Abstract
A 75-year-old man was diagnosed with pulmonary nontuberculous mycobacterial (NTM) infection in February 2005 and was treated with rifampicin, ethambutol, and clarithromycin. However, the infection was resistant to treatment, and his chest radiograph showed an abnormality that gradually seemed to aggravate. The patient’s sputum was positive for Mycobacteria. Moreover, the patient had dyspnea and an underlying chronic inflammation in the lungs. He visited our hospital because of dyspnea and leg edema in June 2011. Laboratory evaluation on admission revealed proteinuria (6 g/day) and decreased serum total protein (5.8 g/dL) and albumin (1.6 g/dL) levels, indicating nephrotic syndrome. Percutaneous renal biopsy revealed membranoproliferative glomerulonephritis (MPGN) in the acute stage and AA amyloidosis of mild degree. AA amyloidosis was also diagnosed histologically on gastric and colonic biopsy, in addition to renal biopsy. His renal function decreased gradually, and therefore, he underwent hemodialysis therapy in January 2012. However, his gastrointestinal-related symptoms persisted, and his appetite diminished, because of which he had become severely malnourished; he died 8 months later. This is a rare case of a patient with two different renal lesions (MPGN and AA amyloidosis) complicated with NTM. Our case suggests that MPGN and amyloidosis should be considered in elderly patients with nephrotic syndrome onset and chronic inflammation.
Keywords: Nontuberculous mycobacterial infection, Membranoproliferative glomerulonephritis, Amyloidosis
Introduction
Patients with nontuberculous mycobacterial (NTM) infection usually have longer periods of clinical illness prior to diagnosis and respond less well to therapy than patients with infectious tuberculosis; this is because such patients need longer treatment courses and have lower success rates with treatment [1, 2]. The number of patients with NTM has been gradually increasing [3].
Among elderly patients with nephrotic syndrome, 4.4 % have membranoproliferative glomerulonephritis (MPGN) [4]. MPGN is the result of an immune-complex-mediated infection or an autoimmune disease and is characterized by glomerulonephritis, with light microscopic findings showing thickening of the glomerular capillary wall and lesions with a lobular appearance [5, 6]. Furthermore, 7.6 % of elderly patients with nephrotic syndrome have concomitant renal amyloidosis [4]. AA amyloidosis is a complicated disease characterized by chronic systemic inflammation and infection, in which the deposition of amyloid protein causes various systemic organ failures; patients with this condition have poor prognosis. We report a rare case in which a patient had MPGN and AA amyloidosis, as well as NTM.
Case report
A 75-year-old man was diagnosed with NTM in February 2005, and since then, he had been treated with rifampicin 450 mg/day, ethambutol 750 mg/day, and clarithromycin 800 mg/day in Nagasaki, Japan. However, the infection was resistant to treatment, and the chest radiograph indicated an abnormality that gradually seemed to aggravate. The smear of the patient’s sputum was positive for Mycobacteria. The patient had dyspnea and seemed to have an underlying chronic inflammation. He visited our hospital because of dyspnea and leg edema in June 2011. Laboratory evaluation revealed proteinuria (6 g/day) and decreased serum total protein (5.8 g/dL) and albumin (1.6 g/dL) levels. He was diagnosed to have nephrotic syndrome; in July 2011, he was admitted to our hospital owing to worsening edema.
The patient’s height was 165.7 cm; weight, 47.6 kg; body temperature, 37.4 °C; blood pressure, 113/77 mmHg; and pulse, 85 beats/min on admission. There was no evidence of anemia in the palpebral conjunctiva or of jaundice in the bulbar conjunctiva. There was no swelling of the lymph nodes of the neck. Respiratory sounds were diminished in the upper right lung field. A heart murmur was not noted. The abdomen was soft and flat without any tenderness. Edema was present in both legs. Blood and serum laboratory data are shown in Table 1. He was diagnosed to have nephrotic syndrome. In addition, serum creatinine (Cr) level was elevated to 1.4 mg/dL and C-reactive protein level to 6.5 mg/dL. The sputum examination showed a positive smear, with a Gaffky score of 4, and the sputum was Mycobacterium intracellulare positive on polymerase chain reaction. A chest radiograph and a computed tomography scan showed infiltration into the lungs of both sides (Fig. 1). An abdominal computed tomography scan showed no kidney atrophy.
