Abstract
A sensitive and reliable ultra high performance liquid chromatography with tandem mass spectrometry (UHPLC-MS/MS) method was developed and validated for simultaneous determination of L-tetrahydropalmatine (L-THP) and its active metabolites L-isocorypalmine (L-ICP) and L-corydalmine (L-CD) in rat plasma,. The analytes were extracted by liquid-liquid extraction and separated on a Bonshell ASB C18 column (2.1 mm×100 mm; 2.7 μm; Agela) using acetonitrile-formic acid aqueous as mobile phase at a flow rate of 0.2 mL/min in gradient mode. The method was validated over the concentration range of 4.00–2,500 ng/mL for L-THP, 0.400–250 ng/mL for L-ICP and 1.00–625 ng/mL for L-CD, respectively. Intra- and inter-day accuracy and precision were within the acceptable limits of less than 15% at all concentrations. Correlation coefficients (r) for the calibration curves were more than 0.99 for all analytes. The quantitative method was successfully applied for simultaneous determination of L-THP and its active metabolites in a pharmacokinetic study after oral administration with L-THP at the dose of 15 mg/kg to rats.
Keywords: UHPLC-MS/MS, L-THP, Metabolites, Pharmacokinetics
1. Introduction
L-tetrahydropalmatine (L-THP) is a biologically active ingredient isolated from a traditional Chinese herb Rhizoma Corydalis yanhusuo (Pharmacopoeia commission of RPC, 2000; Gao et al., 2014). it has been used as an analgesic agent in China with minor hypnotic side effect for morre than 40 years. The structure of L-THP resembles two dopamine (DA) molecule fused into a tetracyclic alkaloid. It has low toxicity and a high therapeutic index. Animal studies indicated that L-THP exerts its tranquilizing action by blocking postsynaptic DA receptors (Zhu, 1998). Recently, it has been reported that L-THP is effective for treating cocaine addiction because of its high-affinity at D1 receptor as a partial agonist and antagonist at D2 receptor and it also interacts with D5 receptor as an agonist (Mantsch et al., 2007; Wu et al., 1990). In a clinical trial in China, treatment of patients addicted only to heroin with L-THP for one month significantly reduced craving and withdrawal symptoms, and enhanced abstinence rate by three-fold three months after discharge. L-THP is being evaluated in a clinical trial (phase I) for treatment of cocaine addiction in the US (Yang et al., 2008).
Metabolism studies of L-THP in rats revealed that 30 urinary metabolites were identified after administration of L-THP using UPLC-QTOF/MS, and the major metabolites are L-isocorypalmine (L-ICP), L-corypalmine (L-CP), L-corydalmine (L-CD) and L-tetrahydropalmatrubine, with mono demethylation at C-2, C-3, C-9 and C-10 position, respectively (Zhang et al., 2014). Among them (Ma et al., 2008; Wang et al., 2012; Sasaki et al., 2010), L-ICP was recently characterized to reduce behavioral sensitization and rewarding effects of cocaine in mice. Further study (Xu et al., 2013) proved that L-ICP had much higher affinity for the DA receptors than L-THP, presenting not only a high-affinity partial agonist of D1 and D5 receptors but also a moderate-affinity antagonist of D2, D3 and D4 receptors. L-CD was confirmed to have better analgesic property and less significant sedative and hypnotic effects than L-THP. Because of their active charactistics and higher affinities to DA receptors, it is necessary to provide a better insight into underlying metabolite processes of L-THP. The chemical structures of L-THP and the two metabolites are shown in Fig. 1.
Fig. 1.
