Abstract
Integrated positron emission tomography/computed tomography (PET/CT) with 2-[18F]fluoro-2-deoxy-D-glucose (18F-FDG) has emerged as a powerful tool for the combined metabolic and anatomic evaluation of many cancers. In urological oncology, however, the use of 18F-FDG has been limited by a generally low tumor uptake, and physiological excretion of FDG through the urinary system. 18F-FDG PET/CT is useful when applied to specific indications in selected patients with urological malignancy. New radiotracers and positron emission tomography/magnetic resonance imaging (PET/MRI) are expected to further improve the performance of PET in uro-oncology.
Keywords: Positron emission tomography/computed tomography (PET/CT), Positron emission tomography/ magnetic resonance imaging (PET/MRI), Urological cancer, Kidney cancer, Bladder cancer, Prostate cancer, Testicular cancer
Introduction
In the late 1990s, positron emission tomography (PET) with 2-[18F]fluoro-2-deoxy-D-glucose (18F-FDG), which exploits the increased utilization and high uptake of glucose by malignant cells, opened a new field in clinical oncologic imaging. The radiopharmaceutical most commonly used for PET in oncology is 18F-FDG, an analog of glucose that is preferentially taken up and trapped inside malignant cells. For more than a decade, integrated positron emission tomography/computed tomography (PET/CT), in which a full-ring-detector clinical PET scanner and multidetector-row helical CT scanner are combined, has made it possible to acquire both metabolic and anatomic imaging data using a single device in a single diagnostic session, and provides precise anatomic localization of suspicious areas of increased 18F-FDG uptake. When used in a clinical setting, 18F-FDG PET/CT leads to a significant improvement in diagnostic accuracy and has had a considerable impact on patient management, including diagnosis, initial staging, optimization of treatment, restaging, monitoring of the response to therapy, and prognostication of many malignant tumors. In urological oncology, however, the use of 18F-FDG has been limited by a generally low tumor uptake, and physiological excretion of 18F-FDG through the urinary system masking 18F-FDG uptake by the primary urological carcinoma. Several new PET tracers are currently under investigation for potential use in the staging and monitoring of response to therapy in urological malignancies. We herein review the current and future roles of PET/CT in the management of urological cancer (kidney, bladder, urinary tract, prostate, adrenal gland, and testicular cancer), discussing its usefulness and limitations for imaging in these patients.
Kidney cancer
Contrast-enhanced CT provides accurate information for detection and local staging of renal cell carcinoma (RCC). Because extrarenal metastasis is detected more easily than renal lesions by 18F-FDG PET/CT [1], this technique is useful for staging (Fig. 1) [2, 3] and restaging [4–8] of advanced RCC and can facilitate treatment optimization [3, 5, 6, 8].
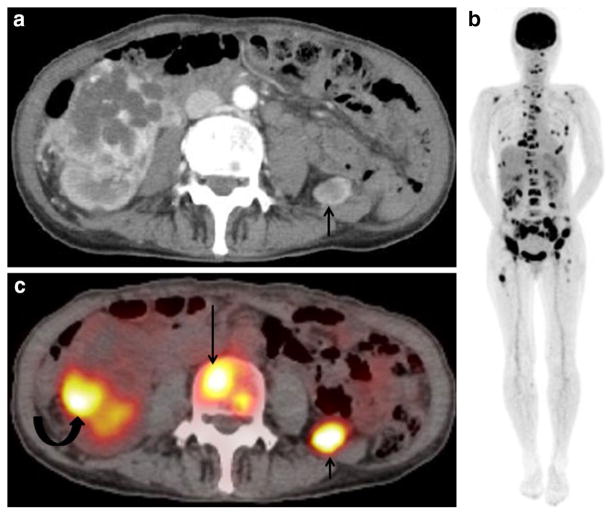
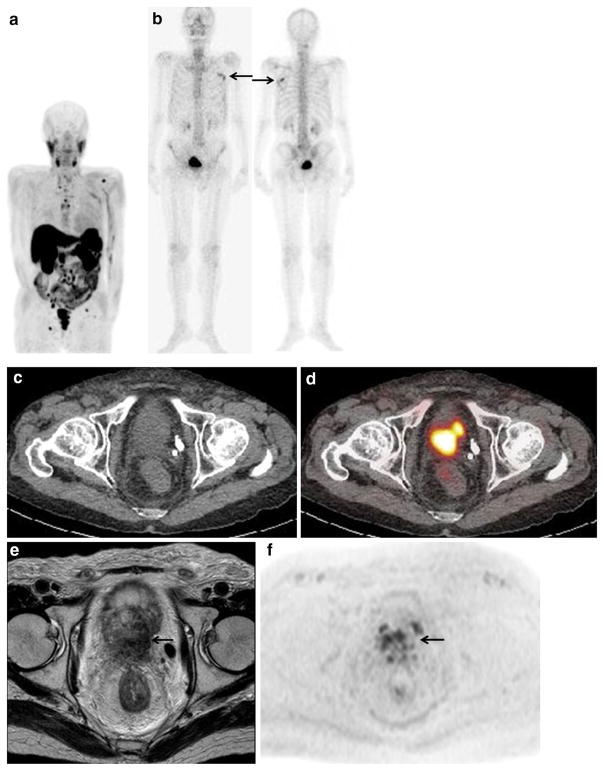
Fig. 1.
A 62-year-old woman with multiple bone metastases and a retroperitoneal metastasis arising from renal cell carcinoma. a Contrast-enhanced CT shows a 5-cm mass with cystic degeneration at the right kidney and a well-enhanced 1.5-cm mass at the left retroperitoneum (arrow), suggesting cystic renal cell cancer and retroperitoneal metastasis. b Maximum intensity projection (MIP) of FDG-PET shows abnormal FDG uptake in multiple skeletal bones. c FDG-PET/ CT at the same level as (a) shows moderate FDG uptake corresponding to the peripheral solid portion of right renal tumor (SUVmax: 5.5, curved arrow), a retroperitoneal mass (SUVmax: 8.2, short arrow), and L4 spine vertebral body (SUVmax: 5.6, long arrow), suggesting renal cell carcinoma with metastasis to bone and retroperitoneum. Because the histopathological specimen of needle biopsy at the renal tumor and iliac bony lesion revealed clear cell renal cell carcinoma and bony metastasis, the patient underwent tyrosine kinase inhibitors
In a meta-analysis of 14 studies including 517 patients, Wang et al. showed that the pooled sensitivity of 18F-FDG PET or PET/CT was 79 % for intrarenal tumors and 84 % for extrarenal metastases, and that 18F-FDG PET/ CT increased the pooled sensitivity to 91 % [1]. Alongi et al. evaluated the clinical impact of 18F-FDG PET/CT for restaging 104 patients after surgery and demonstrated that (1) the sensitivity and specificity were 74 % and 80 %, respectively, (2) 18F-FDG PET/CT findings influenced therapeutic management in 45/104 cases (43 %), and (3) in analysis of overall survival (OS), positive versus negative 18F-FDG-PET/CT findings were associated with poorer cumulative survival rates over a 5-year period (19 vs 69 %, respectively; p < 0.05) [8].
In recent years, it has been reported that 18F-FDG PET/ CT findings may correlate well with pathological prognostic parameters [2, 9–12] and be useful for prognostication [12–18] and monitoring of the response to therapy [14–17, 19, 20].
Several groups have analyzed the relationship between clinicopathological features and 18F-FDG uptake in patients with RCC who underwent nephrectomy and pre-operative 18F-FDG PET/CT and showed that FDG uptake was higher in RCC patients with a higher Furman grade, venous (V) and lymphatic (L) invasion, and a higher TNM stage [2, 9–12]. In a study involving 77 patients with RCC, Mizuno et al. demonstrated that a higher maximum standardized uptake value (SUVmax) on 18F-FDG PET was associated with elevated tumor levels of phosphorylated-Akt, phosphorylated-S6 protein, aggressive behavior and metastatic potential, early relapse, and shorter OS after radical nephrectomy. Their Kaplan–Meier survival analysis indicated that patients with a high SUVmax (≥4.35) had a significantly lower OS rate than those with a low SUV-max (<4.35) irrespective of the presence of distant metastasis before surgery, and among 52 patients without distant metastasis before surgery, those with a high SUVmax (≥3.50) had a significantly lower recurrence-free survival rate than those with a low SUVmax (<3.50) [12]. In another study by Kayani et al., multivariate analysis demonstrated that a high SUVmax (≥7.1) and an increased number of PET-positive lesions (8 or more) demonstrated by 18F-FDG PET/CT before treatment were significantly correlated with shorter OS [hazard ratio (HR): 3.30 and 3.67, respectively] in 44 patients with metastatic clear cell RCC who were treated with sunitinib [14].
Two major groups of targeted drugs currently approved for use against metastatic RCC are multikinase inhibitors and mammalian target of rapamycin (mTOR) inhibitors. Sorafenib and sunitinib are two representatives of the former, inhibiting tyrosine kinase vascular endothelial growth factor (VEGF) receptor 2 and platelet-derived growth factor receptor β in endothelial cells and pericytes, respectively [21]. Because expression of Glut is a downstream result of HIF transcriptional activity, it is conceivable that the intensity of 18F-FDG uptake on PET may reflect the activity of the entire pathway. This means that the variable intensity of 18F-FDG uptake on PET by clear cell RCC may reflect the variable strength of the HIF signaling pathway and expression of its downstream products, thus being predictive of the effects of inhibitors of this pathway.
A recent study by Ueno et al. involving 35 patients with advanced RCC evaluated the response to tyrosine kinase inhibitors (TKI) (sunitinib 19 cases, sorafenib 16 cases) in terms of tumor size and 18F-FDG uptake using 18F-FDG PET/CT before and 1 month after treatment [16]. They showed that PET was able to predict not only the duration of response to TKIs, but also survival duration (OS and progression-free survival [PFS]), and that early assessment by 18F-FDG PET/CT provided useful information for determining individual patient management strategies [16]. In another study, Faenebo et al. [20] determined whether early changes in the glucose metabolism of metastatic RCC assessed by 18F-FDG PET according to the PERCIST 1.0 criteria [22] after 14 and 28 days of treatment with TKIs (sunitinib 18 cases, sorafenib 19 cases, or pazopanib 2 cases) were able to predict OS and PFS in 39 patients. They found that early changes in SUVpeak (peak standardized uptake normalized to lean body mass) and total lesion glycolysis after only 14 days of TKI treatment were significantly correlated with both PFS and OS [20].