Table 1.
Laboratory data on admission
| Complete blood count | Blood chemistry | Serological test | |||
|---|---|---|---|---|---|
| WBC | 7940/μL | Na | 143 mEq/L | C3c | 78.1 mg/dL |
| RBC | 324 × 104/μL | K | 4.3 mEq/L | C4 | 20.0 mg/dL |
| Hb | 10.3 g/dL | CI | 103 mEq/L | IgG | 2040 mg/dL |
| Hct | 31.8 % | Ca | 8.1 mg/dL | IgA | 470 mg/dL |
| PLT | 21.9 × 104/μL | BUN | 24 mg/dL | IgM | 50.3 mg/dL |
| Urinary test | Cr | 1.4 mg/dL | ANA | <20 | |
| pH | 6.5 | eGFR | 42.2 mL/min | MPO-ANCA | <1.3 U/mL |
| Protein | (3+) | UA | 6.3 mg/dL | PR3-ANCA | <3.5 U/mL |
| 6 g/day | TP | 5.8 g/dL | Anti-GBM Ab | <10 EU | |
| OB | (3+) | Alb | 1.6 g/dL | Cryoglobulin | (−) |
| RBC | 50 ~ 99/HPF | T-Chol | 142 mg/dL | Infection | |
| NAG | 59.1 IU/L | AST | 14 IU/L | HBs-Ag | (−) |
| ABG (room air) | ALT | 9 IU/L | HCV-Ab | (−) | |
| pH | 7.44 | ALP | 181 IU/L | β-d-glucan | 8.2 pg/mL |
| PCO2 | 42 mmHg | γ-GTP | 37 IU/L | Mycobacterium | Gaffky4 |
| PO2 | 96 mmHg | LDH | 215 IU/L | Smear | |
| HC03 | 28 mEq/L | CK | 51 IU/L | NTM-PCR | |
| ABE | 4.6 mEq/L | Glu | 129 mg/dL | M. avium | (−) |
| AaDO2 | 1.5 torr | CRP | 6.5 mg/dL | M. intracellulare | (+) |
Fig. 1.
Chest X-ray and CT findings showing infiltration into the lungs of both sides
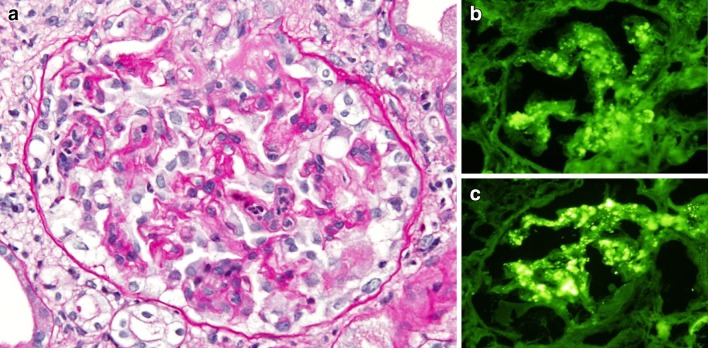
Renal biopsy was performed to determine the cause of nephrotic syndrome. The light microscopic section contained 20 glomeruli, and showed mesangial and endocapillary proliferation with occasional lobular structures. Three glomeruli had cellular crescents. The glomerular basement membrane was focally thickened, forming features of double contour. Immunofluorescence microscopy showed glomerular deposition of IgG, IgA, C3, and fibrinogen in the mesangium and partly in the capillary wall (Fig. 2). Electron microscopy showed electron dense deposits in the subepithelial, subendothelial, and mesangial areas, associated with occasional mesangial interposition (Fig. 3). Reviewing all the findings of light and electron microscopy and immunofluorescence, the patient was diagnosed as MPGN. There was progression of renal dysfunction (Cr 2.2 mg/dL); therefore, he was treated with a corticosteroid (prednisolone 30 mg/day), in addition to NTM treatment. The Cr levels and proteinuria improved to 1.2 mg/dL and 2 g/day, respectively.
