Abstract
Nanomedicine is one of the most promising therapeutic modalities researchers are working on. It involves development of drugs and devices that work at the nanoscale (10–9 m). Coronary artery disease (CAD) is responsible for more than a third of all deaths in age group >35 years. With such a huge burden of mortality, CAD is one of the diseases where nanomedicine is being employed for preventive and therapeutic interventions. Nanomedicine can effectively deliver focused drug payload at sites of local plaque formation. Non-invasive strategies include thwarting angiogenesis, intra-arterial thrombosis and local inflammation. Invasive strategies following percutaneous coronary intervention (PCI) include anti-restenosis and healing enhancement. However, before practical application becomes widespread, many challenges need to be dealt with. These include manufacturing at the nanoscale, direct nanomaterial cellular toxicity and visualization.
Keywords: Nanomedicine, Coronary artery disease, Atherosclerosis, Nanotechnology
1. Introduction
The term ‘Nano’ stems from the Greek word for dwarf. As the name suggests, Nanotechnology involves the study and design of miniscule materials and machines, whose functional organization is measured in nanometers.1, 2 In the metric system, a nanometer (nm) is defined as one billionth of a meter.3 Imperceptible to the human eye, this is the world of the living cell. Nanotechnology uses technological machinery at the molecular scale. For consideration, a nanometer is roughly 10 water molecules or 6 carbon atoms wide.4 A ribosome is about 20 nm in diameter; a nucleus is about 6 μm across, and a single strand of DNA 2 nm wide (Fig. 1).
Fig. 1.

Size comparison at nanoscale.
The curious field of Nanotechnology has witnessed a growth explosion in the past decade and exciting innovations are happening as you read this.5, 6, 7, 8 Though the ever expanding field of nano-medicine is enormous, this particular review is focused on Application of Nano-Medicine in Coronary Artery Disease. The review is structured into the following subsections: historical context, nanomedicine based drug delivery, global mortality burden of coronary artery disease (CAD), nanomedicine therapeutic options for CAD (non-invasive and invasive therapy), nanomedicine and imaging, challenges facing routine nanomedicine application in treating CAD and the future of nanomedicine in CAD treatment.
2. Historical context
Richard Feynman, one of the most illustrious physicists of all time delivered a milestone lecture on quantum mechanics at the California Institute of Technology in 1959. Feynman is widely hailed as the father of quantum mechanics and nanotechnology. He did not realize at the time that this lecture would become the cornerstone of technological and scientific innovation in the field of nanotechnology. Feynman postulated writing all 24 volumes of the Encyclopedia Britannica on the head of a mere pin.9
A revolutionary idea suggested by his friend Albert Hibbs was the creation of micro machines, dubbed as the “swallowable surgeon,” that could be controlled from the outside to perform surgery at the cellular level. This device could be used to eliminate malignant neoplasms at their inception or repair defective heart valves. The benefits of working at the nano level are the minimal friction and mechanical wear and tear. Moreover, as the mass of the device is negligible, gravitational forces in turn become trivial as well.
3. Role of nanomedicine in targeted drug delivery
The anatomical peculiarity and inflammatory changes in diseased tissue produce many opportunities for the application of nanomedicine. Local and site specific inflammation can be utilized for focused nanodrug delivery10 (Fig. 2). Inflammation induced vascular permeability and decreased lymphatic drainage causes increased retention and duration of action of nanomedicines11, 12 (Fig. 3).
Fig. 2.
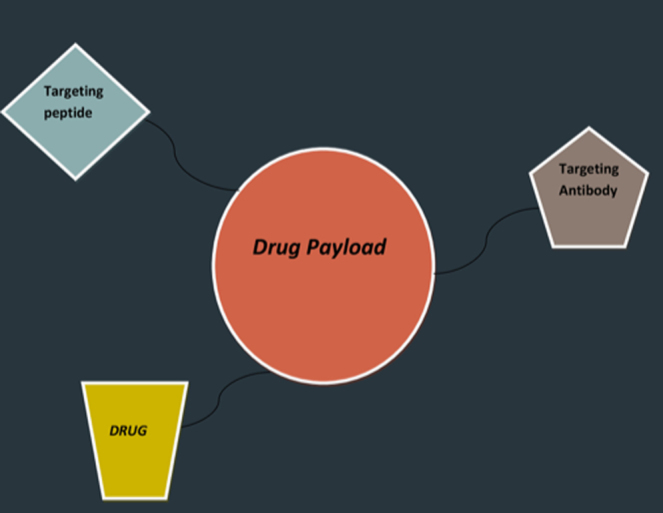
A typical drug payload delivery system.
Fig. 3.

Porous silica nanoparticles encapsulated by lipid bilayers for drug delivery. Creator: Mona Aragon, Carlee Ashley, Ph.D., and Jeffrey Brinker, Ph.D., National Cancer Institute.
4. The global burden of coronary artery disease (CAD)
Undoubtedly, CAD is the leading cause of morbidity and mortality of our generation. Though global CAD mortality has reduced over the last forty years, CAD causes more than 30% of all deaths in people aged 35 and above.13, 14, 15
According to the Global Burden of Disease Study 2013, 17.3 million deaths across the world in 2013 were related to cardiovascular diseases. There was an increment of 41% since 1990.16 CAD is the leading cause of death in adults in high, middle and low income countries.17
In early 2000, it was estimated that CAD mortality in developed nations could rise by around 48% in men and 29% in women between 1990 and 2020. For developing nations, the estimation was 137% in men and 120% in women.18 Above the 40-year age group, the lifetime risk of developing CAD for males is 49% and for females is 32%. For people around 70 years old, the lifetime risk is 35% for males and 24% for females.19 With globally increasing incidence of metabolic syndrome, hypertension and obesity, CAD poses a major risk for all populations. Innovative therapies like targeted nanomedicine, potentially hold some promise in curbing the rising rates of CAD complications, like myocardial infarction, heart failure, and sudden cardiac death.
5. Coronary artery disease and nanomedicine
Atherosclerosis starts at a young age, with progressive plaque deposition in the major arteries of the body. When such a plaque becomes big enough in the coronary artery, myocardial ischemia or infarction can follow. Since the pathogenesis of atherosclerosis starts at the cellular level, only an effective intervention at this level can thwart its progression (Fig. 4). Thus, it makes sense that nanomedicine can be an effective strategy for CAD treatment.
Fig. 4.