Several new PET tracers are currently under investigation for potential use in the staging and monitoring of response to therapy in patients with RCC. These new tracers exploit various cellular process that are altered in malignant cells, including cellular proliferation [18F-fluoro-thymidine (18F-FLT)], aerobic metabolism (11C-acetate), cell membrane synthesis (11C-choline, 18F-fluorocholine), hypoxia [18F-fluoromisonidazole (18F-FMISO)], and amino acid transport (11C-methionine, anti-[18F]fluorocyclobutane carboxylic acid (anti-3-18F-FACBC)) (Table 1).
Table 1.
PET tracer in urological oncology
| Biological analog | Target process | Measured effect | |
|---|---|---|---|
| 18F-FDG | Glucose | Glucose transporters and hexokinases | Aerobic and anaerobic glycolysis, glucose consumption |
| 11C-choline | Choline | Choline kinase | Cell membrane metabolism, tumor proliferation |
| 18F-choline | Choline | Choline kinase | Cell membrane metabolism, tumor proliferation |
| 11C-acetate | Acetate | Tricarboxylic acid cycle and fatty acid synthase | Lipid synthesis |
| 18F-FDHT | Testosterone | Androgen receptor | Measures androgen receptor |
| 18F-NaF | Fluoride | Hydroxyl and bicarbonate ions of bone hydroxyapatite | Measures bone status |
| 18F-FMISO | NA | Measures hypoxia | Tumor hypoxia |
| 18F-FLT | NA | Thymidine kinase | Nucleic acid synthesis, tumor proliferation |
| 18F-FACBC | Amino-fluorocyclobutane-carboxylic acid | Neutral A–A type amino acid uptake and protein synthesis | Protein synthesis |
| 68Ga-PSMA | Prostate specific membrane antigen | Prostate cell surface protein | Tumor aggressiveness, androgen independence |
NA not applicable
Hugonnet et al. used 18F-FMISO PET/CT in 53 patients with metastatic RCC at the baseline and 1 month after sunitinib treatment and demonstrated that patients with initially hypoxic targets had shorter PFS than the others, and that target lesions showed decreased 18F-FMISO uptake during 1 month after sunitinib treatment, suggesting that sunitinib decreased the intensity of tumor hypoxia [23]. Horn et al. compared 18F-FLT and 18F-FDG for early measurement of response to sunitinib treatment in 20 patients with metastatic RCC and demonstrated that while FLT-PET could be used to identify response as early as 1 week after the start of treatment, 18F-FDG PET was more effective at a later time point of 3–4 weeks, suggesting that inhibition of VEGF signaling with sunitinib exerts an early effect on tumor proliferation, which is then followed by a reduction in tumor metabolism [24]. Two groups have reported the utility of 18F-fluoroethylcholine PET [25] and 11C-acetate PET [26] for staging and monitoring of the response of advanced RCC to therapy. Furthermore, one preliminary report has demonstrated that anti-3-18F-FACBC PET showed better conspicuity for papillary RCC than for clear cell RCC [27].
Bladder cancer
Bladder cancer is a heterogeneous disease, 70 % of patients presenting with superficial tumors that tend to recur but are generally not life-threatening, and 30 % presenting with muscle-invasive disease associated with a high risk of death from distant metastases [28]. The standard method of diagnosing bladder cancer continues to be based on direct visualization of the bladder by cystoscopy including biopsy/ resection with histological examination of the tissue. Magnetic resonance imaging (MRI) is an accurate technique for local staging of bladder cancer due to its superior spatial and contrast resolution.
18F-FDG PET has not been widely used to detect and locally stage bladder cancer because physiological 18F-FDG activity excreted through the urinary system interferes with visualization of the primary bladder cancer and locoregional lymph nodes (LNs). A meta-analysis showed that the sensitivity and specificity of 18F-FDG PET/CT for detecting bladder cancer was 80 % (95 % CI 71–87 %) and 84 % (95 % CI 69–93 %), respectively [29]. Various mechanisms for improving the diagnostic capabilities of 18F-FDG PET have been investigated, including adequate hydration, delayed pelvic imaging after oral hydration, and forced diuresis using furosemide with a sensitivity of 85–96 % [30, 31].
18F-FDG PET/CT is useful for staging and restaging (detection of metastatic lesions) of bladder cancer (Fig. 2); a systematic review and meta-analysis of 6 studies involving 236 patients showed that the pooled sensitivity and specificity for staging and restaging (metastatic lesions) were 0.82 (95 % CI: 0.72–0.89) and 0.89 (95 % CI: 0.81–0.95), respectively [32].
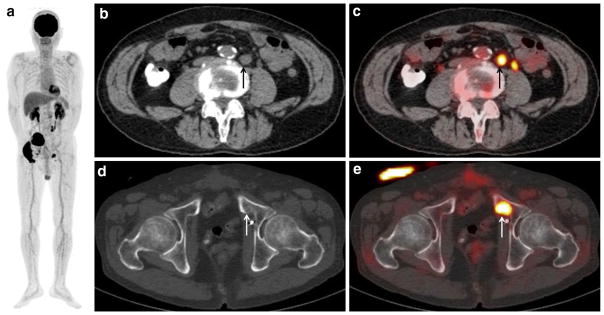
Fig. 2.
A 66-year-old man with bladder cancer (cT3N1) after neoad-juvant chemotherapy and radical cystectomy, who developed lymph node and bony metastases. a MIP of FDG-PET shows two areas of abnormal FDG uptake in the pelvis. b CT of FDG-PET/CT and c PET/CT show moderate FDG uptake (SUVmax: 7.2) corresponding to a 1.5-cm swollen para-aortic lymph node (arrow), suggesting lymph nodal recurrence. A tiny FDG uptake which is located to the left of that nodal recurrent lesion is physiologic excretion in the left ureter. d CT of FDG-PET/CT and e PET/CT show intense FDG uptake (SUVmax: 13.0) corresponding to the mild sclerotic change of the left pubic bone (arrow), suggesting bone metastasis. The detection of this bony metastasis by only (d) CT is difficult
In recent years, several authors have demonstrated that 18F-FDG PET/CT can be used successfully for treatment optimization [33–35], restaging [36], evaluation of metastatic disease response [37], early assessment of treatment response [38], and prognostication [39] of bladder cancer. Mertens et al. [34] demonstrated that 18F-FDG PET/CT provided important additional staging information, which influenced the treatment of muscle-invasive bladder cancer, in almost 20 % of 96 cases in comparison with contrast-enhanced CT. Giannatempo et al. [38] demonstrated that, in comparison with early CT, the response after 2 cycles of first-line chemotherapy detected by 18F-FDG PET was associated with longer PFS and OS in 31 patients with advanced transitional cell carcinoma. Mertens et al. [39] demonstrated that the presence of extravesical 18F-FDG-avid lesions on PET/CT was an independent indicator of mortality [OS and disease free survival (DFS)] in 211 patients with muscle-invasive bladder cancer.
There have been several reports of new PET tracers for imaging of bladder cancer. Although 11C-actate PET/CT may be useful for staging of bladder cancer, assessment of the response to neoadjuvant chemotherapy, especially intravesical instillation of bacillus Calmette-Guérin (BCG), is difficult using this modality because of false positive uptake by areas of inflammation or granulomatous infection [40, 41]. Although 11C-choline PET/CT is useful for restaging of bladder cancer in cases of suspected relapse, especially for evaluation of LN or distant metastases [42], its efficacy for preoperative LN staging [43, 44] and prognostication [45] is clinically limited.
Pelvic and ureter cancer
Local evaluation of the primary tumor on 18F-FDG PET is often limited by the obscuring effect of excreted FDG, particularly if the tumor is small and is confined to the mucosa or wall, but assessment of metabolic activity may still be possible through close correlation with CT images [46]. 18F-FDG PET/CT may also be helpful in the detection of primary tumor [47], nodal or distant metastasis [48], the assessment of recurrent disease [49], and the early assessment of treatment response [38]. Tanaka et al. [48] demonstrated that the clinicians changed their assessments of disease extent and management plans in 32 and 20 % of patients, respectively, based on the 18F-FDG PET/CT results after diagnostic CT for initial staging (n = 47) and for restaging (n = 9).
Prostate cancer
Various PET tracers have been used for PET/CT imaging of prostate cancer.
Fdg
It is well known that the ability of 18F-FDG PET to detect cancer is based on increased expression of cellular membrane glucose transporter 1 (GLUT1) and enhanced hexokinase II enzyme activity within tumor cells. Preclinical studies have examined the expression of GLUT1 in prostate cancer cell lines and found that it is higher in those that are poorly differentiated in comparison with well-differentiated hormone-sensitive cell lines [50]. This suggests that GLUT1 transporter expression increases with increasing disease grade, and may explain the higher 18F-FDG accumulation in castration-resistant than in castration-sensitive tumors, and also the modulatory effect of androgen on the glucose metabolism of castration-sensitive tumors.
18F-FDG PET/CT is of limited value for detection and localization of primary prostate cancer and initial staging of disease, as most primary tumors are slow-growing, well differentiated, multiple, and small, with tumor uptake levels that can overlap with those of normal tissue, prostatitis, or benign prostatic hyperplasia (BPH). 18F-FDG PET/ CT may be most useful for detection of aggressive disease, evaluation of tumor extent (Fig. 3), response of metastatic disease to treatment, and prognostication in patients with castration-resistant tumors [51, 52]. Jadvar demonstrated that tumor 18F-FDG uptake decreased with successful chemohormonal treatment, although imaging findings were sometimes discordant with those of other disease manifestations including changes in the level of serum prostate specific antigen (PSA) or the number of circulating tumor cells [51]. Clearly, additional studies are needed in order to determine the optimal combination of relevant data that can most accurately reflect the effect of various current and novel therapies. Among SUVmax of the most active lesion, the sum of SUVmax, and the average SUVmax for all metabolically active lesions after subtraction of patient-specific background-liver average SUV, Jadvar et al. demonstrated that the sum of SUVmax derived from baseline 18F-FDG PET/CT yielded independent prognostic information on OS in 87 men with castration-resistant metastatic prostate cancer, and suggested that this information might be useful for assessing the comparative effectiveness of various conventional and emerging treatment strategies [52].