Fig. 2.
a Light microscopy showing lobulation of glomerular tufts, endocapillary proliferative change, double contour of glomerular basement membrane, increase in mesangial cells and mesangial matrix (PAS staining, ×200). b, c Immunofluorescence microscopy showing deposition of IgG (b) and C3 (c) in mesangium and partly capillary wall
Fig. 3.
a Electron microscopy (×3000) showing mesangial interposition and deposits in the subepithelial, subendothelial, and mesangial area. b Electron microscopy (×25000) showing amyloid fibrils
As fibrillar structures were also observed in a small area of a small blood vessel on electron microscopy, we suspected amyloidosis (Fig. 3). Re-examination of the renal biopsy using Congo-red staining and direct fast scarlet staining (Fig. 4) was performed. Amyloid deposition was observed sporadically in some of the blood vessels and in the small segments of only four glomeruli among the 20 glomeruli observed. The amyloid deposits disappeared when the tissue was treated with potassium permanganate (Fig. 4), indicating a diagnosis of secondary renal amyloidosis. The patient’s serum amyloid A (SAA) level was elevated at 69.3 μg/mL.
Fig. 4.
a, b, c Light microscopy showing the presence of amyloid deposits of glomeruli, gastric mucosa, and intestinal mucosa (congo-red staining, kidney ×200, intestinum ×100). d, e, f The presence of amyloid deposits disappear when treated with potassium permanganate (KMnO4) was disclosed. g Direct fast scarlet staining, kidney, low-power image
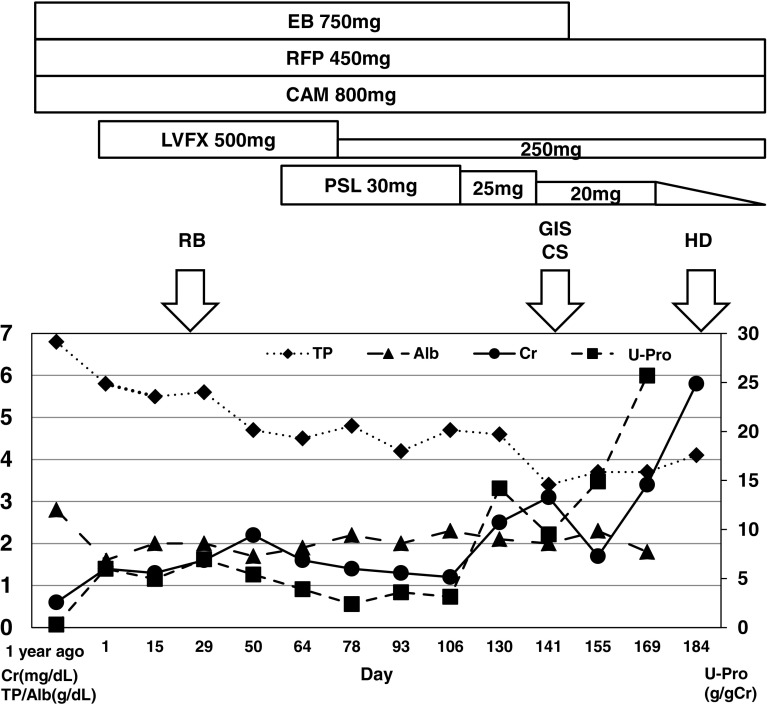
The patient then presented loss of appetite and diarrhea; therefore, upper and lower intestinal endoscopy were performed, and frequent erosions were noted. A biopsy that was performed revealed amyloid A protein deposits in the stomach, duodenum, terminal ileum, and rectum. Echocardiography did not show any findings suggestive of cardiac amyloidosis. Thereafter, the patient’s renal function decreased gradually, and he underwent hemodialysis in January 2012. Despite these, his gastrointestinal symptoms persisted, and his appetite diminished because of which he had become severely malnourished; he died 8 months later. The clinical course of the patient’s condition is shown in Fig. 5.
