Abstract
Aberrant gene expression that drives human cancer can arise from epigenetic dysregulation. While much attention has focused on altered activity of transcription factors and chromatin-modulating proteins, proteins that act post-transcriptionally can potently affect expression of oncogenic signaling proteins. The RNA-binding proteins (RBPs) Musashi-1 (MSI1) and Musashi-2 (MSI2) are emerging as regulators of multiple critical biological processes relevant to cancer initiation, progression, and drug resistance. Following identification of Musashi as regulators of progenitor cell identity in Drosophila, the human Musashi proteins were initially linked to control of maintenance of hematopoietic stem cells, then stem cell compartments for additional cell types. More recently, the Musashi proteins were found to be overexpressed and prognostic of outcome in numerous cancer types, including colorectal, lung, and pancreatic cancers, glioblastoma, and several leukemias. MSI1 and MSI2 bind and regulate the mRNA stability and translation of proteins operating in essential oncogenic signaling pathways, including NUMB/Notch, PTEN/mTOR, TGF-β/SMAD3, MYC, cMET, and others. Based on these activities, MSI proteins maintain cancer stem cell populations and regulate cancer invasion, metastasis and development of more aggressive cancer phenotypes, including drug resistance. While RBPs are viewed as difficult therapeutic targets, initial efforts to develop MSI-specific inhibitors are promising and RNA interference-based approaches to inhibiting these proteins have had promising outcomes in preclinical studies. In the interim, understanding the function of these translational regulators may yield insight into the relationship between mRNA expression and protein expression in tumors, guiding tumor profiling analysis. This review provides a current overview of Musashi as a cancer driver and novel therapeutic target.
Keywords: RNA-binding proteins, Musashi-1, Musashi-2, cancer, leukemia, stem cells
Introduction
The Drosophila musashi (msi) gene was first described in 1994 by Makoto Nakamura and colleagues (1), based on its role in regulating asymmetrical division of Drosophila sensory organ precursor (SOP) cells. Whereas SOPs normally undergo a program of cell division that culminates in production of neuronal precursors, a bristle shaft and a socket cell, loss of msi leads to a double-bristle phenotype. The gene name reflects similarity of this phenotype to martial portrayals of the two-sword fighting style originated by the Japanese national hero Miyamoto Musashi. The msi gene is evolutionarily conserved: as a consequence of earlier gene duplication, humans have two related genes, Musashi-1 (MSI1) and Musashi-2 (MSI2). MSI1 and MSI2 share roughly 75% amino acid identity in overall structure and like msi, belong to a family of RNA-binding proteins (2). Since their first identification, the Musashi genes have attracted considerable interest as regulators of stem and progenitor cell characteristics. Subsequent study of msi demonstrated that it post-transcriptionally regulated expression of genes such as numb, a component of the Notch signaling cascade, a critical regulator of asymmetric cell division in cell progenitors (3). Further studies in Drosophila demonstrated roles for msi in development of spermatogenesis and germline stem cells (4–6).
Following the identification of murine and human MSI1 (7,8) and MSI2 (2), profiling of gene expression and direct functional assessments in mouse models first demonstrated an evolutionarily conserved role in guiding the appropriate differentiation of neuronal progenitor cells (7,9,10). This was followed by additional studies showing roles for the Musashi proteins in regulating normal cell differentiation and organ development for other tissue types (5,11–15), emphasizing the essential nature of this protein signaling over great evolutionary distance. For example, in early studies in mice, Msi1 was found to be highly expressed in the brain, and Msi1 deficiency caused hydrocephaly (16). MSI2 is critical regulator of hematopoietic stem cells, where it plays a function distinct from MSI1 (11,12). While MSI2 deletion leads to decrease in hematopoietic stem cells, overexpression of MSI2 leads to an increase in progenitor and stem cells (11,12). MSI2 also regulates spermatogenesis and embryogenesis (4–6,15).
Cancer is a disease characterized by changes in cell symmetry, differentiation, and self-renewal capacity. Unsurprisingly, for a number of genes originally identified as regulators of stem and progenitor cell capacity, altered function has been linked to cancer. The Musashi proteins were first linked to cancer in 2001 based on studies demonstrating the elevated expression of MSI1 in gliomas (17), medulloblastomas (18), and hepatomas (19). In 2003, MSI2 was identified as part of a translocation event with HoxA9 in chronic myeloid leukemias (CMLs) that preserved MSI2 RNA binding motifs (20), also implicating this paralogue in cancer development. The past several years have been marked by a surge of reports elucidating the frequency and mechanisms of involvement of the Musashi proteins, and particularly MSI2, in multiple forms of human cancer. This review focuses on these new findings, including the potential for drugging MSI2 as a therapeutic target. For comprehensive summaries of Musashi biology, the reader is directed to recent excellent articles, including (14,21,22).
Elevated Musashi protein expression characterizes solid and liquid tumors: evidence for prognostic biomarker status
Since the first implication of MSI1 and MSI2 as potential contributors to cancer pathology, elevated expression of these proteins has been identified in many types of solid tumor (Table 1), arising from organ sites including the brain, breast, pancreas, colon, lung, ovary, bladder, and others (17,23–33). In addition, elevated expression of Musashi proteins has been found in chronic myelogenous leukemia (CML), acute myelogenous leukemia (AML), and acute lymphoblastic leukemia (ALL) (11,12,34–36). For both Musashi proteins, expression is typically recognized as higher in tumor cells referenced to matched normal tissues, and associated with low differentiation status, poor prognosis, lymph node invasion and metastasis for solid tumors, and expression of additional markers of stem cells.
Table 1. Summary of Musashi-1 and Musashi-2 signaling effectors.
AML, acute myeloid leukemia; ALL, acute lymphoblastic leukemia; HSC, hematopoietic stem cells; NSCLC, non-small cell lung cancer.
| Gene | Target gene | Direction of effect on target | Protein function | Cell type | Reference |
|---|---|---|---|---|---|
| Musashi-1/2 | BRD4 | ↑ | Receptor tyrosine kinase; cell invasion | Pancreatic adenocarcinoma | (23) |
| cMET | ↑ | Epigenetic controls, DNA repair | |||
| HMGA2 | ↑ | Notch ligand | |||
| Jagged 1 | ↓ | Notch antagonist | Breast Cancer | (54) | |
| NUMB | ↓ | Cell cycle regulation | Neural progenitor cells | (3) (11) |
|
| p21 (CDKN1A) | ↓ | Receptor tyrosine kinase; cell invasion | Embryonic kidney cells Intestinal epithelium |
(37,92) | |
| Musashi-1 | Doublecortin | ↓ | Microtubule bundling | Neural progenitor cells | (90) |
| Musashi-2 | ↓ | Translation regulation | Testis germ cells | (6) | |
| NF-YA | ↓ | Transcription factor | Breast Cancer | (59) | |
| Tetraspanin 3 | ↑ | Formation of supramolecular membrane complexes | AML | (36) | |
| Musashi-2 | AHR | ↑ | Transcription factor; aryl hydrocarbon receptor | HSC | (91) |
| ESR1 | ↑ | Hormonal receptor | Breast cancer | (30) | |
| Hoxa9 | ↑ | Transcription factors; lymphocyte development | ALL/AML | (53) | |
| Ikzf2 | ↑ | ||||
| Myc | ↑ | Transcription factor, cell cycle progression | |||
| PTEN | ↓ | Phosphatase and inhibitor of PDK–AKT–mTORC1 Negative regulator of tyrosine kinase signaling |
Intestinal epithelium | (37) | |
| LRIG1 | ↓ | ||||
| SMAD3 | ↓ | Transmembrane threonine kinase for TGFβ superfamily members | HSC, NSCLC | (13,26) | |
| TGF-β RI | ↓ |
Control of Musashi protein expression
Our current knowledge regarding upstream signals/pathways regulating MSI proteins expression is limited. Some factors currently defined as influencing Musashi expression are represented in Figure 1. The studies summarized above tie expression of MSI1 and MSI2, at least indirectly, to factors linked to maintenance of stem cell characteristics or induction of cancer. These include loss of APC and induction of TCF/LEF binding in colon cancer (37,38), and repression by KLF4 in pancreatic cancer (24). MSI1 expression, along with other stem cell markers, was negatively regulated in GBM xenografts and in depleted stem-like GBM cells treated with the c-MET inhibitor crizotinib (39). In colon cancer, both stimulation by the NOTCH ligand DLL4 and expression of activated NOTCH3 increased MSI1 levels, whereas silencing of NOTCH3 by short hairpin RNA reduced MSI1 levels in cancer cells and tumor xenografts (40). In lung-infiltrating breast cancer cells, upregulation of MSI1 was dependent on expression of tenascin C (TNC), an extracellular matrix component that protects stem cells(31).
Figure 1. Musashi protein organization and signaling networks.