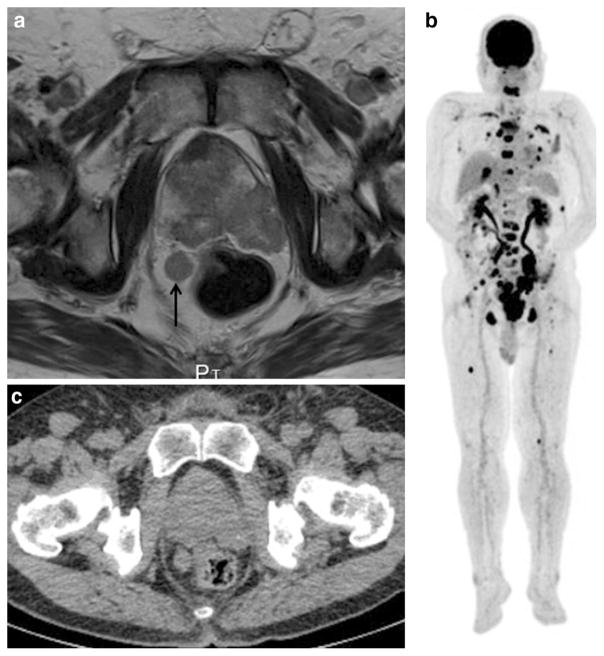
Fig. 3.
A 67-year-old man with multiple bony, lymph node, hepatic, and lung metastases from prostate cancer. a T2-weighted MRI shows a slight hypointense mass occupying the prostate, extending through the prostate capsule and invading the left seminal vesicle, consistent with advanced prostate cancer. One 14 × 16 mm swollen lymph node is also seen near the rectum (arrow), suggesting the presence of nodal cancer spread. b MIP of FDG-PET shows multiple areas of abnormal FDG uptake in the whole body. c CT of FDG-PET/CT and d PET/ CT show intense FDG uptake corresponding to the prostate (SUV-max: 13.3), a para-rectal lymph node swelling, the left pubic and ischial bones, suggesting prostate cancer with lymph node and bone metastases. However, accurate evaluation of primary local staging by FDG-PET/CT is difficult. The detection of these bony metastases by only (c) CT is difficult. e CT of FDG-PET/CT and f PET/CT show intense FDG uptake corresponding to the spine and sternum, consistent with multiple bone metastases. The detection of these multiple bony metastases by only (e) CT is difficult
Any accidentally detected increase of 18F-FDG uptake in the prostate gland should probably be evaluated further to rule out prostate cancer. In a study of 20,422 patients, Bertagna et al. identified prostate incidental uptake (PIU) in 280 patients (1.4 %), among whom 63 (22.5 %) underwent PSA determination and biopsy to clarify the nature of the incidental uptake. They found that in 35 (55.5 %) of these 63 patients, the PIU was malignant, whereas it was benign in the remainder [53]. A systematic review and meta-analysis reported a pooled prevalence of PIU of 1.8 % (95 % CI 1.3–2.3 %) and a pooled risk of malignancy among patients further evaluated or verified by biopsy of 17 % (95 % CI 12–23 %) and 62 % (95 % CI 54–71 %), respectively [54]. Although the prevalence of PIU is not high, patients with elevated serum levels of PSA have a high incidence of prostate cancer [55, 56]. Therefore, patients with PIU should undergo further evaluation, including measurement of the serum PSA concentration, and those found to have a high serum PSA level should undergo prostate MRI [57] or biopsy [56].
Currently, 99mTc-methylene disphosphonate bone scintigraphy is the standard method for detection of bone metastases in patients with prostate cancer. Although 18F-FDG PET is less sensitive than conventional bone scintigraphy for identification of osteoblastic bony metastases, 18F-FDG PET may be able to distinguish between metabolically active lesions from non-viable bony lesions [58].
Choline
11C-choline and 18F-fluorocholine are well established tracers routinely employed at some PET centers for imaging of prostate cancer in Western countries.
Focal choline uptake in the prostate could make one suspect prostate cancer, but non-malignant causes, such as high-grade prostatic intraepithelial neoplasia, prostatitis, BPH and normal tissue, can also be sources of false-positive focal activity. Although a few groups have shown that increased choline uptake in primary prostate cancer is correlated with histological surrogate markers of aggressiveness, such as the Gleason score and MIB-1/Ki-67 labeling index [59], many groups have failed to find a significant correlation between choline uptake and the serum PSA level, Gleason score or tumor grade [60, 61]. Multiparametric MRI, a combination of high-resolution T2-weighted imaging and functional MR techniques such as dynamic contrast-enhanced imaging and diffusion-weighted imaging, remains the gold standard imaging technique for detection and local staging of untreated prostate cancer, showing features such as capsular and seminal vesicle invasion. Although choline PET/CT has limitations for detection of primary prostate cancer, it might be useful for a minority of newly diagnosed patients in whom distant metastatic disease is highly suspected on the basis of clinical data (e.g., serum PSA level >20 ng/ml, Gleason score 8–10, locally advanced tumor evident by palpation and/or MRI) [62].
Although choline PET/CT shows better performance than conventional CT and MR imaging for detection of locoregional LN metastasis, it does not reach an optimal detection rate in comparison with lymphadenectomy, and therefore cannot replace this procedure. In the largest pre-operative series (n = 210) of intermediate- and high-risk patients who had undergone radical prostatectomy (RP) with surgical LN dissection, Poulsen et al. reported that the patient-based sensitivity and specificity for detection of pelvic LN metastases were 73 and 88 %, with a node-based sensitivity and specificity of 56 and 94 %, respectively [63].
Choline PET/CT is a useful modality for detection of bone metastasis. In a patient-based analysis of patients with PSA progression after primary treatment, Picchio et al. directly compared 11C-choline PET/CT and bone scintigraphy for detection of bone metastasis and showed that the former had better sensitivity and specificity [64]. In patients with biochemical failure, imaging plays a critical role in distinguishing between local recurrence and distant spread of disease (mostly bone and LN) when formulating appropriate treatment strategies. Choline PET/CT is a powerful tool for restaging of biochemically recurrent prostate cancer, particularly when the PSA level becomes significantly elevated. Many reports have discussed the usefulness of choline PET/CT for detecting sites of recurrence in patients with PSA failure (Fig. 4), and choline PET/CT is employed routinely for this purpose at a number of PET centers in Western countries.
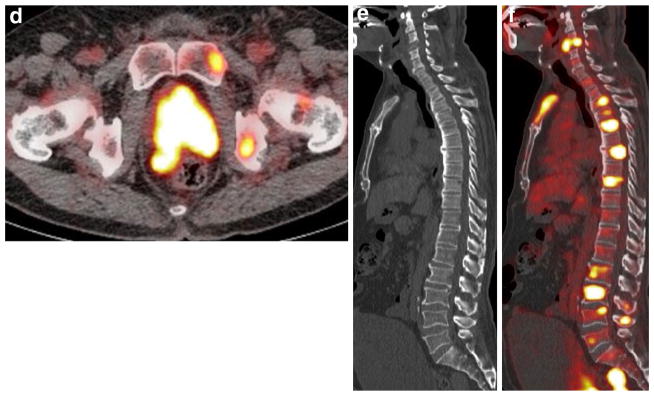
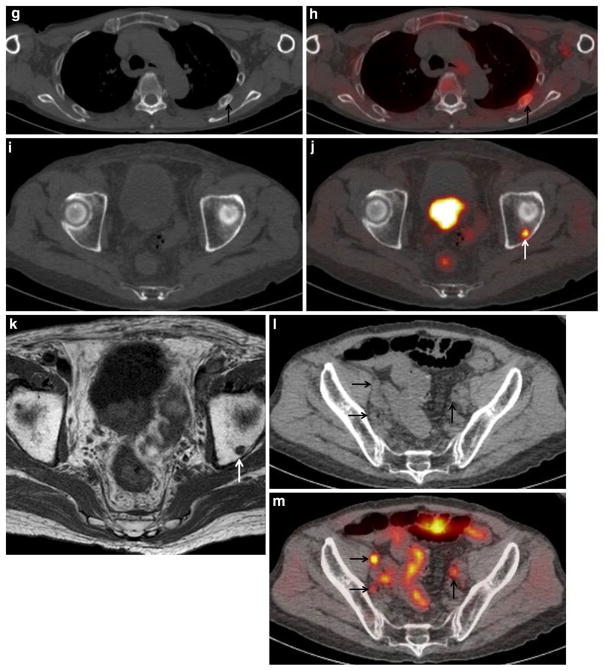
Fig. 4.
An 84-year-old man with PSA level of 43 ng/ml, who underwent androgen-deprivation therapy. a MIP of 11C-choline PET shows multiple areas of abnormal choline uptake in the prostate, whole-body lymph node and bone. b Bone scintigraphy shows abnormal uptake in the left fourth and fifth ribs (arrow), suggesting bone metastases. c CT of 11C-choline PET/CT and d PET/CT show intense 11C-choline uptake corresponding to the prostate and seminal vesicle (SUVmax: 9.7), suggesting local recurrence in the prostate and seminal vesicle. e T2-weighted and f diffusion-weighted MRI show a slight hypointense mass with restricted water diffusion occupying the prostate and seminal vesicle (arrows), consistent with local recurrence. g CT of 11C-choline PET/CT and h PET/CT show mild 11C-choline uptake corresponding to the mild sclerotic change of the left fifth rib (arrow), suggesting bone metastasis, similar to (b) bone scintigraphy. i CT of 11C-choline PET/CT and j PET/CT show tiny and mild 11C-choline uptake corresponding to the left acetabulum (arrow), suggesting bone metastasis. k T1-weighted MRI shows a hypointense spot in the left acetabulum (arrow), consistent with bone metastasis. The detection of this bony metastasis by only (i) CT or (b) bone scintigraphy is impossible. l CT of FDG-PET/CT and m PET/ CT show moderate FDG uptake corresponding to the three swollen pelvic lymph nodes (arrows), suggesting lymph node metastases. The detection of these tiny nodal metastases by only (l) CT is difficult
Giovacchini et al. evaluated the findings of 11C-choline PET/CT in 358 patients (PSA 0.23–45.0 ng/ml, median 1.27 ng/ml) treated with RP, and reported that patient-based sensitivity, specificity, and accuracy for restaging of prostate cancer were 85, 93, and 89 %, respectively; the percentage of positive scans was 19 % when the PSA level was 0.23–1 ng/ml, 46 % for 1–3 ng/ml, and 82 % for >3 ng/ ml [65]. In a multivariate analysis of 1000 patients with biochemical evidence of recurrence after various treatments (PSA 1.15–11.0 ng/ml, median 3.30 ng/ml), Cimitan et al. revealed that only older age, a Gleason score greater than or equal to 7, systemic chemotherapy, and a serum PSA level greater than or equal to 1 ng/ml were independent predictors of 18F-choline PET/CT positivity [66]. One systematic review and meta-analysis including 19 selected studies with a total of 1555 patients revealed a pooled sensitivity of 85.6 % and pooled specificity of 92.6 % [67]. In recent years, two groups have clarified that multiparametric MRI with an endorectal coil is superior for detection of local recurrence after RP relative to choline PET/CT, and therefore, strictly speaking, the combination of multiparametric MRI and choline PET/CT would be an ideal tool for restaging of patients with PSA failure [68, 69]. In the future, wider application of integrated PET/MRI can be expected.