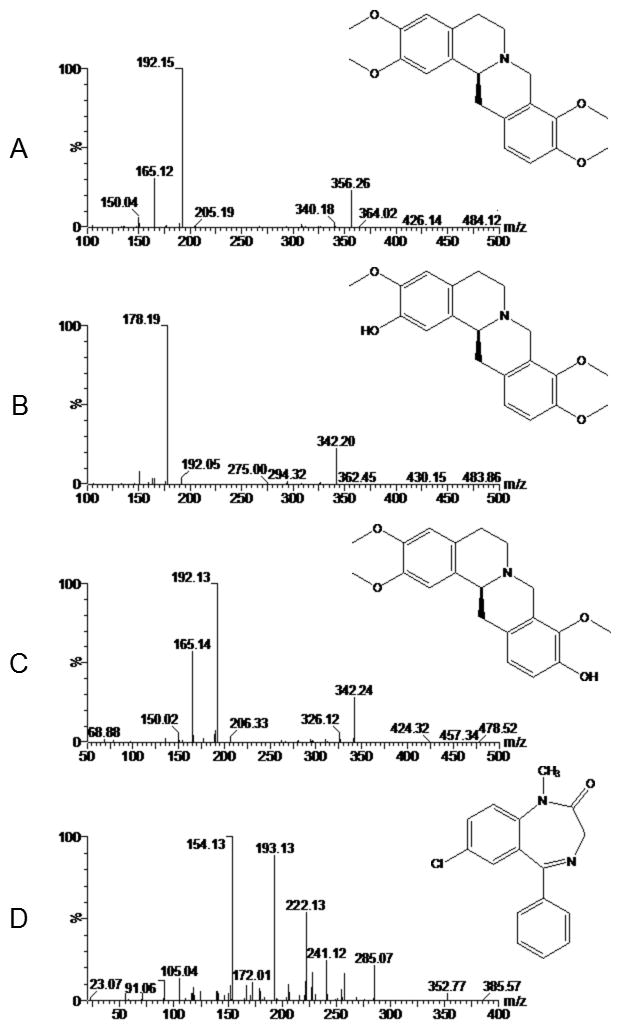
The structure of L-THP (A), L-ICP (B), L-CD (C) and diazepam (D) and their full scan product ions.
Although earlier research have described method for determination of several metabolites of L-THP by LC-MS/MS (Xiao et al., 2014), they mainly focued on the urinary excretion. The quantitative determination and pharmacokinetic profile for active metabolites in plasma had not been thoroughly studied. Investigations on the plasma exposure and clearance pathway of these active metabolites are crucial for better understanding of the pharmacological mechanism of L-THP. In this paper, we described a simple, selective and highly sensitive ultra high performance liquid chromatography with tandem mass spectrometry (UHPLC-MS/MS) method for determination of L-THP and its active metabolites (L-ICP, L-CD) in rat plasma. To the best of our knowledge, no prior reports have researched L-THP as well as its active metabolites from plasma. The ability to analyze these two metabolites together with L-THP makes this assay a very useful tool in preclinical pharmacokinetic studies which would be an important part for medications development of L-THP for the treatment of cocaine addiction in the future.
2. Experimental
2.1. Chemical and reagents
Internal standard (IS) of diazepam was purchased from National Institutes for Food and Drug Control (Beijing, China). L-THP (purity ≥99.5%, measured by HPLC ), L-ICP (purity ≥99.5%, measured by HPLC) and L-CD (purity ≥99.5%, measured by HPLC) were provided by Dr. Lee’s laboratory (McLean Hospital, MA, USA). Acetonitrile of HPLC grade was purchased from Fisher Scientific (Fair Lawn, NJ, USA). Distilled water was obtained from Wahaha Co., Ltd. (Hangzhou, China). Formic acid, ethyl acetate and isopropanol of HPLC grade were provided by Shandong Yuwang Industrial Co., Ltd. (Yucheng, China). Other chemicals were of analytical grade.
Sprague-Dawley rats weighing (180–220) g were kindly provided by the Experimental Animal Center of Shenyang Pharmaceutical University (Shenyang, China). The animal study was carried out in accordance with the Guideline for Animal Experimentation of Shenyang Pharmaceutical University, and the protocol was approved by the Animal Ethics Committee of the institution.
2.2. Instrumentation and analytical conditions
Chromatographic analysis was performed on an ACQUITY UHPLC system (Waters, Milford, MA, USA), including a binary pump solvent manager, a column oven and an autosampler. Three analytes and IS were separated on an Bonshell ASB C18 column (2.1 mm × 100 mm; 2.7 μm; Agela) by gradient elution with the mobile phase of 0.1% formic acid aqueous (A) and acetonitrile (B) at the flow rate of 0.2 mL/min. The gradient program was as follows: 0–2 min, 20–26%B; 2–4 min, 26%B; 4–6 min, 26–80%B; 6–8 min, 80%B; 8–10 min, 20%B. The column temperature was set at 30°C and the autosampler was conditioned at 4°C. An injection volume of 10 μL was applied for analysis.