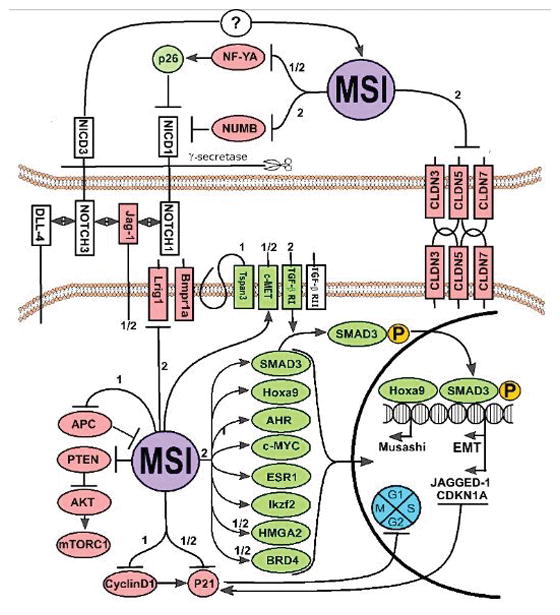
Selected cancer-relevant MSI1 and/or MSI2 regulatory proteins, and direct downstream binding targets are shown. Green, proteins positively regulated and red, genes negatively regulated, as reported for MSI1 (1) MSI2 (2) or both (1/2).
Post-transcriptionally, the MSI1 3′ UTR is regulated by direct interaction with another important RNA-binding protein (RBP), HuR, which positively regulated Msi1 expression in glioblastoma: expression of both proteins was correlated in primary GBM tumor samples (41). The Msi1 3′UTR region is also potentially targeted by several tumor suppressor miRNAs in glioblastoma and neuroblastoma (miR-34a, -101, -128, -137, and -138) (41). The finding of regulation by the tumor suppressive miR-137 was independently made in CRCs (42).
Finally, in insights from non-cancer models that may be relevant to suggesting cancer-relevant changes, in osteoclast precursors, MSI2 expression is upregulated by receptor activator of NF-κB ligand (RANKL) during osteoclast differentiation (43). Intriguingly, in mouse spermatogonia cells, MSI2 expression appears to be directly targeted and negatively regulated by MSI1 at the post-transcriptional level before nuclear translocation, suggesting cross-regulation between the two paralogues (6). These and other factors controlling Musashi expression are discussed at length in Fox et al (21).
Demonstration of driver roles for Musashi expression in oncogenesis
The first demonstrations of driver activity for Musashi overexpression in cancer etiology emerged in 2010, coupled with initial identification of Musashi-relevant cancer targets that suggested mechanisms of action. Ito et al used mouse models of CML to demonstrate that levels of MSI2 increased in CML blasts at the stage of blast crisis (11). This work linked directly back to the earlier observation that msi negatively regulated numb in Drosophila (3), as in CML. Ito and colleagues demonstrated that expression of the NUP98-HOXA9 fusion protein, an oncogene typically expressed at blast crisis, induces MSI2 expression, and that MSI2 expression is essential for Numb suppression and the cellular dedifferentiation that characterizes blast crisis. In the same year, Kharas and colleagues independently confirmed the specific role of MSI2, but not MSI1, in human myeloid leukemia cell lines and a BCR-ABL driven mouse model of CML and AML, and showed that MSI2 expression was required to support proliferation and depress apoptosis in these models (12). Also in 2010, Rezza and colleagues investigated MSI1 in the context of intestinal progenitor stem cells and colorectal cancer (CRC). This work also confirmed a functional requirement for a Musashi protein in supporting proliferation and inducing tumorigenic growth in xenograft assays. Mechanistically, this analysis demonstrated upregulation of MSI1 in cancer was induced by the WNT pathway (strongly linked to etiology of CRC), through direct transcriptional activation of MSI1 mediated by the WNT-dependent transcriptional activators Tcf/Lef. It also demonstrated that MSI1 was responsible for inducing the Notch pathway, and sustaining activity of the WNT pathway, suggesting a “feedforward” signaling function(44). These studies were followed by a number of others, similarly identifying activity of MSI1 and MSI2 in supporting proliferation and apoptosis of cancers of various types: for example, studies of MSI1 in medulloblastoma, glioblastoma and breast cancer identified a large number of potential targets that support oncogenesis, and others validated a role for MSI1 in metastasis and stemness in breast and colon cancer (31,41,45–48) (Table 2, Figure 1).
Table 2. MSI1 and MSI2 as prognostic biomarkers in human malignancies.
ALL, acute lymphocytic leukemia; CML, chronic myelogenous leukemia; HCC, hepatocellular carcinoma; IHC, immunohistochemistry; MDS, myelodysplastic syndrome; NSCLC, non-small cell lung cancer; PDAC, pancreatic adenocarcinoma; RT-qPCR, real-time quantitative polymerase chain reaction; SCLC, small cell lung cancer; TCGA, The Cancer Genome Atlas; TMA, tissue Microarray; WB, western blot.
| Gene | Cancer type | Type of Evidence | Reference |
|---|---|---|---|
| MSI-1/2 | Colon Cancer | TMA of 203 patient specimens | (110) |
| RNA-seq data for 26 paired tumor and normal tissue patient specimens (TCGA) | (55) (37) |
||
| WB analysis of 20 matched patient specimens | (25) | ||
| TMA and IHC of 146 patient specimens | (111) | ||
| SCLC | IHC of 85 patient lung tumors specimens | (112) | |
| NSCLC | IHC of 14 paired patient NSCLC specimens | (112) | |
| TMA of 123 NSCLC versus 22 normal lung tissue patient specimens | (26) | ||
| Musashi-1 | Barrett’s Esophagus and Esophageal Adenocarcinoma | IHC and RTqPCR of metaplastic and dysplastic patient specimens vs normal esophagus and adenocarcinoma | (93) |
| Bladder Cancer | RTqPCR of 70 patient specimens of bladder cancer vs normal tissue | (94) | |
| Breast Cancer | IHC 140 patient breast tumors; WB 20 breast cancer cell lines Methylation, IHC in 91 breast tumors |
(32) (33) |
|
| Endometrial Cancer | IHC and RTqPCR of 46 patient specimens | (95) | |
| RTqPCR of 35 tumor vs 15 normal tissue patient specimens | (96) | ||
| Gastric Cancer | IHC of gastric cancer vs normal tissue | (97) | |
| Gliomas | IHC of 241 patient specimens | (98) | |
| IHC and RTqPCR of 72 patient astrocytoma specimens vs normal brain | (99) | ||
| RTqPCR of 32 tumor vs nonneoplastic patient specimens | (100) | ||
| IHC of 73 patient glioma specimens; WB analysis 7 glioma and 8 astrocytoma vs normal brain patient specimens | (17) | ||
| Hepatoma | IHC of 24 matched patient specimens | (101) | |
| Medulloblastoma | TMA of 259 tumor vs 10 normal cerebellar patient specimens | (41) | |
| RTqPCR of 25 medulloblastoma patient specimens | (102) | ||
| RTqPCR for gene expression of patient brain tumor specimens | (18) | ||
| Ovarian Cancer | IHC analysis of 80 patient ovarian adenocarcinoma specimens vs normal ovarian tissue | (103) | |
| Retinoblastoma | IHC of 3 human RB tumor specimens; ICH of Y79 and WER1-RB27 cells | (104) | |
| Musashi-2 | ALL | RTqPCR 140 patient specimens with B-ALL | (105) |
| AML | Microarray of 16 AML patient specimens | (12) | |
| TMA of 120 AML patient specimens | (106) | ||
| RtqPCR 454 AML and 38 normal patient specimens | (107) | ||
| Microarray of 363 patient specimens | (53) | ||
| Bladder Cancer | WB of bladder cancer cell lines, IHC of 167 patient bladder cancer specimens vs normal bladder tissue | (28) | |
| CML | RTqPCR of 152 CML patients comparing advanced disease versus chronic phase CML | (108) | |
| RTqPCR of 90 patient specimens | (11) | ||
| Hepatocellular Carcinoma | IHC of 149 HCC vs 40 normal hepatic tissue specimens | (51) | |
| MDS | RTqPCR of low (19) and high (30) risk MDS vs AML (35) and normal patient blood samples (19) | (109) | |
| Microarray of 183 MDS vs 17 normal patient specimens | (58) | ||
| Pancreatic Cancer | Microarray of 130 PDAC vs 20 normal patient specimens | (24) |
For most solid tumors, a critical transition from localized to invasive or metastatic involves a transition in cell state in which epithelial features and lateral cell-cell connections are lost, replaced by mesenchymal features that support cell migration. This epithelial-mesenchymal transition (EMT) is a target of much study (49), and is closely linked not only to higher stage, but also to stem cell status and drug resistance (50). In 2014, studies of hepatocellular carcinomas (HCC) showed upregulation of MSI2 in invasive tumors of poor prognosis, and demonstrated that knockdown of MSI2 reduced HCC cell invasion and repressed expression of biomarkers of EMT: results echoed in analysis of clinical samples (51). This work was subsequently confirmed and extended in an independent study of hepatitis B virus (HBV)-dependent HCC (38). Importantly, two groups performed transcriptome and/or ribosomal profiling to identify genes regulated by the Musashi proteins, followed by extensive functional analysis (52–54). These studies demonstrated direct Musashi-dependent regulation of expression of genes in transforming growth factor beta (TGFβ) signaling pathways, linking Musashi for the first time to a major regulator of EMT transitions, independent of its role in regulation of Numb. They also demonstrated direct support by Musashi of EMT processes in cultured mammary tumor cells and in mammary growth in vivo (54), and in support of stem-like properties of hematopoietic stem cells (HSCs)(13).