Acetate
As well as choline PET/CT, 11C-acetate PET/CT has also been useful for identification of disease recurrence in the setting of PSA failure, with a reported sensitivity of 59–83 % [70]. Wachter et al. evaluated the clinical impact of 11C-acetate PET/CT fused imaging on treatment decision-making in 50 patients with recurrent prostate cancer after definitive treatment, and found that in 14 patients (28 %) the treatment plan was changed on the basis of the11C-acetate PET/CT findings [71].
NaF
PET with 18F-sodium fluoride (18F-NaF), a highly sensitive radiotracer for skeletal metastases, whose uptake reflects the increased regional blood flow and bone turnover in malignant bone lesions, has been reported to be more sensitive for detection of skeletal metastases than bone scintigraphy [72, 73] and 18F-FDG PET [74]. It appears that 18F-NaF PET/CT may exploit the high sensitivity of 18F-NaF PET, reducing the risk of false positivity by determining the morphology of scintigraphic lesions demonstrated by the CT component of PET/CT [72]. Recently, combined administration of 18F-NaF and 18F-FDG in a single PET/ CT scan for cancer detection has been advocated for detection of both extraskeletal and skeletal lesions [75], and it has been reported that 18F-NaF/FDG PET/CT is superior to whole-body MRI and bone scintigraphy for evaluating the extent of skeletal disease [76].
FDHT
Androgen receptor imaging with PET radiotracers, such as 16β-18F-fluoro-5α-dihydrotestosterone (18F-FDHT), has been developed to exploit its important role in the assessment of growth and proliferation of prostate carcinoma as well as modulation of androgen status. Antiandrogen therapy is widely used in the treatment of prostate carcinoma, and therefore may be useful for monitoring of treatment response. In order to effectively study the biology of prostate cancer and develop therapies that target the androgen receptor, methods for assessing androgen receptor expression and functionality are needed. However, such analyses are difficult because the dominant site of metastases is bone, which is notoriously problematic to biopsy. A small pilot study of patients with progressive clinically metastatic prostate carcinoma demonstrated excellent tumor localization and retention, and treatment resulted in a reduction of 18F-FDHT uptake at the tumor sites [77].
FACBC
In recent years, the investigational synthetic L-leucine analog anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid (anti-3-18F-FACBC) has been proposed as a possible alternative radiopharmaceutical for detection of prostate cancer relapse [78]. Anti-3-18F-FACBC uptake is related to the functional activity of two different amino acid transporters (ASC and LAT1) [79], which appear to be upregulated when prostate cancer progresses to metastatic disease. The levels of many amino acids are increased within prostate cancer cells in comparison with noncancerous tissues, many of which exceeded the increases observed for choline. Nanni et al. demonstrated that anti-3-18F-FACBC PET/CT may be superior to 11C-choline PET/CT for identification of disease recurrence in the setting of biochemical failure [80, 81].
PSMA
Prostate-specific membrane antigen (PSMA) has been used as a relevant target for imaging of prostate cancer. PSMA is a cell-surface protein expressed at higher levels in prostate carcinoma tissue than in other tissues, and its expression is associated with tumor aggressiveness, androgen independence, metastatic disease, and disease recurrence [82]. Mostly in Europe, PSMA-targeting ligands have been labeled with 68Ga for PET imaging, and this approach shows some promise for detection of prostate carcinoma recurrence. Eiber et al. [83] analyzed the findings of 68Ga-PSMA PET/CT in 248 patients (PSA 0.2–59.4 ng/ ml, median 1.99 ng/ml) treated with RP and found that the patient-based detection rate was 89.5 %; the detection rates for PSA levels of ≥2, 1 to <2, 0.5 to <1, and 0.2 to <0.5 ng/ ml were 96.8, 93.0, 72.7, and 57.9 %, respectively. Importantly, 68Ga-PSMA PET/CT reveals a high degree of positivity within the clinically important range of low PSA values (<0.5 ng/ml), which in many cases can substantially influence further clinical management [83]. Moreover, two groups have demonstrated that 68Ga-PSMA PET/CT may be superior to 18F-choline PET/CT [84, 85] and conventional imaging modalities (bone scintigraphy and contrast-enhanced CT of the chest, abdomen, and pelvis) [86] for identification of disease recurrence in the setting of biochemical failure. Rowe et al. evaluated 13 patients with primary prostate cancer who underwent imaging with MRI and PET/CT with N-[N-[(S)-1,3-dicarboxypropyl]carbamoyl]-4-(18)F-fluorobenzyl-L-cysteine (18F-DCFBC), a low-molecular-weight radiotracer that targets the PSMA, prior to RP. They demonstrated that although the sensitivity of 18F-DCFBC for primary prostate cancer was less than that of MRI, 18F-DCFBC PET was able to detect more clinically significant high-grade and larger-volume tumors (Gleason score 8 and 9) with higher specificity than MRI. They also found relatively low 18F-DCFBC PET uptake in BPH lesions, compared with cancer in the prostate, possibly allowing more specific detection of primary prostate cancer by 18F-DCFBC PET. Furthermore, 18F-DCFBC uptake in primary tumors was positively correlated with the Gleason score (ρ = 0.64; PSMA expression, ρ = 0.47; and PSA, ρ = 0.52) [87].
As PSMA-617 can be labeled with 68Ga, 177Lu, 111In, or 90Y, it can be used for PET imaging as well as for radioligand-based therapy. Recently, 177Lu-PSMA-617 has been used increasingly for radioligand therapy of metastatic prostate cancer at several centers [88].
Adrenal malignant tumor
Malignant adrenal disease tends to show higher 18F-FDG uptake than benign adrenal lesion and one systematic review and meta-analysis including 21 selected studies with a total of 1391 lesions (824 benign, 567 malignant) in 1217 patients revealed a pooled sensitivity of 97 % (95 % CI 93–98 %) and specificity of 91 % (95 % CI 87–94 %) of 18F-FDG PET/CT (SUVmax or standard uptake ratio) for differentiating between malignant and benign adrenal disease [89]. False negativities (partial volume effect in evaluating small lesions, necrotic or hemorrhagic metastases, and metastases from RCC or malignant neuroendocrine tumor) and false positives (adenoma, hyperplasia, tuberculosis, pheochromocytoma, or hemorrhage) are also reported [90].
A multicenter study demonstrated that 18F-FDG PET/ CT showed the better diagnostic accuracy of diagnosing primary adrenal malignancy (adrenocortical carcinoma, malignant pheochromocytoma, neuroblastoma, and lymphoma) than contrast-enhanced CT [91]. Adrenocortical carcinoma is a rare and aggressive disease and 18F-FDG PET/CT may be a complementary tool to morphological imaging techniques, useful in diagnosis and detecting disease recurrence or metastatic sites, and in evaluation of treatment response of adrenocortical carcinoma [92, 93]. The diagnostic performance of 18F-FDG PET/CT for differentiating metastatic adrenal tumors from adrenal adenoma is high (sensitivity, specificity, and accuracy are 82, 92, and 90 %) as well as adrenal protocol CT (unenhanced, early and delayed contrast-enhanced CT) and, moreover, the combination of 18F-FDG PET/CT and adrenal protocol CT can improve the 18F-FDG PET/CT or adrenal protocol CT diagnostic accuracy for differentiation of metastatic adrenal tumors from adrenal adenoma, reducing false-positive cases [94].
Testicular cancer
18F-FDG PET/CT is clinically useful for initial staging of testicular cancer and determining the viability of residual masses after completion of treatment, especially in patients with seminoma (Fig. 5).
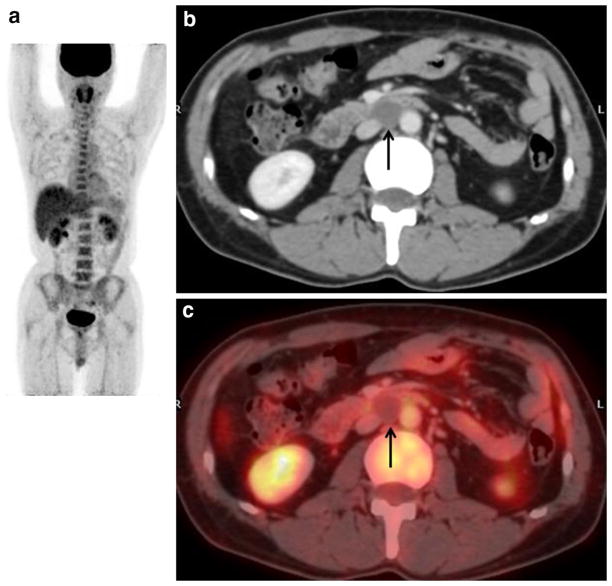
Fig. 5.