Triple-quadrupole tandem mass spectrometric detection was carried out on a Micromass Quattro Micro™ API mass spectrometer (Waters, Milford, MA, USA) with an electrospray ionization (ESI) interface. The ESI source was operated in positive ionization mode. The optimal MS parameters were as follows: capillary voltage, 3.5 kV; source temperature, 120°C; desolvation temperature, 400°C; desolvation gas flow, 600 L/h; cone gas flow, 50 L/h. For multiple reaction monitoring (MRM) conditions, the analytes were optimized by infusion of reference standard, as summarized in Table 1. All data were acquired in centroid mode by Masslynx V4.1 software (Waters, Milford, MA, USA).
Table 1.
MRM transition, cone voltage and collision energy for the determination of four analytes.
| Component | Parent ion | Daughter ion | Cone voltage(V) | Collision energy (eV) | Polarity |
|---|---|---|---|---|---|
| L-THP | 356.26 | 192.15 | 50 | 24 | Positive |
| L-ICP | 342.20 | 178.19 | 50 | 23 | Positive |
| L-CD | 342.28 | 192.13 | 40 | 24 | Positive |
| Diazepam | 285.08 | 154.13 | 50 | 28 | Positive |
2.3. Preparation of stock solutions, calibration standards and quality control samples
Standard stock solutions of L-THP, L-ICP and L-CD were prepared in acetonitrile with a final concentration of 100 μg/mL. The IS stock solution was prepared at a concentration of 100 μg/mL and further diluted to achieve a final concentration of 500 ng/mL with acetonitrile. Analytical standards for L-THP, L-ICP and L-CD were prepared by serial dilution with acetonitrile over a concentration range of 40.0–25,000 ng/mL, 4.00–2,500 ng/mL and 10.0–6,250 ng/mL, respectively. All the solutions were stored at 4°C.
The mixed calibration standards were prepared by spiking 100 μL aliquots of blank plasma with 10 μL of the corresponding mixed standard solutions to obtain the final concentrations in the range of 4.00–2,500 ng/mL for L-THP, 0.400–250 ng/mL for L-ICP, 1.00–625 ng/mL for L-CD. Quality control (QC) samples (4.00, 8.00, 100 and 2,000 ng/mL for L-THP; 0.400, 0.800, 10.0 and 200 ng/mL for L-ICP; 1.00, 2.00, 25.0 and 500 ng/mL for L-CD) were prepared in the same fashion.
2.4. Sample preparation
Plasma samples were removed from −80°C storage and thawed in a water bath under ambient conditions. 100 μL of plasma samples spiked with 10 μL IS and 10 μL acetonitrile were vortexed for 30 s. Then, liquid-liquid extraction with 1 mL ethyl acetate and isopropanol (1:1, v/v) was performed by placing the samples on a rotary device for 2 min. After vortex mixing, the samples were centrifuged at 12,000 rpm for 5 min. The upper organic layer was transferred to a clean centrifuge tube and evaporated to dryness under a gentle stream of nitrogen at 30°C. The residue was redissolved in 50 μL acetonitrile and water (1:1, v/v), followed by injection of 10 μL aliquot into UHPLC-MS/MS for analysis.