The years 2015 and 2016 have been marked by a great expansion in studies that have both emphasized the central role of Musashi proteins as oncogenic drivers, and have elucidated mechanisms through the integration of transcriptomic and other profiling techniques, and use of sophisticated in vivo models. Two elegant 2015 studies expanded on the role of Musashi proteins in the intestinal epithelium, demonstrating that either MSI1 or MSI2 can act as an oncogenic driver and is induced by the loss of the tumor suppressor Apc, and identifying these proteins as regulators of the central PDK-AKT-mTORC1 signaling axis, which was defined as essential for Musashi-dependent transformation (37,55). In other work in solid tumors, MSI2 (but not MSI1) was identified as upregulated in, and a driver of, metastasis in non-small cell lung cancer (NSCLC), with activity in directly supporting expression of the TGFβ receptor TGFBR1, and TGFβ effector SMAD3, and indirectly downregulating tight junction–associated claudins to increase tumor cell invasion and metastasis (26). MSI2 targets were found to include many genes associated with cell adhesion and motility and Musashi proteins to promote cell migration in multiple cell types (45,56). Both MSI1 and MSI2 were identified as drivers of pancreatic cancers, responsible for the transition from pancreatic intraepithelial neoplasias (PanINs) to overt pancreatic adenocarcinoma, and required for aggressive disease (23,24). KLF4 was identified as a transcriptional repressor of MSI2 in pancreatic tumors, and loss of expression of KLF4 results in overexpression of MSI2, promoting PDAC progression and metastasis (24). MSI2 was found to promote aggressive breast cancer in part by supporting the protein expression of the estrogen receptor, ESR1, by indirect regulation of WNT and NOTCH in part by downregulation of Jagged1, and by induction of proliferin and tachykinin (TAC1) and inhibition of DKK3 (30,33,46,54,57), and to promote migratory and invasive behavior of bladder cancers in part by stimulating signaling through the JAK2/STAT3 signaling pathway(28). MSI1 was found to drive an extensive program of genes associated with proliferation and cell adhesion controls in glioblastoma (45) and BRD4, cMET, and HMGA2, and others, in pancreatic cancer (21).
In the context of liquid tumors, MSI2 induction of tetraspannin3 (Tspan3), a cell membrane protein that coordinates multiple signaling cascade, was found to be essential for pathogenesis of AML, with deletion of Tspan3 eliminating cell responsiveness to CXCL12/SDF1 chemokine signaling (36). MSI2 was for the first time linked to roles in supporting stem cell gene signatures and disease progression in myelodysplastic syndromes (MDS) (58). Overall, these and other studies have identified a large number of targets of Musashi-dependent expression, which besides those mentioned above include Hoxa9, Myc, Ikzf2 (53), NF-YA, a regulator of the proteasome (59), and Jagged1 (54). Musashi proteins have also recently been reported influence expression of lin28A, joining other studies connecting these proteins to control of cancer-associated micro-RNAs (miRs) (60,61). Factors of general interest for many cancers are summarized in Table 2.
Mechanisms of post-transcriptional regulation by Musashi proteins
Understanding how Musashi proteins regulate the expression of targets in cancer versus under physiological growth conditions, and potentially therapeutic targeting of Musashi proteins, depends on accurate understanding of the biochemical function of the protein structure. The Musashi proteins have been classified as members of class A/B heterogeneous nuclear ribonucleoproteins (hnRNPs) (2). For each Musashi protein, two N-terminal RNA recognition motifs (RRM) mediate Musashi binding to a group of target mRNAs (Figure 2A). Biochemical and structural studies have suggested that RRM1 contributes the majority of the binding energy and specificity, while RRM2 has a more supportive role. The RRMs are the most highly conserved part of the Musashi sequence, with 85% sequence identity between MSI1 and MSI2 in mice, and are highly conserved across species (2). In Musashi-targeted genes, RRM1 and RRM2 recognition sequences are found in a close proximity at the 3′-end of the mRNAs (7). MSI1 specifically recognizes motifs (Figure 2B, 3A) variously defined as [(G/A)U(n)AGU (n =1–3)] (3), r(GUAG) and r(UAG) (62), (UAG) (54,63) and other poly-U motifs(55). MSI2 has been suggested to preferentially interact with an ACCUUUUUAGAA′ motif, and other poly-U sequences(37), UAG motifs(63) and UAG containing motifs +/− additional flanking nucleotides(56). In general, it is thought that both proteins recognize similar sequences and thus likely regulate similar mRNA targets (2,62). Several groups have performed high throughput studies of MSI1 or MSI2 binding targets, with results confirming partial but incomplete overlap (13,23,41,53,54,56,64) (Table 3).
Figure 2. Musashi protein interactions.
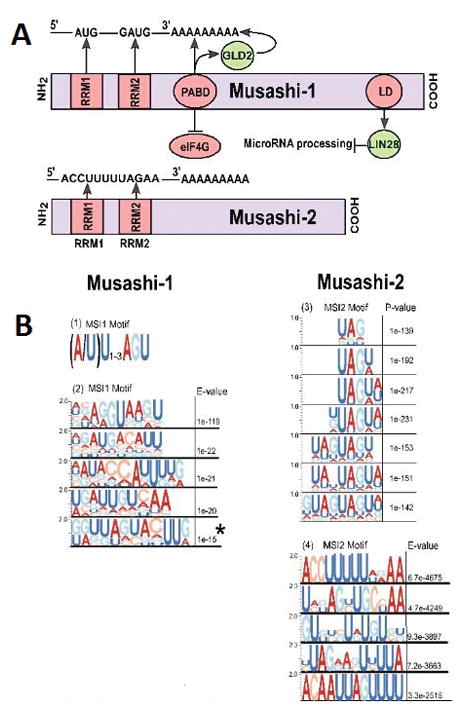
(A) Schematic representation of Musashi-1 and 2 molecular interaction domains for interactions with RNA and with RNA-binding proteins. RRM - RNA recognition motif; LD, LIN28 interaction domain; PABD - Poly-A binding domain. (B) Summary of recognition motifs identified for MSI1 in 1) Imai T et al (3), 2) Li et al (55) or MSI2 in 3) Wang S et al (37) and 4) Bennet CG et al (56). Sequence motifs are reprinted from original publications by permission of the authors and publishers. Motifs are represented in the Selex format, with size of letter reflecting preference for indicated nucleotides. (Adapted from Imai T et al. “The neural RNA-binding protein Musashi1 translationally regulates mammalian numb gene expression by interacting with its mRNA.” Mollecular and Cellular Bioliology, 2001 June; Volume 21, Issue 12. Pages 3888–900; Copyright [2001] by permission from American Society for Microbiology. Adapted from Cell Reports, Volume 13, Issue 11. Li N et al. “The Msi Family of RNA-Binding Proteins Function Redundantly as Intestinal Oncoproteins.” 2015 December 22; Pages No. 2440–55, Copyright [2015], with permission from Elsevier. Adapted by permission from Macmillan Publishers Ltd on behalf of Cancer Research UK: Nature Communications (37), copyright [2015]. Adapted from Bennett CG et al. “Genome-wide analysis of Musashi-2 targets reveals novel functions in governing epithelial cell migration.” Nucleic Acids Research. 2016 May 5; Volume 44, Issue 8, pages 3788–800, Copyright [2016] by permission of Oxford University Press.)
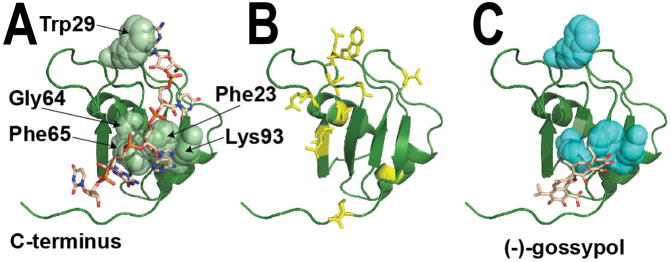
Figure 3. The NMR structure of RRM1 from mouse Msi1.
(A) The structure of this RRM domain (indicated as a green ribbon) was determined in complex with a short segment of cognate RNA (sequence GUAGU, indicated as brown sticks), PDB ID 2rs2 (62). The RRM domain is a conserved fold that packs two α-helices against one face of a four-stranded β-sheet. As with other “canonical” RRMs (88), the opposite face of this β-sheet is then used to bind a single-stranded segment of RNA, as shown. The residues with the largest differences in NMR HSQC peak intensities upon addition of RNA include Phe23, Trp29, Gly64, Phe65, and Lys93 (shown in spheres), confirming the role of these residues in Msi1 binding to RNA (76,78). Structures of other proteins containing tandem RRM domains often show these domains in an arrangement that presents a single deep RNA-binding cleft (88,89). In this Msi1-RRM1 structure, the C-terminus of the RRM1 domain makes close contact with the RNA beyond simply the canonical interactions on the β-sheet. Thus, the structural basis for Musashi-RNA association may also be strongly affected by interactions between the two RRM domains that are not captured in the complex shown here. (B) Upon addition of oleic acid to the Musashi RRM domain, the NMR HSQC peak intensity for residues throughout the structure exhibit diminished intensity (cyan sticks), suggesting extensive structural changes consistent with allosteric inhibition (76). (C) Upon addition of (−)-gossypol to the RRM domain, the NMR HSQC peak intensity is diminished primarily for residues involved in RNA binding (cyan spheres), suggesting direct inhibition and supporting the docked model shown (78).
Table 3. Summary of high throughput experiments identifying direct MSI1 and MSI2 targets.