A 51-year-old man with residual retroperitoneal mass after undergoing chemotherapy treatment for seminoma. a MIP of FDG-PET shows no abnormal FDG uptake. Mild and diffuse FDG uptake in the whole-body skeleton may be reactive uptake due to the hyper-hematopoietic function of the bone marrow. b Contrast-enhanced CT of FDG-PET/CT shows a 1.5-cm residual retroperitoneal mass (arrow). c FDG-PET/CT shows no increased activity in residual mass (arrow). Subsequent follow-up over 1 year has shown both no evidence of recurrent disease and no elevated tumor markers
Ambrosini et al. [95] evaluated the clinical impact of 18F-FDG PET/CT for staging and restaging of testicular tumors, reviewing 51 cases of seminoma and 70 cases of non-seminoma. PET/CT showed good sensitivity and specificity for detection of seminoma lesions (92 and 84 %, respectively), but its sensitivity was lower for non-seminoma forms (77 and 95 %, respectively); it influenced the clinical management of 47 out of 51 (92 %) seminomas and 59 out of 70 (84 %) non-seminomas. One systematic review and meta-analysis including 4 selected studies with a total of 130 patients evaluated the diagnostic accuracy of 18F-FDG PET/CT for prediction of viable residual tumors after chemotherapy in patients with metastatic seminoma and demonstrated that it was superior to CT for determining tumor size and predicting tumor viability: sensitivity was 72 vs 63 %, specificity was 92 vs 59 %, positive predictive value (PPV) was 70 vs 28 %, and negative predictive value (NPV) was 93 vs 86 %, respectively [96]. Another recent systematic review and meta-analysis including 9 selected studies with a total of 375 patients showed that 18F-FDG PET/CT had a pooled sensitivity of 78 % (95 % CI: 67–87 %), a specificity of 86 % (95 % CI: 81–89 %), a PPV of 58 % (95 % CI: 48–68 %), a NPV of 94 % (95 % CI: 90–96 %), and an accuracy of 84 % (95 % CI: 80–88 %) [97]. Based on these results, it can be concluded that negative 18F-FDG PET/CT findings warrant follow-up if only to avoid inappropriate subsequent treatment (surgery, chemotherapy, or radiotherapy), whereas positive 18F-FDG PET/CT findings suggest a high possibility of residual seminoma, though a false-positive result cannot be excluded in view of the low PPV.
However, there are several major limitations to 18F-FDG PET: (1) lesions of <1 cm are often not detected due to the limited spatial resolution of PET. (2) Mature teratoma has variable low 18F-FDG uptake or no uptake, and cannot be distinguished from necrosis or fibrosis, and therefore 18F-FDG PET is not recommended for characterization of residual masses after therapy for non-seminomatous germ cell cancers, which frequently harbor residual foci of mature teratoma [98]. (3) There should be a minimum of 6 weeks between the end of chemotherapy and acquisition of an 18F-FDG PET/CT scan to avoid any false-positive findings due to chemotherapy-induced inflammatory and granulomatous tissues or false-negative results due to temporary suppression of tumor cell activities [99].
PET/MRI
In recent years, there has been increasing interest in the development of integrated PET/MRI systems, which are now commercially available. PET/MRI would have a number of advantages over PET/CT, such as improved soft-tissue contrast, the possibility of performing truly simultaneous instead of sequential acquisitions, and the availability of sophisticated MRI sequences, such as diffusion and perfusion imaging, functional MRI, and MR spectroscopy, which can add important information. Moreover, the use of PET/MRI would allow a significant decrease in radiation exposure, which is of foremost importance for serial follow-up and pediatric imaging. Several groups have demonstrated the usefulness of PET/MRI in patients with RCC using 18F-FLT [100], bladder cancer using 18F-FDG [101], prostate cancer using choline [102, 103] or PSMA [104], and adrenal pheochromocytoma using 18F-fluorodihydro-xyphenylalanine (18F-DOPA) [105].
Conclusion
The use of PET/CT in oncology is rapidly expanding with 18F-FDG, the most commonly used radiotracer. Although 18F-FDG in urological oncology is a challenge, 18F-FDG PET/CT was recently demonstrated to be useful when applied to specific indications in selected patients. The rapid development of new metabolic PET tracers with more favorable properties has improved the ability to visualize these urological malignancies, and several advances in PET/CT have been made in recent years. New radiotracers and new techniques of PET/CT and PET/MRI are expected to further improve the performance of PET in uro-oncology. Thus, the use of PET/CT and PET/MRI for patients with urological malignancies will continue to expand. Larger clinical trials are needed to further establish the role of PET/CT and PET/MRI in the management of urological malignancy.
Footnotes
Compliance with ethical standards
Conflict of interest We declare that we have no financial support or relationship that may pose a conflict of interest.
References
- 1.Wang HY, Ding HJ, Chen JH, Chao CH, Lu YY, Lin WY, et al. Meta-analysis of the diagnostic performance of [18F]FDG-PET and PET/CT in renal cell carcinoma. Cancer Imaging. 2012;12:464–74. doi: 10.1102/1470-7330.2012.0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ferda J, Ferdova E, Hora M, Hes O, Finek J, Topolcan O, et al. FDG-PET/CT in potentially advanced renal cell carcinoma: a role in treatment decisions and prognosis estimation. Anticancer Res. 2013;33:2665–72. [PubMed] [Google Scholar]
- 3.Win AZ, Aparici CM. Clinical effectiveness of 18F-fluorode-oxyglucose positron emission tomography/computed tomography in management of renal cell carcinoma: a single institution experience. World J Nucl Med. 2015;14:36–40. doi: 10.4103/1450-1147.150535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Park JW, Jo MK, Lee HM. Significance of 18F-fluorodeoxyglu-cose positron-emission tomography/computed tomography for the postoperative surveillance of advanced renal cell carcinoma. BJU Int. 2009;103:615–9. doi: 10.1111/j.1464-410X.2008.08150.x. [DOI] [PubMed] [Google Scholar]
- 5.Nakatani K, Nakamoto Y, Saga T, Higashi T, Togashi K. The potential clinical value of FDG-PET for recurrent renal cell carcinoma. Eur J Radiol. 2011;79:29–35. doi: 10.1016/j.ejrad.2009.11.019. [DOI] [PubMed] [Google Scholar]
- 6.Rodríguez Martínez de Llano S, Jiménez-Vicioso A, Mahmood S, Carreras-Delgado JL. Clinical impact of 18F-FDG PET in management of patients with renal cell carcinoma. Rev Esp Med Nucl. 2010;29:12–9. doi: 10.1016/j.remn.2009.11.008. [DOI] [PubMed] [Google Scholar]
- 7.Fuccio C, Ceci F, Castellucci P, Spinapolice EG, Palumbo R, D’Ambrosio D, et al. Restaging clear cell renal carcinoma with 18F-FDG PET/CT. Clin Nucl Med. 2014;39:e320–4. doi: 10.1097/RLU.0000000000000382. [DOI] [PubMed] [Google Scholar]
- 8.Alongi P, Picchio M, Zattoni F, Spallino M, Gianolli L, Saladini G, et al. Recurrent renal cell carcinoma: clinical and prognostic value of FDG PET/CT. Eur J Nucl Med Mol Imaging. 2016;43:464–73. doi: 10.1007/s00259-015-3159-6. [DOI] [PubMed] [Google Scholar]
- 9.Noda Y, Kanematsu M, Goshima S, Suzui N, Hirose Y, Matsunaga K, et al. 18-F fluorodeoxyglucose uptake in positron emission tomography as a pathological grade predictor for renal clear cell carcinomas. Eur Radiol. 2015;25:3009–16. doi: 10.1007/s00330-015-3687-2. [DOI] [PubMed] [Google Scholar]
- 10.Takahashi M, Kume H, Koyama K, Nakagawa T, Fujimura T, Morikawa T, et al. Preoperative evaluation of renal cell carcinoma by using 18F-FDG PET/CT. Clin Nucl Med. 2015;40:936–40. doi: 10.1097/RLU.0000000000000875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nakajima R, Abe K, Kondo T, Tanabe K, Sakai S. Clinical role of early dynamic FDG-PET/CT for the evaluation of renal cell carcinoma. Eur Radiol. 2015 doi: 10.1007/s00330-015-4026-3. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 12.Mizuno T, Kamai T, Abe H, Sakamoto S, Kitajima K, Nishihara D, et al. Clinically significant association between the maximum standardized uptake value on 18F-FDG PET and expression of phosphorylated Akt and S6 kinase for prediction of the biological characteristics of renal cell cancer. BMC Cancer. 2015;15:1097. doi: 10.1186/s12885-015-1097-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Namura K, Minamimoto R, Yao M, Makiyama K, Murakami T, Sano F, et al. Impact of maximum standardized uptake value (SUVmax) evaluated by 18-Fluoro-2-deoxy-D-glucose positron emission tomography/computed tomography (18F-FDG-PET/ CT) on survival for patients with advanced renal cell carcinoma: a preliminary report. BMC Cancer. 2010;10:667. doi: 10.1186/1471-2407-10-667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kayani I, Avril N, Bomanji J, Chowdhury S, Rockall A, Sahdev A, et al. Sequential FDG-PET/CT as a biomarker of response to sunitinib in metastatic clear cell renal cancer. Clin Cancer Res. 2011;17:6021–8. doi: 10.1158/1078-0432.CCR-10-3309. [DOI] [PubMed] [Google Scholar]