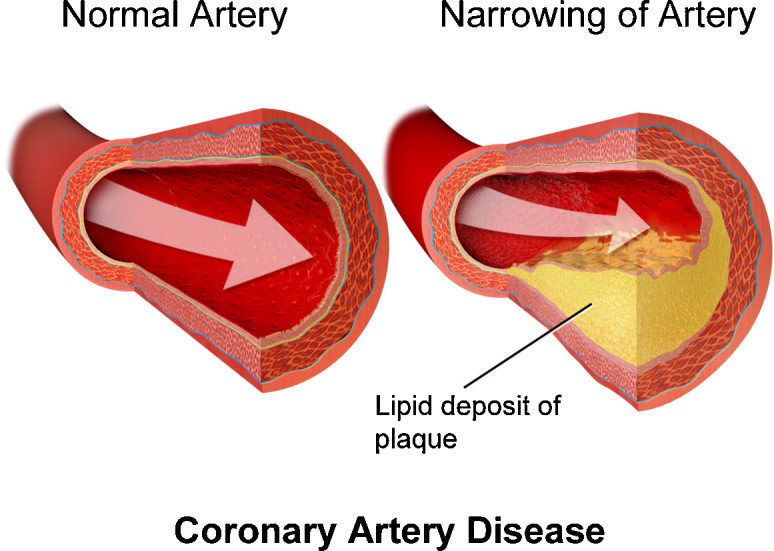
Development of coronary artery disease. By Bruce Blaus, “Blausen gallery 2014”, Wikiversity Journal of Medicine.
There are several areas in the treatment of coronary artery disease where nanotechnology can be used. New and exciting therapeutic modalities may possibly stem from this approach. There are broadly two existing treatment options for CAD: (a) non-invasive management with medical therapy and (b) invasive therapy involving mechanical revascularization (PCI or CABG) (Fig. 5).
Fig. 5.
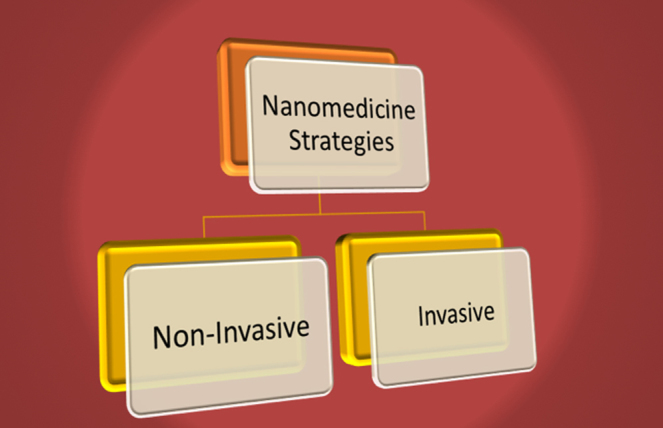
Nano-medicine treatment strategies for CAD.
5.1. Non-invasive nanomedicine strategies
Atherosclerosis is a chronic condition in which the arterial wall thickens and becomes inflamed as a result of atheromatous plaque formation.20 Existing non-invasive techniques seek to reduce atherosclerotic plaques and fix the ones likely to dislodge or thrombose.
5.1.1. Affecting lipid levels
Lipoprotein carriers are required for the transport of cholesterol in the body. These lipoproteins are themselves the size of nanoparticles. Excessive LDL, that carries cholesterol from liver to peripheral tissues, soon becomes oxidized and starts to deposit on the vessel wall. This in turn causes endothelial injury and triggers the inflammatory cycle, which in turn triggers atherosclerosis.21
Currently used pharmacological treatments chiefly consist of statins, which have consistently shown reductions in morbidity and mortality in CAD.22 Anti-inflammatory and anti-oxidant effects of high dose statin therapy have also been observed.23 High dose statin therapy is limited due to dose-dependent side effects. One study evaluated the efficacy of a targeted vesicle system for direct administration of high dose statin.24 The researchers used pravastatin-loaded vesicles which were surface functionalized by oligonucleotides. These oligonucleotides had an affinity for inflammatory macrophages, thus reducing systemic toxicity. It demonstrated 15-fold reductions in cytotoxicity to muscle cells. This study portends a bright future for nanoparticle based drug delivery systems in CAD. HDL transports cholesterol from peripheral tissues to the liver, along with exerting anti-inflammatory athero-protective effects. The HDL levels in the blood may be increased by using biomimetic nanoparticle based synthetic HDL.25 A liposomal formulation with dimyristoyl phosphatidylcholine (DPMC) can help extract cholesterol from peripheral tissues. DPMC has been known to be an HDL surface molecule. In another study, DPMC liposomes were infused in cholesterol fed rabbits. The results show a reduced aortic plaque volume as well as cholesterol content.26
5.1.2. Affecting angiogenesis
Neo-vessel generation within atherosclerotic plaques occurs in advanced disease states. It is postulated that plaque angiogenesis may encourage plaque growth, plaque hemorrhage, and plaque rupture.27 Thus therapies targeting angiogenesis have been tried as well. Fumagillin is known to be a potent anti-angiogenic drug. It is hypothesized that therapies which inhibit angiogenesis may stabilize or regress atherosclerotic plaques. Fumagillin delivery via integrin targeted nanoparticles has been tried, at the site of atherogenic angiogenesis. The adverse systemic effects of Fumagillin (neurocognitive impairment) can be effectively reduced using target specific nanoparticle drug delivery.28
In another study by the same group, cholesterol fed rabbit models were given statin therapies along with nanoparticle based Fumagillin. The results showed sustained anti-angiogenic effects.29
5.1.3. Affecting inflammation
Inflammation in atherosclerosis involves cytokine up-regulation and recruitment of monocytes. These monocytes then travel to the vessel wall and become macrophages, which play the primary cellular role in the pathology of atherosclerosis.20 These macrophages absorb oxidized LDLs, and convert into lipid-filled foam cells, which in turn express inflammatory cytokines. Hence the inflammatory cycle continues. Many methods have been tried to thwart this cycle. Nakashiro et al. offered an interesting hypothesis; nanoparticle-mediated focused delivery of peroxisome proliferator-activated receptor-γ (PPAR-γ) agonist pioglitazone into circulating monocytes, can inhibit plaque rupture in experimental mouse model. Their study results showed decreased macrophage activation as well as atherosclerotic plaque rupture in hyperlipidemic ApoE(−/−) mice. More testing is required but initial results seem promising as a potential therapeutic nano strategy against CAD.30
In one study it was reported that after nanoparticle-assisted systemic delivery of a short interfering RNA (siRNA) silencing CCR2, plaque burden reduced significantly.31
Artificially produced siRNA can effectively decrease its target protein production. However, it has a problem crossing the cell membrane, as it is a large molecule with a negative charge but cannot readily cross the cell membrane due to its large size and negative charge.32
Large scale application has been hampered due to this challenge of siRNA delivery. Nanoparticles may thus be used as effective delivery agents for siRNA to thwart the inflammatory cycle of atherosclerosis.31
Light activatable nano-agents have been shown to directly ablate macrophages and decrease plaque inflammation.33 Iron oxide nanoparticles coated with dextran were loaded with phototoxic agents. An ApoE knockout mouse model was used to demonstrate that the nanoparticles were selectively taken up by macrophages inside atherosclerotic plaques, thus inducing widespread death of macrophages when irradiated, without skin toxicity. Thus light-activated nano-carrier systems may be a potentially safe option to check inflammation by killing macrophages.