2.5. Method validation procedure
Full method validation was carried out following the currently accepted US-FDA Bioanalytical Method Validation Guidance and European Medicines Agency Guideline on Bioanalytical Method Validation with respect to specificity, linearity, lower limit of quantification (LLOQ), carryover, precision and accuracy, recovery, matrix effect and stability (Shah et al., 2000; US Food and Drug Administration, 2013)
The specificity was assessed by comparing chromatograms of six individual blank rat plasma samples, blank plasma spiked with L-THP, L-ICP, L-CD and IS, and a plasma sample after oral administration of L-THP. The linearity of the assay was assessed by analyzing the calibration curves using least-squares linear regression of the peak area ratios of the analytes to the IS versus the nominal concentration of the calibration standard with a weighed factor (1/χ2). QC samples at LLOQ, low, medium and high concentration were analyzed on three consecutive validation days with six replicates at each concentration to determine the accuracy and precision. Precision was defined as the relative standard deviation (RSD, %) and accuracy as relative error (RE, %). The extraction recovery was determined at three QC levels (low, medium and high) by comparing the analyte-to-IS peak area ratio obtained from blank plasma spiked with analytes before extraction with the ratio from samples which analytes were added after extraction. The matrix effect was measured by “IS-normalized MF” and was evaluated as follows: for each of the all analytes and the IS, the matrix factor (MF) was calculated as the ratio of peak area with matrix (measured by analyzing blank matrix spiked after extraction with analytes at three concentration levels subtracted those of blank samples) divided by IS and peak area without matrix (pure standard solutions of the analytes at three concentration levels) divided by IS; with acceptable IS normalized MF less than 15% deviation. Stability studies in bio-samples were also conducted at three QC levels in several different storage conditions: at room temperature for 12 h, at −80°C for at least 2 weeks, after three freeze-thaw cycles, and for 12 h in processed samples at 4°C in auto-sampler tray.
2.6. Application to pharmacokinetic study
Six male rats were orally administered L-THP at the dose of 15 mg/kg. All the male rats were fasted for 12 h, with free access to water prior to the experiments. Blood samples (about 0.3 mL) were collected into heparinized centrifuge tubes via the postorbital venous plexus vein from each rat before administration and 0.083, 0.167, 0.333, 0.5, 0.75, 1, 2, 4, 6, 8, 12 and 24 h after administration. These samples were separated for plasma by centrifugation for 10 min at 4,000 rpm and then stored at −80°C until analysis. These samples were analyzed for simultaneous quantitation of the levels of L-THP, L-ICP and L-CD.
The pharmacokinetic parameters of analytes were calculated by the noncompartmental analysis using the DAS 2.1 software package supplied by the Pharmacological Society of China (Beijing, China). All data were expressed as mean±SD.
3. Results and discussion
3.1. Optimization of UHPLC-MS/MS condition
Initially, ESI in both positive and negative modes were tested in the method development, and greater specificity was observed in the positive mode than in the negative mode for all analytes. At the same time, mass spectrometric parameters were optimized with infusion and flow injection analysis. Their full scan product ions are shown in Fig. 1. In order to find the most sensitive analytical condition, several parameters were investigated including composition of mobile phase, and type and concentration of additives. It was found that acetonitrile gave better resolution and lower background noise than methanol. Moreover, the concentration of formic acid in water phase was optimized from 0.05 to 0.2%. Finally, addition of 0.1% formic acid into water phase worked best, it dramatically enhanced the response of the alkaloids with an adequate LLOQ. To obtain chromatograms with satisfying resolution and appropriate retention time, gradient elution was adopted to separate compounds.
3.2. Optimization of sample preparation
The method of protein precipitation by methanol or acetonitrile was tested at first, whereas, it showed extremely low recoveries for all analytes. In the study of liquid-liquid extraction, several organic solvents were tested including ether, ethyl acetate, isopropanol, methyl tert-butyl ether and different ratios of ethyl acetate and isopropanol (from 1:9 to 9:1, v/v). The study showed that their extraction recoveries were significantly increased when a mixture of ethyl acetate and isopropanol was used. Finally ethyl acetate-isopropanol (1:1, v/v) showed optimum recovery for all analytes with no matrix effects. Meanwhile, the plasma sample preparation parameters including volume of extraction solvent, centrifugation rotation speed and centrifugation time were also screened to obtain the ultimate parameters.
3.3. Method validation
3.3.1. Specificity, the lower limit of quantification and carry over
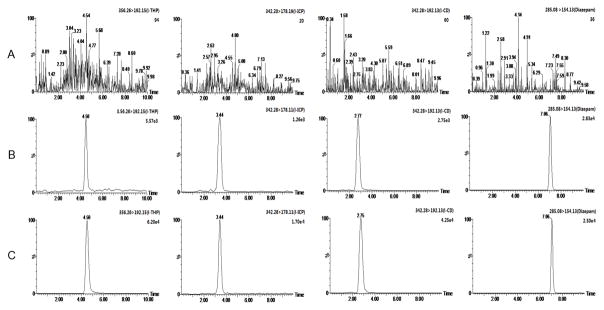
The specificity of the method was established with blank plasma, plasma spiked with the three analytes and IS, and rat plasma samples 1 h after oral administration of L-THP at a dose of 15 mg/kg. The results showed no interference with L-THP, L-ICP, L-CD and IS, as shown in Fig. 2.