Fig. 5.
Clinical course
Discussion
Compared with M. tuberculosis, NTM is less virulent and is not transmitted by human-to-human contact. Among the various causative agents of NTMs, the Mycobacterium avium complex (MAC) is the most common cause of pulmonary disease worldwide. In Japan, the most common nontuberculous species causing human disease are the slowly growing MAC and M. kansasii species. MAC includes two species M. avium and M. intracellulare. These organisms are genetically similar and generally cannot be differentiated in a clinical microbiology laboratory. Chemotherapy regimens for MAC are based on triple therapy with rifampicin, ethambutol, and clarithromycin. If necessary, streptomycin or kanamycin may be added. NTM progresses slowly, and patients with NTM have relatively good prognosis. Patients with NTM disease usually have chronic clinical illness prior to diagnosis and respond less well to therapy than patients with tuberculosis; this is because patients with NTM need much longer treatment courses and have lower success rates with treatment [1, 2]. The average number of patients with MAC per year was reported to be 5.12 from 1993 to 2002, 10.98 from 2002 to 2006, and 15.87 from 2006 to 2009. The number of patients with NTM has been gradually increasing [3]. In most cases, treating patients with NTM is difficult, especially in cases of persistent and recurrent disease.
The Japan Renal Biopsy Registry, which was started in 2007, reported that, in Japan, a renal biopsy was performed in 36.3 % of the elderly population with nephrotic syndrome to investigate its cause. Particularly, 7.6 and 4.4 % of the elderly patients with nephrotic syndrome had renal amyloidosis and MPGN; the ratio is higher in the elderly than that in young people [4]. Therefore, it is necessary to consider the possibility of renal amyloidosis and MPGN in elderly patients with nephrotic syndrome, as reported in this case.
MPGN has traditionally been classified as MPGN types I, II, and III depending on the findings observed on electron microscopy. However, Sethi et al. [5, 6] recently proposed a new classification scheme of MPGN lesions: those with C3 and immunoglobulin on immunofluorescence called immune-complex-mediated MPGN versus those with C3 staining alone on immunofluorescence called complement-mediated MPGN. Immune-complex-mediated MPGN is characterized by the presence of immune complexes and complement components, while complement-mediated MPGN is characterized by the presence of complement components in the absence of immune complexes. Immune-complex-mediated MPGN occurs because of chronic antigenemia and/or circulating immune complexes and is observed in chronic infections, autoimmune diseases, and monoclonal gammopathies. An underlying cause can be found in most cases. In chronic infections, immune-complex-mediated MPGN is most commonly secondary to hepatitis C virus (HCV) infections in Japan. HCV-induced MPGN is typically associated with mixed cryoglobulinemia. In addition to HCV, chronic bacterial (e.g., mycobacterium endocarditis, shunt nephritis, and abscesses) and fungal infections can cause MPGN. In idiopathic MPGN, a causal etiology cannot be found, whereas in secondary cases, the associated systemic disease or infection is known [5, 6]. Idiopathic MPGN is almost limited to young people between 8 and 30 years of age, and secondary MPGN is common in the elderly [7–10]. In the present case, the onset was during old age; therefore, the possibility of secondary infection is high rather than that of idiopathic MPGN; further, there was an immune-complex-mediated MPGN as determined by the C3 and immunoglobulin deposits observed on immune-fluorescence microscopy. Although immune-complex-mediated MPGN associated with NTM is rare, in this case, the possibility of this association is high [11, 12]. Our patient received appropriate NTM treatment with rifampicin, ethambutol, and clarithromycin; however, due to the progression of renal dysfunction, corticosteroid therapy with prednisolone 30 mg/day was started. The effectiveness of steroid treatment for MPGN should be carefully examined. According to the guidelines for the treatment of nephrotic syndrome in Japan, idiopathic MPGN should be treated with steroids, but secondary MPGN should be treatment for the primary disease [10]. Despite NTM treatment, our patient developed nephrotic syndrome, and the possibility of idiopathic MPGN cannot be excluded. Furthermore, drug interactions result from the ability of rifampicin to induce the cytochrome P450 isoenzyme 3A4, potentially decreasing the serum concentration of prednisolone. Our patient was carefully managed by administering the effect of prednisolone 20 mg/day. We frequently assessed the patient’s general condition and chest radiographs. There was no progression of NTM as a side effect of corticosteroid therapy.