- 15.Revheim ME, Winge-Main AK, Hagen G, Fjeld JG, Fosså SD, Lilleby W. Combined positron emission tomography/computed tomography in sunitinib therapy assessment of patients with metastatic renal cell carcinoma. Clin Oncol (R Coll Radiol) 2011;23:339–43. doi: 10.1016/j.clon.2010.11.006. [DOI] [PubMed] [Google Scholar]
- 16.Ueno D, Yao M, Tateishi U, Minamimoto R, Makiyama K, Hayashi N, et al. Early assessment by FDG-PET/CT of patients with advanced renal cell carcinoma treated with tyrosine kinase inhibitors is predictive of disease course. BMC Cancer. 2012;12:162. doi: 10.1186/1471-2407-12-162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen JL, Appelbaum DE, Kocherginsky M, Cowey CL, Rathmell WK, McDermott DF, et al. FDG-PET as a predictive biomarker for therapy with everolimus in metastatic renal cell cancer. Cancer Med. 2013;2:545–52. doi: 10.1002/cam4.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yoon HJ, Paeng JC, Kwak C, Park YH, Kim TM, Lee SH, et al. Prognostic implication of extrarenal metabolic tumor burden in advanced renal cell carcinoma treated with targeted therapy after nephrectomy. Ann Nucl Med. 2013;27:748–55. doi: 10.1007/s12149-013-0742-4. [DOI] [PubMed] [Google Scholar]
- 19.Vercellino L, Bousquet G, Baillet G, Barré E, Mathieu O, Just PA, et al. 18F-FDG PET/CT imaging for an early assessment of response to sunitinib in metastatic renal carcinoma: preliminary study. Cancer Biother Radiopharm. 2009;24:137–44. doi: 10.1089/cbr.2008.0527. [DOI] [PubMed] [Google Scholar]
- 20.Farnebo J, Grybäck P, Harmenberg U, Laurell A, Wersäll P, Blomqvist LK, et al. Volumetric FDG-PET predicts overall and progression-free survival after 14 days of targeted therapy in metastatic renal cell carcinoma. BMC Cancer. 2014;14:408. doi: 10.1186/1471-2407-14-408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Signoretti S, Regan M, Atkins M. Carbonic anhydrase IX as a predictive biomarker of response to kidney cancer therapy. BJU Int. 2008;101:31–5. doi: 10.1111/j.1464-410X.2008.07646.x. [DOI] [PubMed] [Google Scholar]
- 22.Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50:1220S–50S. doi: 10.2967/jnumed.108.057307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hugonnet F, Fournier L, Medioni J, Smadja C, Hindié E, Huchet V, et al. Hypoxia in Renal Cancer Multicenter Group: metastatic renal cell carcinoma: relationship between initial metastasis hypoxia, change after 1 month’s sunitinib, and therapeutic response: an 18F-fluoromisonidazole PET/CT study. J Nucl Med. 2011;201:1048–55. doi: 10.2967/jnumed.110.084517. [DOI] [PubMed] [Google Scholar]
- 24.Horn KP, Yap JT, Agarwal N, Morton KA, Kadrmas DJ, Beard-more B, et al. FDG and FLT-PET for early measurement of response to 37.5 mg daily sunitinib therapy in metastatic renal cell carcinoma. Cancer Imaging. 2015;15:15. doi: 10.1186/s40644-015-0049-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Middendorp M, Maute L, Sauter B, Vogl TJ, Grünwald F. Initial experience with 18F-fluoroethylcholine PET/CT in staging and monitoring therapy response of advanced renal cell carcinoma. Ann Nucl Med. 2010;24:441–6. doi: 10.1007/s12149-010-0375-9. [DOI] [PubMed] [Google Scholar]
- 26.Oyama N, Takahara N, Hasegawa Y, Tanase K, Miwa Y, Akino H, et al. Assessment of therapeutic effect of sunitinib by 11C-Acetate PET compared with FDG PET imaging in a patient with metastatic renal cell carcinoma. Nucl Med Mol Imaging. 2011;45:217–9. doi: 10.1007/s13139-011-0084-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schuster DM, Nye JA, Nieh PT, Votaw JR, Halkar RK, Issa MM, et al. Initial experience with the radiotracer anti-1-amino-3-[18F]fluorocyclobutane-1-carboxylic acid (anti-[18F] FACBC) with PET in renal carcinoma. Mol Imaging Biol. 2009;11:434–8. doi: 10.1007/s11307-009-0220-5. [DOI] [PubMed] [Google Scholar]
- 28.Kaufman DS, Shipley WU, Feldman AS. Bladder cancer. Lancet. 2009;374:239–49. doi: 10.1016/S0140-6736(09)60491-8. [DOI] [PubMed] [Google Scholar]
- 29.Wang N, Jiang P, Lu Y. Is fluorine-18 fluorodeoxyglucose positron emission tomography useful for detecting bladder lesions? A meta-analysis of the literature. Urol Int. 2014;92:143–9. doi: 10.1159/000351964. [DOI] [PubMed] [Google Scholar]
- 30.Lodde M, Lacombe L, Friede J, Morin F, Saourine A, Fradet Y. Evaluation of fluorodeoxyglucose positron-emission tomography with computed tomography for staging of urothelial carcinoma. BJU Int. 2010;106:658–63. doi: 10.1111/j.1464-410X.2010.09212.x. [DOI] [PubMed] [Google Scholar]
- 31.Nayak B, Dogra PN, Naswa N, Kumar R. Diuretic 18F-FDG PET/CT imaging for detection and locoregional staging of urinary bladder cancer: prospective evaluation of a novel technique. Eur J Nucl Med Mol Imaging. 2013;40:386–93. doi: 10.1007/s00259-012-2294-6. [DOI] [PubMed] [Google Scholar]
- 32.Lu YY, Chen JH, Liang JA, Wang HY, Lin CC, Lin WY, et al. Clinical value of FDG PET or PET/CT in urinary bladder cancer: a systemic review and meta-analysis. Eur J Radiol. 2012;81:2411–6. doi: 10.1016/j.ejrad.2011.07.018. [DOI] [PubMed] [Google Scholar]
- 33.Apolo AB, Riches J, Schöder H, Akin O, Trout A, Milowsky MI, et al. Clinical value of fluorine-18 2-fluoro-2-deoxy-D-glucose positron emission tomography/computed tomography in bladder cancer. J Clin Oncol. 2010;28:3973–8. doi: 10.1200/JCO.2010.28.7052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mertens LS, Fioole-Bruining A, Vegt E, Vogel WV, van Rhijn BW, Horenblas S. Impact of 18F-fluorodeoxyglucose (FDG)-positron-emission tomography/computed tomography (PET/ CT) on management of patients with carcinoma invading bladder muscle. BJU Int. 2013;112:729–34. doi: 10.1111/bju.12109. [DOI] [PubMed] [Google Scholar]
- 35.Kollberg P, Almquist H, Bläckberg M, Cronberg C, Garpered S, Gudjonsson S, et al. [18F]Fluorodeoxyglucose - positron emission tomography/computed tomography improves staging in patients with high-risk muscle-invasive bladder cancer scheduled for radical cystectomy. Scand J Urol. 2015;49:296–301. doi: 10.3109/21681805.2014.990053. [DOI] [PubMed] [Google Scholar]
- 36.Öztürk H, Karapolat I. Efficacy of 18F-fluorodeoxyglucose-positron emission tomography/computed tomography in restaging muscle-invasive bladder cancer following radical cystectomy. Exp Ther Med. 2015;9:717–24. doi: 10.3892/etm.2015.2187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mertens LS, Fioole-Bruining A, van Rhijn BW, Kerst JM, Bergman AM, Vogel WV, et al. FDG-positron emission tomography/ computerized tomography for monitoring the response of pelvic lymph node metastasis to neoadjuvant chemotherapy for bladder cancer. J Urol. 2013;189:1687–91. doi: 10.1016/j.juro.2012.11.009. [DOI] [PubMed] [Google Scholar]
- 38.Giannatempo P, Alessi A, Miceli R, Raggi D, Farè E, Nicolai N, et al. Interim fluorine-18 fluorodeoxyglucose positron emission tomography for early metabolic assessment of therapeutic response to chemotherapy for metastatic transitional cell carcinoma. Clin Genitourin Cancer. 2014;12:433–9. doi: 10.1016/j.clgc.2014.03.007. [DOI] [PubMed] [Google Scholar]
- 39.Mertens LS, Mir MC, Scott AM, Lee ST, Fioole-Bruining A, Vegt E, et al. 18F-fluorodeoxyglucose–positron emission tomography/computed tomography aids staging and predicts mortality in patients with muscle-invasive bladder cancer. Urology. 2014;83:393–8. doi: 10.1016/j.urology.2013.10.032. [DOI] [PubMed] [Google Scholar]
- 40.Vargas HA, Akin O, Schöder H, Olgac S, Dalbagni G, Hricak H, et al. Prospective evaluation of MRI, 11C-acetate PET/CT and contrast-enhanced CT for staging of bladder cancer. Eur J Radiol. 2012;81:4131–7. doi: 10.1016/j.ejrad.2012.06.010. [DOI] [PubMed] [Google Scholar]
- 41.Schöder H, Ong SC, Reuter VE, Cai S, Burnazi E, Dalbagni G, et al. Initial results with 11C-acetate positron emission tomography/computed tomography (PET/CT) in the staging of urinary bladder cancer. Mol Imaging Biol. 2012;14:245–51. doi: 10.1007/s11307-011-0488-0. [DOI] [PubMed] [Google Scholar]
- 42.Graziani T, Ceci F, Lopes FL, Chichero J, Castellucci P, Schiavina R, et al. 11C-choline PET/CT for restaging of bladder cancer. Clin Nucl Med. 2015;40:e1–5. doi: 10.1097/RLU.0000000000000573. [DOI] [PubMed] [Google Scholar]
- 43.Maurer T, Souvatzoglou M, Kübler H, Opercan K, Schmidt S, Herrmann K, et al. Diagnostic efficacy of 11C-choline positron emission tomography/computed tomography compared with conventional computed tomography in lymph node staging of patients with bladder cancer prior to radical cystectomy. Eur Urol. 2012;61:1031–8. doi: 10.1016/j.eururo.2011.12.009. [DOI] [PubMed] [Google Scholar]
- 44.Brunocilla E, Ceci F, Schiavina R, Castellucci P, Maffione AM, Cevenini M, et al. Diagnostic accuracy of 11C-choline PET/CT in preoperative lymph node staging of bladder cancer: a systematic comparison with contrast-enhanced CT and histologic findings. Clin Nucl Med. 2014;39:e308–12. doi: 10.1097/RLU.0000000000000342. [DOI] [PubMed] [Google Scholar]