Fig. 2.
MRM chromatograms. (A) A blank rat plasma sample; (B) a blank rat plasma spiked with L-THP (LLOQ), L-ICP (LLOQ), L-CD (LLOQ) and IS; (C) a rat plasma 1 h after oral administration of L-THP at the dose 15 mg/kg.
The LLOQ for L-THP, L-ICP and L-CD in plasma with the signal-to-noise over 10 was 4.00, 0.400 and 1.00 ng/mL, respectively. At these concentrations, the intra- and inter-day CV were less than 10.7%, and the RE were within ±7.9% for both L-THP and its metabolites, meeting the requirements.
Chromatographic carryover was evaluated by running a blank or wash after the highest standard and assessing the relative peak area of this blank versus the signal at LLOQ. Carryover was not observed at the retention time of each analyte.
3.3.2. Linearity
Regression equation, correlation coefficients (r) and linearity range are shown in Table 2. They all exhibited good linearity. All standards met the criteria of <15% deviation from nominal concentration.
Table 2.
Summary of calibration curve, correlation coefficient and linearity range of L-THP, L-ICP and L-CD in rat plasma.
| Compound | Regression equation | Correlation coefficient (r) | Linearity range (ng/mL) |
|---|---|---|---|
| L-THP | y=0.3374x+0.0229 | 0.9965 | 4.00–2,500 |
| L-ICP | y=0.2626x+0.0498 | 0.9952 | 0.400–250 |
| L-CD | y=0.3940x+0.0073 | 0.9987 | 1.00–625 |
3.3.3. Precision and accuracy
Table 3 shows the results obtained for the the intra- and inter-day precision and accuracy of L-THP, L-ICP and L-CD. The precision (% RSD) for all these analytes under investigation did not exceed 15%. Accuracy (% RE) for all analytes was in the range from −7.9% to 11.6% at any of the concentrations studied and met the requirements of validation.
Table 3.
Precision and accuracy for analysis of L-THP, L-ICP and L-CD in rat plasma (n=6).
| Compound | Concentration spiked (ng/mL) | Intra-day
|
Inter-day
|
||
|---|---|---|---|---|---|
| RSD % | RE % | RSD % | RE % | ||
| L-THP | 4.00 | 5.4 | −0.5 | 9.6 | −0.5 |
| 8.00 | 6.5 | −1.4 | 13.3 | −1.4 | |
| 100 | 5.7 | 2.6 | 2.6 | 8.7 | |
| 2,000 | 4.8 | −6.1 | 1.4 | 5.8 | |
| L-ICP | 0.400 | 4.6 | 0.7 | 8.4 | 0.7 |
| 0.800 | 3.9 | −0.1 | 8.3 | −0.1 | |
| 10.0 | 12.6 | 11.6 | 12.7 | 7.4 | |
| 200 | 5.9 | 2.7 | 7.0 | −1.2 | |
| L-CD | 1.00 | 10.7 | −7.9 | 8.1 | 0.1 |
| 2.00 | 8.6 | 1.8 | 12.2 | −2.7 | |
| 25.0 | 1.9 | 8.8 | 4.1 | −6.1 | |
| 500 | 7.4 | −2.2 | 12.7 | −2.2 | |
3.3.4. Recovery and matrix effect
The recovery ranged from 88.9 to 95.8% for L-THP, 88.9 to 98.7% for L-ICP and 95.0 to 99.4% for L-CD, respectively. The recovery of the IS was 88.9% at a concentration of 50 ng/mL. The matrix effect was within the range 99.3–104.6% for L-THP, 94.9–107.7% for L-ICP and 94.9–105.7% for L-CD, indicating that no significant matrix effect was observed for L-THP, L-ICP and L-CD. The results are shown in Table 4.