Delivery of glucocorticoids may be achieved using nanoparticles. Glucocorticoids reduce macrophage accumulation in atherosclerotic plaques. This was effectively shown in a cholesterol-fed rabbit model.34
Due to a large volume of distribution and a rapid rate of clearance, frequent administrations are needed. Therefore, steroid induced side effects are a matter of concern. This problem may be solved by increasing the circulatory half lives which in turn will increase drug concentration in the vascular endothelium.
In one study it was found that inflammation markedly reduced after liposomal glucocorticoid therapy.35 Hence it may be a potential therapeutic option in the future.
5.1.4. Affecting intra-arterial thrombosis
Growing atherosclerotic plaques develop a necrotic lipid core and a fibrous cap. Intra coronary thrombosis occurs due to subsequent plaque degeneration and rupture. This results in myocardial ischemia and in advanced cases, infarction.20
Hirudin (a natural anti-thrombotic) encapsulated micellar nanoparticles have been tried in vivo to target fibrin rich clots.36 The anti-thrombotic efficacy of the formulation is under testing.
Thrombin is a rate-limiting factor in the clotting cascade. Nanoparticles coupled with irreversible thrombin inhibitor, (d-phenylalanyl-l-prolyl-l-arginyl-chloromethyl ketone) (PPACK), were used in mouse arterial thrombosis models.37 The study showed optimistic results in thwarting intra-arterial thrombosis.
5.2. Invasive nanomedicine therapeutic strategies
Obstructive CAD treatment has been revolutionized by percutaneous coronary interventions (PCI).38 Bare metal stents (BMS) provide mechanical advantage for vascular patency but also result in significant arterial wall injury. This causes neointimal hyperplasia resulting in restenosis. To solve this problem Drug eluting stents (DES) were created. DES though highly effective are not without their adverse effects. Delayed endothelialization and high risk of thrombogenesis require extended anti-platelet drug therapies.39 In 3–20% of patients after DES, in-stent restenosis is found.40 Therefore, biodegradable/bioabsorbable stents using antibody coated nanoparticles which recruit endothelial progenitor cells are under study41 (Fig. 6).
Fig. 6.

Antibody coated nanowires for stent based drug therapy. Creator: Z.L. Wang, Ph.D., National Cancer Institute.
Anti-proliferative drugs can be delivered using nanoparticles specifically at the site of PCI, to thwart neo-intimal genesis. Nakano et al. showed that nanoparticle eluting stent can be an effective drug delivery system and serve as a platform for delivery of nano-machinery targeting cardiovascular pathology.42 Nanoparticle based active recruitment of endothelial cells, at the site of intervention can be used for healing and endothelial cell generation. This may prevent neo-intimal hyperplasia as well as thrombogenesis.
5.2.1. Anti-restenosis strategy following PCI
Nanoparticle based drug delivery has been tried in preventing arterial stenosis. Bisphosphonate is a potent inhibitor of monocytes and macrophages. However bisphosphonate has poor cell membrane permeability thus needing high systemic doses liposomal nanoparticles encapsulated with bisphosphonate have been shown to effectively penetrate macrophages and monocytes, thereby reducing proliferation.43 During vascular insult, transient inactivation of macrophages by liposomal bisphosphonates could thwart subsequent restenosis, as it is characteristically caused by excessive inflammation. In a lipid fed rabbit model, liposomal alendronate administration showed significantly decreased neointimal genesis as well as arterial stenosis.44
A phase II, dose-finding, randomized, multi-center, prospective, double blinded study of liposomal alendronate is currently in progress.
Albumin-based-nanoparticle delivery of paclitaxel has also been tried. Paclitaxel is a mitotic inhibitor with strong anti-proliferative effects. Thus it has been used extensively for restenosis prevention in drug-eluting stents. Albumin reversibly binds paclitaxel and has a natural affinity for vessel walls by attaching with glycoprotein VI-receptors on endothelial cells. In this manner it can be used to transport drugs effectively across endothelial cells.45 Dose-dependent reduction in stent restenosis was seen after administration of albumin-based, paclitaxel-coated nanoparticles (nab-paclitaxel).46 In patients undergoing BMS placement the systemic delivery of Nab-Paclitaxel was tested. The results showed optimal drug delivery without significant toxicity.47
5.2.2. Prevention of in-stent restenosis
Nanoparticle drug delivery directly to vascular injury sites is a promising option. Potential targets for injured endothelium include v3 integrin, VCAM-1, tissue-factor, and subendothelial extracellular matrix proteins such as collagen IV or chondroitin sulfate proteoglycans (CSPGs).48
Prednisolone-encapsulated liposomal formulation directed at CSPGs showed preferential drug concentration to sites of stent induced injury. Decreased rates of restenosis were observed in atherosclerotic rabbits.49 Similarly paclitaxel-encapsulated polymeric nanoparticles have been used in rat carotid injury models50 (Fig. 7). Similar anti-restenotic effects with paclitaxel loaded nano-particles were observed.51
Fig. 7.
Direct control over nanoparticle shape and size aids in focused drug delivery. Joseph DeSimone, Ph.D., National Cancer Institute.
Stents themselves can be used as targets for nanoparticle-assisted drug delivery. A paclitaxel loaded magnetic nanoparticle that has an affinity for the stent struts as well as adjacent arterial tissues has been developed. Upon application of a magnetic field, these nanoparticles can be maneuvered thereby thwarting in-stent restenosis.52
Tsukie et al. found that in a porcine coronary artery model, pitavastatin-NP-eluting stents reduced in-stent stenosis as effectively as sirolimus eluting stents (SES). The statin eluting stent also did not show delayed endothelial healing effects typical of SES.53 Such a nanotechnology platform holds developmental potential for a more efficacious and safer device in the future.