HITS-CLIP, high-throughput sequencing of RNA isolated by crosslinking immunoprecipitation; CLIP-seq, cross-linking immunoprecipitation; RIP-ChIP, RNA-binding protein immunoprecipitation-microarray chip profiling; RNA-IP, RNA immunoprecipitation; Ribo-Seq, ribosome profiling; RIP-qPCR, RNA immunoprecipitation-quantitative polymerase chain reaction.
| Gene | Reference | Experiment | Validated targets | Total genes identified | Cell type |
|---|---|---|---|---|---|
| MSI-1/2 | (23) | RIP-qPCR, CLIP-seq | c-MET, BRD4, HMGA2 | 73329 MSI1 tags | Mouse pancreatic adenocarcinoma |
| (55) | CLIP-Seq | 97 | Mouse intestinal epithelial cells | ||
| Musashi-1 | (113) | mRNA co-association assay | Mos (c-Mos) | N/A | Xenopus laevis oocytes |
| (64) | Microarray analysis, Rip-Chip | 64 genes | HEK 293T | ||
| (41) | Microarray analysis, RIP-chip | N/A | 52 genes | Daoy cells | |
| (54) | Ribo-Seq | Jagged1 | Unstated | Mouse neural stem cells | |
| (114) | Immunoprecipitation (RIP) assay | sima, HIF-1a | Unstated | Schneider (S2R+) Drosophila cells | |
| (45) | iCLIP, RNA-seq | 86 | 9319 | Glioma cells | |
| Musashi-2 | (13) | Microarray, HITS-CLIP | 48 genes | 668 gene | Hematopoietic stem cells |
| (53) | Microarray, HITS-CLIP | 11 genes | 283 genes | Mouse Lin−Sca+Kit+ Leukemia stem cells | |
| (37) | CLIP-Seq | PTEN, Lrig1, Bmpr1 | Unstated | Mouse intestinal epithelial cells | |
| (56) | HITS-CLIP, Ribo-seq | 88 genes | Mouse keratinocytes |
The C-terminal region of Musashi proteins harbor additional protein-protein interaction sequences, which have been best defined through studies of MSI1, and allow the proteins to both induce and repress protein translation (Figure 2A) (21,62,65–67). A poly-A binding protein (PABP)-interaction domain immediately C-terminal to RRM2 disrupts the ability of PABP to interact with the eukaryotic translation initiation factor 4 gamma (eIF4G) binding, and thus represses initiation of translation of a subset of Musashi targets. Separately, studies in Xenopus and mouse models have identified binding between the C-terminal domain of MSI1 and the poly-(A) polymerase GLD2 (PAPD4) (68–70). This results in increased polyadenylation, mRNA stabilization, and higher protein expression in GLD2-rich oocytes. In contrast, overexpression of either Xenopus or mouse MSI1 in the context of GLD2-depleted mouse cells led to translational inhibition. Connecting Musashi proteins to control of miRS, discrete LIN28-binding motif has been identified in the C-terminus of MSI1 (9): while this motif is absent in MSI2, recent studies have nevertheless shown biological activity of MSI2 in regulating Lin28A (23,61). Additionally, both MSI1 and MSI2 have been described to regulate alternative splicing in mouse retinal photoreceptor and neural stem cells, with mechanisms still to be exactly defined (54,71). Understanding how the protein structure and signaling downstream of MSI1 and MSI2 are linked to their function in different cellular contexts remains an important area for future work.
Perhaps because the dual ability to stimulate and repress translation, and differences in the abundance of as yet undefined additional partner proteins, the activity of Musashi proteins to regulate specific mRNAs differs depending on cellular context. For example, several groups reported that both MSI1 and MSI2 bound NUMB mRNA in vivo and in vitro (3,72–74). However, while Musashi proteins repressed NUMB consistently in CNS tumors and some hematologic malignancies, HSCs lacking Msi2 have unchanged levels of the Numb protein(13). Katz et al. did not identify significant MSI1-dependent changes in NUMB RNA expression by ribosome profiling in neural stem cells upon MSI1 manipulation (54), and no consistent pattern of change in NUMB protein levels was detected upon MSI2 overexpression or depletion in human and murine NSCLC cells (26).
Musashi proteins in tumor responses to chemotherapy and radiation therapy
As expected for proteins shown to control stem cell identity and EMT, overexpression of Musashi proteins has increasingly been linked to therapeutic resistance in cancer. As some examples, elevated expression of MSI2 induced resistance to paclitaxel in ovarian cancer cells in vitro (27). MSI2 silencing in AML cells sensitized these cells to treatment with daunorubicin, accompanied by induction of cell cycle arrest and induction of apoptosis, mediated by downregulation of BCL2 and upregulation of BAX (35). MSI1 was recently described as a regulator of response to radiation therapy in glioblastoma. In this study, depletion of MSI1 led to decreased expression of the catalytic subunit of DNA-PK. This resulted in an increase in DNA damage due to reduced capacity for non-homologous end-joining (NHEJ)-based repair (75). These and other studies have increased interest in regulating the expression and biological activities of the Musashi proteins, to potentially achieve therapeutic benefit.
Musashi proteins as therapeutic targets in cancer
The important role of both MSI1 and MSI2 in various cancers has motivated three independent groups to attempt to develop small-molecule inhibitors of these proteins (76–78). All three groups used similar fluorescence polarization (FP) competition assays to search for compounds that would disrupt the binding of Musashi proteins to a short fluorescein-labeled RNA, and all three identified compounds in pilot screens that inhibit RNA-binding; the compounds themselves are quite distinct, however, reflecting the composition of the screening libraries selected by each group.
In screening against MSI1, Clingman and colleagues (76) used a traditional compound library augmented by a set of known bioactive compounds. While the traditional library did not yield useful hits, the latter collection yielded oleic acid as an initial hit. Further studies showed that several other ω-9 monounsaturated fatty acids also inhibit Msi1 binding to RNA, with ~1 μM Ki values. Oleic acid was shown to bind the Musashi RRM (Figure 3B) and to inhibit proliferation of a cell line expressing Msi1, though the mechanism of action was not fully established. Most interestingly, these studies pointed to potential feedback between Msi1 activity and fatty acid biosynthesis, potentially positioning these compounds as useful probes to discover new biology.
Minuesa and colleagues performed parallel small pilot screens of a 6208 compound library against Msi1 and Msi2 (77). Of the small number of compounds that were identified as hits in one screen, most did not emerge from the other, suggesting either different structural requirements for disrupting MSI1 and MSI2 RNA binding, or issues with false discovery rate. However, three dual-activity compounds were ultimately validated through secondary screen, each with ~5 μM IC50 values. The activity of these compounds in cells has not yet been explored.
Lan and colleagues used only the RRM1 of MSI1 to carry out their screen of the NCI diversity set of 2000 compounds, including natural products and approved oncology drugs (78). Among the top hits in this screen was (−)-gossypol, a natural product extracted from cottonseed that has already completed Phase IIb clinical trials for prostate cancer. This compound also inhibited RNA binding by full-length MSI1, with a Ki value ~500 nM, and via a direct interaction with the RNA-binding groove of RRM1 (Figure 3C). (−)-gossypol was shown to reduce Notch/Wnt signaling in colon cancer cell lines, and suppress tumor growth in a mouse xenograft model, which may be relevant to its control of MSI1 activity. The basis for earlier clinical development of (−)-gossypol, however, was primarily for its known inhibition of the anti-apoptotic Bcl-family proteins (Bcl-xL, Bcl-2, and Mcl-1) (79,80); additional targets may exist, confounding efforts to identify Musashi-specific activities. Disappointingly, (−)-gossypol showed only minimal activity in combination with prednisone against prostate cancer and no activity in combination with docetaxel in NSCLC in Phase I/II studies (81,82); however, explicit optimization of (−)-gossypol to target Musashi may provide new avenues for development of this compound.
While attempts to develop Musashi inhibitors are thus still in the early stages, these three studies support the druggability of Msi1/Msi2 via small molecules. The potential utility of Msi1 as a therapeutic target, meanwhile, is strongly supported by recent proof-of-principle studies demonstrating that antisense oligonucleotides targeting Musashi effectively block tumor growth in preclinical models of pancreatic cancer (23). Collectively then, these studies position the Musashi proteins as tractable and valuable therapeutic targets for further drug discovery efforts.
Conclusions and Perspectives
The Musashi proteins are now firmly established as modulators of oncogenic initiation, progression, and therapeutic response, for many clinically intractable cancers. While inhibitors of the Musashi proteins are still years from the clinic, such agents may prove valuable in cancer therapy, or as regulators of responses to chemotherapy, targeted therapy and potentially radiation and other treatment modalities. Technical approaches such as PAR-CLIP, which utilizes a photoactivatable nucleoside for more efficient and specific crosslinking, may help provide insight into specific direct targets regulated by MSI proteins(83). In the interim, the Musashi proteins serve as valuable indicators of stem cell reservoirs within tumors, and can be used to gain sophisticated insights into the activity of these populations: for example, a MSI2-based reporter system has recently been useful as a probe for in vivo imaging of HSC dynamics and movement (84). In addition, considering the expression and activity of MSI1 and MSI2 in individual tumors may yield insight into the relationship between transcriptomic signatures and the actual protein signaling networks operative in tumor cells. For example, in comparing two lung tumors with comparable SMAD3 mRNA expression, the tumor that also has high MSI2 mRNA expression is perhaps more likely to have abundant SMAD3 protein available to support metastasis than is the tumor lacking MSI2. As with the epigenetic regulatory machinery, proteins such as MSI1 and MSI2 that operate post-transcriptionally are only now emerging as valuable hubs for broad-based control of oncogenic signaling networks. Finally, beyond cancer, manipulation of the Musashi proteins may be useful in other clinical applications. For example, it may be useful to transiently enhance Musashi function when enlarging stem or progenitor pools for therapeutic purposes, such as bone marrow replacement. As other examples, MSI1 is induced as part of the damage response process in models for arthritis(85), elevated in atherosclerotic plaques(86), and critical for survival of photoreceptors and appropriate cell function in the retina (71,87). Clearly, more work is needed to refine the role of MSI proteins in these and other tissues, with the goal of ameliorating pathologic conditions. The next several years promise to be fruitful for research discovery.