Table 4.
Recovery and matrix effect for analysis of L-THP, L-ICP, L-CD in rat plasma (n=6).
| Compound | Concentration spiked (ng/mL) | Recovery (%, mean±SD) | RSD(%) | Matrix effect (%, mean±SD) | RSD(%) |
|---|---|---|---|---|---|
| L-THP | 8.00 | 88.9±9.9 | 11.1 | 102.8±9.6 | 9.3 |
| 100 | 95.8±9.2 | 9.6 | 99.3±12.8 | 12.9 | |
| 2,000 | 90.0±1.7 | 1.9 | 104.6±11.2 | 10.7 | |
| L-ICP | 0.800 | 90.3±10.6 | 11.8 | 107.7±10.6 | 9.8 |
| 10.0 | 88.9±9.2 | 10.4 | 105.0±8.4 | 8.0 | |
| 200 | 98.7±7.3 | 7.4 | 94.9±12.9 | 13.6 | |
| L-CD | 2.00 | 97.3±7.7 | 7.9 | 105.7±7.2 | 6.8 |
| 25.0 | 99.4±4.6 | 4.7 | 94.9±12.1 | 12.8 | |
| 500 | 95.0±3.8 | 4.0 | 101.9±9.6 | 9.6 |
3.3.5. Stability
All results for the stability samples tested are summarized in Table 5, and they were within the acceptable criteria of ±15%, indicating that L-THP, L-ICP and L-CD were stable under the conditions described above.
Table 5.
Stability of L-THP, L-ICP and L-CD in rat plasma at different conditions (n=3).
| Compound | Concentration spiked (ng/mL) | 12h, room temperature
|
2 weeks, −80°C
|
Three freeze-thaw cycles
|
12h, 4°C
|
||||
|---|---|---|---|---|---|---|---|---|---|
| RE% | RSD% | RE% | RSD% | RE% | RSD% | RE% | RSD% | ||
| L-THP | 8.00 | 9.1 | 11.5 | 8.6 | 3.4 | 1.4 | 7.0 | −1.7 | 7.6 |
| 100 | −3.2 | 4.3 | 7.4 | 10.3 | −0.5 | 3.7 | −5.7 | 5.3 | |
| 2,000 | −8.9 | 12.5 | 9.8 | 14.3 | 3.1 | 6.0 | −2.3 | 8.8 | |
| L-ICP | 0.800 | 2.9 | 5.1 | 2.6 | 13.0 | −5.4 | 6.6 | 2.1 | 9.0 |
| 10.0 | −5.9 | 9.4 | −5.6 | 9.1 | −7.0 | 10.0 | −5.1 | 9.7 | |
| 200 | 6.3 | 3.3 | 10.7 | 1.8 | 8.7 | 2.1 | 5.0 | 4.2 | |
| L-CD | 2.00 | −1.6 | 5.5 | 0.8 | 5.6 | −5.4 | 10.9 | 0.9 | 6.3 |
| 25.0 | 2.2 | 4.0 | −3.3 | 5.0 | 2.1 | 8.7 | 7.5 | 3.1 | |
| 500 | 2.6 | 3.8 | −0.5 | 3.0 | −6.1 | 2.4 | 2.4 | 3.3 | |
3.4. Application to a pharmacokinetic study
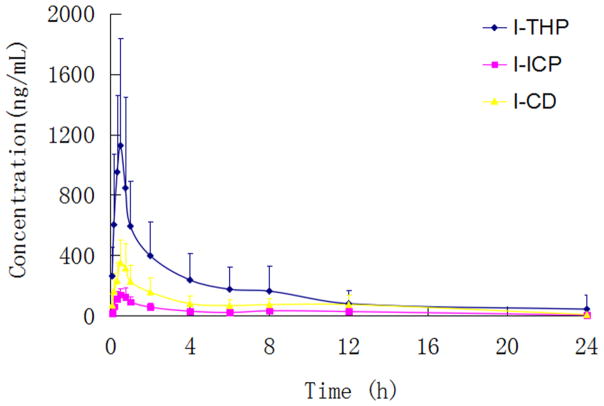
The method described above was successfully applied to a pharmacokinetic study of L-THP and its active metabolites, which the plasma concentration were determined after oral administration of L-THP in male Sprague-Dawley rats. The plasma concentration-time profiles of L-THP, L-ICP and L-CD were shown in Fig. 3, and the pharmacokinetic parameters of L-THP, L-ICP and L-CD including half-time (T1/2), maximum plasma concentration (Cmax), time to reach the maximum concentration (Tmax), area under concentration-time curve (AUC0−t and AUC0−∞) calculated by noncompartmental are listed in Table 6.