5.2.3. Healing enhancement strategy following PCI
Endothelium is essential for the healthy functioning of the coronary arteries.54 Following PCI, near-complete denudation of endothelium occurs. Therapies have been tried to promote endothelial regeneration on stent surface. A self-assembled nanofibrous matrix that mimics an endothelial extracellular matrix has been developed. In this study, a nanofibrous matrix was used to attract endothelial cells.55 It also acted as a surrogate reservoir of nitric oxide (NO), which is known to inhibit smooth muscle cell proliferation as well as platelet adhesion.
This NO-releasing nanofibrous matrix resulted in significantly improved endothelial cell formation. Smooth muscle cell proliferation and platelet cells adhesion were also inhibited in vitro.
Peptide amphiphile nanofibers were conjugated with (a) an REDV epitope that selectively encourages endothelial cell adhesion and spreads over smooth muscle cells and (2) a Dopa molecule that creates a potent hydrogen bond with hydrophilic surfaces of stainless steel, thus securing the nanofiber on the stent surface.56
Improvement in endothelial cell adhesion, spread and proliferation was seen with a nanofiber-coated stent as compared to a bare stent. Further research is required to evaluate clinical applicability. However this nanofibrous matrix offers great potential for bioactive material development and endothelial cell recovery.
In one study involving magnetic nanoparticles, endothelial cells were loaded with magnetic nanoparticles (MNP) and infused into rats with stainless steel stents placed in their carotid arteries. On the application of a magnetic field, these MNP-loaded cells selectively targeted to the stent and remained attached.57, 58 Further studies are required to ascertain long-term endothelial cell viability after stent delivery.
5.2.4. CABG biosynthetic grafts using nanotechnology
In coronary artery bypass graft (CABG) surgery, the patient's healthy vascular graft is used to bypass the obstructed coronary vessels in order to restore coronary blood flow.59 Sometimes patient's own blood vessels are unavailable as they might be diseased. Nanotechnology based tissue engineered vascular grafts (TEVGs) which are flexible like the normal arteries are a potential solution to this problem.
An artificial vessel functioning as a small conduit has been developed.60
Mesenchymal stem cells seeded to electro-spun biodegradable nano-fibrous scaffolds showed cellular graft synthesis, and were comparable to healthy normal vessels.60
6. Nanomedicine and imaging
Magnetic nanoparticles have a role in many biomedical applications for imaging, sensing, tagging and separation. Ferromagnetic iron oxide particles with poly-dispersive properties are being used to accentuate the contrast for MRI. Drug delivery at lower magnetic field gradients is also being pursued. Such poly-dispersive iron particles have reduced the concentration of nanoparticles required for cell separation.61
With decreasing size, the nanoparticle surface area to volume ratio rises significantly. Reporter molecules like radiotracers can be conjugated to nanoparticles thereby increasing signal to noise ratio for imaging. Photoacoustic imaging involves gold nanoparticles in the form of contrast agents.62, 63, 64, 65
Due to their strong light scattering properties, gold nanoparticles can be used in optical imaging of coronary blood vessels.66
Nanoparticle based blood pool contrast agents are used for visualization of vasculature in vivo. Quantum dots can be used for cell tracking studies and membrane protein labeling.67, 68
Enzymatic activity in the pathogenesis of atherosclerosis can be tracked via fluoroscence resonance energy transfer (FRET).69, 70
7. Challenges in application of nanomedicine
Cardiovascular disease poses unique problems for nanotechnology implementation. Inflammation, infection, neoplasia, autoimmunity, and degeneration are important harbingers of mortality. Through innovation and scientific development, we have created novel technological wonders such as nanotubes, nanowires, and nano-spheres. However, the problems facing their implementation are many (Fig. 8).
Fig. 8.
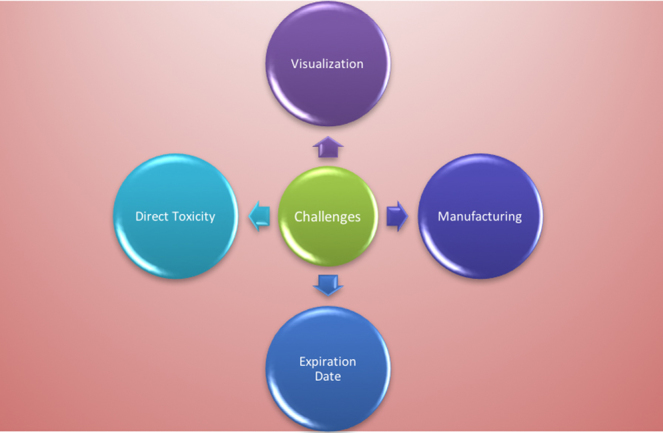
Challenges in application.
7.1. Development
Any production unit that hopes to mass produce nano-machines needs to be extremely clean. The tiniest contaminants in the manufacturing process can result in impaired nano-infrastructure. This may translate into in vivo adverse effects. In 1986, Drexler, one of the great pioneers of nanotechnology, postulated that such nanomaterials can be used for self-manufacture and assembly, thus creating a “billion tiny factories.”