Acknowledgments
Grants Support The authors were supported by R21CA191425 from the NIH and AHEPA Foundation funding (to EAG), a DOD Career Development Award LC140074 (to YB), R01CA178831 (to JK) and NCI Core Grant P30 CA006927 (to Fox Chase Cancer Center).
We thank Alexander Deneka for technical assistance with the EndNote and Inkscape software.
Footnotes
Author Contributions. All named authors took part in researching information and writing the manuscript. Figures were generated by AK and JK.
Conflict of Interest The authors declare no conflict of interests.
References
- 1.Nakamura M, Okano H, Blendy JA, Montell C. Musashi, a neural RNA-binding protein required for Drosophila adult external sensory organ development. Neuron. 1994;13(1):67–81. doi: 10.1016/0896-6273(94)90460-x. [DOI] [PubMed] [Google Scholar]
- 2.Sakakibara S, Nakamura Y, Satoh H, Okano H. Rna-binding protein Musashi2: developmentally regulated expression in neural precursor cells and subpopulations of neurons in mammalian CNS. J Neurosci. 2001;21(20):8091–107. doi: 10.1523/JNEUROSCI.21-20-08091.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Imai T, Tokunaga A, Yoshida T, Hashimoto M, Mikoshiba K, Weinmaster G, et al. The neural RNA-binding protein Musashi1 translationally regulates mammalian numb gene expression by interacting with its mRNA. Mol Cell Biol. 2001;21(12):3888–900. doi: 10.1128/MCB.21.12.3888-3900.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Siddall NA, McLaughlin EA, Marriner NL, Hime GR. The RNA-binding protein Musashi is required intrinsically to maintain stem cell identity. Proceedings of the National Academy of Sciences of the United States of America. 2006;103(22):8402–7. doi: 10.1073/pnas.0600906103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sutherland JM, Fraser BA, Sobinoff AP, Pye VJ, Davidson TL, Siddall NA, et al. Developmental expression of Musashi-1 and Musashi-2 RNA-binding proteins during spermatogenesis: analysis of the deleterious effects of dysregulated expression. Biol Reprod. 2014;90(5):1–12. doi: 10.1095/biolreprod.113.115261. [DOI] [PubMed] [Google Scholar]
- 6.Sutherland JM, Sobinoff AP, Fraser BA, Redgrove KA, Davidson TL, Siddall NA, et al. RNA binding protein Musashi-1 directly targets Msi2 and Erh during early testis germ cell development and interacts with IPO5 upon translocation to the nucleus. FASEB J. 2015;29(7):2759–68. doi: 10.1096/fj.14-265868. [DOI] [PubMed] [Google Scholar]
- 7.Sakakibara S, Imai T, Hamaguchi K, Okabe M, Aruga J, Nakajima K, et al. Mouse-Musashi-1, a neural RNA-Binding protein highly enriched in the mammalian CNS stem cell. Developmental Biology. 1996;176(2):230–42. doi: 10.1006/dbio.1996.0130. [DOI] [PubMed] [Google Scholar]
- 8.Good P, Yoda A, Sakakibara S, Yamamoto A, Imai T, Sawa H, et al. The human Musashi homolog 1 (MSI1) gene encoding the homologue of Musashi/Nrp-1, a neural RNA-binding protein putatively expressed in CNS stem cells and neural progenitor cells. Genomics. 1998;52(3):382–4. doi: 10.1006/geno.1998.5456. [DOI] [PubMed] [Google Scholar]
- 9.Kawahara H, Okada Y, Imai T, Iwanami A, Mischel PS, Okano H. Musashi1 cooperates in abnormal cell lineage protein 28 (Lin28)-mediated let-7 family microRNA biogenesis in early neural differentiation. J Biol Chem. 2011;286(18):16121–30. doi: 10.1074/jbc.M110.199166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shibata S, Umei M, Kawahara H, Yano M, Makino S, Okano H. Characterization of the RNA-binding protein Musashi1 in zebrafish. Brain Res. 2012;1462:162–73. doi: 10.1016/j.brainres.2012.01.068. [DOI] [PubMed] [Google Scholar]
- 11.Ito T, Kwon HY, Zimdahl B, Congdon KL, Blum J, Lento WE, et al. Regulation of myeloid leukaemia by the cell-fate determinant Musashi. Nature. 2010;466(7307):765–8. doi: 10.1038/nature09171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kharas MG, Lengner CJ, Al-Shahrour F, Bullinger L, Ball B, Zaidi S, et al. Musashi-2 regulates normal hematopoiesis and promotes aggressive myeloid leukemia. Nature Medicine. 2010;16(8):903–8. doi: 10.1038/nm.2187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Park SM, Deering RP, Lu Y, Tivnan P, Lianoglou S, Al-Shahrour F, et al. Musashi-2 controls cell fate, lineage bias, and TGF-beta signaling in HSCs. J Exp Med. 2014;211(1):71–87. doi: 10.1084/jem.20130736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sutherland JM, McLaughlin EA, Hime GR, Siddall NA. The Musashi family of RNA binding proteins: master regulators of multiple stem cell populations. Adv Exp Med Biol. 2013;786:233–45. doi: 10.1007/978-94-007-6621-1_13. [DOI] [PubMed] [Google Scholar]
- 15.Sutherland JM, Sobinoff AP, Gunter KM, Fraser BA, Pye V, Bernstein IR, et al. Knockout of RNA Binding Protein MSI2 Impairs Follicle Development in the Mouse Ovary: Characterization of MSI1 and MSI2 during Folliculogenesis. Biomolecules. 2015;5(3):1228–44. doi: 10.3390/biom5031228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sakakibara S, Nakamura Y, Yoshida T, Shibata S, Koike M, Takano H, et al. RNA-binding protein Musashi family: Roles for CNS stem cells and a subpopulation of ependymal cells revealed by targeted disruption and antisense ablation. Proceedings of the National Academy of Sciences of the United States of America. 2002;99(23):15194–9. doi: 10.1073/pnas.232087499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kanemura Y, Mori K, Sakakibara S, Fujikawa H, Hayashi H, Nakano A, et al. Musashi1, an evolutionarily conserved neural RNA-binding protein, is a versatile marker of human glioma cells in determining their cellular origin, malignancy, and proliferative activity. Differentiation. 2001;68(2–3):141–52. doi: 10.1046/j.1432-0436.2001.680208.x. [DOI] [PubMed] [Google Scholar]
- 18.Hemmati HD, Nakano I, Lazareff JA, Masterman-Smith M, Geschwind DH, Bronner-Fraser M, et al. Cancerous stem cells can arise from pediatric brain tumors. Proc Natl Acad Sci U S A. 2003;100(25):15178–83. doi: 10.1073/pnas.2036535100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shu HJ, Saito T, Watanabe H, Ito JI, Takeda H, Okano H, et al. Expression of the Musashi1 gene encoding the RNA-binding protein in human hepatoma cell lines. Biochem Biophys Res Commun. 2002;293(1):150–4. doi: 10.1016/S0006-291X(02)00175-4. [DOI] [PubMed] [Google Scholar]
- 20.Barbouti A, Hoglund M, Johansson B, Lassen C, Nilsson PG, Hagemeijer A, et al. A novel gene, MSI2, encoding a putative RNA-binding protein is recurrently rearranged at disease progression of chronic myeloid leukemia and forms a fusion gene with HOXA9 as a result of the cryptic t(7;17)(p15;q23) Cancer Res. 2003;63(6):1202–6. [PubMed] [Google Scholar]
- 21.Fox RG, Park FD, Koechlein CS, Kritzik M, Reya T. Musashi signaling in stem cells and cancer. Annu Rev Cell Dev Biol. 2015;31:249–67. doi: 10.1146/annurev-cellbio-100814-125446. [DOI] [PubMed] [Google Scholar]
- 22.Gunter KM, McLaughlin EA. Translational control in germ cell development: A role for the RNA-binding proteins Musashi-1 and Musashi-2. IUBMB Life. 2011;63(9):678–85. doi: 10.1002/iub.499. [DOI] [PubMed] [Google Scholar]
- 23.Fox RG, Lytle NK, Jaquish DV, Park FD, Ito T, Bajaj J, et al. Image-based detection and targeting of therapy resistance in pancreatic adenocarcinoma. Nature. 2016;534(7607):407–11. doi: 10.1038/nature17988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guo K, Cui J, Quan M, Xie D, Jia Z, Wei D, et al. A novel KLF4-MSI2 signaling pathway regulates growth and metastasis of pancreatic cancer. Clin Cancer Res. 2016 doi: 10.1158/1078-0432.CCR-16-1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gao C, Han C, Yu Q, Guan Y, Li N, Zhou J, et al. Downregulation of Msi1 suppresses the growth of human colon cancer by targeting p21cip1. Int J Oncol. 2015;46(2):732–40. doi: 10.3892/ijo.2014.2749. [DOI] [PubMed] [Google Scholar]