- 45.Maurer T, Horn T, Souvatzoglou M, Eiber M, Beer AJ, Heck MM, et al. Prognostic value of 11C-choline PET/CT and CT for predicting survival of bladder cancer patients treated with radical cystectomy. Urol Int. 2014;93:207–13. doi: 10.1159/000357686. [DOI] [PubMed] [Google Scholar]
- 46.Patil VV, Wang ZJ, Sollitto RA, Chuang KW, Konety BR, Hawkins RA, et al. 18F-FDG PET/CT of transitional cell carcinoma. AJR Am J Roentgenol. 2009;193:W497–504. doi: 10.2214/AJR.08.1945. [DOI] [PubMed] [Google Scholar]
- 47.Asai S, Fukumoto T, Tanji N, Miura N, Miyagawa M, Nishimura K, et al. Fluorodeoxyglucose positron emission tomography/ computed tomography for diagnosis of upper urinary tract urothelial carcinoma. Int J Clin Oncol. 2015;20:1042–7. doi: 10.1007/s10147-015-0817-4. [DOI] [PubMed] [Google Scholar]
- 48.Tanaka H, Yoshida S, Komai Y, Sakai Y, Urakami S, Yuasa T, et al. Clinical value of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in upper tract urothelial carcinoma: impact on detection of metastases and patient management. Urol Int. 2016;96:65–72. doi: 10.1159/000381269. [DOI] [PubMed] [Google Scholar]
- 49.Kitajima K, Yamamoto S, Fukushima K, Yamakado K, Katsuura T, Igarashi Y, et al. FDG-PET/CT as a post-treatment restaging tool in urothelial carcinoma: comparison with contrast-enhanced CT. Eur J Radiol. 2016;85:593–8. doi: 10.1016/j.ejrad.2015.12.017. [DOI] [PubMed] [Google Scholar]
- 50.Jadvar H, Xiankui L, Shahinian A, Park R, Tohme M, Pinski J, et al. Glucose metabolism of human prostate cancer mouse xenografts. Mol Imaging. 2005;4:91–7. doi: 10.1162/15353500200505118. [DOI] [PubMed] [Google Scholar]
- 51.Jadvar H. Imaging evaluation of prostate cancer with 18F-fluoro-deoxyglucose PET/CT: utility and limitations. Eur J Nucl Med Mol Imaging. 2013;40:S5–10. doi: 10.1007/s00259-013-2361-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jadvar H, Desai B, Ji L, Conti PS, Dorff TB, Groshen SG, et al. Baseline 18F-FDG PET/CT parameters as imaging biomarkers of overall survival in castrate-resistant metastatic prostate cancer. J Nucl Med. 2013;54:1195–201. doi: 10.2967/jnumed.112.114116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bertagna F, Piccardo A, Dib B, Bertoli M, Fracassi F, Bosio G, et al. Multicentre study of 18F-FDG-PET/CT prostate incidental uptake. Jpn J Radiol. 2015;33:538–46. doi: 10.1007/s11604-015-0453-y. [DOI] [PubMed] [Google Scholar]
- 54.Bertagna F, Sadeghi R, Giovanella L, Treglia G. Incidental uptake of 18F-fluorodeoxyglucose in the prostate gland. Systematic review and meta-analysis on prevalence and risk of malignancy. Nuklearmedizin. 2014;53:249–58. doi: 10.3413/Nukmed-0668-14-05. [DOI] [PubMed] [Google Scholar]
- 55.Hwang I, Chong A, Jung SI, Hwang EC, Kim SO, Kang TW, et al. Is further evaluation needed for incidental focal uptake in the prostate in 18-fluoro-2-deoxyglucose positron emission tomography-computed tomography images? Ann Nucl Med. 2013;27:140–5. doi: 10.1007/s12149-012-0663-7. [DOI] [PubMed] [Google Scholar]
- 56.Kwon T, Jeong IG, You D, Hong JH, Ahn H, Kim CS. Prevalence and clinical significance of incidental 18F-fluoro-2-deoxyglucose uptake in prostate. Korean J Urol. 2015;56:288–94. doi: 10.4111/kju.2015.56.4.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Brown AM, Lindenberg ML, Sankineni S, Shih JH, Johnson LM, Pruthy S, et al. Does focal incidental 18F-FDG PET/CT uptake in the prostate have significance? Abdom Imaging. 2015;40:3222–9. doi: 10.1007/s00261-015-0520-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Morris MJ, Akhurst T, Osman I, Nunez R, Macapinlac H, Siedlecki K, et al. Fluorinated deoxyglucose positron emission tomography imaging in progressive metastatic prostate cancer. Urology. 2002;59:913–8. doi: 10.1016/s0090-4295(02)01509-1. [DOI] [PubMed] [Google Scholar]
- 59.Piert M, Park H, Khan A, Siddiqui J, Hussain H, Chenevert T, et al. Detection of aggressive primary prostate cancer with 11C-choline PET/CT using multimodality fusion techniques. J Nucl Med. 2009;50:1585–93. doi: 10.2967/jnumed.109.063396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Reske SN, Blumstein NM, Neumaier B, Gottfried HW, Finsterbusch F, Kocot D, et al. Imaging prostate cancer with 11C-choline PET/CT. J Nucl Med. 2006;47:1249–54. [PubMed] [Google Scholar]
- 61.Beheshti M, Imamovic L, Broinger G, Vali R, Waldenberger P, Stoiber F, et al. 18F Choline PET/CT in the preoperative staging of prostate cancer in patients with intermediate or high risk of extracapsular disease: a prospective study of 130 patients. Radiology. 2010;254:925–33. doi: 10.1148/radiol.09090413. [DOI] [PubMed] [Google Scholar]
- 62.Kitajima K, Murphy RC, Nathan MA. Choline PET/CT for imaging prostate cancer: an update. Ann Nucl Med. 2013;27:581–91. doi: 10.1007/s12149-013-0731-7. [DOI] [PubMed] [Google Scholar]
- 63.Poulsen MH, Bouchelouche K, Hoilund-Carlsen PF, Petersen H, Gerke O, Steffansen SI, et al. [18F]-Fluorocholine positron-emission/computed tomography for lymph node staging of prostate cancer: a prospective study of 210 patients. BJU Int. 2012;110:1666–71. doi: 10.1111/j.1464-410X.2012.11150.x. [DOI] [PubMed] [Google Scholar]
- 64.Picchio M, Spinapolice EG, Fallanca F, Crivellaro C, Giovacchini G, Gianolli L, et al. [11C]Choline PET/CT detection of bone metastases in patients with PSA progression after primary treatment for prostate cancer: comparison with bone scintigraphy. Eur J Nucl Med Mol Imaging. 2012;39:13–26. doi: 10.1007/s00259-011-1920-z. [DOI] [PubMed] [Google Scholar]
- 65.Giovacchini G, Picchio M, Coradeschi E, Bettinardi V, Gianolli L, Scattoni V, et al. Predictive factors of [11C]choline PET/CT in patients with biochemical failure after radical prostatectomy. Eur J Nucl Med Mol Imaging. 2010;37:301–9. doi: 10.1007/s00259-009-1253-3. [DOI] [PubMed] [Google Scholar]
- 66.Cimitan M, Evangelista L, Hodolič M, Mariani G, Baseric T, Bodanza V, et al. Gleason score at diagnosis predicts the rate of detection of 18F-choline PET/CT performed when biochemical evidence indicates recurrence of prostate cancer: experience with 1,000 patients. J Nucl Med. 2015;56:209–15. doi: 10.2967/jnumed.114.141887. [DOI] [PubMed] [Google Scholar]
- 67.Evangelista L, Zattoni F, Guttilla A, Saladini G, Zattoni F, Colletti PM, et al. Choline PET or PET/CT and biochemical relapse of prostate cancer: a systematic review and meta-analysis. Clin Nucl Med. 2013;38:305–14. doi: 10.1097/RLU.0b013e3182867f3c. [DOI] [PubMed] [Google Scholar]
- 68.Panebianco V, Sciarra A, Lisi D, Galati F, Buonocore V, Catalano C, et al. Prostate cancer: 1HMRS-DCEMR at 3T versus [18F]choline PET/CT in the detection of local prostate cancer recurrence in men with biochemical progression after radical retropubic prostatectomy (RRP) Eur J Radiol. 2012;81:700–8. doi: 10.1016/j.ejrad.2011.01.095. [DOI] [PubMed] [Google Scholar]
- 69.Kitajima K, Murphy RC, Nathan MA, Froemming AT, Hagen CE, Takahashi N, et al. Detection of recurrent prostate cancer after radical prostatectomy: comparison of 11C-choline PET/CT with pelvic multiparametric MR imaging with endorectal coil. J Nucl Med. 2014;55:223–32. doi: 10.2967/jnumed.113.123018. [DOI] [PubMed] [Google Scholar]
- 70.Kitajima K, Murphy RC, Nathan MA, Sugimura K. Update on positron emission tomography for imaging of prostate cancer. Int J Urol. 2014;21:12–23. doi: 10.1111/iju.12250. [DOI] [PubMed] [Google Scholar]
- 71.Wachter S, Tomek S, Kurtaran A, Wachter-Gerstner N, Djavan B, Becherer A, et al. 11C-acetate positron emission tomography imaging and image fusion with computed tomography and magnetic resonance imaging with recurrent prostate cancer. J Clin Oncol. 2006;24:2513–9. doi: 10.1200/JCO.2005.03.5279. [DOI] [PubMed] [Google Scholar]
- 72.Even-Sapir E, Metser U, Mishani E, Lievshitz G, Lerman H, Leibovitch I. The detection of bone metastases in patients with high-risk prostate cancer: 99mTc-MDP Planar bone scintigraphy, single- and multi-field-of-view SPECT, 18F-fluoride PET, and 18F-fluoride PET/CT. J Nucl Med. 2006;47:287–97. [PubMed] [Google Scholar]
- 73.Tateishi U, Morita S, Taguri M, Shizukuishi K, Minamimoto R, Kawaguchi M, et al. A meta-analysis of 18F-fluoride positron emission tomography for assessment of metastatic bone tumor. Ann Nucl Med. 2010;24:523–31. doi: 10.1007/s12149-010-0393-7. [DOI] [PubMed] [Google Scholar]
- 74.Jadvar H, Desai B, Ji L, Conti PS, Dorff TB, Groshen SG, et al. Prospective evaluation of 18F-NaF and 18F-FDG PET/CT in detection of occult metastatic disease in biochemical recurrence of prostate cancer. Clin Nucl Med. 2012;37:637–43. doi: 10.1097/RLU.0b013e318252d829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Iagaru A, Mittra E, Yaghoubi SS, Dick DW, Quon A, Goris ML, et al. Novel strategy for a cocktail 18F-fluoride and 18F-FDG PET/CT scan for evaluation of malignancy: results of the pilot-phase study. J Nucl Med. 2009;50:501–5. doi: 10.2967/jnumed.108.058339. [DOI] [PubMed] [Google Scholar]