7.2. Age of nanomaterials in vivo
The age of a nanomaterial in a biological cell is undetermined. No long-term information is present to estimate a definite answer. The potential adverse effects of a foreign body inside a living cell are left to speculation as well.71
7.3. Biological side effects
The adverse effects of nano-machines at the cellular level might translate into symptoms manifesting at the level of the patient. The fact is that no consistent information is present about the biological safety of nanoparticles. Safety protocols and tests have been written but currently hazard and risk identification is done on a case-by-case basis.72
7.4. Direct toxicity
The chemical makeup of the nanoparticle can cause direct toxicity inside the living cell. Toxicity of nanoparticles is based on their chemical structure.73 For example, the toxicity of carbon nanotubes is a direct manifestation of the mechanical structure, dimensions, type of carbon isotope used, surface coating, and relative carbon concentrations.74
DNA damage has also been reported in some studies. In one study in mouse embryonic stem cells, the mutation frequency doubled after injection of multi-walled carbon nanotubes.75
8. Future of nanomedicine in CAD therapy
Great potential for nanomedicine exists in the treatment of CAD. Effective nano-drug delivery systems for different drugs are under development. Systemic toxicity is limited using this approach. Quick endothelial regeneration after stent placement is facilitated by biomimetic nanofibrous scaffolds. Tissue engineered graft materials for CABG can be developed using nano-spinning and nano-patterning. Endothelial cells incorporated with polylactide-based magnetic nanoparticles (MNP) have shown strong magnetic responsiveness, and a potential for swift expansion. Disintegration of internalized MNP in proliferating and contact-inhibited states has also been shown.76 MNP-based gene and cell therapy combined with magnetic fields was used to produce circumferential re-endothelialization of blood vessels.77
Magnetic cell targeting is a unique approach to tackling the problem of cell delivery in regenerative medicine.78 Despite of preclinical experiment, the scalability of this technique is questionable. Ferromagnetic implants like stents, have shown some degree of clinical scalability. Upon application of a homogenous magnetic field, ferromagnetic implants can be used for cell targeted drug delivery (Fig. 9). However, such ferromagnetic stents cannot be placed everywhere, especially in long vessels with turbulent blood flow. Chondrogenesis has been reported in the vessel, due to rise in cytokine concentration.79 Gene expression has also been found to be upregulated for up to 3 weeks, after ferro-magnetic material implantation.80 Despite the side effects, a scalable magnetic targeting system has been shown to accentuate nano-medicine cell retention and decrease restenosis rate in vessels. Magnetic delivery of endothelial cells has been shown to be a good strategy to prevent lumen narrowing after angioplasty.81
Fig. 9.
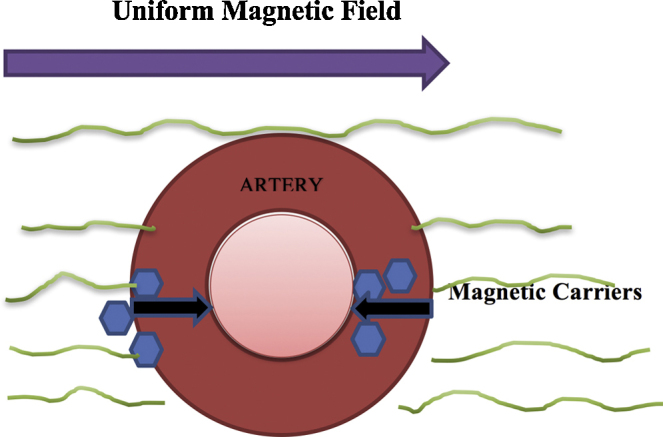
Cell targeted nano-medicine therapy using magnetic field.
Duplex stainless steel stents have shown weak ferromagnetic properties. Due to rapid capture of endothelial outgrowth cells, such stents can be utilized to accelerate vascular healing. Studies are being conducted to apply this technique in vivo for clinical application.
Nanoparticle labeled autologous endothelial cell delivery has shown patent devices and a thin, uniform neo-intima, without any thrombosis or inflammation after 7 days.82 Clinical application of nanotechnology needs to be carefully evaluated using randomized trials. There is a dearth of reliable data on nanotechnology safety. Concerted research effort between biomedical engineers and practicing clinicians is essential to develop practical and efficacious therapeutic nano-modalities for CAD. All in all, momentous strides in nanomedicine have remarkably enhanced current treatment approaches for coronary artery disease. As therapeutic modalities continue to shrink in size and scientific curiosity continues to expand, the future of CAD treatment indeed seems very exciting.
Conflicts of interest
The authors have none to declare.
References
- 1.Emerich D.F., Thanos C.G. Nanotechnology and medicine. Expert Opin Biol Ther. 2003;3:655–663. doi: 10.1517/14712598.3.4.655. [DOI] [PubMed] [Google Scholar]
- 2.Sahoo S.K., Labhasetwar V. Nanotech approaches to drug delivery and imaging. Drug Discov Today. 2003;8:1112–1120. doi: 10.1016/s1359-6446(03)02903-9. [DOI] [PubMed] [Google Scholar]
- 3.Sahoo S.K., Parveen S., Panda J.J. The present and future of nanotechnology in human health care. Nanomed Nanotechnol Biol Med. 2007;3(1):20–31. doi: 10.1016/j.nano.2006.11.008. [DOI] [PubMed] [Google Scholar]
- 4.Whitesides G.M. The ‘right’ size in nanobiotechnology. Nat Biotechnol. 2003;21:1161–1165. doi: 10.1038/nbt872. [DOI] [PubMed] [Google Scholar]
- 5.Matsudai M., Hunt G. Nanotechnology and public health. Nippon Koshu Eisei Zasshi. 2005;52:923–927. [PubMed] [Google Scholar]
- 6.Chan W.C. Bionanotechnology progress and advances. Biol Blood Marrow Transplant. 2006;12:87–91. doi: 10.1016/j.bbmt.2005.10.004. [DOI] [PubMed] [Google Scholar]
- 7.Williams D. Nanotechnology: a new look. Med Dev Technol. 2004;15:9–10. [PubMed] [Google Scholar]
- 8.Cheng M.M., Cuda G., Bunimovich Y.L. Nanotechnologies for biomolecular detection and medical diagnostics. Curr Opin Chem Biol. 2006;10:11–19. doi: 10.1016/j.cbpa.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 9.Feynman R. There's plenty of room at the bottom. Eng Sci. 1960;23:22–36. [Google Scholar]
- 10.Vasir J.K., Labhasetwar V. Targeted drug delivery in cancer therapy. Technol Cancer Res Treat. 2005;4:363–374. doi: 10.1177/153303460500400405. [DOI] [PubMed] [Google Scholar]