- 26.Kudinov AE, Deneka A, Nikonova AS, Beck TN, Ahn YH, Liu X, et al. Musashi-2 (MSI2) supports TGF-beta signaling and inhibits claudins to promote non-small cell lung cancer (NSCLC) metastasis. Proc Natl Acad Sci U S A. 2016;113(25):6955–60. doi: 10.1073/pnas.1513616113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee J, An S, Choi YM, Lee J, Ahn KJ, Lee JH, et al. Musashi-2 is a novel regulator of paclitaxel sensitivity in ovarian cancer cells. Int J Oncol. 2016;5(49):1945–52. doi: 10.3892/ijo.2016.3683. [DOI] [PubMed] [Google Scholar]
- 28.Yang C, Zhang W, Wang L, Kazobinka G, Han X, Li B, et al. Musashi-2 promotes migration and invasion in bladder cancer via activation of the JAK2/STAT3 pathway. Lab Invest. 2016;96(9):950–8. doi: 10.1038/labinvest.2016.71. [DOI] [PubMed] [Google Scholar]
- 29.Wang XY, Penalva LOF, Yuan HY, Linnoila RI, Lu JC, Okano H, et al. Musashi1 regulates breast tumor cell proliferation and is a prognostic indicator of poor survival. Molecular Cancer. 2010;9:221–233. doi: 10.1186/1476-4598-9-221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kang MH, Jeong KJ, Kim WY, Lee HJ, Gong G, Suh N, et al. Musashi RNA-binding protein 2 regulates estrogen receptor 1 function in breast cancer. Oncogene. 2016:1–8. doi: 10.1038/onc.2016.327. [DOI] [PubMed] [Google Scholar]
- 31.Oskarsson T, Acharyya S, Zhang XH, Vanharanta S, Tavazoie SF, Morris PG, et al. Breast cancer cells produce tenascin C as a metastatic niche component to colonize the lungs. Nat Med. 2011;17(7):867–74. doi: 10.1038/nm.2379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kagara N, Huynh KT, Kuo C, Okano H, Sim MS, Elashoff D, et al. Epigenetic regulation of cancer stem cell genes in triple-negative breast cancer. Am J Pathol. 2012;181(1):257–67. doi: 10.1016/j.ajpath.2012.03.019. [DOI] [PubMed] [Google Scholar]
- 33.Wang XY, Penalva LO, Yuan H, Linnoila RI, Lu J, Okano H, et al. Musashi1 regulates breast tumor cell proliferation and is a prognostic indicator of poor survival. Molecular cancer. 2010;9:221–33. doi: 10.1186/1476-4598-9-221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Griner LN, Reuther GW. Aggressive myeloid leukemia formation is directed by the Musashi 2/Numb pathway. Cancer Biol Ther. 2010;10(10):979–82. doi: 10.4161/cbt.10.10.14010. [DOI] [PubMed] [Google Scholar]
- 35.Han Y, Ye A, Zhang Y, Cai Z, Wang W, Sun L, et al. Musashi-2 Silencing Exerts Potent Activity against Acute Myeloid Leukemia and Enhances Chemosensitivity to Daunorubicin. PLoS One. 2015;10(8):e0136484. doi: 10.1371/journal.pone.0136484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kwon HY, Bajaj J, Ito T, Blevins A, Konuma T, Weeks J, et al. Tetraspanin 3 Is Required for the Development and Propagation of Acute Myelogenous Leukemia. Cell Stem Cell. 2015;17(2):152–64. doi: 10.1016/j.stem.2015.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang S, Li N, Yousefi M, Nakauka-Ddamba A, Li F, Parada K, et al. Transformation of the intestinal epithelium by the MSI2 RNA-binding protein. Nat Commun. 2015;6:6517. doi: 10.1038/ncomms7517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang MH, Qin SY, Zhang SG, Li GX, Yu ZH, Wang K, et al. Musashi-2 promotes hepatitis Bvirus related hepatocellular carcinoma progression via the Wnt/beta-catenin pathway. Am J Cancer Res. 2015;5(3):1089–100. [PMC free article] [PubMed] [Google Scholar]
- 39.Rath P, Lal B, Ajala O, Li Y, Xia S, Kim J, et al. In Vivo c-Met Pathway Inhibition Depletes Human Glioma Xenografts of Tumor-Propagating Stem-Like Cells. Transl Oncol. 2013;6(2):104–11. doi: 10.1593/tlo.13127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pasto A, Serafin V, Pilotto G, Lago C, Bellio C, Trusolino L, et al. NOTCH3 signaling regulates MUSASHI-1 expression in metastatic colorectal cancer cells. Cancer Res. 2014;74(7):2106–18. doi: 10.1158/0008-5472.CAN-13-2022. [DOI] [PubMed] [Google Scholar]
- 41.Vo DT, Subramaniam D, Remke M, Burton TL, Uren PJ, Gelfond JA, et al. The RNA-binding protein Musashi1 affects medulloblastoma growth via a network of cancer-related genes and is an indicator of poor prognosis. Am J Pathol. 2012;181(5):1762–72. doi: 10.1016/j.ajpath.2012.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Smith AR, Marquez RT, Tsao WC, Pathak S, Roy A, Ping J, et al. Tumor suppressive microRNA-137 negatively regulates Musashi-1 and colorectal cancer progression. Oncotarget. 2015;6(14):12558–73. doi: 10.18632/oncotarget.3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fujiwara T, Zhou J, Ye S, Zhao H. RNA-binding protein Musashi2 induced by RANKL is critical for osteoclast survival. Cell Death Dis. 2016;7:e2300. doi: 10.1038/cddis.2016.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rezza A, Skah S, Roche C, Nadjar J, Samarut J, Plateroti M. The overexpression of the putative gut stem cell marker Musashi-1 induces tumorigenesis through Wnt and Notch activation. J Cell Sci. 2010;123(Pt 19):3256–65. doi: 10.1242/jcs.065284. [DOI] [PubMed] [Google Scholar]
- 45.Uren PJ, Vo DT, de Araujo PR, Potschke R, Burns SC, Bahrami-Samani E, et al. RNA-Binding Protein Musashi1 Is a Central Regulator of Adhesion Pathways in Glioblastoma. Mol Cell Biol. 2015;35(17):2965–78. doi: 10.1128/MCB.00410-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nahas GR, Murthy RG, Patel SA, Ganta T, Greco SJ, Rameshwar P. The RNA-binding protein Musashi 1 stabilizes the oncotachykinin 1 mRNA in breast cancer cells to promote cell growth. FASEB J. 2016;30(1):149–59. doi: 10.1096/fj.15-278770. [DOI] [PubMed] [Google Scholar]
- 47.Cambuli FM, Correa BR, Rezza A, Burns SC, Qiao M, Uren PJ, et al. A Mouse Model of Targeted Musashi1 Expression in Whole Intestinal Epithelium Suggests Regulatory Roles in Cell Cycle and Stemness. Stem cells. 2015;33(12):3621–34. doi: 10.1002/stem.2202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Maria Cambuli F, Rezza A, Nadjar J, Plateroti M. Brief report: musashi1-eGFP mice, a new tool for differential isolation of the intestinal stem cell populations. Stem cells. 2013;31(10):2273–8. doi: 10.1002/stem.1428. [DOI] [PubMed] [Google Scholar]
- 49.Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139(5):871–90. doi: 10.1016/j.cell.2009.11.007. [DOI] [PubMed] [Google Scholar]
- 50.Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene. 2010;29(34):4741–51. doi: 10.1038/onc.2010.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.He L, Zhou X, Qu C, Hu L, Tang Y, Zhang Q, et al. Musashi2 predicts poor prognosis and invasion in hepatocellular carcinoma by driving epithelial-mesenchymal transition. J Cell Mol Med. 2014;18(1):49–58. doi: 10.1111/jcmm.12158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Park CY, Min KN, Son JY, Park SY, Nam JS, Kim DK, et al. An novel inhibitor of TGF-beta type I receptor, IN-1130, blocks breast cancer lung metastasis through inhibition of epithelial-mesenchymal transition. Cancer Lett. 2014;351(1):72–80. doi: 10.1016/j.canlet.2014.05.006. [DOI] [PubMed] [Google Scholar]