AA amyloidosis involves the deposition of amyloid protein in tissues. The amyloid fibrils are derived from N-terminal cleavage fragments of the hepatic acute-phase reactant SAA protein. SAA is an apolipoprotein composed of high-density lipoprotein, which is synthesized by hepatocytes under the transcriptional regulation of the proinflammatory cytokines IL-6 and TNF-alpha. SAA is a highly dynamic acute-phase response protein. Sustained high circulating levels of SAA are a prerequisite for the development of AA amyloidosis. AA amyloidosis is a multisystem disease and amyloid deposits can be found in almost all sites on biopsy. Patients almost always present with proteinuric renal dysfunction. More extensive disease can produce hepatosplenomegaly and gut dysfunction, usually causing diarrhea and weight loss. Because the deposition of amyloid in the kidney was localized and very mild, amyloidosis was not thought to be the cause of nephrotic syndrome at the time of biopsy. Around 60 % of AA amyloidosis cases are caused by rheumatoid arthritis (RA) [13, 14]; the other major conditions associated with AA amyloidosis are ankylosing spondylitis, psoriatic arthritis, chronic pyogenic infections (bronchiectasis, osteomyelitis, paraplegics, or tuberculosis), Crohn’s disease, Castleman’s disease, neoplasms (lymphoma, mesothelioma), and familial Mediterranean fever [15]. AA amyloidosis by NTM is very rare [16–18]. The time from the diagnosis of NTM to the diagnosis of AA amyloidosis is approximately 5–8 years in most cases. The time to death after being diagnosed with AA amyloidosis associated with NTM disease is 1 year or less. Decreased production of SAA was associated with stabilization or regression of amyloid deposits and prolonged survival [15]. However, patients with AA amyloidosis associated with refractory NTM, as in this case, are considered to have a poorer prognosis than those with AA amyloidosis associated with RA.
In the acute stage of MPGN, mesangial and endocapillary proliferation with occasional lobular structures and cellular crescents are observed. The glomerular basement membrane was focally thickened, forming features of double contour. In early and mild lesions, amyloid is deposited sporadically in some of the blood vessels and in the small segments of only four glomeruli among 20 glomeruli. Therefore, in the present case, the onset was considered to occur relatively simultaneously, and the patient was suggested to have different renal lesions, MPGN and amyloidosis, as well as NTM.
Thus, this was a rare case of immune-complex-mediated MPGN and AA amyloidosis complicated with NTM. Our case suggests that MPGN and amyloidosis should be considered in elderly patients with nephrotic syndrome onset and chronic inflammation.
Conflict of interest
The authors have declared that no conflict of interest exists.