- 11.Matsumura Y., Maeda H. A new concept for macromolecular. Therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986;46:6387–6392. [PubMed] [Google Scholar]
- 12.Maeda H., Wu J., Sawa T., Matsumura Y., Hori K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release. 2000;65:271–284. doi: 10.1016/s0168-3659(99)00248-5. [DOI] [PubMed] [Google Scholar]
- 13.Lloyd-Jones D., Adams R.J., Brown T.M. Executive summary: heart disease and stroke statistics – 2010 update: a report from the American Heart Association. Circulation. 2010;121:948. doi: 10.1161/CIRCULATIONAHA.109.192666. [DOI] [PubMed] [Google Scholar]
- 14.Nichols M., Townsend N., Scarborough P., Rayner M. Cardiovascular disease in Europe 2014: epidemiological update. Eur Heart J. 2014;35:2950. doi: 10.1093/eurheartj/ehu299. [DOI] [PubMed] [Google Scholar]
- 15.Rosamond W., Flegal K., Furie K. American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2008;117(4):e25. doi: 10.1161/CIRCULATIONAHA.107.187998. [DOI] [PubMed] [Google Scholar]
- 16.Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. AU GBD 2013 Mortality and Causes of Death Collaborators. Lancet. 2015;385(9963):117. doi: 10.1016/S0140-6736(14)61682-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lopez A.D., Mathers C.D., Ezzati M., Jamison D.T., Murray C.J. Global and regional burden of disease and risk factors, 2001: systematic analysis of population health data. Lancet. 2006;367:1747. doi: 10.1016/S0140-6736(06)68770-9. [DOI] [PubMed] [Google Scholar]
- 18.Yusuf S., Reddy S., Ounpuu S., Anand S. Global burden of cardiovascular diseases. Part I. General considerations, the epidemiologic transition, risk factors, and impact of urbanization. Circulation. 2001;104:2746. doi: 10.1161/hc4601.099487. [DOI] [PubMed] [Google Scholar]
- 19.Lerner D.J., Kannel W.B. Patterns of coronary heart disease morbidity and mortality in the sexes: a 26-year follow-up of the Framingham population. Am Heart J. 1986;111:383. doi: 10.1016/0002-8703(86)90155-9. [DOI] [PubMed] [Google Scholar]
- 20.Libby P., Ridker P.M., Hansson G.K. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473:317–325. doi: 10.1038/nature10146. [DOI] [PubMed] [Google Scholar]
- 21.Ross R. Atherosclerosis – an inflammatory disease. N Engl J Med. 1999;340:115–126. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- 22.Shepherd J., Cobbe S.M., Ford I. West of Scotland Coronary Prevention Study Group. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. N Engl J Med. 1995;333:1301–1307. doi: 10.1056/NEJM199511163332001. [DOI] [PubMed] [Google Scholar]
- 23.Cannon C.P., Steinberg B.A., Murphy S.A., Mega J.L., Braunwald E. Meta-analysis of cardiovascular outcomes trials comparing intensive versus moderate statin therapy. J Am Coll Cardiol. 2006;48:438–445. doi: 10.1016/j.jacc.2006.04.070. [DOI] [PubMed] [Google Scholar]
- 24.Broz P., Ben-Haim N., Grzelakowski M., Marsch S., Meier W., Hunziker P. Inhibition of macrophage phagocytotic activity by a receptor-targeted polymer vesicle-based drug delivery formulation of pravastatin. J Cardiovasc Pharmacol. 2008;51:246–252. doi: 10.1097/FJC.0b013e3181624aed. [DOI] [PubMed] [Google Scholar]
- 25.Lan Hsia S., Duncan R., Schob A.H. Serum levels of high-density lipoprotein phospholipids correlate inversely with severity of angiographically defined coronary artery disease. Atherosclerosis. 2000;152:469–473. doi: 10.1016/s0021-9150(99)00499-2. [DOI] [PubMed] [Google Scholar]
- 26.Cho B.H., Park J.R., Nakamura M.T., Odintsov B.M., Wallig M.A., Chung B.H. Synthetic dimyristoylphosphatidylcholine liposomes assimilating into high-density lipoprotein promote regression of atherosclerotic lesions in cholesterol-fed rabbits. Exp Biol Med (Maywood) 2010;235:1194–1203. doi: 10.1258/ebm.2010.009320. [DOI] [PubMed] [Google Scholar]
- 27.Moreno P.R., Purushothaman K.R., Fuster V. Plaque neovascularization is increased in ruptured atherosclerotic lesions of human aorta: implications for plaque vulnerability. Circulation. 2004;110:2032–2038. doi: 10.1161/01.CIR.0000143233.87854.23. [DOI] [PubMed] [Google Scholar]
- 28.Winter P.M., Neubauer A.M., Caruthers S.D. Endothelial alpha(v)beta3 integrin-targeted fumagillin nanoparticles inhibit angiogenesis in atherosclerosis. Arterioscler Thromb Vasc Biol. 2006;26:2103–2109. doi: 10.1161/01.ATV.0000235724.11299.76. [DOI] [PubMed] [Google Scholar]
- 29.Winter P.M., Caruthers S.D., Zhang H., Williams T.A., Wickline S.A., Lanza G.M. Antiangiogenic synergism of integrin-targeted fumagillin nanoparticles and atorvastatin in atherosclerosis. JACC Cardiovasc Imaging. 2008;1:624–634. doi: 10.1016/j.jcmg.2008.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nakashiro S., Matoba T., Umezu R. Pioglitazone-incorporated nanoparticles prevent plaque destabilization and rupture by regulating monocyte/macrophage differentiation in ApoE−/− mice. Arterioscler Thromb Vasc Biol. 2016;36:491–500. doi: 10.1161/ATVBAHA.115.307057. [DOI] [PubMed] [Google Scholar]
- 31.Leuschner F., Dutta P., Gorbatov R. Therapeutic siRNA silencing in inflammatory monocytes in mice. Nat Biotechnol. 2011;29:1005–1010. doi: 10.1038/nbt.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Blow N. Small RNAs: delivering the future. Nature. 2007;450:1117–1120. doi: 10.1038/4501117a. [DOI] [PubMed] [Google Scholar]
- 33.McCarthy J.R., Korngold E., Weissleder R., Jaffer F.A. A light-activated theranostic nanoagent for targeted macrophage ablation in inflammatory atherosclerosis. Small. 2010;6:2041–2049. doi: 10.1002/smll.201000596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Poon M., Gertz S.D., Fallon J.T. Dexamethasone inhibits macrophage accumulation after balloon arterial injury in cholesterol fed rabbits. Atherosclerosis. 2001;155:371–380. doi: 10.1016/s0021-9150(00)00605-5. [DOI] [PubMed] [Google Scholar]