- 53.Park SM, Gonen M, Vu L, Minuesa G, Tivnan P, Barlowe TS, et al. Musashi2 sustains the mixed-lineage leukemia-driven stem cell regulatory program. J Clin Invest. 2015;125(3):1286–98. doi: 10.1172/JCI78440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Katz Y, Li F, Lambert NJ, Sokol ES, Tam WL, Cheng AW, et al. Musashi proteins are post-transcriptional regulators of the epithelial-luminal cell state. Elife. 2014;3:e03915. doi: 10.7554/eLife.03915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Li N, Yousefi M, Nakauka-Ddamba A, Li F, Vandivier L, Parada K, et al. The Msi Family of RNA-Binding Proteins Function Redundantly as Intestinal Oncoproteins. Cell Rep. 2015;13(11):2440–55. doi: 10.1016/j.celrep.2015.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bennett CG, Riemondy K, Chapnick DA, Bunker E, Liu X, Kuersten S, et al. Genome-wide analysis of Musashi-2 targets reveals novel functions in governing epithelial cell migration. Nucleic Acids Res. 2016;44(8):3788–800. doi: 10.1093/nar/gkw207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang XY, Yin Y, Yuan H, Sakamaki T, Okano H, Glazer RI. Musashi1 modulates mammary progenitor cell expansion through proliferin-mediated activation of the Wnt and Notch pathways. Mol Cell Biol. 2008;28(11):3589–99. doi: 10.1128/MCB.00040-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Taggart J, Ho TC, Amin E, Xu H, Barlowe TS, Perez AR, et al. MSI2 is required for maintaining activated myelodysplastic syndrome stem cells. Nat Commun. 2016;7:10739. doi: 10.1038/ncomms10739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lagadec C, Vlashi E, Frohnen P, Alhiyari Y, Chan M, Pajonk F. The RNA-binding protein Musashi-1 regulates proteasome subunit expression in breast cancer- and glioma-initiating cells. Stem cells. 2014;32(1):135–44. doi: 10.1002/stem.1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Choudhury NR, de Lima Alves F, de Andres-Aguayo L, Graf T, Caceres JF, Rappsilber J, et al. Tissue-specific control of brain-enriched miR-7 biogenesis. Genes Dev. 2013;27(1):24–38. doi: 10.1101/gad.199190.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fang T, Lv H, Wu F, Wang C, Li T, Lv G, et al. Musashi 2 contributes to the stemness and chemoresistance of liver cancer stem cells via LIN28A activation. Cancer Lett. 2016;384:50–9. doi: 10.1016/j.canlet.2016.10.007. [DOI] [PubMed] [Google Scholar]
- 62.Ohyama T, Nagata T, Tsuda K, Kobayashi N, Imai T, Okano H, et al. Structure of Musashi1 in a complex with target RNA: the role of aromatic stacking interactions. Nucleic Acids Res. 2012;40(7):3218–31. doi: 10.1093/nar/gkr1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zearfoss NR, Deveau LM, Clingman CC, Schmidt E, Johnson ES, Massi F, et al. A conserved three-nucleotide core motif defines Musashi RNA binding specificity. J Biol Chem. 2014;289(51):35530–41. doi: 10.1074/jbc.M114.597112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.de Sousa Abreu R, Sanchez-Diaz PC, Vogel C, Burns SC, Ko D, Burton TL, et al. Genomic analyses of musashi1 downstream targets show a strong association with cancer-related processes. J Biol Chem. 2009;284(18):12125–35. doi: 10.1074/jbc.M809605200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Miyanoiri Y, Kobayashi H, Imai T, Watanabe M, Nagata T, Uesugi S, et al. Origin of higher affinity to RNA of the N-terminal RNA-binding domain than that of the C-terminal one of a mouse neural protein, musashi1, as revealed by comparison of their structures, modes of interaction, surface electrostatic potentials, and backbone dynamics. J Biol Chem. 2003;278(42):41309–15. doi: 10.1074/jbc.M306210200. [DOI] [PubMed] [Google Scholar]
- 66.Nagata T, Kurihara Y, Matsuda G, Saeki J, Kohno T, Yanagida Y, et al. Structure and interactions with RNA of the N-terminal UUAG-specific RNA-binding domain of hnRNP D0. J Mol Biol. 1999;287(2):221–37. doi: 10.1006/jmbi.1999.2616. [DOI] [PubMed] [Google Scholar]
- 67.Okabe M, Imai T, Kurusu M, Hiromi Y, Okano H. Translational repression determines a neuronal potential in Drosophila asymmetric cell division. Nature. 2001;411(6833):94–8. doi: 10.1038/35075094. [DOI] [PubMed] [Google Scholar]
- 68.Cragle C, MacNicol AM. Musashi protein-directed translational activation of target mRNAs is mediated by the poly(A) polymerase, germ line development defective-2. J Biol Chem. 2014;289(20):14239–51. doi: 10.1074/jbc.M114.548271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.MacNicol MC, Cragle CE, MacNicol AM. Context-dependent regulation of Musashi-mediated mRNA translation and cell cycle regulation. Cell Cycle. 2011;10(1):39–44. doi: 10.4161/cc.10.1.14388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rutledge CE, Lau HT, Mangan H, Hardy LL, Sunnotel O, Guo F, et al. Efficient translation of Dnmt1 requires cytoplasmic polyadenylation and Musashi binding elements. PLoS One. 2014;9(2):e88385. doi: 10.1371/journal.pone.0088385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Murphy D, Cieply B, Carstens R, Ramamurthy V, Stoilov P. The Musashi 1 Controls the Splicing of Photoreceptor-Specific Exons in the Vertebrate Retina. PLoS genetics. 2016;12(8):e1006256. doi: 10.1371/journal.pgen.1006256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Okano H, Imai T, Okabe M. Musashi: a translational regulator of cell fate. J Cell Sci. 2002;115(Pt 7):1355–9. doi: 10.1242/jcs.115.7.1355. [DOI] [PubMed] [Google Scholar]
- 73.Takahashi T, Suzuki H, Imai T, Shibata S, Tabuchi Y, Tsuchimoto K, et al. Musashi-1 post-transcriptionally enhances phosphotyrosine-binding domain-containing m-Numb protein expression in regenerating gastric mucosa. PLoS One. 2013;8(1):e53540. doi: 10.1371/journal.pone.0053540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Jadhav S, Ajay AK, Trivedi P, Seematti J, Pellegrini K, Craciun F, et al. RNA-binding Protein Musashi Homologue 1 Regulates Kidney Fibrosis by Translational Inhibition of p21 and Numb mRNA. J Biol Chem. 2016;291(27):14085–94. doi: 10.1074/jbc.M115.713289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.de Araujo PR, Gorthi A, da Silva AE, Tonapi SS, Vo DT, Burns SC, et al. Musashi1 Impacts Radio-Resistance in Glioblastoma by Controlling DNA-Protein Kinase Catalytic Subunit. Am J Pathol. 2016;186(9):2271–8. doi: 10.1016/j.ajpath.2016.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Clingman CC, Deveau LM, Hay SA, Genga RM, Shandilya SM, Massi F, et al. Allosteric inhibition of a stem cell RNA-binding protein by an intermediary metabolite. Elife. 2014;3:1–26. doi: 10.7554/eLife.02848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Minuesa G, Antczak C, Shum D, Radu C, Bhinder B, Li Y, et al. A 1536-well fluorescence polarization assay to screen for modulators of the MUSASHI family of RNA-binding proteins. Comb Chem High Throughput Screen. 2014;17(7):596–609. doi: 10.2174/1386207317666140609122714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lan L, Appelman C, Smith AR, Yu J, Larsen S, Marquez RT, et al. Natural product (-)-gossypol inhibits colon cancer cell growth by targeting RNA-binding protein Musashi-1. Mol Oncol. 2015;9(7):1406–20. doi: 10.1016/j.molonc.2015.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Oliver CL, Miranda MB, Shangary S, Land S, Wang S, Johnson DE. (-)-Gossypol acts directly on the mitochondria to overcome Bcl-2- and Bcl-X(L)-mediated apoptosis resistance. Mol Cancer Ther. 2005;4(1):23–31. [PubMed] [Google Scholar]
- 80.Etxebarria A, Landeta O, Antonsson B, Basanez G. Regulation of antiapoptotic MCL-1 function by gossypol: mechanistic insights from in vitro reconstituted systems. Biochemical pharmacology. 2008;76(11):1563–76. doi: 10.1016/j.bcp.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 81.Sonpavde G, Matveev V, Burke JM, Caton JR, Fleming MT, Hutson TE, et al. Randomized phase II trial of docetaxel plus prednisone in combination with placebo or AT-101, an oral small molecule Bcl-2 family antagonist, as first-line therapy for metastatic castration-resistant prostate cancer. Ann Oncol. 2012;23(7):1803–8. doi: 10.1093/annonc/mdr555. [DOI] [PubMed] [Google Scholar]
- 82.Ready N, Karaseva NA, Orlov SV, Luft AV, Popovych O, Holmlund JT, et al. Double-blind, placebo-controlled, randomized phase 2 study of the proapoptotic agent AT-101 plus docetaxel, in second-line non-small cell lung cancer. J Thorac Oncol. 2011;6(4):781–5. doi: 10.1097/JTO.0b013e31820a0ea6. [DOI] [PubMed] [Google Scholar]