References
- 1.Morimoto K, Iwai K, Ohmori M, et al. Nontuberculous mycobacteriosis mortality in Japan. Kekkaku. 2011;86:547–552. [PubMed] [Google Scholar]
- 2.The Nontuberculous Mycobacteriosis Control Committee of the Japanese Society for Tuberculosis Scientific Assembly for Infection and Tuberculosis of the Japanese Respiratory Society. Guidelines for chemotherapy of pulmonary nontuberculous mycobacterial disease—2012 revised version. Kekkaku. 2013;88:29–32. [PubMed] [Google Scholar]
- 3.Osoreda H, Kobayashi H. Chronological change of primary type pulmonary Mycobacterium avium-intracellulare complex infection. Kekkaku. 2010;85:447–452. [PubMed] [Google Scholar]
- 4.Yokoyama H, Sugiyama H, Sato H, et al. Committee for the Standardization of Renal Pathological Diagnosis and for Renal Biopsy and Disease Registry of the Japanese Society of Nephrology, and the Progressive Renal Disease Research of the Ministry of Health, Labour and Welfare of Japan. Clin Exp Nephrol. 2012;16:903–920. doi: 10.1007/s10157-012-0673-8. [DOI] [PubMed] [Google Scholar]
- 5.Bomback AS, Appel GB. Pathogenesis of the C3 glomerulopathies and reclassification of MPGN. Nat Rev Nephrol. 2012;8:634–642. doi: 10.1038/nrneph.2012.213. [DOI] [PubMed] [Google Scholar]
- 6.Sethi S, Nester CM, Smith RJ. Membranoproliferative glomerulonephritis and C3 glomerulopathy: resolving the confusion. Kidney Int. 2012;81:434–441. doi: 10.1038/ki.2011.399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.West CD. Childhood membranoproliferative glomerulonephritis: an approach to management. Kidney Int. 1986;29:1077–1093. doi: 10.1038/ki.1986.110. [DOI] [PubMed] [Google Scholar]
- 8.Cameron JS, Turner DR, Heaton J, Williams DG, et al. Idiopathic mesangiocapillary glomerulonephritis. Comparison of types I and II in children and adults and long-term prognosis. Am J Med. 1983;74:175–192. doi: 10.1016/0002-9343(83)90606-X. [DOI] [PubMed] [Google Scholar]
- 9.Rennke HG. Secondary membranoproliferative glomerulonephritis. Kidney Int. 1995;47:643–656. doi: 10.1038/ki.1995.82. [DOI] [PubMed] [Google Scholar]
- 10.Matsuo S, Imai E, et al. Guidelines for the treatment of nephrotic syndrome. Nihon Jinzo Gakkai Shi. 2011;53:78–122. [PubMed] [Google Scholar]
- 11.Pecchini F, Bufano G, Ghiringhelli P. Membranoproliferative glomerulonephritis secondary to tuberculosis. Clin Nephrol. 1997;47:63–64. [PubMed] [Google Scholar]
- 12.Meyrier A, Valensi P, Sebaoun J. Mesangio-capillary glomerulonephritis and the nephrotic syndrome in the course of disseminated tuberculosis. Nephron. 1988;49:341–342. doi: 10.1159/000185091. [DOI] [PubMed] [Google Scholar]
- 13.Toyoshima H, Kusaba T, Yamaguchi M. Cause of death in autopsied RA patients. Ryumachi. 1993;33:209–214. [PubMed] [Google Scholar]
- 14.Gertz MA, Kyle RA. Secondary systemic amyloidosis: response and survival in 64 patients. Medicine (Baltimore) 1991;70:246–256. doi: 10.1097/00005792-199107000-00002. [DOI] [PubMed] [Google Scholar]
- 15.Lachmann HJ, Goodman HJ, Gilbertson JA, et al. Natural history and outcome in systemic AA amyloidosis. N Engl J Med. 2007;356:2361–2371. doi: 10.1056/NEJMoa070265. [DOI] [PubMed] [Google Scholar]
- 16.Zaizen Yukihiro, Arita Kazuhiro, Kawano Hiroshi, et al. Case report of AA amyloidosis associated with cystic infection during the course of non-tuberculous mycobacteriosis. Kokyu Junkan. 2007;55:237–241. [Google Scholar]
- 17.Shinozuka N, Kasamatsu N, Seto T, et al. A fatal case of pulmonary non-tuberculous mycobacteriosis with reactive AA amyloidosis. Nihon Kokyuki Gakkai Zasshi. 2007;45:636–642. [PubMed] [Google Scholar]
- 18.Haga Takahiro, Kasamatsu Norio, Kobayashi Takeshi, et al. A case of pulmonary nontuberculous mycobacteriosis with reactive AA amyloidosis exacerbated in spite of various antimicrobial therapy. Jpn J Chest Dis. 2009;68:245–251. [Google Scholar]