- 35.Lobatto M.E., Fayad Z.A., Silvera S. Multimodal clinical imaging to longitudinally assess a nanomedical anti-inflammatory treatment in experimental atherosclerosis. Mol Pharm. 2010;7:2020–2029. doi: 10.1021/mp100309y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Peters D., Kastantin M., Kotamraju V.R. Targeting atherosclerosis by using modular, multifunctional micelles. Proc Natl Acad Sci U S A. 2009;106:9815–9819. doi: 10.1073/pnas.0903369106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Myerson J., He L., Lanza G., Tollefsen D., Wickline S. Thrombin-inhibiting perfluorocarbon nanoparticles provide a novel strategy for the treatment and magnetic resonance imaging of acute thrombosis. J Thromb Haemost. 2011;9:1292–1300. doi: 10.1111/j.1538-7836.2011.04339.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Serruys P.W., Kutryk M.J., Ong A.T. Coronary–artery stents. N Engl J Med. 2006;354:483–495. doi: 10.1056/NEJMra051091. [DOI] [PubMed] [Google Scholar]
- 39.Stone G.W., Moses J.W., Ellis S.G. Safety and efficacy of sirolimus and paclitaxel-eluting coronary stents. N Engl J Med. 2007;356:998–1008. doi: 10.1056/NEJMoa067193. [DOI] [PubMed] [Google Scholar]
- 40.Dangas G.D., Claessen B.E., Caixeta A., Sanidas E.A., Mintz G.S., Mehran R. In-stent restenosis in the drug-eluting stent era. J Am Coll Cardiol. 2010;56:1897–1907. doi: 10.1016/j.jacc.2010.07.028. [DOI] [PubMed] [Google Scholar]
- 41.Pendyala L.K., Matsumoto D., Shinke T. Nobori stent shows less vascular inflammation and early recovery of endothelial function compared with Cypher stent. JACC Cardiovasc Interv. 2012;5:436–444. doi: 10.1016/j.jcin.2011.11.013. [DOI] [PubMed] [Google Scholar]
- 42.Nakano K., Egashira K., Masuda S. Formulation of nanoparticle-eluting stents by a cationic electrodeposition coating technology: efficient nano-drug delivery via bioabsorbable polymeric nanoparticle-eluting stents in porcine coronary arteries. JACC Cardiovasc Interv. 2009;2:277–283. doi: 10.1016/j.jcin.2008.08.023. [DOI] [PubMed] [Google Scholar]
- 43.Danenberg H.D., Fishbein I., Gao J. Macrophage depletion by clodronate-containing liposomes reduces neointimal formation after balloon injury in rats and rabbits. Circulation. 2002;106:599–605. doi: 10.1161/01.cir.0000023532.98469.48. [DOI] [PubMed] [Google Scholar]
- 44.Danenberg H.D., Golomb G., Groothuis A. Liposomal alendronate inhibits systemic innate immunity and reduces in-stent neointimal hyperplasia in rabbits. Circulation. 2003;108:2798–2804. doi: 10.1161/01.CIR.0000097002.69209.CD. [DOI] [PubMed] [Google Scholar]
- 45.Stinchcombe T.E. Nanoparticle albumin-bound paclitaxel: a novel Cremphor-EL-free formulation of paclitaxel. Nanomedicine (Lond) 2007;2:415–423. doi: 10.2217/17435889.2.4.415. [DOI] [PubMed] [Google Scholar]
- 46.Kolodgie F.D., John M., Khurana C. Sustained reduction of in-stent neointimal growth with the use of a novel systemic nanoparticle paclitaxel. Circulation. 2002;106:1195–1198. doi: 10.1161/01.cir.0000032141.31476.15. [DOI] [PubMed] [Google Scholar]
- 47.Margolis J., McDonald J., Heuser R. Systemic nanoparticle paclitaxel (nab-paclitaxel) for in-stent restenosis I (SNAPIST-I): a first-in-human safety and dose-finding study. Clin Cardiol. 2007;30:165–170. doi: 10.1002/clc.20066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Brito L., Amiji M. Nanoparticulate carriers for the treatment of coronary restenosis. Int J Nanomed. 2007;2:143–161. [PMC free article] [PubMed] [Google Scholar]
- 49.Joner M., Morimoto K., Kasukawa H. Site-specific targeting of nanoparticle prednisolone reduces in-stent restenosis in a rabbit model of established atheroma. Arterioscler Thromb Vasc Biol. 2008;28:1960–1966. doi: 10.1161/ATVBAHA.108.170662. [DOI] [PubMed] [Google Scholar]
- 50.Chan J.M., Rhee J.W., Drum C.L. In vivo prevention of arterial restenosis with paclitaxel encapsulated targeted lipid-polymeric nanoparticles. Proc Natl Acad Sci U S A. 2011;108:19347–19352. doi: 10.1073/pnas.1115945108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lanza G.M., Yu X., Winter P.M. Targeted antiproliferative drug delivery to vascular smooth muscle cells with a magnetic resonance imaging nanoparticle contrast agent: implications for rational therapy of restenosis. Circulation. 2002;106:2842–2847. doi: 10.1161/01.cir.0000044020.27990.32. [DOI] [PubMed] [Google Scholar]
- 52.Chorny M., Fishbein I., Yellen B.B. Targeting stents with local delivery of paclitaxel-loaded magnetic nanoparticles using uniform fields. Proc Natl Acad Sci U S A. 2010;107:8346–8351. doi: 10.1073/pnas.0909506107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tsukie N., Nakano K., Matoba T. Pitavastatin-incorporated nanoparticle-eluting stents attenuate in-stent stenosis without delayed endothelial healing effects in a porcine coronary artery model. J Atheroscler Thromb. 2013;20:32–45. doi: 10.5551/jat.13862. [DOI] [PubMed] [Google Scholar]
- 54.Kipshidze N., Dangas G., Tsapenko M. Role of the endothelium in modulating neointimal formation: vasculoprotective approaches to attenuate restenosis after percutaneous coronary interventions. J Am Coll Cardiol. 2004;44:733–739. doi: 10.1016/j.jacc.2004.04.048. [DOI] [PubMed] [Google Scholar]
- 55.Kushwaha M., Anderson J.M., Bosworth C.A. A nitric oxide releasing, self assembled peptide amphiphile matrix that mimics native endothelium for coating implantable cardiovascular devices. Biomaterials. 2010;31:1502–1508. doi: 10.1016/j.biomaterials.2009.10.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ceylan H., Tekinay A.B., Guler M.O. Selective adhesion and growth of vascular endothelial cells on bioactive peptide nanofiber functionalized stainless steel surface. Biomaterials. 2011;32:8797–8805. doi: 10.1016/j.biomaterials.2011.08.018. [DOI] [PubMed] [Google Scholar]
