples were normalized to the levels in DMSO-treated NS cells. (PDF 26 kb)
Effect of apigenin on the expression of major SASP factors in BJ fibroblasts. Using qPCR we determined the mRNA levels of major SASP factors in DMSO- and apigenin-treated non-senescent and senescent BJ fibroblasts (IL-6, IL-8, and GROA). IL-1A and IL-1B were also investigated at the mRNA level. (PDF 15 kb)
Apigenin reduces the senescence-induced inflammatory pathway activation. (A) IL-6 secretion of cells infected with a lentivirus NF-κB-luciferase reporter construct, induced to senesce by IR in the presence of apigenin, is shown. (B) NF-κB reporter activation in the senescent cells in (A) was measured by luminescence using the Promega Luciferase Assay System. (C) Induction of IL-6 secretion in NS and senescent HCA2 cells upon administration of three concentrations of recombinant IL-1A was measured by AlphaLISA. (PDF 18 kb)