Fig. 3.
The mean plasma concentration time profiles of L-THP, L-ICP and L-CD after oral administration of 15 mg/kg, respectively (mean±SD; n=6).
Table 6.
The main pharmacokinetic parameters of L-THP and its metabolites after oral administration of 15 mg/kg (mean±SD; n=6).
| Compound | AUC(0-t)(μg·h/L) | AUC(0-∞) (μg·h/L) | t1/2z (h) | Tmax (h) | Cmax (μg/L) |
|---|---|---|---|---|---|
| L-THP | 4119.09±3598.68 | 4120.55±3599.18 | 4.49±0.83 | 0.44±0.08 | 1217.19±734.58 |
| L-ICP | 722.52±406.61 | 729.00±407.19 | 6.27±2.14 | 0.56±0.16 | 148.79±51.40 |
| L-CD | 1807.73±1118.53 | 1808.86±1119.03 | 3.66±0.40 | 0.58±0.13 | 386.40±170.78 |
After oral administration of L-THP, it was found that L-THP, L-ICP and L-CD can be detected at all of the time points. As shown in the results, the Tmax of L-ICP and L-CD were within the range of 0.5–1 h, and the values of t1/2z were about 6.27 h and 3.66 h. It was demonstrated that it took a short time for L-THP to transform to L-ICP and L-CD. The Cmax for L-THP, L-ICP and L-CD were (1217.19±734.58) μg/L, (148.79±51.40) μg/L and (386.40±170.78) μg/L, and the AUC(0–t) value for them were (4119.09±3598.68) μg·h/L, (722.52±406.61) μg·h/L and (1807.73±1118.53) μg·h/L, respectively. The phenomenon above suggests that L-THP is only partially converted to metabolites, the prototype drug is still present in the blood. The AUC(0–t) of L-CD is about two to three fold higher than that of L-ICP, which may be due to their different structure.
It has already been published that L-THP and its four monodesmethyl metabolites were determined in rat urine by an enzymatic hydrolysis procedure [12]. But in our study, there were no L-CP and l-tetrahydropalmatrubine peaks detected in rat plasma even tracking retention time and mass spectrometry data with the reference substances. Similar result was also obtained from the plasma of intraperitoneal injection taken L-THP . It suggested that L-ICP and L-CD as main metabolites in the plasma, presented different clearance pathway of urine.
4. Conclusions
In this study, a sensitive UHPLC-MS/MS method was developed for the simultaneous determination of L-THP and its two active metabolites (L-ICP and L-CD) in rat plasma. This method requires a simple liquid-liquid extraction procedure and the analysis time was 10 min per sample. After validation, the method has been successfully applied to the pharmacokinetic study. This is the first essay of L-THP, L-ICP and L-CD studied simutaneously in the field of pharmacokinetic study after a single oral administration of L-THP at 15 mg/kg to rats. These results would be necessary in the development of L-THP as a clinical candidate, particulary in our attempt to develop L-THP as a therapeutic agent for treatment of cocaine addiction, an urgent need to meet global challenge of addiction problems.
Acknowledgments
This study was supported by the R01 (US-NIH-AT-006899: DYW, Lee).