- 57.Polyak B., Fishbein I., Chorny M. High field gradient targeting of magnetic nanoparticle-loaded endothelial cells to the surfaces of steel stents. Proc Natl Acad Sci U S A. 2008;105:698–703. doi: 10.1073/pnas.0708338105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pislaru S.V., Harbuzariu A., Gulati R. Magnetically targeted endothelial cell localization in stented vessels. J Am Coll Cardiol. 2006;48:1839–1845. doi: 10.1016/j.jacc.2006.06.069. [DOI] [PubMed] [Google Scholar]
- 59.Stankus J.J., Soletti L., Fujimoto K., Hong Y., Vorp D.A., Wagner W.R. Fabrication of cell microintegrated blood vessel constructs through electrohydrodynamic atomization. Biomaterials. 2007;28:2738–2746. doi: 10.1016/j.biomaterials.2007.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hashi C.K., Zhu Y., Yang G.Y. Antithrombogenic property of bone marrow mesenchymal stem cells in nanofibrous vascular grafts. Proc Natl Acad Sci U S A. 2007;104:11915–11920. doi: 10.1073/pnas.0704581104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Qiang Y., Antony J., Marino M.G. Synthesis of core–shell nanoclusters with high magnetic moment for biomedical applications. IEEE Trans Magn. 2004;40:3538–3540. [Google Scholar]
- 62.Wang Y., Xie X., Wang X. Photoacoustic tomography of a nanoshell contrast agent in the in vivo rat brain. Nano Lett. 2004;4:1689–1692. [Google Scholar]
- 63.Yang X., Skrabalak S.E., Li Z.Y., Xia Y., Wang L.V. Photoacoustic tomography of a rat. Nano Lett. 2007;7:3798–3802. doi: 10.1021/nl072349r. [DOI] [PubMed] [Google Scholar]
- 64.Mallidi S., Larson T., Aaron J., Sokolov K., Emelianov S. Molecular specific optoacoustic imaging with plasmonic nanoparticles. Opt Express. 2007;15:6583–6588. doi: 10.1364/oe.15.006583. [PubMed] [DOI] [PubMed] [Google Scholar]
- 65.Yang X., Stein E.W., Ashkenazi S., Wang L.V. Nanoparticles for photoacoustic imaging. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1:360–368. doi: 10.1002/wnan.42. [DOI] [PubMed] [Google Scholar]
- 66.Jain P.K., Huang X.H., El-Sayed I.H., El-Sayed M.A. Noble metals on the nanoscale: optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc Chem Res. 2008;41:1578–1586. doi: 10.1021/ar7002804. [DOI] [PubMed] [Google Scholar]
- 67.Howarth M., Takao K., Hayashi Y., Ting A.Y. Targeting quantum dots to surface proteins in living cells with biotin ligase. Proc Natl Acad Sci U S A. 2005;102:7583–7588. doi: 10.1073/pnas.0503125102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bonasio R., Carman C.V., Kim E. Specific and covalent labeling of a membrane protein with organic fluorochromes and quantum dots. Proc Natl Acad Sci U S A. 2007;104:14753–14758. doi: 10.1073/pnas.0705201104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chang E., Miller J.S., Sun J. Protease-activated quantum dot probes. Biochem Biophys Res Commun. 2005;334:1317–1321. doi: 10.1016/j.bbrc.2005.07.028. [DOI] [PubMed] [Google Scholar]
- 70.Xu C., Xing B., Rao J. A self-assembled quantum dot probe for detecting beta-lactamase activity. Biochem Biophys Res Commun. 2006;344:931–935. doi: 10.1016/j.bbrc.2006.03.225. [DOI] [PubMed] [Google Scholar]
- 71.Zhang L.W., Monteiro-Riviere N.A. Mechanisms of quantum dot nanoparticle cellular uptake. Toxicol Sci. 2009;110:138–155. doi: 10.1093/toxsci/kfp087. [DOI] [PubMed] [Google Scholar]
- 72.Hoet P., Legiest B., Geys J., Nemery B. Do nanomedicine require novel safety assessments to ensure their safety for long-term human use? Drug Saf. 2009;32:625–636. doi: 10.2165/00002018-200932080-00002. [DOI] [PubMed] [Google Scholar]
- 73.Ambesh P., Angeli D.G. Nanotechnology in neurology: genesis, current status, and future prospects. Ann Indian Acad Neurol. 2015;18(4):382–386. doi: 10.4103/0972-2327.169535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Magrez A., Kasas S., Salicio V. Cellular toxicity of carbon-based nanomaterials. Nano Lett. 2006;6:1121–1125. doi: 10.1021/nl060162e. [DOI] [PubMed] [Google Scholar]
- 75.Zhu L., Chang D.W., Dai L., Hong Y. DNA damage induced by multiwalled carbon nanotubes in mouse embryonic stem cells. Nano Lett. 2007;7:3592–3597. doi: 10.1021/nl071303v. [DOI] [PubMed] [Google Scholar]
- 76.Adamo R.F., Fishbein I., Zhang K. Magnetically enhanced cell delivery for accelerating recovery of the endothelium in injured arteries. J Controlled Release. 2016;222:169–175. doi: 10.1016/j.jconrel.2015.12.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Haldar R., Kaushal A., Samanta S., Ambesh P., Srivastava S., Singh P.K. Contemporary social network sites: relevance in anesthesiology teaching, training, and research. J Anaesthesiol Clin Pharmacol. 2016;32(3):382–385. doi: 10.4103/0970-9185.188821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Riegler J., Liew A., Hynes S.O. Superparamagnetic iron oxide nanoparticle targeting of MSCs in vascular injury. Biomaterials. 2013;34:1987–1994. doi: 10.1016/j.biomaterials.2012.11.040. [DOI] [PubMed] [Google Scholar]
- 79.Soni N., Ambesh P., Sunil K., Jena M. Abernethy malformation with inferior vena cava stenosis: a cryptic cause of cyanosis. Indian Heart J. 2015;67(6):543–545. doi: 10.1016/j.ihj.2015.06.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kedziorek D.A., Muja N., Walczak P. Gene expression profiling reveals early cellular responses to intracellular magnetic labeling with superparamagnetic iron oxide nanoparticles. Magn Reson Med. 2010;63:1031e43. doi: 10.1002/mrm.22290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Polyak B., Medved M., Lazareva N. Magnetic nanoparticle-mediated targeting of cell therapy reduces in-stent stenosis in injured arteries. ACS Nano. 2016;10(10):9559–9569. doi: 10.1021/acsnano.6b04912. [DOI] [PubMed] [Google Scholar]
- 82.Tefft B.J., Uthamaraj S., Harburn J.J. Magnetizable stent-grafts enable endothelial cell capture. J Magn Magn Mater. 2016 doi: 10.1016/j.jmmm.2016.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]