- 76.Minamimoto R, Loening A, Jamali M, Barkhodari A, Mosci C, Jackson T, et al. Prospective comparison of 99mTc MDP scintigraphy, combined 18F-NaF and 18F-FDG PET/CT and whole-body MRI in patients with breast and prostate cancers. J Nucl Med. 2015;56:1862–8. doi: 10.2967/jnumed.115.162610. [DOI] [PubMed] [Google Scholar]
- 77.Larson SM, Morris M, Gunther I, Beattie B, Humm JL, Akhurst TA, et al. Tumor localization of 16beta-18Ffluoro-5alpha-dihy-drotestosterone versus 18F-FDG in patients with progressive, metastatic prostate cancer. J Nucl Med. 2004;45:366–73. [PubMed] [Google Scholar]
- 78.Oka S, Okudaira H, Yoshida Y, Schuster DM, Goodman MM, Shirakami Y. Transport mechanisms of trans-1-amino-3-fluoro[1-(14)C]cyclobutanecarboxylic acid in prostate cancer cells. Nucl Med Biol. 2012;39:109–19. doi: 10.1016/j.nucmedbio.2011.06.008. [DOI] [PubMed] [Google Scholar]
- 79.Shoup TM, Olson J, Hoffman JM, Votaw J, Eshima D, Eshima L, et al. Synthesis and evaluation of [18F]1-amino-3-fluorocy-clobutane-1-carboxylic acid to image brain tumors. J Nucl Med. 1999;40:331–8. [PubMed] [Google Scholar]
- 80.Nanni C, Schiavina R, Boschi S, Ambrosini V, Pettinato C, Brunocilla E, et al. Comparison of 18F-FACBC and 11C-choline PET/CT in patients with radically treated prostate cancer and biochemical relapse: preliminary results. Eur J Nucl Med Mol Imaging. 2013;40:S11–7. doi: 10.1007/s00259-013-2373-3. [DOI] [PubMed] [Google Scholar]
- 81.Nanni C, Schiavina R, Brunocilla E, Borghesi M, Ambrosini V, Zanoni L, et al. 18F-FACBC compared with 11C-choline PET/ CT in patients with biochemical relapse after radical prostatectomy: a prospective study in 28 patients. Clin Genitourin Cancer. 2014;12:106–10. doi: 10.1016/j.clgc.2013.08.002. [DOI] [PubMed] [Google Scholar]
- 82.Bouchelouche K, Turkbey B, Choyke P, Capala J. Imaging prostate cancer: an update on positron emission tomography and magnetic resonance imaging. Curr Urol Rep. 2010;11:180–90. doi: 10.1007/s11934-010-0105-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Eiber M, Maurer T, Souvatzoglou M, Beer AJ, Ruffani A, Haller B, et al. Evaluation of hybrid 68Ga-PSMA ligand PET/ CT in 248 patients with biochemical recurrence after radical prostatectomy. J Nucl Med. 2015;56:668–74. doi: 10.2967/jnumed.115.154153. [DOI] [PubMed] [Google Scholar]
- 84.Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, et al. Comparison of PET imaging with a 68Ga-labelled PSMA ligand and 18F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2014;41:11–20. doi: 10.1007/s00259-013-2525-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Morigi JJ, Stricker PD, van Leeuwen PJ, Tang R, Ho B, Nguyen Q, et al. Prospective comparison of 18F-fluoromethylcholine versus 68Ga-PSMA PET/CT in prostate cancer patients who have rising PSA after curative treatment and are being considered for targeted therapy. J Nucl Med. 2015;56:1185–90. doi: 10.2967/jnumed.115.160382. [DOI] [PubMed] [Google Scholar]
- 86.Rowe SP, Macura KJ, Ciarallo A, Mena E, Blackford A, Nadal R, et al. Comparison of PSMA-based 18F-DCFBC PET/CT to conventional imaging modalities for detection of hormone-sensitive and castration-resistant metastatic prostate cancer. J Nucl Med. 2016;57:46–53. doi: 10.2967/jnumed.115.163782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rowe SP, Gage KL, Faraj SF, Macura KJ, Cornish TC, Gonzalez-Roibon N, et al. 18F-DCFBC PET/CT for PSMA-based detection and characterization of primary prostate cancer. J Nucl Med. 2015;56:1003–10. doi: 10.2967/jnumed.115.154336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kratochwil C, Giesel FL, Eder M, Afshar-Oromieh A, Benešová M, Mier W, et al. [177Lu]Lutetium-labelled PSMA ligand induced remission in a patient with metastatic prostate cancer. Eur J Nucl Med Mol Imaging. 2015;42:987–8. doi: 10.1007/s00259-014-2978-1. [DOI] [PubMed] [Google Scholar]
- 89.Boland GW, Dwamena BA, Jagtiani Sangwaiya M, Goehler AG, Blake MA, Hahn PF, et al. Characterization of adrenal masses by using FDG PET: a systematic review and meta-analysis of diagnostic test performance. Radiology. 2011;259:117–26. doi: 10.1148/radiol.11100569. [DOI] [PubMed] [Google Scholar]
- 90.Dong A, Cui Y, Wang Y, Zuo C, Bai Y. 18F-FDG PET/CT of adrenal lesions. AJR Am J Roentgenol. 2014;203:245–52. doi: 10.2214/AJR.13.11793. [DOI] [PubMed] [Google Scholar]
- 91.Cistaro A, Niccoli Asabella A, Coppolino P, Quartuccio N, Altini C, Cucinotta M, et al. Diagnostic and prognostic value of 18F-FDG PET/CT in comparison with morphological imaging in primary adrenal gland malignancies—a multicenter experience. Hell J Nucl Med. 2015;18:97–102. doi: 10.1967/s002449910202. [DOI] [PubMed] [Google Scholar]
- 92.Takeuchi S, Balachandran A, Habra MA, Phan AT, Bassett RL, Jr, Macapinlac HA, Chuang HH. Impact of 18F-FDG PET/CT on the management of adrenocortical carcinoma: analysis of 106 patients. Eur J Nucl Med Mol Imaging. 2014;41:2066–73. doi: 10.1007/s00259-014-2834-3. [DOI] [PubMed] [Google Scholar]
- 93.Ardito A, Massaglia C, Pelosi E, Zaggia B, Basile V, Brambilla R, et al. 18F-FDG PET/CT in the post-operative monitoring of patients with adrenocortical carcinoma. Eur J Endocrinol. 2015;173:749–56. doi: 10.1530/EJE-15-0707. [DOI] [PubMed] [Google Scholar]
- 94.Park SY, Park BK, Kim CK. The value of adding 18F-FDG PET/ CT to adrenal protocol CT for characterizing adrenal metastasis (≥10 mm) in oncologic patients. AJR Am J Roentgenol. 2014;202:W153–60. doi: 10.2214/AJR.13.10873. [DOI] [PubMed] [Google Scholar]
- 95.Ambrosini V, Zucchini G, Nicolini S, Berselli A, Nanni C, Allegri V, et al. 18F-FDG PET/CT impact on testicular tumours clinical management. Eur J Nucl Med Mol Imaging. 2014;41:668–73. doi: 10.1007/s00259-013-2624-3. [DOI] [PubMed] [Google Scholar]
- 96.Müller J, Schrader AJ, Jentzmik F, Schrader M. Assessment of residual tumours after systemic treatment of metastatic seminoma: 18F-2-fluoro-2-deoxy-D-glucose positron emission tomography—meta-analysis of diagnostic value. Urologe A. 2011;50:322–7. doi: 10.1007/s00120-010-2469-3. [DOI] [PubMed] [Google Scholar]
- 97.Treglia G, Sadeghi R, Annunziata S, Caldarella C, Bertagna F, Giovanella L. Diagnostic performance of fluorine-18-fluorode-oxyglucose positron emission tomography in the postchemotherapy management of patients with seminoma: systematic review and meta-analysis. Biomed Res Int. 2014;2014:852681. doi: 10.1155/2014/852681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Oechsle K, Hartmann M, Brenner W, Venz S, Weissbach L, Franzius C, et al. [18F]Fluorodeoxyglucose positron emission tomography in nonseminomatous germ cell tumors after chemotherapy: the German multicenter positron emission tomography study group. J Clin Oncol. 2008;26:5930–5. doi: 10.1200/JCO.2008.17.1157. [DOI] [PubMed] [Google Scholar]
- 99.Bachner M, Loriot Y, Gross-Goupil M, Zucali PA, Horwich A, Germa-Lluch JR, et al. 2-18Fluoro-deoxy-D-glucose positron emission tomography (FDG-PET) for postchemotherapy seminoma residual lesions: a retrospective validation of the SEM-PET trial. Ann Oncol. 2012;23:59–64. doi: 10.1093/annonc/mdr052. [DOI] [PubMed] [Google Scholar]
- 100.Antunes J, Viswanath S, Rusu M, Valls L, Hoimes C, Avril N, et al. Radiomics analysis on FLT-PET/MRI for characterization of early treatment response in renal cell carcinoma: a proof-of-concept study. Transl Oncol. 2016;9:155–62. doi: 10.1016/j.tranon.2016.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Rosenkrantz AB, Balar AV, Huang WC, Jackson K, Friedman KP. Comparison of coregistration accuracy of pelvic structures between sequential and simultaneous imaging during hybrid PET/MRI in patients with bladder cancer. Clin Nucl Med. 2015;40:637–41. doi: 10.1097/RLU.0000000000000772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Souvatzoglou M, Eiber M, Martinez-Moeller A, Fürst S, Holzapfel K, Maurer T, et al. PET/MR in prostate cancer: technical aspects and potential diagnostic value. Eur J Nucl Med Mol Imaging. 2013;40:79–88. doi: 10.1007/s00259-013-2445-4. [DOI] [PubMed] [Google Scholar]
- 103.Wetter A, Lipponer C, Nensa F, Heusch P, Rübben H, Altenbernd JC, et al. Evaluation of the PET component of simultaneous [18F]choline PET/MRI in prostate cancer: comparison with [18F]choline PET/CT. Eur J Nucl Med Mol Imaging. 2014;41:79–88. doi: 10.1007/s00259-013-2560-2. [DOI] [PubMed] [Google Scholar]
- 104.Freitag MT, Radtke JP, Hadaschik BA, Kopp-Schneider A, Eder M, Kopka K, et al. Comparison of hybrid 68Ga-PSMA PET/ MRI and 68Ga-PSMA PET/CT in the evaluation of lymph node and bone metastases of prostate cancer. Eur J Nucl Med Mol Imaging. 2016;43:70–83. doi: 10.1007/s00259-015-3206-3. [DOI] [PubMed] [Google Scholar]
- 105.Magnaldi S, Mayerhoefer ME, Khameneh A, Schuetz M, Javor D, Mitterhauser M, et al. 18F-DOPA PET/CT and MRI: description of 12 histologically-verified pheochromocytomas. Anticancer Res. 2014;34:791–5. [PubMed] [Google Scholar]