- 83.Corcoran DL, Georgiev S, Mukherjee N, Gottwein E, Skalsky RL, Keene JD, et al. PARalyzer: definition of RNA binding sites from PAR-CLIP short-read sequence data. Genome biology. 2011;12(8):R79. doi: 10.1186/gb-2011-12-8-r79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Koechlein CS, Harris JR, Lee TK, Weeks J, Fox RG, Zimdahl B, et al. High-resolution imaging and computational analysis of haematopoietic cell dynamics in vivo. Nat Commun. 2016;7:12169. doi: 10.1038/ncomms12169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.O’Valle F, Peregrina M, Crespo-Lora V, Galindo-Moreno P, Roman M, Padial-Molina M, et al. Osteoarticular Expression of Musashi-1 in an Experimental Model of Arthritis. BioMed research international. 2015;2015:681456. doi: 10.1155/2015/681456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bobryshev YV, Tran D, Botelho NK, Lord RV, Orekhov AN. Musashi-1 expression in atherosclerotic arteries and its relevance to the origin of arterial smooth muscle cells: histopathological findings and speculations. Atherosclerosis. 2011;215(2):355–65. doi: 10.1016/j.atherosclerosis.2011.01.013. [DOI] [PubMed] [Google Scholar]
- 87.Susaki K, Kaneko J, Yamano Y, Nakamura K, Inami W, Yoshikawa T, et al. Musashi-1, an RNA-binding protein, is indispensable for survival of photoreceptors. Experimental eye research. 2009;88(3):347–55. doi: 10.1016/j.exer.2008.06.019. [DOI] [PubMed] [Google Scholar]
- 88.Daubner GM, Clery A, Allain FH. RRM-RNA recognition: NMR or crystallography.. and new findings. Current opinion in structural biology. 2013;23(1):100–8. doi: 10.1016/j.sbi.2012.11.006. [DOI] [PubMed] [Google Scholar]
- 89.Wang H, Zeng F, Liu Q, Liu H, Liu Z, Niu L, et al. The structure of the ARE-binding domains of Hu antigen R (HuR) undergoes conformational changes during RNA binding. Acta crystallographica Section D, Biological crystallography. 2013;69(Pt 3):373–80. doi: 10.1107/S0907444912047828. [DOI] [PubMed] [Google Scholar]
- 90.Horisawa K, Imai T, Okano H, Yanagawa H. 3′-Untranslated region of doublecortin mRNA is a binding target of the Musashi1 RNA-binding protein. FEBS letters. 2009;583(14):2429–34. doi: 10.1016/j.febslet.2009.06.045. [DOI] [PubMed] [Google Scholar]
- 91.Rentas S, Holzapfel NT, Belew MS, Pratt GA, Voisin V, Wilhelm BT, et al. Musashi-2 attenuates AHR signalling to expand human haematopoietic stem cells. Nature. 2016;532(7600):508–11. doi: 10.1038/nature17665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Battelli C, Nikopoulos GN, Mitchell JG, Verdi JM. The RNA-binding protein Musashi-1 regulates neural development through the translational repression of p21WAF-1. Mol Cell Neurosci. 2006;31(1):85–96. doi: 10.1016/j.mcn.2005.09.003. [DOI] [PubMed] [Google Scholar]
- 93.Bobryshev YV, Freeman AK, Botelho NK, Tran D, Levert-Mignon AJ, Lord RV. Expression of the putative stem cell marker Musashi-1 in Barrett’s esophagus and esophageal adenocarcinoma. Dis Esophagus. 2010;23(7):580–9. doi: 10.1111/j.1442-2050.2010.01061.x. [DOI] [PubMed] [Google Scholar]
- 94.Nikpour P, Mowla SJ, Forouzandeh-Moghaddam M, Ziaee SA. The stem cell self-renewal gene, Musashi 1, is highly expressed in tumor and non-tumor samples of human bladder. Indian J Cancer. 2013 Jul-Sep;50(3):214–8. doi: 10.4103/0019-509X.118735. [DOI] [PubMed] [Google Scholar]
- 95.Gotte M, Wolf M, Staebler A, Buchweitz O, Kelsch R, Schuring AN, et al. Increased expression of the adult stem cell marker Musashi-1 in endometriosis and endometrial carcinoma. The Journal of pathology. 2008;215(3):317–29. doi: 10.1002/path.2364. [DOI] [PubMed] [Google Scholar]
- 96.Ma L, Xu YL, Ding WJ, Shao HF, Teng YC. Prognostic value of Musashi-1 in endometrioid adenocarcinoma. Int J Clin Exp Pathol. 2015;8(5):4564–72. [PMC free article] [PubMed] [Google Scholar]
- 97.Kuang RG, Kuang Y, Luo QF, Zhou CJ, Ji R, Wang JW. Expression and significance of Musashi-1 in gastric cancer and precancerous lesions. World journal of gastroenterology. 2013;19(39):6637–44. doi: 10.3748/wjg.v19.i39.6637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Dahlrot RH, Hansen S, Herrstedt J, Schroder HD, Hjelmborg J, Kristensen BW. Prognostic value of Musashi-1 in gliomas. Journal of neuro-oncology. 2013;115(3):453–61. doi: 10.1007/s11060-013-1246-8. [DOI] [PubMed] [Google Scholar]
- 99.Ma YH, Mentlein R, Knerlich F, Kruse ML, Mehdorn HM, Held-Feindt J. Expression of stem cell markers in human astrocytomas of different WHO grades. Journal of neuro-oncology. 2008;86(1):31–45. doi: 10.1007/s11060-007-9439-7. [DOI] [PubMed] [Google Scholar]
- 100.Toda M, Iizuka Y, Yu W, Imai T, Ikeda E, Yoshida K, et al. Expression of the neural RNA-binding protein Musashi1 in human gliomas. Glia. 2001;34(1):1–7. doi: 10.1002/glia.1034. [DOI] [PubMed] [Google Scholar]
- 101.Chen K, Gao Q, Zhang W, Liu Z, Cai J, Liu Y, et al. Musashi1 regulates survival of hepatoma cell lines by activation of Wnt signalling pathway. Liver international : official journal of the International Association for the Study of the Liver. 2015;35(3):986–98. doi: 10.1111/liv.12458. [DOI] [PubMed] [Google Scholar]
- 102.Yokota N, Mainprize TG, Taylor MD, Kohata T, Loreto M, Ueda S, et al. Identification of differentially expressed and developmentally regulated genes in medulloblastoma using suppression subtraction hybridization. Oncogene. 2004;23(19):3444–53. doi: 10.1038/sj.onc.1207475. [DOI] [PubMed] [Google Scholar]
- 103.Chen PX, Li QY, Yang Z. Musashi-1 Expression is a Prognostic Factor in Ovarian Adenocarcinoma and Correlates with ALDH-1 Expression. Pathol Oncol Res. 2015;21(4):1133–40. doi: 10.1007/s12253-015-9943-6. [DOI] [PubMed] [Google Scholar]
- 104.Seigel GM, Hackam AS, Ganguly A, Mandell LM, Gonzalez-Fernandez F. Human embryonic and neuronal stem cell markers in retinoblastoma. Mol Vis. 2007;13:823–32. [PMC free article] [PubMed] [Google Scholar]
- 105.Aly RM, Ghazy HF. Prognostic significance of MSI2 predicts unfavorable outcome in adult B-acute lymphoblastic leukemia. Int J Lab Hematol. 2015;37(2):272–8. doi: 10.1111/ijlh.12284. [DOI] [PubMed] [Google Scholar]
- 106.Byers RJ, Currie T, Tholouli E, Rodig SJ, Kutok JL. MSI2 protein expression predicts unfavorable outcome in acute myeloid leukemia. Blood. 2011;118(10):2857–67. doi: 10.1182/blood-2011-04-346767. [DOI] [PubMed] [Google Scholar]
- 107.Thol F, Winschel C, Sonntag AK, Damm F, Wagner K, Chaturvedi A, et al. Prognostic significance of expression levels of stem cell regulators MSI2 and NUMB in acute myeloid leukemia. Ann Hematol. 2013;92(3):315–23. doi: 10.1007/s00277-012-1637-5. [DOI] [PubMed] [Google Scholar]
- 108.Kaeda J, Ringel F, Oberender C, Mills K, Quintarelli C, Pane F, et al. Up-regulated MSI2 is associated with more aggressive chronic myeloid leukemia. Leuk Lymphoma. 2015;56(7):2105–13. doi: 10.3109/10428194.2014.981175. [DOI] [PubMed] [Google Scholar]
- 109.Pereira JK, Traina F, Machado-Neto JA, da Duarte AS, Lopes MR, Saad ST, et al. Distinct expression profiles of MSI2 and NUMB genes in myelodysplastic syndromes and acute myeloid leukemia patients. Leuk Res. 2012;36(10):1300–3. doi: 10.1016/j.leukres.2012.06.010. [DOI] [PubMed] [Google Scholar]
- 110.Li D, Peng X, Yan D, Tang H, Huang F, Yang Y, et al. Msi-1 is a predictor of survival and a novel therapeutic target in colon cancer. Ann Surg Oncol. 2011;18(7):2074–83. doi: 10.1245/s10434-011-1567-9. [DOI] [PubMed] [Google Scholar]
- 111.Zong Z, Zhou T, Rao L, Jiang Z, Li Y, Hou Z, et al. Musashi2 as a novel predictive biomarker for liver metastasis and poor prognosis in colorectal cancer. Cancer medicine. 2016;5(4):623–30. doi: 10.1002/cam4.624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Moreira AL, Gonen M, Rekhtman N, Downey RJ. Progenitor stem cell marker expression by pulmonary carcinomas. Mod Pathol. 2010;23(6):889–95. doi: 10.1038/modpathol.2010.68. [DOI] [PubMed] [Google Scholar]
- 113.Charlesworth A, Wilczynska A, Thampi P, Cox LL, MacNicol AM. Musashi regulates the temporal order of mRNA translation during Xenopus oocyte maturation. The EMBO journal. 2006;25(12):2792–801. doi: 10.1038/sj.emboj.7601159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Bertolin AP, Katz MJ, Yano M, Pozzi B, Acevedo JM, Blanco-Obregon D, et al. Musashi mediates translational repression of the Drosophila hypoxia inducible factor. Nucleic Acids Res. 2016;44(16):7555–67. doi: 10.1093/nar/gkw372. [DOI] [PMC free article] [PubMed] [Google Scholar]