References
- Gao Y, Hu S, Li L, Lin Y. Simultaneous determination of four alkaloids in mice plasma and brain by LC-MS/MS for pharmacokinetic studies after administration of Corydalis Rhizoma and Yuanhu Zhitong extracts. Journal of Pharmaceutical and Biomedical Analysis. 2014;92:6–12. doi: 10.1016/j.jpba.2013.12.037. [DOI] [PubMed] [Google Scholar]
- Guidance for Industry: Bioanalytical Method Validation. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Veterinary Medicine (CVM); 2013. [Google Scholar]
- Mantsch JR, Li SJ, Risinger R, Awad S, Kstz E, Baker DA, Yang Z. Levo-tetrahydropalmatine attenuates cocaine self-administration and cocaine-induced reinstatement in rats. Psychopharmacology. 2007;192:581–91. doi: 10.1007/s00213-007-0754-7. [DOI] [PubMed] [Google Scholar]
- Ma ZZ, Xu W, Jensen NH, Roth BL, Liu-Chen LY, Lee DY. Isoquinoline alkaloids isolated from Corydalis yanhusuo and their binding affinities at the dopamine D1 receptor. Molecules. 2008;13:2303–12. doi: 10.3390/molecules13092303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pharmacopoeia commission of RPC, Chinese pharmacopoeia (Part I, 2000 edition) Beijing: Chemical Industry Publishing House; 2000. pp. 139–140. [Google Scholar]
- Sasaki T, Fukushima T, Yamashita K, Toyo’oka T. Stereoselective effect of kynurenine enantiomers on the excretion of serotonin and its metabolite in rat urine. Chirality. 2010;22:258–61. doi: 10.1002/chir.20737. [DOI] [PubMed] [Google Scholar]
- Shah VP, Midha KK, Findlay JW, Hill HM, Hulse JD, McGileray IJ, McKay G, Miller KJ, Patnaik RN, Powell ML, Tonelli A, Viswanathan CT, Yacobi A. Bioanalytical method validation--a revisit with a decade of progress. Pharmaceutical Research. 2000;17:1551–7. doi: 10.1023/a:1007669411738. [DOI] [PubMed] [Google Scholar]
- Wang C, Li S, Tang Y, Wang S, Zhang Y, Fan G, Li L, Zhang Y. Microdialysis combined with liquid chromatography-tandem mass spectrometry for the determination of levo-tetrahydropalmatine in the rat striatum. Journal of Pharmaceutical and Biomedical Analysis. 2012;64:1–7. doi: 10.1016/j.jpba.2012.01.016. [DOI] [PubMed] [Google Scholar]
- Wu G, Jiang JW, Wu GC, Cao XD. Effects of four dopamine agonists on l-tetrahydropalmatine-induced analgesia and electroacupuncture analgesia in rabbits. Zhongguo Yao Li Xue Bao. 1990;11:196–200. [PubMed] [Google Scholar]
- Xiao W, Zhuang X, Shen G, Zhong Y, Yuan M, Li H. Simultaneous quantification of l-tetrahydropalmatine and its urine metabolites by ultra high performance liquid chromatography with MS/MS. Journal of Separation Science. 2014;37:696–703. doi: 10.1002/jssc.201301352. [DOI] [PubMed] [Google Scholar]
- Xu W, Wang Y, Ma Z, Chiu YT, Huang P, Rasakham K, Unterwald E, Lee DY, Liu-Chen LY. L-isocorypalmine reduces behavioral sensitization and rewarding effects of cocaine in mice by acting on dopamine receptors. Drug and Alcohol Dependence. 2013;133:693–703. doi: 10.1016/j.drugalcdep.2013.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z, Shao YC, Li SJ, Qi JL, Zhang MJ, Hao W, Jin GZ. Medication of l-tetrahydropalmatine significantly ameliorates opiate craving and increases the abstinence rate in heroin users: a pilot study. Acta Pharmacologica Sinica. 2008;29:781–8. doi: 10.1111/j.1745-7254.2008.00817.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Dong X, Le J, Wen J, Lin Z, Liu Y, Lou Z, Chai Y, Hong Z. A practical strategy for characterization of the metabolic profile of chiral drugs using combinatory liquid chromatography-mass spectrometric techniques: Application to tetrahydropalmatine enantiomers and their metabolites in rat urine. Journal of Pharmaceutical and Biomedical Analysis. 2014;94:152–62. doi: 10.1016/j.jpba.2014.01.045. [DOI] [PubMed] [Google Scholar]
- Zhu YP. Chinese materia medica: chemistry, pharmacology, and applications. CRC Press; 1998. [Google Scholar]